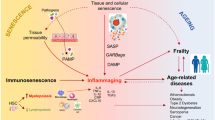
Abstract
Older people display increasing rates and severity of infections and reduced vaccine responses. The innate immune system is the first line of defense that protects the host from invading pathogens. Recognition of microbial components by Toll-Like Receptor (TLR)-dependent activation of antigen-presenting cells, such as dendritic cells, macrophages, or monocytes, is a crucial step for the ensuing initiation of the adaptive immune response. Several studies suggest that cytokine production upon stimulation by TLR ligands are affected in older individuals. Herein we discuss the different mechanisms that could account for the alterations of TLR-responsiveness in the geriatric population, focusing on the clinical characteristics of this age group. We suggest that frailty-associated factors, such as malnutrition, dysbiosis, and neurohormonal changes, could limit cytokine production by myeloid cells thereby contributing to immunosenescence.
Similar content being viewed by others
References
Agrawal A et al (2007a) Altered innate immune functioning of dendritic cells in elderly humans: a role of phosphoinositide 3-kinase-signaling pathway. J Immunol 1950(178):6912–6922
Agrawal A, Agrawal S, Gupta S (2007b) Dendritic cells in human aging. Exp Gerontol 42:421–426
Ahluwalia N et al (2001) Cytokine production by stimulated mononuclear cells did not change with aging in apparently healthy, well-nourished women. Mech Ageing Dev 122:1269–1279
Akgun KM, Crothers K, Pisani M (2012) Epidemiology and management of common pulmonary diseases in older persons. J Gerontol A Biol Sci Med Sci 67:276–291
Anpalahan M, Gibson SJ (2008) Geriatric syndromes as predictors of adverse outcomes of hospitalization. Intern Med J 38:16–23
Arunachalam K, Gill HS, Chandra RK (2000) Enhancement of natural immune function by dietary consumption of Bifidobacterium lactis (HN019). Eur J Clin Nutr 54:263–267
Asha NJ, Tompkins D, Wilcox MH (2006) Comparative analysis of prevalence, risk factors, and molecular epidemiology of antibiotic-associated diarrhea due to Clostridium difficile, Clostridium perfringens, and Staphylococcus aureus. J Clin Microbiol 44:2785–2791
Bakker RC, Osse RJ, Tulen JHM, Kappetein AP, Bogers AJJC (2012) Preoperative and operative predictors of delirium after cardiac surgery in elderly patients. Eur J Cardio-Thorac Surg 41:544–549
Beasley JM et al (2010) Protein intake and incident frailty in the Women’s health initiative observational study. J Am Geriatr Soc 58:1063–1071
Boge T et al (2009) A probiotic fermented dairy drink improves antibody response to influenza vaccination in the elderly in two randomised controlled trials. Vaccine 27:5677–5684
Brewster DR, Manary MJ, Menzies IS, O’Loughlin EV, Henry RL (1997) Intestinal permeability in kwashiorkor. Arch Dis Child 76:236–241
Bringiotti R et al (2014) Intestinal microbiota: the explosive mixture at the origin of inflammatory bowel disease? World J Gastrointest Pathophysiol 5:550–559
Britanova OV et al (2014) Age-related decrease in TCR repertoire diversity measured with deep and normalized sequence profiling. J Immunol 1950(192):2689–2698
Canaday DH et al (2010) Influenza-induced production of interferon-alpha is defective in geriatric individuals. J Clin Immunol 30:373–383
Cani PD et al (2007) Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes 56:1761–1772
Chang SS, Weiss CO, Xue Q-L, Fried LP (2012) Association between inflammatory-related disease burden and frailty: results from the Women’s health and aging studies (WHAS) I and II. Arch Gerontol Geriatr 54:9–15
Ciaramella A et al (2011) Effect of age on surface molecules and cytokine expression in human dendritic cells. Cell Immunol 269:82–89
Clegg A, Young J, Iliffe S, Rikkert MO, Rockwood K (2013) Frailty in elderly people. Lancet 381:752–762
Colonna-Romano G et al (2009) A double-negative (IgD-CD27-) B cell population is increased in the peripheral blood of elderly people. Mech Ageing Dev 130:681–690
Colonna-Romano G et al (2010) B cells compartment in centenarian offspring and old people. Curr Pharm Des 16:604–608
Compte N et al (2013a) Increased basal and alum-induced interleukin-6 levels in geriatric patients are associated with cardiovascular morbidity. PLoS One 8:e81911
Compte N et al (2013b) Frailty in old age is associated with decreased interleukin-12/23 production in response to toll-like receptor ligation. PLoS One 8:e65325
Compte N, Bailly B, De Breucker S, Goriely S, Pepersack T (2015) Study of the association of total and differential white blood cell counts with geriatric conditions, cardio-vascular diseases, seric IL-6 levels and telomere length. Exp Gerontol 61:105–112
Cros J et al (2010) Human CD14dim monocytes patrol and sense nucleic acids and viruses via TLR7 and TLR8 receptors. Immunity 33:375–386
Cullum S, Metcalfe C, Todd C, Brayne C (2008) Does depression predict adverse outcomes for older medical inpatients? A prospective cohort study of individuals screened for a trial. Age Ageing 37:690–695
de Vos AF et al (2009) In vivo lipopolysaccharide exposure of human blood leukocytes induces cross-tolerance to multiple TLR ligands. J Immunol 1950(183):533–542
Della Bella S et al (2007) Peripheral blood dendritic cells and monocytes are differently regulated in the elderly. Clin Immunol 122:220–228
Derhovanessian E et al (2010) Hallmark features of immunosenescence are absent in familial longevity. J Immunol 1950(185):4618–4624
Ershler WB (2001) The value of the SENIEUR protocol: distinction between ‘ideal aging’ and clinical reality. Mech Ageing Dev 122:134–136
Ewig S et al (2012) Nursing-home-acquired pneumonia in Germany: an 8-year prospective multicentre study. Thorax 67:132–138
Feldblum I et al (2007) Characteristics of undernourished older medical patients and the identification of predictors for undernutrition status. Nutr J 6:37
Ferrucci L et al (2005) The origins of age-related proinflammatory state. Blood 105:2294–2299
Franceschi C et al (2000) Inflamm-aging. An evolutionary perspective on immunosenescence. Ann N Y Acad Sci 908:244–254
Freeman SL et al (2010) Dairy proteins and the response to pneumovax in senior citizens: a randomized, double-blind, placebo-controlled pilot study. Ann N Y Acad Sci 1190:97–103
Fried LP et al (2001) Frailty in older adults: evidence for a phenotype. J Gerontol A Biol Sci Med Sci 56:M146–M156
Fried LP, Ferrucci L, Darer J, Williamson JD, Anderson G (2004) Untangling the concepts of disability, frailty, and comorbidity: implications for improved targeting and care. J Gerontol A Biol Sci Med Sci 59:255–263
Fukushima Y et al (2007) Improvement of nutritional status and incidence of infection in hospitalised, enterally fed elderly by feeding of fermented milk containing probiotic Lactobacillus johnsonii La1 (NCC533). Br J Nutr 98:969–977
Fulop T et al (2010) Aging, frailty and age-related diseases. Biogerontology 11:547–563
Fulop T, Larbi A, Pawelec G (2013) Human T cell aging and the impact of persistent viral infections. Front Immunol 4:271
Gabriel P, Cakman I, Rink L (2002) Overproduction of monokines by leukocytes after stimulation with lipopolysaccharide in the elderly. Exp Gerontol 37:235–247
Gibson KL et al (2009) B-cell diversity decreases in old age and is correlated with poor health status. Aging Cell 8:18–25
Hamza SA et al (2012) Immune response of 23-valent pneumococcal polysaccharide vaccinated elderly and its relation to frailty indices, nutritional status, and serum zinc levels. Geriatr Gerontol Int 12:223–229
Hearps AC et al (2012) Aging is associated with chronic innate immune activation and dysregulation of monocyte phenotype and function. Aging Cell 11:867–875
Hotamisligil GS (2010) Endoplasmic reticulum stress and atherosclerosis. Nat Med 16:396–399
Ichinohe T et al (2011) Microbiota regulates immune defense against respiratory tract influenza A virus infection. Proc Natl Acad Sci USA 108:5354–5359
Inouye SK, Studenski S, Tinetti ME, Kuchel GA (2007) Geriatric syndromes: clinical, research, and policy implications of a core geriatric concept. J Am Geriatr Soc 55:780–791
Jing Y et al (2009) Aging is associated with a numerical and functional decline in plasmacytoid dendritic cells, whereas myeloid dendritic cells are relatively unaltered in human peripheral blood. Hum Immunol 70:777–784
Kagansky N et al (2005) Poor nutritional habits are predictors of poor outcome in very old hospitalized patients. Am J Clin Nutr 82:784–791.-; quiz 913-4
Kong K-F et al (2008) Dysregulation of TLR3 impairs the innate immune response to West Nile virus in the elderly. J Virol 82:7613–7623
Langkamp-Henken B et al (2006) Nutritional formula improved immune profiles of seniors living in nursing homes. J Am Geriatr Soc 54:1861–1870
Ligthart GH (2001) The SENIEUR protocol after 16 years: the next step is to study the interaction of ageing and disease. Mech Ageing Dev 122:136–140
Ligthart GJ et al (1984) Admission criteria for immunogerontological studies in man: the SENIEUR protocol. Mech Ageing Dev 28:47–55
Liman TG et al (2012) Impact of low mini-mental status on health outcome up to 5 years after stroke: the Erlangen stroke project. J Neurol 259:1125–1130
Lung TL, Saurwein-Teissl M, Parson W, Schonitzer D, Grubeck-Loebenstein B (2000) Unimpaired dendritic cells can be derived from monocytes in old age and can mobilize residual function in senescent T cells. Vaccine 18:1606–1612
Manzur A et al (2011) Influence of prior pneumococcal and influenza vaccination on outcomes of older adults with community-acquired pneumonia. J Am Geriatr Soc 59:1711–1716
Metcalf TU et al (2015) Global analyses revealed age-related alterations in innate immune responses after stimulation of pathogen recognition receptors. Aging Cell 14:421–432
Mysliwska J, Bryl E, Foerster J, Mysliwski A (1998) Increase of interleukin 6 and decrease of interleukin 2 production during the ageing process are influenced by the health status. Mech Ageing Dev 100:313–328
Naylor K et al (2005) The influence of age on T cell generation and TCR diversity. J Immunol 1950(174):7446–7452
Nilsson B-O et al (2003) Morbidity does not influence the T-cell immune risk phenotype in the elderly: findings in the Swedish NONA immune study using sample selection protocols. Mech Ageing Dev 124:469–476
Nyugen J, Agrawal S, Gollapudi S, Gupta S (2010) Impaired functions of peripheral blood monocyte subpopulations in aged humans. J Clin Immunol 30:806–813
Oliveira MRM, Fogaca KCP, Leandro-Merhi VA (2009) Nutritional status and functional capacity of hospitalized elderly. Nutr J 8:54
Ongradi J, Kovesdi V (2010) Factors that may impact on immunosenescence: an appraisal. Immun Ageing A 7:7
Panda A et al (2010) Age-associated decrease in TLR function in primary human dendritic cells predicts influenza vaccine response. J Immunol 1950(184):2518–2527
Pawelec G et al (2005) Human immunosenescence: is it infectious? Immunol Rev 205:257–268
Pepersack T, Turneer M, De Breucker S, Stubbe M, Beyer I (2005) Tetanus immunization among geriatric hospitalized patients. Eur J Clin Microbiol Infect Dis 24:495–496
Poynter L, Kwan J, Sayer AA, Vassallo M (2011) Does cognitive impairment affect rehabilitation outcome? J Am Geriatr Soc 59:2108–2111
Pratt RH, Winston CA, Kammerer JS, Armstrong LR (2011) Tuberculosis in older adults in the United States, 1993–2008. J Am Geriatr Soc 59:851–857
Pussinen PJ, Havulinna AS, Lehto M, Sundvall J, Salomaa V (2011) Endotoxemia is associated with an increased risk of incident diabetes. Diabetes Care 34:392–397
Qian C et al (2006) TLR agonists promote ERK-mediated preferential IL-10 production of regulatory dendritic cells (diffDCs), leading to NK-cell activation. Blood 108:2307–2315
Rajagopalan S (2001) Tuberculosis and aging: a global health problem. Clin Infect Dis 33:1034–1039
Reichert TA et al (2004) Influenza and the winter increase in mortality in the United States, 1959–1999. Am J Epidemiol 160:492–502
Reis e Sousa C (2004) Toll-like receptors and dendritic cells: for whom the bug tolls. Semin. Immunol 16:27–34
Ridda I et al (2009) Immunological responses to pneumococcal vaccine in frail older people. Vaccine 27:1628–1636
Sadeghi HM, Schnelle JF, Thoma JK, Nishanian P, Fahey JL (1999) Phenotypic and functional characteristics of circulating monocytes of elderly persons. Exp Gerontol 34:959–970
Sagawa M, Kojimahara N, Otsuka N, Kimura M, Yamaguchi N (2011) Immune response to influenza vaccine in the elderly: association with nutritional and physical status. Geriatr Gerontol Int 11:63–68
Sansoni P et al (2014) New advances in CMV and immunosenescence. Exp Gerontol 55:54–62
Saurwein-Teissl M, Schonitzer D, Grubeck-Loebenstein B (1998) Dendritic cell responsiveness to stimulation with influenza vaccine is unimpaired in old age. Exp Gerontol 33:625–631
Schiffrin EJ, Morley JE, Donnet-Hughes A, Guigoz Y (2010) The inflammatory status of the elderly: the intestinal contribution. Mutat Res 690:50–56
Schmader KE et al (2010) Effect of a zoster vaccine on herpes zoster-related interference with functional status and health-related quality-of-life measures in older adults. J Am Geriatr Soc 58:1634–1641
Schreibelt G et al (2010) Toll-like receptor expression and function in human dendritic cell subsets: implications for dendritic cell-based anti-cancer immunotherapy. Cancer Immunol Immunother 59:1573–1582
Seidler S, Zimmermann HW, Bartneck M, Trautwein C, Tacke F (2010) Age-dependent alterations of monocyte subsets and monocyte-related chemokine pathways in healthy adults. BMC Immunol 11:30
Shaw AC et al (2011) Dysregulation of human toll-like receptor function in aging. Ageing Res Rev 10:346–353
Shaw AC, Goldstein DR, Montgomery RR (2013) Age-dependent dysregulation of innate immunity. Nat Rev Immunol 13:875–887
Shodell M, Siegal FP (2002) Circulating, interferon-producing plasmacytoid dendritic cells decline during human ageing. Scand J Immunol 56:518–521
Shu Z, Ma J, Tuerhong D, Yang C, Upur H (2013) How intestinal bacteria can promote HIV replication. AIDS Rev 15:32–37
Sridharan A et al (2011) Age-associated impaired plasmacytoid dendritic cell functions lead to decreased CD4 and CD8 T cell immunity. Age (Dordr) 33:363–376
Steger MM, Maczek C, Grubeck-Loebenstein B (1996) Morphologically and functionally intact dendritic cells can be derived from the peripheral blood of aged individuals. Clin Exp Immunol 105:544–550
Strindhall J et al (2007) No immune risk profile among individuals who reach 100 years of age: findings from the Swedish NONA immune longitudinal study. Exp Gerontol 42:753–761
Strindhall J et al (2013) The inverted CD4/CD8 ratio and associated parameters in 66-year-old individuals: the Swedish HEXA immune study. Age (Dordr) 35:985–991
Suzuki T (2013) Regulation of intestinal epithelial permeability by tight junctions. Cell Mol Life Sci 70:631–659
Thompson WW et al (2003) Mortality associated with influenza and respiratory syncytial virus in the United States. JAMA 289:179–186
Tsai AC, Lee L-C, Wang J-Y (2013) Complementarity of the mini-nutritional assessment and activities of daily living for predicting follow-up mortality risk in elderly Taiwanese. Br J Nutr 109:658–666
van Duin D, Shaw AC (2007) Toll-like receptors in older adults. J Am Geriatr Soc 55:1438–1444
van Duin D et al (2007a) Prevaccine determination of the expression of costimulatory B7 molecules in activated monocytes predicts influenza vaccine responses in young and older adults. J Infect Dis 195:1590–1597
van Duin D et al (2007b) Age-associated defect in human TLR-1/2 function. J Immunol 1950(178):970–975
Verschoor CP et al (2014a) Alterations to the frequency and function of peripheral blood monocytes and associations with chronic disease in the advanced-age, frail elderly. PLoS One 9:e104522
Verschoor CP, Johnstone J, Loeb M, Bramson JL, Bowdish DME (2014b) Anti-pneumococcal deficits of monocyte-derived macrophages from the advanced-age, frail elderly and related impairments in PI3K-AKT signaling. Hum Immunol 75:1192–1196
Walston J et al (2006) Research agenda for frailty in older adults: toward a better understanding of physiology and etiology: summary from the American Geriatrics Society/National Institute on Aging research conference on frailty in older adults. J Am Geriatr Soc 54:991–1001
Wikby A et al (2006) The immune risk phenotype is associated with IL-6 in the terminal decline stage: findings from the Swedish NONA immune longitudinal study of very late life functioning. Mech Ageing Dev 127:695–704
Wu J et al (2011) Age-dependent alterations of HLA-DR expression and effect of lipopolysaccharide on cytokine secretion of peripheral blood mononuclear cells in the elderly population. Scand J Immunol 74:603–608
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Section Editor information
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG
About this entry
Cite this entry
Compte, N., Pepersack, T., Goriely, S. (2018). Frailty in Old Age is Associated with Altered Cytokine Production in Response to TLR Ligation. In: Fulop, T., Franceschi, C., Hirokawa, K., Pawelec, G. (eds) Handbook of Immunosenescence. Springer, Cham. https://doi.org/10.1007/978-3-319-64597-1_152-1
Download citation
DOI: https://doi.org/10.1007/978-3-319-64597-1_152-1
Received:
Accepted:
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-64597-1
Online ISBN: 978-3-319-64597-1
eBook Packages: Springer Reference Biomedicine and Life SciencesReference Module Biomedical and Life Sciences