Abstract
The various polyphenol families present in wine are important for a number of technological properties of wine such as clarity, hue, and palatal taste. Dietary polyphenols are associated with a wide range of health benefits, protecting against chronic diseases and promoting healthy aging. However, basic and clinical science is showing that the reality is much more complex than this and that several issues, notably daily intake, bioavailability, or in vivo antioxidant activity, are yet to be resolved. The concentration of phenolic compounds in wine is determined by viticulture and vinification practices, peculiar of different countries. Interesting are the effects of different yeast strains on the final concentration of polyphenols in red wine. We here summarize the recent findings concerning the effects of specific classes of polyphenol (soluble acids, flavonols, and stilbenes) on human health and propose future directions for research to increase the amount of these healthy compounds in wine.
Access provided by Autonomous University of Puebla. Download reference work entry PDF
Similar content being viewed by others
Keywords
1 Introduction
Wine contains a large and diverse class of phenolic compounds known variously as “polyphenols” or “biophenols.” These compounds contribute to the characteristic colors and flavors of wine and act as natural wine preservatives that allows a long aging process [1]. Polyphenols are extracted during crushing and fermentation when the juice is in contact with the grape skins and seeds. The amount of phenolic compounds in red wine is about six times higher than that in white wine because red juice has longer contact time with the grape skins and seeds. The concentration of polyphenolic compounds in red wine is approximately 1800–3000 mg/L [2].
There is emerging evidence that a functional diet can help in modulating the immune system responses to the inflammation processes through a variety of mechanisms based on the absorption and utilization by the human metabolism of specific compounds.
Polyphenols are the principal compounds related to the wine consumption benefits due to their antioxidant and free radical scavenging properties. This class of compounds has been proven to exert important health effects, acting against cancer pathologies [3] as well as reactive oxygen species (ROS) which are considered the main cause of different cardiovascular and neurodegenerative diseases.
Grapevine (Vitis spp) is the most cultivated fruit crop in the world, with an area dedicated to viticulture. The berries are harvested primarily for winemaking but also to provide fresh table grapes, raisins, and other minor products. Phenolic composition of grape is genetically driven and greatly affected by environmental factors. A major challenge for breeding of grapevine cultivars adapted to climate change and with high potential for winemaking is to dissect the complex plant metabolic response involved in adaptation mechanisms. Among plant products are the polyphenols, a large family of secondary metabolites, which are involved in plant responses to biotic and abiotic stresses. The most represented polyphenols in plants are the flavonoids, the cinnamic and benzoic acids, and the stilbenes. They derive from the phenylpropanoid metabolism, but flavonoids are ubiquitous in plants, whereas stilbenes are specific to certain plant families.
Other polyphenols, as the phytoalexins, are antimicrobial compounds synthesized in response to pathogen or herbivore attack. However, other roles have been described for stress-induced polyphenols, including the defense signaling of responses and protection against ultraviolet (UV) light damage [4, 5].
Phenolic compounds are secondary metabolites synthesized [4] during normal development of the berry grape in response to stress conditions.
A large-scale experiment involving cultivation of an association panel of a large number (more than 200) V. vinifera cultivars designed to represent the genetic and phenotypic variation encountered in cultivated grapevine and metabolomics analysis targeted to a large number of polyphenolic compounds (polyphenomics) was performed. Chemometrics analysis of the data showed large differences in polyphenol composition related to genetic factors, environmental factors (i.e., water stress), and genetic-environment interactions. Correlation networks shed light on the relationships between the different polyphenol metabolites and related biosynthetic pathways. In addition, detailed polyphenomics analysis confirmed that polyphenol reactions described in wine take place in the berries.
Finally, was reported a large-scale study demonstrating an influence of environmental influence (water stress) on the different classes of polyphenols but also cultivar differences in the types and extents of drought responses, with different molecules affected either positively or negatively and different impacts on grape and wine quality [6].
Grape phenolic compounds comprise several families, divided between non-flavonoids (hydroxybenzoic acids, hydroxycinnamic acids, and stilbenes) and flavonoids, based on the same C6-C3-C6 skeleton (flavonols, dihydroflavonols, flavan-3-ols, and anthocyanins). Each family is represented by several compounds differing by their hydroxylation level and by substitution of the hydroxy groups (methylation, glycosylation, acylation). For example, anthocyanins, the red grape pigments, are based on six aglycones, which can be mono- or di-glucosylated and further acylated with acetic, p-coumaric, and caffeic acid, giving rise to a large number of compounds [6]. Grape flavan-3-ols also show high diversity. They include several monomers (catechin, epicatechin, gallocatechin, epigallocatechin, and epicatechin 3-gallate) that are the constitutive units of oligomers and polymers (proanthocyanidins or condensed tannins), with degrees of polymerization ranging from 2 to over 100 in grape skin [6].
The flavonoid family includes the flavonols, such as myricetin, quercetin, and kaempferol, which exist both as aglycones and sugar conjugates. The non-flavonoids encompass hydroxybenzoic acids such as gallic acid, hydroxycinnamic acids, including p-coumaric, caffeic, and caftaric acids, and the stilbenes, such as trans-resveratrol and cis-resveratrol [5]. The synthesis of stilbenes in grape berry tissues is activated in response to fungal attack, to berry injury, and to ultraviolet irradiation [5].
The healthy physiological effects are especially associated to flavonoids and stilbenes [7], namely, quercetin, (+)-catechin, gallic acid, and trans-resveratrol [8]. The stilbene trans-resveratrol has gained great attention, and a number of scientific papers have appeared related to the moderate consumption of red wine for its ability to inhibit platelet aggregation and LDL oxidation and its beneficial effects in health. Since trans-resveratrol is postulated to be involved in the health benefits associated with a moderate consumption of red wine, it is one of the most extensively studied natural products.
The various polyphenol families present in wine [7, 9] are important for a number of technological properties of wine [7]. The knowledge about their qualitative and quantitative profile in grapes is very important to predict wine aging attitude and can help to solve problems related to color stability, especially in the case of red wines that are destined to long aging periods [10]. The wine aging also changes the phenolic composition, as these compounds can suffer diverse transformations, like oxidation processes, condensation and polymerization reactions, and extraction from wood, usually associated to the changes in color and colloidal stability [11], flavor, bitterness, and astringency [12, 13]. The polyphenolic fingerprint can be useful for the classification of wines, since it can give us information about the variety, the geographic and winery origin, and even the applied winemaking technology [14].
During winemaking, only a fraction of the grape flavonoids is selectively transferred to the wine and a final yield strongly depending on the management of the contact of the liquid must, containing berry skin and seeds, with the solid parts of the grape bunches and on the grape variety [10].
The data concerning the extractable phenolics of the grape cannot be simply generalized to predict the wine composition, since a high variability in the extraction yield from grape to wine is introduced by the technological factors governing the winemaking process (such as temperature, duration and intensity of the liquid-solid contact, final ethanol concentration).
Many conditions (i.e., genetic, agronomic, technological, storage, etc.) linked to each other by complex and multifactorial phenomena affect both profiles and concentrations of bioactive compounds, either in grape or in wine [15].
2 Soluble Acids, Flavonols, and Stilbenes: Bioactive Compounds in Wine
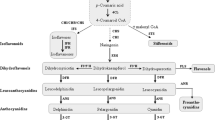
Wine is a complex mixture of hundreds of molecules, some of them showing important biological properties, while others are mainly associated with its organoleptic characteristics. In particular, we describe specific classes of polyphenols such as phenolic acids (hydroxybenzoic and hydroxycinnamic acids), flavonols, and stilbenes (Fig. 1).
2.1 Phenolic Acids
In wine, there are two groups of phenolic acids: hydroxybenzoic acids and hydroxycinnamic acids [16]. Hydroxybenzoic acids, including gallic acid, protocatechuic acid, gentisic acid, p-hydroxybenzoic acid, vanillic acid, and syringic acid, derive from benzoic acid. Chlorogenic acid, as the main constituent of the hydroxycinnamic acid derivative group, increased with harvest time delay, and the same occurred with sinapic acid. The converse was true of caffeic acid and ferulic acid, which were also esterified with tartaric acid as the known compounds caftaric acid and fertaric acid, respectively [16, 17]. Hydroxycinnamic acids have gained an increasing interest in health because they are known to be potent antioxidants.
These compounds have been described as chain-breaking antioxidants acting through radical scavenging activity that is related to their hydrogen or electron donating capacity and to the ability to delocalize/stabilize the resulting phenoxyl radical within their structure. The free radical scavenger ability of antioxidants can be predicted from standard one-electron potentials.
Phenolic acids represent important fraction of wine phenolics, but their biological effects have been scarcely investigated. The interrelationship between antioxidative capacity and vasodilatory activity, two potentially beneficial biological effects, of phenolic acids from wine was examined. Antioxidative and vasodilatory effects of phenolic acids relate to the number of hydroxyl groups in the phenyl ring, degree of compactness and branching of molecules, and three-dimensional distributions of atomic polarizability of the tested molecules [18]. Caffeic acid has been shown to have neuroprotective effects against injury induced by 5-S-cysteinyl-dopamine, against Aβ-induced neurotoxicity and by inhibiting peroxynitrite-induced neuronal injury [19, 20]. Ferulic acid has been cited as an antidiabetic effect by lowering blood glucose and by increasing plasma insulin [21].
2.2 Flavonols
Four aglycones belong to the flavonols class: myricetin, quercetin, kaempferol, and isorhamnetin. The diversity in the flavonols structure is due to changes in the basic skeleton introduced by enzymes such as glycosyl transferase, methyl transferase, and rhamnosyl-transferase. In one plant species, dozens of different flavonoids may be present, and many of these are conjugated to various types of sugars. Both the basic structure and the level of glycosylation determine the biological function and bioavailability of polyphenols in the human and animal intestines. Depending on their structure, these molecules have diversified activities.
Flavonols are found throughout the plant foods. The best-known flavonols are quercetin and kaempferol. Quercetin, kaempferol, myricetin, and isorhamnetin are flavonols presents in grape skins and stems as several different glycosides. Quercetin accumulates in grape skins to protect against damage from ultraviolet (UV) light. There are high concentrations of quercetin in wine made from sun-exposed grapes. Quercetin is readily extracted from grape skins during fermentation. Stems may contribute additional flavonols in whole cluster fermentations.
Quercetin glycosides may be hydrolyzed in wine to form quercetin aglycone. This process is similar to the hydrolysis that can occur with anthocyanins. Unlike anthocyanins, flavonol aglycones are stable in wine and can be used to monitor hydrolysis reactions. Quercetin may cause problems as a precipitate in bottled wines. Flavonols can interact with anthocyanins, enhancing their red color in a process known as co-pigmentation. This process may also help anthocyanin color stability.
Dietary flavonols inhibit LDL oxidation and so reduce the primary risk factor for atherosclerosis and related diseases. The animal studies are supported by human epidemiological studies, which show inverse correlations between the occurrence of CVD, certain cancers, and age-related degenerative diseases and the consumption of flavonol-rich diets [22,23,24,25]. Flavonols have been linked to protective effects against several specific cancers, including leukemia and pancreatic, breast, cervical, prostate, uterine, and urinary tract cancers. Subjects with regular flavonol intake have a 10–60% lower incidence of these types of cancer compared with subjects with low flavonol intake.
This protective activity results from both the action of flavonols as stimulators of antioxidant defenses and their direct inhibitory effects on cellular proliferation. Quercetin consumption has been reported to be inversely associated with breast cancer incidence [26].
2.3 Stilbenes
Stilbenes are a class of compounds with multiple pharmaceutically relevant properties. The stilbenes production in grape berry tissues is considered to be a part of the general defense mechanism since they display strong antifungal and antimicrobial activities [27].They are a group of plant phytoalexin polyphenols found in high concentrations in grapes, berries, nuts, and teas. In plants, their main function is to protect the plants against pathogens and fungi; therefore, their content is highly variable and increases with stress exposure. UV radiation, heavy metal exposure, and fungal infection may thus be utilized to enrich grapes with stilbenoids [28, 29].
Resveratrol (3,4_,5-trihydroxystilbene) may be found especially in red wine, grapes, and berries, at a concentration ranging from 0.1 to 15 mg/L. Nowadays, the primary source of resveratrol is the roots of perennial plant Polygonum cuspidatum (Japanese knotweed) [29]. Due to the positive health effects attributed to resveratrol through so-called French paradox, its biological activity has been extensively studied. Most biological effects are assigned to trans-resveratrol, the more stable of the two isoforms. However, also cis-resveratrol, which is formed from trans-resveratrol upon UV light exposure, exhibits certain anti-inflammatory activity. Unless otherwise stated, the activities mentioned here apply to trans-resveratrol. Stilbenes are non-nitrogen polyphenols with acidic and amphiphilic character which causes their enrichment in biomembranes, where many of their targets occur (COX, 5-LOX, protein kinase B) [30]. Structurally, stilbenoids possess two aromatic rings connected by an ethylene or ethene bridge with a variety of substituents. Even though the presence of double bond suggests that stilbenoids exist in cis- as well as trans-form, the trans-form is more stable and the biologically relevant form. In nature, stilbenoids are synthesized from phenylalanine through multiple enzymatic reactions [31]. Stilbenoids are heterogeneously spread throughout the plant kingdom. They are especially abundant in Gnetaceae, Pinaceae, Cyperaceae, Fabaceae, Dipterocarpaceae, and Vitaceae families [32]. Resveratrol has a function of phytoalexin produced by specific plant species in response to biotic and abiotic challenges. It is thought to be one of the principal agents in the health-promoting effects of red wine [33]. Results of clinical studies show that the most important source of resveratrol and piceid are wines (98.4%) and grape and grape juices (1.6%), whereas peanuts, pistachios, and berries contribute to less than 0.01% [34]. Wine is the major source of resveratrol and piceid to the diet, ranging from 95% and 98.7% for trans-resveratrol and trans-piceid to 99.9% and 99.7% for cis-resveratrol and cis-piceid, respectively. Other food items such as grapes contribute by amount of 3.8% of total trans-resveratrol, whereas other contributors such as pistachios or berries provide less than 1% of the dietary total amount of trans-resveratrol and trans-piceid.
Resveratrol possesses numerous important bioactivities, including anti-inflammatory, antioxidant, anti-aggregatory functions, and modulation of lipoprotein metabolism [35,36,37]. It has also been shown to detain chemo preventive properties against certain forms of cancer and cardiovascular disorders and to have positive effects on longevity [38,39,40,41,42].
Anticancer activity of this compound is mainly due to induction of apoptosis via several pathways, as well as alteration of gene expressions, leading to a decrease in tumor initiation, promotion, and progression [43]. Resveratrol blocks the growth of lymphoma cells and increases their rate of cell death [44]. Resveratrol sensitizes chemotherapy-resistant lymphoma cells to treatment with paclitaxel-based chemotherapy [45], also reduces the production of growth factors, such as vascular endothelial growth factor and interleukin [33], and may reduce the ability of lymphoma cells to spread to other organs [46]. Finally, it was demonstrated that in vitro administration of resveratrol favorably altered gene expression in the androgen axis and in cell cycle regulators, providing potential anticancer benefit for prostate cancer [47]. Moreover, trans-resveratrol appears to protect against diabetes [48] and neurodegenerative disorders [49], due to induction of sirtuin 1 genes [50]. Trans-resveratrol might also contribute to increasing the life span of metazoans and mice by miming the effect of caloric restriction and thus decreasing age-related signs [51, 52]. Experimental studies have shown that resveratrol exhibits both an anti-inflammatory and cardioprotective potential by inhibiting the expression of inflammatory mediators and the monocyte adhesion to vascular endothelial cells [53, 54]. Although resveratrol exhibits potent anticancer activities against transformed cells, its effectiveness is limited by its poor bioavailability, and as a dietary phytonutrients, it is most effective against tumors with which it comes in direct contact, such as skin cancers and tumors of the gastrointestinal tract. Furthermore, inhibition of sirtuin 1 by both pharmacological and genetic means abolished protein de-acetylation and autophagy as stimulated by resveratrol, but not by piceatannol, indicating that these compounds act through distinct molecular pathways. In support of this notion, resveratrol and piceatannol synergized in inducing autophagy as well as in promoting cytoplasmic protein de-acetylation [55].
3 Technological Approaches to Enhance Polyphenol Content and Antioxidant Activity in Wine
The winemaking steps determine the phenolic content of red wines that able the extraction of phenolic compounds from the grape berries. Numerous winemaking procedures have been developed to enhance the extraction of phenolic molecules, by preventing the several motives that affect the release of polyphenols from the berry tissues [56, 57]. Several investigations have studied the effect of the fermentation temperature during the winemaking process on the extraction of polyphenols, thus demonstrating that their concentration increased when wines were produced at higher fermentation temperatures [58, 59]. Moreover, it has recently shown the efficacy of thermo-vinification to improve a number of factors, such as the antioxidant potential and the polyphenolic and resveratrol content, in Pinot noir, Prokupac, Merlot, and Cabernet Sauvignon wines [60].
The impact of maceration time and of the utilization of enological additives (enzymes, sulfur dioxide) on the polyphenol content has been studied [57]. Gambuti and coworkers [61] have examined the effects of those factors during vinification of Aglianico grape must. The authors indicated that the simultaneous addition, during the pre-fermentation stage, of pectolytic enzymes and SO2 increased the release of these molecules from grape tissues, thus resulting in a higher concentration of polyphenols in the produced wine. Comparable evidences were recently obtained by investigating experimental vinification of grape must from the Vranec cultivar [62, 63]. Also in this case, both increased maceration time and SO2 addition augmented the final concentration of total polyphenols, anthocyanins, flavonoids, and flavan-3-ols in the final product, this effect action not dependent by the wine aging.
The action of diverse winemaking approaches in determining polyphenolic profile of red wines obtained from the Italian red cultivar “Negroamaro” has been recently studied. Were compared three different pre-fermentative steps: the traditional (7 days of maceration at 25 °C), the cryomaceration, (24 h at 0 °C using dry ice), and ultrasound (37 kHz, 150 W, 15 min at 30 °C) [64]. The authors demonstrated that the ultrasound action enhanced the release of all polyphenol classes, whereas the cryomaceration only improved the anthocyanin content in the produced wine.
This evidence has been recently supported by a recent study of Ferraretto and Celotti [65]. They evaluated the effect of high-power ultrasound (20 kHz) on the phenolic structure of red wines and demonstrated that this physical treatment promote the polymerization of the phenolic compounds as the wine matures and consequently speed up the aging course of wines.
Another investigation [66] has evaluated the consequence of different technological approaches on the polyphenolic profile and the antioxidant activity of wine produced from grape of the Italian cultivar Primitivo. The addition of tannins was more efficient in enhancing the concentration of phenolic molecule when compared in musts to the other considered technologies and the aging to get better wine antioxidant activity. Furthermore, a recent report indicated that protracted post-fermentation maceration up to 50 days increased the polyphenol concentration and, consequently, the potential healthy effect of the obtained wine [67]. However, a number of reports do not agree with this hypothesis.
However in contrast with the above findings, Mulero and coworkers [68] did not detect variation in the concentrations of the different types of phenolic compounds in three wines from Monastrell grapes, produced by separately adopting the protracted maceration, must fermentation by adding enzymes, or the traditional procedure above three technological approaches.
The pulsed electric field (PEF) technology represents a very promising approach because of its ability to enhance the extraction of polyphenolic compounds throughout the vinification process. The PEF pretreatments on Cabernet Sauvignon must gave substantial differences in the produced wines, since the same PEF-treated must showed an increase of 97% in the content of total flavonols of 32% in the content of total phenolics and of 62%, in the color, when compared to the untreated control [59]. The above findings were confirmed by similar investigations that analyzed the vinification of Cabernet [69], Merlot [70], and Syrah [71] grape musts after the application of the PEF step. Moreover, PEF treatment allowed the acceleration and enhancement in the extraction of phenolic compounds throughout the maceration step of winemaking process [72].
An interesting research have recently assessed the effects of a novel fermentation technologies based on the “Ganimede” that is able to trap the carbon dioxide (CO2) generated during the alcoholic fermentation on the phenolic contents of Cabernet Sauvignon wines [73] indicating that this device was able to increase the concentration of anthocyanins superior in the wines produced.
The mechanisms through which yeast influences the color and content of polyphenolic compounds of wine are currently being researched, but three modes of interaction between the yeasts and the polyphenolic component have been already identified. Some strains of yeast adsorb polyphenols on the cell wall. However, although it has been shown that yeast is one of the factors able of inducing the reduction of the polyphenol concentration in wines, it is unknown whether their adsorption on the cell walls is the only mechanism responsible of this behavior. The amount of biomass produced during the alcoholic fermentation is capable to adsorb on the cell walls a significant amount of polyphenols. This capacity is likely to be strain-specific, since different yeast strains have a different composition of the cell wall. In fact, it could be hypothesized that specific yeast strains could perform a “differentiated” adsorption of the diverse polyphenol classes. The second type of interaction between yeast strains and the wine polyphenol is related to the microbial β-glucosidase enzymatic activity. In fact, the greatest part of anthocyanins is found in the wine as glucosidase derivatives (linked to a sugar); thus, in this state they are much less sensitive to chemical or enzymatic oxidation; therefore the β-glucosidase activity decreases color and stability, since it produces free anthocyanins in wine. The third mechanism regards the strain-specific secretion throughout the alcoholic fermentation, by some wine yeast, of polysaccharides capable to combine with the polyphenols and to form with them stabile complexes.
It has been shown that different yeast metabolites, including pyruvic acid, can react with the anthocyanins of the grapes giving rise to stable pigments during the maturation and aging of red wines [74]. Taken together, the above considerations highlight the pivotal role played by yeasts in modifying the polyphenolic profile of wines.
Indeed, several investigations have highlighted the ability of different wine yeast strains and of enological additives of microbial origin to improve the phenolic profile of red wines. The addition of β-glucanases or other yeast-derived enzyme preparations as enological additives increased the antioxidant potential of sparkling wines [75]. On the opposite, yeast lees have been demonstrated to lower the amounts of polyphenolic compounds [76] and anthocyanins [74] in wines, because they formed stabile complexes with the mannoproteins released by yeasts after their yeast.
Even though previous studies did not show any effect of yeast starters on the polyphenolic profile of Pinot noir [77], Cabernet Franc, and Merlot [78] wines, several recent reports indicated a correlation between the utilized yeast strain and the antioxidant capacity of the produced wine.
Brandolini and coworkers [79] carried out an investigation by evaluating the properties of wines produced by the separate inoculation of 19 strains of Saccharomyces cerevisiae in the same grape must. The antioxidant capacity and the polyphenol profile detected in the different wines were extremely different, thus showing the strain-specific yeast feature to differentially adsorb polyphenols during the vinification process. Kostadinović and coworkers [62] showed analogous evidence on Vranec and Merlot wines in Macedonia. The authors used different starter cultures to carry out vinification tests, where the strains demonstrated that they were capable to affect specifically the trans-resveratrol concentration and antioxidant activities in the final wines.
The employment of different yeast starter strains allowed the production of Pinot noir wines with a dissimilar polyphenolic content [80]. This study has analyzed how yeast selection can modify phenolic content in Pinot noir wine. In fact, five different yeast starters were tested in multiple vinifications, where the Saccharomyces cerevisiae strain RC212 was able to raise conspicuously the concentrations of total pigment, anthocyanin, and tannins. Recently, Carrascosa and collaborators [81] have recently shown that different yeast strains were able to produce Albariño wines denoted with a specific polyphenol composition. The above findings were further confirmed by a recent report [82], where an unambiguous correlation between the yeast starter utilized to promote the fermentation process and chemical profile of wine was recognized, thus underlining the strain-specific yeast property to modify the color and the polyphenolic composition of the final product.
Moreover, a recent study has produced the identification of yeast starter cultures able to enhance the quality of the wine produced from the Italian red cultivar “Gaglioppo” a cultivar with reduced synthesis of anthocyanins [83]. The obtained evidences further evidenced the strain-specific capacity of wine yeast to modify the final amounts of total anthocyanins, total polyphenols, and total tannins.
Recently, Giovinazzo and coworkers (manuscript in preparation) highlighted a positive role of autochthonous yeast starter cultures for the enhancement of polyphenol content throughout the industrial production of Negroamaro wine. The statistical assessment of the experiment showed that the use of autochthonous strains increased the concentrations of several classes of polyphenols in the produced wines when compared to wines produced from the Sama grape must with commercial starter strain.
Taken together, the above-described scientific outcomes emphasize the relevance of the development and the industrial application of innovative biotechnological approaches in order to exalt the presence of healthy molecules in wine, thus improving “functional parameters” with the consequential enhancement of the final wine quality.
4 Wine Polyphenol Mechanism of Action Against Cardiovascular Diseases
Wine polyphenols have garnered much attention, especially with regard to their potential role in the protection against cardiovascular diseases. Indeed, red wine is thought to be responsible for the “French paradox” [84], a term used to describe the low incidence of cardiovascular disease in the French population despite their high intake of saturated fats.
Many preclinical and some clinical studies have identified a number of mechanisms and targets by which specific wine polyphenols could exert benefits against cardiovascular diseases. Wine polyphenols, including flavonols and resveratrol, have been shown to modulate the expression of inflammation-related genes involved in the atherosclerotic process as well as in chronic degenerative diseases.
Flavonols (quercetin, kaempferol, and myricetin) significantly downregulate the coordinated expression of the endothelial adhesion molecules, E-selectin, vascular cell adhesion molecule (VCAM)-1, and intercellular adhesion molecule (ICAM)-1, in human cultured endothelial cells activated by inflammatory triggers [85], thus decreasing the adhesion and subsequent trans-endothelial migration of monocytes into the intima of the vascular wall, i.e., processes that constitute the initial steps in the development of atherosclerosis. Flavonols have also been reported to significantly downregulate the expression of monocyte chemoattractant protein (MCP)-1 and macrophage colony-stimulating factor (M-CSF); both pro-inflammatory endothelial proteins guide monocytes into the subendothelial space, during inflammatory state [85]. The anti-inflammatory flavonol effects were mediated by the reduced activation of NF-κB and AP-1, whose binding sites are present in the promoter region of the pro-inflammatory genes including VCAM-1, ICAM-1, E-selectin, as well as MCP-1 and M-CSF [86]. Furthermore, flavonols also affected the inflammatory response by downregulating the expression of inflammation-related genes, like interleukin (IL)-6, IL-4, and tumor necrosis factor (TNF)-α. In human vascular endothelial cells, our research group reported that quercetin reduced inflammatory angiogenesis, a key pathogenic process contributing to atherosclerotic lesion formation, progression, and vulnerability, through inhibition of the pro-inflammatory enzyme cyclooxygenase (COX)-2 and gelatinases, the matrix metalloproteinase (MMP)-9 [87].
A limitation of the anti-inflammatory effect by flavonols in these in vitro studies is the use of flavonols at supraphysiological concentrations and as aglycone forms. It has been proven that after oral absorption, flavonols are rapidly converted to circulating conjugates through glucuronidation, sulfidation, or methylation. This accounts for the very low aglycone concentrations in human plasma (in the nanomolar range).
Few studies have investigated the effects of flavonol metabolites on vascular cell function. It has been shown that human quercetin plasma metabolites at physiologically significant concentrations were able to inhibit COX-2 expression in human lymphocytes. Furthermore, Tribolo et al. showed that quercetin and its phase II metabolites affected the expression of VCAM-1, ICAM-1, and MCP-1 in inflamed endothelial cells [88]. However, at 10 μM, quercetin metabolites showed a reduced ability to decrease the stimulated expression of these genes when compared with quercetin. This suggested that the chemical transformation of quercetin during phase II metabolism resulted in a reduction of bioactivity, at least with respect to regulation of inflammatory gene expression. However, at a vascular level, quercetin glucuronides can be freed of their sugar moiety by a deconjugation process performed by β-glucuronidases, and the free aglycone is delivered to tissues, particularly under inflammatory conditions [89]. In a vascular co-culture model represented by human arterial smooth muscle cells and endothelial cells, quercetin and its phase II metabolites at physiologically relevant concentration significantly decreased the stimulated expression of the vasoconstrictor endothelin-1 [90], involved in the endothelial regulation of vascular tone.
Many results obtained in cell culture studies have been replicated in animal model but not in human trials. However, several human studies have shown that quercetin can reduce blood pressure in hypertensive patients [91], although the precise mechanisms have not been elucidated.
In addition to flavonols, the anti-inflammatory action of wine polyphenols is exerted by resveratrol.
Resveratrol, like quercetin, has been reported to modulate the expression of inflammation-related genes involved in the cellular processes that control adhesion and migration of monocytes to vascular endothelium [92, 93]. We reported that resveratrol decreased monocytoid cell adhesion to human endothelial cells via reduction of VCAM-1 gene expression and by suppressing VCAM-1 promoter activity (see Figs. 2 and 3) [15]. In addition to VCAM-1, resveratrol also inhibited ICAM-1 and E-selectin, as well as MCP-1 and M-CSF [85, 93]. In human endothelial cells, monocytes, and smooth muscle cells, resveratrol strongly inhibited the expression of MMPs [85, 93, 94], responsible for the degradation of extracellular matrix, an essential event in atherosclerotic process, thus preventing plaque development, progression, and vulnerability. Furthermore, in endothelial and mononuclear cells, resveratrol inhibited, in a dose-dependent manner, the stimulated expression of tissue factor [96], a key regulator in the extrinsic pathway of blood coagulation. These anti-inflammatory and anti-thrombotic effects of resveratrol were at least in part mediated by lowered levels of intracellular ROS and the reduced activation of redox-sensitive transcription factors, NF-κB and AP-1 [85, 93, 96]. Part of the beneficial effects of resveratrol was also mediated by the upregulation of endothelial nitric oxide synthase (eNOS), involved in the regulation of vascular homeostasis. Most of the studies about the cardiovascular beneficial effect of resveratrol were performed by using its aglycone form; however it has been shown that piceid, a glycosidic form of resveratrol, also preserved the vascular anti-inflammatory properties, although at a lesser extent [85, 94].
Characterization of Primitivo wine polyphenol extract (PWPE) and Negramaro wine polyphenol extract (NWPE) polyphenol content and chemical structure of polyphenols. (a) Polyphenol content PWPE and NWPE (10 μg/mL). (b) Chemical structures of polyphenol groups identified in red wine extracts: hydroxycinnamic acids (CFF caffeic acid, CMR p-coumaric acid, CFT caftaric acid), flavonols (KMP kaempferol, QRC quercetin, MYR myricetin), and stilbenes (RSV trans-resveratrol, PCD trans-piceid)
Inhibitory effects of PWPE and NWPE on the monocyte adhesion to endothelial monolayer and on the expression of endothelial adhesion molecules. (a, b) HUVEC were pretreated with Primitivo wine polyphenol extract (PWPE) and Negramaro wine polyphenol extract (NWPE) (1–50 μg/mL) or vehicle (control) for 1 h and then stimulated with LPS 0.5 μg/mL for 16 h. HUVEC were co-cultured with calcein AM-labeled U937 monocytes for 1 h. The number of adherent U937 cells was monitored by fluorescence microscope (a) or measured by the fluorescence plate reader (b). (c–e) Cell surface expression of ICAM-1 (c), VCAM-1 (d), and E-Selectin (e) was analyzed by cell surface EIA. Each experiment was performed in triplicate. Data are expressed as the percentage of LPS-induced expression (mean ± SD). #p <0.01 versus control; *p <0.05; **p <0.01 versus lipopolysaccharide (LPS) alone
As a critical point of in vitro studies, the cardio-vasculo-protective effects of resveratrol have been shown to occur at supraphysiological (10 μM) concentrations, which cannot be achievable through dietary intake. However, some beneficial properties of resveratrol have been also observed at dietary doses. In human endothelial cells, resveratrol at physiological concentrations decreased the stimulated expression of VCAM-1, ICAM-1, and MCP-1 [54], as well as the cytokines, IL-6 and CCXL2. A significant increase in eNOS expression in HUVEC has been reported also at lower concentration of resveratrol (1 μM), following repeated stimulation for 6 days [97]. Physiological concentrations (0.1–1 μM) of resveratrol have also been reported to modulate the expression of genes involved in cell proliferation, blood pressure regulation, oxidative stress response, and autophagy in endothelial cells [98].
Another critical point for resveratrol efficacy is its low bioavailability. It is rapidly absorbed, metabolized, and excreted; however, in spite of its low bioavailability, evidence that beneficial activities occur in humans is beginning to emerge, and this phenomenon has been described as the “resveratrol paradox.” This paradox could thus be related to a possible action of resveratrol metabolites [99] and/or to a synergistic effect of resveratrol with other polyphenols or micronutrients. Accordingly, resveratrol as a blend of polyphenols from grape extracts exhibited a greater inhibitory effects on the expression of inflammatory markers in vascular cell than resveratrol alone [85, 95], suggesting the occurrence of a synergism among resveratrol and other polyphenols.
Though resveratrol’s potential utility in preventive medicine has been demonstrated using in vitro models, few clinical trials have also evaluated the effects of resveratrol on clinically relevant biomarkers. In healthy individuals, Agarwal and collaborators evaluated the effects of 1-month resveratrol treatment on endothelial response and plasma biomarkers [100]. Exposing cultured human coronary artery endothelial cells to plasma drawn post-resveratrol supplement resulted in significantly lower mRNA expression of VCAM-1, ICAM-1, and IL-8 than plasma drawn from the same subjects at baseline. This clinical trial highlighted for improved gene expression in vascular endothelium by resveratrol. A triple-blind, randomized, placebo-controlled, 1-year treatment with a resveratrol-containing grape supplement on stable patients with coronary artery disease [101] showed dose-dependently an increase of the anti-inflammatory serum adiponectin and a decrease of plasminogen activator inhibitor-1. Moreover, the transcriptional profiling showed a downregulation of pro-inflammatory genes and a modulation of inflammatory transcription factors, confirming previous in vitro findings [102].
Moreover, in peripheral blood mononuclear cells of type 2 diabetes and hypertensive patients with coronary artery disease [102], the supplementation with resveratrol-containing grape supplement significantly reduced the expression of the pro-inflammatory cytokines CCL3, IL-1, and TNF-α and modulated inflammatory-related microRNAs. These clinical studies support the conclusion of beneficial anti-inflammatory and immunomodulatory effects of grape extract enriched in resveratrol for secondary prevention of patients with coronary artery diseases.
5 Conclusion
The precise nature of the role played by polyphenols in human health has been largely highlighted in these last years. A better knowledge concerning the composition and dynamics of polyphenol profile in red grape will help vinedresser and winemakers in producing grape-derived products and wines with high content of phenolic antioxidants and considerable antioxidant activity, maintaining optimal organoleptic properties and a significant link with the original terroir.
Even though, a better understanding is still requested about the several different cellular mechanisms and complex pathways involved in wine polyphenol metabolism, the present findings suggest that the contribution of antioxidant phenols through a reasonable daily drinking of red wines may offer additional protection against in vivo oxidation and other damages of human cell components.
References
Yung JY, Saliba AJ, Prenzler PD (2010) Should red wine be considered a functional food? Compr Rev Food Sci Food Saf 9:530–551
Goldberg DM, Tsang E, Karumanchiri A, Diamandis EP, Soleas G, Ng E (1996) Method to assay the concentrations of phenolic constituents of biological interest in wines. Anal Chem 68:1688–1694
Giovinazzo G, Grieco F (2015) Functional properties of grape and wine polyphenols. Plant Foods Hum Nutr 70:454–462
Chong MF, Macdonald R, Lovegrove JA (2010) Fruit polyphenols and CVD risk: a review of human intervention studies. Br J Nutr 104(S3):S28–S39
Jandet P, Douillet-Breuil AC, Bessis R, Debord S, Sbaghi M, Adrian M (2002) Phytoalexins from the vitaceae, biosynthesis, phytoalexin gene expression in transgenic plants, antifungal activity, and metabolism. J Agric Food Chem 50:2731–2741
Pinasseau L, Vallverdú-Queralt A, Verbaere A, Roques M, Meudec E, Le Cunff L, Péros J-P, Ageorges A, Sommerer N, Boulet J-C, Terrier N, Cheynier V (2017) Cultivar diversity of grape skin polyphenol composition and changes in response to drought investigated by LC-MS based metabolomics. Front Plant Sci 8:1826
Paixao N, Pereira V, Marques JC, Camara JS (2008) Quantification of polyphenols with potential antioxidant properties in wines using reverse phase HPLC. J Sep Sci 31:2189–2198
Soleas GJ, Diamandis EP, Goldberg DM (2001) The world of resveratrol. In: American Institute for Cancer Research (eds) Nutrition and Cancer Prevention. Advances in Experimental Medicine and Biology, vol 492. Springer, Boston, MA, pp 159–182
Arnous A, Makris DP, Kefalas P (2001) Effect of principal polyphenolic components in relation to antioxidant characteristics of aged red wines. J Agric Food Chem 49:5736–5742
Mattivi F, Zulian C, Nicolini G, Valenti L (2002) Wine, biodiversity, technology, and antioxidants. Ann N Y Acad Sci 957:37–56
Saucier C, Little D, Glories Y (1997) First evidence of acetaldehyde-flavanol condensation products in red wine. Am J Enol Vitic 48:370–373
Sánchez-Iglesias M, González-Sanjosé ML, Pérez-Magariño S, Ortega-Heras M, González-Huerta C (2009) Effect of micro-oxygenation and wood type on the phenolic composition and colour of an aged red wine. J Agric Food Chem 57:11498–11509
Berta Baca-Bocanegra J, Nogales-Bueno J, Hernández-Hierro M, Heredia FJ (2018) Evaluation of extractable polyphenols released to wine from cooperage byproduct by near infrared hyperspectral imaging. Food Chem 20:206–212
Spranger MI, Climaco MC, Sun BS, Eiriz N, Fortunato C, Nunes A, Leandro MC, Avelar ML, Belchior AP (2004) Differentiation of red winemaking technologies by phenolic and volatile composition. Anal Chim Acta 513:151–161
Restuccia D, Sicari V, Pellicanò TM, Spizzirri UG, Loizzo MR (2017) The impact of cultivar on polyphenol and biogenic amine profiles in Calabrian red grapes during winemaking. Food Res Int 102:303–312
Cabrita MJ, Torres M, Palma V, Alves E, Patão R, Costa Feritas AM (2008) Impact of malolactic fermentation on low molecular weight phenolic compounds. Talanta 74:1281–1286
Gil-Muñoz R, Gómez-Plaza E, Martínez A, López-Roca JM (1999) Evolution of phenolic compounds during wine fermentation and post-fermentation: influence of grape temperature. J Food Compos Anal 12:259–272
Teixeira J, Gaspar A, Garrido EM, Garrido J, Borges F (2013) Hydroxycinnamic acid antioxidants: an electrochemical overview. Bio Med Res Int 2013:Article ID 251754. 11 pages
Sul D, Kim HS, Lee D, Joo SS, Hwang KW, Park SY (2009) Protective effect of caffeic acid against beta-amyloid-induced neurotoxicity by the inhibition of calcium influx and tau phosphorylation. Life Sci 84:257–262
Vauzour D, Houseman EJ, George TW, Corona G, Garnotel R, Jackson KG, Sellier C, Gillery P, Kennedy OB, Lovegrove JA, Spencer JP (2010) Moderate champagne consumption promotes an acute improvement in acute endothelial-independent vascular function in healthy human volunteers. Br J Nutr 103:1168–1178
Jung EH, Ran Kim S, Hwang IK, Youl HT (2007) Hypoglycemic effects of a phenolic acid fraction of rice bran and ferulic acid in C57BL/KsJ-db/db mice. J Agric Food Chem 55:9800–9804
Vina J, Gomez-Cabrera MC, Borras C (2007) Fostering antioxidant defences, up-regulation of antioxidant genes or antioxidant supplementation. Br J Nutr 98:S36–S40
Mink PJ, Scrafford CG, Barraj LM (2007) Flavonoid intake and cardiovascular disease mortality, a prospective study in post-menopausal women. Am J Clin Nutr 85:895–909
Seeram NP, Adams LS, Hardy ML, Heber D (2004) Total cranberry extract versus its phytochemical constituents, anti-proliferative and synergistic effects against human tumour cell lines. J Agric Food Chem 52:2512–2517
Soobratte MA, Bahorun T, Aruoma OI (2006) Chemopreventive actions of polyphenolic compounds in cancer. Biofactors 27:19–35
Fink BN, Steck SE, Wolff MS, Britton JA, Kabat GC, Schroeder JC, Teitelbaum SL, Neugut AI, Gammon MD (2007) Dietary flavonoid intake and breast cancer risk among women on Long Island. Am J Epidemiol 165:514–523
Zhu L, Zhang Y, Lu J (2012) Phenolic contents and compositions in skins of red wine grape cultivars among various genetic backgrounds and originations. Int J Mol Sci 13:3492–3510
Dvorakova M, Landa P (2017) Anti-inflammatory activity of natural stilbenoids: a review. Pharm Res 124:126–145
Piotrowska H, Kucinska M, Murias M (2012) Biological activity of piceatannol: leaving the shadow of resveratrol. Mutat Res 750:60–82
Neves AR, Lúcio M, Lima JLC, Reis S (2012) Resveratrol in medicinal chemistry: a critical review of its pharmacokinetics drug-delivery, and membrane interactions. Curr Med Chem 19:1663–1681
Poulose SM, Thangthaeng N, Miller MG, Shukitt-Hale B (2015) Effects of pterostilbene and resveratrol on brain and behaviour. Neurochem Int 89:227–233
Rivière C, Pawlus AD, Mérillon JM (2012) Natural stilbenoids: distribution in the plant kingdom and chemotaxonomic interest in Vitaceae. Nat Prod Rep 29:1317–1333
Baur JA, Sinclair DA (2006) Therapeutic potential of resveratrol: the in vivo evidence. Nat Rev Drug Discov 5:493–506
Zamora-Ros R, Andres-Lacueva C, Lamuela-Raventos RM, Berenguer T, Jakszyn P, Martinez C, Sanchez M, Navarro C, Chirlaque M, Tormo M-J, Quiros J, Amiano P, Dorronsoro M, Larranaga N, Barricarte A, Ardanaz E, Gonzalez C (2008) Concentrations of resveratrol and derivatives in foods and estimation of dietary intake in a Spanish population, European Prospective Investigation into Cancer and Nutrition (EPIC)-Spain cohort. Br J Nutr 100:188–196
Gerard E, Mullin MD (2011) Red wine, grapes, and better health-resveratrol. Nutr Clin Pract 26:722–723
D’Introno A, Paradiso A, Scoditti E, D’Amico L, De Paolis A, Carluccio MA, Nicoletti I, DeGara L, Santino A, Giovinazzo G (2009) Anti-oxidant and anti-inflammatory properties of tomato fruit synthesising different amount of stilbenes. Plant Biotechnol J 7:422–429
Frei B (2004) Efficacy of dietary antioxidants to prevent oxidative damage and inhibit chronic disease. J Nutr 134:3196–3198
Kundu JK, Surth YJ (2008) Cancer chemopreventive and therapeutic potential of resveratrol, mechanistic perspectives. Cancer Lett 269:243–261
Nguyen A, Martinez M, Stamos MJ, Moyer MP, Planutis K, Hope C, Holcombe RF (2009) Results of a phase I pilot clinical trial examining the effect of plant-derived resveratrol and grape powder on Wnt pathway target gene expression in colonic mucosa and colon cancer. Cancer Manag Res 1:25–37
Fonar Y, Frank D (2011) FAK and WNT signaling: the meeting of two pathways in cancer and development. Anti Cancer Agents Med Chem 11:600–606
Kaminski BM, Steinhilber D, Stein JM, Ulrich S (2011) Phytochemicals resveratrol and sulforaphane as potential agents for enhancing the anti-tumor activities of conventional cancer therapies. Curr Pharm Biotechnol 67:1167–1178
Vergara D, Simeone P, Toraldo D, Del Boccio P, Vergaro V, Leporatti S, Pieragostino D, Tinelli A, De Domenico S, Alberti S, Urbani A, Salzet M, Santino A, Maffia M (2012) Resveratrol down regulates Akt/GSK and ERK signalling pathways in OVCAR-3 ovarian cancer cells. Mol Biosyst 8:1078–1087
Udenigwe CC, Ramprasath VR, Aluko RE, Jones PJ (2008) Potential of resveratrol in anticancer and anti-inflammatory therapy. Nutr Rev 66:445–454
Bruno R, Ghisolfi L, Priulla M, Nicolin A, Bertelli A (2003) Wine and tumours: study of resveratrol. Drugs Exp Clin Res 29:257–261
Jazirehi AR, Bonavida B (2004) Resveratrol modifies the expression of apoptotic regulatory proteins and sensitizes non-Hodgkin’s lymphoma and multiple myeloma cell lines to paclitaxel-induced apoptosis. Mol Cancer Ther 3:71–84
Dulak J (2005) Nutraceuticals as anti-angiogenic agents: hopes and reality. J Physiol Pharmacol 56:51–67
Jones SB, DePrimo SE, Whitfield ML, Brooks JD (2005) Resveratrol induced gene expression profiles in human prostate cancer cells. Cancer Epidemiol Biomark Prev 14:596–604
Sharma S, Chopra K, Kulkarni SK (2007) Effect of insulin and its combination with resveratrol or curcumin in attenuation of diabetic neuropathic pain, participation of nitric oxide F TNF-alpha. Phytother Res 21:278–283
Anekonda TS (2006) Resveratrol a boon for treating Alzheimer’s disease? Brain Res Rev 52:316–326
Wodd JG, Rogina B, Lavu S, Howitz K, Hefland SL, Tatar M, Sinclair D (2004) Sirtuin activators mimic caloric restriction and delay ageing in metazoans. Nature 430:686–689
Park SJ, Ahmad F, Philp A, Baar K, Williams T, Luo H, Ke H, Rehmann H, Taussig R, Brown AL, Kim MK, Beaven MA, Burgin AB, Manganiello V, Chung JH (2012) Resveratrol ameliorates aging-related metabolic phenotypes by inhibiting cAMP phosphodiesterases. Cell 14:421–433
Pearson KJ1, Baur JA, Lewis KN, Peshkin L, Price NL, Labinskyy N, Swindell WR, Kamara D, Minor RK, Perez E, Jamieson HA, Zhang Y, Dunn SR, Sharma K, Pleshko N, Woollett LA, Csiszar A, Ikeno Y, Le Couteur D, Elliott PJ, Becker KG, Navas P, Ingram DK, Wolf NS, Ungvari Z, Sinclair DA, de Cabo R (2008) Resveratrol delays age-related deterioration and mimics transcriptional aspects of dietary restriction without extending life span. Cell Metab 8:157–168
Carluccio MA, Ancora MA, Massaro M, Carluccio M, Scoditti E, Distante A, Storelli C, De Caterina R (2007) Homocysteine induces VCAM-1 gene expression through NF-kappa B and NAD(P)H oxidase activation, protective role of Mediterranean diet polyphenolic antioxidants. Am J Physiol Heart Circ Physiol 293:2344–2354
Csiszar A, Smith K, Labinskyy N, Orosz Z, Rivera A, Ungvari Z (2006) Resveratrol attenuates TNF-α-induced activation of coronary arterial endothelial cells: role of NF-κB inhibition. Am J Physiol-Heart Circ Physiol 291:H1694–H1699
Pietrocola F, Mariño G, Lissa D, Vacchell IE, Malik SA, Niso-Santano M, Zamzami N, Galluzzi L, Maiuri MC, Kroemer G (2012) Pro-autophagic polyphenols reduce the acetylation of cytoplasmic proteins. Cell Cycle 11:3851–3860
Amri A, Chaumeil JC, Sfar S, Charrueau C (2012) Administration of resveratrol, what formulation solutions to bioavailability limitations ? J Control Rel 158:182–193
González-Neves G, Gil G, Favre G, Baldi C, Hernández N, Traverso S (2013) Influence of winemaking procedure and grape variety on the color and composition of young red wines. S Afr J Enol Vitic 34:138–146
Gambacorta G, Antonacci D, Pati S, la Gatta M, Faccia M, Coletta A, La Notte E (2011) Influence of winemaking technologies on phenolic composition of Italian red wines. Eur Food Res Technol 233:1057–1066
El Darra N, Turk MF, Ducasse MA, Grimi N, Maroun RG, Louka N, Vorobiev E (2016) Changes in polyphenol profiles and color composition of freshly fermented model wine due to pulsed electric field, enzymes and thermos vinification pretreatments. Food Chem 194:944–950
Atanacković M, Petrović A, Jović S, Gojković-Bukarica L, Bursać M, Cvejić J (2012) Influence of winemaking techniques on the resveratrol content, total phenolic content and antioxidant potential of red wines. Food Chem 131:513–518
Gambuti A, Strollo D, Erbaggio A, Lecce L, Moio L (2007) Effect of winemaking practices on color indexes and selected bioactive phenolics of Aglianico wine. J Food Sci 72:S623–S628
Kostadinović S, Wilkens A, Stefova M, Ivanova V, Vojnoski B, Mirhosseini H, Winterhalter P (2012) Stilbene levels and antioxidant activity of Vranec and Merlot wines from Macedonia: effect of variety and enological practices. Food Chem 135:3003–3009
Ivanova V, Vojnoski B, Stefova M (2012) Effect of winemaking treatment and wine aging on phenolic content in Vranec wines. J Food Sci Technol 492:161–172
Coletta A, Trani A, Faccia M, Punzi R, Dipalmo T, Crupi P, Antonacci D, Gambacorta G (2013) Influence of viticultural practices and winemaking technologies on phenolic composition and sensory characteristics of Negroamaro red wines. Int J Food Sci Technol 4811: 2215–2227
Ferraretto P, Celotti E (2016) Preliminary study of the effects of ultrasound on red wine polyphenols. CyTA-J Food 14:529–535
Baiano A, Terracone C, Gambacorta G, La Notte E (2009) Phenolic content and antioxidant activity of Primitivo wine: comparison among winemaking technologies. J Food Sci 74: 258–267
Francesca N, Romano R, Sannino C, Le Grottaglie L, Settanni L, Moschetti G (2014) Evolution of microbiological and chemical parameters during red wine making with extended post-fermentation maceration. Int J Food Microbiol 17:84–93
Mulero J, Zafrilla P, Cayuela JM, Martínez-Cachá A, Pardo F (2011) Antioxidant activity and phenolic compounds in organic red wine using different winemaking techniques. J Food Sci 76:C436–C440
Cholet CL, Delsart C, Petrel M, Gontier E, Grimi N, L’Hyvernay A, Ghidossi R, Vorobiev E, Mietton-Peuchot M, Geny L (2014) Structural and biochemical changes induced by pulsed electric field treatments on Cabernet Sauvignon grape berry skins: impact on cell wall total tannins and polysaccharides. J Agric Food Chem 62:2925–2934
Delsart C, Ghidossi R, Poupot C, Cholet C, Grimi N, Vorobiev E, Milisic V, Peuchot MM (2012) Enhanced extraction of phenolic compounds from merlot grapes by pulsed electric field treatment. Am J Enol Vitic 63:205–211
Puértolas E, Saldaña G, Alvarez I, Raso J (2010) Effect of pulsed electric field processing of red grapes on wine chromatic and phenolic characteristics during aging in oak barrels. J Agric Food Chem 58:2351–2357
Luengo E, Alvarez I, Raso J (2015) Phenolic extraction: pulsed electric fields: a technology for improving phenolic extraction in red wines. Wine Vitic J 301:17–21
Bai B, He F, Yang L, Chen F, Reeves MJ, Li J (2013) Comparative study of phenolic compounds in Cabernet Sauvignon wines made in traditional and Ganimede fermenters. Food Chem 1414:3984–3992
Morata A, Gomez-Cordoves C, Subervolia J, Bartolome B, Colomo B, Suarez JA (2003) Adsorption of anthocyanins by yeast cell walls during fermentation of red wines. J Agric Food Chem 51:4084–4088
Rodriguez-Nogales JM, Fernández-Fernández E, Gómez M, Vila-Crespo J (2012) Antioxidant properties of sparkling wines produced with β-glucanases and commercial yeast preparations. J Food Sci 77:1005–1010
Mazauric JP, Salmon JM (2005) Interactions between yeast lees and wine polyphenols during simulation of wine aging: I analysis of remnant polyphenolic compounds in the resulting wines. J Agric Food Chem 53:5647–5653
Girard B, Yuksel D, Cliff MA, Delaquis P, Reynolds AG (2001) Vinification effects on the sensory, colour, and GC profiles of Pinot noir wines from British Colombia. Food Res Int 34:483–499
Mazza G, Fukumoto L, Delaquis P, Girard B, Ewert B (1999) Anthocyanins, phenolics, and color of Cabernet franc, Merlot, and Pinot noir wines from British Colombia. J Agric Food Chem 47:4009–4017
Brandolini V, Fiore C, Maietti A, Tedeschi P, Romano P (2007) Influence of Saccharomyces cerevisiae strains on wine total antioxidant capacity evaluated by photo-chemiluminescence. World J Microbiol Biotechnol 23:581–586
Carew AL, Smith P, Close DC, Curtin C, Dambergs RG (2013) Yeast effects on Pinot noir wine phenolics, color, and tannin composition. J Agric Food Chem 6141:9892–9898
Carrascosa AV, Bartolome B, Robredo S, Leon A, Cebollero E, Juega M, Nunez YP, Martinez MC, Martinez-Rodriguez AJ (2012) Influence of locally-selected yeast on the chemical and sensorial properties of Albariño white wines. LWT-Food Sci Technol 46: 319–325
Tufariello M, Chiriatti MA, Grieco F, Perrotta C, Capone S, Rampino P, Tristezza M, Mita G, Grieco F (2014) Influence of autochthonous Saccharomyces cerevisiae strains on volatile profile of Negroamaro wines. LWT Food Sci Technol 58:35–48
Caridi A, De Bruno A, De Salvo E, Piscopo A, Poiana M, Sidari R (2017) Selected yeasts to enhance phenolic content and quality in red wine from low-pigmented grapes. Eur Food Res Technol 2433:367–378
Renaud S, de Lorgeril M (1992) Wine, alcohol, platelets, and the French paradox for coronary heart disease. Lancet 339:1523–1526
Calabriso N, Scoditti E, Massaro M, Pellegrino M, Storelli C, Ingrosso I, Giovinazzo G, Carluccio MA (2016) Multiple anti-inflammatory and anti-atherosclerotic properties of red wine polyphenolic extracts: differential role of hydroxycinnamic acids, flavonols and stilbenes on endothelial inflammatory gene expression. Eur J Nutr 55:477–489
Collins T, Read MA, Neish AS, Whitley MZ, Thanos D, Maniatis T (1995) Transcriptional regulation of endothelial cell adhesion molecule: NF-kB and cytokine-inducible enhancer. FASEB J 9:899–909
Scoditti E, Calabriso N, Massaro M, Pellegrino M, Storelli C, Martines G, De Caterina R, Carluccio MA (2012) Mediterranean diet polyphenols reduce inflammatory angiogenesis through MMP-9 and COX-2 inhibition in human vascular endothelial cells: a potentially protective mechanism in atherosclerotic vascular disease and cancer. Arch Biochem Biophys 527:81–89
Tribolo S, Lodi F, Connor C, Suri S, Wilson VG, Taylor MA, Needs PW, Kroon PA, Hughes DA (2008) Comparative effects of quercetin and its predominant human metabolites on adhesion molecule expression in activated human vascular endothelial cells. Atherosclerosis 197:50–56
Shimoi K, Saka N, Nozawa R, Sato M, Amano I, Nakayama T, Kinae N (2001) Deglucuronidation of a flavonoid, luteolin monoglucuronide, during inflammation. Drug Metab Dispos 29:1521–1524
Lodi F, Winterbone MS, Tribolo S, Needs PW, Hughes DA, Kroon PA (2012) Human quercetin conjugated metabolites attenuate TNF-alpha-induced changes in Vasomodulatory molecules in a HUASMCs/HUVECs co-culture model. Planta Med 78:1571–1573
Boomgaarden I, Egert S, Rimbach G, Wolffram S, Muller MJ, Doring F (2010) Quercetin supplementation and its effect on human monocyte gene expression profiles in vivo. Br J Nutr 104:336–345
Tome-Carneiro J, Larrosa M, Gonzalez-Sarrias A, Tomas-Barberan FA, Garcia-Conesa MT, Espin JC (2013) Resveratrol and clinical trials: the crossroad from in vitro studies to human evidence. Curr Pharm Des 19:6064–6093
Carluccio MA, Siculella L, Ancora MA, Massaro M, Scoditti E, Storelli C, Visioli F, Distante A, De Caterina R (2003) Olive oil and red wine antioxidant polyphenols inhibit endothelial activation: antiatherogenic properties of Mediterranean diet phytochemicals. Arterioscler Thromb Vasc Biol 23:622–629
Lee B, Moon SK (2005) Resveratrol inhibits TNF-alpha-induced proliferation and matrix metalloproteinase expression in human vascular smooth muscle cells. J Nutr 135:2767–2773
Calabriso N, Massaro M, Scoditti E, Pellegrino M, Ingrosso I, Giovinazzo G, Carluccio MA (2016) Red grape skin polyphenols blunt matrix metalloproteinase-2 and-9 activity and expression in cell models of vascular inflammation: protective role in degenerative and inflammatory diseases. Molecules 21(9):1147
Pendurthi UR, Williams JT, Rao LVM (1999) Resveratrol, a polyphenolic compound found in wine, inhibits tissue factor expression in vascular cells – a possible mechanism for the cardiovascular benefits associated with moderate consumption of wine. Arterioscler Thromb Vasc Biol 19:419–426
Takizawa Y, Kosuge Y, Awaji H, Tamura E, Takai A, Yanai T, Yamamoto R, Kokame K, Miyata T, Nakata R, Inoue H (2013) Up-regulation of endothelial nitric oxide synthase (eNOS), silent mating type information regulation 2 homologue 1 (SIRT1) and autophagy-related genes by repeated treatments with resveratrol in human umbilical vein endothelial cells. Br J Nutr 110:2150–2155
Ungvari Z, Bagi Z, Feher A, Recchia FA, Sonntag WE, Pearson K, de Cabo R, Csiszar A (2010) Resveratrol confers endothelial protection via activation of the antioxidant transcription factor Nrf2. Am J Phys Heart Circ Phys 299:H18–H24
Bresciani L, Calani L, Bocchi L, Delucchi F, Savi M, Ray S, Brighenti F, Stilli D, Del Rio D (2014) Bioaccumulation of resveratrol metabolites in myocardial tissue is dose-time dependent and related to cardiac hemodynamics in diabetic rats. Nutr Metab Cardiovasc Dis 24:408–415
Agarwal B, Campen MJ, Channell MM, Wherry SJ, Varamini B, Davis JG, Baur JA, Smoliga JM (2013) Resveratrol for primary prevention of atherosclerosis: clinical trial evidence for improved gene expression in vascular endothelium. Int J Cardiol 166:246–248
Tome-Carneiro J, Gonzálvez M, Larrosa M, Yáñez-Gascón MJ, García-Almagro FJ, Ruiz-Ros JA, Tomás-Barberán FA, García-Conesa MT, Espín JC et al (2013) Grape resveratrol increases serum adiponectin and downregulates inflammatory genes in peripheral blood mononuclear cells: a triple-blind, placebo-controlled, one-year clinical trial in patients with stable coronary artery disease. Cardiovasc Drug Ther 27:37–48
Tome-Carneiro J, Larrosa M, Yáñez-Gascón MJ, Dávalos A, Gil-Zamorano J, Gonzálvez M, García-Almagro FJ, Ruiz Ros JA, Tomás-Barberán FA, Espín JC, García-Conesa MT (2013) One-year supplementation with a grape extract containing resveratrol modulates inflammatory-related microRNAs and cytokines expression in peripheral blood mononuclear cells of type 2 diabetes and hypertensive patients with coronary artery disease. Pharmacol Res 72:69–82
Acknowledgments
This research was partially supported by the Apulia Region in the framework of the Projects NEWINE (Bando “Ricerca e sperimentazione in Agricoltura”; Project code PRS_042), SOLBIOGRAPE (Bando “Ricerca e sperimentazione in Agricoltura”; Project code PRS_053), and DOMINA APULIAE (POR Puglia FESR – FSE 2014-2020-Azione 1.6. – InnoNetwork; Project code AGBGUK2).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Switzerland AG
About this entry
Cite this entry
Giovinazzo, G., Carluccio, M.A., Grieco, F. (2019). Wine Polyphenols and Health. In: Mérillon, JM., Ramawat, K.G. (eds) Bioactive Molecules in Food. Reference Series in Phytochemistry. Springer, Cham. https://doi.org/10.1007/978-3-319-78030-6_81
Download citation
DOI: https://doi.org/10.1007/978-3-319-78030-6_81
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-78029-0
Online ISBN: 978-3-319-78030-6
eBook Packages: Chemistry and Materials ScienceReference Module Physical and Materials ScienceReference Module Chemistry, Materials and Physics