Abstract
Nodular lymphocyte predominant Hodgkin lymphoma (NLPHL) is a rare subtype of Hodgkin lymphoma representing around 5% of all Hodgkin lymphomas. It usually affects middle-aged men and has an indolent clinical behavior. In contrast to classical Hodgkin lymphoma, the tumor cells of NLPHL—called LP cells—express B-cell antigens. Different histopathological growth patterns related to the clinical behavior of NLPHL can be observed. In around 1/3 of NLPHL, the LP cells harbor translocations involving the BCL6 locus. Furthermore, somatic mutations of SOCS1, JUNB, DUSP2, and SGK1 are frequently found in LP cells. In this chapter we discuss the clinical behavior, morphologic growth patterns, immunophenotype, and molecular and genetic features of NLPHL.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
- Nodular lymphocyte predominant Hodgkin lymphoma
- Immunophenotype
- Genetic lesions
- Cell of origin
- Microenvironment
History
Nodular lymphocyte predominant Hodgkin lymphoma (NLPHL) was first recognized as a special subtype of Hodgkin lymphoma (HL) in the classification by Jackson and Parker in 1947 (Jackson and Parker 1947) and termed lymphocytic and histiocytic variant by Lukes and Butler in 1966 (Lukes and Butler 1966). Lennert distinguished already in 1974 (Lennert and Mohri 1974) between a nodular and a diffuse type of NLPHL. The tumor cells of NLPHL were previously called L and H cells, according to the lymphocytic and histiocytic appearance of the infiltrate. In the WHO classification of 2008 (Swerdlow et al. 2008) their name was revised to “lymphocyte predominant” or LP cells.
Clinical Characteristics
NLPHL has a predilection for the male gender, which is affected in about 75% of cases (Anagnostopoulos et al. 2000; Jackson et al. 2010). Male gender does not only represent an increased risk for the development of NLPHL but bears furthermore an even sixfold higher risk for relapse in diseased patients (Hartmann et al. 2013a). Usually, NLPHL affects middle-aged patients around 40 years. However, the age range is broad, including pediatric patients from approximately the age of 8 years up to elderly persons. In most cases, NLPHL is diagnosed in early stage, usually stage I or II (Jackson et al. 2010). Axillary and cervical lymph nodes are most frequently affected. Only a small subgroup of patients presents with advanced disease. These patients have often liver and spleen involvement. Although NLPHL is generally an indolently behaving lymphoma, relapses are much more frequently observed than in classical HL (cHL) (Anagnostopoulos et al. 2000). These can occur after a long latency of about 10 years. Some patients even present with multiple relapses, and in long standing disease, there is an important risk of transformation into an aggressive diffuse large B-cell lymphoma (DLBCL) (Biasoli et al. 2010; Al-Mansour et al. 2010), which can be fatal. The histological criteria when to diagnose transformation are not well defined, and small sheets of blasts do not seem to impact the clinical outcome (Hartmann et al. 2013a). Due to this reason, the outcome of patients with NLPHL and transformation into DLBCL is heterogeneous (Biasoli et al. 2010; Al-Mansour et al. 2010; Huang et al. 2004; Sundeen et al. 1988; Hansmann et al. 1989; Hartmann et al. 2014a).
Pathology
Growth Patterns
NLPHL can generally be divided into cases with nodular and diffuse growth patterns (Fig. 6.1a and b), (Hansmann et al. 1991; Boudova et al. 2003). Apart from their growth pattern, these also differ in the composition of the microenvironment. Cases with a predominant nodular pattern usually have a high content of reactive B cells, whereas in cases with a predominant diffuse growth pattern, ill-defined follicular dendritic cell meshworks in a T-cell-rich background can be observed (Hansmann et al. 1991). In 2003 a minute analysis of the different growth patterns and their combination was performed by Fan et al. (2003). Six different growth patterns were described (Fig. 6.2) including nodular patterns with a T-cell-rich background as well as a rare purely diffuse growth pattern with a B-cell-rich background. It was furthermore noted that the cases with a purely diffuse growth pattern and morphologic features resembling T-cell/histiocyte-rich large B-cell lymphoma (THRLBCL), i.e., diffusely distributed scattered large blasts in a background poor of B cells, had a higher risk of recurrence. This observation was confirmed in a large study involving 413 NLPHL patients from the German Hodgkin Study Group (Hartmann et al. 2013a), in which the cases with an atypical growth pattern presented significantly more frequently with an advanced clinical stage and a higher relapse rate. In some cases, the growth pattern can very closely resemble THRLBCL , and the revised WHO classification from 2016 (Swerdlow et al. 2016) suggests to label these cases NLPHL with THRLBCL-like transformation.
Schematic representation of NLPHL growth patterns (a–f), modified after Fan et al. (2003)
Tumor Cells: The LP Cells
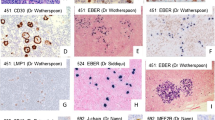
Although the growth pattern in NLPHL can vary, the LP cells , the tumor cells in NLPHL, almost always consist of mononucleated large blasts with folded, popcorn-like nuclei and usually one nucleolus (Fig. 6.3). The cytoplasm is usually a small rim. Tumor cells resembling classical Hodgkin-Reed-Sternberg (HRS) cells can only rarely be found in NLPHL. LP cells generally show a scattered distribution in the tissue with hot spot areas in the nodules of the typical nodular variants. LP cells derive from germinal center B cells and usually show a preserved B-cell phenotype, which can be slightly downregulated. They are positive for CD20, CD79a, OCT2, PAX5, BOB.1 (Fig. 6.4), and the germinal center markers BCL6 , HGAL, and CD75 (Anagnostopoulos et al. 2000; Natkunam et al. 2005; Carbone and Gloghini 2014; Kraus and Haley 2000). LP cells are furthermore positive for J chain (Stein et al. 1986) and EMA (Delsol et al. 1984) in a fraction of cases (Table 6.1). CD19 expression, which is found by almost all reactive B cells, is lost in the LP cells in more than half of the cases (Nathwani et al. 2013). A subset of NLPHL cases expresses IgD in the LP cells (Fig. 6.4) (Prakash et al. 2006). These are often pediatric male patients (Prakash et al. 2006; Huppmann et al. 2014). Furthermore, LP cells express the immunoglobulin kappa light chains more frequently than lambda light chains (Schmid et al. 1991). Infection by the Epstein-Barr virus (EBV) is only very rarely observed in LP cells (Anagnostopoulos et al. 2000; Huppmann et al. 2014).
Differential Diagnoses
Differential diagnoses to NLPHL include all kinds of lymphomas which present with scattered single B-cell blasts. This includes on the one hand cHL with CD20 expression, which occurs in rare cases. However, in these cases usually CD30 is much stronger expressed than CD20, and additionally CD15 is positive. Despite the fact that CD20 can be expressed by HRS cells, the expression intensity is mostly weak, and other B-cell antigens are negative in HRS cells of cHL. Both CD15 and CD30 can be expressed in rare cases of NLPHL, but usually they are not coexpressed (Hartmann et al. 2014a). Moreover, EBV is present in the HRS cells of cHL in around 30–40% of the cases in the Western world. This, however, depends very much on the country of origin, and EBV infection of HRS cells is more frequently encountered in developing nations. Other lymphomas that can be a differential diagnosis to NLPHL are all kinds of nodal peripheral T-cell lymphomas with Hodgkin-like cells (Quintanilla-Martinez et al. 1999; Moroch et al. 2012). Hodgkin-like cells in peripheral T-cell lymphomas usually do not belong to the T-cell lymphoma clone and are frequently EBV-infected B cells with a preserved B-cell phenotype and CD30 expression. Frequently the background infiltrate of nodal peripheral T-cell lymphomas can contain nodular areas of reactive B cells, resembling atypical variants of NLPHL. However, the most striking difference to NLPHL is the aberrant immunophenotype of the T cells , usually of T-helper cell origin and the clonality of the T cells, which helps to confirm an underlying T-cell neoplasia.
Another differential diagnosis to NLPHL is the progressive transformation of germinal centers (PTGC) (Poppema et al. 1979; Hansmann et al. 1990). Like in NLPHL, large nodular areas composed of naive B cells can occur in lymph nodes with PTGC. However, the important difference to NLPHL is that within these nodules still germinal center residues of variable size exist, which are completely destroyed in NLPHL. Moreover, although in PTGC scattered centroblasts can be found within germinal center residues, LP cells surrounded by rosetting T cells do not occur. PTGC follows exact morphological patterns (Hartmann et al. 2015a). Knowledge of these patterns can help to distinguish PTGC from NLPHL. However, in every lymph node with PTGC, a close workup is necessary, since sometimes NLPHL can coexist with PTGC in the same lymph node.
Cellular Origin
Applying single-cell PCR from micromanipulated LP cells, it was shown that LP cells are clonal and that they have ongoing somatic hypermutation of their immunoglobulin genes (Braeuninger et al. 1997). LP cells furthermore present an aberrant somatic hypermutation of the genes PIM1, PAX5, RhoH/TTF, and MYC (Liso et al. 2006) which are also mutated in germinal center B cells. Some of these mutations show intraclonal diversity in the LP cells, consistent with ongoing aberrant somatic hypermutation. All these data suggest that LP cells derive from germinal center B cells. This is furthermore consistent with the observed expression of germinal center B-cell markers in the LP cells. Interestingly, also in THRLBCL the tumor cells have clonal immunoglobulin gene rearrangements and show ongoing somatic hypermutation (Bräuninger et al. 1999), further supporting the close relationship between NLPHL and THRLBCL. When microdissected LP cells were investigated by gene expression profiling (Brune et al. 2008), they showed a similar degree of relationship to germinal center B cells and memory B cells, suggesting that they resemble an intermediate developmental stage in the transition between germinal center and memory B cells.
NLPHL Cell Line DEV
The NLPHL cell line DEV was established in 1985 by Poppema et al. (1985). Originally, it was assumed to be derived from cHL and only later reclassified as NLPHL (Poppema et al. 1989). The DEV cell line expresses B-cell antigens like CD20 and CD19 but is additionally positive for CD30. Moreover, it has an alternative BCL6 break and complex translocations involving chromosome 3 (Atayar et al. 2006). It furthermore displays a mutation in the start codon of the B2M gene, resulting in very low levels of B2M expression (Liu et al. 2014). DEV is negative for both human leukocyte antigens (HLA) classes I and II due to complex genomic rearrangements, including the gene locus of the HLA class II transactivator gene CIITA (Liu et al. 2014; Mottok et al. 2015).
Deregulated Transcription Factor Networks and Signaling Pathways
Primary LP cells have an active JAK-STAT signaling related to frequent mutations in SOCS1 (Mottok et al. 2007), which is a negative regulator of JAKs. Mutations in SOCS1 were usually found in motifs of somatic hypermutation, and they presented intraclonal diversity, in line with ongoing somatic hypermutation and the germinal center B-cell derivation of LP cells. JAK2 is phosphorylated in approximately 39% of NLPHL cases, whereas phosphorylation of STAT6 occurrs in 49% of NLPHL (Mottok et al. 2009). In contrast, phosphorylation of STAT3 and STAT5 was not seen in LP cells. It was furthermore observed by gene expression profiling that LP cells have a constitutive active NF-kappaB signaling, and a subset of cases shows activation of ERK (Brune et al. 2008). NF-kappaB activity in NLPHL is usually not related to mutations in NFKBIA or TNFAIP3, which are frequent in cHL (Schumacher et al. 2010).
Genetic Lesions
A variety of genetic lesions has been observed in NLPHL (Table 6.2). LP cells are always strongly positive for BCL6 , and translocations involving both the BCL6 locus and the immunoglobulin loci have been identified in up to 30% of NLPHL (Wlodarska et al. 2003; Renné et al. 2005). Additional NLPHL carries BCL6 translocations with diverse non-Ig locus partners (Wlodarska et al. 2003). By classic comparative genomic hybridization (CGH), a high number of genomic imbalances were detected both in NLPHL and THRLBCL (Franke et al. 2001, 2002). Surprisingly, in these studies, the number of aberrations was higher in NLPHL when compared with THRLBCL. However, the NLPHL cases investigated were not subtyped according to their growth patterns. Using array CGH , the number of aberrations was higher in THRLBCL and atypical NLPHL variants compared with typical NLPHL (Hartmann et al. 2015b). Despite the differences in the number of aberrations, common genomic events, e.g., gains of the REL locus, were recurrently detected in both NLPHL and THRLBCL.
Whole genome sequencing of two DLBCL derived from and clonally related to NLPHL and a subsequent targeted analysis of primary NLPHL revealed frequent mutations in SOCS1, DUSP2, JUNB, and SGK1 (Hartmann et al. 2016). Whereas JUNB works as an oncogene in classical HL, it was frequently affected by heterozygous stop mutations in NLPHL, leading to a very weak expression of the wild-type allele in the LP cells and suggesting a tumor suppressor function in NLPHL. SGK1 was strongly expressed both in the LP cells of primary NLPHL and the NLPHL cell line DEV. Application of a specific SGK1 inhibitor resulted in a high rate of apoptotic DEV cells, suggesting that SGK1 may act as an oncogene in NLPHL.
Familial NLPHL
There are several reports on families with accumulation of NLPHL (Merli et al. 2013; Saarinen et al. 2011, 2013). In one Finnish study, this was associated with a small germline deletion of serine 724 of the NPAT gene (Saarinen et al. 2011). However, there were also family members which were not affected by NLPHL. This small germline NPAT deletion was furthermore also observed in non-familial NLPHL patients, suggesting an increased risk for the development of NLPHL. Furthermore, genetic syndromes leading to a defect in the innate and adaptive immune system as well as the autoimmune lymphoproliferative syndrome, which can occur in children, have been associated with the development of NLPHL (Lorenzi et al. 2013; van den Berg et al. 2002).
Microenvironment
The microenvironment is considerably different in NLPHL with a typical nodular growth pattern compared to cases with a predominantly diffuse growth pattern. In typical NLPHL , where LP cells are mainly located within the B-cell nodules, the microenvironment has a strong similarity to that observed in normal germinal centers. Rosetting T cells around the LP cells are frequently observed, and these represent follicular T-helper cells, which are usually PD1 positive (Fig. 6.5) (Nam-Cha et al. 2008; Churchill et al. 2010). Furthermore a high number of CD57-positive cells are observed within the nodules of typical NLPHL (von Wasielewski et al. 1997). These CD57-positive cells are in the majority also CD4 and PD1 positive (Sattarzadeh et al. 2015). Other components in the microenvironment represent epithelioid cells which can be so abundant that they form granulomas (Fig. 6.6). A prominent epithelioid cell reaction was associated with a tendency to show less frequent relapses (Hartmann et al. 2014b).
In NLPHL with mainly diffuse areas, PD1- and CD57- as well as MUM1-positive rosetting T cells were less frequently observed (Churchill et al. 2010; Hartmann et al. 2013b). However, even rare cases of THRLBCL showed PD1-positive rosetting T cells. The NLPHL cases with predominant diffuse areas resembling THRLBCL present a high content of macrophages, like observed in THRLBCL (Hartmann et al. 2013b). Furthermore, a low lymphocyte-monocyte ratio in the peripheral blood was observed to be an independent risk factor for progression-free and overall survival in NLPHL (Porrata et al. 2012), likely reflecting the composition in the nodal compartment.
A specific double-positive T-cell population was observed in the microenvironment of NLPHL. This population consists of CD4+CD8+ double-positive T cells and occurs both in NLPHL and in PTGC (Rahemtullah et al. 2006, 2008). It can be detected by flow cytometry and can be helpful in the diagnosis of NLPHL. This double-positive T-cell population usually constitutes a minority of the T cells in the microenvironment (10–38% of the T cells) and probably represents an activated T-cell population.
Relationship to THRLBCL
Already a long time ago, it was noticed that both NLPHL and THRLBCL occur predominantly in middle-aged men, the tumor cells have the same immunophenotype , and NLPHL patients exist, who present with relapses under the morphologic picture of THRLBCL (Rüdiger et al. 2002). Vice versa, it has also been observed that THRLBCL patients developed NLPHL in the relapse situation (Rüdiger et al. 2002). The tumor cells in NLPHL and THRLBCL are both of germinal center origin (Braeuninger et al. 1997) and they have a high similarity in their gene expression patterns (Brune et al. 2008; Hartmann et al. 2013b). Furthermore they share common genetic events (Hartmann et al. 2015b). However, the clinical behavior of THRLBCL is usually aggressive (Achten et al. 2002) and distinct from that of typical NLPHL. Therefore, THRLBCL seems to be a lymphoma entity which is closely related to NLPHL but which may represent a tumor-cell poor transformation like a DLBCL with an abundant microenvironment. To date, the relationship between NLPHL and THRLBCL is not fully understood, and further workup is necessary.
Abbreviations
- CGH:
-
Comparative genomic hybridization
- cHL:
-
Classical Hodgkin lymphoma
- DLBCL:
-
Diffuse large B-cell lymphoma
- HLA:
-
Human leukocyte antigen
- NF-kappaB:
-
Nuclear factor kappa B
- NLPHL:
-
Nodular lymphocyte predominant Hodgkin lymphoma
- PTGC:
-
Progressive transformation of germinal centers
- THRLBCL:
-
T cell/histiocyte rich large B-cell lymphoma
References
Achten R, Verhoef G, Vanuytsel L, De Wolf-Peeters C (2002) T-cell/histiocyte-rich large B-cell lymphoma: a distinct clinicopathologic entity. J Clin Oncol 20:1269–1277
Al-Mansour M, Connors JM, Gascoyne RD, Skinnider B, Savage KJ (2010) Transformation to aggressive lymphoma in nodular lymphocyte-predominant Hodgkin’s lymphoma. J Clin Oncol 28:793–799
Anagnostopoulos I, Hansmann ML, Franssila K, Harris M, Harris NL, Jaffe ES et al (2000) European Task Force on Lymphoma project on lymphocyte predominance Hodgkin disease: histologic and immunohistologic analysis of submitted cases reveals 2 types of Hodgkin disease with a nodular growth pattern and abundant lymphocytes. Blood 96:1889–1899
Atayar C, Kok K, Kluiver J, Bosga A, van den Berg E, van der Vlies P et al (2006) BCL6 alternative breakpoint region break and homozygous deletion of 17q24 in the nodular lymphocyte predominance type of Hodgkin's lymphoma-derived cell line DEV. Hum Pathol 37:675–683
Biasoli I, Stamatoullas A, Meignin V, Delmer A, Reman O, Morschhauser F et al (2010) Nodular, lymphocyte-predominant Hodgkin lymphoma: a long-term study and analysis of transformation to diffuse large B-cell lymphoma in a cohort of 164 patients from the Adult Lymphoma Study Group. Cancer 116:631–639
Boudova L, Torlakovic E, Delabie J, Reimer P, Pfistner B, Wiedenmann S et al (2003) Nodular lymphocyte-predominant Hodgkin lymphoma with nodules resembling T-cell/histiocyte-rich B-cell lymphoma: differential diagnosis between nodular lymphocyte-predominant Hodgkin lymphoma and T-cell/histiocyte-rich B-cell lymphoma. Blood 102:3753–3758
Braeuninger A, Küppers R, Strickler JG, Wacker HH, Rajewsky K, Hansmann ML (1997) Hodgkin and Reed-Sternberg cells in lymphocyte predominant Hodgkin disease represent clonal populations of germinal center-derived tumor B cells. Proc Natl Acad Sci U S A 94:9337–9342
Bräuninger A, Küppers R, Spieker T, Siebert R, Strickler JG, Schlegelberger B et al (1999) Molecular analysis of single B cells from T cell-rich B-cell lymphoma shows the derivation of the tumor cells from mutating germinal center B cells and exemplifies means by which immunoglobulin genes are modified in germinal center B cells. Blood 93:2679–2687
Brune V, Tiacci E, Pfeil I, Doring C, Eckerle S, van Noesel CJ et al (2008) Origin and pathogenesis of nodular lymphocyte-predominant Hodgkin lymphoma as revealed by global gene expression analysis. J Exp Med 205:2251–2268
Carbone A, Gloghini A (2014) CD75: a B-cell marker which should not be forgotten in lymphocyte predominant Hodgkin lymphoma. Am J Hematol 89:449
Churchill HR, Roncador G, Warnke RA, Natkunam Y (2010) Programmed death 1 expression in variant immunoarchitectural patterns of nodular lymphocyte predominant Hodgkin lymphoma: comparison with CD57 and lymphomas in the differential diagnosis. Hum Pathol 41:1726–1734
Delsol G, Gatter KC, Stein H, Erber WN, Pulford KA, Zinne K et al (1984) Human lymphoid cells express epithelial membrane antigen. Implications for diagnosis of human neoplasms. Lancet 2:1124–1129
Fan Z, Natkunam Y, Bair E, Tibshirani R, Warnke RA (2003) Characterization of variant patterns of nodular lymphocyte predominant hodgkin lymphoma with immunohistologic and clinical correlation. Am J Surg Pathol 27:1346–1356
Franke S, Wlodarska I, Maes B, Vandenberghe P, Delabie J, Hagemeijer A et al (2001) Lymphocyte predominance Hodgkin disease is characterized by recurrent genomic imbalances. Blood 97:1845–1853
Franke S, Wlodarska I, Maes B, Vandenberghe P, Achten R, Hagemeijer A et al (2002) Comparative genomic hybridization pattern distinguishes T-cell/histiocyte-rich B-cell lymphoma from nodular lymphocyte predominance Hodgkin's lymphoma. Am J Pathol 161:1861–1867
Hansmann ML, Stein H, Fellbaum C, Hui PK, Parwaresch MR, Lennert K (1989) Nodular paragranuloma can transform into high-grade malignant lymphoma of B type. Hum Pathol 20:1169–1175
Hansmann ML, Fellbaum C, Hui PK, Moubayed P (1990) Progressive transformation of germinal centers with and without association to Hodgkin’s disease. Am J Clin Pathol 93:219–226
Hansmann ML, Stein H, Dallenbach F, Fellbaum C (1991) Diffuse lymphocyte-predominant Hodgkin's disease (diffuse paragranuloma). A variant of the B-cell-derived nodular type. Am J Pathol 138:29–36
Hartmann S, Eichenauer DA, Plutschow A, Mottok A, Bob R, Koch K et al (2013a) The prognostic impact of variant histology in nodular lymphocyte-predominant Hodgkin lymphoma: a report from the German Hodgkin Study Group (GHSG). Blood 122:4246–4252
Hartmann S, Doring C, Jakobus C, Rengstl B, Newrzela S, Tousseyn T et al (2013b) Nodular lymphocyte predominant hodgkin lymphoma and T cell/histiocyte rich large B cell lymphoma—endpoints of a spectrum of one disease? PLoS One 8:e78812
Hartmann S, Eray M, Doring C, Lehtinen T, Brunnberg U, Kujala P et al (2014a) Diffuse large B cell lymphoma derived from nodular lymphocyte predominant Hodgkin lymphoma presents with variable histopathology. BMC Cancer 14:332
Hartmann S, Eichenauer DA, Plutschow A, Mottok A, Bob R, Koch K et al (2014b) Histopathological features and their prognostic impact in nodular lymphocyte-predominant Hodgkin lymphoma—a matched pair analysis from the German Hodgkin Study Group (GHSG). Br J Haematol 167:238–242
Hartmann S, Winkelmann R, Metcalf RA, Treetipsatit J, Warnke RA, Natkunam Y et al (2015a) Immunoarchitectural patterns of progressive transformation of germinal centers with and without nodular lymphocyte-predominant Hodgkin lymphoma. Hum Pathol 46:1655–1661
Hartmann S, Doring C, Vucic E, Chan FC, Ennishi D, Tousseyn T et al (2015b) Array comparative genomic hybridization reveals similarities between nodular lymphocyte predominant Hodgkin lymphoma and T cell/histiocyte rich large B cell lymphoma. Br J Haematol 169:415–422
Hartmann S, Schuhmacher B, Rausch T, Fuller L, Doring C, Weniger M et al (2016) Highly recurrent mutations of SGK1, DUSP2 and JUNB in nodular lymphocyte predominant Hodgkin lymphoma. Leukemia 30:844–853
Huang JZ, Weisenburger DD, Vose JM, Greiner TC, Aoun P, Chan WC et al (2004) Diffuse large B-cell lymphoma arising in nodular lymphocyte predominant Hodgkin lymphoma: a report of 21 cases from the Nebraska Lymphoma Study Group. Leuk Lymphoma 45:1551–1557
Huppmann AR, Nicolae A, Slack GW, Pittaluga S, Davies-Hill T, Ferry JA et al (2014) EBV may be expressed in the LP cells of nodular lymphocyte-predominant Hodgkin lymphoma (NLPHL) in both children and adults. Am J Surg Pathol 38:316–324
Jackson H Jr, Parker F Jr (1947) Hodgkin’s disease and allied disorders. Oxford University Press, New York
Jackson C, Sirohi B, Cunningham D, Horwich A, Thomas K, Wotherspoon A (2010) Lymphocyte-predominant Hodgkin lymphoma—clinical features and treatment outcomes from a 30-year experience. Ann Oncol 21:2061–2068
Kraus MD, Haley J (2000) Lymphocyte predominance Hodgkin’s disease: the use of bcl-6 and CD57 in diagnosis and differential diagnosis. Am J Surg Pathol 24:1068–1078
Lennert K, Mohri N (1974) [Histological classification and occurrence of Hodgkin’s disease]. Internist (Berl) 15:57–65
Liso A, Capello D, Marafioti T, Tiacci E, Cerri M, Distler V et al (2006) Aberrant somatic hypermutation in tumor cells of nodular-lymphocyte-predominant and classic Hodgkin lymphoma. Blood 108:1013–1020
Liu Y, Abdul Razak FR, Terpstra M, Chan FC, Saber A, Nijland M et al (2014) The mutational landscape of Hodgkin lymphoma cell lines determined by whole-exome sequencing. Leukemia 28:2248–2251
Lorenzi L, Tabellini G, Vermi W, Moratto D, Porta F, Notarangelo LD et al (2013) Occurrence of nodular lymphocyte-predominant hodgkin lymphoma in hermansky-pudlak type 2 syndrome is associated to natural killer and natural killer T cell defects. PLoS One 8:e80131
Lukes RJ, Butler JJ (1966) The pathology and nomenclature of Hodgkin’s disease. Cancer Res 26:1063–1083
Merli M, Maffioli M, Ferrario A, Passamonti F (2013) Looking for familial nodular lymphocyte-predominant Hodgkin lymphoma. Am J Hematol 88:719–720
Moroch J, Copie-Bergman C, de Leval L, Plonquet A, Martin-Garcia N, Delfau-Larue MH et al (2012) Follicular peripheral T-cell lymphoma expands the spectrum of classical Hodgkin lymphoma mimics. Am J Surg Pathol 36:1636–1646
Mottok A, Renne C, Willenbrock K, Hansmann ML, Brauninger A (2007) Somatic hypermutation of SOCS1 in lymphocyte-predominant Hodgkin lymphoma is accompanied by high JAK2 expression and activation of STAT6. Blood 110:3387–3390
Mottok A, Renne C, Seifert M, Oppermann E, Bechstein W, Hansmann ML et al (2009) Inactivating SOCS1 mutations are caused by aberrant somatic hypermutation and restricted to a subset of B cell lymphoma entities. Blood 114:4503–4506
Mottok A, Woolcock B, Chan FC, Tong KM, Chong L, Farinha P et al (2015) Genomic alterations in CIITA are frequent in primary mediastinal large B cell lymphoma and are associated with diminished MHC class II expression. Cell Rep 13:1418–1431
Nam-Cha SH, Roncador G, Sanchez-Verde L, Montes-Moreno S, Acevedo A, Dominguez-Franjo P et al (2008) PD-1, a follicular T-cell marker useful for recognizing nodular lymphocyte-predominant Hodgkin lymphoma. Am J Surg Pathol 32:1252–1257
Nathwani B, Vornanen M, Winkelmann R, Kansal R, Döring C, Hartmann S et al (2013) Intranodular clusters of activated cells with T follicular helper (TFH) phenotype in nodular lymphocyte predominant Hodgkin lymphoma: a pilot study of 32 cases from Finland. Hum Pathol 44:1737–1746
Natkunam Y, Lossos IS, Taidi B, Zhao S, Lu X, Ding F et al (2005) Expression of the human germinal center-associated lymphoma (HGAL) protein, a new marker of germinal center B-cell derivation. Blood 105:3979–3986
Poppema S, Kaiserling E, Lennert K (1979) Nodular paragranuloma and progressively transformed germinal centers. Ultrastructural and immunohistologic findings. Virchows Archiv B 31:211–225
Poppema S, De Jong B, Atmosoerodjo J, Idenburg V, Visser L, De Ley L (1985) Morphologic, immunologic, enzymehistochemical and chromosomal analysis of a cell line derived from Hodgkin’s disease. Evidence for a B-cell origin of Sternberg-Reed cells. Cancer 55:683–690
Poppema S, Visser L, De Jong B, Brinker M, Atmosoerodjo J, Timens W (1989) The typical Reed-Sternberg phenotype and Ig gene rearrangement of Hodgkin's disease derived cell line ZO indicating a B cell origin. In: Diehl V, Pfreundschuh M, Loeffler M (eds) New aspects in the diagnosis and treatment of Hodgkin's disease. Springer, Berlin
Porrata LF, Ristow K, Habermann TM, Witzig TE, Colgan JP, Inwards DJ et al (2012) Peripheral blood lymphocyte/monocyte ratio at diagnosis and survival in nodular lymphocyte-predominant Hodgkin lymphoma. Br J Haematol 157:321–330
Prakash S, Fountaine T, Raffeld M, Jaffe ES, Pittaluga S (2006) IgD positive L&H cells identify a unique subset of nodular lymphocyte predominant Hodgkin lymphoma. Am J Surg Pathol 30:585–592
Quintanilla-Martinez L, Fend F, Moguel LR, Spilove L, Beaty MW, Kingma DW et al (1999) Peripheral T-cell lymphoma with Reed-Sternberg-like cells of B-cell phenotype and genotype associated with Epstein-Barr virus infection. Am J Surg Pathol 23:1233–1240
Rahemtullah A, Reichard KK, Preffer FI, Harris NL, Hasserjian RP (2006) A double-positive CD4+CD8+ T-cell population is commonly found in nodular lymphocyte predominant Hodgkin lymphoma. Am J Clin Pathol 126:805–814
Rahemtullah A, Harris NL, Dorn ME, Preffer FI, Hasserjian RP (2008) Beyond the lymphocyte predominant cell: CD4+CD8+ T-cells in nodular lymphocyte predominant Hodgkin lymphoma. Leuk Lymphoma 49:1870–1878
Renné C, Martin-Subero JI, Hansmann ML, Siebert R (2005) Molecular cytogenetic analyses of immunoglobulin loci in nodular lymphocyte predominant Hodgkin's lymphoma reveal a recurrent IGH-BCL6 juxtaposition. J Mol Diagn 7:352–356
Rüdiger T, Gascoyne RD, Jaffe ES, de Jong D, Delabie J, De Wolf-Peeters C et al (2002) Workshop on the relationship between nodular lymphocyte predominant Hodgkin’s lymphoma and T cell/histiocyte-rich B cell lymphoma. Ann Oncol 13(Suppl 1):44–51
Saarinen S, Aavikko M, Aittomaki K, Launonen V, Lehtonen R, Franssila K et al (2011) Exome sequencing reveals germline NPAT mutation as a candidate risk factor for Hodgkin lymphoma. Blood 118:493–498
Saarinen S, Pukkala E, Vahteristo P, Makinen MJ, Franssila K, Aaltonen LA (2013) High familial risk in nodular lymphocyte-predominant Hodgkin lymphoma. J Clin Oncol 31:938–943
Sattarzadeh A, Diepstra A, Rutgers B, van den Berg A, Visser L (2015) CD57+ T-cells are a subpopulation of T-follicular helper cells in nodular lymphocyte predominant Hodgkin lymphoma. Exp Hematol Oncol 4:27
Schmid C, Sargent C, Isaacson PG (1991) L and H cells of nodular lymphocyte predominant Hodgkin’s disease show immunoglobulin light-chain restriction. Am J Pathol 139:1281–1289
Schumacher MA, Schmitz R, Brune V, Tiacci E, Doring C, Hansmann ML et al (2010) Mutations in the genes coding for the NF-{kappa}B regulating factors I{kappa}B{alpha} and A20 are uncommon in nodular lymphocyte-predominant Hodgkin lymphoma. Haematologica 95:153–157
Stein H, Hansmann ML, Lennert K, Brandtzaeg P, Gatter KC, Mason DY (1986) Reed-Sternberg and Hodgkin cells in lymphocyte-predominant Hodgkin’s disease of nodular subtype contain J chain. Am J Clin Pathol 86:292–297
Sundeen JT, Cossman J, Jaffe ES (1988) Lymphocyte predominant Hodgkin’s disease nodular subtype with coexistent “large cell lymphoma”. Histological progression or composite malignancy? Am J Surg Pathol 12:599–606
Swerdlow SH, International Agency for Research on Cancer, World Health Organization (2008) WHO classification of tumours of haematopoietic and lymphoid tissues, 4th edn. International Agency for Research on Cancer, Lyon, France, pp 317–334
Swerdlow SH, Campo E, Pileri SA, Harris NL, Stein H, Siebert R et al (2016) The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood 127:2375–2390
van den Berg A, Maggio E, Diepstra A, de Jong D, van Krieken J, Poppema S (2002) Germline FAS gene mutation in a case of ALPS and NLP Hodgkin lymphoma. Blood 99(4):1492
von Wasielewski R, Werner M, Fischer R, Hansmann ML, Hubner K, Hasenclever D et al (1997) Lymphocyte-predominant Hodgkin’s disease. An immunohistochemical analysis of 208 reviewed Hodgkin's disease cases from the German Hodgkin Study Group. Am J Pathol 150:793–803
Wlodarska I, Nooyen P, Maes B, Martin-Subero JI, Siebert R, Pauwels P et al (2003) Frequent occurrence of BCL6 rearrangements in nodular lymphocyte predominance Hodgkin lymphoma but not in classical Hodgkin lymphoma. Blood 101:706–710
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG
About this chapter
Cite this chapter
Hartmann, S., Hansmann, ML. (2018). Pathobiology of Nodular Lymphocyte Predominant Hodgkin Lymphoma. In: Hudnall, S., Küppers, R. (eds) Precision Molecular Pathology of Hodgkin Lymphoma . Molecular Pathology Library. Springer, Cham. https://doi.org/10.1007/978-3-319-68094-1_6
Download citation
DOI: https://doi.org/10.1007/978-3-319-68094-1_6
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-68093-4
Online ISBN: 978-3-319-68094-1
eBook Packages: MedicineMedicine (R0)