Abstract
Several treatment approaches for shoulder instability are possible, from conservative to surgical. Still, many surgical techniques have been described to address this condition, each with different indications according to the pathological findings, patient’s age, activity level and expectations. Besides the capsulolabral detachment from the glenoid, a plastic deformation of the capsule also occurs every time a shoulder dislocates, particularly at the first event, with variable degrees of bony lesions. It is this variability in the type and extent of the imaging and arthroscopic findings, along with the natural clinical history of each unstable shoulder, that make it of utmost importance to precisely define the instability pattern in order to select the most appropriate treatment. We present an overview of the current indications for the capsulolabral repair and the latest techniques and hardware available for that purpose.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Shoulder instability is a common pathology and several treatment approaches are possible, from conservative to surgical. Still, several surgical techniques have been described to address this condition, each with different indications according to the pathological findings, patient’s age, activity level and expectations [1].
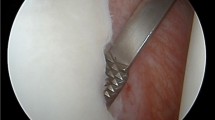
Anterior instability of the shoulder results from different types of soft tissue alterations (Fig. 11.1), with or without bony defects. They may range from a classic Bankart lesion to other variants of capsulolabral lesions such as the Perthes lesion, a labral peel off to the glenoid neck also occurring with acute anterior instability, and the anterior labro-ligamentous periosteal sleeve avulsion (ALPSA) that has also been termed ‘medialized Bankart lesion’, which is more common in cases of recurrent than with first-time traumatic dislocations of the shoulder. The gleno labral articular disruption (GLAD) lesion is present when a superficial tear of the antero-inferior labrum is combined with a portion of articular cartilage of the contiguous quadrant of the glenoid and the humeral avulsion of glenohumeral ligaments (HAGL) lesion does not involve the labro-ligamentous complex at the glenoid, but represents an isolated tear of the IGHL at its humeral insertion following vigorous shoulder dislocation.
Besides the capsulolabral detachment from the glenoid leading to these well-described labral lesions, a plastic deformation of the capsule also occurs every time a shoulder dislocates, particularly at the first event. It is a phenomenon that is similar to what happens to a simple plastic bag that is stretched with the fingers. The deformation of the plastic after the first time it is stretched will never recover back to the previous condition.
It is this variability in the type and extent of the imaging and arthroscopic findings, along with the natural clinical history of each unstable shoulder, that makes it of utmost importance to precisely define the instability pattern in order to select the most appropriate treatment.
2 Background
Surgical treatment of the unstable shoulder has evolved significantly since the first descriptions of open techniques. With the increasing popularity of arthroscopy and improved techniques and implants, arthroscopic stabilization has very rapidly become an appealing and effective choice for the treatment of traumatic shoulder instability.
Early series on the results of arthroscopic repairs reported failure rates that were quite high, despite initial success rates [2, 3]. Later studies with longer follow-ups and updated surgical techniques comparing open and arthroscopic approaches reported variable results, from 3 to 18% after open techniques and 9 to 31% after arthroscopic techniques [4,5,6,7,8]. However, many others would state that there are no statistical differences between the two groups [9, 10]. One reason for such differences in results is the heterogeneity of the groups that were studied, considering that the indications for simple labral reconstruction are, for some, controversial. Furthermore, the techniques and implants used may have varied, offering today a higher potential for success.
3 Indications for Capsulolabral Repair
Whichever surgical technique is performed to address shoulder instability, the potential success of the arthroscopic or open procedures is similar as long as the surgeon is able to recognize and address all underlying relevant contributory pathologies. Many of the failures after isolated Bankart repair reported in the literature are probably due to improper patient selection and one must bear this fact in mind when interpreting scientific evidence.
Several pre-operative risk factors for failure after surgery have been recognized, namely, younger patient age, involvement in contact sports, important bony lesions in the glenoid and/or humeral head, hyperlaxity and concomitant rotator cuff or deltoid insufficiency. Still, even with correct recognition and consideration of these factors, it seems that underestimated capsular tears and deformation are the most common cause of failure after arthroscopic Bankart repair [1, 11,12,13] along with inadequate correction of an excessively large anteroinferior capsular pouch and detached capsulolabral complex with poor quality tissue, more common after multiple episodes of dislocations or subluxations.
Bony lesion assessment is of utmost importance. The presence of a bony Bankart defect is very frequent in revision patients [13] and one should clearly distinguish between loss of glenoid contour such as the ‘classic inverted pear glenoid’ and an avulsion fracture of the anterior glenoid. While the latter may be treated arthroscopically by an anatomical reconstruction with no major increase in the failure rate [1], the former often has an associated attenuation of the anteroinferior capsulolabral complex that contribute to further erosion of the anteroinferior glenoid. In such cases, it is generally accepted that when a bony loss of over 20% is present, surgery should ideally include a bony reconstruction procedure [14,15,16,17].
Besides glenoid bony amputations, bone defects on the humeral head side, which are present in virtually all cases of shoulder dislocations, can also contribute to recurrent instability. The volume and the location of a Hill–Sachs lesion will interfere in the likelihood of repetitive dislocations, and several attempts to quantify it in the most effective way have been described.
The concept of an ‘engaging’ Hill–Sachs was introduced in order to qualify the humeral head lesion as one at a higher risk of recurrence if treated with a classic arthroscopic capsulolabral repair [18], which only addresses the restoration of the anteroinferior soft tissues. Such ‘engagement’ would have to be checked under anaesthesia or arthroscopically as the locking of the humeral head bone defect on the anterior glenoid rim in external rotation and abduction of the shoulder. In fact, as many would say, all dislocating shoulders are ‘engaging’ before performing the Bankart repair, voiding this concept of its major value and potentially leading to overtreatment of ‘engaging’ Hill–Sachs lesions. Yamamoto [19] therefore introduced the ‘glenoid track’ concept, which evaluates the zone of contact between the glenoid and the humeral head that is modified according to the arm position. The need for specific calculations under imaging, such as MRI or CT scan with 3D reconstruction, has certainly compromised wide acceptance of this method for routine usage despite allowing an objective identification of those patients with bipolar lesions at a higher risk of recurrence following isolated Bankart repair.
4 Techniques and Hardware
Surgical repair of a Bankart lesion follows steps that have been thoroughly described. The main discussion today is over the correct indications and whether there is place or not for associated procedures. However, enhancements in the technique and evolutions on implants and instrumentation have offered the orthopaedic surgeon a broader set of options to manage this lesion.
Current evidence would argue that there is hardly room for open repair of the labrum today. Still, variations in the arthroscopic approach may be necessary to be able to offer the most safe and effective method.
Both lateral decubitus and beach chair positioning allow for excellent visualization but the former may be advantageous for intra-articular procedures—which include instability repairs—due to the permanent double traction to the arm (Fig. 11.2), which will maintain the head retracted with a spacious joint.
A standard posterior viewing portal is established, which allows for a first intra-articular observation and diagnosis, followed by one or two additional anterior portals.
An anterior–inferior portal, ideally chosen using an outside-in technique with a needle, is located right superior to the subscapularis tendon through the rotator interval and slightly lateral to the glenoid plan, in a fashion that permits drilling and placement of an anchor at around 45° angulation in respect to the glenoid surface and as low as possible on the anteroinferior glenoid rim (Fig. 11.3).
Anterior-inferior rotator interval portal in a left shoulder; (a) located using an outside-in technique with a needle, right superior to the subscapularis tendon and slightly lateral to the glenoid plan, (b) allowing drilling and placement of the most inferior anchor at around 45° angulation in respect to the glenoid surface
A second anterior portal may be useful for both instrumentation and viewing (Fig. 11.4). It is placed at the superior border of the rotator interval, right behind the long biceps tendon, or directly through the pulley on top of the long biceps. Viewing from this portal may identify anterior labrum lesions more properly, allowing an easy mobilization and tensioning of the soft tissues, invaluable for a proper capsulolabral plication.
A second anterior portal may be useful for both instrumentation and viewing at the superior border of the rotator interval, right behind the long biceps tendon, or directly through the pulley on top of the long biceps. Viewing from this portal may identify anterior labrum lesions more properly, allowing an easy mobilization and tensioning of the soft tissues, invaluable for a proper capsulolabral plication
An alternative to this, in case a SLAP lesion repair is planned, is using a transcuff approach instead (Fig. 11.5), which can be used for both instrumentation and anchor placement in the superior labrum.
The use of a percutaneous 5:30 o’clock portal [20] through the subscapularis muscle is an option that can be very useful to place the lowest anterior anchor. It allows a safe drilling into the glenoid vault and avoids the risks of marginal drilling when using a rotator interval portal for that purpose.
It has been demonstrated that drilling for the most inferior anchor from a standard rotator interval portal will most likely perforate the far cortex on the inferior glenoid neck [21], with risks of iatrogenic lesion to the axillary nerve and of impairment of the anchor fixation. Following the placement of the anchor through this portal, subsequent handling of the sutures and soft tissue repairs are performed in a classical way using other portals.
To minimize this risk of missing the best drilling direction for the most inferior anchor, some companies offer the possibility of using a curved guide and a flexible drill, enabling an effective perforation of a tunnel inside bone, dispensing the 5:30 portal.
Fixation of the capsulolabral tissue to the glenoid rim can be effectively achieved by the usage of different types of anchors and suture configurations. Evolutions on these have been the rule since the advent of shoulder arthroscopy, with various reports contributing to a better knowledge of the biomechanical properties of the fixation today.
The recognition of the capsulolabral footprint led to the description of double-row fixations on the glenoid by Lafosse et al.—the Cassiopeia technique—and later by other surgeons [22,23,24,25], with significant improvement in functional outcomes with no major complications. However, in spite of laboratorial studies and a few low-strength studies with patients showing the time-zero strength of this technique, there is no clinical evidence that this option has advantages over the single row and the higher risks of complications and increased costs must not be underestimated.
Other Bankart repair configurations are widely used and have deserved a dedicated comparison (Fig. 11.6). Classical knotted and knotless fixations, simple vertical stitch, horizontal mattress stitch, Mason–Allen (a combination of a mattress and single stitch with a double-loaded anchor) [11, 26], double-row and purse-string [27] techniques have all been presented as viable options but there is a lack of strong clinical evidence of advantages of one over another.
Bankart repair configurations: (a) vertical stitch with double-loaded anchor, (b) simple vertical stitch with single-loaded anchor (knotted or knotless), (c) horizontal mattress stitch, (d) Mason–Allen, a combination of a mattress and single stitch with a double-loaded anchor [11, 26], (e) double-row and (f) purse-string [27] techniques
Double-row repair techniques have been shown to provide better coverage of the native footprint of the labrum but have not provided superior biomechanical properties in the lab compared to single-row repair techniques. There is no clear difference in footprint coverage, gapping, stiffness or biomechanical strength between the simple suture and horizontal mattress suture repair techniques [28]. Likewise, the same authors did not find any additional strength by using labral tape in double-row fixations.
Mattress type repairs are reportedly more effective in achieving a more anatomical reconstruction of the labral stump, potentially more similar to the native labral slope with a bumper effect, but they have been shown to offer identical biomechanical characteristics when compared to simple suture repairs [29]. In fact, laboratorial and post-operative studies with MRI have shown that the labral slope, height and morphology are reliably restored at 15 months after using bioabsorbable knotless anchors, similar to controls and other reports on simple knotted repairs [30]. There is also no difference in the radiological and clinical outcomes at, respectively, 6 months and at least 2 years after surgery between using a simple stitch and a modified Mason-Allen stitch in arthroscopic Bankart repairs [26].
The availability of different types of anchors in the market for this purpose today is very wide and they definitely deserve an overview, considering their role in the final clinical result. Major evolutions have taken place concerning this matter since the advent of shoulder arthroscopy in order to increase its success and limit the risk of complications.
Despite the good results of the first reports on arthroscopic Bankart repairs, the rate, pattern and extent of the complications due to the usage of metallic suture anchors in the glenoid lead to a shift from metallic to bioabsorbable implants [31,32,33]. The acceptance of ‘arguably’ biodegradable suture anchors, such as the slowly degrading pure PLLA (poly-l-lactic acid) or more rapidly degrading biocomposite PLLA/β-tricalcium phosphate-based anchors was, for that reason, very high among orthopaedic surgeons. Nevertheless, even these are not risk-free, with reports on breakage, osteolysis, chondrolysis and synovitis after using them [34] and therefore the constant evolution in implant types and profiles, with older anchors and techniques being replaced with newer ones as technology develops. Besides biodegradable lactide-containing suture anchors, other innovations included the use of polyetheretherketone (PEEK) as the anchor material, the addition of multiple high-strength sutures made in part or entirely with ultrahigh molecular weight polyethylene (UHMWPE) and the development of ‘knotless’ designs.
PEEK is a radiolucent but not biodegradable plastic suitable for a variety of implants that has the advantages of being high strength, enabling good post-operative imaging and facilitating revision surgery because it is soft enough to be drilled through [35]. However, complications due to the fact that it is a rigid implant are not negligible and therefore the appeal for newer options, such as the all-suture anchors.
All-suture anchors are made using UHMWPE—the material of which virtually all the anchors’ sutures currently in the market are made of—and perform very well in terms of pullout strength in the lab, in some cases better than their rigid counterparts [35, 36]. However, some biomechanical concerns have been reported with the use of these newer anchors, namely, the first-generation ones [37], concerning load to failure and bone cyst formation [38]. A direct comparison between an all-soft and a rigid biocomposite glenoid anchor revealed a histologic and biomechanical response in dogs [38] that brought some concern about the former, by means of a large cyst-like cavity formation with a rim of dense lamellar bone in the anchor sites. This potential risk for clinical failure has motivated further studies and another group found satisfying radiological and clinical outcomes after arthroscopic instability surgery using first-generation all-suture anchors in human patients [39]. Unlike the canine models of the previous report, these patients followed a classical post-op protocol that included immobilization, and imaging at early follow-up (12–28 months) revealed good labral healing without important bony reaction or the formation of large cysts.
In spite of some differences in displacement after cyclic loading between different all-soft anchors for the glenoid [40], it has been demonstrated that this phenomenon can be minimized by slightly reducing the insertion depth for the anchor [41] which will minimize the amount of bone stock that is destroyed with a deep drilling. At the end of the day, its overall efficacy compares favourably to standard solid anchors for labral repairs.
Knotless anchors for labral repair have been an appealing option for some time now and have been subject to several comparisons in the literature. They offer the advantage of a quicker and easier repair, diminishing the potential for errors, and absence of a bulky knot stack that may lead to early osteoarthritis when present and rubbing against the chondral surface. Furthermore, the rate of glove and skin lacerations is lower, recognized as a risk for both the patient and the surgeon when tying knots. [42]
While some studies report no significant differences between the two options [43, 44], others report worse clinical results using knotless anchors when compared to classical knot-tying suture anchors [45].
But one must be judicious when interpreting these scientific conclusions. Generally speaking, there are two different kinds of knotless labral anchors available, demanding either an ‘anchor first’ or ‘suture first’ technique for their usage. All the comparisons available in the literature consider the ‘suture first’ technique anchors, which, as recurrently reported, do not allow the best estimation of the tension to give the sutures and respective soft tissues fixation.
However, an ‘anchor first’ technique, due to its different method of application and tissue fixation, does not present with the same issue and may be a valuable option without such limitation (Fig. 11.7).
Regardless of the type of anchor that is used, a satisfactory capsular shift is mandatory whenever there is a need to reduce the capsular volume, which is normally the case. Previous reports have demonstrated that a minimum of three double-loaded suture anchors had to be used for that purpose [1] but another one states that one or two anchors could be enough, as long as the capsulolabral tissue is plicated as desired [27].
It seems today that all-soft anchors, being less invasive and requiring a significantly smaller bone tunnel than rigid suture anchors, may reduce the risk of hardware complications such as secondary joint damage or glenoid fracture, and at the same time allow a safer drilling for multiple anchors into a small anatomic area, in case of failure of a previous one or in the revision setting. Besides, in the event of marginal tunnel drilling or perforation of the far cortex, the fact that these anchors only require an intact cortical surface for proper seating will likely diminish the risk of their loosening (Fig. 11.8).
The thin tunnel for all-soft anchors allows multiple anchors into a small anatomic area, in case of failure of a previous one or in the revision setting. In the event of marginal tunnel drilling or perforation of the far cortex, the sole requirement for an intact cortical surface for proper seating, usage of a soft anchor will likely diminish the risk of their loosening
These facts may eventually lead to better clinical results in the long run and widen the indications for soft tissue repairs, compared to bony procedures.
Still, objective criteria are necessary in order to take a better-informed decision on the treatment of an unstable shoulder.
5 Discussion and Conclusion
Given the subjectivity and lack of consensus on the surgical management of shoulder instability, Balg and Boileau [46] proposed a simple ten-point scale Instability Severity Index Score (ISIS) to determine the risk of recurrence following isolated arthroscopic Bankart repair. It considers several prognostic factors, which, if present, add up 1 or 2 points to the final score: age below 20, being into competitive, contact or overhead sports, hyperlaxity and important bony losses either on the humeral head (Hill–Sachs lesion) or the glenoid. A score of 3 or less was associated with a 5% recurrence rate and a score above 4 was associated with an unacceptable recurrence rate after an isolated Bankart repair and therefore a bony reconstruction should be performed. In spite of some recognized weaknesses [47, 48], the ISIS is a useful tool for the surgeon to choose the optimal surgical treatment and minimize the risk of recurrent instability.
Still, many questions remain unanswered concerning this matter, despite substantial progress made in the understanding of risk factors for recurrence following surgical treatment of anterior shoulder instability.
Arthroscopic remplissage for anterior instability has become an adjunct to Bankart repairs since it was first described in 2008 as a means to augment the labral repair in patients with subcritical glenoid bone loss.
However, the critical level of glenoid bone loss requiring bone grafting or coracoid transfer is not clearly defined, ranging from 10 to 25% according to different researchers [49]. This led to the definition of the glenoid track concept as a means for defining the need for isolated Bankart with or without remplissage versus Latarjet [50] as described in another chapter of this book. It is a valid tool to guide the surgeon but, like other tools available for the same purpose, has limitations, since it is often difficult, inaccurate and not very practical to calculate and does not account for soft tissue quality and patient factors such as age and sex.
Taking this into account as well as evidence that the soft tissues repair is of undeniable importance, even when performing a Latarjet [51], it is clear that a proper capsulolabral reconstruction has a major place in shoulder instability treatment today.
References
Boileau P, Villalba M, Héry JY, Balg F, Ahrens P, Neyton L. Risk factors for recurrence of shoulder instability after arthroscopic Bankart repair. J Bone Joint Surg Am. 2006;88(8):1755–63.
Grana WA, Buckley PD, Yates CK. Arthroscopic Bankart suture repair. Am J Sports Med. 1993;21:348–53.
Green MR, Christensen KP. Arthroscopic Bankart repair: two to five year follow-up with clinical correlation to severity of glenoid labral lesion. Am J Sports Med. 1995;23:276–81.
Bacilla P, Field LD, Savoie FH. Arthroscopic Bankart repair in a high demand patient population. Arthroscopy. 1997;13:51–60.
Gartsman GM, Roddey TS, Hammerman SM. Arthroscopic treatment of anterior-inferior glenohumeral instability. J Bone Joint Surg. 2000;82:991–1003.
Cole BJ, L’Insalata J, Irrgang J, Warner JJP. Comparison of arthroscopic and open anterior shoulder stabilization. J Bone Joint Surg. 2000;82:1108–14.
Sperber A, Hamberg P, Karlsson J, et al. Comparison of an arthroscopic and an open procedure for posttraumatic instability of the shoulder: a prospective randomized multicenter study. J Shoulder Elbow Surg. 2001;10:105–8.
Provencher MT. Editorial commentary: is it time to take a stand? When arthroscopic Bankart repair is no longer a viable option for anterior shoulder instability. Arthroscopy. 2018;34(9):2537–40.
Fabbriciani C, Milano G, Demontis A, Fadda S, Ziranu F, Mulas PD. Arthroscopic versus open treatment of Bankart lesion of the shoulder: a prospective randomized study. Arthroscopy. 2004;20:456–62.
Harris JD, Gupta AK, Mall NA, et al. Long-term outcomes after Bankart shoulder stabilization. Arthroscopy. 2013;29(5):920–33.
Castagna A, Conti M, Moushine E, Delle Rose G, Massazza G, Garofalo R. A new technique to improve tissue grip and contact force in arthroscopic capsulolabral repair: the MIBA stitch. Knee Surg Sports Traumatol Arthrosc. 2008;16:415–9.
Mologne TF, McBride MT, Lapoint JM. Assessment of failed arthroscopic anterior labral repairs: findings at open surgery. Am J Sports Med. 1997;25:813–7.
Tauber M, Resch H, Forstner R, Raffl M, Schauer J. Reasons for failure after surgical repair of anterior instability. J Shoulder Elbow Surg. 2004;13(3):279–85.
Arce G, Arcuri F, Ferro D, Pereira E. Is selective arthroscopic revision beneficial for treating recurrent anterior shoulder instability? Clin Orthop Relat Res. 2012;479(4):965–71.
Bartl C, Schumann K, Paul J, Vogt S, Imhoff AB. Arthroscopic capsulolabral revision repair for recurrent anterior shoulder instability. Am J Sports Med. 2011;39:511–8.
Mologne TS, Provencher MT, Menzel KA, et al. Arthroscopic stabilization in patients with an inverted pear glenoid: results in patients with bone loss of the anterior glenoid. Am J Sports Med. 2007;35(8):1276–83.
Provencher MT, Arciero RA, Burkhart SS, Levine WN, Ritting AW, Romeo AA. Key factors in primary and revision surgery for shoulder instability. Instr Course Lect. 2010;59:227–44.
Burkhart SS, De Beer JF. Traumatic glenohumeral bone defects and their relationship to failure of arthroscopic Bankart repairs: significance of the inverted-pear glenoid and the humeral engaging Hill–Sachs lesion. Arthroscopy. 2000;16:677–94.
Yamamoto N, Itoi E, Abe H, et al. Contact between the glenoid and the humeral head in abduction, external rotation, and horizontal extension: a new concept of glenoid track. J Shoulder Elbow Surg. 2007;16(5):649–56.
Imhoff AB, Ansah P, Tischer T, et al. Arthroscopic repair of anterior-inferior glenohumeral instability using a portal at the 5:30 o’clock position: analysis of the effects of age, fixation method, and concomitant shoulder injury on surgical outcomes. Am J Sports Med. 2010;38(9):1795–803.
Lim TK, Koh KH, Lee SH, Shon MS, Bae TS, Park WH, Yoo JC. Inferior anchor cortical perforation with arthroscopic bankart repair: a cadaveric study. Arthroscopy. 2013;29(1):31–6.
Lafosse L, Baier GP, Jost B. Footprint fixation for arthroscopic reconstruction in anterior shoulder instability: the Cassiopeia double-row technique. Arthroscopy. 2006;22(2):231.e1–6.
Iwaso H, Uchiyama E, Sakakibara S, Fukui N. Modified double-row technique for arthroscopic Bankart repair: surgical technique and preliminary results. Acta Orthop Belg. 2011;77(2):252–7.
Wade R, Reddy PVB. Functional outcome of arthroscopic double row repair for Bankart lesion. J Orthop. 2018;15(3):792–7. https://doi.org/10.1016/j.jor.2018.03.021. eCollection 2018 Sep
McDonald LS, Thompson M, Altchek DW, McGarry MH, Lee TQ, Rocchi VJ, Dines JS. Doublerow capsulolabral repair increases load to failure and decreases excessive motion. Arthroscopy. 2016;32(11):2218–25. https://doi.org/10.1016/j.arthro.2016.03.025. Epub 2016 May 18
Park JY, Chung SW, Lee JS, Oh KS, Lee JH. Comparison of clinical and radiographic outcomes of vertical simple stitch versus modified Mason-Allen Stitch in arthroscopic Bankart repairs: a prospective randomized controlled study. Am J Sports Med. 2019;47(2):398–407.
Witney-Lagen C, Perera N, Rubin S, Venkateswaran B. Fewer anchors achieves successful arthroscopic shoulder stabilization surgery: 114 patients with 4 years of follow-up. J Shoulder Elbow Surg. 2014;23(3):382–7.
Judson CH, Voss A, Obopilwe E, Dyrna F, Arciero RA, Shea KP. An anatomic and biomechanical comparison of Bankart repair configurations. Am J Sports Med. 2017;45(13):3004–9.
Boddula MR, Adamson GJ, Gupta A, McGarry MH, Lee TQ. Restoration of labral anatomy and biomechanics after superior labral anterior-posterior repair - comparison of mattress versus simple technique. Am J Sports Med. 2012;40(4):875–81.
Stein T, Mehling AP, Reck C, Buckup J, Efe T, Hoffmann R, Jaeger A, Welsch F. MRI assessment of the structural labrum integrity after Bankart repair using knotless bio-anchors. Knee Surg Sports Traumatol Arthrosc. 2011;19:1771–9.
Papalia R, Franceschi F, Diaz Balzani L, D’Adamio S, Denaro V, Maffulli N. The arthroscopic treatment of shoulder instability: bioabsorbable and standard metallic anchors produce equivalent clinical results. Arthroscopy. 2014;30(9):1173–83.
Silver MD, Daigneault JP. Symptomatic interarticular migration of glenoid suture anchors. Arthroscopy. 2000;16(1):102–5.
Ozbaydar M, Elhassan B, Warner JJ. The use of anchors in shoulder surgery: a shift from metallic to bioabsorbable anchors. Arthroscopy. 2007;23(10):1124–6.
Cobaleda AF, Sanders EJ, Barber FA. Adverse events associated with biodegradable lactide-containing suture anchors. Arthroscopy. 2014;30(5):555–60.
Barber FA, Herbert MA, Hapa O, Rapley JH, Barber CA, Bynum JA, Hrnack SA. Biomechanical analysis of pullout strengths of rotator cuff and glenoid anchors: 2011 update. Arthroscopy. 2011;27(7):895–905.
Barber FA, Herbert MA. Cyclic loading biomechanical analysis of the pullout strengths of rotator cuff and glenoid anchors: 2013 update. Arthroscopy. 2013;29(5):832–44.
Erickson J, Chiarappa F, Haskel J, Rice J, Hyatt A, Monica J, Dhawan A. Biomechanical comparison of a first- and a second-generation all-soft sutureglenoid anchor. Orthop J Sports Med. 2017;5(7):2325967117717010. https://doi.org/10.1177/2325967117717010. eCollection 2017 Jul
Pfeiffer FM, Smith MJ, Cook JL, Kuroki K. The histologic and biomechanical response of two commercially available small glenoid anchors for use in labral repairs. J Shoulder Elbow Surg. 2014;23(8):1156–61.
Willemot L, Elfadalli R, Jaspars KC, Mark H, Ahw MH, Peeters J, Jansen N, Declerq G, Verborgt O. Radiological and clinical outcome of arthroscopic labral repair with all-suture anchors. Acta Orthop Belg. 2016;82:174–8.
Ruder JA, Dickinson EY, Peindl RD, Habet NA, Fleischli JE. Cyclic and load-to-failure properties of all-suture anchors in human cadaveric shoulder glenoid bone. Arthroscopy. 2019. pii: S0749-8063(19)30061-1.
Ruder JA, Dickinson EY, Habet N, Peindl RD, D’Alessandro DF, Fleischli JE. Slight reduction in the insertion depth for an all-suture anchor decreases cyclic displacement in the shoulder glenoid. Arthroscopy. 2018;34(5):1384–90.
Ng DZ, Kumar VP. Arthroscopic Bankart repair using knot-tying versus knotless suture anchors: is there a difference? Arthroscopy. 2014;30(4):422–7.
Martinez A, Han Y, Sardar ZM, Beckman L, Steffen T, Miller BS, Martineau PA. Risk of glove perforation with arthroscopic knot tying using different surgical gloves and high-tensile strength sutures. Arthroscopy. 2013;29(9):1552–8.
Kocaoglu B, Guven O, Nalbantoglu U, Aydin N, Haklar U. No difference between knotless sutures and suture anchors in arthroscopic repair of Bankart lesions in collision athletes. Knee Surg Sports Traumatol Arthrosc. 2009;17(7):844–9.
Cho NS, Lubis AM, Ha JH, Rhee YG. Clinical results of arthroscopic Bankart repair with knot-tying and knotless suture anchors. Arthroscopy. 2006;22(12):1276–82.
Balg F, Boileau P. The instability severity index score. A simple pre-operative score to select patients for arthroscopic or open shoulder stabilisation. J Bone Joint Surg Br. 2007;89(11):1470–7.
Rouleau DM, Hebert-Davies J, Djahangiri A, Godbout V, Pelet S, Balg F. Validation of the instability shoulder index score in a multicenter reliability study in 114 consecutive cases. Am J Sports Med. 2013;41(2):278–82.
Ruiz Ibán MA, Asenjo Gismero CV, Moros Marco S, Ruiz Díaz R, Del Olmo Hernández T, Del Monte Bello G, García Navlet M, Ávila Lafuente JL, Díaz Heredia J. Instability severity index score values below 7 do not predict recurrence after arthroscopic Bankart repair. Knee Surg Sports Traumatol Arthrosc. 2019;27(12):3905–11. https://doi.org/10.1007/s00167-019-05471-w.
Lazarides AL, Duchman KR, Ledbetter L, Riboh JC, Garrigues GE. Arthroscopic remplissage for anterior shoulder instability: a systematic review of clinical and biomechanical studies. Arthroscopy. 2019;35:617–28.
Di Giacomo G, Itoi E, Burkhart SS. Evolving concept of bipolar bone loss and the Hill-Sachs lesion: from “engaging/ non-engaging” lesion to “on-track/off-track” lesion. Arthroscopy. 2014;30:90–8.
Cuéllar A, Cuéllar R, de Heredia PB. Arthroscopic revision surgery for failure of open latarjet technique. Arthroscopy. 2017;33(5):910–7.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 ESSKA
About this chapter
Cite this chapter
Gomes, N., Aramberri, M., Fonte, H. (2020). Arthroscopic Bankart Repair: How It Looks Today. In: Brzóska, R., Milano, G., Randelli, P., Kovačič, L. (eds) 360° Around Shoulder Instability. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-662-61074-9_11
Download citation
DOI: https://doi.org/10.1007/978-3-662-61074-9_11
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-662-61073-2
Online ISBN: 978-3-662-61074-9
eBook Packages: MedicineMedicine (R0)