Abstract
Allergenicity depends on several factors determined by the very physicochemical nature of the molecules themselves, i.e., their capacity to penetrate the horny layer, lipophilicity, and chemical reactivity. The sensitizing property of the majority of contact allergens could be predicted from these characteristics (Patlewicz et al., Contact Dermatitis 50:91–97, 2004; Gerberick et al., Altern Lab Anim 36(2):215–242, 2008). Two other factors, however, further contribute to the allergenicity of chemicals, namely, their pro-inflammatory activity and capacity to induce maturation of LC. These issues will be dealt with in more detail in the following sections.
Along with their migration and settling within the draining lymph nodes, haptenized LC further mature, as characterized by their increased expression of costimulatory and antigen-presentation molecules (Cumberbatch et al., Arch Dermatol Res 289:277–284, 1997; Heufler et al. J Exp Med 167:700–705, 1988). In addition, they adopt a strongly veiled, interdigitating appearance, thus maximizing the chances of productive encounters with naive T lymphocytes and recognition of altered self (Steinman et al., J Invest Dermatol 105:2S–7S, 1995; Furue et al., Br J Dermatol 135:194–198, 1996; Schuler and Steinman, J Exp Med 161:526–546, 1985).
The intricate structure of lymph node paracortical areas, the differential expression of chemokines and their receptors, the characteristic membrane ruffling of IDC, and the predominant circulation of naïve T lymphocytes through these lymph node areas provide optimal conditions for T-cell-receptor binding, i.e., the first signal for induction of T-cell activation (Banchereau and Steinman, Nature 392:245–252, 1998). Intimate DC–T-cell contacts are further strengthened by secondary signals, provided by sets of cellular adhesion molecules, and growth-promoting cytokines (reviewed in Hommel, Immunol Cell Biol 82:62–66, 2004; Cella et al., Curr Opin Immunol 9:10–16, 1997).
In healthy individuals, primary skin contacts with contact allergens lead to differentiation and expansion of allergen-specific effector T cells displaying Th1, Th2, and/or Th17 cytokine profiles. The same allergens, if encountered along mucosal surfaces, favor the development of allergen-specific Th2 and Th17 effector cells, and/or Th3 and Tr1 allergen-specific regulatory T cells. Whereas the first two subsets may assist or replace Th1 cells in pro-inflammatory effector functions, the latter two subsets are mainly known for downregulating immune responsiveness. For most, if not all allergens, along with prolonged allergenic contacts, the role of Th2 cells as effector cells gradually increases given reduced longevity of Th1 responses.
The respective contributions of similar subsets of allergen-specific CD8 + T cells are still unknown, but distinct effector roles of allergen-specific Tc1 and Tc2 have been postulated.
Priming via the skin results in CLA positive T cells, which upon inflammatory stimuli preferentially enter the skin; on the other hand, gut homing T cells have been primed and generated along mucosal surfaces. Upon priming, T cells loose much of their capacity to recirculate via the lymph nodes, but gain the capacity to enter the tissues. In particular recently activated T cells will enter skin inflammatory sites. ACD reactions are primarily infiltrated by CD4 and/or CD8 pro-inflammatory cells, later reactions may be dominated by Th2 cells and regulatory T cells. Skin infiltation by T cells is fine-tuned by sets of adhesion molecules and chemokine receptors, whose expression is not rigid, but can be modulated by micro-environmental factors.
After antigenic activation the progeny of primed T cells is released via the efferent lymphatics into the bloodstream. Circulating allergen-specific cells can be challenged in vitro to provide diagnostic parameters for contact hypersensitivity. At least for water-soluble allergens, like metal salts, the degree of allergen-specific proliferation and cytokine production, in particular type-2 cytokines, correlate with clinical allergy. For routine application of a broad spectrum of allergens, culture conditions still need to be improved. For mechanistic in vitro studies in ACD, however, with selected sets of relatively nontoxic allergens, peripheral blood provides an excellent source of lymphocytes and antigen-presenting cells.
ACD reactions can be mediated by classical effector cells, i.e., allergen-specific CD4+ type-1 T cells which, upon triggering by allergen-presenting cells, produce IFN-γ to activate nonspecific inflammatory cells like macrophages. However, CD8 + T cells, and other cytokines, including IL-4, IL-17, and IL-22 can also play major roles in ACD. The conspicuous difference with DTH reactions induced by intradermal administration of protein antigens, i.e., the epidermal infiltrate, can largely be attributed to hapten-induced chemokine release by keratinocytes.
Access provided by Autonomous University of Puebla. Download reference work entry PDF
Similar content being viewed by others
Keywords
- Activation-induced cell death
- Bandrowski’s base
- Effector memory T cells
- Imprinting process
- Langerhans cells
- Prehaptens
- Prohaptens
- Tetrachlorosalicylanilide
1 Core Messages
-
Allergenicity depends on several factors determined by the very physicochemical nature of the molecules themselves, i.e., their capacity to penetrate the horny layer, lipophilicity, and chemical reactivity. The sensitizing property of the majority of contact allergens could be predicted from these characteristics (Patlewicz et al. 2004; Gerberick et al. 2008). Two other factors, however, further contribute to the allergenicity of chemicals, viz. their pro-inflammatory activity and capacity to induce maturation of LC. These issues will be dealt with in more detail in the following sections.
-
Along with their migration and settling within the draining lymph nodes, haptenized LC further mature, as characterized by their increased expression of costimulatory and antigen-presentation molecules (Cumberbatch et al. 1997; Heufler et al. 1988). In addition, they adopt a strongly veiled, interdigitating appearance, thus maximizing the chances of productive encounters with naive T lymphocytes and recognition of altered self (Steinman et al. 1995; Furue et al. 1996; Schuler and Steinman 1985).
-
The intricate structure of lymph node paracortical areas, the differential expression of chemokines and their receptors, the characteristic membrane ruffling of IDC, and the predominant circulation of naïve T lymphocytes through these lymph node areas provide optimal conditions for T-cell-receptor binding, i.e., the first signal for induction of T-cell activation (Banchereau and Steinman 1998). Intimate DC–T-cell contacts are further strengthened by secondary signals, provided by sets of cellular adhesion molecules, and growth-promoting cytokines (reviewed in Hommel (2004), Cella et al. (1997)).
-
In healthy individuals, primary skin contacts with contact allergens lead to differentiation and expansion of allergen-specific effector T cells displaying Th1, Th2, and/or Th17 cytokine profiles. The same allergens, if encountered along mucosal surfaces, favor the development of allergen-specific Th2 and Th17 effector cells, and/or Th3 and Tr1 allergen-specific regulatory T cells. Whereas the first two subsets may assist or replace Th1 cells in pro-inflammatory effector functions, the latter two subsets are mainly known for downregulating immune responsiveness. For most, if not all allergens, along with prolonged allergenic contacts, the role of Th2 cells as effector cells gradually increases given reduced longevity of Th1 responses.
-
The respective contributions of similar subsets of allergen-specific CD8+ T cells are still unknown, but distinct effector roles of allergen-specific Tc1 and Tc2 have been postulated.
-
Priming via the skin results in CLA positive T cells, which upon inflammatory stimuli preferentially enter the skin; on the other hand, gut homing T cells have been primed and generated along mucosal surfaces. Upon priming, T cells loose much of their capacity to recirculate via the lymph nodes, but gain the capacity to enter the tissues. In particular recently activated T cells will enter skin inflammatory sites. ACD reactions are primarily infiltrated by CD4 and/or CD8 pro-inflammatory cells, later reactions may be dominated by Th2 cells and regulatory T cells. Skin infiltation by T cells is fine-tuned by sets of adhesion molecules and chemokine receptors, whose expression is not rigid, but can be modulated by micro-environmental factors.
-
After antigenic activation the progeny of primed T cells is released via the efferent lymphatics into the bloodstream. Circulating allergen-specific cells can be challenged in vitro to provide diagnostic parameters for contact hypersensitivity. At least for water-soluble allergens, like metal salts, the degree of allergen-specific proliferation and cytokine production, in particular type-2 cytokines, correlate with clinical allergy. For routine application of a broad spectrum of allergens, culture conditions still need to be improved. For mechanistic in vitro studies in ACD, however, with selected sets of relatively nontoxic allergens, peripheral blood provides an excellent source of lymphocytes and antigen-presenting cells.
-
ACD reactions can be mediated by classical effector cells, i.e., allergen-specific CD4+ type-1 T cells which, upon triggering by allergen-presenting cells, produce IFN-γ to activate nonspecific inflammatory cells like macrophages. However, CD8+ T cells, and other cytokines, including IL-4, IL-17, and IL-22 can also play major roles in ACD. The conspicuous difference with DTH reactions induced by intradermal administration of protein antigens, i.e., the epidermal infiltrate, can largely be attributed to hapten-induced chemokine release by keratinocytes.
2 Introduction
During the past few decades, the understanding of why, where, and when allergic contact dermatitis (ACD) might develop has rapidly increased. Critical discoveries include the identification of T cells as mediators of cell-mediated immunity, their thymic origin and recirculation patterns, and the molecular basis of their specificity to just one or few allergens out of the thousands of allergens known (Janeway 2008). Progress has also resulted from the identification of genes that determine T-cell function, and the development of monoclonal antibodies that recognize their products. Moreover, the production of large amounts of recombinant products, e.g., cytokines and chemokines, and the breeding of mice with disruptions in distinct genes (knockout mice) or provided with additional genes of interest (transgenic mice), have allowed in-depth analysis of skin-inflammatory processes, such as those taking place in ACD.
Although humoral antibody-mediated reactions can be a factor, ACD depends primarily on the activation of allergen-specific T cells (Bergstresser 1989), and is regarded as a prototype of delayed hypersensitivity, as classified by Turk (1975) and Gell and Coombs (type IV hypersensitivity) (Gell et al. 1975). Evolutionarily, cell-mediated immunity has developed in vertebrates to facilitate eradication of microorganisms and toxins. Elicitation of ACD by usually nontoxic doses of small molecular weight allergens indicates that the T-cell repertoire is often slightly broader than one might wish. Thus, ACD can be considered to reflect an untoward side effect of a well-functioning immune system.
Subtle differences can be noted in macroscopic appearance, time course, and histopathology of allergic contact reactions in various vertebrates, including rodents and man (Mestas and Hughes 2004). Nevertheless, essentially all basic features are shared. Since both mouse and guinea pig models, next to clinical studies, have greatly contributed to our present knowledge of ACD, both data sets provide the basis for this chapter.
In ACD, a distinction should be made between induction (also known as sensitization or primary) and effector (also known as elicitation or secondary) phases (Saint-Mezard et al. 2003) (Fig. 1). The induction phase includes the events following a first contact with the allergen and is complete when the individual is sensitized and capable of giving a positive ACD reaction. The effector phase begins upon elicitation (challenge) and results in clinical manifestation of ACD. The entire process of the induction phase requires at least 4 days to several weeks, whereas the effector phase reaction is fully developed within 1–4 days. Main episodes in the induction phase (steps 1–5) and effector phase (step 6) are:
-
1.
Binding of allergen to skin components. The allergen penetrating the skin readily associates with all kinds of skin components, including major histocompatibility complex (MHC) proteins. These molecules, in humans encoded for by histocompatibility antigen (HLA) genes, are abundantly present on epidermal antigen-presenting cells, called Langerhans cells (LC) (Lepoittevin 2006; Gerberick et al. 2008; Divkovic et al. 2005).
-
2.
Hapten-induced activation of allergen-presenting cells. Allergen-carrying LC become activated, mature, and travel via the afferent lymphatics to the regional lymph nodes, where they settle as so-called interdigitating cells (IDC) in the paracortical T-cell areas.
-
3.
Recognition of allergen-modified LC by specific T cells. In nonsensitized individuals the frequency of T cells with certain specificities is usually far below 1 per million. Within the paracortical areas, conditions are optimal for allergen-carrying IDC to encounter naive T cells that specifically recognize the allergen–MHC molecule complexes. The dendritic morphology of these allergen-presenting cells strongly facilitates multiple cell contacts, leading to binding and activation of allergen-specific T cells.
-
4.
Proliferation of specific T cells in draining lymph nodes. Supported by interleukin (IL)-1, released by the allergen-presenting cells, activated T cells start producing several growth factors, including IL-2 (Hoyer et al. 2008). A partly autocrine cascade follows since at the same time receptors for IL-2 are upregulated in these cells, resulting in vigorous blast formation and proliferation within a few days.
-
5.
Systemic propagation of the specific T-cell progeny. The expanded progeny is subsequently released via the efferent lymphatics into the blood flow and begins to recirculate. Thus, the frequency of specific effector-memory T cells in the blood may rise to as high as one in a thousand, whereas most of these cells display receptor molecules facilitating their migration into peripheral tissues. In the absence of further allergen contacts, their frequency gradually decreases in subsequent weeks or months, but does not return to the low levels found in naive individuals.
-
6.
Effector phase. By renewed allergen contact, the effector phase is initiated, which depends not only on the increased frequency of specific T cells, and their altered migratory capacities, but also on their low activation threshold. Thus, within the skin, allergen-presenting cells and specific T cells can meet, and lead to plentiful local cytokine and chemokine release. The release of these mediators, many of which have a pro-inflammatory action, causes the arrival of more inflammatory cells, thus further amplifying local mediator release. This leads to a gradually developing eczematous reaction that reaches its maximum after 18–72 h and then declines.
Immunological events in allergic contact dermatitis (ACD). During the induction phase (left), skin contact with a hapten triggers migration of epidermal Langerhans cells (LC) via the afferent lymphatic vessels to the skin-draining lymph nodes. Haptenized LC home into the T cell-rich paracortical areas. Here, conditions are optimal for encountering naïve T cells that specifically recognize allergen–MHC molecule complexes. Hapten-specific T cells now expand abundantly and generate effector and memory cells, which are released via the efferent lymphatics into the circulation. With their newly acquired homing receptors, these cells can easily extravasate peripheral tissues. Renewed allergen contact sparks off the effector phase (right). Due to their lowered activation threshold, hapten-specific effector T cells are triggered by various haptenized cells, including LC and keratinocytes (KC), to produce proinflammatory cytokines and chemokines. Thereby, more inflammatory cells are recruited further amplifying local inflammatory mediator release. This leads to a gradually developing eczematous reaction, reaching a maximum within 18–48 h, after which reactivity successively declines
The following sections discuss these six main episodes of the ACD reaction in more detail. Furthermore, the chapter will discuss local hyperreactivity, such as flare-up and retest reactivity, and hyporeactivity, i.e., upon desensitization or tolerance induction.
3 Binding of Contact Allergens to Skin Components
Chemical Nature of Contact Allergens. Most contact allergens are small, chemically reactive molecules with a molecular weight less than 500 Da (Bos and Meinardi 2000) (Fig. 2). Since these molecules are too small to be antigenic themselves, contact sensitizers are generally referred to as haptens.
Hapten presentation by epidermal Langerhans cells. Allergen penetrating into the epidermis readily associates with all kinds of skin components, including major histocompatibility complex (MHC) proteins, abundantly present on epidermal Langerhans cells (LC). Both MHC class I and class II molecules may be altered directly or via intracellular hapten processing and, subsequently, be recognized by allergen-specific CD8+ and CD4+ T cells
Upon penetration through the epidermal horny layer, haptens readily conjugate to endogenous epidermal and dermal molecules. Sensitizing organic compounds may covalently bind to protein nucleophilic groups, such as thiol, amino, and hydroxyl groups, as is the case with poison oak/ivy allergens (reviewed in Roberts and Lepoittevin 1998). Examples of contact allergens containing electrophilic components include aldehydes, ketones, amides, or polarized bonds. Metal ions, e.g., nickel cations, instead form stable metal–protein chelate complexes by coordination bonds (Budinger and Hertl 2000). The most reactive nucleophilic side chains are those found in the amino acids lysine, cysteine, and histidine (Gerberick et al. 2008). Of note, their degree of ionization and hence nucleophilicity is dependent on the pH of the microenvironment which is influenced by surrounding amino acids as well as protein location within the epithelium (Divkovic et al. 2005). Predicting the chemicals that can function as haptens in allergic contact dermatitis as well as identifying cutaneous proteins involved in hapten–protein complexes is the subject of current intense investigations (Gerberick et al. 2008; Mutschler et al. 2009; Lepoittevin 2006).
Hapten Presentation by Langerhans cells (LC). Sensitization is critically dependent on direct association of haptens with epidermal LC-bound MHC molecules, or peptides present in the groove of these molecules. Both MHC class I and class II molecules may be altered this way, and thus give rise to allergen-specific CD8+ and CD4+ T cells, respectively. Distinct differences between allergens can, however, arise from differences in chemical reactivity and lipophilicity (Fig. 2), since association with MHC molecules may also result from internalization of the haptens, followed by their intracellular processing as free hapten molecules or hapten–carrier complexes. Lipophilic haptens can directly penetrate into LC, conjugate with cytoplasmic proteins and be processed along the “endogenous” processing route, thus favoring association with MHC class I molecules (Blauvelt et al. 2003). In contrast, hydrophilic allergens such as nickel ions may, after conjugation with skin proteins, be processed along the “exogenous” route of antigen processing and thus favor the generation of altered MHC class II molecules. Thus, the chemical nature of the haptens can determine to what extent allergen-specific CD8+ and/or CD4+ T cells will be activated (Kimber et al. 2002; Liberato et al. 1981; Kalish et al. 1994; Toebak et al. 2006).
Pre- and Prohaptens. Whereas most contact allergens can form hapten–carrier complexes spontaneously, some need activation first. Contact allergens requiring activation outside the body, e.g., by UV-light or oxygen, are called prehaptens (Naisbitt 2004; Lepoittevin 2006). The typical photoallergen tetrachlorosalicylanilide is a prototype of this. Tetrachlorosalicylanilide, which undergoes photochemical dechlorination with UV irradiation, ultimately provides photoadducts with skin proteins (Epling et al. 1988). Contact allergens dependent on activation inside the body, e.g., by enzyme-induced metabolic conversion, are referred to as prohaptens. A classical prohapten is p-phenylenediamine, which needs to be oxidized by N-acetyltransferases to a reactive metabolite which can form a trimer, known as Bandrowski’s base (Krasteva et al. 1993; Merk et al. 2004). Reduced enzyme activity in certain individuals, related to genetic enzyme polymorphisms, explains the reduced risk of sensitization to prohaptens that need enzymatic activation (Schnuch et al. 1998; Karlberg et al. 2008; Blömeke et al. 2009).
4 Hapten-Induced Activation of Allergen-Presenting Cells
Physiology of Langerhans Cells. Although originally thought to be neurons based on their staining properties and cellular morphology (Langerhans 1868), LC subsequently were surmised to function as “professional” antigen-presenting cells (Wilson and Villadangos 2004). They form a contiguous network within the epidermis and represent 2–5% of the total epidermal cell population (Hoath and Leahy 2003). Their principal functions are internalization, processing, transport, and presentation of skin-encountered antigens (Breathnach 1988; Romani et al. 2003). As such, LC play a pivotal role in the induction of cutaneous immune responses to infectious agents as well as to contact sensitizers (Kimber and Dearman 2003; Inaba et al. 1986; Kimber and Cumberbatch 1992). Recent studies of LC indicate that this cell type has direct epidermal innervations and can respond to a number of neurotransmitters (among them are calcitonin gene-related peptide, α-melanocyte stimulating hormone and substance P) (Luger 2002). Most of the experimental evidence to date indicates a suppressive effect of the neurohormones and neuropeptides on Langerhans cell function and cutaneous inflammation, but it has become evident lately that the timing of exposure to a stimulus is critical to the outcome of the immune response (Romagnani 2006). Thus, administration of a stress hormone or exposure to a stressor before the LC encounters an allergen may diminish the immune response toward that substance, while a stressor may enhance immune function when acting on a maturing LC or before reexposure to the allergen (Seiffert and Granstein 2006). LC originate from CD34+ bone marrow progenitors, entering the epidermis via the blood stream (Dieu et al. 1998). Their continuous presence in the epidermis is also assured by local proliferation (Stingl et al. 1978; Czernielewski and Demarchez 1987). They reside as relatively immature DC, characterized by a high capacity to gather antigens by macropinocytosis, whereas their capacity to stimulate naïve T cells is still underdeveloped at this stage (Streilein and Grammer 1989). Their prominent dendritic morphology and the presence of distinctive Birbeck granules were observed long ago (Langerhans 1868; Birbeck 1961; Braathen 1980). In the last decade, their pivotal function in the induction of skin immune responses was explained by high expression of molecules mediating antigen presentation (e.g., MHC class I and II, CD1), as well as of cellular adhesion and costimulatory molecules (e.g., CD54, CD80, CD86, and cutaneous lymphocyte antigen [CLA]) (Kimber et al. 1998, 2002; Park et al. 1998).
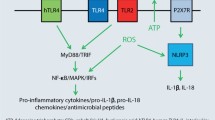
Hapten-Induced LC Activation. Upon topical exposure to contact sensitizers, or other appropriate stimuli (e.g., trauma, irradiation), up to 40% of the local LC become activated (Weinlich et al. 1998; Richters et al. 1994), leave the epidermis, and migrate, via afferent lymphatic vessels, to the draining lymph nodes (Jakob et al. 2001) (Fig. 3). This process of LC migration results from several factors, including contact allergen-induced production of cytokines favoring LC survival (Ozawa et al. 1996; Wong et al. 1997; Aiba and Tagami 1999) and loosening from surrounding keratinocytes (Inaba et al. 1993; Jakob and Udey 1998; Schwarzenberger and Udey 1996). Thus, within 15 min after exposure to a contact sensitizer, production of IL-1β mRNA is induced (Enk 1992; Enk et al. 1993). Along with this, caspase-1, formerly known as interleukin-1-converting enzyme, is activated and cleaves the active IL1 β cytokine from the translated precursor-IL1β protein. Caspase-1 activates also IL-18 from its precursor form. These inflammatory processes are now viewed at as making up the “inflammasome” (Iversen and Johansen 2008). IL-1β in concert with IL-18 stimulates release of tumor necrosis factor (TNF)-α and granulocyte-macrophage colony-stimulating factor (GM-CSF) from keratinocytes (Iversen and Johansen 2008). Together, these three cytokines facilitate migration of LC from the epidermis toward the lymph nodes (Wang et al. 2003; Steinman et al. 1995). IL-1β and TNF-α downregulate membrane-bound E-cadherin expression and thus cause disentanglement of LC from surrounding keratinocytes (Fig. 3) (Schwarzenberger and Udey 1996; Tang et al. 1993; Jakob and Udey 1998). Simultaneously, adhesion molecules are upregulated promoting LC migration by mediating interactions with the extracellular matrix and dermal cells, such as CD54, α 6 integrin, and CD44 variants (Ma et al. 1994; Rambukhana et al. 1995; Price et al. 1997; Weiss et al. 1997; Brand et al. 1999). Also, production of the epidermal basement membrane degrading enzyme metalloproteinase-9 is upregulated in activated LC (Kobayashi 1997).
(a–d) Hapten-induced migration of Langerhans cells. (a) In a resting state, epidermal Langerhans cells (LC) reside in suprabasal cell layers, tightly bound to surrounding keratinocytes (KC), e.g., by E-cadherin. (b) Early after epidermal hapten exposure, LC produce IL-1β and IL-18, which induces the release of IL-1α, TNF-α and GM-CSF from keratinocytes. Together, these three cytokines facilitate migration of LC from the epidermis toward the lymph nodes. (c) Emigration of LC starts with cytokine-induced disentanglement from surrounding keratinocytes (e.g., by downregulation of E-cadherin) and production of factors facilitating penetration of the basal membrane (e.g., matrix metalloproteinases) and interactions with extracellular matrix and dermal cells (e.g., integrins and integrin ligands). (d) Once in the dermis, LC migration is directed toward the draining afferent lymphatic vessels, guided by local production of chemokines (e.g., CCL19 and CCL21) acting on newly expressed chemokine receptors, such as CCR7, on activated LC. Along their journey, haptenized LC further mature as characterized by their increased dendritic morphology and expression of costimulatory and antigen-presentation molecules
Next, LC migration is directed by hapten-induced alterations in chemokine receptor levels (Randolph 2001). Upon maturation, LC downregulate expression of receptors for inflammatory chemokines (e.g., CCR1, 2, 5, and 6), whereas others (including CCR4, 7, and CXCR4) are upregulated (Fig. 3) (reviewed by Sallusto (1998a), Zlotnik et al. (1999), Caux et al. (2000), Sallusto et al. (1999)). Notably, CCR7 may guide maturing LC into the draining lymphatics and the lymph node paracortical areas, since two of its ligands (CCL19 and 21) are produced by both lymphatic and high endothelial cells (Saeki et al. 1999; Gunn et al. 1998). Importantly, the same receptor-ligand interactions cause naive T cells, which also express CCR7, to accumulate within the paracortical areas (Kim and Broxmeyer 1999). Migratory responsiveness of both cell types to CCR7 ligands is promoted by leukotriene C4, released from these cells via the transmembrane transporter molecule Abcc1 (previously called MRP1) (Randolph 2001; Robbiani et al. 2000; Honig et al. 2003; van de Ven et al. 2009). Interestingly, Abcc1 belongs to the same superfamily as the transporter associated with antigen-processing TAP, known to mediate intracellular peptide transport in the “endogenous route” which favors peptide association with MHC Class I molecules. Final positioning of the LC within the paracortical T-cell areas may be due to another CCR7 ligand, EBI1-ligand chemokine (ELC, CCL19), produced by resident mature DC (Sallusto 1998).
5 Recognition of Allergen-Modified Langerhans’ Cells by Specific T Cells
Homing of Naive T Cells into Lymph Nodes. More than 90% of naive lymphocytes present within the paracortical T-cell areas have entered the lymph nodes by high endothelial venules (HEV) (Haig et al. 1999). These cells are characterized not only by CCR7 but also by the presence of a high molecular weight isoform of CD45 (CD45RA) (Haig et al. 1999; Altin and Sloan 1997). Entering the lymph nodes via HEV is established by the lymphocyte adhesion molecule l-selectin (CD62L), which allows rolling interaction along the vessel walls by binding to peripheral node addressins (PNAd), such as GlyCAM-1 or CD34 (Schon et al. 2003; von Andrian and Mrini 1998; Vestweber and Blanks 1999). Next, firm adhesion is mediated by the interaction of CD11a/CD18 with endothelial CD54, resulting in subsequent endothelial transmigration. Extravasation and migration of naïve T cells to the paracortical T-cell areas is supported by chemokines such as CCL18, 19, and 21 produced locally by HEV and by hapten-loaded and resident DC (Robbiani et al. 2000; Adema et al. 1997; Ngo et al. 1998; Nagira et al. 1997). In non-sensitized individuals, frequencies of contact-allergen-specific T cells are very low, and estimates vary from 1 per 109 to maximally 1 per 106 (Haig et al. 1999; Rustemeyer et al. 1999). Nevertheless, the preferential homing of naive T cells into the lymph node paracortical areas, and the large surface area of interdigitating cells, make allergen-specific T-cell activation likely with only few dendritic cells exposing adequate densities of haptenized MHC molecules (Crivellato et al. 2004; Itano and Jenkins 2003).
Activation of Hapten-Specific T Cells. As outlined in “Binding of Contact Allergens to Skin Components,” the chemical nature of the hapten determines its eventual cytoplasmic routing in antigen-presenting cells (APC), and thus whether presentation will be predominantly in context of MHC class I or II molecules (Fig. 2). T cells, expressing CD8 or CD4 molecules can recognize hapten-MHC class I or II complexes showing stabilized MHC membrane expression (Griem et al. 1998; Moulon et al. 1995). Chances of productive interactions with T cells are high since each MHC-allergen complex can trigger a high number of T-cell receptor (TCR) molecules (“serial triggering”) (Li et al. 2004). Moreover, after contacting specific CD4+ T cells, hapten-presenting DC may reach a stable super-activated state, allowing for efficient activation of subsequently encountered specific CD8+ T cells (Schoenberger et al. 1998). The actual T-cell activation is executed by TCRξ-chain mediated signal transduction, followed by an intracellular cascade of biochemical events, including protein phosphorylation, inositol phospholipid hydrolysis, increase in cytosolic Ca2+ (Gascoigne and Zal 2004; Cantrell 1996), and activation of transcription factors, ultimately leading to gene activation (Fig. 4) (Kuo and Leiden 1999).
Activation of hapten-specific T cells. T-cell receptor (TCR) triggering by hapten–major histocompatibility complex (MHC) complexes (“signal 1”) is insufficient for T-cell activation. But “professional” antigen-presenting cells (APC), such as Langerhans cells, can provide the required costimulation (“signal 2”), involving secreted molecules, such as cytokines, or sets of cellular adhesion molecules present on the outer cellular membranes of APC and T cells. T cells, stimulated in this way, activate nuclear responder elements (e.g., CD28RE). Together with nuclear transcription factors (NF), produced upon TCR triggering, these nuclear responder elements enable transcription of T-cell growth factors, e.g., IL-2. APC–T cell interaction gives rise to mutual activation (“amplification”): on APC, ligation of CD40 with CD154 molecules on T cells induces overexpression of several costimulatory molecules, including CD80 and CD86. In turn, these molecules bind to and increase expression of CD28 on T cells. This interaction stabilizes CD154 expression, causing amplified CD154–CD40 signaling, and preserves strong IL-2 production, finally resulting in abundant T-cell expansion
For activation and proliferation, TCR triggering (“signal 1”) is insufficient, but hapten-presenting APC also provide the required costimulation (“signal 2”; Fig. 5) (Davis and van der Merwe 2003; Trautmann and Randriamampita 2003). The costimulatory signals may involve secreted molecules, such as cytokines (IL-1), or sets of cellular adhesion molecules (CAMs) and their counter-structures present on the outer cellular membranes of APC and T cells. Expression levels of most of these CAMs vary with their activational status, and thus can provide positive stimulatory feedback loops. For example, as mentioned above, after specific TCR binding and ligation of CD40L (CD154) on T cells with CD40 molecules, APC reach a superactivated state, characterized by overexpression of several CAMs, including CD80 and CD86 (Fig. 4) (Acuto and Michel 2003; Quezada et al. 2004). In turn, these molecules bind to and increase expression of CD28 on T cells. This interaction stabilizes CD154 expression, causing amplified CD154–CD40 signaling (Dong et al. 2003).
Spectrum of allergen driven CD4+ T cell differentiation: current schematic view. Depending on the immunological microenvironment (amount of allergen, danger signals, and other soluble mediators), activated naïve T cells are skewed into distinct phenotypes. The presence of allergen and sufficient danger signals leads to the development of effector T cell phenotypes of ACD. Presence of IL-6, TGF-β, IL-21, and IL-23 stimulates the generation of TH17/Th23 cells. Development of TH1 cells is stimulated by the presence of IL-12 and IFN-γ, and the development of TH2 is favored by IL-4. The absence of sufficient danger signals stimulates the development of tolerogenic phenotypes, including TH3 and Tr1 (Larsen 2009; Dong 2008; Zhu 2008; Duhen 2009; Ward 2009; Cavani 2008; Sallusto 2009)
The activational cascade is, as illustrated above, characterized by mutual activation of both hapten-presenting APC and hapten-reactive T cells. Whereas this activation protects the APC from apoptotic death and prolongs their life to increase the chance of activating their cognate T cells, only the latter capitalize on these interactions by giving rise to progeny. As discussed below, to promote T-cell growth, cellular adhesion stimuli need to be complimented by a broth of cytokines, many of which are released by the same APC. Together, elevated expression levels of (co-)stimulatory molecules on APC and local abundance of cytokines overcome the relatively high activation threshold of naive T cells (Viola and Lanzavecchia 1996).
6 Proliferation and Differentiation of Specific T Cells
T-Cell Proliferation. Upon their activation, naive allergen-specific T cells start producing several cytokines, including IL-2, the classical T-cell growth factor (Crispin 2009; Malek 2009). In particular, ligation of T cell-bound CD28 receptors unleashes full-scale IL-2 production in T cells by increasing IL-2 transcription and mRNA stabilization (Pei 2008). T-cell IL-2 production peaks within 24 h and declines subsequently (Villarino 2007). Concomitant upregulation of the IL-2 receptor α-chain facilitates the assembly of high-affinity IL-2 receptor complexes which augment autocrine T-cell responsiveness, thus providing a positive feedback loop leading to T-cell clonal expansions up to 1,000 fold (Lan 2008). The process of proliferation can be visible as an impressive, sometimes painful lymph node swelling.
T-Cell Differentiation. Whereas allergen specificity remains strictly conserved along with their proliferation, within few days T cells show distinct expression of transcription factors associated with varying cytokine production profiles and surface marker expression (Zhu and Paul 2008; Sallusto 2009; Dong 2008; Oboki 2008). Thus, the recent offspring of allergen-specific CD4+ T cells can show at least five distinct cytokine profiles, generally associated with helper/effector or regulatory/suppressive functions (Fig. 5). Type-1 Th cells are characterized by a predominant release of IFN-γ, IL-2, and TNF-β, all known as prototypical pro-inflammatory and cytotoxic cytokines. Type-2 Th cells secrete IL-4, IL-5, IL-13, which have distinct pro-inflammatory activities but are most prominent in promoting humoral antibody production, e.g., along mucosal surfaces where IgA contributes to exclusion of microbial entry (Dabbagh and Lewis 2003; Faria and Weiner 2006). Next, the Th3 subset is distinguished by its release of transforming growth factor (TGF)-β, which displays anti-inflammatory activities (Allan et al. 2008; Faria and Weiner 2006). Recently, Th-17 cells have been recognized as a separate lineage of pro-inflammatory T cells, characterized by production of IL-17A and IL-17E, as well as IL-22, all of which play pivotal roles in auto-immune diseases, e.g., by recruiting neutrophils and macrophages (Korn et al. 2009; Louten 2009). Finally, still another subset of CD4+ T cells is recognized for its strong regulatory role in controlling inflammatory reactivities, i.e., the Tr1 cells or “inducible Tregs,” characterized by the secretion of IL-10 (Zhu and Paul 2008; Allan et al. 2008; Sallusto 2009). This CD4+ T-cell population is phenotypically remarkably heterogeneous, with part of the cells expressing high amounts of the high affinity IL-2 receptor (“CD25 high”), either or not accompanied by expression of the transcription factor FoxP3 (Romagnani 2006; Feuerer 2009; Cavani 2008) (Fig. 5). Tr1 cells have essential roles in maintenance of immune homeostasis, regulating effector T-cell responses and preventing their potentially pathogenic effects by various indirect ways, e.g., by suppressing macrophage functions (Wu et al. 2007; Shevach 2009). Each of these five cytokine profiles is under control of distinct sets of transcription factors which are shown in Fig. 5, but discussed further elsewhere (e.g., Zhu and Paul 2008; Wilson 2009; Zhou and Littman 2009; Basso et al. 2009; Dong 2008).
To some extent the same distinct cytokine profiles may develop in CD8+ T cells, where at least type 1 and 2 cytokine-releasing CD8+ cells are known to contribute to allergic contact dermatitis (Kimber and Dearman 2002; Coulter et al. 2010).
Several factors are thought to contribute to the above-described polarized cytokine production profiles in allergen-specific T cells, including (1) the site and cytokine environment of first allergenic contact, (2) the molecular nature and concentrations of the allergen, and (3) the neuroendocrine factors.
Cytokine Environment. In the skin-draining lymph nodes, allergen-activated LC and dermal dendritic cells rapidly produce large amounts of IL-12, switching off IL-4 cytokine production, thus promoting the differentiation of Th1 cells (Nakamura et al. 1997b; Kang et al. 1996; Pulendran 2004). Of note, since Th1 cells retain, next to IL12R, high IL-4R expression they remain sensitive to IL-4 as a growth factor (Kubo et al. 1997). Thus, they also retain the capacity to shift cytokine production toward the type-2 profile. In contrast, type-2 T cells, e.g., developing in mucosa-draining lymph nodes, rapidly lose the genes encoding the IL-12-R β2 chain and thus, type-2 differentiation is irreversible (Rogge et al. 1997; Zhou et al. 2009).
Early differentiation of type-1 T cells is promoted by microbial danger-signal-induced IL-12 and IL-18, leading to IFN-γ release by nonspecific “bystander” cells, e.g., DC and NK cells, within the lymph nodes (Nakamura et al. 1997; Orange and Biron 1996). IFN-γ interferes with skewing toward other cytokine profiles. Since Th1 cells rapidly lose functional IFN-γR expression, these cells, in contrast to e.g., Th2, Th3 and Th17 cells, become refractory to the growth-inhibitory effects of IFN-γ (Groux et al. 1997; Gajewski and Fitch 1988; Takatori et al. 2008). Interestingly, T-cell skewing may also be facilitated by primary contact-mediated signals, e.g., Th1 skewing by CD154 ligation through APC-bound CD40 [124], or Th2 skewing by ligation of CD134 (OX40) through APC-bound CD252 (Ohshima et al. 1997; Croft et al. 2009).
In the process of T-cell skewing toward the other major cytokine profiles, TGF-β plays a central role. TGF-β can be produced by various cell types, including Th3 cells themselves, but is most prominently produced by mucosal epithelial cells (Zhu and Paul 2008; Takatori et al. 2008; Iliev et al. 2009). Apparently, in conjunction with IL-10 production, e.g., produced by mucosal B cells, allergen-stimulated T cells rapidly initiate endogenous TGF-β production thus revealing the Th3 phenotype (Izcue et al. 2009). These cells may stimulate IgA production along the mucosae, but elsewhere immunosuppressive activities prevail. Interestingly, in conjunction with abundant local IL-2 production, such as induced by strong antigenic stimulation involving most effective CD28 triggering, TGF-β favors skewing toward IL-10 production, thus providing an effective immunoregulatory feedback loop (Hoyer et al. 2008; Letourneau 2009). Still, in the presence of strong and persistant microbial molecule-induced danger/growth signals, e.g., IL-6, IL-21, and IL-23, TGF-β induces the development of Th17 and/or Th22 cells, which both have been postulated to contribute to various allergic and autoimmune disorders (Larsen 2009; Oboki 2008; Louten 2009; Oukka 2008; Takatori et al. 2008) (Fig. 5).
Thus, ACD may be caused by any combination of at least three distinct types of effector T cells, releasing type-1, -2, and -17/22 cytokines, respectively. Considering that contact allergens will mainly enter via the skin, type-1 pro-inflammatory T cells are thought to represent the primary effector cells in ACD (Edele et al. 2007; Fyhrquist-Vanni et al. 2007). Nevertheless, in sensitized individuals, type-2 T cells also play a role, as shown by both IL-4 production and allergen-specific type-2 T cells in the blood and at ACD reaction sites (see Sect. 7) (Werfel et al. 1997; Probst et al. 1995; Grewe et al. 1998). Their role may increase along with the longevity of sensitization, since several factors contribute to shifting type-1 to type-2 responses, including reversibility of the former and not of the latter T cells, as mentioned above (Perez et al. 1995; Ulrich et al. 2001). Still, other sets of cytokines, including IL-17 and/or IL-22, are important in immune defense mechanisms, and thus Th17 and or Th22 cells have also been found to mediate allergic and autoimmune disorders (van Beelen et al. 2007). Given rapid local release of both IL-4 and TGF-β within mucosal tissues, mucosal allergen contacts, if accompanied by strong danger signals, may lead in particular to Th2 and Th17 effector cells. Without these signals, rather immunoregulatory subsets (Th3, Tr1) would develop, as is observed in the induction of “oral tolerance” (see below) (Mucida and Salek-Ardakani 2009).
Nature of the Allergen. A second factor in determining T-cell cytokine-production profiles, although still poorly understood, is the molecular character of the contact allergen itself, and the resulting extent of TCR triggering (Constant and Bottomly 1997; Constant et al. 1995). For both protein and peptide antigens, high doses of antigen might favor type-2 responses, whereas intermediate/low doses would induce type-1 T-cell responses (Constant and Bottomly 1997; Bretscher et al. 1997). Strong antigenic stimulation was also shown to upregulate CD40L expression on T cells and, in combination with microbial-induced IL-6, to promote, Th17 differentiation (Lezzi 2009). To what extent this translates to contact allergens is still unclear. Certainly, endogenous capacities of contact allergens to provide danger signals and activate the “inflammasome,” in combination with their capacity to induce differentiation-skewing cytokines (in particular IL-4, IL-6, IL-12, and IL-23), will affect the outcome (Toebak and Moed 2006; Watanabe 2008). In this respect, some contact allergens are notorious for inducing type-2 responses, even if their primary contact is by the skin route, e.g., trimellitic acid, which is also known as a respiratory sensitizer (Kanerva et al. 1997).
Neuroendocrine Factors. Diverse neuroendocrine factors co-determine T-cell differentiation (Geenen and Brilot 2003; Luger and Lotti 1998, 2002). An important link has been established between nutritional deprivation and decreased T-cell-mediated allergic contact reactions (Lord et al. 1998). Apparently, adipocyte-derived leptin, a hormone released by adequately nourished and functioning fat cells, is required for type-1 T-cell differentiation. Administration of leptin to mice restored ACD reactivity in mice during starvation (Lord et al. 1998). Also, androgen hormones and adrenal cortex-derived steroid hormones, e.g., dehydroepiandrosterone (DHEA), promote type-1 T-cell and ACD reactivity. DHEA, like testosterone, may favor differentiation of type-1 T cells by promoting IFN-γ and suppressing IL-4 release (Morfin et al. 2000; Cutolo et al. 2002). In contrast, the female sex hormone progesterone furthers the development of type-2 CD4+ T cells and even induces, at least transient, IL-4 production and CD30 expression in established type-1 T cells (Kidd 2003; Piccinni 1995). Type-2 T-cell polarization is also facilitated by adrenocorticotrophic hormone (ACTH) and glucocorticosteroids (Vieira et al. 1998), and by prostaglandin (PG)E2 (Calder et al. 1992). PGE 2, released from mononuclear phagocytes, augments intracellular cAMP levels, resulting in inhibition of pro-inflammatory cytokine production, like IFN-γ and TNF-α (Uotila 1996; Demeure et al. 1997; Abe et al. 1997; Kalinski et al. 1997), and thus can influence the development of effector T cells in ACD.
7 Systemic Propagation of the Specific T-Cell Progeny
T-Cell Recirculation. Upon sensitization via the skin, the progeny of primed T cells is released via the efferent lymphatic vessels of the skin draining lymph nodes and the thoracic duct into the blood (Fig. 6). If the first encounter with allergen occurs via the intestinal route (e.g., along with induction of oral tolerance), priming will take place in the Peyer’s patches and mesenteric lymph nodes, and primed T cells will be released from there to the circulation. The subsequent recirculation and homing pattern of primed T cells is guided by adhesion molecules and chemokine receptors which they express on the cell membrane (Table 1). As outlined below, expression of these molecules is determined by the site of priming, as well as by the activational state of the T cells. In addition, there is a distinct relationship between the sets of chemokine and homing receptors expressed by T cells and their type of differentiation.
Systemic propagation of hapten-specific T cells. From the skin-draining lymphoid tissue, the progeny of primed T cells is released via the efferent lymphatic vessels and the thoracic duct (DT) into the blood and becomes part of the circulation. Like their naïve precursors, these CCR7 + effector/memory T cells can still enter lymphoid tissues and settle in paracorticale areas by binding to its ligands CCL19 and CCL21. But increased expression of skin-homing molecules, e.g., cutaneous lymphocyte antigen (CLA), facilitates their spontaneous migration in the skin
First, primed T cells have different homing receptors depending on the site of priming, a process called “imprinting” (Woodland 2009; Edele 2008). During priming of allergen-specific T cells in the skin draining lymph nodes, both CD4+ and CD8+ T cells are stimulated to express cutaneous lymphocyte antigen (CLA; Fuhlbrigge et al. 1997) and the chemokine receptors CCR4 and CCR10, a phenotype that predisposes for eventual migration to the skin. In the mesenteric lymph nodes, on the other hand, T cells are stimulated to express the integrin α4:β7 and the chemokine receptor CCR9, a phenotype which predisposes for gut homing. An instructive role of the peripheral tissues in this imprinting process was demonstrated in a mouse model on T cell priming by dendritic cells, where either dermal or intestinal cells were added to the cultures, resulting in T cells expressing mouse “CLA” or α4:β7 integrin, respectively (Edele 2008). Whereas for the imprint of gut homing retinoic acid was identified as a crucial factor, for the imprint of skin homing, the active metabolite of vitamin D3 was shown to be essential, because it induces CCR10 expression in T cells (Sigmundsdottir 2008). Still, for induction of CLA and thus for establishing the full skin homing profile, cell-cell contact and/or other mediators like IL-12 seem to be required (Edele 2008).
After priming and imprinting, circulating gut homing memory T lymphocytes, bearing the α4:β7 integrin, can attach to intestinal endothelial cells by binding to the mucosal vascular addressin MAdCAM-1. Further infiltration in the mucosa is guided by chemokines, such as CCL25, produced by small intestinal epithelial cells (Miles 2008). Thus, along the gut, T lymphocyte progeny is attracted that has been generated in other mucosal tissues. Likewise, in the skin, CLA-positive cells are attracted that have been generated in skin draining lymph nodes. CLA binds to E-selectin (CD62E) on dermal endothelial cells, while CCR4 and CCR10 expression allow the lymphocytes to migrate in the skin toward CCL17 and CCL27 produced by keratinocytes in the epidermis.
At least as important for the recirculation and homing characteristics of T cells is the activational state of the cells. In this respect, primed T cells can be divided into two main subsets: the central memory T cells (T CM) and the effector memory T cells (T EM). Like their naive precursors, T CM can still enter the peripheral lymphoid tissues, due to the fact that they continue to express CD62L and CCR7, allowing for binding to high endothelial venules in the lymph nodes and migration into the paracortical areas. T EM, on the other hand, have lost these molecules and migrate, due to simultaneous upregulation of several other adhesion molecules, preferentially to peripheral inflamed tissues. T EM are characterized by rapid effector function upon antigenic stimulation, but, in the absence of antigenic stimuli, T EM eventually convert to T CM by reacquiring CCR7 and CD62L. In turn, T CM may convert to T EM upon antigenic restimulation (Sallusto et al. 1999, 2004; Woodland 2009; Sallusto 2009).
Peripheral endothelial binding and extravasation of T cells to inflamed tissues requires the expression of both selectins and integrins on the T cell membrane, such as LFA-1, VLA-4, and PSGL-1. The vascular expression of their respective ligands (Table 1) is strongly increased by cytokines released at inflammatory sites. The density of adhesion molecules on the T cell membrane is generally upregulated upon activation, in particular in T EM. Since their expression is highest only for short periods after activation, only recently activated T cells show a unique propensity to enter skin sites and exert effector functions.
Third, the differentiation of T cells (Th1, Th2, etc.) is clearly associated with distinct homing chracteristics. T cells biased toward a pro-inflammatory phenotype show a higher propensity to enter skin sites, as compared to mucosal tissues (Austrup et al. 1997; Fuhlbrigge et al. 1997; Larsen 2009; Duhen 2009). In mice, the early influx of type-1 T cells into delayed-type hypersensitivity (DTH) reactions was found to be more efficient than that of type-2 T cells, although both cell types expressed CLA. Here, CD162, highly expressed by type-1 T cells, was found to be important for this preferential homing (Borges et al. 1997). Also the pattern of chemokine receptors differs between the Th subsets (Table 1). Some receptors, such as CXCR3, are preferentially expressed on Th1 cells, whereas others like CCR4 and CCR8 are in particular expressed by Th2 cells (Ward 2009; Cavani 2008; Hudak 2002; Sallusto 2009). The latter chemokine receptors are not only overexpressed on type-2 cytokine-producing T cells, but also on basophils and eosinophils. Together, these cells strongly contribute to local immediate allergic hyperresponsiveness. The more recently described Th17 and Th22 lymphocyte subsets expressing CCR4, CCR6, and CCR10 (Ward 2009; Duhen 2009) are attracted to the skin by epidermal CCL17, CCL20, and CCL27, respectively (Table 1, Fig. 5). Overall, results obtained thus far favor the view that the pro-inflammatory subsets (Th1 and Th17/22) will be the first to enter skin sites upon local inflammatory stimuli, their primary function being an early control of antigenic pressure, e.g., through amplification of macrophage effector functions. The ACD reaction is, however, a dynamic process, in which the first influx of cells influences the local chemokine environment and determines the type of subsequent infiltrating cells. Thus, upon repeated exposure to contact allergens gradually Th2 cells and regulatory cells may dominate (Kitagaki 1997). Interestingly, also at the T-cell level modulation of the cytokine and chemokine receptor profiles may occur, thereby maintaining plasticity of the immune response (Wilson 2009; Sallusto 2009). Of note, the actual composition of the T-cell infiltrate in ACD skin lesions does not only depend on the influx of lymphocytes, but should rather be regarded as the resultant of infiltration, apoptosis, and retention of lymphocytes, next to their emigration to the lymphatics.
Finally, the antigen specificity of T cells contributes to their migration pattern. Allergens penetrated via the epidermis and displayed at the dermal endothelial surface may be recognized by allergen-specific T cells, thus resulting in activation, immobilization, and transendothelial migration of these cells at sites of allergen exposure (Ward 2009).
Allergen-Specific T-Cell Recirculation: Options for In Vitro Testing. The dissemination and recirculation of primed, allergen-specific T cells in the body suggests that peropheral blood offers a most useful and accessible source for T cell based in vitro assays for ACD (Fig. 6). A major advantage of in vitro testing would be the noninterference with the patient’s immune system, thus eliminating any potential risk of primary sensitization and boosting by in vivo skin testing. Although such tests have found several applications in fundamental research, e.g., on recognition of restriction elements, cross-reactivities and cytokine-profile analyses, their use for routine diagnostic purposes is still limited. Even in highly sensitized individuals, frequencies of contact allergen-specific memory/effector cells may still be below 1 per 104 (Cavani et al. 1998; Lindemann 2008; Kalish 1990). Given the relatively small samples of blood obtainable by venepuncture (at only one or a few time points), numbers of specific T cells in any culture well used for subsequent in vitro testing would typically be below 100 cells/well. For comparison, in vivo skin test reactions recruit at least 1,000 times more specific T cells from circulating lymphocytes passing by for the period of testing, i.e., at least 24 h (von Blomberg et al. 1991).
Therefore, the sensitivity of in vitro assays, e.g., allergen-induced proliferation or cytokine production may not always be sufficient to pick up weak sensitization. Intermediate or strong sensitization is, however, readily detected in vitro by both proliferation and cytokine production assays (Lindemann 2008; Bordignon 2008; Minang 2005; Moed 2005). With respect to the latter, both the “Elispot” assay, where allergen induced cytokine production is evaluated at the single cell level, and the cytokine evaluation in allergen-stimulated culture supernatants provide adequate information (Rustemeyer et al. 2004; Spiewak 2007; Minang 2005). Notably with respect to cytokine production, type-2 cytokines appear to provide most specific parameters for contact sensitization in these assays (Rustemeyer et al. 2004; Minang 2008), although generally both Th1 and Th2 cytokines are being produced in vitro by allergic individuals upon allergen exposure (Moed 2005; Minang 2006).
Importantly, most of the above-mentioned, successful in vitro studies evaluated hydrophilic allergens, like nickel, chromium, and palladium salts. Reports on successful in vitro assays with other hydrophobic and more toxic allergens are scarce (Moed 2005; Wahlkvist 2007; Skazik 2008). Appropriate allergen presentation is a major hurdle in in vitro studies, because of the broad range of requirements for different allergens with unique solubilities, toxicities, and reactivity profiles. Moreover, in the absence of LC, monocytes are the major source of APC, and their numbers in peripheral blood vary substantially within and between donors. Of note, optimal APC function is particularly critical for in vitro activation of resting memory T cells, since, in the absence of repeated allergenic contacts, activated effector memory T cells (T EM) may finally revert to a more naïve phenotype, with a higher threshold for triggering (Sallusto et al. 2004; Boyman 2009). Supplementing in vitro test cultures with appropriate mixtures of cytokines may, however, compensate for suboptimal APC function (Rustemeyer et al. 1999, 2004; Moed 2005).
8 The Effector Phase of Allergic Contact Dermatitis
Elicitation of ACD. Once sensitized, individuals can develop ACD upon reexposure to the contact allergen. Positive patch test reactions mimic this process of allergen-specific skin hyperreactivity. Thus, skin contacts induce an inflammatory reaction that, in general, is maximal within 2–3 days and, without further allergen supply, declines thereafter (Fig. 7). Looked at superficially, the mechanism of this type of skin hyperreactivity is straightforward: allergen elicitation or challenge leads to the (epi)dermal accumulation of contact allergen-specific memory/effector T lymphocytes which, upon encountering allergen-presenting cells, are reactivated to release pro-inflammatory cytokines. These, in turn, spark the inflammatory process, resulting in macroscopically detectable erythema and induration. As compared to immediate allergic reactions, developing within a few minutes after mast-cell degranulation, ACD reactions show a delayed time course, since both the migration of allergen-specific T cells from the dermal vessels and local cytokine production need several hours to become fully effective. Still, the picture of the rise and fall of ACD reactions is far from clear. Some persistent issues are discussed below, notably: (1) irritant properties of allergens, (2) role of early-phase reactivity, (3) T-cell patrol and specificity, (4) effector T-cell phenotypes, and (5) down regulatory processes.
The effector phase of allergic contact dermatitis. (a) 0 h: In resting skin relatively few randomly patrolling, skin-homing CLA+ T cells are present. (b) 0–4 h: Reexposure of the contact allergen, binding to (epi)dermal molecules and cells, induces release of proinflammatory cytokines. (c) 2–6 h: Influenced by inflammatory mediators, activated epidermal Langerhans cells (LC) start migrating toward the basal membrane and endothelial cells express increased numbers of adhesion molecules. Endothelial cell-bound hapten causes preferential extravasation of hapten-specific T cells, which are further guided by inflammatory chemokines. (d) 4–8 h: Hapten-activated T cells release increasing amounts of inflammatory mediators, amplifying further cellular infiltration. (e) 12–48 h: The inflammatory reaction reaching its maximum, characterized by (epi)dermal infiltrates, oedema, and spongiosis. (f) 48–120 h: Gradually, downregulatory mechanisms take over, leading to decreased inflammation and disappearance of the cellular infiltrate. Finally, primordial conditions are reconstituted except for a few residual hapten-specific T cells causing the local skin memory. KC keratinocyte, DC dendritic cell
Irritant Properties of Allergens. Within a few hours after allergenic skin contact, immunohistopathological changes can be observed, including vasodilatation, upregulation of endothelial adhesion molecules (Goebeler et al. 1993, 1995), mast-cell degranulation (Walsh et al. 1990; Waldorf et al. 1991), keratinocyte cytokine and chemokine production (Spiekstra 2005; Meller 2007), influx of leucocytes (Bangert et al. 2003; Houck et al. 2004), and LC migration toward the dermis (Rambukhana et al. 1995; Silberberg-Sinakin et al. 1976; Hill et al. 1990; Toebak 2009). These pro-inflammatory phenomena, which are also observed in non-sensitized individuals (Sterry et al. 1991) and in T-cell-deficient nude mice (Herzog et al. 1989), strongly contribute to allergenicity (Saint-Mezard et al. 2003). Clearly most, if not all, of these effects can also be caused by irritants and, therefore, do not unambiguously discriminate between irritants and contact allergens (Willis et al. 1986; Hoefakker et al. 1995; Brasch et al. 1992; Spiekstra 2005). Apparently, true differences between these types of compounds depend on whether or not allergen-specific T cells become involved. Thus, only after specific T-cell triggering distinctive features might be observed, e.g., local release of certain chemokines, such as the Th1 associated chemokines CXCL9, CXCL10 (IP-10), and CXCL11 (I-TAC/IP-9) ([206], Meller 2007) or the Th2-related chemokines CCL11, CCL17, and CCL22 (Flier et al. 1999; Meller 2007). Certainly, pro-inflammatory effects of contact allergens increase, in many ways, the chance of allergen-specific T cells meeting their targets. The first cells affected by skin contact, i.e., keratinocytes and LC, are thought to represent major sources of pivotal mediators such as IL-1β and TNF-α (Enk 1992; Kondo and Sauder 1995). First, as described in “Hapten-Induced Activation of Allergen-Presenting Cells,” these cytokines cause hapten-bearing LC to mature and migrate toward the dermis (Kimber et al. 1998; Steinman et al. 1995; Toebak 2009). But, these cytokines also cause (over)expression of adhesion molecules on dermal postcapillary endothelial cells and loosen intercellular junctions. Thereby, extravasation of leucocytes, including allergen-specific T cells, is strongly promoted (Wardorf et al. 1991; Pober et al. 1986; Shimizu et al. 1991; Ward 2009). Moreover, haptens can stimulate nitric oxide (NO) production of the inducible NO-synthase (iNOS) of LC and keratinocytes, which contributes to local edema, vasodilatation, and cell extravasation (Ross et al. 1998; Rowe et al. 1997; Virag et al. 2002). In addition to these pathomechanisms that contribute to inflammatory responses, toll-like receptors (TLR) can be involved in triggering sensitization. Recent studies have indicated that at least the contact allergen nickel can specifically trigger human TLR-4, which adds to the release of unspecific danger signals (Rothenberg 2010; Schmidt et al. 2010).
Histopathological analyses support the view that the major causative events take place in the papillary dermis, close to the site of entry of allergen-specific T cells, for instance at hair follicles, where haptens easily penetrate and blood capillaries are nearby (Szepietowski et al. 1997). Here, perivascular mononuclear cell infiltrates develop, giving the highest chance of encounters between allergen-presenting cells and specific T cells. Once triggered, extravasated T cells will readily enter the lower epidermal layers, in which haptenized keratinocytes produce lymphocyte-attracting chemokines, like CXCL9/10, CCL17, CCL20, and CCL27 (Flier et al. 1999; Larsen 2009; Meller 2007; Woodland 2009, Table 1). Subsequently, since effector memory T cells can also be triggered by “non-professional” APC, including KC, fibroblasts, and infiltrating mononuclear cells, ACD reactivity is amplified in the epidermis (Hommel 2004; Viola and Lanzavecchia 1996). Together, these events result in the characteristic epidermal damage seen in ACD, such as spongiosis and hyperplasia. Notably, in ongoing ACD reactions, the production of chemokines attracting lymphocytes and monocytes/macrophages, in addition to the production of cytokines, adds to the nonspecific recruitment and activation of leucocytes (Yu et al. 1994; Buchanan and Murphy 1997). Thus, like the very early events in the effector phase reaction, the final response to a contact allergen is antigen-non-specific. It is therefore not surprising that allergic and irritant reactions are histologically alike.
Early Phase Reactivity. In the elicitation phase allergen-specific T cells are triggered by MHC-bound allergen, just like in the afferent phase. The role of Langerhans cells in allergen presentation upon elicitation is, however, less prominent, and also other cells, like mast cells, macrophages and keratinocytes may now contribute, since effector T cells are easily triggered and do not require professional antigen presentation. The role of keratinocytes in the onset of the ACD reaction is important, because of the cytokines and chemokines they produce upon hapten application (Meller 2007; Spiekstra 2005), thereby facilitating the influx of effector T cells. In addition, a variety of other cells and mediators may contribute to the initiation of the ACD reaction, as summarized below.
The role of neutrophils in the onset of ACD reactions has not been well established, though recent studies in mice demonstrate that skin reactivity to haptens largely depends on CXCL1, released from endothelial cells when the first hapten-specific CD8 T cells encounter the allergen and produce IL-17. CXCL1 may then attract neutrophils to the elicitation site, thus facilitating further influx of allergen-specific T cells (Kish 2009). In the human system, neutrophil infiltration was also observed in skin biopsies from nickel patch tests, presumably as a result of IL-17/IL-22-mediated inflammation (Larsen 2009). Moreover, it has been shown that IL-8/CXCL8, a potent neutrophil chemoattractant, is readily produced by human antigen presenting cells upon hapten exposure (Toebak et al. 2006); this could also contribute to an early influx of neutrophils in ACD reactions.
The role of an antibody-mediated early phase reaction in the development of ACD is still unclear in man, although Askenase and his colleagues have generated robust data to support this view in murine models (Askenase 2000): Hapten-specific IgM, produced upon sensitization by distant hapten-activated B-1 cells, can bind antigen early after challenge and activate complement. The resulting C5a causes the release of serotonin and TNF-α from local mast cells and platelets, leading to vascular dilatation and permeabilization, detectable as an early ear swelling peaking at 2 h (Van Loweren et al. 1983). Furthermore, C5a and TNF-α induce the upregulation of adhesion molecules on local endothelial cells (Foreman et al. 1994; Groves et al. 1995), thereby contributing to the recruitment of T cells in hapten challenge sites (Groves et al. 1995; Tsuji et al. 1997). In addition, human T cells were found to express the C5a receptor and are chemoattracted to endothelium-bound C5a (Nataf et al. 1999). However, against most contact allergens, including nickel, no antibodies have been detected in man, arguing against humoral mechanisms playing more than a minor role in clinical ACD (Wilkinson et al. 1994; Shirakawa et al. 1992). Interestingly in mice, immunoglobulin light chains, which have long been considered as the meaningless remnants of a spillover in the regular immunoglobulin production of B cells, were discovered to mediate very early hypersensitivity reactions by mast cell activation (Redegeld and Nijkamp 2003; vd Heyden 2006).
In addition to an auxiliary role of B cells and antibodies, natural killer (NK) cells have been reported to play a role in the onset of ACD reactions. Mice lacking both T- and B-cells (RAG2−/−) could still be sensitized to contact allergens and Thy1+ NK cells were identified here as effector cells with a prominent role for the activating NK receptor NKG2D (O’Leary 2006). Interestingly, another NK-like cell, the invariant NKT cell, that recognizes CD1d bound glycolipids resulting in rapid IL-4 and IFN-γ release, was also found to play a role in the elicitation of contact sensitivity in mice: Blocking of CD1d prevented both sensitization and elicitation by contact allergens (Nieuwenhuis 2005). Notably, in human ACD reactions relatively high frequencies of invariant NKT cells have been observed, ranging from 1.7% to 33% of total infiltrating T cells, which is 10–100 fold higher than the frequency found in the circulation (Gober 2008). Also other T cells with relatively restricted TCR repertoire such as Tγδ cells have been reported to contribute in a non-antigen-specific, probably non-MHC-restricted manner, to (early) elicitation responses (Dieli et al. 1998).
To conclude, using various mouse models, different types of early allergen specific reactivity have been claimed to play initiating roles in ACD, but clinical evidence for such mechanisms is still lacking.
T-Cell Patrol and Specificity of T-Cell Infiltrates. Whereas early nonspecific skin reactivity to contact allergens is pivotal for both sensitization and elicitation, full-scale development of ACD, of course, depends on allergen-specific T cells within the (epi)dermal infiltrates. In healthy skin there is a constant flow of memory T cells ending up in the draining lymph nodes: about 200 T cells/h/cm 2 skin (Brand et al. 1999, Fig. 6). Since one single antigen-specific T cell can already trigger visible skin inflammation (Milon et al. 1981; Marchal et al. 1982), randomly skin-patrolling memory/effector T cells might account for the initiation of the allergen-specific effector phase. However, since frequencies of hapten-specific T cells in sensitized individuals may still remain below 1 in 10,000, this does not seem to be a realistic scenario. Thus, augmented random and/or specific T-cell infiltration accompanies the development of ACD. Apparently, local chemokine release upon allergen contact is pivotal in this respect (see T-Cell recirculation; Meller 2007). Chemokine gene expression evaluated 48 h after NiSO4 application was increased for both Th1-related cytokines (CXCL9, CXCL10, and CXCL11) and Th2-related cytokines (CCL11, CCL17, and CCL22). CCL27, on the other hand, which attracts preferentially CCR10 bearing Th17/22 cells is constitutively produced in resting skin, but is rapidly released upon allergen contact to accumulate in the draining lymph nodes (Huang 2008).
The question concerning the specificity of ACD T-cell infiltrates has so far received little attention. In a guinea pig model, preferential entry of dinitrochlorobenzene (DNCB)-specific T cells was observed within 18 h after elicitation of skin tests with DNCB, as compared to non-related compounds (Scheper et al. 1985). Probably, extravasation of hapten-specific T cells benefits from T cell receptor-mediated interactions with endothelial MHC molecules, presenting hapten penetrated from the skin (Ward 2009). Within minutes after epicutaneous application, hapten can indeed be found in dermal tissues and on endothelial cells (Goebeler et al. 1993; Macatonia et al. 1987; Lappin et al. 1996). Indeed, the frequency of allergen-specific cells in positive patch tests to urushiol was found to be 10- to 100-fold higher than in the blood (Kalish 1990). Interestingly, whereas preferential entry may already contribute to relatively high frequencies of allergen-specific T cells (within 48 h up to 10%) (Probst et al. 1995; Milon et al. 1981), at later stages, when the ACD reaction fades away, the local frequency of allergen-specific T cells may increase even further, due to allergen-induced proliferation and rescue from apoptosis. Thus, at former skin reaction sites these cells can generate “local skin memory” (see Sect. 8).
Effector T-Cell Phenotypes. The debate on phenotypes of effector T cells in ACD is still ongoing and the number of T cell subsets potentially involved is growing every year (Fig. 7). Consensus exists, however, on the phenotype of the skin homing T cell, i.e., CLA postive. This molecule enables binding to cutaneous endothelial cells via E-selectin (CD62E) and thus migration into the dermis.
Since cutaneous infiltrates show a clear preponderance of CD4+ T cells, it is not surprising that these cells have most often been held responsible for mediating ACD. In nickel allergic individuals, indeed, allergen-responding cells were found to be CD4+CLA+ memory T cells (Moed 2004a, 2004b). Other studies, however, revealed CD8+CLA+ nickel reactive T cells as most discriminating for allergic individuals, since CD4+ nickel-reactive T cells were also found in healthy controls (Cavani et al. 1998). While the effector mechanism of CD4+ T cells is mainly based on cytokine production, CD8+ T cells may mediate skin inflammation also through killing of hapten-bearing target cells. In mice, generally CD8+ T cells are found to cause contact sensitivity reactions, certainly to strong allergens, like DNFB (Kish 2009; Gober 2008). In mice CD4+ T cells are rather found to be regulatory, as shown by the fact that contact sensitization to weak allergens succeeded only after depletion of the CD4+ T cells (Vocanson 2006). Of note, most model allergens studied in mice are hydrophobic molecules like DNFB and oxazolone, whereas in human studies very often water soluble metal salts, such as NiSO 4 are used as model allergen. This could, at least partly explain the different T-cell subsets involved (Figs. 2 and 5 MHCI/II presentation). So, taken together, it has become clear that both CD4+ and CD8+ T cells can act as effector cells in DTH and ACD reactions. Likewise, neither of these subsets can be regarded simply as regulatory or suppressor cells, although both of these subsets may, depending on the allergen models and read-out assays, play such roles (Kimber et al. 2002; Abe et al. 1996).
An essentially similar conclusion holds true for T-cell subsets (whether CD4+ or CD8+), releasing type-1, type-2, or type-17 cytokines or combinations thereof. Whereas type-1 cytokines, in particular IFN-γ, display well-established pro-inflammatory effects by increasing MHC and ICAM-1 expression (Saulnier 1995; Kish 2009) thereby contributing to improved allergen presentation and infiltration, IL-4, a hallmark type-2 cytokine, can cause erythema and induration, when released in the skin (Rowe and Bunker 1998; Asherson et al. 1996). Indeed, blockage of IL-4 can interfere with ACD (Asherson et al. 1996). IL-17 plays a role in recruitment and activation of neutrophils. It was shown to be produced both by CD8+ T cells (in mouse models with DNFB; Kish 2009) and by CD4+ T cells (in human nickel patch tests; Larsen 2009). The latter study shows, interestingly, that within a few hours after challenge CCL20 expression is upregulated in the skin, attracting CCR6 positive cells. Since all Th17 cells do express this receptor, an early preferential influx of Th17, and as a consequence IL-17 and IL-22 production, could be an essential early event in the development of the ACD reaction.
Thus, a picture emerges in which ACD reactions can be caused both by allergen-specific type-1, type-2, and type-17 T cells (Cavani et al. 1998; Rustemeyer et al. 2004; Moed 2004; Larsen 2009; Gober 2008; Oboki 2008). In retrospect, the down regulatory effects of IL-4 on ACD reactions observed earlier in some mouse models (Asada et al. 1997) might be ascribed to accelerated allergen-clearance rather than to blunt suppression. Still, both with time and repeated allergen-pressure, type-2 responsiveness may rapidly take over (Kitagaki et al. 1995; Kitagaki 1997). Allergen-specific T cells isolated from skin test sites of sensitized individuals, as compared to blood, showed a strong bias toward type-2 cytokine profiles (Werfel et al. 1997). Additional local IFN-γ release seems, however, indispensable, since for a broad panel of contact allergens, clinical ACD reactions were characterized by increased expression of mRNA encoding IFN-γ-inducible chemokines (Flier et al. 1999). In addition, transgenic mice expressing IFN-γ in the epidermis showed strongly increased ACD reactivity (Carroll et al. 1997).
Downregulatory Processes. Resolution of ACD reactions and risk factors for the development of chronicity are not yet fully understood. Of course, if the allergen source is limited, as with skin testing, local concentrations of allergen usually rapidly decrease, thus taking away the critical trigger of the ACD reaction cascade. Since even ACD reactions due to chronic exposure to allergen seldom result in permanent tissue destruction and scarification, immunoregulatory factors most likely contribute to prevention of excessive cytotoxicity and fatal destruction of the basal membrane. Both IL-1 and heparinase, secreted from activated keratinocytes and T cells, protect keratinocytes from TNF-α-induced apoptosis (Lider et al. 1995; Kothny-Wilkes et al. 1998). Moreover, activated effector T cells can undergo activation-induced cell death (AICD) during the resolution phase (Orteu et al. 1998). Notably, pro-inflammatory type-1 T cells, expressing high levels of Fas-ligand (CD95L) and low amounts of apoptosis-protecting FAP-1 protein, are more susceptible to AICD than type-2 cells (Zhang et al. 1997). This may partly explain the shift toward type-2 reactivity that is observed upon prolonged allergen exposure (Kitagaki et al. 1995). Moreover, during the late phase of ACD, keratinocytes, infiltrated macrophages and T cells start producing IL-10 (Enk 1992; Schwarz et al. 1994; Berg et al. 1995), which has many anti-inflammatory activities, including suppression of antigen-presenting cell and macrophage functions ([110], Lalani et al. 1997; Morel and Oriss 1998). In addition, the release of factors, such as PGE2 and TGF-β, derived from activated keratinocytes and infiltrated leucocytes, e.g., type-3 T cells, contribute to dampening of the immune response (Epstein et al. 1991; Lawrence et al. 1997). Release of PGE2, on the one hand, inhibits production of pro-inflammatory cytokines (Kalinski et al. 1997; Walker et al. 1983) and, on the other hand, activates basophils (Weston and Peachell 1998). These may constitute up to 5–15% of infiltrating cells in late phase ACD reactions (Dvorak et al. 1976) and are also believed to contribute to down regulation of the inflammatory response (Marone et al. 1994; Lundeberg et al. 1999). TGF-β silences activated T cells and inhibits further infiltration by down regulating the expression of adhesion molecules on both endothelial and skin cells (Sallusto et al. 2004). Regulatory cells producing these suppressive mediators might even predominate in skin sites, frequently exposed to the same allergen, and known to show local (allergen-specific) hypo-responsiveness [283]. It is of interest in this context that CD4+ memory T cells expanded from late DTH reactions could be educated to become CD4+CD25++ regulatory T cells expressing Foxp3 (Vukmanovic 2008).
9 Flare-Up and Retest Reactivity
Local allergen-retention. Flare-up reactivity of former ACD and patch test reaction sites is sometimes observed (Jensen et al. 2003; Hindsen et al. 2001; Larsson et al. 1997). From the basic mechanisms of ACD, it can be inferred that allergen-specific flare-up reactions depend either on local allergen or T-cell retention at these skin sites. Upon short-lasting, low-dose contacts, e.g., by skin testing, local allergen retention usually does not exceed a 2-week period, which is actually long enough to exceed the time required for active sensitization. In experimental guinea-pig studies we observed that skin tests with DNCB, chromium or penicillin could become positive even if primary sensitization was postponed to 1 week after skin testing. Apparently effector T cells released into the circulation at that late time still detected sufficient residual allergen at the former skin test sites to cause flare-up reactivity (Scheper et al. unpublished results). Maximum allergen-persistence for around 14 days was also reported by Saint-Mezard et al. (2003), using the hapten fluorescein-isothiocyanate in a mouse model for flare-up reactivity. Also in humans flare-up reactions due to locally persisting allergen can be observed, when from about 4–6 days after primary sensitization, peripheral effector T-cell frequency increases (Skog 1976). Clinically, this phenomenon can explain anomalous results from patch testing with multiple contact allergens. When a patient suspected for penicillin allergy was patch tested with cross-reactive penicillin derivatives, a regular 24–72 h reaction was only observed to one of the penicillins, but all others also became positive from about 8–9 days after skin testing. The first penicillin derivative turned out to release formaldehyde to which the patient was found to be allergic. Positive reactivity to formaldehyde apparently had potentiated primary sensitization to penicillin, causing the other, previously negative reaction sites to flare up (Neering, personal communication). Thus, skin test sites may occasionally flare-up if the testing dose itself led to the release or activation of sufficiently high numbers of effector T cells in the circulation.
Local T-cell retention. In contrast, allergen-specific T cells may persist for at least several months in the skin causing “local skin memory”(Fig. 8) (Scheper et al. 1983; Moed 2004). Thus, locally increased allergen-specific hyperreactivity, detectable through either accelerated “retest” reactivity (after repeated allergenic contact at the same skin site) or flare-up reactivity (after allergen entry from the circulation, e.g., derived from food ingestion), may be observed for long periods of time at former skin reaction sites (Christensen et al. 1985; Hindsen and Christensen 1992). Typically, the erythematous reactions peak between 2 and 6 h after contact with the allergen. Histological examination of such previously positive skin reaction sites shows that the majority of remaining T cells is CD4+ CCR10+ (Moed 2004). The remarkable flare-up reactivity at such sites can be understood by considering that just one specific effector T cell can be sufficient to generate macroscopic reactivity (Marchal et al. 1982). Moreover, a very high frequency of the residual T cells may be specific for the allergen, as discussed above in “The Effector Phase of Allergic Contact Dermatitis.” Apparently, local specific T-cell retention is highly advantageous in combating microbial infections, since memory T cells localized in peripheral tissues contribute to robust protection e.g., to viral infections (Woodland 2009). Only in highly sensitized individuals unrelated skin test sites may also show flare-up reactions (Scheper et al. 1983) and even generalized erythematous macular eruptions can be observed with higher allergen doses (Polak 1968). The latter reactivities probably relate to the fact that recently activated T cells show strong expression of adhesion and homing molecules, e.g., CLA and chemokine receptors, such as CCR5, facilitating random migration into peripheral tissues and thus allergen-specific T-cell patrol in the skin (Moser et al. 1998; Woodland 2009). Upon subsequent allergen entry from the circulation, these allergen-specific T cells could mediate generalized erythematous reactions (Hindsen et al. 2001).
Local skin memory. In former allergic contact dermatitis sites a few hapten-specific T cells can remain, mainly close to dermal dendritic cells (DC). Retest reaction: renewed hapten contact can induce a rapid onset of an erythematous reaction, sparked off by the residual hapten-specific T cells. KC keratinocyte, LC Langerhans cell
Interestingly, local allergen-specific T-cell retention/“local skin memory” can be clinically exploited to discriminate between simultaneous sensitization to different sensitizers (“concomitant sensitization”) and cross-reactivity between different sensitizers (Rustemeyer et al. 2002; Matura 1998; Inerot and Moller 2000). Using several different combinations of contact allergens in a guinea pig model, we retested guinea pigs previously sensitized to DNCB and methyl methacrylate (MMA), with the same allergens and some other methacrylate congeners. Accelerated retest reactivities were observed with the latter congeners on the former MMA, but not DNCB, patch test sites (Rustemeyer et al. 2002). Thus, with preferential local retention of MMA-specific T cells at the MMA skin test site, no accelerated retest reactivity could be elicited with DNCB, but to varying degrees with all four MMA-related compounds. In clinical practice using this approach, Matura et al. (1998) confirmed positive cross-retest reactions for cloprednol and tixocortol pivalate, both belonging to group A, and budesonide, amcinonide, and triamcinolone, all belonging to group B corticosteroids (see also Isaksson and Bruze 2003).
10 Hyporeactivity: Tolerance and Desensitization
Of course, uncontrolled development and expression of T-cell-mediated immune function would be detrimental to the host. During evolution, several mechanisms developed to curtail lymph node hyperplasia or to prevent excessive skin damage upon persisting antigen exposure.
Regulation of Immune Responses. First, allergen contacts, e.g., by oral or intravenous administration, may lead to large-scale presentation of allergen by cells other than skin DC (Fig. 9). In the absence of appropriate costimulatory signals (as described above in “Recognition of Allergen-Modified Langerhans’ Cells”) allergen presented by e.g., immature Langerhans’ cells may anergize naive T cells, i.e., cause receptor-downregulation associated with an unresponsive state, eventually leading to their death by apoptosis (Fig. 10) (Zinkernagel 2004; Piccirillo and Thornton 2004; Benson and Whitacre 1997). With increasing densities of MHC-antigen complexes on the surface of professional APC, at least three different levels of T-cell tolerance may be induced, characterized by active suppression, anergy or deletion (Ferber et al. 1994; Morgan et al. 1999). Unresponsiveness of T cells, induced by allergenic contacts at skin sites where LC/DC functions have been damaged, e.g., by UV irradiation, or are naturally absent, e.g., in the tail skin of mice, may be ascribed to T-cell anergy, frequently associated with TCR/CD4 or CD8 down regulation, and apoptosis/deletion (Shreedhar et al. 1998; Semma and Sagami 1981). Whereas anergy and deletion reflect “passive” unresponsiveness, tolerance by active suppression may also be induced under similar circumstances (Taams et al. 1999). Actually, with increasing dose and exposure times, even regular epicutaneous allergenic contacts not only induce T effector cells but also lymphocytes controlling T-cell proliferation (afferently acting regulatory cells) and/or causing decreased skin reactivity (regulatory cells of effector phase). Thus, allergic contact hypersensitivity is the resultant of a delicate balance between effector and regulatory mechanisms (Boerrigter and Scheper 1987; Girolomoni et al. 2004).
The character of the APC–T-cell interaction determines the immunological outcome. Sensitization: Naïve T cells, activated by antigen-presenting cells (APC) providing both hapten-specific (“signal 1”) and appropriate costimulatory (“signal 2”) signals, develop into effector T cells, characterized by Th-17/22, -1, and -2 cytokine secretion profiles. Tolerance: In the absence of appropriate costimulatory signals, immunological tolerance may develop. With increasing density of MHC-hapten complexes on the surface of APC, activating “signal 1” T-cell pathways, multiple levels of T-cell tolerance might be induced
Cellular Basis of Active Tolerance. Upon preferential stimulation of regulatory cells, e.g., by feeding non-primed, naïve individuals with contact allergens, strong and stable allergen-specific, active tolerance may develop (Mayer et al. 2001; Weiner 1997; Wang 2008). The concept of active regulatory (“suppressor”) cells controlling ACD is based on the fact that, in experimental animal models, such allergen-specific tolerance can be transferred by lymphoid cells from tolerant to naive animals (Dieli et al. 1998; Rustemeyer et al. 2001). Active suppression, as revealed by these adoptive cell transfers, is a critical event in regulating T-cell responses to contact sensitizers, and to all possible peptide/protein antigens, including bacterial, autoimmune, and graft rejection antigens (Miller et al. 1977; Polak 1980; Weiner 1997).
Like effector T cells in ACD, regulatory cells are not a single subpopulation of cells. As outlined above, depending on e.g., the nature of the allergen and route of exposure, ACD can be mediated by both CD4+ and CD8+ T cells, either or both releasing Th1, Th2, Th3, Th17/22 cytokines. With distinct effector phenotypes for particular allergens, each of the other phenotypes can act as regulatory cells (Weigle and Romball 1997; Arnaboldi 2009). Notwithstanding, type-2 cytokine producing cells are prominent in regulating ACD, with allergic contact hypersensitivity enhanced and tolerance reversed by interfering with type-2 T-cell functions (Zembala and Ashershon 1973; Boerrigter and Scheper 1984; Mokyr et al. 1998). Also, interferons and IL-12, both impairing Th2 and Th17/22 cells, were shown to inhibit regulatory cells and to stimulate effector-cell functions in mouse models (Knop et al. 1982; Zhang and Michael 1990; Claessen et al. 1996). In particular after mucosal allergen contact stimulation, T cells producing IL-10 and/or TGF-β (type-3 cytokine profile), many of which co-expressing CD4, CD25 and the transcription factor Foxp3 (Treg), may act as regulatory cells (Bridoux et al. 1997; Cavani et al. 2003; Feuerer 2009). These T cells promote anti-inflammatory immunity, e.g., by switching antibody production to IgA, which mediates secretory immunity and thus contributes to antigen exclusion in the lumen, e.g., of the gastro-intestinal tract (Hafler et al. 1997). Of note, TGF-β strongly suppresses development of both type-1 and -2 effector T cells, and can silence T cells in a semi-naïve state (O’Garra 1998).
Regulatory Mechanisms of the Effector Phase. A critical feature of the regulatory principles involving mutual regulation of T-cell subpopulations by Th1, Th2, Th3 and Th17/22 cytokines, is that regulatory functions are most effective during initiation of immune responses (Fig. 5). Thus, once established, effector T cell and cytokine profiles show remarkable stability and refractoriness to regulatory forces. Down-regulation of allergic skin reactions may, therefore, take considerable time. Of course, the preliminary factor facilitating decreased allergic skin reactivity is removal of hapten by exudate and innate immune cells of the inflammatory infiltrate. But, at chronically exposed sites also specific regulatory mechanisms can be involved, such as CD8+ T cells, acting either as regulator/suppressor (CD28−CD11b+) or cytotoxic (CD28+CD11b−) T cells (Lonati et al. 1998; De panfilis 1998), which may downregulate skin reactivity by targeting allergen-presenting DC [331]. Multiplicity and reduncancy of regulatory mechanisms have thus far hampered development of robust clinical treatments exploiting regulatory T-cell functions to provide for allergen-specific downregulation of the effector phase of ACD. The development of potential therapeutic applications of regulatory cells in various disorders, such as allergic contact dermatitis and autoimmune diseases, therefore, needs much more time than envisioned earlier (Ilan 2009).
Redundancy of Tolerance Mechanisms. Besides regulatory T cells, producing different cytokines, or exerting distinct cytotoxicities, other mechanisms may also contribute to immune regulation and tolerance. Clearly, the risk of excessive immune reactivity should be very low. These mechanisms involve allergen-specific T cells shedding truncated T-cell receptors, acting as antagonists and blocking allergen presentation (Kuchroo 1998), and high-dose allergen-induced anergic T cells (Morgan et al. 1999). Possibly, the latter cells, by actively suppressing DC functions, can function as “active” suppressor cells (Taams et al. 1998, 2000). Interestingly, DC, becoming suppressive by this mechanism (Taams et al. 1998) or by suppressive cytokines like IL-10 and PGE 2 (Kumar and Sercarz 1998; Kalinski et al. 1998), can, in turn, act themselves as suppressor cells by conferring antigen-specific anergy to subsequently encountered T cells (Steinbrink et al. 1999; Taams et al. 2000). Although, at present, consensus has been reached about a critical role of regulatory/suppressor cells in the development and expression of ACD, the relative contributions of each of the various mechanisms are still far from clear.
Induction of Lasting Tolerance Only in Naive Individuals. Both clinical and experimental findings indicate that full and persistent tolerance can only be induced prior to any sensitizing allergen contacts (Van Hoogstraten et al. 1989; Strobel and Mowat 1998; Wang 2008). Upon primary allergenic contacts, naive T cells differentiate to produce polarized cytokine profiles (Figs. 5 and 9). Once polarized, however, T-cell profiles are irreversible, due to loss of cytokine (receptor) genes, or at least very stable, due to the mutually suppressive activities of T-cell cytokines. An important corollary of the latter concept of active suppression is the bystander effect, in which the response to any antigen can be downregulated by immunosuppressive cytokines acting in a local tolerogenic microenvironment (von Herrath 1997). The latter was observed for both protein antigens (Fowler and Weiner 1997) and methacrylate contact allergens (Rustemeyer et al. 2001). Stable polarization/skewing may also explain why even low, non-sensitizing doses of nickel applied to the skin prevented subsequent tolerance induction by feeding the metal allergen (Van Hoogstraten et al. 1994). Apparently, the progeny of naïve allergen-specific cells, once “on the stage,” have been triggered to a “subclinical” degree toward effector cell differentiation and become refractory to regulatory cell action (Fig. 10). This may also have contributed to incomplete tolerance induction in earlier clinical studies when feeding with poison ivy-/oak-derived allergens (Epstein 1987). Indeed, to our knowledge, permanent reversal of existing ACD in healthy individuals has, as yet, never been achieved. Nevertheless, as described above, effector cells still seem susceptible, though transiently, to the downregulation of allergen reactivity, as was observed in desensitization procedures (Van Hoogstraten et al. 1994; Morris 1998).
Transient Desensitization in Primed Individuals. For dermatologists, methods by which patients might be desensitized for existing ACD would be a welcome addition to the currently prevailing symptomatic therapies, and investigators have made a wide variety of attempts to achieve this goal. Unfortunately, as mentioned above, therapeutic protocols involving ingestion of poison ivy allergen, penicillin, or nickel sulfate were of only transient benefit to the patients (Epstein 1987; Wendel et al. 1985; Panzani et al. 1995; Tammaro et al. 2009). Similarly in animal models, only a limited and transient degree of hyposensitization was obtained by Chase (1946) when feeding DNCB-contact-sensitized guinea pigs with the allergen, whereas, for achieving persistent chromium-unresponsiveness in presensitized animals, Polak and Turk (1968a, b) needed a rigorous protocol involving up to lethal doses of the allergen. As outlined above, mechanisms underlying specific desensitization in ACD probably depend on direct interference of allergen with effector T-cell function, by blocking or downregulating T-cell receptors, leading to anergy and apoptosis (Polak and Rinck 1978). As the onset of desensitization is immediate, no suppressor mechanisms may initially be involved. Apparently in the absence of LC, MHC class II-positive keratinocytes can serve as APC and are very effective in rendering allergen-specific effector cells anergic (Gaspari et al. 1988). Moreover, at later stages active suppression may come into play resulting from secondary inactivation of DC function by anergized T cells (Shreedhar et al. 1998). Nevertheless, major problems with in vivo desensitization procedures relate to the refractoriness of effector T cells to regulatory cell functions, and the rapid replacement of anergized effector cells by naïve T cells from relatively protected peripheral lymphoid tissues provides a source of new effector cells upon sensitizing allergen contacts. The same conclusions can be drawn from attempts to achieve local desensitization. It was found that local desensitization by repeatedly applying allergen at the same skin site did not result from local skin hardening or LC inactivation, as local reactivity to an unrelated allergen at the site was unimpaired (Boerrigter and Scheper 1987). Persistence of cellular infiltrates, in the absence of erythematous reactivity, at a desensitized skin site could reflect local anergy, but also locally active regulatory cells. Upon discontinuation of allergen exposure, however, local unresponsiveness was rapidly (within 1 week) lost. Collectively, this data illustrates the problems encountered in attempting to eradicate established effector-T-cell function, not only in ACD but also in autoimmune diseases (Weiner 1997; Wang 2008).
11 Summary and Conclusions
Extensive research has led to a better understanding of the mechanisms of ICD and ACD. The primary role of innate immune cells in coping with exogenous potential harmful threats is rapidly being uncovered. Also, the basic immunology of ACD is now well-defined, including T-cell migratory patterns, recognition of distinct allergens, interactions with other inflammatory cells to generate inflammation, and cytokine profiles. But new complexities have emerged. For instance, in contrast to earlier belief, many of the currently known T-cell subpopulations can act either or both as effector and regulatory cells, depending on the nature of the allergen, the route of entry, frequency of exposure, and many other, still ill-defined factors. In particular, the poor understanding of regulatory mechanisms in ACD still hampers further therapeutic progress. So far, no methods of permanent desensitization have been devised.
Nevertheless, next to the established anti-inflammatory drugs, recently defined cellular interaction molecules and mediators provide promising targets for new generations of anti-inflammatory drugs, some of which have already entered clinical trials. Clearly, drugs found to be effective in preventing severe T-cell-mediated conditions, e.g., rejection of a vital organ graft, should be very safe before their use in ACD would seem appropriate. To date, prudence favors alternative measures to prevent ICD and ACD, be it through legal action to outlaw the use of certain materials or through avoiding personal contact with these materials. In the meantime, for difficult-to-avoid allergens, further studies on the potential value of tolerogenic treatments prior to possible sensitization seem warranted.
References
Abe M, Kondo T, Xu H, Fairchild RL (1996) Interferon-gamma inducible protein (IP-10) expression is mediated by CD8+ T cells and is regulated by CD4+ T cells during the elicitation of contact hypersensitivity. J Invest Dermatol 107:360–366
Abe N, Katamura K, Shintaku N, Fukui T, Kiyomasu T, Lio J, Ueno H, Tai G, Mayumi M, Furusho K (1997) Prostaglandin E2 and IL-4 provide naïve CD4+ T cells with distinct inhibitory signals for the priming of IFN-gamma production. Cell Immunol 181:86–92
Acuto O, Michel F (2003) CD28-mediated co-stimulation: a quantitative support for TCR signalling. Nat Rev Immunol 3:939–951
Adema GJ, Hartgers F, Verstraten R, de Vries E, Marland G, Menon S, Foster J, Xu Y, Nooyen P, McClanahan T, Bacon KB, Figdor CG (1997) A dendritic-cell-derived C–C chemokine that preferentially attracts naïve T cells. Nature 387:713–717
Aiba S, Tagami H (1999) Dendritic cell activation induced by various stimuli, e.g. exposure to microorganisms, their products, cytokines, and simple chemicals as well as adhesion to extracellular matrix. J Dermatol Sci 20:1–13
Allan SE, Broady R, Gregori S, Himmel ME, Locke N, Roncarolo MG, Bacchetta R, Levings MK (2008) CD4+ T-regulatory cells: toward therapy for human diseases. Immunol Rev 223:391–421
Altin JG, Sloan EK (1997) The role of CD45 and CD45-associated molecules in T cell activation. Immunol Cell Biol 75:430–445
Asada H, Linton J, Katz SI (1997) Cytokine gene expression during the elicitation phase of contact sensitivity – regulation by endogenous IL-4. J Invest Dermatol 108:406–411
Asherson GL, Dieli F, Sireci G, Salerno A (1996) Role of IL-4 in delayed type hypersensitivity. Clin Exp Immunol 103:1–4
Austrup F, Vestweber D, Borges E, Lohning M, Brauer R, Herz U, Renz H, Hallmann R, Scheffold A, Radbruch A, Hamann A (1997) P- and E selectin mediate recruitment of T-helper-1 but not T-helper-2 cells into inflamed tissues. Nature 385:81–83
Banchereau J, Steinman RM (1998) Dendritic cells and the control of immunity. Nature 392:245–252
Bangert C, Friedl J, Stary G, Stingl G, Kopp T (2003) Immunopathologic features of allergic contact dermatitis in humans: participation of plasmacytoid dendritic cells in the pathogenesis of the disease? J Invest Dermatol 121:1409–1418
Basso AS, Cheroutre H, Mucida D (2009) More stories on Th17 cells. Cell Res 19(4):399–411
Benson JM, Whitacre CC (1997) The role of clonal deletion and anergy in oral tolerance. Res Immunol 148:533–554
Berg DJ, Leach MW, Kuhn R, Rajewsky K, Muller W, Davidson NJ, Rennick D (1995) Interleukin 10 but not interleukin 4 is a natural suppressant of cutaneous inflammatory responses. J Exp Med 182:99–108
Bergstresser PR (1989) Sensitization and elicitation of inflammation in contact dermatitis. In: Norris DA (ed) Immune mechanisms in cutaneous disease. Dekker, New York, pp 219–246
Birbeck M (1961) An electron microscope study of basal melanocytes and high level clear cells (Langerhans cells) in vitiligo. J Invest Dermatol 37:51–56
Blauvelt A, Hwang ST, Udey MC (2003) Allergic and immunologic diseases of the skin. J Allergy Clin Immunol 111:S560–S570
Blömeke B, Brans R, Coenraads PJ, Dickel H, Bruckner T, Hein DW, Heesen M, Merk HF, Kawakubo Y (2009) Para-phenylenediamine and allergic sensitization: risk modification by N-acetyltransferase 1 and 2 genotypes. Br J Dermatol 161(5):1130–1135
Boerrigter GH, Scheper RJ (1984) Local administration of the cytostatic drug 4-hydroperoxy-cyclophosphamde (4-HPCY) facilitates cell mediated immune reactions. Clin Exp Immunol 58:161–166
Boerrigter GH, Scheper RJ (1987) Local and systemic desensitization induced by repeated epicutaneous hapten application. J Invest Dermatol 88:3–7
Boerrigter GH, de Groot J, Scheper RJ (1986) Intradermal administration of 4-hydoperoxy-cyclophosphamde during contact sensitization potentiates effector T cell responsiveness in draining lymph nodes. Immunopharmacology 11(1):13–20
Borges E, Tietz W, Steegmaier M, Moll T, Hallmann R, Hamann A, Vestweber D (1997) P-selectin glycoprotein ligand-1 (PSGL-1) on T helper 1 but not on T helper 2 cells binds to P-selectin and supports migration into inflamed skin. J Exp Med 185:573–578
Bos JD, Meinardi MMHM (2000) The 500 Dalton rule for the skin penetration of chemical compounds and drugs. Exp Dermatol 9:165–169
Braathen LR (1980) Studies on human epidermal Langerhans cells. III. Induction of T lymphocyte response to nickel sulphate in sensitized individuals. Br J Dermatol 103:517–526
Brand CU, Hunger RE, Yawalkar N, Gerber HA, Schaffner T, Braathen LR (1999) Characterization of human skin-derived CD1a-positive lymph cells. Arch Dermatol Res 291:65–72
Brasch J, Burgard J, Sterry W (1992) Common pathways in allergic and irritant contact dermatitis. J Invest Dermatol 98:166–170
Breathnach SM (1988) The Langerhans cell. Centenary review. Br J Dermatol 119:463–469
Bretscher PA, Ogunremi O, Menon JN (1997) Distinct immunological states in murine cutaneous leishmaniasis by immunizing with different amounts of antigen: the generation of beneficial, potentially harmful, harmful and potentially extremely harmful states. Behring Inst Mitt 98:153–159
Bridoux F, Badou A, Saoudi A, Bernard L, Druet E, Pasquier R, Druet P, Pelletier L (1997) Transforming growth factor beta (TGF-beta)-dependent inhibition of T helper cell 2 (Th2)-induced autoimmunity by self-major histocompatibility complex (MHC) class II-specific, regulatory CD4+ T cell lines. J Exp Med 185:1769–1775
Buchanan KL, Murphy JW (1997) Kinetics of cellular infiltration and cytokine production during the efferent phase of a delayed-type hypersensitivity reaction. Immunology 90:189–197
Budinger L, Hertl M (2000) Immunologic mechanisms in hypersensitivity reactions to metal ions: an overview. Allergy 55:108–115
Calder PC, Bevan SJ, Newsholme EA (1992) The inhibition of T-lymphocyte proliferation by fatty acids is via an eicosanoid-independent mechanism. Immunology 75:108–115
Cantrell D (1996) T cell receptor signal transduction pathways. Annu Rev Immunol 14:259–274
Carroll JM, Crompton T, Seery JP, Watt FM (1997) Transgenic mice expressing IFN-gamma in the epidermis have eczema, hair hypopigmentation, and hair loss. J Invest Dermatol 108:412–422
Caux C, Ait-Yahia S, Chemin K, de Bouteiller O, Dieu-Nosjean MC, Homey B, Massacrier C, Vanbervliet B, Zlotnik A, Vicari A (2000) Dendritic cell biology and regulation of dendritic cell trafficking by chemokines. Springer Semin Immunopathol 22:345–369
Cavani A, Mei D, Guerra E, Corinti S, Giani M, Pirrotta L, Puddu P, Girolomoni G (1998) Patients with allergic contact dermatitis to nickel and nonallergic individuals display different nickel-specific T cell responses. Evidence for the presence of effector CD8+ and regulatory CD4+ T cells. J Invest Dermatol 111:621–628
Cavani A, Nasorri F, Ottaviani C, Sebastiani S, De Pita O, Girolomoni G (2003) Human CD25+ regulatory T cells maintain immune tolerance to nickel in healthy, nonallergic individuals. J Immunol 171:5760–5768
Cella M, Sallusto F, Lanzavecchia A (1997) Origin, maturation and antigen presenting function of dendritic cells. Curr Opin Immunol 9:10–16
Chase MW (1946) Inhibition of experimental drug allergy by prior feeding of the sensitizing agent. Proc Soc Exp Biol Med 61:257–259
Christensen OB, Beckstead JH, Daniels TE, Maibach HI (1985) Pathogenesis of orally induced flare-up reactions at old patch sites in nickel allergy. Acta Derm Venereol 65:298–304
Claessen AME, von Blomberg BME, de Groot J, Wolvers DAE, Kraal G, Scheper RJ (1996) Reversal of mucosal tolerance by subcutaneous administration of interleukin-12 at the site of attempted sensitization. Immunology 88:363–367
Constant SL, Bottomly K (1997) Induction of Th1 and Th2 CD4+ T cell responses: the alternate approaches. Annu Rev Immunol 15:297–322
Constant SL, Pfeiffer C, Woodard A, Pasqualini T, Bottomly K (1995) Extent of T cell receptor ligation can determine the functional differentiation of naïve CD4+ T cells. J Exp Med 5:1591–1596
Coulter EM, Jenkinson C, Farrell J, Lavergne SN, Pease C, White A, Aleksic M, Basketter D, Williams DP, King C, Pirmohamed M, Park BK, Naisbitt DJ (2010) Measurement of CD4+ and CD8+ T-lymphocyte cytokine secretion and gene expression changes in p-phenylenediamine allergic patients and tolerant individuals. J Invest Dermatol 130:161–174
Crivellato E, Vacca A, Ribatti D (2004) Setting the stage: an anatomist’s view of the immune system. Trends Immunol 25:210–217
Croft M, So T, Duan W, Soroosh P (2009) The significance of OX40 and OX40L to T-cell biology and immune disease. Immunol Rev 229(1):173–191
Cumberbatch M, Dearman RJ, Kimber I (1997) Interleukin 1-beta and the stimulation of Langerhans cell migration – comparisons with tumour necrosis factor alpha. Arch Dermatol Res 289:277–284
Cutolo M, Seriolo B, Villaggio B, Pizzorni C, Craviotto C, Sulli A (2002) Androgens and estrogens modulate the immune and inflammatory responses in rheumatoid arthritis. Ann N Y Acad Sci 966:131–142
Czernielewski SM, Demarchez M (1987) Further evidence for the self-reproducing capacity of Langerhans cells in human skin. J Invest Dermatol 88:17–20
Dabbagh K, Lewis DB (2003) Toll-like receptors and T-helper-1/T-helper-2 responses. Curr Opin Infect Dis 16:199–204
Davis SJ, van der Merwe PA (2003) TCR triggering: co-receptor-dependent or -independent? Trends Immunol 24:624–626
De Panfilis G (1998) CD8+ cytolytic T lymphocytes and the skin. Exp Dermatol 7:121–131
Demeure CE, Yang LP, Desjardins C, Raynauld P, Delespesse G (1997) Prostaglandin E-2 primes naïve T cells for the production of anti-inflammatory cytokines. Eur J Immunol 27:3526–3531
Dieli F, Ptak W, Sireci G, Romano GC, Potestio M, Salerno A, Asherson GL (1998) Cross-talk between V-beta-8(+) and gamma-delta(+) T lymphocytes in contact sensitivity. Immunology 93:469–477
Dieu M-C, Vanbervliet B, Vicari A, Bridon J-M, Oldham E, Ait-Yahia S, Brière F, Zlotnik A, Lebecque S, Caux C (1998) Selective recruitment of immature and mature dendritic cells by distinct chemokines expressed in different anatomic sites. J Exp Med 188:373–386
Divkovic M, Pease CK, Gerberick GF, Basketter DA (2005) Hapten-protein binding: from theory to practical application in the in vitro prediction of skin sensitization. Contact Dermatitis 53(4):189–200
Dong C (2008) TH17 cells in development: an updated view of their molecular identity and genetic programming. Nat Rev Immunol 8(5):337–348
Dong C, Nurieva RI, Prasad DV (2003) Immune regulation by novel costimulatory molecules. Immunol Res 28:39–48
Dvorak HF, Mihm MC Jr, Dvorak AM (1976) Morphology of delayed-type hypersensitivity reactions in man. J Invest Dermatol 64:391–401
Edele F, Esser PR, Lass C, Laszczyk MN, Oswald E, Strüh CM, Rensing-Ehl A, Martin SF (2007) Innate and adaptive immune responses in allergic contact dermatitis and autoimmune skin diseases. Inflamm Allergy Drug Targets 6(4):236–244
Eliasson E, Kenna JG (1996) Cytochrome P450 2E1 is a cell surface autoantigen in halothane hepatitis. Mol Pharmacol 50:573–582
Enk AH, Katz SI (1992a) Early molecular events in the induction phase of contact sensitivity. Proc Natl Acad Sci U S A 89:1398–1402
Enk AH, Katz SI (1992b) Identification and induction of keratinocyte-derived IL-10. J Immunol 149:92–95
Enk AH, Angeloni VL, Udey MC, Katz SI (1993) An essential role for Langerhans cell-derived IL-1b in the initiation of primary immune responses in skin. J Immunol 150:3698–3704
Epling GA, Wells JL, Yoon UC (1988) Photochemical transformations in salicylanilide photoallergy. Photochem Photobiol 47:167–171
Epstein WL (1987) The poison ivy picker of Pennypack Park: the continuing saga of poison ivy. J Invest Dermatol 88:7–9
Epstein SP, Baer RL, Thorbecke GJ, Belsito DV (1991) Immunosuppressive effects of transforming growth factor beta: inhibition of the induction of Ia antigen on Langerhans cells by cytokines and of the contact hypersensitivity response. J Invest Dermatol 96:832–837
Faria AM, Weiner HL (2006) Oral tolerance and TGF-beta-producing cells. Inflamm Allergy Drug Targets 5(3):179–190
Ferber I, Schönrich G, Schenkel J, Mellor AL, Hämmerling GJ, Arnold B (1994) Levels of peripheral T cell tolerance induced by different doses of tolerogen. Science 263:674–676
Flier J, Boorsma DM, Bruynzeel DP, van Beek PJ, Stoof TJ, Scheper RJ, Willemze R, Tensen CP (1999) The CXCR3 activating chemokines IP-10, MIG and IP-9 are expressed in allergic but not in irritant patch test reactions. J Invest Dermatol 113:574–578
Foreman KE, Vaporciyan AA, Bonish BK, Jones ML, Johnson KJ, Glovsky MM, Eddy SM, Ward PA (1994) C5a-induced expression of P-selectin in endothelial cells. J Clin Invest 94:1147–1155
Fowler E, Weiner HL (1997) Oral tolerance: elucidation of mechanisms and application to treatment of autoimmune diseases. Biopolymers 43:323–335
Fuhlbrigge RC, Kieffer JD, Armerding D, Kupper TS (1997) Cutaneous lymphocyte antigen is a specialized form of PSGL-1 expressed on skin-homing T cells. Nature 389:978–981
Furue M, Chang CH, Tamaki K (1996) Interleukin-1 but not tumor necrosis factor a synergistically upregulates the granulocyte-macrophage colony-stimulating factor-induced B7–1 expression of murine Langerhans cells. Br J Dermatol 135:194–198
Fyhrquist-Vanni N, Alenius H, Lauerma A (2007) Contact dermatitis. Dermatol Clin 25(4):613–623
Gajewski TF, Fitch FW (1988) Antiproliferative effect of IFN-gamma in immune regulation. I. IFN-gamma inhibits the proliferation of Th2 but not Th1 murine helper T lymphocyte clones. J Immunol 140:4245–4252
Gascoigne NR, Zal T (2004) Molecular interactions at the T cell-antigen-presenting cell interface. Curr Opin Immunol 16:114–119
Gaspari AA, Jenkins MK, Katz SI (1988) Class II MCH-bearing keratinocytes induce antigen-specific unresponsiveness in hapten-specific TH1 clones. J Immunol 141:2216–2220
Geenen V, Brilot F (2003) Role of the thymus in the development of tolerance and autoimmunity towards the neuroendocrine system. Ann N Y Acad Sci 992:186–195
Gell PDH, Coombs RRA, Lachman R (1975) Clinical aspects of immunology, 3rd edn. Blackwell, London
Gerberick F, Aleksic M, Basketter D, Casati S, Karlberg AT, Kern P, Kimber I, Lepoittevin JP, Natsch A, Ovigne JM, Rovida C, Sakaguchi H, Schultz T (2008) Chemical reactivity measurement and the predicitve identification of skin sensitisers. The report and recommendations of ECVAM Workshop 64. Altern Lab Anim 36(2):215–242
Girolomoni G, Gisondi P, Ottaviani C, Cavani A (2004) Immunoregulation of allergic contact dermatitis. J Dermatol 31:264–270
Goebeler M, Meinardus-Hager G, Roth J, Goerdt S, Sorg C (1993) Nickel chloride and cobalt chloride, two common contact sensitizers, directly induce expression of intercellular adhesion molecule-1 (ICAM-1), vascular cell adhesion molecule-1 (VCAM-1), and endothelial leukocyte adhesion molecule (ELAM-1) by endothelial cells. J Invest Dermatol 100:759–765
Goebeler M, Roth J, Brocker EB, Sorg C, Schulze-Osthoff K (1995) Activation of nuclear factor-kappa B and gene expression in human endothelial cells by the common haptens nickel and cobalt. J Immunol 155:2459–2467
Grewe M, Bruijnzeel-Koomen CA, Schöpf E, Thepen T, Langeveld-Wildschut AG, Ruzicka T, Krutmann J (1998) A role for Th1 and Th2 cells in the immunopathogenesis of atopic dermatitis. Immunol Today 19(8):359–361
Griem P, Wulferink M, Sachs B, Gonzales JB, Gleichmann E (1998) Allergic and autoimmune reactions to xenobiotics: how do they arise? Immunol Today 19:133–141
Groux H, Sornasse T, Cottrez F, de Vries JE, Coffman RL, Roncarolo MG, Yssel H (1997) Induction of human T helper cell type-1 differentiation results in loss of IFN-g receptor b-chain expression. J Immunol 158:5627–5631
Groves RW, Allen MH, Ross EL, Barker JN, MacDonald DM (1995) Tumor necrosis factor alpha is pro-inflammatory in normal human skin and modulates cutaneous adhesion molecule expression. Br J Dermatol 132:345–352
Gunn MD, Tangemann K, Tam C, Cyster JG, Rosen SD, Williams LT (1998) A chemokine expressed in lymphoid high endothelial venules promotes the adhesion and chemotaxis of naïve T lymphocytes. Proc Natl Acad Sci U S A 95:258–263
Hafler DA, Kent SC, Pietrusewicz MJ, Khoury SJ, Weiner HL, Fukaura H (1997) Oral administration of myelin induces antigen-specific TGF-beta 1 secreting T cells in patients with multiple sclerosis. Ann N Y Acad Sci 835:120–131
Haig DM, Hopkins J, Miller HRP (1999) Local immune responses in afferent and efferent lymph. Immunology 96:155–163
Herzog WR, Meade R, Pettinicchi A, Ptak W, Askenase PW (1989) Nude mice produce a T cell-derived antigen-binding factor that mediates the early component of delayed-type hypersensitivity. J Immunol 142:1803–1812
Heufler C, Koch F, Schuler G (1988) Granulocyte/macrophage colony-stimulating factor and interleukin 1 mediate the maturation of epidermal Langerhans cells into potent immunostimulatory dendritic cells. J Exp Med 167:700–705
Hill S, Edwards AJ, Kimber I, Knight SC (1990) Systemic migration of dendritic cells during contact sensitization. Immunology 71:277–281
Hindsen M, Christensen OB (1992) Delayed hypersensitivity reactions following allergic and irritant inflammation. Acta Derm Venereol 72:220–221
Hindsen M, Bruze M, Christensen OB (2001) Flare-up reactions after oral challenge with nickel in relation to challenge dose and intensity and time of previous patch test reactions. J Am Acad Dermatol 44:616–623
Hoath SB, Leahy DG (2003) The organization of human epidermis: functional epidermal units and phi proportionality. J Invest Dermatol 121:1440–1446
Hoefakker S, Caubo M, van Herve EHM, Roggeveen MJ, Boersma WJA, Van’t Joost T, Notten WRF, Claassen E (1995) In vivo cytokine profiles in allergic and irritant contact dermatitis. Contact Dermatitis 33:258–266
Hommel M (2004) On the dynamics of T-cell activation in lymph nodes. Immunol Cell Biol 82:62–66
Honig SM, Fu S, Mao X, Yopp A, Gunn MD, Randolph GJ, Bromberg JS (2003) FTY720 stimulates multidrug transporter- and cysteinyl leukotriene-dependent T cell chemotaxis to lymph nodes. J Clin Invest 111:627–637
Houck G, Saeed S, Stevens GL, Morgan MB (2004) Eczema and the spongiotic dermatoses: a histologic and pathogenic update. Semin Cutan Med Surg 23:39–45
Hoyer KK, Dooms H, Barron L, Abbas AK (2008) Interleukin-2 in the development and control of inflammatory disease. Immunol Rev 226:19–28
Iliev ID, Mileti E, Matteoli G, Chieppa M, Rescigno M (2009) Intestinal epithelial cells promote colitis-protective regulatory T-cell differentiation through dendritic cell conditioning. Mucosal Immunol 2(4):340–350
Inaba K, Schuler G, Witmer MD, Valinsky J, Atassi B, Steinman RM (1986) Immunologic properties of purified epidermal Langerhans cells. Distinct requirements for stimulation of unprimed and sensitized T lymphocytes. J Exp Med 164:605–613
Inaba K, Schuler G, Steinman RM (1993) GM-CSF – a granulocyte/macrophage/dendritic cell stimulating factor. In: Van Furth R (ed) Hemopoietic growth factors and mononuclear phagocytes. Karger, Basel, pp 187–196
Inerot A, Moller H (2000) Symptoms and signs reported during patch testing. Am J Contact Dermatol 11:49–52
Isaksson M, Bruze M (2003) Late patch-test reactions to budesonide need not be a sign of sensitization induced by the test procedure. Am J Contact Dermatol 14:154–156
Itano AA, Jenkins MK (2003) Antigen presentation to naive CD4 T cells in the lymph node. Nat Immunol 4:733–739
Iversen L, Johansen C (2008) Inflammasomes and inflammatory caspases in skin inflammation. Expert Rev Mol Diagn 8(6):697–705
Izcue A, Coombes JL, Powrie F (2009) Regulatory lymphocytes and intestinal inflammation. Annu Rev Immunol 27:313–338
Jakob T, Udey MC (1998) Regulation of E-cadherin mediated adhesion in Langerhans cell-like dendritic cells by inflammatory mediators that mobilize Langerhans cells in vivo. J Immunol 160:4067–4073
Jakob T, Ring J, Udey MC (2001) Multistep navigation of Langerhans/dendritic cells in and out of the skin. J Allergy Clin Immunol 108:688–696
Jensen CS, Menne T, Lisby S, Kristiansen J, Veien NK (2003) Experimental systemic contact dermatitis from nickel: a dose–response study. Contact Dermatitis 49:124–132
Kalinski P, Hilkens CMU, Snijders A, Snijdewint FGM, Kapsenberg ML (1997) IL-12 deficient dendritic cells, generated in the presence of prostaglandin E2, promote type-2 cytokine production in maturing human naïve T helper cells. J Immunol 159:28–35
Kalinski P, Schuitemaker JH, Hilkens CM, Kapsenberg ML (1998) Prostaglandin E2 induces the final maturation of IL-12 deficient CD1a + CD83+ dendritic cells. J Immunol 161:2804–2809
Kalish RS, Wood JA, LaPorte A (1994) Processing of urushiol (poison ivy) hapten by both endogenous and exogenous pathways for presentation to T cells in vitro. J Clin Invest 93:2039–2047
Kanerva L, Hyry H, Jolanki R, Hytonen M, Estlander T (1997) Delayed and immediate allergy caused by methylhexahydrophthalic anhydride. Contact Dermatitis 36:34–38
Kang KF, Kubin M, Cooper KD, Lessin SR, Trinchieri G, Rook AH (1996) IL-12 synthesis by human Langerhans cells. J Immunol 156:1402–1407
Karlberg AT, Bergström MA, Börje A, Luthman K, Nilsson JL (2008) Allergic contact dermatitis–formation, structural requirements, and reactivity of skin sensitizers. Chem Res Toxicol 21(1):53–69
Kidd P (2003) Th1/Th2 balance: the hypothesis, its limitations, and implications for health and disease. Altern Med Rev 8:223–246
Kim CH, Broxmeyer HE (1999) Chemokines: signal lamps for trafficking of T and B cells for development and effector function. J Leukoc Biol 65:6–15
Kimber I, Cumberbatch M (1992) Dendritic cells and cutaneous immune responses to chemical allergens. Toxicol Appl Pharmacol 117:137–146
Kimber I, Dearman RJ (2002) Allergic contact dermatitis: the cellular effectors. Contact Dermatitis 46:1–5
Kimber I, Dearman RJ (2003) What makes a chemical an allergen? Ann Allergy Asthma Immunol 90:28–31
Kimber I, Dearman RJ, Cumberbatch M, Huby RJ (1998) Langerhans cells and chemical allergy. Curr Opin Immunol 10:614–619
Kimber I, Basketter DA, Gerberick GF, Dearman RJ (2002) Allergic contact dermatitis. Int Immunopharmacol 2:201–211
Kitagaki H, Fujisawa S, Watanabe K, Hayakawa K, Shiohara T (1995) Immediate-type hypersensitivity response followed by late reaction is induced by repeated epicutaneous application of contact sensitizing agents in mice. J Invest Dermatol 105:749–755
Knop J, Stremmer R, Neumann C, Dc Maeyer D, Macher E (1982) Interferon inhibits the suppressor T cell response of delayed-type hypersensitivity. Nature 296:775–776
Kobayashi Y (1997) Langerhans cells produce type IV collagenase (MMP-9) following epicutaneous stimulation with haptens. Immunology 90:496–501
Kondo S, Sauder DN (1995) Epidermal cytokines in allergic contact dermatitis. J Am Acad Dermatol 33:786–800
Korn T, Bettelli E, Oukka M, Kuchroo VK (2009) IL-17 and Th17 cells (review). Annu Rev Immunol 27:485–517. PMID: 19132915
Kothny-Wilkes G, Kulms D, Poppelmann B, Luger TA, Kubin M, Schwarz T (1998) Interleukin-1 protects transformed keratinocytes from tumor necrosis factor-related apoptosis-inducing ligand. J Biol Chem 273:29247–29253
Krasteva M, Nicolas JF, Chabeau G, Garrigue JL, Bour H, Thivolet J, Schmitt D (1993) Dissociation of allergenic and immunogenic functions in contact sensitivity to para-phenylenediamine. Int Arch Allergy Immunol 102:200–204
Kubo M, Ransom J, Webb D, Hashimoto Y, Tada T, Nakayama T (1997) T-cell subset-specific expression of the IL-4 gene is regulated by a silencer element and STAT6. EMBO J 16(13):4007–4020
Kuchroo VK, Byrne MC, Atsumi Y, Greenfeld E, Connol JH, Whitters MJ, O’Hara RM, Collins M, Dorf ME (1991) T cell receptor alpha chain plays a critical role in antigen-specific suppressor cell function. Proc Natl Acad Sci U S A 88:8700–8704
Kumar V, Sercarz E (1998) Induction or protection from experimental autoimmune encephalomyelitis depends on the cytokine secretion profile of TCR peptide-specific regulatory CD4 T cells. J Immunol 161:6585–6591
Kuo CT, Leiden JM (1999) Transcriptional regulation of T lymphocyte development and function. Annu Rev Immunol 17:149–187
Lalani I, Bhol K, Ahmed AR (1997) Interleukin-10 biology, role in inflammation and autoimmunity. Ann Allergy Asthma Immunol 79:469–484
Langerhans P (1868) Über die nerven der menschlichen haut. Virchows Arch Pathol Anat 44:325–337
Lappin MB, Kimber I, Norval M (1996) The role of dendritic cells in cutaneous immunity. Arch Dermatol Res 288:109–121
Larsson A, Moller H, Björkner B, Bruze M (1997) Morphology of endogenous flare-up reactions in contact allergy to gold. Acta Derm Venereol 77:474–479
Lawrence JN, Dickson FM, Benford DJ (1997) Skin irritant-induced cytotoxicity and prostaglandin E-2 release in human skin keratinocyte cultures. Toxicol Vitro 11:627–631
Lepoittevin JP (2006) Metabolism versus chemical transformation or pro- versus prehaptens? Contact Dermatitis 54(2):73–74
Li QJ, Dinner AR, Qi S, Irvine DJ, Huppa JB, Davis MM, Chakraborty AK (2004) CD4 enhances T cell sensitivity to antigen by coordinating Lck accumulation at the immunological synapse. Nat Immunol 5:791–799
Liberato DJ, Byers VS, Ennick RG, Castagnoli N (1981) Region specific attack of nitrogen and sulfur nucleophiles on quinones from poison oak/ivy catechols (urushiols) and analogues as models for urushiol-protein conjugate formation. J Med Chem 24:28–33
Lider O, Cahalon L, Gilat D, Hershkoviz R, Siegel D, Margalit R, Shoseyov O, Cohen IR (1995) A disaccharide that inhibits tumor necrosis factor alpha is formed from the extracellular matrix by the enzyme heparinase. Proc Natl Acad Sci U S A 92:5037–5041
Lonati A, Licenziati S, Marcelli M, Canaris D, Pasolini G, Caruso A, de Panfilis G (1998) Quantitative analysis “at the single cell level” of the novel CD28-CD11b- subpopulation of CD8+ T lymphocytes. ESDR meeting, Cologne
Lord GM, Matarese G, Howard LK, Baker RJ, Bloom SR, Lechler RI (1998) Leptin modulates the T-cell immune response and reverses starvation-induced immunosuppression. Nature 394:897–901
Luger TA (2002) Neuromediators–a crucial component of the skin immune system. J Dermatol Sci 30(2):87–93
Luger TA, Lotti T (1998) Neuropeptides: role in inflammatory skin diseases. J Eur Acad Dermatol Venereol 10:207–211
Lundeberg L, Mutt V, Nordlind K (1999) Inhibitory effect of vasoactive intestinal peptide on the challenge phase of allergic contact dermatitis in humans. Acta Derm Venereol 79:178–182
Ma J, Wing J-H, Guo Y-J, Sy M-S, Bigby M (1994) In vivo treatment with anti-ICAM-1 and antiLFA-1 antibodies inhibits contact sensitization-induced migration of epidermal Langerhans cells to regional lymph nodes. Cell Immunol 158:389–399
Macatonia SE, Knight SC, Edwards AJ, Griffiths S, Fryer P (1987) Localization of antigen on lymph node dendritic cells after exposure to the contact sensitizer fluorescein isothiocyanate. Functional and morphological studies. J Exp Med 166:1654–1667
Marchal G, Seman M, Milon G, Truffa-Bachi P, Zilberfarb V (1982) Local adoptive transfer of skin delayed-type hypersensitivity initiated by a single T lymphocyte. J Immunol 129:954–958
Marone G, Spadaro G, Patella V, Genovese A (1994) The clinical relevance of basophil releasability. J Allergy Clin Immunol 94:1293–1303
Matura M (1998) Contact allergy to locally applied corticosteroids. Thesis, Leuven
Mayer L, Sperber K, Chan L, Child J, Toy L (2001) Oral tolerance to protein antigens. Allergy 56:12–15
Merk HF, Abel J, Baron JM, Krutmann J (2004) Molecular pathways in dermatotoxicology. Toxicol Appl Pharmacol 195:267–277
Mestas J, Hughes CC (2004) Of mice and not men: differences between mouse and human immunology. J Immunol 172:2731–2738
Miller SD, Sy M-S, Claman HN (1977) The induction of hapten-specific T cell tolerance using hapten-modified lymphoid membranes. II. Relative roles of suppressor T cells and clone inhibition in the tolerant state. Eur J Immunol 7:165–170
Milon G, Marchal G, Seman M, Truffa-Bachi P (1981) A delayed-type hypersensitivity reaction initiated by a single T lymphocyte. Agents Actions 11:612–614
Moed H, Boorsma DM, Stoof TJ, von Blomberg BM, Bruynzeel DP, Scheper RJ, Gibbs S, Rustemeyer T (2004a) Nickel-responding T cells are CD4+ CLA+ CD45RO+ and express chemokine receptors CXCR3, CCR4 and CCR10. Br J Dermatol 151:32–41
Moed H, Boorsma DM, Tensen CP, Flier J, Jonker MJ, Stoof TJ, Von Blomberg BM, Bruynzeel DP, Scheper RJ, Rustemeyer T, Gibbs S (2004b) Increased CCL27-CCR10 expression in allergic contact dermatitis: implications for local skin memory. J Pathol 204:39–46
Mokyr MB, Kalinichenko T, Gorelik L, Bluestone JA (1998) Realization of the therapeutic potential of CTLA-4 blockade in low-dose chemotherapy-treated tumor-bearing mice. Cancer Res 58:5301–5304
Morel PA, Oriss TB (1998) Crossregulation between Th1 and Th2 cells. Crit Rev Immunol 18:275–303
Morfin R, Lafaye P, Cotillon AC, Nato F, Chmielewski V, Pompon D (2000) 7 alpha-hydroxy-dehydroepiandrosterone and immune response. Ann N Y Acad Sci 917:971–982
Morgan DJ, Kreuwel HTC, Sherman LA (1999) Antigen concentration and precursor frequency determine the rate of CD8(+) T cell tolerance to peripherally expressed antigens. J Immunol 163:723–727
Morris DL (1998) Intradermal testing and sublingual in desensitization for nickel. Cutis 61:129–132
Moser B, Loetscher M, Piali L, Loetscher P (1998) Lymphocyte responses to chemokines. Int Rev Immunol 16:323–3244
Moser K, Kriwet K, Naik A, Kalia YN, Guy RH (2001) Passive skin penetration enhancement and its quantification in vitro. Eur J Pharm Biopharm 52:103–112
Moulon C, Vollmer J, Weltzien H-U (1995) Characterization of processing requirements and metal crossreactivities in T cell clones from patients with allergic contact dermatitis to nickel. Eur J Immunol 25:3308–3315
Mucida D, Salek-Ardakani S (2009) Regulation of TH17 cells in the mucosal surfaces. J Allergy Clin Immunol 123(5):997–1003
Murphy K, Travers P, Walport M (eds) (2008) Janeway’s immunobiology, 7th edn. Garland Science/Taylor & Francis Group, New York/Oxford
Mutschler J, Giménez-Arnau E, Foertsch L, Gerberick GF, Lepoittevin JP (2009) Mechanistic assessment of peptide reactivity assay to predict skin allergens with Kathon CG isothiazolinones. Toxicol Vitro 23(3): 439–446
Nagira M, Imai T, Hieshima K, Kusuda J, Ridanpaa M, Takagi S, Nishimura M, Kakizaki M, Nomiyama H, Yoshie O (1997) Molecular cloning of a novel human CC chemokine secondary lymphoid-tissue chemokine that is a potent chemoattractant for lymphocytes and mapped to chromosome 9p13. J Biol Chem 272:19518–19524
Naisbitt DJ (2004) Drug hypersensitivity reactions in skin: understanding mechanisms and the development of diagnostic and predictive tests. Toxicology 194:179–196
Nakamura T, Kamogawa Y, Bottomly K, Flavell RA (1997a) Polarization of IL-4- and IFN-gamma-producing CD4(+) T cells following activation of naive CD4(+) T cells. J Immunol 158:1085–1094
Nakamura T, Lee RK, Nam SY, Podack ER, Bottomly K, Flavell RA (1997b) Roles of IL-4 and IFN-g in stabilizing the T helper cell type-1 and 2 phenotype. J Immunol 158:2648–2653
Nataf S, Davoust N, Ames RS, Barnum SR (1999) Human T cells express the C5a receptor and are chemoattracted to C5a. J Immunol 162:4018–4023
Ngo VN, Tang LH, Cyster JG (1998) Epstein-Barr virus-induced molecule 1 ligand chemokine is expressed by dendritic cells in lymphoid tissues and strongly attracts naïve T cells and activated B cells. J Exp Med 188:181–191
O’Garra A (1998) Cytokines induce the development of functionally heterogeneous T helper cell subsets. Immunity 8:275–283
Ohshima Y, Tanaka Y, Tozawa H, Takahashi Y, Maliszewski C, Delespesse C (1997) Expression and function of OX40 ligand on human dendritic cells. J Immunol 159:3838–3848
Orange JS, Biron CA (1996) An absolute and restricted requirement for IL-12 in natural killer cell IFN-g production and antiviral defense. J Immunol 156:1138–1142
Orteu CH, Poulter LW, Rustin MHA, Sabin CA, Salmon M, Akbar AN (1998) The role of apoptosis in the resolution of T cell-mediated cutaneous inflammation. J Immunol 161:1619–1629
Oukka M (2008) Th17 cells in immunity and autoimmunity. In: Ann Rheum Dis 67(Suppl 3):iii26–iii29
Ozawa H, Nakagawa S, Tagami H, Aiba S (1996) Interleukin-1b and granulocyte-macrophage colony stimulating factor mediate Langerhans cell maturation differently. J Invest Dermatol 106:441–445
Panzani RC, Schiavino D, Nucera E, Pellegrino S, Fais G, Schinco G, Patriarca G (1995) Oral hyposensitization to nickel allergy: preliminary clinical results. Int Arch Allergy Immunol 107:251–254
Park SH, Chiu YH, Jayawardena J, Roark J, Kavita U, Bendelac A (1998) Innate and adaptive functions of the CD1 pathway of antigen presentation. Semin Immunol 10:391–398
Patlewicz GY, Basketter DA, Pease CK, Wilson K, Wright ZM, Roberts DW, Bernard G, Arnau EG, Lepoittevin JP (2004) Further evaluation of quantitative structure–activity relationship models for the prediction of the skin sensitization potency of selected fragrance allergens. Contact Dermatitis 50:91–97
Perez VL, Lederer JA, Lichtman AH, Abbas AK (1995) Stability of Th1 and Th2 populations. Int Immunol 7:869–875
Piccirillo CA, Thornton AM (2004) Cornerstone of peripheral tolerance: naturally occurring CD4+ CD25+ regulatory T cells. Trends Immunol 25:374–380
Pober JS, Bevilacqua MP, Mendrick DL, Lapierre LA, Fiers W, Gimbrone MA Jr (1986) Two distinct monokines, interleukin 1 and tumor necrosis factor, each independently induce biosynthesis and transient expression of the same antigen on the surface of cultured human vascular endothelial cells. J Immunol 136:1680–1687
Polak L (1980) Immunological aspects of contact sensitivity. An experimental study. Monogr Allergy 15:4–60
Polak L, Rinck C (1978) Mechanism of desensitization in DNCH-contact sensitive guinea pigs. J Invest Dermatol 70:98–104
Polak L, Turk JL (1968a) Studies on the effect of systemic administration of sensitizers in guinea-pigs with contact sensitivity to inorganic metal compounds. II. The flare-up of previous test sites of contact sensitivity and the development of a generalized rash. Clin Exp Immunol 3:253–262
Polak L, Turk SL (1968b) Studies on the effect of systemic administration of sensitizers in guinea pigs with contact sensitivity to inorganic metal compounds. I. The induction of immunological unresponsiveness in already sensitized animals. Clin Exp Immunol 3:245–251
Price AA, Cumberbatch M, Kimber I (1997) α6 integrins are required for Langerhans cell migration from the epidermis. J Exp Med 186:1725–1735
Probst P, Küntzlin D, Fleischer B (1995) TH2-type infiltrating T cells in nickel-induced contact dermatitis. Cell Immunol 165:134–140
Pulendran B (2004) Modulating TH1/TH2 responses with microbes, dendritic cells, and pathogen recognition receptors. Immunol Res 29:187–196
Quezada SA, Jarvinen LZ, Lind EF, Noelle RJ (2004) CD40/CD154 interactions at the interface of tolerance and immunity. Annu Rev Immunol 22:307–328
Rambukhana A, Bos JD, Irik D, Menko WJ, Kapsenberg ML, Das PK (1995) In situ behaviour of human Langerhans cells in skin organ culture. Lab Investig 73:521–531
Randolph GJ (2001) Dendritic cell migration to lymph nodes: cytokines, chemokines, and lipid mediators. Semin Immunol 13:267–274
Redegeld FA, Nijkamp FP (2003) Immunoglobulin free light chains and mast cells: pivotal role in T-cell-mediated immune reactions? Trends Immunol 24:181–185
Richters CD, Hoekstra MJ, van Baare J, Du Pont JS, Hoefsmit EC, Kamperdijk EW (1994) Isolation and characterization of migratory human skin dendritic cells. Clin Exp Immunol 98:330–336
Robbiani DF, Finch RA, Jager D, Muller WA, Sartorelli AC, Randolph GJ (2000) The leukotriene C(4) transporter MRP1 regulates CCL19 (MIP-3beta, ELC)-dependent mobilization of dendritic cells to lymph nodes. Cell 103:757–768
Roberts DW, Lepoittevin J-P (1998) Allergic contact dermatitis: the molecular basis. In: Lepoittevin J-P, Basketter DA, Goossens A, Karlberg A-T (eds) . Springer, Berlin/Heidelberg/New York, pp 81–1118
Rogge L, Barberis-Maino L, Biffi M, Passini N, Presky DH, Gubler U, Sinigaglia F (1997) Selective expression of an interleukin-12 receptor component by human T helper 1 cells. J Exp Med 185:825–831
Romagnani S (2006) Regulation of the T cell response. Clin Exp Allergy 36(11):1357–1366
Romani N, Holzmann S, Tripp CH, Koch F, Stoitzner P (2003) Langerhans cells – dendritic cells of the epidermis. Acta Pathol Microbiol Immunol Scand 111:725–740
Ross R, Gilitzer C, Kleinz R, Schwing J, Kleinert H, Forstermann U, Reske-Kunz AB (1998) Involvement of NO in contact hypersensitivity. Int Immunol 10:61–69
Rothenberg ME (2010) Innate sensing of nickel. Nat Immunol 11(9):781–782
Rowe A, Bunker CB (1998) Interleukin-4 and the interleukin-4 receptor in allergic contact dermatitis. Contact Dermatitis 38:36–39
Rowe A, Farrell AM, Bunker CB (1997) Constitutive endothelial and inducible nitric oxide synthase in inflammatory dermatoses. Br J Dermatol 136:18–23
Rustemeyer T, von Blomberg BME, de Ligter S, Frosch PJ, Scheper RJ (1999) Human T lymphocyte priming in vitro by haptenated autologous dendritic cells. Clin Exp Immunol 117:209–216
Rustemeyer T, de Groot J, von Blomberg BME, Frosch PJ, Scheper RJ (2001) Induction of tolerance and cross-tolerance to methacrylate contact sensitizers. Toxicol Appl Pharmacol 176:195–202
Rustemeyer T, de Groot J, von Blomberg BME, Frosch PJ, Scheper RJ (2002) Assessment of contact allergen cross-reactivity by retesting. Exp Dermatol 11:257–265
Rustemeyer T, von Blomberg BME, van Hoogstraten IMW, Bruynzeel DP, Scheper RJ (2004) Analysis of effector and regulatory immune-reactivity to nickel. Clin Exp Allergy 34(9):1458–1466
Saeki H, Moore AM, Brown MJ, Hwan ST (1999) Secondary lymphoid-tissue chemokine (SLC) and CC chemokine receptor 7 (CCR7) participate in the emigration pathway of mature dendritic cells from the skin to regional lymph nodes. J Immunol 162:2472–2475
Saint-Mezard P, Krasteva M, Chavagnac C, Bosset S, Akiba H, Kehren J, Kanitakis J, Kaiserlian D, Nicolas JF, Berard F (2003) Afferent and efferent phases of allergic contact dermatitis (ACD) can be induced after a single skin contact with haptens: evidence using a mouse model of primary ACD. J Invest Dermatol 120:641–647
Sallusto F, Lanzavecchia A, Mackay CR (1998a) Chemokines and chemokine receptors in T-cell priming and Th1/Th2-mediated responses. Immunol Today 19:568–574
Sallusto F, Schaerli P, Loetscher P, Schaniel C, Lenig D, Mackay CR, Qin S, Lanzavecchia A (1998b) Rapid and coordinated switch in chemokine receptor expression during dendritic cell maturation. Eur J Immunol 28:2760–2769
Sallusto F, Palermo B, Lenig D, Miettinen M, Matikainen S, Julkunen I, Forster R, Burgstahler R, Lipp M, Lanzavecchia A (1999) Distinct patterns and kinetics of chemokine production regulate dendritic cell function. Eur J Immunol 29:1617–1625
Sallusto F, Geginat J, Lanzavecchia A (2004) Central memory and effector memory T cell subsets: function, generation, and maintenance. Annu Rev Immunol 22:745–763
Scheper RJ, von Blomberg BME, Boerrigter GH, Bruynzeel D, van Dinther A, Vos A (1983) Induction of local memory in the skin. Role of local T cell retention. Clin Exp Immunol 51:141–148
Scheper RJ, van Dinther-Janssen AC, Polak L (1985) Specific accumulation of hapten-reactive T cells in contact sensitivity reaction sites. J Immunol 134:1333–1336
Schmidt M, Raghavan B, Müller V, Vogl T, Fejer G, Tchaptchet S, Keck S, Kalis C, Nielsen PJ, Galanos C, Roth J, Skerra A, Martin SF, Freudenberg MA, Goebeler M (2010) Crucial role for human Toll-like receptor 4 in the development of contact allergy to nickel. Nat Immunol 11(9):814–819
Schnuch A, Westphal GA, Muller MM, Schulz TG, Geier J, Brasch J, Merk HF, Kawakubo Y, Richter G, Koch P, Fuchs T, Gutgesell T, Reich K, Gebhardt M, Becker D, Grabbe J, Szliska C, Aberer W, Hallier E (1998) Genotype and phenotype of N-acetyltransferase 2 (NAT2) polymorphism in patients with contact allergy. Contact Dermatitis 38:209–211
Schoenberger SP, Toes REM, Vandervoort EIH, Offringa R, Melief CJM (1998) T-cell help for cytotoxic T lymphocytes is mediated by CD40-CD40L interactions. Nature 393:480–483
Schon MP, Zollner TM, Boehncke WH (2003) The molecular basis of lymphocyte recruitment to the skin: clues for pathogenesis and selective therapies of inflammatory disorders. J Invest Dermatol 121:951–962
Schuler G, Steinman RM (1985) Murine epidermal Langerhans cells mature into potent immune-stimulatory dendritic cells in vitro. J Exp Med 161:526–546
Schwarz A, Grabbe S, Riemann H, Aragane Y, Simon M, Manon S, Andrade S, Luger TA, Zlotnik A, Schwarz T (1994) In vivo effects of interleukin-10 on contact hypersensitivity and delayed-type hypersensitivity reactions. J Invest Dermatol 103:211–216
Schwarzenberger K, Udey MC (1996) Contact allergens and epidermal proinflammatory cytokines modulate Langerhans cell E-cadherin expression in situ. J Invest Dermatol 106:553–558
Seiffert K, Granstein RD (2006) Neuroendocrine regulation of skin dendritic cells. Ann N Y Acad Sci 1088(1):195–206
Semma M, Sagami S (1981) Induction of suppressor T cells to DNFB contact sensitivity by application of sensitizer through Langerhans cell-deficient skin. Arch Dermatol Res 271:361–364
Shimizu Y, Newman W, Gopal TV, Horgan KJ, Graber N, Beall LD, van Seventer GA, Shaw S (1991) Four molecular pathways of T cell adhesion to endothelial cells: roles of LFA-1, VCAM-1, and ELAM-1 and changes in pathway hierarchy under different activation conditions. J Cell Biol 113:1203–1212
Shirakawa T, Kusaka Y, Morimoto K (1992) Specific IgE antibodies to nickel in workers with known reactivity to cobalt. Clin Exp Allergy 22:213–218
Shreedhar V, Giese T, Sung VW, Ullrich SE (1998) A cytokine cascade including prostaglandin E2, IL-4, and IL-10 is responsible for UV-induced systemic immune suppression. J Immunol 160:3783–3789
Silberberg-Sinakin I, Thorbecke GJ, Baer RL, Rosenthal SA, Berezowsky V (1976) Antigen-bearing Langerhans cells in skin, dermal lymphatics, and in lymph nodes. Cell Immunol 25:137–151
Skog E (1976) Spontaneous flare-up reactions induced by different amounts of 1,3-dinitro-4-chlorobenzene. Acta Derm Venereol 46:386–395
Steinbrink K, Jonuleit H, Muller G, Schuler G, Knop J, Enk AH (1999) Interleukin-10-treated human dendritic cells induce a melanoma-antigen-specific anergy in CD8(+) T cells resulting in a failure to lyse tumor cells. Blood 93:1634–1642
Steinman RM, Hoffman L, Pope M (1995) Maturation and migration of cutaneous dendritic cells. J Invest Dermatol 105:2S–7S
Sterry W, Künne N, Weber-Matthiesen K, Brasch J, Mielke V (1991) Cell trafficking in positive and negative patch test reactions: demonstration of a stereotypic migration pathway. J Invest Dermatol 96:459–462
Stingl G, Katz SI, Clement L, Green I, Shevach EM (1978) Immunological functions of Ia-bearing epidermal Langerhans cells. J Immunol 121:2005–2013
Streilein JW, Grammer SF (1989) In vitro evidence that Langerhans cells can adopt two functionally distinct forms capable of antigen presentation to T lymphocytes. J Immunol 143:3925–3933
Strobel S, Mowat AM (1998) Immune responses to dietary antigens: oral tolerance. Immunol Today 19:173–181
Szepietowski JC, McKenzie RC, Keohane SG, Walker C, Aldridge RD, Hunter JA (1997) Leukaemia inhibitory factor: induction in the early phase of allergic contact dermatitis. Contact Dermatitis 36:21–25
Taams LS, van Rensen AJML, Poelen MC, van Els CACM, Besseling AC, Wagenaar JPA, van Eden W, Wauben MHM (1998) Anergic T cells actively suppress T cell responses via the antigen presenting cell. Eur J Immunol 28:2902–2912
Taams LS, van Eden W, Wauben MHM (1999) Dose-dependent induction of distinct anergic phenotypes: multiple levels of T cell anergy. J Immunol 162:1974–1981
Taams LS, Boot EPJ, van Eden W, Wauben MHM (2000) Anergic’ T cells modulate the T-cell activating capacity of antigen-presenting cells. J Autoimmun 14:335–341
Takatori H, Kanno Y, Chen Z, O’Shea JJ (2008) New complexities in helper T cell fate determination and the implications for autoimmune diseases. Mod Rheumatol 18(6):533–541
Tammaro A, De Marco G, Persechino S, Narcisi A, Camplone G (2009) Allergy to nickel: first results on patients administered with an oral hyposensitization therapy. Int J Immunopathol Pharmacol 22(3):837–840
Tang A, Amagai M, Granger LG, Stanley JR, Udey MC (1993) Adhesion of epidermal Langerhans cells to keratinocytes mediated by E-cadherin. Nature 361:82–85
Toebak MJ, Moed H, von Blomberg MB, Bruynzeel DP, Gibbs S, Scheper RJ, Rustemeyer T (2006) Intrinsic characteristics of contact and respiratory allergens influence production of polarizing cytokines by dendritic cells. Contact Dermatitis 55(4):238–245
Trautmann A, Randriamampita C (2003) Initiation of TCR signalling revisited. Trends Immunol 24:425–428
Tsuji RF, Geba GP, Wang Y, Kawamoto K, Matis LA, Askenase PW (1997) Required early complement activation in contact sensitivity with generation of local C5-dependent chemotactic activity, and late T cell interferon g: a possible initiating role of B cells. J Exp Med 186:1015–1026
Turk JL (1975) Delayed hypersensitivity, 2nd edn. North-Holland, Amsterdam
Ulrich P, Grenet O, Bluemel J, Vohr HW, Wiemann C, Grundler O, Suter W (2001) Cytokine expression profiles during murine contact allergy: T helper 2 cytokines are expressed irrespective of the type of contact allergen. Arch Toxicol 75(8):470–479
Uotila P (1996) The role of cyclic AMP and oxygen intermediates in the inhibition of cellular immunity in cancer. Cancer Immunol Immunother 43:1–9
van Beelen AJ, Teunissen MB, Kapsenberg ML, de Jong EC (2007) Interleukin-17 in inflammatory skin disorders. Curr Opin Allergy Clin Immunol 7(5):374–381
van de Ven R, Scheffer GL, Scheper RJ, de Gruijl TD (2009) The ABC of dendritic cell development and function. Trends Immunol 30(9):421–429
Van Hoogstraten IMW, Andersen JE, von Blomberg BME, Boden D, Bruynzeel DP, Burrows D, JMG C, Dooms-Goossens A, Lahti A, Menné T, Rycroft R, Todd D, KJJ V, Wilkinson JD, Scheper RJ (1989) Preliminary results of a multicenter study on the incidence of nickel allergy in relationship to previous oral and cutaneous contacts. In: Frosch PJ, Dooms-Goossens A, Lachapelle JM, RJG R, Scheper RJ (eds) Current topics in contact dermatitis. Springer, Berlin/Heidelberg/New York, pp 178–184
Van Hoogstraten IMW, von Blomberg BME, Boden D, Kraal G, Scheper RJ (1994) Non-sensitizing epicutaneous skin tests prevent subsequent induction of immune tolerance. J Invest Dermatol 102:80–83
Van Loweren H, Meade R, Askenase PW (1983) An early component of delayed type hypersensitivity mediated by T cells and mast cells. J Exp Med 157:1604–1617
Vestweber D, Blanks JE (1999) Mechanisms that regulate the function of the selectins and their ligands. Physiol Rev 79:181–213
Vieira PL, Kalinski P, Wierenga EA, Kapsenberg ML, Dejong EC (1998) Glucocorticoids inhibit bioactive IL-12P70 production by in vitro-generated human dendritic cells without affecting their T cell stimulatory potential. J Immunol 161:5245–5251
Viola A, Lanzavecchia A (1996) T cell activation determined by T cell receptor number and tunable thresholds. Science 273:104–106
Virag L, Szabo E, Bakondi E, Bai P, Gergely P, Hunyadi J, Szabo C (2002) Nitric oxide-peroxynitrite-poly(ADP-ribose) polymerase pathway in the skin. Exp Dermatol 11:189–202
Von Andrian UH, Mrini C (1998) In situ analysis of lymphocyte migration to lymph nodes. Cell Adhes Commun 6:85–96
von Blomberg BME, Bruynzeel DP, Scheper RJ (1991) Advances in mechanisms ofallergic contact dermatitis: in vitro and in vivo research. In: Marzulli FN, Maibach HI (eds) Dermatotoxicology, 4th edn. Hemisphere Publishing Corporation, New York, pp 255–362
von Herrath MG (1997) Bystander suppression induced by oral tolerance. Res Immunol 148:541–554
Waldorf HA, Walsh LJ, Schechter NM, Murphy GF (1991) Early molecular events in evolving cutaneous delayed hypersensitivity in humans. Am J Pathol 138:477–486
Walker C, Kristensen F, Bettens F, deWeck AL (1983) Lymphokine regulation of activated (G1) lymphocytes. I. Prostaglandin E2-induced inhibition of interleukin 2 production. J Immunol 130:1770–1773
Walsh LJ, Lavker RM, Murphy GF (1990) Determinants of immune cell trafficking in the skin. Lab Investig 63:592–600
Wang B, Esche C, Mamelak A, Freed I, Watanabe H, Sauder DN (2003) Cytokine knockouts in contact hypersensitivity research. Cytokine Growth Factor Rev 14:381–389
Wardorf HA, Walsh LJ, Schechter NM (1991) Early cellular events in evolving cutaneous delayed hypersensitivity in humans. Am J Pathol 138:477–486
Weigle WO, Romball CG (1997) CD4+ T-cell subsets and cytokines involved in peripheral tolerance. Immunol Today 18:533–538
Weiner HL (1997) Oral tolerance: immune mechanisms and treatment of autoimmune diseases. Immunol Today 18:335–343
Weiner HL, Gonnella PA, Slavin A, Maron R (1997) Oral tolerance: cytokine milieu in the gut and modulation of tolerance by cytokines. Res Immunol 148:528–533
Weinlich G, Heine M, Stössel H, Zanella M, Stoitzner P, Ortner U, Smolle J, Koch F, Sepp NT, Schuler G, Romani N (1998) Entry into afferent lymphatics and maturation in situ of migrating murine cutaneous dendritic cells. J Invest Dermatol 110:441–448
Weiss JM, Sleeman J, Renkl AC, Dittmar H, Termeer CC, Taxis S, Howells N, Hofmann M, Kohler G, Schöpf E, Ponta H, Herrlich P, Simon JC (1997) An essential role for CD44 variant isoforms in epidermal Langerhans cell and blood dendritic cell function. J Cell Biol 137:1137–1147
Wendel GD, Stark BJ, Jamison RB, Molina RD, Sullivan TJ (1985) Penicillin allergy and desensitization in serious infections during pregnancy. N Engl J Med 312:1229–1232
Werfel T, Hentschel M, Kapp A, Renz H (1997) Dichotomy of blood- and skin-derived IL-4-producing allergen-specific T cells and restricted V beta repertoire in nickel-mediated contact dermatitis. J Immunol 158:2500–2505
Weston MC, Peachell PT (1998) Regulation of human mast cell and basophil function by cAMP. Gen Pharmacol 31:715–719
Wilkinson SM, Mattey DL, Beck MH (1994) IgG antibodies and early intradermal reactions to hydrocortisone in patients with cutaneous delayed-type hypersensitivity to hydrocortisone. Br J Dermatol 131:495–498
Willis CM, Young E, Brandon DR, Wilkinson JD (1986) Immunopathological and ultrastructural findings in human allergic and irritant contact dermatitis. Br J Dermatol 115:305–316
Wilson NS, Villadangos JA (2004) Lymphoid organ dendritic cells: beyond the Langerhans cells paradigm. Immunol Cell Biol 82:91–98
Wong BR, Josien R, Lee SY, Sauter B, Li HL, Steinman RM, Choi YW (1997) TRANCE (Tumor necrosis factor [TNF]-related activation-induced cytokine), a new TNF family member predominantly expressed in T cells, is a dendritic cell-specific survival factor. J Exp Med 186:2075–2080
Wu K, Bi Y, Sun K, Wang C (2007) IL-10-producing type 1 regulatory T cells and allergy. Cell Mol Immunol 4(4):269–275
Yu X, Barnhill RL, Graves DT (1994) Expression of monocyte chemoattractant protein-1 in delayed type hypersensitivity reactions in the skin. Lab Investig 71:226–235
Zembala M, Ashershon GL (1973) Depression of T cell phenomenon of contact sensitivity by T cells from unresponsive mice. Nature 244:227–228
Zhang ZY, Michael JG (1990) Orally inducible immune unresponsiveness is abrogated by IFN-gamma treatment. J Immunol 144:4163–4165
Zhang X, Brunner T, Carter L, Dutton RW, Rogers P, Bradley L, Sato T, Reed JC, Green D, Swain SL (1997) Unequal death in T helper cell (Th)1 and Th2 effectors: Th1, but not Th2, effectors undergo rapid Fas/FasL-mediated apoptosis. J Exp Med 185:1837–1849
Zhou L, Littman DR (2009) Transcriptional regulatory networks in Th17 cell differentiation (review). Curr Opin Immunol 21(2):146–152
Zhou L, Chong MM, Littman DR (2009) Plasticity of CD4+ T cell lineage differentiation (review). Immunity 30(5):646–655. PMID: 19464987
Zinkernagel RM (2004) On ‘reactivity’ versus ‘tolerance. Immunol Cell Biol 82:343–352
Zlotnik A, Morales J, Hedrick JA (1999) Recent advances in chemokines and chemokine receptors. Crit Rev Immunol 19:1–47
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this entry
Cite this entry
Rustemeyer, T., van Hoogstraten, I.M.W., von Blomberg, B.M.E., Scheper, R.J. (2020). Mechanisms of Allergic Contact Dermatitis. In: John, S., Johansen, J., Rustemeyer, T., Elsner, P., Maibach, H. (eds) Kanerva’s Occupational Dermatology. Springer, Cham. https://doi.org/10.1007/978-3-319-68617-2_14
Download citation
DOI: https://doi.org/10.1007/978-3-319-68617-2_14
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-68615-8
Online ISBN: 978-3-319-68617-2
eBook Packages: MedicineReference Module Medicine