Abstract
Basilar invagination (BI) and Chiari malformation type I CM-I) are the most common adult craniovertebral junction malformations, and they are frequently associated with each other and present synchronously. The relationship between BI and CM-I has remained incompletely understood, and the choice of surgical strategy has remained controversial. This brief review focuses on the different aspects of BI and CM-I, and further discusses the relationship between these two concomitant pathologies on the basis of the concepts proposed over the last three decades.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
Basilar invagination (BI) and Chiari malformation (CM) type I (CM-I) are the most common adult craniovertebral junction (CVJ) malformations [1]. These two pathologies are frequently associated with each other and present synchronously. According to craniometric studies, it seems that both of these pathologies belong to a spectrum of malformations whose common features include underdevelopment of the occipital bone and consequent neural and cerebrospinal fluid (CSF) flow compression [2]. However, each of these two malformations can present in isolation and be attributed to different aetiological factors and causal mechanisms [3,4,5]. The relationship between BI and CM-I has remained incompletely understood, thus hindering the development of therapeutic strategies.
With the purpose of better understanding these two malformations, this paper first focuses on different aspects of both BI and CM-I, and then discusses the relationship between these two concomitant pathologies on the basis of the concepts proposed over the last three decades.
Basilar Invagination
Definition and Aetiology
Basilar invagination is defined as a developmental anomaly of the CVJ in which the odontoid process abnormally prolapses upward and backward into the foramen magnum. However, several other terms such as ‘basilar impression’, ‘platybasia’ and ‘cranial settling’ have been interchangeably used in the literature to describe BI, causing terminological confusion over the years. In contrast to primary BI, the term ‘basilar impression’ refers to acquired or secondary BI, which may result from softening of the bone around the skull base. ‘Platybasia’ is an anthropological term describing an abnormally obtuse (>140°) angle between the anterior skull base and the clivus [6]. The term ‘cranial settling’ is typically used when BI is associated with rheumatoid arthritis.
Many pathologies may lead to development of BI. The reported aetiological factors include basioccipital/clivus hypoplasia, occipital condyle hypoplasia, atlas hypoplasia, an incomplete ring of C1 with spreading of the lateral masses, achondroplasia and atlanto-occipital assimilation (occipitalization of the atlas) [4, 5, 7, 8].
Clinical Presentations
The clinical presentations of BI are related to compression of neural and vascular structures around the CVJ area, as well as obstruction of CSF circulation. The symptoms and signs are diverse because of the multitude of aetiological factors in BI and the wide range of structures involved, including signs of medullary dysfunction such as nystagmus, dysphagia, ataxia, dysmetria and cranial nerve palsy; and signs of myelopathy such as motor dysfunction (weakness, restricted neck movements), sensory dysfunction (neck pain, paraesthesia) and vegetative dysfunction (bowel and bladder disturbance). The clinical presentation varies depending on the underlying or accompanying pathological process [9].
Craniometric Measurement and Diagnosis
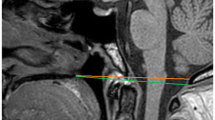
A series of craniometric measurements were employed to detect and diagnose BI in the previous literature. However, only a minor part of them are routinely used in clinical work, since most craniometric measurements lack adequate specificity and sensitivity [10]. There are three reference lines widely used in clinical work: the Chamberlain line, the McGregor line and the McRae line [11]. These three lines have been routinely adopted because of their high specificity, sensitivity and reproducibility. They are all viewed on lateral radiographs of the skull (Fig. 1).
Demonstration of three reference lines for measurement of basilar invagination (BI). (a) The Chamberlain line on a sagittal computed tomography (CT) scan [orange]. (b) The Chamberlain line on T2 magnetic resonance imaging (MRI) [orange]. (c) The McGregor line on a sagittal CT scan [green]. (d) The McGregor line on T2 MRI [green]. (e) The McRae line on a sagittal CT scan [yellow]. (f) The McRae line on T2 MRI [yellow]
The Chamberlain line extends from the posterior portion of the hard palate to the opisthion, which is the midpoint of the posterior margin of the foramen magnum (Fig. 1a, b) [6]. The tip of the odontoid process typically lies below or on the Chamberlain line. It is considered normal for the odontoid process to extend no more than 2.5 mm above this line, although the range varies, depending on the literature source [6, 12,13,14]. Generally, BI is considered present if the extension is greater than 5 mm [10].
Since precise identification of the opisthion on lateral radiographs is always difficult, McGregor proposed a modification of the Chamberlain line—the McGregor line—which extends from the posterior margin of the hard palate to the lowest point of the occipital squamosal surface (Fig. 1c, d) [12]. This line is about 2 mm inferior to the Chamberlain line, and thus it is considered abnormal when the tip of the odontoid process extends more than 7 mm above the line [12, 14].
The McRae line extends from the anterior (basion) to the posterior (opisthion) rim of the foramen magnum and is basically the anteroposterior length of the foramen magnum (Fig. 1d, e) [15]. The tip of the odontoid process should normally lie below this line. Furthermore, this line can also be used to define narrowing of the foramen magnum when the anteroposterior length is less than 19 mm in the sagittal plane [15].
While computed tomography (CT) is ideal for evaluation of osseous anatomy, magnetic resonance imaging (MRI) provides better assessment of soft tissue [8, 10]. In addition to conventional imaging—which includes plain radiography, CT and MRI—the development of dynamic imaging expands the ability to detect associated CVJ instability, functional stenosis of the spinal canal and cord compression [16,17,18,19].
Treatment Strategy
Surgical treatment for BI should be considered when neurological disturbance is present or foreseen. A large proportion of patients with BI present with neurological deficits resulting from compression of the cervicomedullary junction or the upper cervical cord [20,21,22]. The compression can be caused by a prolapsing dens or CVJ instability. Therefore, the definitive treatment for BI includes decompression and stabilization. Otherwise, in some patients without neurological symptoms where BI is an incidental finding, the necessity of surgical treatment should be carefully evaluated, since BI may present as a progressive deterioration and can result in neurological impairment and even sudden death if left untreated [23].
Thus, preoperative evaluation is of great importance. Aside from history taking and physical examination, nutritional status, dental hygiene and pulmonary status should be assessed [24]. Multimodal preoperative imaging of the CVJ, including CT and MRI (and even dynamic imaging), is an integral part of surgical planning [8, 10, 25]. In addition to searching for signs and sites of compression, preoperative assessment mainly focuses on the reducibility of BI, which is the critical factor in selection of the most suitable surgical approach.
A trial of axial cervical traction is usually performed in patients with BI to assess the degree to which the odontoid process might be reduced. It is now generally accepted that a posterior surgical approach alone, including decompression and fusion, can be adapted if the BI is reducible, while anterior decompression combined with posterior stabilization should be performed if the BI is not reducible [4, 23, 25,26,27].
Chiari Malformation Type I
Definition and Aetiology
Chiari malformation type I is characterized by downward herniation of the cerebellar tonsils and is defined as displacement of the cerebellar tonsils by more than 5 mm below the foramen magnum [28]. However, this is merely a radiographic definition, and the distances reported have varied from 3 mm to 5 mm, depending on the source [29, 30]. It has thus been suggested that the radiographic definition is limited to a terminological criterion and is not necessarily associated with clinical symptoms [31]. Therefore, cerebellar tonsil herniation of less than 5 mm or less than 3 mm does not exclude the diagnosis of CM-I.
In essence, CM-I is a disorder of the para-axial mesoderm, which is characterized by underdevelopment of the posterior cranial fossa (PCF) and overcrowding of the normally developed hindbrain [1, 32]. However, it can be associated with other miscellaneous conditions such as craniosynostosis [33], CSF leakage [34], Paget’s disease [35] and intracranial mass lesions [36, 37]. Furthermore, five distinct causal mechanisms of cerebellar tonsil herniation—(1) cranial constriction, (2) cranial settling, (3) spinal cord tethering, (4) intracranial hypertension and (5) intraspinal hypotension—which have been reported to have diagnosis and therapeutic implications, were identified in a previous study [3]. Therefore, CM-I is a disorder of multiple aetiological factors, including genetic predisposition, congenital anomalies and acquisition through trauma or illness, and it should be better defined generically.
Clinical Presentations
Only a small proportion of patients with CM-I are symptomatic [38]. The clinical presentation may be attributed to the original disorders as well as to the secondary pathological changes such as syringomyelia, scoliosis and BI. Taking syringomyelia as an example, it has been reported that patients with syringomyelia present at a slightly younger age and receive earlier diagnoses than patients without syringomyelia [31]. According to a large-scale patient-reported CM-I symptom study, pain is the most common symptom, and headache is the most frequently implicated type of pain [39]. Headache is also an indication for surgical treatment of CM-I in the absence of syringomyelia [38]. Other frequent clinical presentations of CM-I consist of a wide range of non-specific symptoms such as dizziness, sleeping disorder, neck pain, exhaustion and weakness [40].
Radiological Measurement and Diagnosis
According to the definition of CM-I, it is diagnosed radiologically as herniation of the cerebellar tonsils below the plane of the foramen magnum. However, the precise degree of herniation is not well established. It was suggested that in doubtful cases, cardiac-gated cine MRI is valuable in demonstrating a CSF flow obstruction as an indicator of clinically relevant herniation [41,42,43]. Furthermore, as the feature of CM-I, the ectopic tonsil position may result from a wide range of anatomical anomalies—such as craniosynostosis and an underdeveloped PCF—around the CVJ, leading to overcrowding of the PCF [44]. Therefore, analysis of the PCF is critical for better understanding of the pathogenesis and development of CM-I. Investigation of the PCF includes morphological and volumetric analyses, performed with the application of MRI.
Morphological Analysis of the Posterior Cranial Fossa
Measurements are performed on midline sagittal images, and four parameters are used to characterized the morphology of the PCF: (1) the length of the supraocciput, measured from the centre of the internal occipital protuberance to the opisthion; (2) the length of the clivus, measured from the tip of the dorsum sellae to the basion; (3) the slope of the tentorium, measured by calculation of the angle formed by the tentorium and a line drawn between the internal occipital protuberance and the opisthion; and (4) the extent of the cerebellar herniation, measured from the tips of the tonsils to a line drawn between the basion and the opisthion (Fig. 2) [31].
Measurement of posterior cranial fossa (PCF) morphology on T1 sagittal magnetic resonance imaging (MRI). The length of the supraocciput (IO) is measured by the distance from the centre of the internal occipital protuberance (I) to the opisthion (O). The length of the clivus (DB) is measured by the distance from the top of the dorsum sellae (D) to the basion (B). The slope of the tentorium (T) is calculated from the angle (a) formed by the supraocciput and tentorium. The level of the foramen magnum is defined by a line drawn from the basion (B) to the opisthion (O)
Volumetric Analysis of the Posterior Cranial Fossa
The posterior cranial fossa is defined as a space bounded by a series of osseous anatomical structures, including the clivus, occipital bone, tentorium and bilateral petrous ridges. Volumetric measurement is performed by the Cavalieri method. A clear grid with regularly spaced dots (4 mm apart) is placed on each equally spaced consecutive axial image of the posterior fossa. The section thickness is recorded as T, while the number of points that fall on the posterior fossa for each slice is recorded as Pi. Then the distances between the dots on the grid are correlated with the actual distance on the MRI by comparison with the centimetre scale on the image. The area (Ap) between points on the grid is calculated by squaring the actual anatomical distance between each set of two adjacent dots. The volume of the PCF (V) is calculated with the use of the following equation: V = Ap * ∑ Pi * T.
Apart from a reduced volume of the PCF, additional anomalies may involve the joint and discs of the CVJ. Klippel–Feil syndrome, atlanto-occipital assimilation and BI are features to look for in preoperative imaging, since these may indicate CVJ instability [24, 42, 45, 46]. Thus, routine employment of preoperative functional X-rays of the cervical spine in flexion and extension is recommended.
Treatment Strategy
The primary goals of surgery for CM-I include removing the compression from the brainstem and re-establishing CSF circulation. In the patient with syringomyelia, the goal is to prevent any additional neurological deficit and to decrease the size of the syrinx. However, there is no general consensus on the indication for the surgery. In a survey of American Society of Pediatric Neurosurgeons, Rocque et al. proposed that the presence of a syrinx seems to be a reasonable justification for operation [47]; other researchers have suggested that the decision regarding surgery should depend on the likelihood that a fixed neurological deficit as a consequence of the syrinx is more probable than spontaneous syrinx resolution, which has been observed, and the range of time necessary for a CM-I-related syrinx to resolve is yet to be explored [38, 48]. Another opinion from the perspective of symptomatology, held by Klekamp, is that asymptomatic patients have not been considered for surgery whether there was any sign of a syrinx or not, since he stated that he had not encountered an asymptomatic patient with a progressive syrinx [49].
With regard to the surgical strategy, although foramen magnum decompression is widely accepted as the treatment of choice for CM-I, controversies remain as to how this operation should be performed [50, 51]. The ongoing debate focuses on the risk and benefit of posterior fossa decompression alone versus posterior fossa decompression with duraplasty. According to a meta-analysis performed by Durham et al., posterior fossa decompression alone was associated with a significantly higher rate of reoperation (12.6% versus 2.1%) but a lower rate of CSF-related complications (1.8% versus 18.5%) than posterior fossa decompression with duraplasty [52]. In another meta-analysis, no convincing conclusion that one method was superior to the other could be drawn [53]. Complication rates are lower with procedures that leave the dura intact. However, this would be counterbalanced by lower rates of syrinx reduction and higher rates of symptom recurrence. Therefore, it appears that the surgical strategy should be tailored for individual patients according to preoperative evaluations using a series of radiographic tools and intraoperative assessments, such as intraoperative ultrasound [54,55,56].
On the other hand, another completely different surgical strategy has been proposed on the basis that the pathogenesis of CM-I is primary associated with atlantoaxial instability. In his series of studies, Goel suggested that CM-I may be a secondary phenomenon and a natural neural alteration in the face of atlantoaxial instability, and that surgical treatment should aim to restore atlantoaxial stabilization [57,58,59]. This conception was drawn from the observation of frequent concomitant presentation of BI and CM-I and the speculation that BI and CM-I are a continuum of the same pathological phenomenon originating from atlantoaxial instability, which needs to be investigated further.
The Relationship Between Basilar Invagination and Chiari Malformation Type I
In contrast to scenarios in which each of these two clinical entities presents in isolation, when BI is associated with CM-I (which frequently occurs in clinical observation), a variety of aspects—including the clinical presentation, natural progress, treatment strategy and prognosis—become very complicated and different.
Basilar Invagination with or Without Chiari Malformation Type I
The clinical course and symptoms of BI have been reported to be different when it is associated with CM-I. In an early study of 190 patients treated surgically for BI, the symptom onset of patients without CM-I was relatively acute, while the duration of symptoms of those with CM-I was long lasting. The most common presentations in the former population included weakness (100%), torticollis (69%), neck pain (59%), restricted neck movements (59%), posterior column dysfunction (39%), a low hairline (48%), a short neck (41%), bowel and bladder disturbance (28%) and paraesthesia (25%), while the most frequent presentations in the latter population included weakness (94%), paraesthesia (79%), disturbances of the posterior column and spinothalamic tract (56%), a short neck (50%) and ataxia (47%). Accordingly, in that study, Goel et al. presented a classification system for BI, which was divided into two subgroups on the basis of absence of CM (group I) and presence of CM (group II) [26]. In essence, group I included patients with invagination of the odontoid process into the foramen magnum, as well as potential compression of the brainstem. The tip of the odontoid process was distanced from the anterior arch of the atlas or the inferior aspect of the clivus, suggesting the presence of instability of the CVJ. However, the angle of the clivus and the volume of the PCF were not affected. On the other hand, in group II a reduced PCF volume could be noted from the superior migration of the assembly of the odontoid process, the clivus and the anterior arch of the atlas, thus leading to the presence of CM-I. This classification provided a comprehensive understanding of the pathology and pathogenesis of the anomaly and would be helpful in selection of surgical treatment, as well as in prediction of the outcome. On the premise that CM-I in the presence of BI results from reduced PCF volume, it could be deduced that while anterior transoral surgery should be performed in group I patients, posterior foramen magnum decompression should be additionally performed in group II patients. In 2014, Visocchi et al. first reported that clinical and radiological resolution of CVJ compression after transoral correction of BI was evident for up to 2 years postoperation, at which time the child had a relapse of some of the presenting symptoms and follow-up CT and MRI scans showed quite complete regrowth of the odontoid process, partial clival regeneration and recurrence of preoperative CM. Also, in this case the correlation of BI and CM was further confirmed [60].
Chiari Malformation Type I with or Without Basilar Invagination
Different incidence rates of BI in patients with CM-I have been reported, ranging from 12% to 35% [24, 31, 61]. Although the symptoms of CM-I commonly include occipital pain and gait ataxia, several differences in clinical presentation are noted between patients who have CM-I with invagination and those who have CM-I without invagination. Patients with invagination tend to be more affected by caudal cranial nerve deficits and gait disturbances owing to ventral compression, while patients without invagination are more likely to suffer from sensory disorders or neuropathic pain, which may be related to syringomyelia. In comparison with radiological findings in patients without invagination, segmentation anomalies such as assimilation of the atlas or Klippel–Feil syndrome are more common in patients with invagination [61].
The combination with BI makes treatment of CM-I more complicated. Although complication rates have reportedly been significantly higher in patients who have CM-I with BI than in those who have CM-I without BI [61], the issue of whether the combination with BI is associated with a worse long-term outcome of surgical treatment of CM-I remain controversial [61,62,63]. The clinical outcome may depend on the treatment strategy adopted, as well as individual differences, and thus there is a series of subsequent questions that remain to be resolved. As stated above, symptomatic patients with CM-I require surgical decompression of the foramen magnum. However, whether additional operations such as ventral transoral decompression, traction and realignment should be incorporated for treatment of concurrent BI remains controversial, since the presence of ventral compression or instability should be carefully evaluated before or during the operation.
The sign of instability is sometimes difficult to detect on preoperational radiographs or CT scans because the range of motion may be restricted by biomechanical limitations. When tabling the paradoxical discussion on low rates of symptomatic improvement and high rates of recurrence with conservative surgery, or the other way around with aggressive surgery, Goel proposed a series of surgeries aiming at atlantoaxial stabilization by listing the negative long-term outcomes of foramen magnum decompression [64, 65] and claiming that CM-I was caused by atlantoaxial dislocation regardless of the presence or absence of BI [57]. However, in studies with large populations, the coincidence of atlantoaxial instability with CM-I is less frequent [31, 45, 66]. Furthermore, defining instability in complex patients can be challenging. Although intraoperative findings may provide clues regarding instability, no objective criterion is available so far for diagnosing instability during surgery. As demonstrated in Goel’s clinical grading system for atlantoaxial dislocation, no gross physical or radiological abnormality may be present in cases of type III dislocation. It is illogical and even dangerous to perform a stabilization operation alone when instability is not clinically presented.
With regard to compression, the site and degree of compression should be assessed thoroughly for preoperational planning. When CM-I is associated with BI, aside from dorsal compression of the brainstem by herniated cerebellar tonsils, ventral compression by the odontoid peg may also be present. Is there any causal relationship between ventral and dorsal compression, thus making more decompression possible with less surgery? In a comparative study of 323 patients undergoing 350 operations, Klekamp demonstrated good short- and long-term results in patients who had CM-I with or without additional BI, through application of a treatment algorithm. He recommended that patients who had CM-I without BI and those who had BI without ventral compression could be managed by foramen magnum decompression alone, while most patients with ventral compression could be treated by posterior decompression, realignment and stabilization, reserving anterior decompression for patients with profound, symptomatic brainstem compression [61].
With further development of the research on the pathological theory and surgical strategies, the relationship between BI and CM-I—and the potential compression and instability involved in these two concomitant pathologies—should become better understood and managed.
Compliance with Ethical Standards
No financial support was received for this work.
Competing Interests
The authors declare that they have no competing interests.
References
Nishikawa M, Sakamoto H, Hakuba A, Nakanishi N, Inoue Y. Pathogenesis of Chiari malformation: a morphometric study of the posterior cranial fossa. J Neurosurg. 1997;86(1):40–7.
Ferreira JA, Botelho RV. The odontoid process invagination in normal subjects, Chiari malformation and basilar invagination patients: pathophysiologic correlations with angular craniometry. Surg Neurol Int. 2015;6:118.
Milhorat TH, Nishikawa M, Kula RW, Dlugacz YD. Mechanisms of cerebellar tonsil herniation in patients with Chiari malformations as guide to clinical management. Acta Neurochir. 2010;152(7):1117–27.
Klimo P Jr, Rao G, Brockmeyer D. Congenital anomalies of the cervical spine. Neurosurg Clin N Am. 2007;18(3):463–78.
Menezes AH. Craniocervical developmental anatomy and its implications. Childs Nerv Syst. 2008;24(10):1109–22.
Chamberlain WE. Basilar impression (platybasia): a bizarre developmental anomaly of the occipital bone and upper cervical spine with striking and misleading neurologic manifestations. Yale J Biol Med. 1939;11(5):487–96.
Charnas LR, Marini JC. Communicating hydrocephalus, basilar invagination, and other neurologic features in osteogenesis imperfecta. Neurology. 1993;43(12):2603–8.
Smoker WR. Craniovertebral junction: normal anatomy, craniometry, and congenital anomalies. Radiographics. 1994;14(2):255–77.
Pinter NK, McVige J, Mechtler L. Basilar invagination, basilar impression, and platybasia: clinical and imaging aspects. Curr Pain Headache Rep. 2016;20(8):49.
Smoker WR, Khanna G. Imaging the craniocervical junction. Childs Nerv Syst. 2008;24(10):1123–45.
Cronin CG, Lohan DG, Mhuircheartigh JN, Meehan CP, Murphy J, Roche C. CT evaluation of Chamberlain’s, McGregor’s, and McRae’s skull-base lines. Clin Radiol. 2009;64(1):64–9.
Mc GM. The significance of certain measurements of the skull in the diagnosis of basilar impression. Br J Radiol. 1948;21(244):171–81.
VanGilder JC, Menezes AH, Dolan KD. The craniovertebral junction and its abnormalities. New York: Futura; 1987.
Menezes AH, VanGilder JC. Anomalies of the craniovertebral junction. Philadelphia: Saunders; 1990.
McRae DL, Barnum AS. Occipitalization of the atlas. Am J Roentgenol Radium Ther Nucl Med. 1953;70(1):23–46.
El Asri AC, Akhaddar A, Gazzaz M, et al. Dynamic CT scan of the craniovertebral junction: a role in the management of os odontoideum. Neurol Neurochir Pol. 2010;44(6):603–8.
Gupta V, Khandelwal N, Mathuria SN, Singh P, Pathak A, Suri S. Dynamic magnetic resonance imaging evaluation of craniovertebral junction abnormalities. J Comput Assist Tomogr. 2007;31(3):354–9.
Reijnierse M, Breedveld FC, Kroon HM, Hansen B, Pope TL, Bloem JL. Are magnetic resonance flexion views useful in evaluating the cervical spine of patients with rheumatoid arthritis? Skelet Radiol. 2000;29(2):85–9.
Karhu JO, Parkkola RK, Koskinen SK. Evaluation of flexion/extension of the upper cervical spine in patients with rheumatoid arthritis: an MRI study with a dedicated positioning device compared to conventional radiographs. Acta Radiol. 2005;46(1):55–66.
Bassi P, Corona C, Contri P, Paiocchi A, Loiero M, Mangoni A. Congenital basilar impression: correlated neurological syndromes. Eur Neurol. 1992;32(4):238–43.
Erbengi A, Oge HK. Congenital malformations of the craniovertebral junction: classification and surgical treatment. Acta Neurochir. 1994;127(3–4):180–5.
Goel A, Desai K. Surgery for syringomyelia: an analysis based on 163 surgical cases. Acta Neurochir. 2000;142(3):293–301. discussion 301-292.
Sawin PD, Menezes AH. Basilar invagination in osteogenesis imperfecta and related osteochondrodysplasias: medical and surgical management. J Neurosurg. 1997;86(6):950–60.
Smith JS, Shaffrey CI, Abel MF, Menezes AH. Basilar invagination. Neurosurgery. 2010;66(3 Suppl):39–47.
Menezes AH. Evaluation and treatment of congenital and developmental anomalies of the cervical spine. Invited submission from the Joint Section Meeting on Disorders of the Spine and Peripheral Nerves, March 2004. J Neurosurg Spine. 2004;1(2):188–97.
Goel A, Bhatjiwale M, Desai K. Basilar invagination: a study based on 190 surgically treated patients. J Neurosurg. 1998;88(6):962–8.
Menezes AH. Surgical approaches: postoperative care and complications “transoral–transpalatopharyngeal approach to the craniocervical junction”. Childs Nerv Syst. 2008;24(10):1187–93.
Elster AD, Chen MY. Chiari I malformations: clinical and radiologic reappraisal. Radiology. 1992;183(2):347–53.
Aboulezz AO, Sartor K, Geyer CA, Gado MH. Position of cerebellar tonsils in the normal population and in patients with Chiari malformation: a quantitative approach with MR imaging. J Comput Assist Tomogr. 1985;9(6):1033–6.
Armonda RA, Citrin CM, Foley KT, Ellenbogen RG. Quantitative cine-mode magnetic resonance imaging of Chiari I malformations: an analysis of cerebrospinal fluid dynamics. Neurosurgery. 1994;35(2):214–23. discussion 223-214.
Milhorat TH, Chou MW, Trinidad EM, et al. Chiari I malformation redefined: clinical and radiographic findings for 364 symptomatic patients. Neurosurgery. 1999;44(5):1005–17.
Milhorat TH, Bolognese PA, Nishikawa M, et al. Association of Chiari malformation type I and tethered cord syndrome: preliminary results of sectioning filum terminale. Surg Neurol. 2009;72(1):20–35.
Cinalli G, Spennato P, Sainte-Rose C, et al. Chiari malformation in craniosynostosis. Childs Nerv Syst. 2005;21(10):889–901.
Atkinson JL, Weinshenker BG, Miller GM, Piepgras DG, Mokri B. Acquired Chiari I malformation secondary to spontaneous spinal cerebrospinal fluid leakage and chronic intracranial hypotension syndrome in seven cases. J Neurosurg. 1998;88(2):237–42.
Elisevich K, Fontaine S, Bertrand G. Syringomyelia as a complication of Paget’s disease. Case report. J Neurosurg. 1987;66(4):611–3.
Lee M, Rezai AR, Wisoff JH. Acquired Chiari-I malformation and hydromyelia secondary to a giant craniopharyngioma. Pediatr Neurosurg. 1995;22(5):251–4.
Morioka T, Shono T, Nishio S, Yoshida K, Hasuo K, Fukui M. Acquired Chiari I malformation and syringomyelia associated with bilateral chronic subdural hematoma. Case report. J Neurosurg. 1995;83(3):556–8.
Rocque BG, Oakes WJ. Surgical treatment of Chiari I malformation. Neurosurg Clin N Am. 2015;26(4):527–31.
Fischbein R, Saling JR, Marty P, et al. Patient-reported Chiari malformation type I symptoms and diagnostic experiences: a report from the national Conquer Chiari Patient Registry database. Neurol Sci. 2015;36(9):1617–24.
Mueller DM, Oro JJ. Prospective analysis of presenting symptoms among 265 patients with radiographic evidence of Chiari malformation type I with or without syringomyelia. J Am Acad Nurse Pract. 2004;16(3):134–8.
Hofkes SK, Iskandar BJ, Turski PA, Gentry LR, McCue JB, Haughton VM. Differentiation between symptomatic Chiari I malformation and asymptomatic tonsilar ectopia by using cerebrospinal fluid flow imaging: initial estimate of imaging accuracy. Radiology. 2007;245(2):532–40.
Tubbs RS, Lyerly MJ, Loukas M, Shoja MM, Oakes WJ. The pediatric Chiari I malformation: a review. Childs Nerv Syst. 2007;23(11):1239–50.
Panigrahi M, Reddy BP, Reddy AK, Reddy JJ. CSF flow study in Chiari I malformation. Childs Nerv Syst. 2004;20(5):336–40.
Tubbs RS. Definitions and anatomic considerations in Chiari I malformation and associated syringomyelia. Neurosurg Clin N Am. 2015;26(4):487–93.
Tubbs RS, Beckman J, Naftel RP, et al. Institutional experience with 500 cases of surgically treated pediatric Chiari malformation type I. J Neurosurg Pediatr. 2011;7(3):248–56.
Kagawa M, Jinnai T, Matsumoto Y, et al. Chiari I malformation accompanied by assimilation of the atlas, Klippel-Feil syndrome, and syringomyelia: case report. Surg Neurol. 2006;65(5):497–502. discussion 502
Rocque BG, George TM, Kestle J, Iskandar BJ. Treatment practices for Chiari malformation type I with syringomyelia: results of a survey of the American Society of Pediatric Neurosurgeons. J Neurosurg Pediatr. 2011;8(5):430–7.
Doughty KE, Tubbs RS, Webb D, Oakes WJ. Delayed resolution of Chiari I–associated hydromyelia after posterior fossa decompression: case report and review of the literature. Neurosurgery. 2004;55(3):711.
Klekamp J. Surgical treatment of Chiari I malformation—analysis of intraoperative findings, complications, and outcome for 371 foramen magnum decompressions. Neurosurgery. 2012;71(2):365–80. discussion 380.
Sakushima K, Tsuboi S, Yabe I, et al. Nationwide survey on the epidemiology of syringomyelia in Japan. J Neurol Sci. 2012;313(1–2):147–52.
Schijman E, Steinbok P. International survey on the management of Chiari I malformation and syringomyelia. Childs Nerv Syst. 2004;20(5):341–8.
Durham SR, Fjeld-Olenec K. Comparison of posterior fossa decompression with and without duraplasty for the surgical treatment of Chiari malformation type I in pediatric patients: a meta-analysis. J Neurosurg Pediatr. 2008;2(1):42–9.
Hankinson T, Tubbs RS, Wellons JC. Duraplasty or not? An evidence-based review of the pediatric Chiari I malformation. Childs Nerv Syst. 2011;27(1):35–40.
Hayhurst C, Richards O, Zaki H, Findlay G, Pigott TJ. Hindbrain decompression for Chiari–syringomyelia complex: an outcome analysis comparing surgical techniques. Br J Neurosurg. 2008;22(1):86–91.
Menezes AH, Greenlee JD, Donovan KA. Honored guest presentation: lifetime experiences and where we are going: Chiari I with syringohydromyelia–controversies and development of decision trees. Clin Neurosurg. 2005;52:297–305.
Milhorat TH, Bolognese PA. Tailored operative technique for Chiari type I malformation using intraoperative color Doppler ultrasonography. Neurosurgery. 2003;53(4):899–905. discussion 905-896.
Goel A. Is atlantoaxial instability the cause of Chiari malformation? Outcome analysis of 65 patients treated by atlantoaxial fixation. J Neurosurg Spine. 2015;22(2):116–27.
Goel A. Atlantoaxial facetal distraction spacers: indications and techniques. J Craniovertebr Junction Spine. 2016;7(3):127–8.
Goel A. Basilar invagination, Chiari malformation, syringomyelia: a review. Neurol India. 2009;57(3):235–46.
Visocchi M, Trevisi G, Iacopino DG, Tamburrini G, Caldarelli M, Barbagallo GM. Odontoid process and clival regeneration with Chiari malformation worsening after transoral decompression: an unexpected and previously unreported cause of “accordion phenomenon”. Eur Spine J. 2015;24(Suppl 4):S564–8.
Klekamp J. Chiari I malformation with and without basilar invagination: a comparative study. Neurosurg Focus. 2015;38(4):E12.
Aghakhani N, Parker F, David P, et al. Long-term follow-up of Chiari-related syringomyelia in adults: analysis of 157 surgically treated cases. Neurosurgery. 2009;64(2):308–15. discussion 315
Arora P, Behari S, Banerji D, Chhabra DK, Jain VK. Factors influencing the outcome in symptomatic Chiari I malformation. Neurol India. 2004;52(4):470–4.
Klekamp J. Neurological deterioration after foramen magnum decompression for Chiari malformation type I: old or new pathology? J Neurosurg Pediatr. 2012;10(6):538–47.
Naftel RP, Tubbs RS, Menendez JY, Wellons JC 3rd, Pollack IF, Oakes WJ. Worsening or development of syringomyelia following Chiari I decompression: case report. J Neurosurg Pediatr. 2013;12(4):351–6.
Goldstein HE, Anderson RC. Craniovertebral junction instability in the setting of Chiari I malformation. Neurosurg Clin N Am. 2015;26(4):561–9.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer International Publishing AG, part of Springer Nature
About this chapter
Cite this chapter
Liao, C. et al. (2019). The Relationship Between Basilar Invagination and Chiari Malformation Type I: A Narrative Review. In: Visocchi, M. (eds) New Trends in Craniovertebral Junction Surgery. Acta Neurochirurgica Supplement, vol 125. Springer, Cham. https://doi.org/10.1007/978-3-319-62515-7_16
Download citation
DOI: https://doi.org/10.1007/978-3-319-62515-7_16
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-62514-0
Online ISBN: 978-3-319-62515-7
eBook Packages: MedicineMedicine (R0)