Abstract
The prevalence of developmental defects of enamel (DDE) in the permanent dentition in developed countries has been reported to be in the range of 9–68 % and with no gender predilection. Several etiological factors have been implicated as being responsible for DDE in the permanent teeth. Although local, systemic, genetic or environmental factors have been attributed to DDE frequently they are likely to be multifactorial in nature. These factors are discussed in relation to the timing of enamel development with consideration of the evidence, or lack thereof, for the association between the putative etiological factors and the nature of the subsequent abnormalities.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
The first permanent tooth to begin calcification is the first molar. This occurs around the time of birth, while the anterior teeth commence calcification between 4 and 6 months of age in a sequential order from the central incisor to the canine. The maxillary lateral incisor is the exception as calcification of this tooth occurs around 10–12 months of age [1]. At around 6 years of age, the first permanent molar tooth begins to erupt into the oral cavity and by the age of 14 years, most children have all of their permanent teeth erupted except for their third molars (Table 2.1). Many factors have been implicated in the etiology of developmental defects of enamel (DDE) in permanent teeth. This chapter discusses these factors in relation to the timing of enamel development and considers the evidence, or lack thereof, for the association between the etiological factors and the nature of the subsequent abnormalities.
Prevalence of DDE in the Permanent Dentition
Mouth and tooth prevalence are the most commonly used systems to report the prevalence data for DDE. Mouth prevalence is determined by the inclusion of any individual who has been found to have at least one tooth affected by the condition, while tooth prevalence illustrates the percentage of teeth affected per person. Mouth prevalence figures reflect the extent of the distribution of enamel defects in a population group because individuals who are mildly and severely affected are grouped together. Tooth prevalence indicates the proportion of teeth affected and hence reflects the severity of the condition [2].
Table 2.2 summarizes the prevalence of DDE in the permanent dentition as reported in the literature. Wide variations exist in the literature because of the use of various terminologies and the different diagnostic criteria employed to describe the enamel defects in the permanent dentition [21, 22, 24, 26]. Nevertheless, the majority of reports have failed to demonstrate any difference in the prevalence of enamel defects between girls and boys [12, 27]. Furthermore, for all types of enamel defects, the published mouth prevalence in the permanent dentition ranges from 9.8 % to 93 % [8, 26], while tooth prevalence figures range from 2.2 % to 21.6 % [26, 28].
Etiology of DDE in the Permanent Dentition
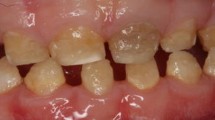
Enamel morphogenesis is a continuous, complex process that starts with the secretion of enamel matrix proteins followed by mineralization and finally maturation. This process has been shown to start at the cusps on the molars and the incisal part of the incisors, progressing to the cervical areas of the teeth [29]. However, there is still limited understanding of how mineralization progresses across the crowns of the teeth. This could be important in determining the timing of defects to relate to specific disturbances caused by systemic disorders. Disturbances in the different stages of enamel formation may result in a range of enamel defects with quite different clinical appearances and structural changes. Defective formation of the enamel matrix results in hypoplasia, a quantitative defect, depicted by generalized thinning or pitting types of defects (Fig. 2.1). Defective calcification of an otherwise normal fully developed organic enamel matrix results in hypomineralization, a qualitative defect (Fig. 2.2). This is seen clinically as changes in color and translucency of the enamel and presents as enamel opacities which can be either demarcated or diffuse [2, 30].
Although the etiology of enamel defects may be attributed to local, systemic, genetic, or environmental factors, most are likely to be multifactorial in nature. This makes it difficult to identify a single cause for many cases of DDE. The time frame of exposure and the mechanism underpinning the causative factors determine the presentation of these defects. Defects on a single tooth or only a few teeth suggest a local etiological factor e.g. a defect in a permanent tooth due to damage (trauma or infection) to its primary predecessor (Fig. 2.3a–c). Alternatively, a systemic factor (both short and longer term) may affect all the teeth that are developing during the time of the insult and lead to what is described as a chronological defect. Genetic factors can be considered separately. Defects caused by genetic factors are most often (although not always) generalized in distribution, affecting both the primary and permanent dentitions. They may present as either enamel defects alone as seen in amelogenesis imperfecta (see Chap. 5) or as enamel defects associated with other general genetic disorders/syndromes (see Chap. 4).
(a) Caries in the mandibular second primary molar has led to the intra-radicular infection which has resulted in hypoplasia of the developing second premolar. (b) Hypoplasia of the maxillary permanent canine as a consequence of infection of the primary predecessor. (c) Hypoplasia in the form of missing enamel is exhibited by this mandibular second premolar following infection of the primary second molar
Enamel defects can be classified clinically as demarcated and diffuse opacities and hypoplasia. The location of isolated defects depends on the stage of amelogenesis at the time of the insult or injury [31]. The general consensus regarding the etiology of isolated opacities, which may be demarcated or diffuse and present as white, creamy, or yellow in color, is that amelogenesis is affected by a disturbance during the mineralization phase. It remains unclear why this would involve only an isolated patch of enamel on the crown and not the whole surface. Conversely, hypoplasia occurs when there is a disturbance during the secretory stage of amelogenesis while the enamel is only partly mineralized. Thus, enamel defects with similar presentations may have been caused by a variety of etiological factors. Furthermore, the same etiological factor can produce enamel defects with different presentations depending on the timing of the insult. Examples of this are commonly seen following primary tooth trauma. When a maxillary anterior primary tooth is intruded in infancy (during the first year of life), the crown of the permanent successor may suffer severe structural damage with missing enamel and even dilaceration of the root or the crown, while an intrusion in the later preschool years may only cause an isolated labial hypomineralized enamel opacity on the permanent successor (Fig. 2.4a, b).
Determining the Etiology
Based on the number of teeth affected, the possible etiological factors for DDE in permanent teeth can be categorized as being local or general. However the evidence is equivocal, with the majority of published reports being animal studies or case reports of children with specific systemic disorders. The putative etiological factors reported in the literature are summarized in Table 2.3. Only a few of these factors have good evidence supporting a direct causal effect, e.g., trauma to a primary predecessor or high levels of ingested fluoride during early childhood.
Localized Enamel Defects
When only one or few adjacent teeth exhibit an enamel defect, it is usually considered to be caused by a very localized or isolated factor rather than a more generalized systemic or genetic factor [32]. The most common causes of localized enamel defects are trauma, chronic radicular infection resulting from pulpal necrosis in a primary predecessor tooth, surgery in the adjacent area or radiation therapy. Other isolated defects with incompletely formed enamel such as those associated with dens invaginatus and dens evaginatus may be due to a genetic influence in certain teeth.
Intrusive and lateral luxation injuries to the primary teeth often result in enamel defects in the succedaneous permanent teeth [33, 34]. This occurs most often in the anterior teeth as they are more likely to suffer the direct impact of trauma from falling or being struck by an object than the posterior teeth. The severe consequences arise because of the direct or nearly direct impact of the apex of the root of a primary incisor on the crown or follicle of the developing permanent successor or may occur as a consequence of post-traumatic complications of inflammation and infection. Furthermore, if the trauma to the primary tooth leads to pulpal necrosis, then there is a greater likelihood of enamel defects occurring in the succedaneous permanent tooth. This can also occur following severe dental caries with pulp exposure leading to untreated chronic infection [35]. Surgical procedures such as extraction of primary teeth, removal of supernumerary teeth, cleft palate repair or distraction osteogenesis have all been reported to cause localized enamel defects in the succedaneous or adjacent permanent teeth [36–39]. Untreated carious lesions extending into the pulp of primary teeth may result in pulpal necrosis and infection which may result in DDE in the succedaneous permanent teeth (Fig. 2.3) [40–42]. The reported defects have ranged from demarcated opacities to hypoplastic defects [43].
Generalized Enamel Defects
Generalized enamel defects are those defects that are seen either on the crowns of groups of teeth or in all the teeth. As mentioned previously, the stage of amelogenesis in the particular tooth germ at the time of the insult or injury is often critical to the resulting location and type of enamel defect. The timing of the disturbances during tooth morphogenesis will determine the location and type of defects and the number of teeth that will be affected. Homologous pairs of teeth will usually have enamel defects in similar locations although the severity of the defects may not always be the same suggesting that even homologous teeth do not always mineralize at exactly the same rate. As the process of enamel development occurs in the different tooth types over different developmental times, the locations of enamel defects will differ between different homologous pairs of teeth [44]. Developmental defects with this type of distribution are referred to as generalized defects of enamel and may be caused by environmental factors or systemic conditions that have either a defined time of influence or an ongoing influence throughout childhood, or they may be caused by genetic factors. These conditions and their association with DDE are discussed more fully in Chaps. 4 and 5.
Environmental Factors
Several environmental factors have been associated with DDE. These are believed to cause systemic disturbances that affect enamel development rather than the environmental agent affecting the ameloblasts directly. Environmental agents such as lead, mercury, bisphenol A (an endocrine-disrupting chemical), some drugs such as anticancer agents and tetracycline and some trace elements including fluoride and strontium have been implicated in DDE. The systemic ingestion of these chemical substances may exert an adverse effect on enamel formation during and after fetal development [45, 46]. Exposure to such substances during amelogenesis may result in the formation of defective enamel depending on the stage of enamel development, the timing of exposure, the length of exposure and the underlying health of the individual [47]. It should be remembered that some of the substances also have a very positive effect, such as the ingestion of low levels of fluoride to improve enamel maturation and decrease dental caries risk.
DDE arising from excess fluoride ingestion have been found in areas with high natural levels of fluoride in the drinking water. Ingestion of excess fluoride during tooth development can result in dental fluorosis, a form of enamel hypomineralization where the white striations contain less mineral and retain more developmental enamel proteins. The hypomineralization can vary from minor white striations to small or more extensive opacities [48, 49]. The first 3 years of life is generally understood to be the window of maximum susceptibility for the development of fluorosis in the permanent maxillary central incisor teeth [50]. Nevertheless, for the whole permanent dentition, excluding the third molars, the first 6–8 years of life is an important period when exposure to appropriate levels of fluoride as defined in local guidelines should be followed [51]. Fluorotic hypomineralization defects do have specific characteristics which allow them to be differentiated from defects caused by other factors [52], and this can be useful for the clinician to consider when diagnosing DDE. The characteristic lesions in fluorosis are dull and chalky in appearance; they may vary in color from chalky white, yellow, or brown, and in some cases there are small pits which accumulate organic matter producing yellow to brown spots (Fig. 2.5). When diagnosing DDE that may be related to fluoride, a careful history of total fluoride ingestion as well as medical and developmental histories should be taken for the appropriate developmental period of the affected teeth. Another trace element that can have an influence on the development of hypomineralization in enamel is strontium which has been shown to be associated with enamel hypomineralization similar to that caused by excess fluoride [53].
Several animal experiments have shown that teeth are very sensitive to the effects of dioxins [54]. The most toxic dioxin, 2,3,7,8-tetrachlorodibenzo-para-dioxin (TCDD), arrests degradation and/or removal of enamel matrix proteins which is a pre-requisite for the completion of enamel mineralization [55]. It has been hypothesized that prolonged breast-feeding might increase the risk of DDE due to the environmental contamination of breast milk with dioxins or dioxin-like compounds [56, 57]. A dose-response relationship between high levels of dioxins or polychlorinated biphenyls (PCBs) exposure (serum concentration) and DDE in permanent teeth has been reported [58, 59]. However the evidence remains mixed with other studies reporting contradictory findings [11] and more recent studies failing to identify any correlation between DDE in children living in areas polluted by dioxins and in those living in areas with low pollution [60, 61]. It has also been reported that there is an increase in incidence of diffuse white/creamy mottling of enamel with elevated levels of chemicals such as fluorine, ammonia and sulfur in the atmosphere [62]; hypervitaminosis D [63]; chronic lead poisoning [64]; diphosphonate poisoning [65]; and polychlorinated biphenyl poisoning [66]. Antineoplastic therapy in the form of radiation treatment and/or chemotherapy can affect any cells including ameloblasts and consequently lead to DDE [67]. It has been reported that central nervous system irradiation with scattered irradiation of 0.72–1.44 Gy to the dental arches can result in a range of enamel defects in developing permanent teeth [68, 69].
Genetic Disorders
Amelogenesis imperfecta (AI) is a heterogeneous group of genetic disorders that affects the development of dental enamel resulting in varying degrees of hypoplasia, hypomineralization and/or hypomaturation [70]. A single-gene defect can occur as X-linked, autosomal dominant, or autosomal recessive inheritance. There is evidence that AI may present as part of a hereditary syndrome, examples of which are epidermolysis bullosa [71, 72], pseudohypoparathyroidism [73], and tricho-dento-osseous syndrome [74]. See Chaps. 4 and 5.
Systemic Conditions
It has been suggested that perinatal and postnatal problems, hypoxia and malnutrition may be related to the occurrence of DDE in permanent teeth. However, the mechanisms are not clearly understood and it is difficult to link any of the conditions directly to the defects. Children with low birth weights have been shown to be more at risk for developing DDE in their primary teeth. However the evidence is weaker in relation to the permanent dentition [75]. Similarly, problems at the time of delivery such as caesarean section and labor in excess of 20 h and poor respiratory response in the postnatal period (hypoxia and respiratory diseases in early childhood) have all been linked with the occurrence of DDE, but currently there is insufficient evidence to be confident about any of these as direct causes of DDE [76].
There are numerous reports of DDE in the permanent dentition being associated with diseases and infectious conditions occurring in early childhood. Infectious diseases occurring during early childhood that may be related to DDE include chickenpox, asthma, measles, mumps, scarlet fever, exanthematous fevers, pneumonia and urinary tract infections. Other conditions such as convulsions, tuberculosis, diphtheria, whooping cough, otitis media, bulbar polio with encephalitis, gastrointestinal disturbances, cyanotic congenital heart disease, neurological disorders and renal disorders have also all been mentioned in association with DDE.
Vitamin D deficiency, hypocalcemia, hypophosphatemia and hyperparathyroidism have also all be implicated in DDE in the permanent dentition. Vitamin D-dependent rickets (VDDR) is a condition which appears to be increasing in prevalence either due to vitamin D deficiency in the mother during pregnancy or vitamin D deficiency in the young child [77]. Vitamin D deficiency contributes to the development of hypocalcemia and hypophosphatemia which is then compounded by secondary hyperparathyroidism which in turn increases renal inorganic phosphate (Pi) clearance, effectively worsening the hypophosphatemia. Consequently the low concentrations of Ca2+ and Pi prevent proper mineralization of the organic bone matrix and this also leads to defects in enamel mineralization [78].
There is a growing body of literature reporting associations between DDE and systemic conditions such as celiac disease, cystic fibrosis and tuberous sclerosis though the mechanisms involved are often not fully understood [79–82]. Hypocalcemia has also associated with the occurrence of enamel hypoplasia in the permanent dentition in hereditary vitamin D-resistant rickets, X-linked hypophosphatemia, and hypoparathyroidism [83].
Despite the numerous reports of DDE in association with all of these conditions, there is little strong evidence to support any condition as being a primary etiological agent responsible for the formation of the enamel defects in the permanent dentition.
Summary
The prevalence of DDE in the permanent dentition (in developed countries) is reported to be between 9 and 68 %. This wide variation can be attributed to the use of different criteria and terminologies to describe enamel defects. The etiology of enamel defects may be local or systemic, genetic, or acquired in origin. The clinical presentation of DDE varies greatly depending on the etiology and severity. Single-tooth defects can be attributed to a local factor, whereas in those of systemic etiology many or all of the teeth that are developing during the time of influence of the etiological factor are affected (chronological defects). Defects with a genetic etiology form a separate entity, usually affecting both the primary and permanent teeth. Identifying the presence of DDE and establishing a diagnosis are essential to inform appropriate treatment planning in both the short and longer term (see Chap. 8).
References
AlQahtani SJ, Hector MP, Liversidge HM. Brief communication: the London atlas of human tooth development and eruption. Am J Phys Anthropol. 2010;142(3):481–90.
King N, Wei S. A review of the prevalence of developmental enamel defects in permanent teeth. J Paleopathol. 1992;2:342–57.
Suckling GW, Brown RH, Herbison GP. The prevalence of developmental defects of enamel in 696 nine-year-old New Zealand children participating in a health and development study. Community Dent Health. 1985;2(4):303–13.
Dooland MB, Wylie A. A photographic study of enamel defects among South Australian school children. Aust Dent J. 1989;34(5):470–3.
Dummer PM, Kingdon A, Kingdon R. Prevalence and distribution by tooth type and surface of developmental defects of dental enamel in a group of 15- to 16-year-old children in South Wales. Community Dent Health. 1990;7(4):369–77.
Nunn JH, Murray JJ, Reynolds P, Tabari D, Breckon J. The prevalence of developmental defects of enamel in 15–16-year-old children residing in three districts (natural fluoride, adjusted fluoride, low fluoride) in the north east of England. Community Dent Health. 1992;9(3):235–47.
Fyffe HE, Deery C, Pitts NB. Developmental defects of enamel in regularly attending adolescent dental patients in Scotland; prevalence and patient awareness. Community Dent Health. 1996;13(2):76–80.
Rugg-Gunn AJ, al-Mohammadi SM, Butler TJ. Effects of fluoride level in drinking water, nutritional status, and socio-economic status on the prevalence of developmental defects of dental enamel in permanent teeth in Saudi 14-year-old boys. Caries Res. 1997;31(4):259–67.
Hiller KA, Wilfart G, Schmalz G. Developmental enamel defects in children with different fluoride supplementation–a follow-up study. Caries Res. 1998;32(6):405–11.
Dini EL, Holt RD, Bedi R. Prevalence of caries and developmental defects of enamel in 9–10 year old children living in areas in Brazil with differing water fluoride histories. Br Dent J. 2000;188(3):146–9.
Jalevik B, Noren JG, Klingberg G, Barregard L. Etiologic factors influencing the prevalence of demarcated opacities in permanent first molars in a group of Swedish children. Eur J Oral Sci. 2001;109(4):230–4.
Zagdwon AM, Toumba KJ, Curzon ME. The prevalence of developmental enamel defects in permanent molars in a group of English school children. Eur J Paediatr Dent. 2002;3(2):91–6.
Ekanayake L, van der Hoek W. Prevalence and distribution of enamel defects and dental caries in a region with different concentrations of fluoride in drinking water in Sri Lanka. Int Dent J. 2003;53(4):243–8.
Cochran JA, Ketley CE, Arnadottir IB, Fernandes B, Koletsi-Kounari H, Oila AM, et al. A comparison of the prevalence of fluorosis in 8-year-old children from seven European study sites using a standardized methodology. Community Dent Oral Epidemiol. 2004;32 Suppl 1:28–33.
Mackay TD, Thomson WM. Enamel defects and dental caries among Southland children. N Z Dent J. 2005;101(2):35–43.
Balmer RC, Laskey D, Mahoney E, Toumba KJ. Prevalence of enamel defects and MIH in non-fluoridated and fluoridated communities. Eur J Paediatr Dent. 2005;6(4):209–12.
Wong HM, McGrath C, Lo EC, King NM. Association between developmental defects of enamel and different concentrations of fluoride in the public water supply. Caries Res. 2006;40(6):481–6.
Hoffmann RH, de Sousa Mda L, Cypriano S. Prevalence of enamel defects and the relationship to dental caries in deciduous and permanent dentition in Indaiatuba, Sao Paulo, Brazil. Cad Saude Publica. 2007;23(2):435–44.
Muratbegovic A, Zukanovic A, Markovic N. Molar-incisor-hypomineralisation impact on developmental defects of enamel prevalence in a low fluoridated area. Eur Arch Paediatr Dent. 2008;9(4):228–31.
Arrow P. Prevalence of developmental enamel defects of the first permanent molars among school children in Western Australia. Aust Dent J. 2008;53(3):250–9.
Kanagaratnam S, Schluter P, Durward C, Mahood R, Mackay T. Enamel defects and dental caries in 9-year-old children living in fluoridated and nonfluoridated areas of Auckland, New Zealand. Community Dent Oral Epidemiol. 2009;37(3):250–9.
Seow WK, Ford D, Kazoullis S, Newman B, Holcombe T. Comparison of enamel defects in the primary and permanent dentitions of children from a low-fluoride District in Australia. Pediatr Dent. 2011;33(3):207–12.
Casanova-Rosado AJ, Medina-Solis CE, Casanova-Rosado JF, Vallejos-Sanchez AA, Martinez-Mier EA, Loyola-Rodriguez JP, et al. Association between developmental enamel defects in the primary and permanent dentitions. Eur J Paediatr Dent. 2011;12(3):155–8.
Robles MJ, Ruiz M, Bravo-Perez M, Gonzalez E, Penalver MA. Prevalence of enamel defects in primary and permanent teeth in a group of schoolchildren from Granada (Spain). Med Oral Patol Oral Cir Bucal. 2013;18(2):e187–93.
Vargas-Ferreira F, Zeng J, Thomson WM, Peres MA, Demarco FF. Association between developmental defects of enamel and dental caries in schoolchildren. J Dent. 2014;42(5):540–6.
Angelillo IF, Romano F, Fortunato L, Montanaro D. Prevalence of dental caries and enamel defects in children living in areas with different water fluoride concentrations. Community Dent Health. 1990;7(3):229–36.
Suckling GW, Pearce EI. Developmental defects of enamel in a group of New Zealand children: their prevalence and some associated etiological factors. Community Dent Oral Epidemiol. 1984;12(3):177–84.
de Liefde B, Herbison GP. Prevalence of developmental defects of enamel and dental caries in New Zealand children receiving differing fluoride supplementation. Community Dent Oral Epidemiol. 1985;13(3):164–7.
Hillson S, Bond S. Relationship of enamel hypoplasia to the pattern of tooth crown growth: a discussion. Am J Phys Anthropol. 1997;104(1):89–103.
King N. Prevalence and characteristics of developmental defects of dental enamel in Hong Kong. PhD thesis, The University of Hong Kong; 1990.
Giro CM. Enamel hypoplasia in human teeth; an examination of its causes. J Am Dent Assoc. 1947;34(5):310–7.
Jorgenson RJ, Yost C. Etiology of enamel dysplasia. J Pedod. 1982;6(4):315–29.
Sleiter R, von Arx T. Developmental disorders of permanent teeth after injuries of their primary predecessors. A retrospective study. Schweiz Monatsschr Zahnmed. 2002;112(3):214–9.
Hall S, Iranpour B. The effect of trauma on normal tooth development. Report of two cases. ASDC J Dent Child. 1968;35(4):291–5.
Holan G, Topf J, Fuks AB. Effect of root canal infection and treatment of traumatized primary incisors on their permanent successors. Endod Dent Traumatol. 1992;8(1):12–5.
Dixon DA. Defects of structure and formation of the teeth in persons with cleft palate and the effect of reparative surgery on the dental tissues. Oral Surg Oral Med Oral Pathol. 1968;25(3):435–46.
Kleine-Hakala M, Hukki J, Hurmerinta K. Effect of mandibular distraction osteogenesis on developing molars. Orthod Craniofac Res. 2007;10(4):196–202.
Ranta R. A review of tooth formation in children with cleft lip/palate. Am J Orthod Dentofacial Orthop. 1986;90(1):11–8.
Williamson JJ. Trauma during exodontia. An aetiologic factor in hypoplastic premolars. Br Dent J. 1966;121(6):284–9.
Kimoto S, Suga H, Yamaguchi M, Uchimura N, Ikeda M, Kakizawa T. Hypoplasia of primary and permanent teeth following osteitis and the implications of delayed diagnosis of a neonatal maxillary primary molar. Int J Paediatr Dent. 2003;13(1):35–40.
McCormick J, Filostrat DJ. Injury to the teeth of succession by abscess of the temporary teeth. J Dent Child. 1967;34(6):501–4.
Turner J. Effects of abscess arising from temporary teeth. Br J Dent Sci. 1906;49:562–4.
Brook AH, Winter GB. Developmental arrest of permanent tooth germs following pulpal infection of deciduous teeth. Br Dent J. 1975;139(1):9–11.
Lunt RC, Law DB. A review of the chronology of calcification of deciduous teeth. J Am Dent Assoc. 1974;89(3):599–606.
Knothe H, Dette GA. Antibiotics in pregnancy: toxicity and teratogenicity. Infection. 1985;13(2):49–51.
Phillips-Howard PA, Wood D. The safety of antimalarial drugs in pregnancy. Drug Saf. 1996;14(3):131–45.
Billings RJ, Berkowitz RJ, Watson G. Teeth. Pediatrics. 2004;113(4 Suppl):1120–7.
Fejerskov O, Larsen MJ, Richards A, Baelum V. Dental tissue effects of fluoride. Adv Dent Res. 1994;8(1):15–31.
Rozier RG. Epidemiologic indices for measuring the clinical manifestations of dental fluorosis: overview and critique. Adv Dent Res. 1994;8(1):39–55.
Hong L, Levy SM, Warren JJ, Broffitt B, Cavanaugh J. Fluoride intake levels in relation to fluorosis development in permanent maxillary central incisors and first molars. Caries Res. 2006;40(6):494–500.
Pendrys DG, Stamm JW. Relationship of total fluoride intake to beneficial effects and enamel fluorosis. J Dent Res. 1990;69 Spec No:529–38. Discussion 56–7.
Cutress TW, Suckling GW. Differential diagnosis of dental fluorosis. J Dent Res. 1990;69 Spec No:714–20. Discussion 21.
Curzon ME, Spector PC. Enamel mottling in a high strontium area of the U.S.A. Community Dent Oral Epidemiol. 1977;5(5):243–7.
Alaluusua S, Lukinmaa PL. Developmental dental toxicity of dioxin and related compounds–a review. Int Dent J. 2006;56(6):323–31.
Gao Y, Sahlberg C, Kiukkonen A, Alaluusua S, Pohjanvirta R, Tuomisto J, et al. Lactational exposure of Han/Wistar rats to 2,3,7,8-tetrachlorodibenzo-p-dioxin interferes with enamel maturation and retards dentin mineralization. J Dent Res. 2004;83(2):139–44.
Alaluusua S, Lukinmaa PL, Koskimies M, Pirinen S, Holtta P, Kallio M, et al. Developmental dental defects associated with long breast feeding. Eur J Oral Sci. 1996;104(5–6):493–7.
Alaluusua S, Lukinmaa PL, Vartiainen T, Partanen M, Torppa J, Tuomisto J. Polychlorinated dibenzo-p-dioxins and dibenzofurans via mother’s milk may cause developmental defects in the child’s teeth. Environ Toxicol Pharmacol. 1996;1(3):193–7.
Alaluusua S, Calderara P, Gerthoux PM, Lukinmaa PL, Kovero O, Needham L, et al. Developmental dental aberrations after the dioxin accident in Seveso. Environ Health Perspect. 2004;112(13):1313–8.
Jan J, Sovcikova E, Kocan A, Wsolova L, Trnovec T. Developmental dental defects in children exposed to PCBs in eastern Slovakia. Chemosphere. 2007;67(9):S350–4.
Kuscu OO, Caglar E, Aslan S, Durmusoglu E, Karademir A, Sandalli N. The prevalence of molar incisor hypomineralization (MIH) in a group of children in a highly polluted urban region and a windfarm-green energy island. Int J Paediatr Dent. 2009;19(3):176–85.
Laisi S, Kiviranta H, Lukinmaa PL, Vartiainen T, Alaluusua S. Molar-incisor-hypomineralisation and dioxins: new findings. Eur Arch Paediatr Dent. 2008;9(4):224–7.
Wozniak K. Developmental abnormalities of mineralization in populations with varying exposure to fluorine compounds. Ann Acad Med Stetin. 2000;46:305–15.
Giunta JL. Dental changes in hypervitaminosis D. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1998;85(4):410–3.
Lawson BF, Stout FW, Ahern DE, Sneed WD. The incidence of enamel hypoplasia associated with chronic pediatric lead poisoning. S C Dent J. 1971;29(11):5–10.
Fouda N, Caracatsanis M, Hammarstrom L. Developmental disturbances of the rat molar induced by two diphosphonates. Adv Dent Res. 1989;3(2):234–40.
Jan J, Vrbic V. Polychlorinated biphenyls cause developmental enamel defects in children. Caries Res. 2000;34(6):469–73.
Minicucci EM, Lopes LF, Crocci AJ. Dental abnormalities in children after chemotherapy treatment for acute lymphoid leukemia. Leuk Res. 2003;27(1):45–50.
Duggal MS, Curzon ME, Bailey CC, Lewis IJ, Prendergast M. Dental parameters in the long-term survivors of childhood cancer compared with siblings. Oral Oncol. 1997;33(5):348–53.
Pajari U, Lanning M. Developmental defects of teeth in survivors of childhood ALL are related to the therapy and age at diagnosis. Med Pediatr Oncol. 1995;24(5):310–4.
Crawford PJ, Aldred M, Bloch-Zupan A. Amelogenesis imperfecta. Orphanet J Rare Dis. 2007;2:17.
Arwill T, Olsson O, Bergenholtz A. Epidermolysis bullosa hereditaria. 3. A histologic study of changes in teeth in the polydysplastic dystrophic and lethal forms. Oral Surg Oral Med Oral Pathol. 1965;19:723–44.
Wright JT, Johnson LB, Fine JD. Development defects of enamel in humans with hereditary epidermolysis bullosa. Arch Oral Biol. 1993;38(11):945–55.
Jensen SB, Illum F, Dupont E. Nature and frequency of dental changes in idiopathic hypoparathyroidism and pseudohypoparathyroidism. Scand J Dent Res. 1981;89(1):26–37.
Spangler GS, Hall KI, Kula K, Hart TC, Wright JT. Enamel structure and composition in the tricho-dento-osseous syndrome. Connect Tissue Res. 1998;39(1–3):165–75. Discussion 87–94.
Jacobsen PE, Haubek D, Henriksen TB, Ostergaard JR, Poulsen S. Developmental enamel defects in children born preterm: a systematic review. Eur J Oral Sci. 2014;122(1):7–14.
Beentjes VE, Weerheijm KL, Groen HJ. Factors involved in the aetiology of molar-incisor hypomineralisation (MIH). Eur J Paediatr Dent. 2002;3(1):9–13.
Bell DS. Protean manifestations of vitamin D deficiency, part 1: the epidemic of deficiency. South Med J. 2011;104(5):331–4.
Foster BL, Nociti Jr FH, Somerman MJ. The rachitic tooth. Endocr Rev. 2014;35(1):1–34.
Flanagan N, O’Connor WJ, McCartan B, Miller S, McMenamin J, Watson R. Developmental enamel defects in tuberous sclerosis: a clinical genetic marker? J Med Genet. 1997;34(8):637–9.
Narang A, Maguire A, Nunn JH, Bush A. Oral health and related factors in cystic fibrosis and other chronic respiratory disorders. Arch Dis Child. 2003;88(8):702–7.
Rasmusson CG, Eriksson MA. Celiac disease and mineralisation disturbances of permanent teeth. Int J Paediatr Dent. 2001;11(3):179–83.
Wierink CD, van Diermen DE, Aartman IH, Heymans HS. Dental enamel defects in children with coeliac disease. Int J Paediatr Dent. 2007;17(3):163–8.
Zambrano M, Nikitakis NG, Sanchez-Quevedo MC, Sauk JJ, Sedano H, Rivera H. Oral and dental manifestations of vitamin D-dependent rickets type I: report of a pediatric case. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2003;95(6):705–9.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Anthonappa, R.P., King, N.M. (2015). Enamel Defects in the Permanent Dentition: Prevalence and Etiology. In: Drummond, B., Kilpatrick, N. (eds) Planning and Care for Children and Adolescents with Dental Enamel Defects. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-662-44800-7_2
Download citation
DOI: https://doi.org/10.1007/978-3-662-44800-7_2
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-662-44799-4
Online ISBN: 978-3-662-44800-7
eBook Packages: MedicineMedicine (R0)