Abstract
Kranz anatomy is very interesting as a perfect example of connection between structure and functional processes in C4 photosynthetic plants. It has been evidenced in the nineteenth century in many Chenpodiaceae species and recognized and nominated as Kranz anatomy later in the history of C4 photosynthesis. It comprises two closed and distinct chlorenchyma tissues: an external one and an inner bundle sheath tissue. These tissues are arranged concentrically with respect to vascular tissues. There are many sub-types described in the frame of this chlorenchymatic arrangement and they are reviewed here in relation to old research findings.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
Generally, the plant leaf is considered the organ with highest plasticity in terms of structural and adaptive value. Indeed, without insisting on the well-known role and functions that leaf plays in the life plant, it can be stated that the leaf represents the physical support for functional processes emerging from the long process of evolution and adaptation of plant facing many harmful environmental factors (Grigore 2008).
In several cases, it was observed that leaves of many species that grows in difficult conditions (salinity, aridity) present peculiar features related to special differential tissue functionality, in the direction of a better adaptation to the environment.
From all of these adaptations, Kranz anatomy is very interesting as a perfect example of connection between structure and functional processes in C4 photosynthetic plants. It has been noticed a long time ago that nervures from Atriplex species are surrounded by a sheath of cubic cells containing chloroplasts, higher than other mesophyll cells (Laetsch 1968).
Generally, it is considered that the botanist Moser (1934) used first time the expression containing the word “Kranz,” in relation to the foliar anatomy of Atriplex tataricum, where he observed that the nervures are surrounded by chlorenchymatic sheaths (Der Kranztypus in der Gattung Atriplex); he also depicted this type of structure. Literally, Kranz means “wreath, corona” that explicitly illustrates the way in which these tissues are arranged around vascular bundles. Thereafter, it was discovered that monocots can also present this kind of leaf structure. In fact, Moser only systematized and deepened this issue, explicitly referring to “Kranztypus in der Gattung Atriplex’” (p. 380), but Volkens in the two papers (1887—mentioned by Moser and another from 1893—consulted by us) mentioned about the arrangement of palisade tissue around nervures in Atriplex species (A. halimus, A. roseum, and A. sibiricum): “Nervenbündel (…), um die sich Palissaden im Kranze herumlegen.” (Volkens 1893, p. 64—see also Fig. 6.1).
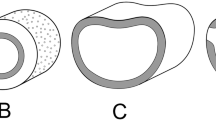
Cross section through the lamina of Zygophyllum simplex (ep epidermis, ext ch external chlorenchyma = palisade tissue, int ch internal chlorenchyma = bundle sheath, el v vascular elements? If yes, then the structure would fit with salsoloid sybtype) (Volkens 1887)
However, since Moser mentioned Volkens’ paper from 1887 and Volkens himself uses this description in the work from 1893, where he introduces drawings from 1887 monograph, we should consider, based on this data, that the first mention of Kranz structure in Chenopodiaceae was made in 1887. Close to this period of time, Arcangeli (1890), Italian botanist, also described a similar structure in Atriplex nummularia.
A close connection was made between this type of structure and C4 plants.
It is well known that in the great majority of C4 plants, functioning of the C4 pathway requires metabolic cooperation of two closed and distinct chlorenchyma tissues: an external one (or photosynthetic carbon assimilative—PCA) and an inner bundle sheath (or photosynthetic carbon reductive—PCR) tissue. These tissues are arranged concentrically with respect to vascular tissues, forming a structural pattern known as Kranz anatomy (Muhaidat et al. 2007). This structural type provides one of the best examples of the intimate connection between plant form and function and represents a suite of structural characters that have evolved repeatedly from C3 ancestors (Dengler and Nelson 1999; Kellog 1999; Sage 2001, 2004).
However, in order to simplify the strictly anatomical language used in this chapter, we will refer to photosynthetic carbon assimilative (PCA) tissue as external chlorenchyma and to photosynthetic carbon reductive (PCR) as internal chlorenchyma, respectively.
This internal structure physically partitions the biochemical events of the C4 pathway into two major phases. In the first step, atmospheric CO2 is initially assimilated into C4 acids by PCA tissue-specific phosphoenolpyruvate carboxylase. In the second phase, these acids diffuse into the PCR compartment, where they are decarboxylated, and the released CO2 is refixed by PCR tissue-specific Rubisco. This biphasic C4 system enhances CO2 levels around Rubisco, suppressing photorespiration and improving plant carbon balance (Kanai and Edwards 1999).
In fact, the biochemical and physiological events associated with C4 photosynthesis are indefinitely much complicated, and in this chapter, attention will be maintained only to its structural support, the Kranz anatomy.
Consequently, three subtypes of C4 biochemical pathways have been described, as follows (Kanai and Edwards 1999):
-
1.
NADP-ME (NADP-malic enzyme)
-
2.
NAD-ME (NAD-malic enzyme)
-
3.
PEP-CK (phosphoenolpyruvate-carboxykinase)
These three types occur in Poaceae, each of which is characterized by a set of “classical” sequence of anatomical and ultrastructural features, which include the number of vascular bundle sheath layers, the position of chloroplasts within internal chlorenchyma cells, the development of chloroplast grana, the size and number of mitochondria, and the occurrence of a suberin lamella within the internal chlorenchyma cell walls (Gutierrez et al. 1974; Hattersley and Browning 1981; Yoshimura et al. 2004; Ueno et al. 2005).
In dicots, only NADP-ME and NAD-ME subtypes can be found. These two subtypes cannot be obviously distinguished on the basis of anatomical characteristics; for example, PCR cell chloroplasts have a centripetal position in both NADP-ME and NAD-ME subtypes, with a few exceptions, where chloroplasts are centrifugally placed. As in C4 grasses, the two subtypes are distinguished by a pronounced cytological dimorphism between cells of the two types of chlorenchyma tissues (Voznesenskaya et al. 1999). Thus, PCR cell chloroplasts in NADP-ME subtype eudicots have greatly reduced grana (associated with a high ratio between photosynthetic systems I and II), while those of PCA cells have well-developed grana (high levels of photosynthetic systems I and II). The higher photosynthetic system I:II ratio in PCR of NADP-ME species reflects a higher proportion of cyclic electron flow, related to linear electron flow. The opposite is true for PCR chloroplasts of NAD-ME C4 plants (Kanai and Edwards 1999; Voznesenskaya et al. 1999; Takabayashi et al. 2005).
Some time ago, anatomical characteristics were considered sufficient to consider a plant as C4 type, i.e., having or not a Kranz anatomy structure type. However, in time, it was demonstrated that these characters should not be generalized, and they are insufficient to make absolute classifications. In addition, it was observed that the presence of these structures is not compulsory for a plant to be considered as C4 (Shomer-Ilan et al. 1975).
Moreover, such correlations made between photosynthetic pathway and anatomical support of processes taking place within it led to classifications of different types of foliar anatomy in the Chenopodiaceae.
Carolin et al. (1975) have conducted the first ultrastructural study in the Chenopodiaceae, identifying and describing different anatomical structures; interestingly, some taxonomic correlations between different groups were done, thus suggesting that certain taxonomic reorganizations might be useful.
After 25 years from Carolin’s study, Jacobs (2001) has reviewed in detail the anatomical types of species from Chenopodiaceae, on structural and ultrastructural considerations. In the next paragraphs, only the structural configurations related to C4 photosynthesis will be described (Jacobs 2001):
-
1.
Atriplicoid (C4) (Carolin et al. 1975). The Kranz cells form a parenchymatous sheath around the bundles, except for a gap on the abaxial side, and the mesophyll is arranged radially. The Kranz cell walls are thicker than those of the mesophyll. The Kranz cell chloroplasts generally have more starch grains than the mesophyll chloroplasts, although chloroplasts in both cell types have well-formed grana. The Kranz cells have larger densities of mitochondria between the elongated plastids.
-
2.
Kochioid (C4) (Carolin et al. 1975). The Kranz cells form arcs along the xylem of peripheral bundles. Most leaves have extensive central aqueous tissue. There is a main bundle and several peripheral bundles with the Kranz cells forming a partial parenchymatic sheath. On the main bundle, this parenchymatic sheath is on the adaxial side but often is not present toward the base of the leaf. In Kochia scoparia, the central aqueous tissue is reduced and the lateral bundles opposite to each other are pressed together and the Kranz cells form a partial parenchymatic sheath interrupted laterally. The Kranz cell walls are thicker than those of the mesophyll, and there is no clear increase in the density of mitochondria. The Kranz cell chloroplasts tend to be centripetal and have ill-developed grana (mostly one or two appressions) and well-developed starch grains. The mesophyll cell chloroplasts have well-defined grana and fewer and smaller starch grains than those of the Kranz chloroplasts. There is usually only one layer of mesophyll cells, and in transection, most of these appear to be in contact with a Kranz cell.
-
3.
Salsoloid (C4). Similar to the Kochioid type except that both the Kranz (inner layer) and mesophyll (outer layer) cells tend to form complete layers around the leaf. There is still a peripheral network of vascular tissue associated with the Kranz cells, but the main bundle is more clearly central in position for most of the length of the leaf. The mesophyll is generally one layered, but there is sometimes a hypodermis. The ultrastructure of both cells is much the same as in the Kochioid type with the exception that the Kranz cells have smaller vacuoles in the Salsoloid type than in the Kochioid type.
-
4.
Kranz-Suaedoid (C4). (Carolin et al. 1975). The Kranz cells form a more or less complete layer between the aqueous tissue and the mesophyll cell layer. The vascular tissue forms a network in the lateral longitudinal plane, and there is no peripheral network. The plastids of the Kranz cell tend to be centripetal and have larger starch grains, and larger and more grana, than those of the mesophyll. The Kranz cells also tend to have a large vacuole and a higher density of mitochondria than the mesophyll cells.
-
5.
Kranz-Halosarcoid (C4). (Carolin et al. 1982). There are two distinct layers of chloroplast-containing cells. The cells of the outer layer have small intercellular spaces except close to the stomata where these spaces are considerably larger. The chloroplasts have well-defined but small grana and rarely hold starch grains. There may be isolated groups of non-photosynthetic cells embedded among the chloroplast-containing cells. The cells of the inner layer tend to be more isodiametric, have thicker walls, and have denser mitochondria than the cells of the outer layer. The chloroplasts of the inner cells tend to be centrifugal, with small grana and usually large starch grains. The peripheral vascular bundles are often close to this inner layer of Kranz cells.
In order to simplify things and to make a more operational classification, this classification should be reformulated in fewer words. Actually, four types of anatomical configuration can be maintained: atriplicoid, kochioid, suaedoid, and salsoloid (Gamaley 1985; Fisher et al. 1997; Kadereit et al. 2003; Voznesenskaya et al. 1999; Pyankov et al. 2001; Muhaidat et al. 2007). The four anatomical types are readily discerned on the basis of photosynthetic tissue arrangement with respect to the vascular and other tissues. In the atriplicoid type, the internal chlorenchyma forms a complete (or nearly complete) sheath around vascular bundles. In the kochioid type, internal chlorenchyma cells are confined to the exterior of peripherally positioned veins and do not form a continuous layer. Both the salsoloid and the suaedoid types are characterized by having a continuous stratum of internal chlorenchyma tissue at the periphery of leaves and photosynthetic stems. These two types are distinguished by the position of the vascular bundles in relation to the chlorenchyma. In the salsoloid type, minor vascular bundles are located adjacent to the internal chlorenchyma, while larger vascular bundles are more deeply embedded in water storage parenchyma. In the suaedoid type, all vascular bundles are centrally placed in water storage tissue and none in direct contact with cells of internal chlorenchyma (Gamaley 1985; Muhaidat et al. 2007; Grigore et al. 2012a, b, 2014).
However, it should be mentioned in respect of salsoloid and suaedoid types that sometimes, when analyzing a permanent slide with leaves of halophytic species, these “continuous” chlorenchymatic layers may present interruptions in some points of the cross section (Grigore et al. 2014). Perhaps, this is due rather to a technical procedure or selected level to be sectioned from leaf.
As already stated, Kranz anatomy types were evidenced in halophytes a long time ago by early botanists; of course, they do not nominate them as species related to C4 photosynthesis or belonging to various types of Kranz anatomy, because of limitations imposed by historic period.
For instance, Volkens in his monograph on flora from Egyptian desert mentioned several halophytic species and gave for them respective drawings of microscopic cross sections: Zygophyllum simplex (Fig. 6.1), Anabasis articulata (Fig. 6.2), Atriplex halimus (Fig. 6.3), Tribulus alatus (Fig. 6.4), Bassia muricata (Fig. 6.5), Halogeton alopecuroides (Fig. 6.6), Salsola longifolia (Fig. 6.7), and Haloxylon schweinfurthii (Fig. 6.8). Drawings with cross sections from chenopods species are then included in the chapter with general characters of Chenopodiaceae (1893). However, he did not explain in detail the nature of pictured tissues; on its figures (and the authors’ following) we included the corresponding explanations. When something was questionable, the “?” is given in the figure explanations. It is included especially in respect of possible vascular elements in direct connection with inner chlorenchyma, which would change between salsoloid and suaedoid subtypes (see above paragraphs).
Cross section through the shoot of Anabasis articulata (ep epidermis, ext ch external chlorenchyma, int ch internal chlorenchyma, el v vascular elements? If yes, then the structure would fit with salsoloid sybtype, otherwise to suaedoid) (Volkens 1887)
Cross section through the lamina of Tribulus alatus (ep epidermis, ext ch external chlorenchyma, int ch internal chlorenchyma, v b vascular bundle; atriplicoid sybtype) (Volkens 1887)
Cross section through the lamina of Bassia muricata (ep epidermis, ext ch external chlorenchyma, int ch internal chlorenchyma, v b vascular bundle; kochioid sybtype) (Volkens 1887)
Cross section through the shoot of Halogeton alopecuroides (ep epidermis, ext ch external chlorenchyma, int ch internal chlorenchyma, hp hypodermis?, el v vascular elements? If yes, then the structure would fit with salsoloid sybtype, otherwise to suaedoid) (Volkens 1887)
Cross section through the shoot of Haloxylon schweinfurthii, general view—left side; detail—right side (ep epidermis, ext ch external chlorenchyma, int ch internal chlorenchyma, hp hypodermis?, el v vascular elements? If yes, then the structure would fit with salsoloid sybtype, otherwise to suaedoid) (Volkens 1887)
However, Muhaidat et al. (2007) include Zygophyllum simplex in the kochioid subtype, and their micrograph (and not drawing) is obviously clear. On their image, the vascular bundles are surrounded incompletely by an internal sheath and support the inclusion of this species in the kochioid subtype.
Monteil (1906) in his consistent study on leaf structure in Chenopodiaceae species evidenced Kranz anatomy in many halophytic taxa. Of course, as in the case of botanists of that time, the expression Kranz anatomy was not used yet. However, he clearly speaks about palisade tissue and “endodermic” sheath, tissues that are clearly identified on drawings he included in the study. In addition, a very subtle but interesting observation is made with allusion to this foliar sheath (nominated not so rigorously with the word “endodermic”): “(…) beneath these palisade cells, we found the sheath already reported in all halophytic chenopods.” (p. 114). Therefore, Kranz anatomy is noticeable based on his drawings observations in Atriplex arenaria (Fig. 6.9), Camphorosma monspeliaca (Fig. 6.10), Kirilovia eriantha (Fig. 6.11), Corispermum orientale (Fig. 6.12), Kochia arenaria (Fig. 6.13), K. scoparia (Fig. 6.14), Chenolea muricata Footnote 1 (Fig. 6.15), Echinopsilon hyssopifolia (Fig. 6.16), Suaeda altissima (Fig. 6.17), Salsola soda (Fig. 6.18), S. kali (Fig. 6.19), and S. tragus (Fig. 6.20).
Cross section through the lamina of Atriplex arenaria (ep epidermis, ext ch external chlorenchyma, int ch internal chlorenchyma, v b vascular bundle; atriplicoid sybtype) (Monteil 1906)
Cross section through the lamina of Camphorosma monspeliaca (h hypodermis, ext ch external chlorenchyma, int ch internal chlorenchyma, v b vascular bundle; salsoloid sybtype) (Monteil 1906)
Cross section through the lamina of Kirilovia eriantha (ext ch external chlorenchyma, int ch internal chlorenchyma, v b vascular bundle; atriplicoid sybtype) (Monteil 1906)
Cross section through the lamina of Corispermum orientale (ext ch external chlorenchyma, int ch internal chlorenchyma, v b vascular bundle; atriplicoid sybtype? The internal and external ch. seem not to be in direct contact) (Monteil 1906)
Cross section through the lamina of Kochia arenaria (ep epidermis, ext ch external chlorenchyma, int ch internal chlorenchyma, v b vascular bundle; kochioid sybtype) (Monteil 1906)
Cross section through the lamina of Kochia scoparia (ep epidermis, ext ch external chlorenchyma, int ch internal chlorenchyma, v b vascular bundle; atriplicoid) (Monteil 1906)
Cross section through the lamina of Echinopsilon hyssopifolia (ep epidermis, ext ch external chlorenchyma, int ch internal chlorenchyma, v b vascular bundle; kochioid subtype) (Monteil 1906)
Cross section through the lamina of Suaeda altissima (ep epidermis, ext ch external chlorenchyma, int ch internal chlorenchyma, v b vascular bundle; suaedoid subtype) (Monteil 1906)
Cross section through the lamina of Salsola soda (ep epidermis, h hypodermis, el v vascular elements, ext ch external chlorenchyma; int ch internal chlorenchyma; v (b) vascular bundle; salsoloid subtype) (Monteil 1906)
Cross section through the lamina of Salsola kali (ep epidermis, el v vascular elements, ext ch external chlorenchyma, int ch internal chlorenchyma, v b vascular bundle; salsoloid subtype) (Monteil 1906)
Cross section through the lamina of Salsola tragus (ep epidermis, el v vascular elements, ext ch external chlorenchyma, int ch internal chlorenchyma, v b vascular bundle; salsoloid subtype) (Monteil 1906)
Chermezon (1910) identified a Kranz anatomy structure in leaf of Atriplex crassifolia (Fig. 6.21); he also made a very important observation, related to the disposition of chlorenchymatic tissues that later will be explicitly designated as Kranz anatomy. Thus, he refers to its foliar anatomy:”(leaf) is different especially by the clearly radial disposition of palisade tissue around chlorophyll sheath of nervures” (p. 236).
Cross section through the lamina of Atriplex crassifolia (ep epidermis, ext ch external chlorenchyma, int ch internal chlorenchyma, v b vascular bundle; atriplicoid subtype) (Chermezon 1910)
In Warming’s (1897) drawings about anatomical structure of halophytes, Kranz anatomy can be noticeable in Haloxylon ammodendron (Fig. 3.39—Succulence chapter—salsoloid type) and in Atriplex farinosa (Fig. 6.22, atriplicoid subtype).
Cross section through the lamina of Atriplex farinosa (ep epidermis, ext ch external chlorenchyma, int ch internal chlorenchyma, v b vascular bundle; atriplicoid subtype; right below—general view of Kranz anatomy) (Warming 1897)
Paulsen (1912) evidenced Kranz anatomy structures in several halophytic species: Anabasis eriopoda (Fig. 6.23), Salsola arbuscula (Fig. 6.24), Horaninowia ulicina (Fig. 6.25), and Suaeda lipskii (Fig. 6.26).
Anabasis eriopoda—Cross section through the stem (b, c) (ep epidermis, ext ch external chlorenchyma, int ch internal chlorenchyma, wt t water storage tissue, C epidermis) (Paulsen 1912)
Salsola arbuscula—Cross section through the lamina (ep epidermis, ext ch external chlorenchyma, int ch internal chlorenchyma, wt t water storage tissue) (Paulsen 1912)
Horaninowia ulicina—Cross section through the lamina (a, general view and b, detail) and stem (c) (ep epidermis, ext ch external chlorenchyma, int ch internal chlorenchyma, wt t water storage tissue) (Paulsen 1912)
Suaeda lipskii—Cross section through the lamina (ep epidermis; ext ch external chlorenchyma, int ch internal chlorenchyma) (Paulsen 1912)
Gamaley (1985) in his study about Kranz variations in plants from Gobi and Karakum deserts evidenced it in many chenopods (Bassia hyssopifolia, Atriplex sibirica, Salsola collina, and Suaeda arcuata).
Our studies in Romanian (Grigore and Toma 2007, 2008; Grigore et al. 2011, 2012a, b), Spanish (Grigore et al. 2011; 2014), and Iranian halophytes (Safiallah et al. 2017) revealed Kranz anatomy in many Chenopodiaceae species: Atriplex tatarica (Figs. 6.27 and 6.28), A. glauca (Fig. 6.29), A. halimus (Figs. 6.30 and 6.31), Petrosimonia oppositifolia (Fig. 6.32), P. triandra (Fig. 6.33), Camphorosma annua (Fig. 6.34), C. monspeliaca (Fig. 6.35), Suaeda splendens (Fig. 6.36), Salsola kali (Fig. 6.37), S. oppositifolia (Fig. 6.38), and Bassia hyssopifolia (Fig. 6.39).
Kranz anatomy has been also evidenced in several halophytic Iranian species of Bassia: B. prostrata (Fig. 6.40), B. pilosa (Fig. 6.41), and B. turkestanica (Fig. 6.42) (photo courtesy of Somayeh Safiallah).
The atriplicoid subtype found by us in Atriplex tatarica was also evidenced by Jacobs (2001) and Muhaidat et al. (2007) and can be found in other species of Atriplex: A. lampa (Pyykkö 1966) and A. buchananii (Troughton and Card 1974).
As we already emphasized, the cross sections should be analyzed carefully, because the continuity/discontinuity of chlorenchyma layers imposes the appurtenance to a subtype or other. For this reason, what we previously considered in Petrosimonia species as belonging to kochioid sybtype—because the chlorenchyma layers seemed on analyzed slides as discontinuous—belong in fact to salsoloid sybtype (Grigore et al. 2014).
The same is true for Camphorosma species, which belongs to salsoloid subtype too.
Muhaidat et al. (2007) reviewed the structural diversity of Kranz anatomy in C4 eudicots. The atriplicoid subtype species has been found in A. rosea and Atriplex polycarpa; kochioid subtype was found in Kochia scoparia and Zygophyllum simplex. Salsoloid subtype was evidenced in Salsola komarovii and suaedoid subtype, in Suaeda vermiculata.
It has been shown that aridity and salinity are important factors promoting stomatal closure and thus reduce intercellular CO2 levels, stimulating photorespiration and aggravating a CO2 substrate deficiency (Guy et al. 1980; Adam 1990). Together, the combination of drought, increased salinity, low humidity, and high temperature produces the greatest potential for photorespiration and CO2 deficiency (Ehleringer and Monson 1993). In addition, drought or salinity stresses further increase CO2 compensation points, because lower stomatal conductance and photosynthetic capacity reduce carbon income, allowing respiration to consume proportionally more of carbon acquired by the plant (Sage 2004).
Evolutionarily speaking, it seems like anatomical modifications (Kranz type) represented a preconditioning step in occurrence of this photosynthetic type (Sage 2004); to evolve an effective CO2 concentration mechanism, the distance between mesophyll and bundle sheath cells has to decline to allow for rapid diffusion of metabolites (Raghavendra 1980; Ehleringer et al. 1997).
Even with all exposed data at our disposal, it is still difficult to find a direct correlation between salinity factor and Kranz anatomy structures. All investigated species by us are xero-halophytes and obligatory halophytes, excepting A. tatarica. In its native distribution area of Middle and western part of Central Asia, this species occupies solonetz sandy and clayey banks of rivers and lakes, coastal solonchaks, and solonetz alluvial trails and is frequently found as a weed in roadside ditches and in villages (Kochánková and Mandák 2008).
C4 species form a particularly high proportion of the herbaceous flora of saline environments, even in cool temperate regions (Long and Mason 1983). Apparently, the inherently higher water use efficiency of C4 species would have two theoretical advantages in saline environments (Long 1999). First, saline soils have a soil water potential of around—2.5 MPa; to extract water, the halophytes must generate a lower water potential, even though this exceeds limits that can apparently be tolerated by many mesophytic vascular plants. Transpiration must be minimal, and the higher water use efficiency of C4 species would confer the advantage of maximizing carbon gain per unit of water lost. Second, plant mineral content is inversely correlated to water use efficiency as an assumed result of increased passive uptake with increased transpiration. For a halophyte, increased transpiration increases the energy needed to exclude Na+ and Cl− (Long and Mason 1983).
It has been suggested that halophytes are, in fact, a special case among xerophytes (Wiessner 1899; Henslow 1895; Schimper 1903; Kearney 1904; Warming 1909; Clements 1920; McDougall 1941; Grigore and Toma 2010). This implies the occurrence of some mechanisms serving to protect the water reserves of the plant in periods of drought or high potential evapotranspiration when soil water potential falls. A cost of xeromorphy is increased resistance to diffusion of CO2 to the mesophyll; because of the low leaf intercellular pressure, necessary to saturate C4 photosynthesis, this cost is minimized in C4 species.
Despite the fact that C4 species represent only about 8000 of the estimated 250,000–300,000 land plants species (Sage et al. 1999), they are major components of biomes that cover more than 35% of the earth’s land surface area. These species are dominant in tropical and subtropical grassland and savanna, warm temperate grassland and savanna, arid steppe, beach dunes, salt marshes, salt desert, hot deserts, and semideserts.
C4 also represents an important ecological strategy in certain desert shrubs, most notably species of Atriplex, particularly in saline soils (Keeley and Rundel 2003). In these species, the key adaptation is the ability to maintain growth under high summer temperatures and drought conditions at a time when C3 species are dormant. The maximal rates of photosynthesis in these desert C4 species are generally no higher than that of concurring C3 species, but the water use efficiency is far greater. In addition, C4 plants have higher nitrogen use efficiency.
Some studies certify the close relationship between C4 photosynthesis and extreme habitats, such as deserts and salinized areas. Thus, Wang (2007) identified among species vegetating in the deserts of China that 36.5% of the Chenopodiaceae species were found with C4 photosynthesis, which was about 48% of the total C4 species. These taxa were predominantly members of the genera Anabasis, Atriplex, Kochia, Salsola, and Suaeda.
Other studies sustain the facts mentioned above: there is a close relationship between some special morphotypes and respective photosynthetic type. In an ecological work, it was observed that halophytes and xerophytes with articulated stems and stem succulents of Anabasis type are exclusively C4. Leaf succulent halophytes and xerophytes are also predominantly C4 (Akhani et al. 1997).
Additional results obtained by Pyankov et al. (2000) referring to C4 plants from Mongolia also suggest the relevance of this photosynthetic pathway on plants growing in extreme environmental conditions. The Chenopodiaceae comprises the greatest number of C4 plants (about 41 species). Additionally, the C4 Chenopodiaceae make up 45% of the total chenopods and are very important ecologically in saline areas and cold arid deserts. NADP-ME tree-like species with a salsoloid type of Kranz anatomy, such as Haloxylon ammodendron and Iljinia regelii, plus shrubby Salsola and Anabasis species, were the plant most resistant to environmental stresses. Most of the annual C4 chenopods species are halophytes, are succulent, and occur in saline and arid habitats.
Notes
- 1.
A serious problem is that related to taxonomical nomenclature. Chenopods species have many synonyms, used by various botanists. For instance, this is synonymous with Bassia muricata, and Echinopsilon hyssopifolia, with Bassia hyssopifolia. However, sometimes slight differences occur between drawings of the same species given by different botanists (see next paragraphs).
References
Adam P (1990) Saltmarsh ecology. Cambridge University Press, Cambridge
Akhani H, Trimborn P, Ziegler H (1997) Photosynthetic pathways in Chenopodiaceae from Africa, Asia and Europe with their ecological, phytogeographical and taxonomical importance. Plant Syst Evol 206(1–4):187–221
Arcanheli G (1890) Sulla struttura delle foglie dell’ Atriplex nummularia Lind. in relazione alla assimilazione. Nuova giorn Ital 22:426–430
Chermezon H (1910) Recherches anatomiques sur les plantes littorales. Ann Sci Nat sér 9 Bot 12:117–313
Carolin RC, Jacobs SWL, Vesk M (1975) Leaf structure in Chenopodiaceae. Bot Jahr Syst Pflanzengeschichte and Pflanyengeographie 95:226–255
Carolin RC, Jacobs SWL, Vesk M (1982) The chlorenchyma of some members of the Salicornieae (Chenopodiaceae). Aust J Bot 30:387–392
Clements FE (1920) Plant indicators: the relation of plant communities to process and practice. Carnegie Institution, Washington
Dengler NG, Nelson T (1999) Leaf structure and development in C4 plants. In: Sage RF, Monson RK (eds) C4 plant biology. Academic, San Diego, pp 133–172
Ehleringer JR, Monson RK (1993) Evolutionary and ecological aspects of photosynthetic pathway variation. Ann Rev Ecol Syst 24:411–439
Ehleringer JR, Cerling TE, Helliker BR (1997) C4 photosynthesis, atmospheric CO2 and climate. Oecologia 112:285–299
Fisher DD, Schenk HJ, Thorsch JA, Ferren WR (1997) Leaf anatomy and subgeneric affiliations of C3 and C4 species of Suaeda (Chenopodiaceae) in North America. Am J Bot 84:1198–1210
Gamaley IB (1985) Variaţii kranţ—anatomii u rastenii pustyni Gobi i Karakumi (The variations of the Kranz-anatomy in Gobi and Karakum plants). Bot Jurn SSSR 70:1302–1314
Grigore M-N (2008) Introducere în Halofitologie. Elemente de Anatomie Integrativă. PIM, Iaşi
Grigore M-N, Toma C (2007) Histo—anatomical strategies of Chenopodiaceae halophytes: adaptive, ecological and evolutionary implications. WSEAS Trans on Biol and Biomed 4:204–218
Grigore M-N, Toma C (2008) Ecological anatomy of halophyte species from the Chenopodiaceae family. Advanced topics on mathematical biology and ecology (Proceedings of the 4th WSEAS International Conference on Mathematical Biology and Ecology—MABE ’08, Acapulco, Mexico, January 25–27, 2008), pp 62–67
Grigore M-N, Toma C (2010) Halofitele. Aspecte de anatomie ecologică. Edit. Univ. “Al. I. Cuza”, Iaşi
Grigore M-N, Toma C, Ivănescu L (2011) Anatomical and ecological observations on Mediterranean halophytes: Suaeda Forssk. ex Scop. genus. Lucr. Şt. (Horticultură). USAMV “Ion Ionescu de la Brad”, Iaşi 54(1):23–28
Grigore M-N, Toma C, Zamfirache M-M, Boscaiu M, Olteanu Z, Cojocaru D (2012a) Ecological anatomy in halophytes with C4 photosynthesis: discussing adaptative features in endangered ecosystems. Carpathian J of Earth and Environ Sci 7(2):13–21
Grigore M-N, Toma C, Zamfirache M-M, Ivănescu L (2012b) A survey of anatomical adaptations in Romanian halophytes. Towards an ecological interpretation. Fres Environ Bull 21(11b):3370–3375
Grigore M-N, Ivănescu L, Toma C (2014) Halophytes: an integrative anatomical study. Springer, Cham, Heidelberg
Guttierez M, Gracen VF, Edwards GE (1974) Biochemical and cytological relationships in C4 plants. Planta 119:279–300
Guy RD, Reid DM, Krouse HR (1980) Shifts in carbon isotope ratios of two C3 halophytes under natural and artificial conditions. Oecologia 44:241–247
Hattersley PW, Browning AJ (1981) Occurrence of the suberized lamella in leaves of grasses of different photosynthetic types. I. In parenchymatous bundle sheats and PCR (“Kranz”) sheaths. Protoplasma 109:371–401
Henslow G (1895) The origin of plant-structures by self-adaptation to the environment. Kegan Paul, Trench, Trübner & Co, Ltd, Paternoster House, Charing Cross Road, London
Jacobs SWL (2001) Review of leaf anatomy and ultrastructure in the Chenopodiaceae (Caryophyllales). J Torrey Bot Soc 128:236–253
Kadereit G, Borsch T, Weising K, Freitag H (2003) Phylogeny of Amaranthaceae and Chenopodiaceae and the evolution of C4 photosynthesis. Int J Plant Sci 164(6):959–986
Kanai R, Edwards GE (1999) The biochemistry of C4 photosynthesis. In: Sage RF, Monson RK (eds) C4 plant biology. Academic, San Diego, pp 59–87
Kearney TH (1904) Are plants of sea and dunes true halophytes? Bot Gaz 37:424–436
Keeley JE, Rundel OW (2003) Evolution of CAM and C4 carbon-concentrating mechanisms. Int J Plant Sci 164(3 Suppl):55–77
Kellog EA (1999) Phylogenetic aspects of the evolution of C4 photosynthesis. In: Sage RF, Monson RK (eds) C4 Plant biology. Academic, San Diego, pp 411–444
Kochánková J, Mandák B (2008) Biological flora of Central Europe: Atriplex tatarica L. Perspect Plant Ecol Evol System 10:217–229
Laetsch WM (1968) Chloroplast specialization in dicotyledons possessing the C4—dicarboxylic acid pathway of photosynthetic CO2 fixation. Am J Bot 55:875–883
Long SP (1999) Environmental responses. In: Sage RF, Monson RK (eds) C4 plant biology. Academic, San Diego, pp 215–249
Long SP, Mason CF (1983) Saltmarsh ecology. Blackie, Glasgow
Mcdougall WB (1941) Plant ecology, 3rd edn. Lea & Febiger, Philadelphia
Monteil P (1906) Anatomie compareé de la feuille des Chenopodiacees. Travaux de Laboratoire de Matiere Medicale de l’École Supérieure de Pharmacie de Paris 4:5–156
Moser H (1934) Untersuchungen űber die Blattstruktur von Atriplex Arten und ihre Beziehungen zur Systematic. Beih Bot Centralbl 52:378–388
Muhaidat R, Sage RF, Dengler NG (2007) Diversity of Kranz anatomy and biochemistry in C4 eudicots. Am J Bot 94(3):362–381
Paulsen O (1912) Studies on the vegetation of the Transcaspian lowlands. The second Danish Pamir expedition conducted by Olufsen O, Copenhagen, Gyldendalske Boghandel, Nordisk Forlag
Pyankov V, Artyusheva EG, Edwards GE, Black CC Jr, Soltis PI (2001) Phylogenetic analysis of tribe Salsoleae (Chenopodiaceae), based on ribosomal ITS sequences: implications for the evolution of photosynthesis types. Am J Bot 88(7):1189–1198
Pyankov VI, Gunin PD, Tsoog S, Black CC (2000) C4 plants in the vegetation of Mongolia: their natural occurrence and geographical distribution in relation to climate. Oecologia 123(1):15–31
Pyykkö M (1966) The leaf anatomy of East Patagonian xeromorphic plants. Ann Bot Fennici 3(3):453–622
Raghavendra AS (1980) Characteristics of plant species intermediate between C3 and C4 pathways of photosynthesis: their focus of mechanism and evolution of C4 syndrome. Photosynthetica 14:271–173
Sage RF (2001) Environmental and evolutionary preconditions for the origin and diversification of C4 photosynthesis syndrome. Plant Biol 3:202–213
Sage RF (2004) The evolution of C4 photosynthesis. New Phytol 161:341–370
Sage RF, Wedin DA, Li M (1999) The biogeography of C4 photosynthesis: patterns and controlling factors. In: Sage RF, Monson RK (eds) C4 plant biology. Academic, San Diego, pp 313–373
Schimper AFW (1903) Plant geography upon a physiological basis. Clarendon, Oxford
Shomer-Ilan A, Beer S, Waisel Y (1975) Suaeda monoica, a C4 plant without typical bundle sheats. Plant Physiol 56:676–679
Safiallah S, Hamdi SMM, Grigore M-N, Sara J (2017) Micromorphology and leaf ecological anatomy of Bassia halophyte species (Amaranthaceae) from Iran. Acta Biologica Szegediensis 61(1):85–93
Takabayashi A, Kishine M, Asada K, Endo T, Sato F (2005) Differential use of two cyclic electron flows around photosystem I for driving CO2-concentration mechanism in C4 photosynthesis. Proc Natl Acad Sci USA 102(46):16898–16903
Troughton JH, Card KA (1974) Leaf anatomy of Atriplex buchananii. New Zeal J Bot 12:167–177
Ueno O, Yoshimura Y, Sentoku N (2005) Variation in the activity of some enzymes of photorespiratory metabolism in C4 grases. Ann Bot 96:863–869
Volkens G (1887) Die Flora der aegyptisch-arabischen Wüste auf Grundlage anatomisch-physiologischer Forschungen. Gebrüder, Borntraeger, Berlin
Volkens G (1893) Chenopodiaceae. In: Engler A, Prantl K (eds) Die Natürlichen Pflanzenfamilien, 3(1a): 36-91
Voznesenskaya EV, Franceschi VR, Pyankov VI, Edwards GE (1999) Anatomy, chloroplast structure and compartmentation of enzymes relative to photosynthetic mechanisms in leaves and cotyledons of species in the tribe Salsoleae (Chenopodiaceae). J Exp Bot 50(341):1779–1795
Wang RZ (2007) C4 plants in the deserts of China: occurrence of C4 photosynthesis and its morphological functional types. Photosynthetica 45(2):167–171
Warming E (1897) Halophyt-studier. D Kgl Danske Vidensk Selsk Skr 6, Raekke, naturvidenskabeling og mathematisk Afd. VIII 4:173–272
Warming E (1909) Oecology of Plants. An introduction to the study of plant-communities. Clarendon, Oxford
Wiessner J (1899) Uber die Formen der Anpassung der Blatter an die Lichtstarke. Biol Centralbl 19:1–14
Yoshimura Y, Kubota F, Ueno O (2004) Structural and biochemical bases of photorespiration in C4 plants: quantification of organelles and glycine decarboxylase. Planta 220:307–317
Author information
Authors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing AG
About this chapter
Cite this chapter
Grigore, MN., Toma, C. (2017). Kranz Anatomy. In: Anatomical Adaptations of Halophytes. Springer, Cham. https://doi.org/10.1007/978-3-319-66480-4_6
Download citation
DOI: https://doi.org/10.1007/978-3-319-66480-4_6
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-66479-8
Online ISBN: 978-3-319-66480-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)