Abstract
Contact urticaria syndrome (CUS) is characterized by the development of itchy wheals and/or eczematous dermatitis that usually appears within minutes after the contact with an eliciting substance. Over the years, advances in the knowledge of the pathogenesis of this condition have caused changes in its classification, diagnosis, and treatment. Management of CUS is similar to that of other diseases caused by hypersensitivity reactions. Thus, the safest and most effective measure is the complete avoidance of the particular allergen. In cases where prevention has failed and symptoms interfere too greatly with the patient’s career and/or quality of life, pharmacological agents could be used to provide symptomatic relief. Nevertheless, this pharmacological therapy could vary depending on the type of immediate contact skin reaction and its pathophysiology. Herein, we discuss the different therapeutic options available for the management of CUS and review the mechanisms by which these agents might achieve their therapeutic effects on patients with CUS.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
- Allergen
- Antihistamines
- Contact urticaria
- Contact urticaria syndrome
- Immediate contact skin reactions
- Prevention
- Protein contact dermatitis
- Urticaria
-
Contact urticaria syndrome (CUS) represents nowadays a worldwide health problem that needs a global approach.
-
A high index of suspicion is needed to elicit the clinical history that would suggest this condition.
-
CUS is treated mainly by prevention.
-
Prognosis of CUS is entirely dependent on the ability of the patient to avoid the etiological substances.
-
No standard recommendations on the use or consensus on the efficacy of pharmacological therapies for CUS currently exist.
Introduction
Contact urticaria syndrome (CUS), contact urticaria (CoU), and protein contact dermatitis (PCD) are conditions characterized by the immediate development of contact inflammatory skin reactions [1,2,3,4,5]. These reactions usually appear within minutes after contact with eliciting substances, and their signs and symptoms are determined by a wide range of factors, such as the route, duration, and extent of exposure; the sensitizing properties of the allergen; and the individual’s inherited and/or acquired susceptibility [2]. Initial presentation of the reaction mainly manifests as wheals and/or dermatitis/eczema, and usually remains in the contact area. However, symptoms connected with CUS (particularly the immunological type) may occasionally spread beyond the initial site of contact and progress to generalized urticaria, and/or systemic symptoms may develop that are similar to those found in angioedema, asthma, or anaphylactic shock [1, 2, 6,7,8] (Table 13.1).
The clinical importance of immediate contact skin reactions , which can be commonly seen in dermatology practice [9], is not only because of the aforementioned risk of developing life-threatening reactions, but also because of their relevance in the occupational setting [10, 11]. Thus, occupational CoU can account for 5% to 10% of reported cases of occupational skin diseases and can have a significant impact on the quality of life of workers, resulting in physical, psychological, and financial hardships [10, 12]. For these reasons, early and proper diagnosis, and appropriate management of patients when they have been correctly diagnosed, is mandatory to avoid such undesirable consequences.
The purpose of this chapter is to discuss the different therapeutic options available for the management of CUS, review the mechanisms by which these treatments might achieve their therapeutic effects on patients with CUS, and propose a simple and effective algorithm for the management and treatment of this condition.
Pathogenic Mechanisms in CUS
To understand the beneficial effects of the treatments available for the management of immediate contact skin reactions , knowledge of the mechanisms involved in the pathogenesis of the disease is of utmost importance. Although such mechanisms are not yet fully understood, the general classification distinguishes these types [7].
-
1.
Immunological contact urticaria (ICoU) . ICoU is a type I immunoglobulin E (IgE)-mediated hypersensitivity reaction in which the patient’s immune system has been previously sensitized to the eliciting substance [1, 9, 13, 14]. Thus, after the initial binding of allergen-bound IgE to mast cells and basophils, histamine (mainly) and other inflammatory mediators are released, causing the itch, inflammation, and swelling in the skin [1, 5, 8]. Therefore, the release of histamine is the central mechanism in the pathogenesis of this type of CoU.
-
2.
Nonimmunological contact urticaria (NICoU) . NICoU is probably the most common form of the disease, does not require presensitization to an allergen, and causes the skin reaction without the involvement of immunological processes [12]. In these cases, it is presumed that some urticants may cause the epidermal release of vasoactive substances, such as prostaglandins and leukotrienes, and a non-IgE-mediated histamine release from mast cells after a direct insult to the local blood vessels [1, 12, 15, 16]. Because of the lack of response to antihistamines , histamine is not considered the main inflammatory mediator involved in this type of CoU [5, 17]. Instead, because oral and topical nonsteroidal antiinflammatory drugs (NSAIDs ) can provide a satisfactory clinical response, prostaglandins and leukotrienes are considered the main agents in NICoU [18,19,20,21].
-
3.
Idiopathic or CoU of unknown origin. A third category of CoU reactions also exists for substances that elicit mixed features of both NICoU and ICoU, or where the mechanism remains uncertain [9, 15].
Furthermore, in cases of PCD , which is a particular type of immediate skin reaction caused by the recurrent exposure to high molecular weight proteins [22], a different molecular mechanism could be identified. Although its pathogenesis remains unclear, it may involve a type I hypersensitivity reaction, type IV (cell-mediated delayed) hypersensitivity reaction, and/or a delayed reaction from IgE-bearing Langerhans cells, similar to that which is observed in atopic dermatitis [22,23,24]. Such mechanisms could explain the clinical features observed in PCD (typically presents with hand and fingertip eczematous dermatitis) and the symptomatic relief achieved with topical corticosteroids or nonsteroidal topical immunomodulators [22, 25]. Finally, it should be also noted that PCD and CoU can be induced by the same allergen through immunological processes and can occasionally be present in the same patient [2].
Management and Treatment of Contact Urticaria Syndrome
Management of CUS is similar to that of other diseases caused by hypersensitivity reactions. Thus, the safest and most effective measure is the complete avoidance of the particular allergen. In other words, CUS is mainly treated by prevention [26]. Therefore, once the culprit substance is identified, the patient should be advised to avoid that substance and potential cross-reacting substances. Complete avoidance of the allergen is not always feasible, however, especially in the occupational setting. In those cases where prevention has failed and the symptoms interfere with the patient’s career and/or quality of life, pharmacological agents could be used to provide symptomatic relief [4]. Importantly, the first-line medications depend on the type of immediate contact skin reaction , but the overall goal is to inhibit the release of the inflammatory mediators involved in the pathogenesis of the disease.
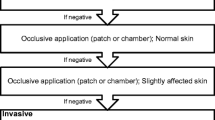
Because preventive measures solve most cases of CUS, there is a lack of published experience regarding the management of this condition with topical or systemic drugs. These therapeutic options, which are discussed next, are similar to those used for chronic urticaria (in cases of CoU) or chronic eczema (in cases of PCD) (Table 13.2). However, no standard recommendations on the use or consensus on the efficacy of these therapies currently exist for CUS. A simple and practical algorithm for the management of CUS is proposed in Fig. 13.1.
Prevention
As previously mentioned, emphasis for treatment of CUS should be placed on prevention, which remains the desideratum of therapy [26]. A thorough history and appropriate clinical testing will help determine the responsible substances. Afterward, patients must be educated on their disorder, understand its possible evolution over time, and be aware of the therapeutic options available. Furthermore, in cases of ICoU, it is also important to stress that recurrent exposures to the eliciting substances can precipitate the progression to subsequent stages of the disease and therefore a greater risk of development systemic symptoms and/or life-threatening reactions [6, 8].
Primary prevention (which aim is to avert the onset of disease, for example, by the replacement of the responsible substances with less harmful materials) and secondary prevention (which aim is to detect and treat early stages of the disease, to prevent relapses or chronicity, and/or to induce behavioral change) are highly recommended for occupational CUS [10, 11, 27]. If the responsible substance cannot be replaced or eliminated, then an adequate control must be implemented to prevent worker exposure to the allergen. Other recommended measures may include providing personal protective equipment to the workers to reduce allergen exposure (e.g., in cases of latex-induced CoU, the use of polyvinyl or nitrile gloves could be a useful alternative if rubber hypersensitivity has been appropriately detected among these patients) and making after-work creams readily available in the workplace and encouraging workers to use them regularly, ensuring physician assessment when appropriate [10, 11].
Importantly, the prognosis of CUS is entirely dependent on the ability of the patient to avoid etiological substances [28]. Thus, even in cases of severe ICoU, the long-term prognosis can be good if patients take an active role in controlling their environment and taking all proper precautions.
Antihistamines
Because histamine release has a key role in the pathogenesis of ICoU, antihistamines are considered the first-line therapy for this disorder, as for chronic urticaria [2, 4, 6, 29,30,31] (Table 13.3). These agents can also provide symptomatic relief in cases of PCD , reducing the itch and burning sensation of the eczematous dermatitis. Antihistamines act as inverse agonists of the histamine receptor, modifying the balance that normally exists between the active and inactive state of the receptor, stabilizing the inactive conformation [32]. Thereby, the local and systemic effects of histamine on target organs are blocked. Some of the antihistamines have additional antiinflammatory properties, reducing the expression of cell adhesion molecules, interfering in the recruitment of inflammatory cells such as neutrophils and eosinophils, and/or inhibiting the secretion of other inflammatory mediators, such as eicosanoids and leukotrienes [33, 34].
Second-generation H1 antihistamines are currently preferred to their predecessors as these have a better safety profile (fewer side effects such as drowsiness, psychomotor impairment, or anticholinergic effects), greater receptor specificity, greater efficacy, and longer duration of action [35]. It should be noted that the dose of H1 antihistamines should be increased (up to fourfold the standard dose) for maximum symptom control before considering alternative therapies, so long as no adverse effects are intolerable [29]. On the other hand, because 15% of histamine receptors in the skin are H2 type, H2 antihistamines may also have some effect on the symptoms of CoU [5, 36]. However, these drugs should not be used as monotherapy because they have only minimal effects on pruritus [33, 37], and current guidelines do not recommend H2 antihistamines as an evidence-based treatment in urticaria [29].
Topical Immunomodulation: Corticosteroids and Calcineurin Inhibitors
Considering that the hands are the site most frequently affected in cases of PCD, current guidelines for the management and treatment of hand eczema could be also used for this condition [38]. Thus, topical immunomodulation with high-potency corticosteroids should be considered the first-line therapy for PCD , as this method may decrease inflammation, accelerate the healing process, and inhibit the nuclear expression of genes that promote the synthesis of proinflammatory interleukins and cytokines [22, 39]. However, the likelihood of developing numerous side effects (such as skin atrophy, purpura, stretch marks, and possible alteration of intrinsic adrenocortical production) make these agents a poor long-term option. In cases of PCD with no satisfactory clinical response to topical steroids and/or with chronic cutaneous symptoms, topical calcineurin inhibitors (TCI) have been also reported as a useful therapeutic alternative [25]. TCI, such as tacrolimus and pimecrolimus, may inhibit the phosphorylase activity of the calcium-dependent serine/threonine phosphatase calcineurin and the dephosphorylation of the nuclear factor of activated T-cell protein necessary for the expression of IL-2, IL-4, IL-5, granulocyte macrophage colony-stimulating factor (GM- CSF) , and tumor necrosis factor (TNF)-α [40], thus decreasing the local inflammation seen in such cases as atopic dermatitis. Furthermore, their mechanism of action in PCD has been also attributed to a decreased expression of the high-affinity Langerhans cells and epidermic dendritic inflammatory cells and to an inhibition of the stimulatory function of these Langerhans cells [25]. Local side effects derived from their use may include stinging, burning, soreness, or itching in the area of treated skin during the first few days of treatment. Headache, acne, folliculitis, flu-like symptoms (e.g., fever, chills, runny nose, sore throat, muscle aches), or increased sensitivity of the skin may also occur.
Nonsteroidal Antiinflammatory Drugs
Some of the most commonly reported causes of NICoU include ingredients of cosmetics and medicaments (e.g., balsam of Peru, benzoic acid, cinnamic alcohol, cinnamic aldehyde), sorbic acid (a preservative used in many foods), dimethyl sulfoxide, metals (cobalt chloride, nickel sulfate, palladium), raw meat, and fish and vegetables [1]. First-line therapies in these cases should include aspirin and NSAIDs , given the important role of prostaglandins (mainly) and leukotrienes in the pathophysiology of this type of CoU [4, 5, 26]. These agents act by blocking the cyclooxygenase enzymes, thus reducing the production of prostaglandins and thromboxanes throughout the body. As noted earlier, antihistamines are unable to inhibit the reactions caused by nonimmunological pathways.
Systemic Corticosteroids
Systemic corticosteroids should be considered when a rapid and complete disease control is necessary. Thus, a short course of oral steroids is recommended for severe cases/exacerbations of CoU [2, 4, 29, 30], generally for a maximum of 10 days, similar to its use in chronic urticaria [29]. They can be also used briefly (for a maximum of 3 weeks) to treat severe cases/exacerbations of eczematous dermatitis in cases of PCD [38]. Long-term therapy should be avoided because of their frequent side effects, such as weight gain, hyperglycemia, hypertension, osteoporosis, cataracts, and gastrointestinal bleeding.
Epinephrine
Rescue therapy, in addition to systemic corticosteroids, may include epinephrine injection, which is considered the first-line treatment in cases of anaphylaxis. For this reason, it is recommended that patients with ICoU always, in addition to medic alert tags detailing their allergens, carry an injectable epinephrine with them and receive appropriate education for its self-administration in cases of life-threatening reactions [4, 5].
Alternative Therapies
Alternative therapies such as phototherapy, leukotriene receptor antagonists, or immunosuppressive drugs, which are used for chronic idiopathic/spontaneous urticaria, have not been extensively studied for CUS. However, they could be used when first-line treatments are not sufficient to ameliorate the symptoms of CUS and these interfere too greatly with the patient’s quality of life [5]. These alternative therapies may include the following options.
-
Phototherapy
Ultraviolet (UV) radiation, which has been successfully used in chronic urticaria, may also be effective in CUS if extensive and/or chronic symptoms are present [41, 42]. Although the mechanism of action by which phototherapy exerts its therapeutic effects in patients with urticaria is not entirely understood, it has been proposed that UVA (long wave, above 340 nm) and UVB (short wave, above 300 nm) radiation may induce T-lymphocyte apoptosis, reduce mast cells and Langerhans cells in the dermis, and also inhibit the release of histamine from mast cells and basophils [43]. Adverse effects may include erythema, hyperpigmentation, polymorphic light eruption, fatigue, and premature aging of the skin.
-
Leukotriene receptor antagonists
Leukotrienes are potent mediators in urticaria that act by intensifying the inflammatory response and recruiting cells to sites of inflammation [44]. Because these mediators are allegedly involved in the pathogenesis of both NICoU and ICoU, leukotriene receptor antagonists such as montelukast and zafirlukast may in part control the symptoms of CUS with relatively few side effects [5, 45]. However, the level of evidence for recommending the use of this treatment is weak [29].
-
Immunosuppressive drugs
For patients with severe and chronic symptoms of CUS in which complete avoidance of the responsible allergens is not possible and prolonged use of systemic corticosteroids is essential, corticosteroid-sparing immunosuppressive modalities could be considered as therapeutic alternatives to ameliorate the symptoms. These agents, such as cyclosporine (3–5 mg/kg/day) and methotrexate (5–20 mg per week), may act by modulating the mast cell response or preventing the initial mast cell activation [31]. For example, it has been demonstrated that cyclosporine can downregulate type 1 helper T cells and inhibit anti-IgE-stimulated histamine release from mast cells and basophils, thereby achieving disease control in patients with chronic urticaria [46, 47]. However, long-term corticosteroid therapy is limited by the adverse side effects, which include hypertension and renal toxicity secondary to cyclosporine and bone marrow suppression and hepatitis secondary to methotrexate [29, 31].
-
Anti-IgE therapy
Omalizumab is a humanized recombinant monoclonal antibody that specifically binds to the Cε3 domain of the IgE heavy chain. This domain is the site at which IgE binds to the high-affinity IgE receptor (FcεRI) on the surface of target cells (mast cells and basophils). Thereby, omalizumab reduces the levels of free IgE and the density of the FcεRI receptor, both of which are essential in the activation (and consequently degranulation) of mast cells and basophils [48, 49]. Omalizumab was approved in 2014 to be administered subcutaneously every 4 weeks for the treatment of chronic spontaneous urticaria refractory to H1 antihistamines . During the past years, its efficacy has been also demonstrated for patients suffering from different types of chronic inducible urticarias, such as symptomatic dermographism, cold urticaria, and solar urticaria [50,51,52]. Similarly, and although there is no current evidence available on CUS, omalizumab may be a potentially interesting treatment for a certain subset of patients with severe and refractory ICoU because IgE has a key role in the pathophysiology of this condition. Side effects derived from its use are usually mild or moderate, including headache, nasopharyngitis, sinusitis, nausea, diarrhea, and local symptoms at the site of injection [49].
-
Immunotherapy
Finally, another alternative treatment that shows promise in controlling symptoms of CUS when complete avoidance of the responsible allergens is not possible is the induction of tolerance through immunotherapy. In this sense, an Italian group reported a satisfactory clinical improvement among bakers and pastry makers with occupational disease from wheat flour sensitization by using specific immunotherapy: 83% of the patients were still at work and 70% claimed only weak or no symptoms during work years after this treatment [53]. Regarding its safety profile, Nettis et al. evaluated the tolerability of sublingual immunotherapy with latex extract among patients with latex-induced CoU in a double-blind, placebo-controlled study [54]. Their results supported the safety of this specific immunotherapy as no statistically significant differences were found between the proportions of adverse events in both the latex-induced CoU group and the placebo group. Nevertheless, these experimental immunotherapies for select urticants are undergoing evaluation and they are not currently available for widespread use.
Conclusions
The avoidance of the responsible/suspected allergen whenever possible is considered the mainstay of the treatment of CUS. First-line medications used to provide symptomatic relief depend on the type of immediate contact skin reaction and its pathophysiology. Thus, antihistamines are considered the best treatment for CoU in which an immunological mechanism is suggested, whereas acetylsalicylic acid and NSAIDs are reserved for cases of NICoU. Topical immunomodulation should be used in cases of CUS presented with eczematous dermatitis (PCD ). Alternative systemic therapies, such as phototherapy, leukotriene receptor antagonists, immunosuppressive drugs, and omalizumab, can be considered in cases of severe or chronic CUS; however, the level of evidence for recommending the use of these agents in CUS is weak. Further biochemical research is certainly required to definitively illustrate the immunologic signaling and cellular mechanisms activated by contact urticants.
References
Wakelin SH. Contact urticaria. Clin Exp Dermatol. 2001;26(2):132–6.
Gimenez-Arnau A, Maurer M, De La Cuadra J, Maibach H. Immediate contact skin reactions, an update of contact urticaria, contact urticaria syndrome and protein contact dermatitis – “A never ending story”. Eur J Dermatol. 2010;20(5):552–62.
Maibach HI, Johnson HL. Contact urticaria syndrome. Contact urticaria to diethyltoluamide (immediate-type hypersensitivity). Arch Dermatol. 1975;111(6):726–30.
Aquino M, Mawhirt S, Fonacier L. Review of contact urticaria syndrome- evaluation to treatment. Curr Treat Options Allergy. 2015;2(4):365–80.
Bhatia R, Alikhan A, Maibach HI. Contact urticaria: present scenario. Indian J Dermatol. 2009;54(3):264–8.
Wang CY, Maibach HI. Immunologic contact urticaria- the human touch. Cutan Ocul Toxicol. 2013;32(2):154–60.
Von Krogh G, Maibach HI. The contact urticaria syndrome- an updated review. J Am Acad Dermatol. 1981;5(3):328–42.
McFadden J. Immunologic contact urticaria. Immunol Allergy Clin N Am. 2014;34(1):157–67.
Verhulst L, Goossens A. Cosmetic components causing contact urticaria: a review and update. Contact Dermatitis. 2016;75(6):333–44.
Nicholson PJ, Llewellyn D, English JS, Guidelines Development Group. Evidence-based guidelines for the prevention, identification and management of occupational contact dermatitis and urticaria. Contact Dermatitis. 2010;63(4):177–86.
Adisesh A, Robinson E, Nicholson PJ, Sen D, Wilkinson M, Standards of Care Working Group. U.K. standards of care for occupational contact dermatitis and occupational contact urticaria. Br J Dermatol. 2013;168(6):1167–75.
Chowdhury MMU. Occupational contact urticaria: a diagnosis not to be missed. Br J Dermatol. 2015;173(6):1364–5.
Amaro C, Goossens A. Immunological occupational contact urticaria and contact dermatitis from proteins: a review. Contact Dermatitis. 2008;58(2):67–75.
Dean AM, Secrest AM, Powell DL. Contact urticaria from occupational exposure to formaldehyde. Dermatitis. 2016;27(4):232.
Saluja SS, Davis CL, Chong TA, Powell DL. Contact urticaria to nickel: a series of 11 patients who were prick test positive and patch test negative to nickel sulfate 2.5% and 5.0%. Dermatitis. 2016;27(5):282–7.
Gomułka K, Panaszek B. Contact urticaria syndrome caused by haptens. Postepy Dermatol Alergol. 2014;31(2):108–12.
Venarske D, deShazo RD. Molecular mechanisms of allergic disease. South Med J. 2003;96(11):1049–54.
Lahti A, Oikarinen A, Viinikka L, Ylikorkala O, Hannuksela M. Prostaglandins in contact urticaria induced by benzoic acid. Acta Derm Venereol. 1983;63(5):425–7.
Lahti A, Väänänen A, Kokkonen EL, Hannuksela M. Acetylsalicylic acid inhibits non-immunologic contact urticaria. Contact Dermatitis. 1987;16(3):133–5.
Johansson J, Lahti A. Topical non-steroidal anti-inflammatory drugs inhibit non-immunologic immediate contact reactions. Contact Dermatitis. 1988;19(3):161–5.
Morrow JD, Minton TA, Awad JA, Roberts LJ. Release of markedly increased quantities of prostaglandin D2 from the skin in vivo in humans following the application of sorbic acid. Arch Dermatol. 1994;130(11):1408–12.
Levin C, Warshaw E. Protein contact dermatitis: allergens, pathogenesis, and management. Dermatitis. 2008;19(5):241–51.
Kanerva L, Estlander T. Immediate and delayed skin allergy from cow dander. Am J Contact Dermat. 1997;8(3):167–9.
Conde-Salazar L, González MA, Guimaraens D. Type I and Type IV sensitization to Anisakis simplex in 2 patients with hand eczema. Contact Dermatitis. 2002;46(6):361.
Mercader P, de la Cuadra-Oyanguren J, Rodríguez-Serna M, Pitarch-Bort G, Fortea-Baixauli JM. Treatment of protein contact dermatitis with topical tacrolimus. Acta Derm Venereol. 2005;85(6):555–6.
Giménez-Arnau A. Contact urticaria and the environment. Rev Env Health. 2014;29(3):207–15.
Alfonso JH, Bauer A, Bensefa-Colas L, Boman A, Bubas M, Constandt L, et al. Minimum standards on prevention, diagnosis and treatment of occupational and work-related skin diseases in Europe - position paper of the COST Action StanDerm (TD 1206). J Eur Acad Dermatol Venereol. 2017;31(Suppl 4):31–43.
Mälkönen T, Jolanki R, Alanko K, Luukkonen R, Aalto-Korte K, Lauerma A, et al. A 6-month follow-up study of 1048 patients diagnosed with an occupational skin disease. Contact Dermatitis. 2009;61(5):261–8.
Zuberbier T, Aberer W, Asero R, Bindslev-Jensen C, Brzoza Z, Canonica GW, et al. The EAACI/GA(2) LEN/EDF/WAO Guideline for the definition, classification, diagnosis, and management of urticaria: the 2013 revision and update. Allergy. 2014;69(7):868–87.
Magerl M, Altrichter S, Borzova E, Giménez-Arnau A, Grattan CE, Lawlor F, et al. The definition, diagnostic testing and management of chronic inducible urticarias – The EAACI/GA(2) LEN/EDF/UNEV consensus recommendations 2016 update and revision. Allergy. 2016;71(6):780–802.
Deza G, Giménez-Arnau AM. Itch in urticaria management. Curr Probl Dermatol. 2016;50:77–85.
Leurs R, Church MK, Taglialatela M. H1-antihistamines: inverse agonism, anti-inflammatory actions and cardiac effects. Clin Exp Allergy. 2002;32(4):489–98.
Jáuregui I, Ferrer M, Montoro J, Dávila I, Bartra J, del Cuvillo A, et al. Antihistamines in the treatment of chronic urticaria. J Investig Allergol Clin Immunol. 2007;17(2):41–52.
Khalaf AT, Li W, Jinquan T. Current advances in the management of urticaria. Arch Immunol Ther Exp. 2008;56(2):103–14.
Simons FE. Advances in H1-antihistamines. N Engl J Med. 2004;351(21):2203–17.
Thurmond RL, Kazerouni K, Chaplan SR, Greenspan AJ. Antihistamines and itch. Handb Exp Pharmacol. 2015;226:257–90.
Lee EE, Maibach HI. Treatment of urticaria. An evidence-based evaluation of antihistamines. Am J Clin Dermatol. 2001;2(1):27–32.
Diepgen TL, Andersen KE, Chosidow O, Coenraads PJ, Elsner P, English J, et al. Guidelines for diagnosis, prevention and treatment of hand eczema. J Dtsch Dermatol Ges. 2015;13(1):e1–22.
Kaplan AP. Treatment of chronic urticaria: approaches other than antihistamines. In: Kaplan A, Greaves M, editors. Urticaria and angioedema. New York: Informa Healthcare; 2009. p. 365–72.
Lebwohl MG, Del Rosso JQ, Abramovits W, Berman B, Cohen DE, Guttman E, et al. Pathways to managing atopic dermatitis: consensus from the experts. J Clin Aesthetic Dermatol. 2013;6(Suppl 7):S2–18.
Kozel MMA, Sabroe RA. Chronic urticaria: aetiology, management and current and future treatment options. Drugs. 2004;64(22):2515–36.
Rombold S, Lobisch K, Katzer K, Grazziotin TC, Ring J, Eberlein B. Efficacy of UVA1 phototherapy in 230 patients with various skin diseases. Photodermatol Photoimmunol Photomed. 2008;24(1):19–23.
Hannuksela M, Kokkonen EL. Ultraviolet light therapy in chronic urticaria. Acta Derm Venereol. 1985;65(5):449–50.
Maxwell DL, Atkinson BA, Spur BW, Lessof MH, Lee TH. Skin responses to intradermal histamine and leukotrienes C4, D4, and E4 in patients with chronic idiopathic urticaria and in normal subjects. J Allergy Clin Immunol. 1990;86(5):759–65.
Sanada S, Tanaka T, Kameyoshi Y, Hide M. The effectiveness of montelukast for the treatment of anti-histamine-resistant chronic urticaria. Arch Dermatol Res. 2005;297(3):134–8.
Altman K, Chang C. Pathogenic intracellular and autoimmune mechanisms in urticaria and angioedema. Clin Rev Allergy Immunol. 2013;45(1):47–62.
Marsland AM, Soundararajan S, Joseph K, Kaplan AP. Effects of calcineurin inhibitors on an in vitro assay for chronic urticaria. Clin Exp Allergy. 2005;35(5):554–9.
Wright JD, Chu HM, Huang CH, Ma C, Chang TW, Lim C. Structural and physical basis for anti-IgE therapy. Sci Rep. 2015;5:11581.
McCormack PL. Omalizumab: a review of its use in patients with chronic spontaneous urticaria. Drugs. 2014;74(14):1693–9.
Aubin F, Avenel-Audran M, Jeanmougin M, Adamski H, Peyron JL, Marguery MC, et al. Omalizumab in patients with severe and refractory solar urticaria: a phase II multicentric study. J Am Acad Dermatol. 2016;74(3):574–5.
Metz M, Schütz A, Weller K, Gorczyza M, Zimmer S, Staubach P, et al. Omalizumab is effective in cold urticaria-results of a randomized placebo-controlled trial. J Allergy Clin Immunol. 2017;140:864. https://doi.org/10.1016/j.jaci.2017.01.043.
Maurer M, Schütz A, Weller K, Schoepke N, Peveling-Oberhag A, Staubach P, et al. Omalizumab is effective in symptomatic dermographism-results of a randomized placebo-controlled trial. J Allergy Clin Immunol. 2017;140:870. https://doi.org/10.1016/j.jaci.2017.01.042.
Cirla AM. Asthma and baker’s allergy: experience with health programs. G Ital Med Lav Ergon. 2011;33(1):20–5.
Nettis E, Di Leo E, Calogiuri G, Milani M, Delle Donne P, Ferrannini A, et al. The safety of a novel sublingual rush induction phase for latex desensitization. Curr Med Res Opin. 2010;26(8):1855–9.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG, part of Springer Nature
About this chapter
Cite this chapter
Deza, G., Giménez-Arnau, A.M. (2018). Management and Treatment of Contact Urticaria Syndrome. In: Giménez-Arnau, A., Maibach, H. (eds) Contact Urticaria Syndrome. Updates in Clinical Dermatology. Springer, Cham. https://doi.org/10.1007/978-3-319-89764-6_13
Download citation
DOI: https://doi.org/10.1007/978-3-319-89764-6_13
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-89763-9
Online ISBN: 978-3-319-89764-6
eBook Packages: MedicineMedicine (R0)