Abstract
While cardiac magnetic resonance imaging is considered the clinical reference technique for the assessment of right ventricular (RV) size and function, echocardiography remains the first-line imaging technique. Recent guidelines for the use of echocardiography recommend the use of quantitative measures of RV dimensions and function. For functional assessment, a combination of fractional area change, tissue Doppler velocities and tricuspid annular planar systolic excursion (TAPSE) provide practical tools that can be used to monitor RV function serially. Novel techniques including three-dimensional echocardiography and strain imaging provide more detailed information that can be helpful in selected patient groups.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
The echocardiographic assessment of RV size and function remains a controversial topic. While most echocardiography laboratories rely predominantly upon qualitative assessment of the RV, recent guidelines and recommendations stress the importance of including quantitative assessment of RV size and function in clinical reports [1, 2]. Different studies have demonstrated clearly the limitations of qualitative assessment when compared to cardiac magnetic resonance imaging (MRI). For example, visual estimation of RV size and function is inaccurate and has wide interobserver variability. Adding quantitative echocardiographic parameters significantly improves accuracy and reliability of echocardiographic assessment [3]. Particularly in pediatric patients with more complex congenital heart disease like hypoplastic left heart syndrome , qualitative assessment of RV function often results in misclassification of the degree of RV dysfunction, even in the hands of experienced echocardiographers [4]. While these observations do not imply that qualitative evaluation is never useful as the first ‘gestalt’ for the echocardiographer, it does suggest that quantitative assessment of RV size and function is essential for quality assurance and accuracy.
Echocardiographic RV imaging is limited by different factors [5]. One of the challenges in the assessment of RV function is the more complex geometry of the RV, which significantly differs from the simpler ellipsoid left ventricle (LV) shape. This makes the RV more difficult to describe in a simple geometric model. Different complex mathematical formula for calculating RV volumes based on 2-D measurements have been proposed but all have limitations and are relatively complicated for application in routine clinical practice [6]. The anterior position in the chest further limits visualization of the RV anterior wall as located in the near field of the echo beam. The wrapping of the RV around the LV further limits two-dimensional imaging of the RV and makes it impossible to image the inflow, trabecular and outflow components in a single view. Assessing the size of the different RV components is especially relevant in conditions where regional RV remodelling occurs. For example, in patients after repair of tetralogy of Fallot (TOF), right ventricular outflow tract patch dilatation can lead to aneurysmal enlargement of the right ventricular outflow tract while pulmonary regurgitation tends to result in progressive enlargement of the RV inflow and apical segments [7]. Accurate determination of RV size in these patients will require imaging the different RV components.
When assessing RV function, the spec ific RV contraction pattern and physiology has to be taken into account. The RV contracts in a more longitudinal direction with a peristaltic wave moving from the RV inflow to the RV outflow, propelling the blood into the pulmonary circulation. This contrasts with the LV which contracts in a more circumferential direction with limited impact of longitudinal shortening on overall ejection.
Despite all these limitations and caveats, echocardiography can provide a reasonable quantitative assessment of RV size and function and should be performed in every patient where assessment of RV function is important.
Measurement of RV Dimensions
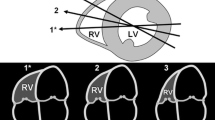
Different measurements of RV size have been proposed and are included in several guideline documents [1, 2, 8, 9]. One of the older measurements of RV size that is no longer included in the guidelines, but is still commonly used in clinical practice, is M-mode measurement of RV end-diastolic dimension from the parasternal long-axis or short-axis view used to assess LV size and function. When measuring the LV, the M-mode also cuts through the RV, close to the right ventricular outflow tract (RVOT) . The RV can be measured at end-diastole (R-wave of the QRS; Fig. 9.1). The problem with the measurement is that it is not based on specific RV anatomical landmarks and measures the RV trabecular part just below the RVOT. Thus, it does not really provide an assessment of different RV components and it can be difficult to avoid an oblique scan. Also, the septum can be activated early, related to right bundle branch block, resulting in abnormal septal motion during the R-wave of the QRS (septal flash). The main reason this measurement is still used in clinical practice, is the availability of pediatric z-scores which allows evaluating changes in RV size over time, independent of patient growth. Zervan et al. demonstrated progressive enlargement of this RV M-mode dimension the first 7 years after TOF repair, with regression after pulmonary valve replacement [10]. This suggests the measurement can be used for tracking progressive RV dilatation.
Measurement of RV dimension from the parasternal short axis M-mode in patients after TOF repair. In the upper panel, the RV is positioned nicely anterior to the LV and the M-mode is a straight line through the RV trabecular part just below the RVOT. In the lower panel it is difficult to obtain an image with the RV positioned anterior to the LV. There is abnormal septal motion related to early septal activation and delayed anterior wall activation resulting in the inward septal motion during the R-wave (small blue arrows)
Recent guidelines recommend measuring RV dimensions from the apical RV-centric view. This view is obtained from the standard apical 4-chamber view by sliding the transducer slightly medially on the chest putting the right ventricular (RV) apex in the middle of the imaging sector. Measurements are obtained in mid-diastole (when the ventricle is largest, Fig. 9.2). The RV transverse diameter is measured at the basal one-third below the tricuspid annulus (RV 1) and in the middle third of RV inflow, approximately halfway between the maximal basal diameter and the apex, at the level of papillary muscles at end-diastole (RV2). RV length is measured from the mid-point of the tricuspid valve annulus to the RV apex. In adults, RV1 > 41 mm and RV2 > 35 mm, indicates RV dilatation. Pediatric z-scores for these RV measurements are currently unavailable, which limits the application of these measurements in children. Additionally the apical RV view only images the RV inflow and trabecular parts and does not include the RV outflow. Especially in patients in whom the RVOT is dilated this results in underestimation of overall RV size. Lai et al. demonstrated that echocardiographic RV dimensions as measured from the apical 4-chamber view are smaller compared to MRI measurements [11]. From the parasternal short and long-axis views, proximal RVOT dimensions can be measured (Fig. 9.3). These measurements are similar to the M-mode measurements obtained from the same windows but are less dependent on alignment of the probe.
RV width and length measurements from an RV-centric apical view . RV1 is measured at the basal level, from the endocardium of the RV free wall to the interventricular septum (IVS). RV2 is measured in the middle third of RV inflow, approximately halfway between the maximal basal diameter and the apex, at the level of papillary muscles at end-diastole (left panel). RV length is measured from the mid-point of the tricuspid valve annulus to the RV apex
From the RV-centric apical view, the RV end-diastolic area can be measured at end-diastole. We include the RV trabeculations in the measurement of RV area as we also use a similar approach when measuring MRI RV v olumes (Fig. 9.4). Especially in adult patients with limited imaging windows, delineation of the RV free wall can be difficult and foreshortening from the RV-centric view should also be avoided. In older patients after TOF repair, RV end-diastolic area indexed for body surface area (RVEDAi) was shown to correlate linearly with indexed RV end-diastolic volume (RVEDVi) as measured by MRI [(RVEDVi = 11.5 + (7 × RVEDAi]) [12]. In children after TOF repair RVEDAi > 20 cm2/m2 predicts RVEDVi > 170 ml/m2 with high sensitivity and specificity [13]. This relatively simple measurement can provide guidance for selecting patients for cardiac MRI for assessment of RV volumes and also for the serial follow-up of RV dimensions. Adding 2-dimensional measurements from additional RV views such as the apical RV 2-chamber and RV 3-chamber views further increase the accuracy of RV size assessment. The feasibility of these views is however influenced by the limited visualization of the anterior RV wall segments, certainly in adults. In newborns, however, our group demonstrated a high feasibility and reproducibility of additional RV measurements from these views [14].
Assessment of RV wall thickness in the assessment of RV hypertrophy remains difficult due to lack of reproducible anatomic landmarks that allow standardization of RV wall thickness measurements. It is recommended to assess RV wall thickness in the RV free wall as imaged from subcostal long-axis views at end-diastole below the tricuspid annulus at a distance approximating the length of the anterior tricuspid leaflet when it is full open and parallel to the RV free wall. The measurement should exclude trabeculae, papillary muscles and epicardial fat. In adults, RV thickness >0.5 cm is considered abnormal. However, no normal pediatric data are available. The recommended measurement technique is difficult and prone to measurement variability as it can be challenging to define the endocardial and epicardial borders related to the RV trabeculations.
Measurement of RV Volumes
Three-dimensional echocardiography is being increasingly adopted for measuring RV end-diastolic and end-systolic volumes. Full volumetric RV data acquisition can be performed from the RV-centric apical view focussing on including the entire RV cavity and all RV walls. This can be difficult in adult patients with significant RV dilatation. Additionally as 3-D spatial resolution is lower than 2-D resolution, visualization of all RV endocardial borders can be challenging. This limits feasibility of 3-D RV volumetric assessment, especially in adult patients after congenital heart surgery. Different studies reported feasibility of only 50–60% [15]. Different software packages are available for RV 3-D volumetric measurements. They require tracing the RV endocardial borders at end-diastole and end-systole. Semi-automated tracing is available but manual adjustments are often required and complicate RV volumetric measurements. RV trabeculations and the moderator band are included in the volume measurements. Intra and interobserver variability between the different methods can be important and should be taken into account when RV 3-D volumes are used. When compared to MRI volumetric data, 3-D echocardiographic volumes generally underestimate RV volumes by 10–25% [15,16,17,18]. This difference seems larger in more dilated RVs. These intertechnique differences should be taken into account when decision-making is based on RV volumes such as for pulmonary valve replacement in patients after TOF repair as most of the cut-off values for RV volumes have been based on MRI measurements.
In adults, normal data on echocardiographic RV volumes are available, and are influenced by age and gender [1]. For adults, RVEDVi of 87 mL/m2 in men and 74 mL/m2 are considered the upper limits of normal. With increasing age, RVEDVi progressively decreases. In children, good, normal echocardiographic 3-D reference data are not available limiting clinical utility of this method in younger age groups.
Other techniques have been proposed such as the 2-D based knowledge reconstruction method which uses magnetic tracking attached to the ultrasound probe for localizing RV 2-D images in 3-D space. On the 2-D images anatomic landmarks are identified and based on a database of RV shapes, RV volumes are reconstructed. While this method has excellent feasibility, reliability and accuracy, its clinical use is limited as image acquisition and analysis is cumbersome and requires specific equipment [16, 19]. Our group found the method difficult to integrate in a high-volume clinical echocardiography laboratory.
Echocardiographic Assessment of RV Systolic Function
Different echocardiographic methods have been proposed for assessing RV systolic function. In most validation studies, RV ejection fraction (RVEF) as measured by MRI is used as the gold-standard reference technique for assessing global RV function. RVEF is influenced by contractile function, size and loading conditions. In the adult population RVEF is also influenced by age and gender. Utilizing RVEF as a parameter for RV function should thus be interpreted carefully, taking the different parameters influencing the measurement into account when used for clinical decision-making.
Three-dimensional echocardiography is the best echocardiographic technique for measuring RVEF , albeit with the limitations mentioned above. Apart from limited feasibility in a post-surgical population, the reproducibility of the method is influenced by variability in measurement of RV systolic volumes. In our experience, RVESV is more difficult to measure, as at end-systole the trabeculations can be difficult to delineate precisely [16]. This influences variability in RVEF calculations. While in adults an RVEF >45% is considered normal, with slightly higher values in women than men, no reliable normative data are available for children. Furthermore, as 3-D echocardiography is not readily available in every laboratory and because the technique is cumbersome, time-consuming and requires specific software and expertise, this method is not widely used in clinical practice and other techniques for assessing RV function have been introduced and validated.
RV fractional area change (FAC) is a reasonable substitute for RVEF. It measures the area change between end-systole and end-diastole from the RV-centric apical 4-chamber view (Fig. 9.5). As for EF, trabeculations are included in the FAC measurement. FAC correlates well with RV EF [20] and for adults FAC >35% is considered normal. No good, normative pediatric data have been established. Our data in newborns suggest that in the neonatal period FAC is lower compared to the adult population with a mean value of 26% for the apical 4-chamber FAC. In this study FAC was also measured from the apical RV 3-chamber view, which resulted in higher values (mean 3-C FAC was 39%). More reference data are required but it is our recommendation to include RV FAC as a standard measurement for assessing RV function.
Measurement of fractional area change in patient with hypoplastic left heart syndrome. From the apical RV-centric view, RV end diastolic area (EDA) and end systole areas (ESA) are measured and FAC is calculated by calculating (EDA-ESA)/EDA. For this patient FAC was estimated as 39% which is within normal rage
Tricuspid annular planar systolic excursion or TAPSE measures systolic longitudinal displacement of the lateral tricuspid annulus from the RV-centric RV view. It can be measured by placing an M-mode cursor through the tricuspid lateral annulus and measuring the distance the annulus travels from end-diastole to end-systole. The M-mode cursor needs to be well aligned with the longitudinal motion often requiring sliding the probe more rightward and lateral. To standardize timing of the measurement and improve the reproducibility, a color tissue Doppler M-mode tracing can be obtained (Fig. 9.6). TAPSE is a parameter assessing RV longitudinal function. If RV dysfunction is global, it correlates well with RVEF but in case of regional RV dysfunction the correlation between TAPSE and RVEF or FAC is much lower. In TOF patients post-repair for instance, TAPSE correlates only moderately with global RVEF as RVOT and anterior wall dysfunction are not captured by TAPSE [21]. In adults, normal TAPSE is >17 mm. Normal values and z-scores have been published for children and infants [22, 23]. In adults with pulmonary hypertension TAPSE has high prognostic value while in children with pulmonary arterial hypertension the prognostic value of TAPSE is less well established [24, 25]. Measurement of TAPSE is influenced by cardiac translation, RV-LV interactions and tricuspid valve regurgitation.
Tricus pid Annular Planar Systolic Excursion (TAPSE) can be measured using the RV-centric apical 4-chamber view. Place the cursor parallel to the RV free wall and activate TDI colour mapping. Activate M-mode and obtain a tracing of the TV annulus. Measure TAPSE from the end-diastolic point (A) to the maximal excursion point at end-systole (B). TAPSE is the distance between A and B in centimeters. The use of TDI colour mapping, though not required, helps define the systolic timing (transition blue-red is start systole; transition red-blue is end systole. TAPSE is measured between the two points
Tricuspid lateral annulus systolic velocity (s’) as measured by tissue Doppler imaging (TDI) is another measurement of RV longitudinal function that is easy to obtain and is highly-reproducible (Fig. 9.7). As for TAPSE alignment with longitudinal motion of the RV lateral wall is important and should be optimized. The pulsed-Doppler cursor is placed at the tricuspid annulus in adults and just below the annulus for children. In adults s’ >9.5 cm/s is considered normal. Normal data for children have been obtained including z-scores [26, 27]. TDI-derived s’ is a regional parameter assessing RV longitudinal function. In the case of global RV dysfunction, s’ can be reduced but is influenced by cardiac translation, tricuspid regurgitation, ventricular interactions and loading conditions (changes in preload and afterload). Typically after cardiac surgery RV s’-values are reduced and remain reduced during follow-up while FAC often normalizes. This is possibly related to reduced cardiac translation after opening the pericardium and the effect of adhesions on RV longitudinal motion. When regional dysfunction is present, s’ also does not correlate well with global RVEF . This was nicely demonstrated in patients after tetralogy of Fallot repair where the relationship between s’ and RVEF as measured by MRI was influenced by RVOT function [28]. All these factors have to be taken into consideration when interpreting RV s’-values. RV s’-velocities have been reported to be reduced after cardiac surgery and heart transplantation while RV ejection fraction is within normal range [29]. In these conditions RV s’ does not seem to represent global RV function.
RV longitudinal strain and strain rate can be measured from the apical 4-chamber view using speckle-tracking echocardiography. Typically RV strain and strain rate are calculated in the RV free wall as the interventricular septal values are generally used for LV longitudinal strain values although the septum should be considered an important interface between RV and LV (Fig. 9.8). Typically end-systolic strain values in the RV basal, mid and apical RV free wall segments are measured. Most strain software applications are not specifically designed to measure RV strain, and differences between values obtained using different vendors have been documented. In adults average lateral wall end-systolic longitudinal strain values < −20 are considered normal. As the negative value can be confusing, this corresponds to an absolute value of >20. Limited normal RV strain data are available in children. Using the apical RV 2-chamber and 3-chamber views, RV longitudinal strain values can also be obtained in the RV inferior and posterior walls [30, 31]. The anterior wall and right ventricular outflow tract are more difficult to track and feasibility of obtaining strain measurements in these RV segments is low even in children with good imaging windows. Thus a ‘global RV strain’ value is more difficult to obtain although different wall segments can be evaluated.
RV longitudinal strain measurements have been investigated in different diseases and have been proven to add additional prognostic information in patients with heart failure [32, 33], pulmonary hypertension in adults [34,35,36] and also in children [37], ischemic heart disease [38,39,40] and aortic stenosis [32, 41]. In congenital heart disease, different studies have looked at RV longitudinal function using strain imaging. After tetralogy of Fallot repair, RV longitudinal strain and strain rate are reduced with the RV apical segments more significantly affected compared to the RV basal parts. The reduction in RV longitudinal strain is associated with exercise capacity in children [42,43,44,45,46] and both RV and LV longitudinal strain values were demonstrated to have prognostic value in adults after TOF repair [47, 48]. Right bundle branch block is common after tetralogy of Fallot repair and causes dyssynchronous RV contraction with early septal activation and delayed RV lateral wall contraction [49]. In adults, biventricular pacing has been suggested to be potentially beneficial in post-operative tetralogy patients with RV dysfunction and right bundle branch block [50,51,52]. Strain imaging has also been proven to provide additional prognostic information for assessment of systemic RV function such as after atrial switch for transposition of the great arteries [53] and after palliative surgery for hypoplastic left heart syndrome [54]. In both conditions it was observed that adequate RV remodelling and adaptation involved a transition in RV mechanics from a predominantly longitudinal to a more circumferential contraction pattern [55]. Further standardization and optimization of RV strain measurements are required for routine clinical utilization but it can be expected to become part of routine RV functional assessment for selected patient groups. More recently three-dimensional strain methods have been proposed for assessing RV global longitudinal strain [56,57,58]. These methods could provide simultaneous assessment of RV volumes, ejection fraction and mechanics.
Assessment of RV Diastolic Function
Assessment of RV diastolic function is even more challenging than assessment of LV diastolic function. Guideline recommendations include assessment of IVC diameter and collapse with respiration, tricuspid inflow patterns, tricuspid annulus tissue Doppler velocities (e’) and E/e’-ratio [2]. Restrictive RV physiology is suggested by late antegrade flow in the pulmonary artery during atrial contraction. This can be measured by pulsed wave velocity with a sample volume placed midway between the pulmonary valve and the bifurcation of the main pulmonary artery and assessing for the presence of late diastolic antegrade flow thoughout inspiration and expiration (Fig. 9.9). There is poor validation of RV diastolic functional assessment especially in the pediatric age group.
Restrictive RV physiology in post-operative tetralogy of Fallot patient. Pulsed wave spectral Doppler trace is obtained between the pulmonary valve leaflets. Late diastolic antegrade forward flow is observed throughout respiration suggestive of RV end-diastolic pressure exceeding pulmonary diastolic pressure
Conclusion
Quantitative assessment of RV size and function should be part of every echocardiographic examination performed in patients in whom RV function can be affected, especially in patients with pulmonary hypertension or patients with congenital heart disease affecting the RV. Two-dimensional measurements of dimensions and RV areas provide simple tools for screening. More accurate assessment of RV size can be performed using three-dimensional echocardiography with the caveat that echocardiographic volumes are lower compared to MRI volumes. For functional RV assessment, measurement of FAC, TAPSE and s’ wave velocity also are good screening tools. Improvements in echocardiographic equipment and analysis software will hopefully result in 3-D and RV strain imaging becoming more easily accessible techniques for the assessment of RV size and function.
References
Lang RM, Badano LP, Mor-Avi V, Afilalo J, Armstrong A, Ernande L, et al. Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Eur Heart J Cardiovasc Imaging. 2015;16(3):233–70.
Rudski LG, Lai WW, Afilalo J, Hua L, Handschumacher MD, Chandrasekaran K, et al. Guidelines for the echocardiographic assessment of the right heart in adults: a report from the American Society of Echocardiography endorsed by the European Association of Echocardiography, a registered branch of the European Society of Cardiology, and the Canadian Society of Echocardiography. J Am Soc Echocardiogr. 2010;23(7):685–713. quiz 86–8.
Ling LF, Obuchowski NA, Rodriguez L, Popovic Z, Kwon D, Marwick TH. Accuracy and interobserver concordance of echocardiographic assessment of right ventricular size and systolic function: a quality control exercise. J Am Soc Echocardiogr. 2012;25(7):709–13.
Bellsham-Revell HR, Simpson JM, Miller OI, Bell AJ. Subjective evaluation of right ventricular systolic function in hypoplastic left heart syndrome: how accurate is it? J Am Soc Echocardiogr. 2013;26(1):52–6.
Mertens LL, Friedberg MK. Imaging the right ventricle—current state of the art. Nat Rev Cardiol. 2010;7(10):551–63.
Helbing WA, Bosch HG, Maliepaard C, Rebergen SA, van der Geest RJ, Hansen B, et al. Comparison of echocardiographic methods with magnetic resonance imaging for assessment of right ventricular function in children. Am J Cardiol. 1995;76(8):589–94.
Sheehan FH, Ge S, Vick GW 3rd, Urnes K, Kerwin WS, Bolson EL, et al. Three-dimensional shape analysis of right ventricular remodeling in repaired tetralogy of Fallot. Am J Cardiol. 2008;101(1):107–13.
Lang RM, Bierig M, Devereux RB, Flachskampf FA, Foster E, Pellikka PA, et al. Recommendations for chamber quantification: a report from the American Society of Echocardiography’s Guidelines and Standards Committee and the Chamber Quantification Writing Group, developed in conjunction with the European Association of Echocardiography, a branch of the European Society of Cardiology. J Am Soc Echocardiogr. 2005;18(12):1440–63.
Lopez L, Colan SD, Frommelt PC, Ensing GJ, Kendall K, Younoszai AK, et al. Recommendations for quantification methods during the performance of a pediatric echocardiogram: a report from the Pediatric Measurements Writing Group of the American Society of Echocardiography Pediatric and Congenital Heart Disease Council. J Am Soc Echocardiogr. 2010;23(5):465–95. quiz 576–7.
Zervan K, Male C, Benesch T, Salzer-Muhar U. Ventricular interaction in children after repair of tetralogy of Fallot: a longitudinal echocardiographic study. Eur J Echocardiogr. 2009;10(5):641–6.
Lai WW, Gauvreau K, Rivera ES, Saleeb S, Powell AJ, Geva T. Accuracy of guideline recommendations for two-dimensional quantification of the right ventricle by echocardiography. Int J Cardiovasc Imaging. 2008;24(7):691–8.
Greutmann M, Tobler D, Biaggi P, Mah ML, Crean A, Oechslin EN, et al. Echocardiography for assessment of right ventricular volumes revisited: a cardiac magnetic resonance comparison study in adults with repaired tetralogy of Fallot. J Am Soc Echocardiogr. 2010;23(9):905–11.
Alghamdi MH, Grosse-Wortmann L, Ahmad N, Mertens L, Friedberg MK. Can simple echocardiographic measures reduce the number of cardiac magnetic resonance imaging studies to diagnose right ventricular enlargement in congenital heart disease? J Am Soc Echocardiogr. 2012;25(5):518–23.
Jain A, Mohamed A, El-Khuffash A, Connelly KA, Dallaire F, Jankov RP, et al. A comprehensive echocardiographic protocol for assessing neonatal right ventricular dimensions and function in the transitional period: normative data and z scores. J Am Soc Echocardiogr. 2014;27(12):1293–304.
Khoo NS, Young A, Occleshaw C, Cowan B, Zeng IS, Gentles TL. Assessments of right ventricular volume and function using three-dimensional echocardiography in older children and adults with congenital heart disease: comparison with cardiac magnetic resonance imaging. J Am Soc Echocardiogr. 2009;22(11):1279–88.
Dragulescu A, Grosse-Wortmann L, Fackoury C, Mertens L. Echocardiographic assessment of right ventricular volumes: a comparison of different techniques in children after surgical repair of tetralogy of Fallot. Eur Heart J Cardiovasc Imaging. 2012;13(7):596–604.
Grewal J, Majdalany D, Syed I, Pellikka P, Warnes CA. Three-dimensional echocardiographic assessment of right ventricular volume and function in adult patients with congenital heart disease: comparison with magnetic resonance imaging. J Am Soc Echocardiogr. 2010;23(2):127–33.
van der Zwaan HB, Geleijnse ML, McGhie JS, Boersma E, Helbing WA, Meijboom FJ, et al. Right ventricular quantification in clinical practice: two-dimensional vs. three-dimensional echocardiography compared with cardiac magnetic resonance imaging. Eur J Echocardiogr. 2011;12(9):656–64.
Dragulescu A, Grosse-Wortmann L, Fackoury C, Riffle S, Waiss M, Jaeggi E, et al. Echocardiographic assessment of right ventricular volumes after surgical repair of tetralogy of Fallot: clinical validation of a new echocardiographic method. J Am Soc Echocardiogr. 2011;24(11):1191–8.
Anavekar NS, Gerson D, Skali H, Kwong RY, Yucel EK, Solomon SD. Two-dimensional assessment of right ventricular function: an echocardiographic-MRI correlative study. Echocardiography. 2007;24(5):452–6.
Koestenberger M, Nagel B, Avian A, Ravekes W, Sorantin E, Cvirn G, et al. Systolic right ventricular function in children and young adults with pulmonary artery hypertension secondary to congenital heart disease and tetralogy of Fallot: tricuspid annular plane systolic excursion (TAPSE) and magnetic resonance imaging data. Congenit Heart Dis. 2012;7(3):250–8.
Koestenberger M, Nagel B, Ravekes W, Urlesberger B, Raith W, Avian A, et al. Systolic right ventricular function in preterm and term neonates: reference values of the tricuspid annular plane systolic excursion (TAPSE) in 258 patients and calculation of Z-score values. Neonatology. 2011;100(1):85–92.
Koestenberger M, Ravekes W, Everett AD, Stueger HP, Heinzl B, Gamillscheg A, et al. Right ventricular function in infants, children and adolescents: reference values of the tricuspid annular plane systolic excursion (TAPSE) in 640 healthy patients and calculation of z score values. J Am Soc Echocardiogr. 2009;22(6):715–9.
Hauck A, Guo R, Ivy DD, Younoszai A. Tricuspid annular plane systolic excursion is preserved in young patients with pulmonary hypertension except when associated with repaired congenital heart disease. Eur Heart J Cardiovasc Imaging. 2016;18(4):459–66.
Kassem E, Humpl T, Friedberg MK. Prognostic significance of 2-dimensional, M-mode, and Doppler echo indices of right ventricular function in children with pulmonary arterial hypertension. Am Heart J. 2013;165(6):1024–31.
Dallaire F, Slorach C, Hui W, Sarkola T, Friedberg MK, Bradley TJ, et al. Reference values for pulse wave Doppler and tissue Doppler imaging in pediatric echocardiography. Circ Cardiovasc Imaging. 2015;8(2):e002167.
Eidem BW, McMahon CJ, Cohen RR, Wu J, Finkelshteyn I, Kovalchin JP, et al. Impact of cardiac growth on Doppler tissue imaging velocities: a study in healthy children. J Am Soc Echocardiogr. 2004;17(3):212–21.
Kutty S, Zhou J, Gauvreau K, Trincado C, Powell AJ, Geva T. Regional dysfunction of the right ventricular outflow tract reduces the accuracy of Doppler tissue imaging assessment of global right ventricular systolic function in patients with repaired tetralogy of Fallot. J Am Soc Echocardiogr. 2011;24(6):637–43.
Raina A, Vaidya A, Gertz ZM, Susan C, Forfia PR. Marked changes in right ventricular contractile pattern after cardiothoracic surgery: implications for post-surgical assessment of right ventricular function. J Heart Lung Transplant. 2013;32(8):777–83.
Forsha D, Risum N, Kropf PA, Rajagopal S, Smith PB, Kanter RJ, et al. Right ventricular mechanics using a novel comprehensive three-view echocardiographic strain analysis in a normal population. J Am Soc Echocardiogr. 2014;27(4):413–22.
Rajagopal S, Forsha DE, Risum N, Hornik CP, Poms AD, Fortin TA, et al. Comprehensive assessment of right ventricular function in patients with pulmonary hypertension with global longitudinal peak systolic strain derived from multiple right ventricular views. J Am Soc Echocardiogr. 2014;27(6):657–65. e3
Galli E, Guirette Y, Feneon D, Daudin M, Fournet M, Leguerrier A, et al. Prevalence and prognostic value of right ventricular dysfunction in severe aortic stenosis. Eur Heart J Cardiovasc Imaging. 2015;16(5):531–8.
Motoki H, Borowski AG, Shrestha K, Hu B, Kusunose K, Troughton RW, et al. Right ventricular global longitudinal strain provides prognostic value incremental to left ventricular ejection fraction in patients with heart failure. J Am Soc Echocardiogr. 2014;27(7):726–32.
Haeck ML, Scherptong RW, Marsan NA, Holman ER, Schalij MJ, Bax JJ, et al. Prognostic value of right ventricular longitudinal peak systolic strain in patients with pulmonary hypertension. Circ Cardiovasc Imaging. 2012;5(5):628–36.
Park JH, Park MM, Farha S, Sharp J, Lundgrin E, Comhair S, et al. Impaired global right ventricular longitudinal strain predicts long-term adverse outcomes in patients with pulmonary arterial hypertension. J Cardiovasc Ultrasound. 2015;23(2):91–9.
van Kessel M, Seaton D, Chan J, Yamada A, Kermeen F, Butler T, et al. Prognostic value of right ventricular free wall strain in pulmonary hypertension patients with pseudo-normalized tricuspid annular plane systolic excursion values. Int J Cardiovasc Imaging. 2016;32(6):905–12.
Okumura K, Humpl T, Dragulescu A, Mertens L, Friedberg MK. Longitudinal assessment of right ventricular myocardial strain in relation to transplant-free survival in children with idiopathic pulmonary hypertension. J Am Soc Echocardiogr. 2014;27(12):1344–51.
Antoni ML, Scherptong RW, Atary JZ, Boersma E, Holman ER, van der Wall EE, et al. Prognostic value of right ventricular function in patients after acute myocardial infarction treated with primary percutaneous coronary intervention. Circ Cardiovasc Imaging. 2010;3(3):264–71.
Chang WT, Liu YW, Liu PY, Chen JY, Lee CH, Li YH, et al. Association of decreased right ventricular strain with worse survival in non-acute coronary syndrome angina. J Am Soc Echocardiogr. 2016;29(4):350–8.e4.
Park SJ, Park JH, Lee HS, Kim MS, Park YK, Park Y, et al. Impaired RV global longitudinal strain is associated with poor long-term clinical outcomes in patients with acute inferior STEMI. JACC Cardiovasc Imaging. 2015;8(2):161–9.
Dahou A, Clavel MA, Capoulade R, Bartko PE, Magne J, Mundigler G, et al. Right ventricular longitudinal strain for risk stratification in low-flow, low-gradient aortic stenosis with low ejection fraction. Heart. 2016;102(7):548–54.
Weidemann F, Eyskens B, Mertens L, Dommke C, Kowalski M, Simmons L, et al. Quantification of regional right and left ventricular function by ultrasonic strain rate and strain indexes after surgical repair of tetralogy of Fallot. Am J Cardiol. 2002;90(2):133–8.
Eyskens B, Brown SC, Claus P, Dymarkowski S, Gewillig M, Bogaert J, et al. The influence of pulmonary regurgitation on regional right ventricular function in children after surgical repair of tetralogy of Fallot. Eur J Echocardiogr. 2010;11(4):341–5.
Dragulescu A, Grosse-Wortmann L, Redington A, Friedberg MK, Mertens L. Differential effect of right ventricular dilatation on myocardial deformation in patients with atrial septal defects and patients after tetralogy of Fallot repair. Int J Cardiol. 2012;168(2):803–10.
Alghamdi MH, Mertens L, Lee W, Yoo SJ, Grosse-Wortmann L. Longitudinal right ventricular function is a better predictor of right ventricular contribution to exercise performance than global or outflow tract ejection fraction in tetralogy of Fallot: a combined echocardiography and magnetic resonance study. Eur Heart J Cardiovasc Imaging. 2013;14(3):235–9.
Friedberg MK, Fernandes FP, Roche SL, Slorach C, Grosse-Wortmann L, Manlhiot C, et al. Relation of right ventricular mechanics to exercise tolerance in children after tetralogy of Fallot repair. Am Heart J. 2013;165(4):551–7.
Diller GP, Kempny A, Liodakis E, Alonso-Gonzalez R, Inuzuka R, Uebing A, et al. Left ventricular longitudinal function predicts life-threatening ventricular arrhythmia and death in adults with repaired tetralogy of fallot. Circulation. 2012;125(20):2440–6.
Orwat S, Diller GP, Kempny A, Radke R, Peters B, Kuhne T, et al. Myocardial deformation parameters predict outcome in patients with repaired tetralogy of Fallot. Heart. 2016;102(3):209–15.
Hui W, Slorach C, Dragulescu A, Mertens L, Bijnens B, Friedberg MK. Mechanisms of right ventricular electromechanical dyssynchrony and mechanical inefficiency in children after repair of tetralogy of fallot. Circ Cardiovasc Imaging. 2014;7(4):610–8.
Thambo JB, Dos Santos P, De Guillebon M, Roubertie F, Labrousse L, Sacher F, et al. Biventricular stimulation improves right and left ventricular function after tetralogy of Fallot repair: acute animal and clinical studies. Heart Rhythm. 2010;7(3):344–50.
Thambo JB, De Guillebon M, Dos Santos P, Xhaet O, Ploux S, Iriart X, et al. Electrical dyssynchrony and resynchronization in tetralogy of Fallot. Heart Rhythm. 2011;8(6):909–14.
Thambo JB, De Guillebon M, Xhaet O, Dos Santos P, Roubertie F, Labrousse L, et al. Biventricular pacing in patients with Tetralogy of Fallot: non-invasive epicardial mapping and clinical impact. Int J Cardiol. 2013;163(2):170–4.
Kalogeropoulos AP, Deka A, Border W, Pernetz MA, Georgiopoulou VV, Kiani J, et al. Right ventricular function with standard and speckle-tracking echocardiography and clinical events in adults with D-transposition of the great arteries post atrial switch. J Am Soc Echocardiogr. 2012;25(3):304–12.
Khoo NS, Smallhorn JF, Kaneko S, Kutty S, Altamirano L, Tham EB. The assessment of atrial function in single ventricle hearts from birth to Fontan: a speckle-tracking study by using strain and strain rate. J Am Soc Echocardiogr. 2013;26(7):756–64.
Pettersen E, Helle-Valle T, Edvardsen T, Lindberg H, Smith HJ, Smevik B, et al. Contraction pattern of the systemic right ventricle shift from longitudinal to circumferential shortening and absent global ventricular torsion. J Am Coll Cardiol. 2007;49(25):2450–6.
Atsumi A, Seo Y, Ishizu T, Nakamura A, Enomoto Y, Harimura Y, et al. Right ventricular deformation analyses using a three-dimensional speckle-tracking echocardiographic system specialized for the right ventricle. J Am Soc Echocardiogr. 2016;29(5):402–11.e2.
Ryo K, Goda A, Onishi T, Delgado-Montero A, Tayal B, Champion HC, et al. Characterization of right ventricular remodeling in pulmonary hypertension associated with patient outcomes by 3-dimensional wall motion tracking echocardiography. Circ Cardiovasc Imaging. 2015;8(6):pii: e003176.
Smith BC, Dobson G, Dawson D, Charalampopoulos A, Grapsa J, Nihoyannopoulos P. Three-dimensional speckle tracking of the right ventricle: toward optimal quantification of right ventricular dysfunction in pulmonary hypertension. J Am Coll Cardiol. 2014;64(1):41–51.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG
About this chapter
Cite this chapter
Mertens, L.L. (2018). Echocardiographic Assessment of the Right Ventricle. In: Friedberg, M., Redington, A. (eds) Right Ventricular Physiology, Adaptation and Failure in Congenital and Acquired Heart Disease. Springer, Cham. https://doi.org/10.1007/978-3-319-67096-6_9
Download citation
DOI: https://doi.org/10.1007/978-3-319-67096-6_9
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-67094-2
Online ISBN: 978-3-319-67096-6
eBook Packages: MedicineMedicine (R0)