Abstract
Proper development and steroidogenic function of the adrenal gland are essential for organism survival. The cortex in the adult adrenal is organized into concentric zones that produce distinct steroid hormones, the zona glomerulosa (zG) produces aldosterone, and the zona fasciculata (zF) produces corticosterone (in rodents) and cortisol (in humans). Homeostasis and regeneration of the cortex are linked to centripetal migration and direct cell fate conversion of cells from the zG to the zF. To define the mechanisms underlying adrenal zonation, it is essential to understand how tissue structure impacts steroidogenic function. This chapter highlights our current knowledge of adrenocortical homeostasis and zonation in the adult, with an emphasis on (1) the adrenal morphology and ultrastructure, (2) the signaling pathways implicated in the control of zonation, and (3) the cellular mechanisms associated with direct cell fate conversion of zG to zF cells, the major pathway underlying centripetal migration and cortical renewal during postnatal homeostasis.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
The adrenal cortex is a major site of steroid hormone production . In adult mammals it is comprised of three concentric layers or zones of steroid-producing cells surrounding the adrenal medulla [1, 2]. The outer layer of the cortex, the zona glomerulosa (zG), represents ~15% of the cortical mass and produces the mineralocorticoid aldosterone, which is essential for sodium retention, intravascular volume, and blood pressure regulation. Excess aldosterone production, as seen in primary aldosteronism, is a major cause of hypertension and cardiovascular damage [3, 4]. The middle layer of the cortex, the zona fasciculata (zF), is ~8 times larger than the zG and produces the glucocorticoid corticosterone (in rodents) and cortisol (in humans), which impacts immunity, metabolism, development , and behavior. A third layer, the zona reticularis (zR), is present in humans, some nonhuman primates (e.g., rhesus macaques, marmosets), ferrets, and the spiny mouse. It lies between the zF and the medulla and produces androgens, such as dehydroepiandrosterone (DHEA) and its sulfated derivative DHEA-S [5]. While the mouse adrenal lacks a true zR, it does contain a transient X-zone (zX), which appears to be a remnant of the fetal adrenal cortex [6] and is thought to be involved in progesterone metabolism [7] (Fig. 1.1).
Embryonic Adrenal Development
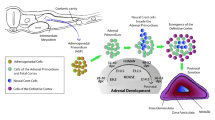
Adrenal embryonic development has been extensively studied [8]. In the mouse, development begins on embryonic day 9 (E9.0), or around 28 days post coitum (28 dpc) in the human, when cells in the coelomic epithelium first express the master transcriptional regulator steroidogenic factor 1 (SF1, also known as NR5A1 and AD4BP), which results in the emergence of the adrenogonadal lineage. SF1+ cells then delaminate into the adjacent mesenchyme giving rise to the adrenogonadal primordium (AGP). AGP cells, marked by expression of the Sf1-fetal adrenal enhancer (FAdE), then give rise to the fetal adrenal anlagen around E10.5 (~33 dpc). Next, cells from the neural crest invade the fetal adrenal ~E12.5 (~48 dpc), which go on to form the adrenal medulla. Subsequently, ~E14.5 (~56 dpc) the fetal cortex slowly begins to regress, while the definitive (adult) cortex emerges beneath the newly formed capsule, though distinct zones are not yet formed (Fig. 1.2). Establishing a connection between the definitive cortex and the fetal cortex, elegant lineage-tracing studies have demonstrated that the definitive cortex is indeed a direct descendent of the fetal cortex [9, 10].
Embryonic adrenal development in the mouse. Schematic illustration of the cellular changes during mouse and human adrenal development. The adrenogonadal primordium (AGP) originates from a thickening of the coelomic epithelium designated by the red dashed circle around E9.0 (28 dpc). At E10.5 (33 dpc), the adrenal anlage separates from the AGP and is then invaded by neural crest cells, precursors of the medullary chromaffin cells around E12.5 (48 dpc). From E14.5 (56 dpc) onward, the fetal cortical cells are slowly replaced by the definitive cortex, which gives rise to functional zones around the time of birth. Once formed, the zones are maintained throughout life
Postnatal Adrenal Development
In contrast to early embryonic development, the mechanisms underlying postnatal adrenal development , which lead to the formation and maintenance of the adrenal’s distinct zones, remain poorly understood. Detailed knowledge of how these mechanisms mediate zonation has important implications for understanding normal homeostatic functions as well as the pathological conditions that arise within the adrenal cortex. For example, it is known that control of steroidogenic output is dependent, in part, on proper maintenance of zonation over time [1]. Consistent with this, impaired zonation has been implicated in a range of conditions, including primary aldosteronism, cortisol-producing adenomas, primary pigmented nodular adrenocortical disease (PPNAD), congenital adrenal hyper- and hypoplasia, and adrenocortical carcinoma [11]. While the precise mechanisms underlying each of these conditions remain to be fully characterized, recent advances in our understanding of the cellular and molecular mechanisms underlying normal tissue homeostasis have made it possible to begin to explore key structure/function relationships within this tissue.
Adrenal Morphology
The adrenal cortex is an epithelial tissue circumscribed by a mesenchymal capsule (Fig. 1.1). The cells of the zG are organized in distinct morphological clusters, known as glomeruli, a highly conserved structure [12], which is surrounded by a basement membrane and a fenestrated capillary network [13]. zG cells are densely packed and contain scant cytoplasm, abundant rough endoplasmic reticulum, and a small number of lipid droplets and mitochondria [12, 14, 15]. In contrast, zF cells are arrayed in cord-like structures and exhibit distinctly different morphological features. zF cells are larger and more loosely packed than zG cells and contain extensive smooth endoplasmic reticulum, large gap junctions, numerous lipid droplets, and mitochondria characterized by tubulovesicular cristae [12, 14]. Also, like in the zG, zF cells are surrounded by a basement membrane and a rich capillary network. While the cells in the zR are morphologically similar to zF cells, they contain fewer lipid droplets with additional lysosomes and lipofuscin pigment granules [16]. In mice, X-zone cells are smaller than zF cells, contain an eosinophilic cytoplasm, and demonstrate a range of mitochondrial shapes with tubular cristae [6, 17].
Signaling Pathways and Zonation
The presence of morphologically distinct, yet physically contiguous, adrenocortical zones suggests tight regulation of each zone’s identity, relative size, and overall function. Recent advances in our understanding of how angiotensin II (AngII), potassium ions (K+), and adrenocorticotropic hormone (ACTH) regulate adrenal homeostasis may ultimately provide key insights into the origins of adrenal zonation and the dynamic regulation of these zones that occurs in response to physiological cues [18,19,20,21,22,23,24,25,26,27]. It is likely that multiple signaling pathways also contribute to adrenal zonation. Considerable progress has been made regarding the role of the canonical Wnt/β-catenin signaling pathway and the role of the ACTH/cyclic adenosine monophosphate (cAMP) pathway in setting the morphological and functional boundaries between the zones [11, 18, 21, 25, 28,29,30,31,32,33,34].
The canonical Wnt signaling pathway is active in the outer region of the cortex, overlapping with the morphological zG, and drives a transcriptional program that facilitates the production of the mineralocorticoid aldosterone [11, 29, 32]. Consistent with this, in vitro and in vivo experiments demonstrate that constitutive activation of the canonical Wnt pathway leads to an upregulation of aldosterone biosynthesis and an expansion of the morphological zG, while inhibition of the pathway leads to inactivation of aldosterone biosynthesis and contraction of the zG [28,29,30,31,32, 34, 35]. In contrast, the ACTH/cAMP signaling pathway is dominant in the zF and mediates the downstream transcriptional effects of ACTH on the synthesis and secretion of glucocorticoids [36,37,38]. Additionally, recent evidence suggests a reciprocal inhibitory effect of these two pathways, whereby Wnt signaling maintains zG zonal identity and size and also serves to inhibit expression of the zF program [30, 34]. Critical mediators of these effects include two key Wnt pathway ligands: Rspo3 (secreted from the capsule) and Wnt4 (expressed in the zG). Consistent with this, ectopic activation of Wnt signaling inactivates the zF steroidogenic program [11, 29, 30]. On the other hand, stabilization of the ACTH/cAMP signaling pathway results in activation of the zF steroidogenic program and inhibition of the Wnt signaling pathway leading to contraction of the zG [30, 33].
The overall significance of these signaling pathways in the regulation of adrenal homeostasis and zonation is made clear by the effects of somatic gain-of-function mutations giving rise to (1) aldosterone-producing adenomas (APAs) (associated with aberrant activation of the Wnt pathway) and (2) PPNAD (arising from mutations in PRKAR1A mutations, which leads to constitutive activation of ACTH/cAMP-dependent signaling) [39, 40].
Centripetal Migration and Cortical Renewal
Once established, the zG and the zF are continuously renewed throughout life and undergo dynamic hormonal feedback regulation . Despite the functional importance of these separate layers, surprisingly little is known about the cellular mechanisms that underlie their formation and ongoing maintenance. Recently, two members of the sonic hedgehog family, GLI1 and SHH, were identified as markers for adrenal progenitor cells that reside in the capsule and subcapsular regions (adjacent to the zG), respectively [41]. Consistent with the classical model of centripetal migration [42], proposed more than 70 years ago, these progenitor cells give rise to terminally differentiated zG cells, which then migrate centripetally and are thought to undergo cell fate conversion into zF cells before undergoing apoptosis at the corticomedullary junction [43].
Generation of Cyp11b2-Cre Mice
To define the molecular and cellular mechanisms underlying adrenal lineage development, we recently targeted the Cyp11b2 (aldosterone synthase) locus in mice, to generate a knock-in/knock-out Cyp11b2-Cre allele (officially known as Cyp11b2tm1.1(cre)Brit). Combined with other strains, these mice facilitate lineage-tracing, cell fate analysis and tissue-specific knock-out studies, specifically within zG cells [20]. CYP11B2 is required for the final steps of aldosterone synthesis, and its gene expression is restricted to terminally differentiated cells in the zG [43], making it a highly specific marker for zG cells. Although given the heterogeneous nature of Cyp11b2 expression with the zG under normal conditions, it is not as sensitive as other validated zG markers (e.g., β-catenin, Dab2, Dlk1) [11, 44,45,46,47]. Importantly, mice heterozygous for the Cyp11b2-Cre allele maintain normal levels of aldosterone and plasma renin activity (PRA), essential components of the renin-angiotensin aldosterone system (RAAS), indicating normal feedback regulation is maintained. In contrast, mice homozygous for the Cyp11b2-Cre allele are aldosterone deficient and demonstrate increase levels of PRA.
Direct Cell Fate Conversion
To investigate whether zG cells undergo direct cell fate conversion to zF cells, lineage-tracing studies were performed by combining Cyp11b2-Cre mice with the Rosa26 lineage reporter strain, which expresses membrane-targeted Tomato at baseline and expresses membrane-targeted green fluorescent protein (GFP) following Cre-mediated recombination (Fig. 1.3a) [20]. These studies revealed activation of the endogenous Cyp11b2 locus around the time of birth, and GFP-marked cells were entirely restricted to the zG. During the first few weeks of postnatal development, the zG was progressively marked by GFP expression (Fig. 1.3b), which subsequently gave rise to zF cells in a radial fashion, ultimately remodeling the entire zF by ~12 weeks of age (Fig. 1.3c) [20]. zG to zF cell fate conversion also functions during adrenal regeneration following dexamethasone suppression [20]. Together, these observations establish that differentiated zG cells give rise to zF cells through a process of direct cell fate conversion during postnatal adrenocortical zonation and regeneration , consistent with the model of centripetal migration.
zG cells give rise to zF cells through direct conversion . (a) Schematic illustration of the Cyp11b2-Cre and the Rosa26-mTmG allele (R26R) alleles before and after Cre-mediated LoxP recombination, which leads to deletion of mTomato and expression of mGFP. (b) Schematic illustration showing centripetal migration of GFP+ cells from the zG to the zF. (c) Representative immunofluorescent images showing centripetal migration of GFP+ cells from the zG (left, 2 weeks of age) to the zF (right, 6 weeks of age) in female mice. Scale bar, 50 μm
Role of SF1 in Cell Fate Conversion and zG Homeostasis
Understanding the mechani sms that regulate cell fate conversion has important implications for both normal and pathological states. The ability of one differentiated cell to be converted into another differentiated cell type, without passing through an undifferentiated state, has been described following the overexpression of specific transcription factors. For example, fibroblasts can be converted into myoblasts following expression of MyoD [48], and embryonic and mesenchymal stem cells can be converted into steroid-producing cells following expression of SF1 [49, 50]. The observations that SF1 plays a critical role during steroidogenic development and is sufficient to activate a steroidogenic program raised the possibility that it may play a role in cell fate conversion. Consistent with this, we observed that deletion of SF1 within zG cells prevented their conversion to zF cells (Fig. 1.4a) [20]. While the overall size of the zG remained essentially unchanged, detailed histological analysis revealed that lineage-marked zG cells had a dramatically altered cell shape, raising the possibility that these cells had undergone dedifferentiation. In addition, functional analysis revealed a state of compensated hypoaldosteronism, indicated by normal aldosterone levels and a nearly threefold increase in the levels of PRA.
Deletion of SF1 impairs zG-to-zF conversion and alters gene expression. (a) Representative immunofluorescent images of wild-type and SF1 KO adrenals demonstrating contribution of GFP+ cells to the zF. Note the absence of GFP+ cells in the zF in SF1 KO adrenals. Both images are t aken from 10-week-old female mice. Scale bar, 50 μm. (b) Heat map representation of differentially expressed genes from w ild-type and SF1 KO whole adrenals. Dendrogr ams represent hierarchical clustering of genes and samples. (c) Select list of genes that are down- or up-regulated in SF1 KO whole adrenals compared to wild-type whole adrenals
To identify the mechanisms by which SF1 regulates cell fate conversion and zG homeostasis, we performed transcriptome analysis comparing total RNA from SF1 KO and wild-type adrenals using Affymetrix microarray analysis. Of 35,556 probe s analyzed, 240 expressed a greater than two-fold difference in expression level and 105 of those contained unique genes (Fig. 1.4b). Among the genes showing the greatest fold changes were members of the Wnt/β-catenin signaling pathway and members of the clock gene family (Fig. 1.4c). Both of these pathways have been implicated in adrenal homeostasis, though what role they play in zG homeostasis and zonation remains largely unknown. Finally, these studies also revealed that zF cells were functionally normal, as evidenced by measurement of basal corticosterone secretion, and indicate that an “alternate (zG-independent) pathway” can contribute to zF formation. Exactly how this alternative pathway directs zF formation as well as whether it functions during normal adrenal homeostasis remains to be determined. One possibility is that when normal tissue homeostasis is severely disrupted, such as in the case of zG-specific SF1 deletion, mesenchymal cells in the capsule harboring stem-/progenitor-like potential may become activated to directly replenish the zF. Changes in gene expression identified in the microarray analysis (Fig. 1.4b) may provide new insight into these mechanisms.
Conclusions and Future Directions
In summary, the mechanisms underlying adrenocortical homeostasis and zonation during postnatal development remain largely unknown, though critical insights have recently been made. It is clear, for example, that direct conversion of zG cells into zF cells represents the major cellular mechanism by which the cortex is maintained under normal homeostatic conditions. However, it remains less clear as to the extent zG cells, alone, sustain long-term cortical renewal or to what degree zG cells rely on replenishment from the capsule, an important signaling center. Genetic lineage-tracing experiments performed by several laboratories have unequivocally demonstrated that the mesenchymal capsule can serve as a source for cellular replenishment for all steroidogenic zones as well as non-steroidogenic stromal cells [9, 41, 51]. However, an important issue raised by these studies is that constant centripetal migration of cells appears to require a much higher cellular turnover rate than provided by capsular cell activity. Hence, it is possible that differentiated cortical cells, especially the more proliferative zG population, may, in fact, play a key role in supporting the self-renewal of this tissue. Understanding which cells underlie adrenocortical self-renewal has important implications for (1) the development of future regenerative medicine strategies and for (2) understanding the pathogenesis of adrenal neoplasms. Going forward, the ability to perform “pulse-chase” lineage-tracing studies utilizing inducible mouse models will help to define the self-renewing potential of mature zG cells and to better understand the mechanisms underlying adrenal homeostasis.
References
Gallo-Payet N, Battista M-C (2014) Steroidogenesis-adrenal cell signal transduction. In: Terjung R (ed) Compr Physiol. Wiley, Hoboken, NJ, pp 889–964.
Yates R, Katugampola H, Cavlan D, Cogger K, Meimaridou E, Hughes C, Metherell L, Guasti L, King P (2013) Adrenocortical development, maintenance, and disease. In: Curr Top Dev Biol. Elsevier, pp 239–312.
Galati S-J, Hopkins SM, Cheesman KC, Zhuk RA, Levine AC. Primary aldosteronism: emerging trends. Trends Endocrinol Metab. 2013;24:421–30. doi:10.1016/j.tem.2013.05.003.
Magill SB (2014) Pathophysiology, diagnosis, and treatment of mineralocorticoid disorders. In: Terjung R (ed) Compr Physiol. Wiley, Hoboken, NJ, pp 1083–1119.
Pihlajoki M, Dörner J, Cochran RS, Heikinheimo M, Wilson DB (2015) Adrenocortical zonation, renewal, and remodeling. Front Endocrinol. doi: 10.3389/fendo.2015.00027.
Morohashi K, Zubair M. The fetal and adult adrenal cortex. Mol Cell Endocrinol. 2011;336:193–7. doi:10.1016/j.mce.2010.11.026.
Hershkovitz L, Beuschlein F, Klammer S, Krup M, Weinstein Y. Adrenal 20alpha-hydroxysteroid dehydrogenase in the mouse catabolizes progesterone and 11-deoxycorticosterone and is restricted to the X-zone. Endocrinology. 2007;148:976–88. doi:10.1210/en.2006-1100.
Xing Y, Lerario AM, Rainey W, Hammer GD. Development of adrenal cortex zonation. Endocrinol Metab Clin N Am. 2015;44:243–74. doi:10.1016/j.ecl.2015.02.001.
Wood MA, Acharya A, Finco I, Swonger JM, Elston MJ, Tallquist MD, Hammer GD. Fetal adrenal capsular cells serve as progenitor cells for steroidogenic and stromal adrenocortical cell lineages in M. musculus. Development. 2013;140:4522–32. doi:10.1242/dev.092775.
Zubair M, Parker KL, Morohashi K. Developmental links between the fetal and adult zones of the adrenal cortex revealed by lineage tracing. Mol Cell Biol. 2008;28:7030–40. doi:10.1128/MCB.00900-08.
Walczak EM, Hammer GD. Regulation of the adrenocortical stem cell niche: implications for disease. Nat Rev Endocrinol. 2014;11:14–28. doi:10.1038/nrendo.2014.166.
Nussdorfer GG. Cytophysiology of the adrenal zona glomerulosa. Int Rev Cytol. 1980;64:307–68.
Otis M, Campbell S, Payet MD, Gallo-Payet N. Expression of extracellular matrix proteins and integrins in rat adrenal gland: importance for ACTH-associated functions. J Endocrinol. 2007;193:331–47. doi:10.1677/JOE-07-0055.
Black VH, Robbins D, McNamara N, Huima T. A correlated thin-section and freeze-fracture analysis of guinea pig adrenocortical cells. Am J Anat. 1979;156:453–503. doi:10.1002/aja.1001560404.
Friend DS, Gilula NB. A distinctive cell contact in the rat adrenal cortex. J Cell Biol. 1972;53:148–63.
Rhodin JA. The ultrastructure of the adrenal cortex of the rat under normal and experimental conditions. J Ultrastruct Res. 1971;34:23–71.
Sato T. (1968) the fine structure of the mouse adrenal X zone. Z Für Zellforsch Mikrosk Anat Vienna Austria. 1948;87:315–29.
Chida D, Nakagawa S, Nagai S, Sagara H, Katsumata H, Imaki T, Suzuki H, Mitani F, Ogishima T, Shimizu C, Kotaki H, Kakuta S, Sudo K, Koike T, Kubo M, Iwakura Y. Melanocortin 2 receptor is required for adrenal gland development, steroidogenesis, and neonatal gluconeogenesis. Proc Natl Acad Sci U S A. 2007;104:18205–10. doi:10.1073/pnas.0706953104.
Deane HW, Shaw JH, Greep RO. The effect of altered sodium or potassium intake on the width and cytochemistry of the zona glomerulosa of the rat’s adrenal cortex. Endocrinology. 1948;43:133–53. doi:10.1210/endo-43-3-133.
Freedman BD, Kempna PB, Carlone DL, Shah MS, Guagliardo NA, Barrett PQ, Gomez-Sanchez CE, Majzoub JA, Breault DT. Adrenocortical zonation results from lineage conversion of differentiated zona glomerulosa cells. Dev Cell. 2013;26:666–73. doi:10.1016/j.devcel.2013.07.016.
Karpac J, Ostwald D, Bui S, Hunnewell P, Shankar M, Hochgeschwender U. Development, maintenance, and function of the adrenal gland in early postnatal proopiomelanocortin-null mutant mice. Endocrinology. 2005;146:2555–62. doi:10.1210/en.2004-1290.
McEwan PE, Vinson GP, Kenyon CJ. Control of adrenal cell proliferation by AT1 receptors in response to angiotensin II and low-sodium diet. Am J Phys. 1999;276:E303–9.
McNeill H. Distribution of extracellular signal-regulated protein kinases 1 and 2 in the rat adrenal and their activation by angiotensin II. J Endocrinol. 2005;187:149–57. doi:10.1677/joe.1.06347.
Nishimoto K, Harris RBS, Rainey WE, Seki T. Sodium deficiency regulates rat adrenal zona glomerulosa gene expression. Endocrinology. 2014;155:1363–72. doi:10.1210/en.2013-1999.
Pulichino A-M, Vallette-Kasic S, Couture C, Gauthier Y, Brue T, David M, Malpuech G, Deal C, Van Vliet G, De Vroede M, Riepe FG, Partsch C-J, Sippell WG, Berberoglu M, Atasay B, Drouin J. Human and mouse TPIT gene mutations cause early onset pituitary ACTH deficiency. Genes Dev. 2003;17:711–6. doi:10.1101/gad.1065603.
Shelton JH, Jones AL. The fine structure of the mouse adrenal cortex and the ultrastructural changes in the zona glomerulosa with low and high sodium diets. Anat Rec. 1971;170:147–81. doi:10.1002/ar.1091700204.
Thomas M, Keramidas M, Monchaux E, Feige J-J. Dual hormonal regulation of endocrine tissue mass and vasculature by adrenocorticotropin in the adrenal cortex. Endocrinology. 2004;145:4320–9. doi:10.1210/en.2004-0179.
Berthon A, Drelon C, Ragazzon B, Boulkroun S, Tissier F, Amar L, Samson-Couterie B, Zennaro M-C, Plouin P-F, Skah S, Plateroti M, Lefèbvre H, Sahut-Barnola I, Batisse-Lignier M, Assié G, Lefrançois-Martinez A-M, Bertherat J, Martinez A, Val P. WNT/β-catenin signalling is activated in aldosterone-producing adenomas and controls aldosterone production. Hum Mol Genet. 2014;23:889–905. doi:10.1093/hmg/ddt484.
Berthon A, Sahut-Barnola I, Lambert-Langlais S, de Joussineau C, Damon-Soubeyrand C, Louiset E, Taketo MM, Tissier F, Bertherat J, Lefrançois-Martinez A-M, Martinez A, Val P. Constitutive beta-catenin activation induces adrenal hyperplasia and promotes adrenal cancer development. Hum Mol Genet. 2010;19:1561–76. doi:10.1093/hmg/ddq029.
Drelon C, Berthon A, Sahut-Barnola I, Mathieu M, Dumontet T, Rodriguez S, Batisse-Lignier M, Tabbal H, Tauveron I, Lefrançois-Martinez A-M, Pointud J-C, Gomez-Sanchez CE, Vainio S, Shan J, Sacco S, Schedl A, Stratakis CA, Martinez A, Val P. PKA inhibits WNT signalling in adrenal cortex zonation and prevents malignant tumour development. Nat Commun. 2016;7:12751. doi:10.1038/ncomms12751.
Heikkilä M, Peltoketo H, Leppäluoto J, Ilves M, Vuolteenaho O, Vainio S. Wnt-4 deficiency alters mouse adrenal cortex function, reducing aldosterone production. Endocrinology. 2002;143:4358–65. doi:10.1210/en.2002-220275.
Kim AC, Reuter AL, Zubair M, Else T, Serecky K, Bingham NC, Lavery GG, Parker KL, Hammer GD. Targeted disruption of beta-catenin in Sf1-expressing cells impairs development and maintenance of the adrenal cortex. Dev Camb Engl. 2008;135:2593–602. doi:10.1242/dev.021493.
Sahut-Barnola I, de Joussineau C, Val P, Lambert-Langlais S, Damon C, Lefrançois-Martinez A-M, Pointud J-C, Marceau G, Sapin V, Tissier F, Ragazzon B, Bertherat J, Kirschner LS, Stratakis CA, Martinez A. Cushing’s syndrome and fetal features resurgence in adrenal cortex-specific Prkar1a knockout mice. PLoS Genet. 2010;6:e1000980. doi:10.1371/journal.pgen.1000980.
Vidal V, Sacco S, Rocha AS, da Silva F, Panzolini C, Dumontet T, Doan TMP, Shan J, Rak-Raszewska A, Bird T, Vainio S, Martinez A, Schedl A. The adrenal capsule is a signaling center controlling cell renewal and zonation through Rspo3. Genes Dev. 2016;30:1389–94. doi:10.1101/gad.277756.116.
Bhandaru M, Kempe DS, Rotte A, Rexhepaj R, Kuhl D, Lang F. Hyperaldosteronism, hypervolemia, and increased blood pressure in mice expressing defective APC. Am J Physiol Regul Integr Comp Physiol. 2009;297:R571–5. doi:10.1152/ajpregu.00070.2009.
Clark AJ, Weber A. Adrenocorticotropin insensitivity syndromes. Endocr Rev. 1998;19:828–43. doi:10.1210/edrv.19.6.0351.
Côté M, Guillon G, Payet MD, Gallo-Payet N. Expression and regulation of adenylyl cyclase isoforms in the human adrenal gland. J Clin Endocrinol Metab. 2001;86:4495–503. doi:10.1210/jcem.86.9.7837.
Gorrigan RJ, Guasti L, King P, Clark AJ, Chan LF. Localisation of the melanocortin-2-receptor and its accessory proteins in the developing and adult adrenal gland. J Mol Endocrinol. 2011;46:227–32. doi:10.1530/JME-11-0011.
Berthon AS, Szarek E, Stratakis CA. PRKACA: the catalytic subunit of protein kinase a and adrenocortical tumors. Front Cell Dev Biol. 2015;3:26. doi:10.3389/fcell.2015.00026.
Boulkroun S, Fernandes-Rosa FL, Zennaro M-C. Molecular and cellular mechanisms of aldosterone producing adenoma development. Front Endocrinol. 2015;6:95. doi:10.3389/fendo.2015.00095.
King P, Paul A, Laufer E. Shh signaling regulates adrenocortical development and identifies progenitors of steroidogenic lineages. Proc Natl Acad Sci. 2009;106:21185–90. doi:10.1073/pnas.0909471106.
Salmon TN, Zwemer RL. A study of the life history of cortico-adrenal gland cells of the rat by means of trypan blue injections. Anat Rec. 1941;80:421–9. doi:10.1002/ar.1090800404.
Kim AC, Barlaskar FM, Heaton JH, Else T, Kelly VR, Krill KT, Scheys JO, Simon DP, Trovato A, Yang W-H, Hammer GD. In search of adrenocortical stem and progenitor cells. Endocr Rev. 2009;30:241–63. doi:10.1210/er.2008-0039.
Eberhart CG, Argani P. Wnt signaling in human development: beta-catenin nuclear translocation in fetal lung, kidney, placenta, capillaries, adrenal, and cartilage. Pediatr Dev Pathol. 2001;4:351–7.
Halder SK, Takemori H, Hatano O, Nonaka Y, Wada A, Okamoto M. Cloning of a membrane-spanning protein with epidermal growth factor-like repeat motifs from adrenal glomerulosa cells. Endocrinology. 1998;139:3316–28. doi:10.1210/endo.139.7.6081.
Pignatti E, Leng S, Carlone DL, Breault DT. Regulation of zonation and homeostasis in the adrenal cortex. Mol Cell Endocrinol. 2017;441:146–55. doi:10.1016/j.mce.2016.09.003.
Romero DG, Yanes LL, de Rodriguez AF, Plonczynski MW, Welsh BL, Reckelhoff JF, Gomez-Sanchez EP, Gomez-Sanchez CE. Disabled-2 is expressed in adrenal zona glomerulosa and is involved in aldosterone secretion. Endocrinology. 2007;148:2644–52. doi:10.1210/en.2006-1509.
Davis RL, Weintraub H, Lassar AB. Expression of a single transfected cDNA converts fibroblasts to myoblasts. Cell. 1987;51:987–1000.
Crawford PA, Sadovsky Y, Milbrandt J. Nuclear receptor steroidogenic factor 1 directs embryonic stem cells toward the steroidogenic lineage. Mol Cell Biol. 1997;17:3997–4006.
Sakai N, Terami H, Suzuki S, Haga M, Nomoto K, Tsuchida N, Morohashi K, Saito N, Asada M, Hashimoto M, Harada D, Asahara H, Ishikawa T, Shimada F, Sakurada K. Identification of NR5A1 (SF-1/AD4BP) gene expression modulators by large-scale gain and loss of function studies. J Endocrinol. 2008;198:489–97. doi:10.1677/JOE-08-0027.
Bandiera R, Vidal VPI, Motamedi FJ, Clarkson M, Sahut-Barnola I, von Gise A, Pu WT, Hohenstein P, Martinez A, Schedl A. WT1 maintains adrenal-gonadal primordium identity and marks a population of AGP-like progenitors within the adrenal gland. Dev Cell. 2013;27:5–18. doi:10.1016/j.devcel.2013.09.003.
Acknowledgments
We thank the members of the Breault laboratory, J. Majzoub, W. Engeland, and P. Barrett, for their helpful discussions. This research was supported by R01 DK 084056.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG
About this chapter
Cite this chapter
Pignatti, E., Leng, S., Carlone, D.L., Breault, D.T. (2018). Adrenal Zonation and Development. In: Levine, A. (eds) Adrenal Disorders. Contemporary Endocrinology. Humana Press, Cham. https://doi.org/10.1007/978-3-319-62470-9_1
Download citation
DOI: https://doi.org/10.1007/978-3-319-62470-9_1
Published:
Publisher Name: Humana Press, Cham
Print ISBN: 978-3-319-62469-3
Online ISBN: 978-3-319-62470-9
eBook Packages: MedicineMedicine (R0)