Abstract
Over the past two decades, multidetector computed tomography (CT) has become a powerful diagnostic tool in emergency medicine relied upon for a variety of conditions. More recently, the development of dual-energy CT technology has enhanced the ability of radiologists to diagnose and distinguish between a variety of conditions, improving accuracy and patient care. The ability to separate material density pairs allows for highly specific observations, and helps to avoid diagnostic pitfalls. Dual-energy CT has benefits in diagnosing conditions of the liver, gallbladder, kidneys, adrenals, pancreas, large and small bowel, as well as vascular structures. It also has benefits in diagnostic evaluation of patients in the setting of trauma. When its benefits are harnessed, dual-energy CT has the potential to significantly improve patient care in the emergency department.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
3.1 Introduction
Use of computed tomography in the evaluation of patients in the emergency department (ED) has grown markedly in recent years [1]. The percentage of patients being seen in the ED who undergo CT as part of their ED visit increased from 2.8% in 1995 to 13.9% by 2007 [2]. The increase in imaging volume has been possible because of several technological advances, namely the widespread use of multi-detector CT (MDCT) scanners, and computer hardware and software which enables rapid multi-planar reformatting. The rapid acquisition and processing have led to the use of CT as a primary modality for the evaluation of a number of acute conditions, both traumatic and nontraumatic. In body imaging, CT is used extensively to evaluate a wide variety of conditions affecting the vasculature, genitourinary, biliary, digestive, and musculoskeletal systems.
Dual-energy CT (DECT) has emerged as a very useful tool in emergency radiology, and has many potential applications in body imaging. Conventional single-energy CT creates images based on the X-ray attenuation properties of anatomic structures from a single energy source. DECT is able to provide the same information as single-energy CT, but has the added benefit of being able to separate materials based on their material density, as different materials demonstrate different attenuation properties when there is a change in the kilovoltage peak (kVp).
In this chapter, the technical aspects of DECT are discussed, along with a variety of conditions for which DECT may be useful to the emergency radiologist. The potential benefits of DECT in the ED are very substantial, both for assisting in the diagnosis of acute conditions, and for problem solving when encountering incidental findings which require characterization (Fig. 3.1).
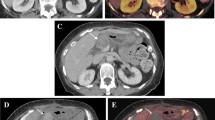
64-year-old woman with a history of cholangiocarcinoma with liver metastases presents with progressive abdominal pain. (a) Axial IV contrast-enhanced CT image performed with dual-energy technique demonstrates heterogeneous hypoattenuation in the inferior vena cava (arrow), which may be interpreted as contrast mixing, bland thrombus, or tumor thrombus. (b) On the corresponding axial iodine image, the finding is more apparent. The iodine image offers additional information as the absence of iodine within the thrombus (arrow) is more consistent with bland thrombus, rather than tumor thrombus
3.2 Technique
Conventional single-energy computed tomography utilizes a single polychromatic X-ray beam, with peak energy levels ranging from 80 to 140 kVp, emitted from a single X-ray tube to a single detector. In contrast, dual-energy CT (DECT) acquires images utilizing two energy spectra. Typically, low-energy images are acquired using 80 kVp, while high-energy images acquired using 140 kVp [3,4,5,6]. Some scanners (e.g., Somatom Force, Siemens Healthcare) allow variations (e.g., low energy 100 kVp, and high energy 150 kVp).
There are three DECT platforms which are currently commercially available: dual-source DECT, single-source (rapid kV switching) DECT, and detector-based spectral CT. Dual-source DECT (Siemens Healthcare) utilizes two perpendicular X-ray tubes and two corresponding sets of detectors for image acquisition. Rapid kV switching DECT, available from several CT vendors, utilizes a single X-ray tube which rapidly alternates (0.5 msc) between low and high energies, along with a single detector. The third mechanism (referred to as “spectral” CT; Philips Healthcare) involves utilization of a single X-ray tube, with a detector with a top layer which absorbs low energy, and a deep layer which absorbs high-energy X-rays [4, 7].
Image contrast is dependent on attenuation of X-ray photons by varying materials, which is in turn influenced by the photon energy level in relation to the k-edge of the material. The k-edge refers to the K-shell electron-binding energy. As photon energy level approaches the k-edge of a given material, there is a sharp increase in the attenuation coefficient [8, 9]. By obtaining information of a tissue at two different energies, dual-energy CT technology can generate image datasets with attenuation information at several virtual monochromatic (VMC) X-ray energy levels, ranging from 40 to 140 kiloelectron volts (keV). Selection of VMC energy level can be used to optimize image contrast. For example, 40–70 keV VMC images can be reconstructed for the purpose of accentuating iodine enhancement [7, 10]. Significant gain in iodine attenuation on 40–60 keV images has been used to reduce iodine dose on DECT exams. Previous investigations have reported 50–70% reduction in iodine doses for CT angiography by using 40–50 kev VMC images from DECT [10,11,12,13]. Despite a current controversy over post-contrast acute kidney injury (AKI), there is still concern about its existence. Therefore, using minimal iodine dose is desirable, especially in the emergency setting, particularly when a patient’s renal function may be unknown. This is particularly relevant in the setting of vascular injury, where a subsequent angiogram may be necessary. Contrast-media dose reduction must be accompanied with adjustments in injection rate in order to yield homogeneous vascular enhancement. Total iodine dose reduction can be achieved with both high- and low-concentration contrast media (CM). Low-concentration CM enable higher volumes on low-contrast computed tomography angiography (CTA) examinations. However, high-concentration CM needs to be mixed with saline to preserve volume [11].
On the other end of the spectrum, 80–140 keV images can also be generated to help reduce beam hardening and photon starvation artifacts related to metallic prostheses or foreign bodies [14, 15]. These artifacts are typically seen on conventional CT as a result of attenuation of low-energy photons in a polychromatic X-ray. Similarly, these images can be used to decrease the pseudo-enhancement which is relatively commonly seen in smaller cysts and soft-tissue nodules [4, 16].
Besides virtual monochromatic images, knowledge of the attenuation characteristics of a tissue at different X-ray energies also allows the creation of material-specific images. Given that different materials have different absorb X-ray photons in a unique way, several material density (MD) images can be created to analyze the composition of various tissues. Currently, the most commonly used MD images are MD-I images (iodine maps) and water-density (virtual non-enhanced) images. MD-I images exclusively show iodine contribution within the image, and do not take into account the inherent attenuation of the tissue. Therefore, iodine maps are helpful for detection of iodine content or enhancement within a nodule or mass [4, 5]. In the emergency setting, this is useful for the appropriate “triaging” of incidental nodules or masses. This will be discussed in further detail.
Virtual non-enhanced (VNE) images can be helpful for detecting the presence of calcium in a nodule or mass. A common misconception is that DECT imparts higher radiation doses than conventional CT. While this may have been true early in the development of DECT, this is not the case with current DECT technology [12]. On the contrary, current DECT technology may allow for radiation dose savings as VNE images can be constructed, which may in turn forgo the need for a conventional non-enhanced acquisition [16] (Table 3.1).
3.3 Gallbladder
3.3.1 Gallstone Disease
Gallstone disease is a common cause for right upper quadrant pain leading to an emergency department visit. Of patients that have gallstone disease, 10–15% will pass calculi into the central bile ducts leading to biliary obstruction, and frequently to subsequent acute cholecystitis or cholangitis [17, 18].
While CT is helpful for detection of hyperattenuating and hypoattenuating calculi, many calculi are essentially isoattenuating to bile within the gallbladder, and are therefore difficult to identify on conventional CT. This can be particularly true when calculi isoattenuating to bile are lodged in the common duct. Dual-energy CT can be helpful in these patients, for calculus detection and characterization (Fig. 3.2). With regard to detection, studies have shown that acquiring imaging with a lower energy (i.e., 40 keV) increases sensitivity for detection of gallstones [18, 19] (Fig. 3.2).
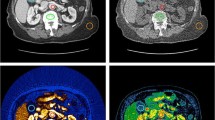
68-year-old man with noncalcified gallstones which are isodense to bile on axial 70 keV (a) and mixed images (b) on dual-energy CT. The presence of calculi is confirmed on an axial T2-weighted single-shot fast spin-echo MR image (c). The calculi decrease in attenuation using virtual monochromatic images at 40 keV (d), and increase in attenuation at 190 keV (e), with corresponding increase in conspicuity relative to surrounding bile. The dependent gallstones have the lowest HU values at 40 keV (d), are isoattenuating at 70 keV (a), and are hyperattenuating at 190 keV (e)
In addition to detection of cholelithiasis, a possible application of DECT is to determine the gallstone composition, as calculi of higher cholesterol content have been shown to have relatively lower attenuation [20, 21]. Given that these calculi may be treated conservatively, prospective characterization may lead clinicians to a more conservative approach [18].
3.3.2 Acute Cholecystitis
Acute cholecystitis is readily diagnosed using conventional CT, with reported sensitivity and specificity greater than 90% [22]. Gangrenous cholecystitis, however, presents a diagnostic challenge. The sensitivity for diagnosing gangrenous cholecystitis on CT is significantly lower at 29% [22]. The implications for missing this diagnosis are potentially serious, as it carries a higher morbidity and mortality. On conventional CT, the main findings suggestive of gangrenous cholecystitis include gas in the wall, intraluminal membranes, and discontinuous or irregular wall and mural striation [22]. While intramural gas in the gallbladder wall is easily detected on conventional CT, the other findings are often more difficult to detect [22]. Dual-energy CT can be helpful in these patients, particularly with iodine maps as areas of enhancement and relative hypoenhancement are easily detected. This may help accentuate areas of wall discontinuity. Intraluminal membranes may also be accentuated by evaluating intraluminal contents with low-keV images, which increases the contrast between the sloughed membranes and the surrounding infected biliary fluid. It may also be easier to see intramural hematoma in the gallbladder wall using VNE images with DECT.
3.4 Pancreas
3.4.1 Acute Pancreatitis
Acute pancreatitis is one of the most commonly encountered conditions in the emergency setting. Most commonly, the etiology is alcohol induced or gallstone related. In the latter scenario, DECT may be helpful in identifying choledocholithiasis. Because low-keV images can be used to increase conspicuity of certain types of calculi (i.e., cholesterol stones), DECT may increase sensitivity for gallstone detection [23], enabling a faster diagnosis of gallstone pancreatitis without having to then perform MR cholangiopancreatography.
Dual-energy CT may also be helpful in the setting of acute pancreatitis, because it can be used to identify complications, including pancreatic necrosis and hemorrhage. Pancreatic necrosis can be difficult to detect on conventional CT, as areas of hypoenhancement may be subtle. When identified, it is often unclear whether these areas represent areas of necrosis or ischemia. Using DECT, iodine maps can be constructed to help detect areas of subtle enhancement, which suggest ischemia rather than necrosis [4, 14]. Necrotic regions appear as areas without any iodine uptake (Fig. 3.3). Patients with necrotizing pancreatitis are generally managed more aggressively, so the distinction is pertinent [4].
54-year-old man with acute epigastric pain. (a) Axial IV contrast-enhanced CT image through the pancreas demonstrating acute pancreatitis with patchy areas of normal enhancement (solid arrow) and hypoenhancing parenchyma (open arrow). On these images, it is unclear whether the hypoenhancement represents ischemia or necrosis. (b) On the corresponding iodine image, there is an area of normal iodine uptake (solid arrow). The hypoenhancing area on CT (open arrow) does demonstrate iodine uptake, suggesting ischemia rather than necrosis. (c) In the same patient, the IV contrast-enhanced image demonstrates a separate area of normal enhancement (solid arrow) and hypoenhancement (open arrow). (d) On the corresponding iodine image, there is an area of normal iodine uptake (solid arrow). The hypoenhancing area on CT (open arrow) demonstrates no iodine uptake, suggesting necrosis rather than ischemia
Hemorrhagic pancreatitis can also be accurately diagnosed with the aid of DECT. On conventional contrast-enhanced CT, hyperattenuating regions may represent hemorrhage or enhancing pancreatic parenchyma. In such patients, review of non-enhanced images would be required to distinguish the two. However, this would require an additional acquisition, which is not routinely performed and would increase radiation dose. Review of VNE images on DECT scans can help to characterize these regions, as hyperattenuating foci on these images would stand out relative to non-enhancing parenchyma, which is consistent with hemorrhage rather than enhancement [4].
An additional benefit of DECT is the reduced radiation dose associated with the examination. Younger patients with acute pancreatitis and its complications often require repeated follow-up imaging examinations [7]. As previously suggested, these patients may undergo DECT, which would obviate the need for a non-contrast scan, as VNE images can be constructed.
3.5 Renal
3.5.1 Nephrolithiasis
Nephrolithiasis is a common condition in the United States, affecting approximately 1 out of 11 adults [1]. There is a slightly higher risk in men than in women, and it is known that dietary factors play a role, namely dehydration and dietary salt intake [24]. When renal calculi become obstructive in the renal pelvis, ureter, or ureterovesical junction, the experience is notoriously painful, and often brings patients to the ED. Additionally, approximately half of patients presenting with symptoms related to kidney calculi will have another episode in the next decade [24].
Over two decades, CT has been recognized as the modality of choice when evaluating patients with clinically suspected nephrolithiasis, or obstructing calculi in the urinary tract [25]. In direct comparison, CT has been shown to be more effective at revealing urolithiasis than ultrasound [26, 27]. Although non-enhanced CT is highly effective in the diagnosis of acute conditions related to urinary tract calculi, there are two areas in which DECT may have additional benefits.
First, many CT examinations of the abdomen and pelvis are acquired with intravenous contrast in the ED. Depending on the protocol for a given scan, and whether or not the patient recently received intravenous contrast for another examination, material density separation may be used to isolate radiodense urinary calculi from iodinated contrast in the collecting system or ureters. A high degree of accuracy is achieved by using material density separation to create VNE images to detect renal or ureteral calculi [28]. Thus, even in patients whose symptoms may not be typical for obstructing calculi and therefore undergo an IV contrast-enhanced examination, the correct diagnosis can still be made using DECT.
Second, dual-energy CT has reliably shown the ability to characterize urinary calculus composition in multiple publications [29,30,31,32,33,34,35]. Specifically, DECT enables the radiologist to determine whether calculi are predominantly made of calcium oxalate, uric acid, or cysteine (Fig. 3.4). The implications for patient therapy are substantial.
Two different patients with renal calculi. (a) Axial VNE CT image through the level of the kidneys demonstrating a left renal calculus (solid arrow). (b) The corresponding iodine image again demonstrates the left renal calculus (solid arrow). Given the persistent finding on both images, these findings represent a calcified calculus. In contrast, a uric acid calculus will be present on the VNE image but not on the iodine image. (c) In a separate patient, axial non-contrast image through the kidneys demonstrating a left renal calculus (solid arrow). (d) Corresponding color overlay DECT image again demonstrating a left renal calculus (solid arrow). (e) Graph demonstrating dual-energy indexes for different calculus composition. Renal calculus composition can be determined by comparing the ratio of attenuation on low- versus high-kVp images
Per the 2014 guidelines from the American Urologic Association, hydration is essential regardless of the type of urinary calculus [24]. For calcium oxalate calculi, recommendations on the dietary intake of sodium, calcium, oxalate, fruits/vegetables, and protein depend on urinary levels of calcium, oxalate, and citrate. Additional pharmacologic therapies may be indicated, such as thiazide diuretics for patients with calcium oxalate calculi or thiol drugs which bind cysteine when dietary modifications fail, or if there is a substantial stone burden. Lastly, differentiating uric acid (UA) calculi from non-uric acid calculi is useful, as uric acid stones may be treated with alkalization of urine [36]. Because UA calculi have increased attenuation at higher peak kVp compared to non-UA calculi which have higher attenuation at lower peak kVp, this distinction is possible with DECT [36].
3.5.2 Focal Renal Mass
Focal renal masses are a commonly encountered entity in abdominal imaging. It is estimated that over half of the adults over 50 years old will be shown to have at least one renal mass on cross-sectional imaging [37]. Fortunately, the vast majority of incidentally encountered renal masses are simple cysts, and can be characterize as such by the absence of enhancement on IV contrast-enhanced examinations. However, a clinical dilemma arises when a focal renal mass is encountered which is not clearly a simple cyst, namely masses with attenuation greater than 20 HU. In this scenario, the most common explanations for a hyperattenuating focal renal mass include a hemorrhagic or proteinaceous cyst, so-called pseudo-enhancement of a renal cyst under 2 cm in diameter, or a solid renal mass. Because the management for these diagnoses is considerably different, it is important to correctly characterize these incidentally encountered renal masses.
Although the majority of abdominopelvic CT scans in the ED are acquired with intravenous contrast in a single phase (portal venous), separation of material density pairs with post-processing enables characterization of incidentally encountered renal masses with VNE [38]. Furthermore, after many investigators have struggled to eliminate pseudo-enhancement of small renal masses [37], DECT has been shown to reliably overcome pseudo-enhancement on virtual monochromatic images [37]. By making the distinction between solid enhancing renal masses and other differential diagnoses, unnecessary follow-up examinations may be avoided, reducing cost, radiation exposure, and patient anxiety.
3.5.3 Detection of Urothelial Masses
Detection of urothelial masses can be difficult, particularly when they grow in a plaque-like manner along the course of the urinary tract. In patients presenting with new hydronephrosis without an obstructing renal calculus, radiologists in the ED may be charged with locating a urothelial mass. DECT urography has been described, and shows promise as a way to evaluate patients with hematuria while possibly reducing radiation exposure [39, 40]. With dual-energy CT, it is possible to use separation of material density pairs to better appreciate the enhancement of urothelial masses which may have caused signs and/or symptoms which have brought the patient to the ED. Although the enhancing masses will commonly be primary transitional cell carcinoma, metastases from other malignancies, including prostate, pancreas, and breast, are also possible.
3.6 Adrenal
3.6.1 Incidental Adrenal Nodules
As the use of cross-sectional imaging has increased markedly over the past 20 years, radiologists working in the emergency department often encounter incidental nodules and masses which are of indeterminate significance. The frequency of incidentally discovered adrenal nodules larger than 1 cm in size, in particular, is estimated to be between 3 and 7% in adults [41]. Adrenal nodules discovered incidentally therefore present one of the common challenges facing radiologists in this setting. In those adult patients without a history of malignancy, the vast majority of adrenal nodules will ultimately prove benign. In one study of 973 patients, with a total of 1049 adrenal nodules, none proved malignant [42]. While the clinical context is important in ascertaining whether further workup is required, such as a history of malignancy or comparison with a prior imaging examination, a certain number of patients will remain indeterminate on the initial evaluation. In this group there is a role for new technology, which can help triage patients with incidental adrenal nodules, possibly avoiding unnecessary further workup.
Because of the inherent ability of dual-energy CT to separate different material densities, VNE images can be created by separating iodine and water components from IV contrast-enhanced images. In recent years, a number of studies have attempted to characterize indeterminate renal mass using DECT [43,44,45,46,47].
Slebocki et al. assessed a group of 63 patients undergoing CT angiography after endovascular repair of an abdominal aortic aneurysm who were evaluated with both pre-contrast CT and single-phase CTA. Of those patients, six had adrenal abnormalities detected, and an excellent correlation was found between the true non-contrast (TNC) and VNE mean attenuation values (12.8 vs. 12.4 HU) [46]. Glazer et al. also found that VNE images can help triage adrenal nodules, and reported excellent interobserver agreement (kappa = 0.92) among multiple radiologists [43]. Gnannt et al. showed that there was no difference in the mean attenuation of adrenal nodules between VNE and TNC images acquired in the portal venous phase of IV contrast-enhanced CT [44]. Botsikas et al. reported a slightly higher mean HU for VNE images made from portal venous-phase images (4.02 HU higher for VNE), but no significant difference from VNE made from 15-min images [45]. Mileto et al. showed that VNE images may be better than TNC images for characterizing lipid-poor adrenal adenomas [47].
In summary, by using material density pairs, a radiologist in the ED can reliably characterize many incidental adrenal nodules on IV contrast-enhanced CT scans. This is true at a variety of kVp settings, and works in both the portal venous and arterial phases of acquisition (Fig. 3.5).
79-year-old man with abdominal pain. (a) Axial non-contrast CT image demonstrating a hyperattenuating subhepatic collection (solid arrow). Given that enteric contrast was previously administered, it was unclear whether this collection represented leakage of contrast. (b) Corresponding axial VNE image shows the same subhepatic collection (solid arrow). (c) Corresponding axial iodine image also demonstrates the same subhepatic collection (solid arrow). (d) Axial calcium image also demonstrates the hyperattenuating subhepatic collection (solid arrow). (e) On the corresponding non-calcium image, the same collection is hypoattenuating (solid arrow). The interpreting radiologist was unable to elicit a history to explain the findings, but the calcification within the collection indicates that it is chronic
3.7 Bowel
3.7.1 Acute Gastrointestinal Hemorrhage
Detection and localization of acute gastrointestinal hemorrhage present a diagnostic challenge for the practicing radiologist. The presence of high-density material within the bowel lumen (i.e., positive oral contrast or other ingested material) may confound CT findings which indicate the presence of acute blood products. Upper endoscopy and optical colonoscopy are essential diagnostic tools in the setting of acute gastrointestinal hemorrhage, and have the additional benefit of providing an opportunity for therapeutic intervention, either through clipping of a bleeding vessel or injection of a sclerosing agent. However, a number of factors, including the presence of a large amount of blood within the bowel lumen, may limit the ability of the gastroenterologist to detect and localize the source of acute bleeding.
As the technology of multi-detector computed tomography (MDCT) has advanced over the past two decades, MDCT has emerged as a valid diagnostic tool in the evaluation of patients with acute gastrointestinal hemorrhage [48,49,50,51,52,53]. Some challenges are inherent to the interpretation of MDCT examinations for the assessment of known or suspected acute gastrointestinal hemorrhage, namely the presence of intrinsically high-density material within the bowel lumen, which may confound the interpretation of a scan. Because of the ability of dual-energy computed tomography (DECT) to separate elements of different attenuation, and therefore to determine the iodine contribution to an image, there is an emerging role for the use of DECT in the evaluation of acute gastrointestinal hemorrhage in the ED setting [48,49,50,51,52,53].
3.7.2 Bowel Inflammation
Patients with inflammatory bowel disease (IBD) may present to the ED with acute or worsening abdominal pain. Some of the CT findings associated with bowel inflammation include hyperenhancement, bowel dilation, and bowel wall thickening, as well as secondary signs of mesenteric fat stranding. By separating material density pairs to create iodine maps, areas of bowel wall hyperenhancement may become more conspicuous and thus may be more readily detected (Fig. 3.6). Iodine maps may be beneficial in common conditions of bowel inflammation including inflammatory bowel disease, acute appendicitis, and diverticulitis [54]. There is ample opportunity for further research in this area.
37-year-old man with a history of Crohn disease presents with abdominal and rectal pain. (a) Axial IV contrast-enhanced CT image demonstrating wall thickening and enhancement involving the hepatic flexure of the colon (arrows). (b) Iodine image at a similar level demonstrating increased conspicuity of the finding (arrows). (c) IV contrast-enhanced CT image demonstrating a right perianal fistula extending towards the right gluteal cleft (arrow). (d) As before, an iodine image at a similar level also shows increased conspicuity of the same finding (arrow)
3.7.3 Bowel Ischemia
Acute bowel ischemia often presents a diagnostic challenge in the ED. Patients presenting with bowel ischemia usually have abdominal pain, signs of a systemic illness such as tachycardia, and/or hematochezia. These symptoms may appropriately prompt an IV contrast-enhanced CT of the abdomen and pelvis. However, “conventional” MDCT may lend to missing as many as 34% of acute mesenteric ischemia [55]. DECT may be useful in acute mesenteric ischemia. In animal models, it has been shown that DECT using a low kVp increases attenuation differences between normally perfused bowel and early ischemic bowel. The authors found this difference to be statistically significant for a lower kVp (51 keV) when compared to conventional 120 kVp images, but also saw improved conspicuity of early bowel ischemia when the kVp was 65 and 70 keV [56] (Fig. 3.7).
68-year-old man with cirrhosis, with increasing abdominal pain. (a) Axial IV contrast-enhanced CT image through the abdomen demonstrates right lower quadrant small bowel loops with wall thickening (solid arrow) and dilated small bowel loops in the left lower quadrant (open arrow). (b) Corresponding axial iodine image demonstrates absence of iodine uptake in the right lower quadrant small bowel loops (solid arrow), strongly suggesting ischemic bowel. In contrast, the left lower quadrant small bowel loops have normal iodine uptake (open arrow)
3.8 Trauma
Abdominopelvic trauma is common, and is a leading cause of morbidity and mortality among younger patients in the emergency setting. CT plays a crucial role in the workup of all hemodynamically stable trauma patients [57]. Portal venous-phase imaging is standard on abdominopelvic CT imaging. However, depending on the severity and mechanism of trauma, protocols can be tailored to the clinical history to include additional phases, i.e., arterial phase or delayed images [57]. In all patients with trauma, it is important to emphasize the need to be highly accurate while minimizing radiation dose to the patient. In this regard, DECT is an ideal technology that can be used to accomplish both of these goals without compromises.
3.8.1 Aortic Injury and Endoleak
In the setting of trauma, the aorta is best evaluated with multiphase imaging, typically including arterial and delayed phases. Non-enhanced images are not routinely acquired at most institutions, but can be helpful for the identification of intramural hematoma. Dual-energy CT can be helpful in these patients as VNE images can be created instead of the conventional non-enhanced acquisition, thus reducing radiation dose. While these images are noisier than the conventional non-enhanced images, they are of diagnostic quality in 95% of patients [6]. Additionally, with further development of DECT technology, image quality has been significantly improved such that the images currently closely resemble acquired non-enhanced images.
Another scenario where DECT has been shown to be helpful is for detection of endoleaks. Endovascular aneurysm repair is a commonly performed procedure for abdominal aortic aneurysms. A common complication of the procedure is subacute to chronic endoleak, which allows for filling of the aneurysm sac, and which is associated with an increased risk of rupture [58]. As previously suggested, CT with arterial and delayed phases is highly sensitive and specific for detection of endoleaks. Unlike in acute aortic injury, non-enhanced images are usually routinely acquired, particularly on the initial post-procedure CT scans. However, sensitivity, specificity, and accuracy may be further increased by using dual-energy technique. This is possible by using lower kVp images, which increases the attenuation of intravenously administered contrast (Fig. 3.8). In one study of 22 patients, endoleaks were shown to be more conspicuous on 80 kVp images when compared to images using 120 kVp [59].
70-year-old man following remote endovascular abdominal aortic aneurysm repair, with clinical concern for a graft endoleak. (a) CT angiogram axial image demonstrating endoleak along the left lateral aspect of the stent graft (arrow). (b) Low-keV image demonstrates increased conspicuity of the same finding despite the same window and level values (arrow). Also note the differences in attenuation on the low-keV images, best seen in the aorta. Intravenous contrast appears brighter on low-keV images. (c) The same finding on the iodine image is much more apparent (arrow)
3.8.2 Solid Organ Injury
Solid organ injury is very common in the setting of trauma. In order of frequency, the most commonly injured organs are spleen, liver, and kidney, with bowel and bladder less commonly injured [57]. The American Association for the Surgery of Trauma (AAST) has developed grading scales for injuries of the solid organs, taking into account size of hematomas and depth of lacerations. CT is an invaluable tool for early detection of these injuries. Typical CT trauma protocols include portal-venous-phase images through the abdomen and pelvis, with or without delayed images [57].
On CT images, lacerations appear as linear areas of hypoattenuation. While these findings may be detected on conventional CT, DECT is useful as it can accentuate the findings (Fig. 3.9). Iodine images can also be constructed to highlight areas of perfusion and areas of hypoenhancement [16, 60]. Other important findings on CT include subcapsular hematoma/hemoperitoneum, active extravasation of contrast, and pseudoaneurysm formation. All of these findings can be made more conspicuous on DECT images.
40-year-old man brought to the emergency department after a motor vehicle collision. (a) Axial IV contrast-enhanced CT image through the abdomen at the level of the pancreas demonstrates heterogeneous enhancement in the pancreatic head and neck (solid arrow), as well as hemoperitoneum and flattening of the inferior vena cava. (b) The iodine image confirms the presence of a laceration through the pancreatic neck (solid arrow)
As previously suggested, trauma CT protocols typically do not include non-enhanced images. While this is sufficient in most patients, there are situations where non-enhanced images can provide valuable information. In the setting of trauma, non-enhanced images may demonstrate subcapsular hematomas of the spleen or liver as a layer of hyperattenuating material overlying and conforming to the contour of the injured organ [60]. Similarly, hemoperitoneum can be subtle, particularly when there are only small amounts adjacent to enhancing bowel loops or other hyperattenuating structures. On post-contrast images, the blood products may be isoattenuating relative to the adjacent organ. Given that non-contrast images are not routinely acquired, these findings can potentially be missed on “routine” IV contrast-enhanced images. However, review of VNE images on DECT will demonstrate increased contrast differentiation between hematoma and adjacent injured organ [60] (Fig. 3.10).
46-year-old woman with known metastatic melanoma presents to the emergency department with abdominal pain. (a) Axial IV contrast-enhanced CT image demonstrating hypoattenuating material layering anterior to the liver (solid arrow). (b) On the corresponding VNE image, the collection appears hyperattenuating (solid arrow), using the fluid seen in the stomach as an internal reference. (c) On the corresponding iodine image, there is no iodine uptake in the collection (solid arrow). This collection represents hemoperitoneum from hepatic melanoma metastases
IV contrast extravasation is a critical finding to identify on CT, as it strongly suggests the need for more invasive management, i.e., angiographic intervention or surgical management. Active arterial (and occasionally venous) contrast extravasation appears as blush of contrast within or adjacent to solid organs or bowel, which should change its morphology and density on delayed images [57]. As these findings can be subtle, DECT can be used to increase conspicuity [60]. Subtle hyperattenuating foci can be more easily detected on iodine images or low-keV images.
A diagnostic dilemma occurs on a single-phase CT examination. In this setting, a single image demonstrating a hyperdense focus may represent contrast extravasation, vascular or gastrointestinal, or other intrinsically hyperdense material. This is further complicated if the finding is seen in the vicinity of a comminuted fracture with osseous fragments or calcified atherosclerotic plaque potentially mimicking contrast extravasation. Dual-energy CT can be very helpful in this scenario, as review of VNE in conjunction with iodine images can help to differentiate between the two. Calcium will be present on both VNE and iodine images. However, extravasated contrast will only be seen on iodine images, and not on VNE images [60] (Fig. 3.11).
69-year-old man after endovascular abdominal aortic aneurysm repair with concern for endoleak. (a) Axial CT angiogram image clearly depicts two areas of hyperattenuation (arrows) within the aneurysm sac—a curvilinear structure on the right and an amorphous structure anteriorly. It is unclear whether these findings represent an endoleak or atherosclerotic calcification. (b) VNE image demonstrating the curvilinear structure in the right side of the aneurysm sac (arrow), but not the hyperattenuating material anteriorly. (c) Iodine images demonstrate both areas of hyperattenuation. There is curvilinear calcification along the right side of the aneurysm sac (arrow), and an endoleak anteriorly (open arrow)
3.8.3 Musculoskeletal Injury
Fractures are commonly seen in the emergency setting. This is true even in the absence of trauma, given that elderly patients can present with insufficiency fractures. While displaced fractures are easily detected on conventional CT images, non-displaced and minimally displaced fractures can often be difficult to detect. In these patients, clinicians typically order MRI for detection of bone marrow edema [61]. While MRI is an excellent tool, it is relatively expensive, time consuming, and sometimes technically difficult to perform in the inpatient setting. An alternative approach is to use DECT, which can be used to subtract calcium from the image. This process removes trabecular bone from the image, allowing bone marrow to be visualized. On the resulting virtual non-calcium images, hyperattenuation in the marrow represents marrow edema and/or hemorrhage [7, 10, 14]. Aside from detection, this technique can also be helpful for characterization of fractures. As previously suggested, elderly patients are often incidentally noted to have spinal compression fractures on CT examinations. In the absence of prior imaging examinations for comparison, these fractures are often interpreted as age indeterminate, leading clinicians to then order MRI or nuclear bone scan for further characterization. As with fracture detection, DECT can be used in these patients to characterize the fractures. In these patients, the presence of marrow edema or hemorrhage is consistent with acute to subacute fracture, while their absence suggests chronicity.
Conclusion
Dual-energy CT has a wide range of applications in diagnostic imaging in the emergency department. Familiarity with the role of DECT in the evaluation of the gallbladder, pancreas, kidneys, bowel, vasculature, and musculoskeletal system can enable timely and accurate diagnoses while frequently eliminating the need for further diagnostic imaging workup.
References
Scales CD Jr, Smith AC, Hanley JM, Saigal CS, Urologic Diseases in America. Prevalence of kidney stones in the United States. Eur Urol. 2012;62(1):160–5.
Larson DB, Johnson LW, Schnell BM, Salisbury SR, Forman HP. National trends in CT use in the emergency department: 1995-2007. Radiology. 2011;258(1):164–73.
Uyeda JW, Sahani DV. Dual-energy CT in the acute abdomen. Curr Radiol Rep. 2015;3:20.
Silva AC, Morse BG, Hara AK, Paden RG, Hongo N, Pavlicek W. Dual-energy (spectral) CT: applications in abdominal imaging. Radiographics. 2011;31(4):1031–46. discussion 1047-1050
Karcaaltincaba M, Aktas A. Dual-energy CT revisited with multidetector CT: review of principles and clinical applications. Diagn Interv Radiol. 2011;17(3):181–94.
Vlahos I, Chung R, Nair A, Morgan R. Dual-energy CT: vascular applications. AJR Am J Roentgenol. 2012;199(5 Suppl):S87–97.
Patino M, Prochowski A, Agrawal MD, Simeone FJ, Gupta R, Hahn PF, et al. Material separation using dual-energy CT: current and emerging applications. Radiographics. 2016;36(4):1087–105.
Heye T, Nelson RC, Ho LM, Marin D, Boll DT. Dual-energy CT applications in the abdomen. AJR Am J Roentgenol. 2012;199(5 Suppl):S64–70.
Marin D, Boll DT, Mileto A, Nelson RC. State of the art: dual-energy CT of the abdomen. Radiology. 2014;271(2):327–42.
Grajo JRPM, Prochowski A, Sahani DV. Dual energy CT in practice: basic principles and applications. Appl Radiol. 2016;45(7):6–12.
Agrawal MD, Oliveira GR, Kalva SP, Pinho DF, Arellano RS, Sahani DV. Prospective comparison of reduced-iodine-dose virtual monochromatic imaging dataset from dual-energy CT angiography with standard-iodine-dose single-energy ct angiography for abdominal aortic aneurysm. AJR Am J Roentgenol. 2016;207(6):W125–32.
Foley WD, Shuman WP, Siegel MJ, Sahani DV, Boll DT, Bolus DN, et al. White paper of the society of computed body tomography and magnetic resonance on dual-energy CT, Part 2: radiation dose and iodine sensitivity. J Comput Assist Tomogr. 2016;40(6):846–50.
Shuman WP, Chan KT, Busey JM, Mitsumori LM, Koprowicz KM. Dual-energy CT aortography with 50% reduced iodine dose versus single-energy CT aortography with standard iodine dose. Acad Radiol. 2016;23(5):611–8.
Aran S, Daftari Besheli L, Karcaaltincaba M, Gupta R, Flores EJ, Abujudeh HH. Applications of dual-energy CT in emergency radiology. AJR Am J Roentgenol. 2014;202(4):W314–24.
Lewis M, Reid K, Toms AP. Reducing the effects of metal artefact using high keV monoenergetic reconstruction of dual energy CT (DECT) in hip replacements. Skelet Radiol. 2013;42(2):275–82.
Yeh BM, Shepherd JA, Wang ZJ, Teh HS, Hartman RP, Prevrhal S. Dual-energy and low-kVp CT in the abdomen. AJR Am J Roentgenol. 2009;193(1):47–54.
Kim JE, Lee JM, Baek JH, Han JK, Choi BI. Initial assessment of dual-energy CT in patients with gallstones or bile duct stones: can virtual nonenhanced images replace true nonenhanced images? AJR Am J Roentgenol. 2012;198(4):817–24.
Shoheiber O, Biskupiak JE, Nash DB. Estimation of the cost savings resulting from the use of ursodiol for the prevention of gallstones in obese patients undergoing rapid weight reduction. Int J Obes Relat Metab Disord. 1997;21(11):1038–45.
Uyeda JW, Richardson IJ, Sodickson AD. Making the invisible visible: improving conspicuity of noncalcified gallstones using dual energy CT. Abdom Radiol. 2017;42(12):2933–9.
Barakos JA, Ralls PW, Lapin SA, Johnson MB, Radin DR, Colletti PM, et al. Cholelithiasis: evaluation with CT. Radiology. 1987;162(2):415–8.
Hickman MS, Schwesinger WH, Bova JD, Kurtin WE. Computed tomographic analysis of gallstones. An in vitro study. Arch Surg. 1986;121(3):289–91.
Bennett GL, Rusinek H, Lisi V, Israel GM, Krinsky GA, Slywotzky CM, et al. CT findings in acute gangrenous cholecystitis. AJR Am J Roentgenol. 2002;178(2):275–81.
Chen AL, Liu AL, Wang S, Liu JH, Ju Y, Sun MY, et al. Detection of gallbladder stones by dual-energy spectral computed tomography imaging. World J Gastroenterol. 2015;21(34):9993–8.
Pearle MS, Goldfarb DS, Assimos DG, Curhan G, Denu-Ciocca CJ, Matlaga BR, et al. Medical management of kidney stones: AUA guideline. J Urol. 2014;192(2):316–24.
Boridy IC, Nikolaidis P, Kawashima A, Sandler CM, Goldman SM. Noncontrast helical CT for ureteral stones. World J Urol. 1998;16(1):18–21.
Ather MH, Jafri AH, Sulaiman MN. Diagnostic accuracy of ultrasonography compared to unenhanced CT for stone and obstruction in patients with renal failure. BMC Med Imaging. 2004;4(1):2.
Ulusan S, Koc Z, Tokmak N. Accuracy of sonography for detecting renal stone: comparison with CT. J Clin Ultrasound. 2007;35(5):256–61.
Scheffel H, Stolzmann P, Frauenfelder T, Schertler T, Desbiolles L, Leschka S, et al. Dual-energy contrast-enhanced computed tomography for the detection of urinary stone disease. Investig Radiol. 2007;42(12):823–9.
Duan X, Li Z, Yu L, Leng S, Halaweish AF, Fletcher JG, et al. Characterization of urinary stone composition by use of third-generation dual-source dual-energy CT with increased spectral separation. AJR Am J Roentgenol. 2015;205(6):1203–7.
Kulkarni NM, Eisner BH, Pinho DF, Joshi MC, Kambadakone AR, Sahani DV. Determination of renal stone composition in phantom and patients using single-source dual-energy computed tomography. J Comput Assist Tomogr. 2013;37(1):37–45.
Leng S, Huang A, Cardona JM, Duan X, Williams JC, McCollough CH. Dual-energy CT for quantification of urinary stone composition in mixed stones: a phantom study. AJR Am J Roentgenol. 2016;207(2):321–9.
Matlaga BR, Kawamoto S, Fishman E. Dual source computed tomography: a novel technique to determine stone composition. Urology. 2008;72(5):1164–8.
Ogawa N, Sato S, Ida K, Kato K, Ariyoshi Y, Wada K, et al. Evaluation of urinary stone composition and differentiation between urinary stones and phleboliths using single-source dual-energy computed tomography. Acta Med Okayama. 2017;71(2):91–6.
Zhang GM, Sun H, Xue HD, Xiao H, Zhang XB, Jin ZY. Prospective prediction of the major component of urinary stone composition with dual-source dual-energy CT in vivo. Clin Radiol. 2016;71(11):1178–83.
Zilberman DE, Ferrandino MN, Preminger GM, Paulson EK, Lipkin ME, Boll DT. In vivo determination of urinary stone composition using dual energy computerized tomography with advanced post-acquisition processing. J Urol. 2010;184(6):2354–9.
Aran S, Shaqdan KW, Abujudeh HH. Dual-energy computed tomography (DECT) in emergency radiology: basic principles, techniques, and limitations. Emerg Radiol. 2014;21(4):391–405.
Silverman SG, Israel GM, Herts BR, Richie JP. Management of the incidental renal mass. Radiology. 2008;249(1):16–31.
Neville AM, Gupta RT, Miller CM, Merkle EM, Paulson EK, Boll DT. Detection of renal lesion enhancement with dual-energy multidetector CT. Radiology. 2011;259(1):173–83.
Ascenti G, Mileto A, Gaeta M, Blandino A, Mazziotti S, Scribano E. Single-phase dual-energy CT urography in the evaluation of haematuria. Clin Radiol. 2013;68(2):e87–94.
Chen CY, Hsu JS, Jaw TS, Shih MC, Lee LJ, Tsai TH, et al. Split-bolus portal venous phase dual-energy CT urography: protocol design, image quality, and dose reduction. AJR Am J Roentgenol. 2015;205(5):W492–501.
Berland LL, Silverman SG, Gore RM, Mayo-Smith WW, Megibow AJ, Yee J, et al. Managing incidental findings on abdominal CT: white paper of the ACR incidental findings committee. J Am Coll Radiol. 2010;7(10):754–73.
Song JH, Chaudhry FS, Mayo-Smith WW. The incidental adrenal mass on CT: prevalence of adrenal disease in 1,049 consecutive adrenal masses in patients with no known malignancy. AJR Am J Roentgenol. 2008;190(5):1163–8.
Glazer DI, Maturen KE, Kaza RK, Francis IR, Keshavarzi NR, Parker RA, et al. Adrenal Incidentaloma triage with single-source (fast-kilovoltage switch) dual-energy CT. AJR Am J Roentgenol. 2014;203(2):329–35.
Gnannt R, Fischer M, Goetti R, Karlo C, Leschka S, Alkadhi H. Dual-energy CT for characterization of the incidental adrenal mass: preliminary observations. AJR Am J Roentgenol. 2012;198(1):138–44.
Botsikas D, Triponez F, Boudabbous S, Hansen C, Becker CD, Montet X. Incidental adrenal lesions detected on enhanced abdominal dual-energy CT: can the diagnostic workup be shortened by the implementation of virtual unenhanced images? Eur J Radiol. 2014;83(10):1746–51.
Slebocki K, Kraus B, Chang DH, Hellmich M, Maintz D, Bangard C. Incidental findings in abdominal dual-energy computed tomography: correlation between true noncontrast and virtual noncontrast images considering renal and liver cysts and adrenal masses. J Comput Assist Tomogr. 2017;41(2):294–7.
Mileto A, Nelson RC, Marin D, Roy Choudhury K, Ho LM. Dual-energy multidetector CT for the characterization of incidental adrenal nodules: diagnostic performance of contrast-enhanced material density analysis. Radiology. 2015;274(2):445–54.
Ernst O, Bulois P, Saint-Drenant S, Leroy C, Paris JC, Sergent G. Helical CT in acute lower gastrointestinal bleeding. Eur Radiol. 2003;13(1):114–7. https://doi.org/10.1007/s00330-002-1442-y.
Yamaguchi T, Yoshikawa K. Enhanced CT for initial localization of active lower gastrointestinal bleeding. Abdom Imaging. 2003;28(5):634–6.
Rajan R, Dhar P, Praseedom RK, Sudhindran S, Moorthy S. Role of contrast CT in acute lower gastrointestinal bleeding. Dig Surg. 2004;21(4):293–6.
Yoon W, Jeong YY, Shin SS, Lim HS, Song SG, Jang NG, et al. Acute massive gastrointestinal bleeding: detection and localization with arterial phase multi-detector row helical CT. Radiology. 2006;239(1):160–7.
Jaeckle T, Stuber G, Hoffmann MH, Jeltsch M, Schmitz BL, Aschoff AJ. Detection and localization of acute upper and lower gastrointestinal (GI) bleeding with arterial phase multi-detector row helical CT. Eur Radiol. 2008;18(7):1406–13.
Sodhi JS, Zargar SA, Rashid W, Shaheen F, Singh M, Javid G, et al. 64-section multiphase CT enterography as a diagnostic tool in the evaluation of obscure gastrointestinal bleeding. Indian J Gastroenterol. 2012;31(2):61–8.
Fulwadhva UP, Wortman JR, Sodickson AD. Use of dual-energy CT and iodine maps in evaluation of bowel disease. Radiographics. 2016;36(2):393–406.
Firetto MC, Lemos AA, Marini A, Avesani EC, Biondetti PR. Acute bowel ischemia: analysis of diagnostic error by overlooked findings at MDCT angiography. Emerg Radiol. 2013;20(2):139–47.
Potretzke TA, Brace CL, Lubner MG, Sampson LA, Willey BJ, Lee FT Jr. Early small-bowel ischemia: dual-energy CT improves conspicuity compared with conventional CT in a swine model. Radiology. 2015;275(1):119–26.
Soto JA, Anderson SW. Multidetector CT of blunt abdominal trauma. Radiology. 2012;265(3):678–93.
Hong C, Heiken JP, Sicard GA, Pilgram TK, Bae KT. Clinical significance of endoleak detected on follow-up CT after endovascular repair of abdominal aortic aneurysm. AJR Am J Roentgenol. 2008;191(3):808–13.
Chandarana H, Godoy MC, Vlahos I, Graser A, Babb J, Leidecker C, et al. Abdominal aorta: evaluation with dual-source dual-energy multidetector CT after endovascular repair of aneurysms--initial observations. Radiology. 2008;249(2):692–700. https://doi.org/10.1148/radiol.2492080359.
Nicolaou S, Eftekhari A, Sedlic T, Hou DJ, Mudri MJ, Aldrich J, et al. The utilization of dual source CT in imaging of polytrauma. Eur J Radiol. 2008;68(3):398–408.
Feldman F, Staron R, Zwass A, Rubin S, Haramati N. MR imaging: its role in detecting occult fractures. Skelet Radiol. 1994;23(6):439–44.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG, part of Springer Nature
About this chapter
Cite this chapter
Yu, H., Bates, D.D.B., Sahani, D.V. (2018). Dual-Energy CT in Patients with an Acute Abdomen. In: Patlas, M., Katz, D., Scaglione, M. (eds) MDCT and MR Imaging of Acute Abdomen. Springer, Cham. https://doi.org/10.1007/978-3-319-70778-5_3
Download citation
DOI: https://doi.org/10.1007/978-3-319-70778-5_3
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-70777-8
Online ISBN: 978-3-319-70778-5
eBook Packages: MedicineMedicine (R0)