Abstract
After varroa invaded Europe in the mid of twentieth century, a few populations of honeybee colonies have been found to survive the mite. This chapter describes the case of natural selection of honeybees in France against varroa. Different hypotheses have been tested to explain this phenomenon, such as resistance of the bees to the mite or to the associated viruses and the lower virulence of the mites. We found that the reproduction of the mite and/or the varroa sensitive hygiene are probably key factors in the survival of those bees. Other varroa resistant honeybee populations have been found in several other countries and are also described as well as the putative mechanisms of survival. Finally, we discuss the interest of those bees for scientists and beekeepers in the framework of honeybee selection and describe the successful approaches lead by scientists for honeybee selection on a specific trait.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
When the varroa mite started to invade Europe in the mid of twentieth century, untreated honeybee colonies could not survive more than 1 or 3 years as the number of mites could sometimes exceed 10.000 per colony. As a result, many untreated colonies, particularly feral colonies, died. Few acaricides were used to control the mite and, as it does happen commonly in pest control, the mite became resistant to fluvalinate, a pyrethroid previously very efficient (Milani 1995). Up to now, the mite has become resistant to most of the chemical acaricides, except for amitraz in France. Having only one acaricide efficient for controlling the mite is a stressful situation which requires investments in the setting up of other acaricides with different targets to allow the rotation of treatment and so avoid varroa resistance. Indeed, other varroa control methods have been set up, including physical or mechanical controls as well as the use of more ‘natural’ chemical compounds such as acids and essential oils. But those methods are usually time-consuming and with a variable efficacy. Interestingly, since the mite invaded Europe, its biology and the varroa/honeybee relationships have been extensively studied leading to the publication of many scientific articles and making this host–parasite model one of the most extensively studied (Rosenkranz et al. 2010).
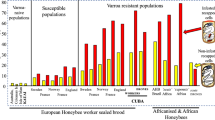
Nevertheless, in 1994, feral and abandoned untreated colonies were observed to have survived for a few years in the west of France. To confirm this phenomenon, with the help of beekeepers, we collected 70 honeybee colonies which had been untreated for at least 3 years and looked well developed and healthy. Colonies collected in the north of France were placed in an apiary in the west of France close to Le Mans, and colonies collected in the south were placed in Avignon. The 70 colonies were observed for their survival, swarming, and honey production. They were not managed for honey production but just left by themselves. The queens were individually marked, and the colonies cheeked twice a month for health and development. Interestingly, those colonies survived, on average, for 6.5 years, the best being 15 years (Le Conte et al. 2007). This ability of bees to survive varroa may be due to a honeybee tolerance or resistance to the mite or its associated virus, to a lower mite virulence or to the environment.
To test the hypothesis of less virulent mites, we set up population molecular markers, mitochondrial and nuclear (microsatellites) (Solignac et al. 2003; Navajas et al. 2002) and sampled varroa mite populations in France, Europe, and few different other countries. We did not find genetic variability and concluded that the mites had a clonal population structure at this time (Solignac et al. 2005). It means that if the mites were less virulent, this would have been based on a limited number of genes. We also looked at the viruses present in the surviving bees compared to sensitive ones and found that the surviving bees had fewer viruses. We injected the bees with virus and could not find differences in survival between the two kinds of bees, suggesting that the surviving bees had fewer viruses because they had fewer varroa mites (see below), as described in Büchler et al. (2010).
Interestingly, when the mite first invaded our country, we could find high numbers of mites (up to 10.000) in the honeybee colonies and limited deformed wing virus (DWV) symptoms. A few years later we observed the opposite, i.e., lower numbers of mites in the colonies and higher DWV symptoms (Le Conte personal communication). It has been demonstrated that the varroa mite can actively modify DWV population structure in honeybee colony populations (Martin et al. 2012) as it can also do for other viruses (Mondet et al. 2014). More recently, we looked at the DWV in our resistant populations in the west of France compared to sensitive honeybee colonies and found a different recombination event between this virus and the Varroa destructor virus (VDV), which is very close to DWV (Dalmon et al., accepted in Scientific Reports). This virus could have evolved into a less virulent form which could explain part of the survival ability of those bees. To conclude this virus story, we must acknowledge that there are strong interactions between the mite, the viruses, and the honeybee host, and that the viruses can evolve and mutate reducing or even increasing their virulence to the bees. So nothing is fixed in those interactions which change over time.
We also tested the resistance hypothesis of those varroa surviving bees. Resistant hosts are able to maintain the parasite population at lower levels than what susceptible hosts face. Varroa population dynamics are much more important in sensitive colonies compared to our surviving colonies (Buechler et al. 2010). This may be due, at least partly, to the development of social immune strategies by the colonies. Indeed, the varroa mite reproduces less in our varroa-resistant bees compared to sensitive bees from colonies set in the same apiaries (Locke et al. 2012) and on many occasions female mites fail to effectively reproduce. This trait is known as Suppressed Mite Reproduction (SMR, see Harbo and Harris 2005). In the USA, a population has been bred for survival to varroa, initially through selection of lower mite growth in the colonies (see below). It has been shown that the colonies present the SMR trait, and this phenomenon is due to a behavior of adult bees that actively target and remove brood cells that are infested by varroa. This mechanism is called Varroa-Sensitive Hygiene (VSH) and is a specific form of hygienic behavior. However, it is possible that colonies display signs of mite reproduction failures (SMR) if infested bee larvae or pupae inhibit mite reproduction. This mechanism and VSH are being investigated in the French surviving populations and will help decipher the mechanisms underlying the SMR trait (Mondet et al. 2016).
In addition, based on the grooming behavior, we developed controlled behavioral experiments to test the ability of the bees to recognize and attack the mite. We found that resistant bees are doing much better compared to sensitive bees (Martin et al. 2001). Gene expression analysis has also revealed that the resistant bees overexpress genes related to stimuli and olfaction (Navajas et al. 2008), which fit with the fact that their antennae are more sensitive to varroa odorant compound compared to sensitive bees (Martin et al. 2001). It is interesting to notice that antennae of bees which express VSH behavior overexpress genes related to olfaction (for instance, odorant-binding proteins) (Mondet et al. 2015).
Moreover, comparing propolis harvested by sensitive or resistant colonies in the same location had shown that concentration of caffeic acid and caffeates was higher in propolis collected by our surviving colonies. Those compounds have pronounced and diverse biological properties on honeybee health (Popova et al. 2014). More studies are needed to confirm the hypothesis that surviving bees would be more capable to go to the ‘pharmacy’ to fight diseases.
What has happened to these bees since we published those results in 2007? Once every two years, we graft queen larvae from the three best colonies in each apiary (west and south of France) to get 20 colonies. The queens are naturally mated by local drones. About 30–35% of the colonies die within 18 months, but the rest of the colonies are good candidates for surviving to the mite, so the stock still survives efficiently.
We are focusing on the varroa mite survival, but it should be clear that those colonies are also resistant to other pathogens as they are not treated or managed against any disease. Those survival colonies swarmed (about 40% depending on the year) and similar varroa-treated colonies produced 1.7 times more honey (Le Conte et al. 2007). The next step in this research is to investigate how those colonies would behave, regarding varroa load and honey production, when they are managed under standard beekeeping management. Preliminary trials show that they can survive in a professional beekeeping environment. Further, breeding efforts or compromises may be required to get the surviving colonies to the standard of honey production expected by the industry. Nevertheless, the surviving populations give evidence that untreated local honeybee colonies can survive the mite, which can provide an important basis for integrated varroa management. Moreover, those honeybee populations are interesting for the beekeeping industry, but also at an ecological point of view, as they are potential sources for generating feral colonies which are keys for pollination especially in areas where domestic beekeeping is not concentrated.
There are other honeybee populations naturally surviving Varroa. The best example would be the Africanized bees (AFB) which invaded the Americas from Brazil to the southern states of the USA. Initially considered as a pest because of their aggressiveness, they survive numerous stresses. It has been shown that varroa offspring mortality is a major component of this resistance phenomenon (Mondragon et al. 2006). More recently, Rivera-Marchand et al. (2012) described similar AFB populations in Puerto Rico. They are surviving the mite, but do not show similar aggressiveness. It is surprising to notice that beekeepers in America are not interested in taking advantage of those naturally varroa surviving bees as they could try to select against aggressiveness, or even import bees from Puerto Rico.
Other naturally surviving populations have been recently identified in Norway and in the Netherlands. At the moment, European scientists are exchanging queens to look at the effect of the environment on the survival of the bees (COLOSS, Ricola Foundation Program; The Persephone Charitable and Environmental Trust).
Other varroa-resistant honeybee populations have been obtained through human selection. This is the case in Sweden (Fries et al. 2006) and in France (Kefuss et al. 2004). The scientists used what they called the ‘Bond’ test: ‘Live and let die.’ Basically, they brought a large number of colonies from different strains in the same location and observed the survival. It is a slightly different approach than ours as in our case, the colonies were already observed as surviving in a local environment. This approach was successful and led to numerous scientific publications (Locke 2016).
Another step in the selection of bees resistant to the mite is to choose a trait to select for, hypothesizing that it would lead to resistant bees. As an example, this quantitative genetic approach has been successfully developed by the USDA in Baton Rouge (USA) using the SMR (Harbo and Harris 2005) and the VSH traits (Harbo and Harris 2009) as a basis for selection.
It is well known that Apis cerana (the original host of varroa) is varroa resistant since colonies of Asian honeybees do not die from mite infestation; we now know that it can be the case also for Apis mellifera as naturally Varroa surviving honeybee colonies occur in different places. Different causes can explain that phenomenon, such as individual and social immunity, olfaction, propolis, viruses, varroa reproduction, swarming. The causes may not be the same in the different bee populations, but the good news is that Apis mellifera can survive Varroa mite infestations without treatments. We have surviving bee populations available from naturally surviving, ‘bond test’ and trait-based selected populations. This is an enormous chance that we have to take and go further in selecting varroa-resistant bees from the populations we want to work with.
Different tools should be used in the future to help beekeeping on this task in the framework of IPM. One could be to identify the compounds involved in the recognition of the mite by the honeybees and use them to evaluate the ability of the colony to destroy the mites in beekeeping. We have recently identified chemicals which are good candidates in this framework. Another possibility is to search for genomic markers, as SNPs, which could be linked to the SMR and/or VSH behavior. Those could also be used as markers to select resistant bees.
References
Buechler R, Berg S, Le Conte Y (2010) Breeding for resistance to Varroa destructor in Europe. Apidologie 41(3):393–408
Fries I, Imdorf A, Rosenkranz P (2006) Survival of mite infested (Varroa destructor) honey bee (Apis mellifera) colonies in a Nordic climate. Apidologie 37(5):564–570
Harbo JR, Harris JW (2005) Suppressed mite reproduction explained by the behaviour of adult bees. J Apic Res 44(1):21–23
Harbo JR, Harris JW (2009) Responses to Varroa by honey bees with different levels of Varroa Sensitive Hygiene. J Apic Res 48(3):156–161
Kefuss J, Vanpoucke J, De Lahitte JD, Ritter W (2004) Varroa tolerance in France of intermissa bees from Tunisia and their naturally mated descendants: 1993-2004. Am Bee J 144(7):563–568
Le Conte Y, De Vaublanc G, Crauser D, Jeanne F, Rousselle JC, Becard JM (2007) Honey bee colonies that have survived Varroa destructor. Apidologie 38(6):566–572
Locke B (2016) Natural Varroa mite-surviving Apis mellifera honeybee populations. Apidologie 47(3):467–482
Locke B, Le Conte Y, Crauser D, Fries I (2012) Host adaptations reduce the reproductive success of Varroa destructor in two distinct European honey bee populations. Ecol Evol 2(6):1144–1150
Martin C, Provost E, Roux M, Bruchou C, Crauser D, Clement JL, LeConte Y (2001) Resistance of the honey bee, Apis mellifera to the acarian parasite Varroa destructor: behavioural and electroantennographic data. Physiol Entomol 26(4):362–370
Martin SJ, Highfield AC, Brettell L, Villalobos EM, Budge GE, Powell M, Nikaido S, Schroeder DC (2012) Global honey bee viral landscape altered by a parasitic mite. Science 336(6086):1304–1306
Milani N (1995) The resistance of Varroa-jacobsoni Oud to pyrethroids—a laboratory assay. Apidologie 26(5):415–429
Mondet F, de Miranda JR, Kretzschmar A et al (2014) On the front line: quantitative virus dynamics in honeybee (Apis mellifera L.) colonies along a new expansion front of the parasite Varroa destructor. PLoS Pathog 10(8)
Mondet F, Alaux C, Severac D, Rohmer M, Mercer AR, Le Conte Y (2015) Antennae hold a key to Varroa-sensitive hygiene behaviour in honey bees. Sci Rep 5:10454
Mondet F, Kim SH, de Miranda JR, Beslay D, Le Conte Y, Mercer AR (2016) Specific cues associated with honey bee social defence against Varroa destructor infested brood. Sci Rep 6:25444
Mondragon L, Martin S, Vandame R (2006) Mortality of mite offspring: a major component of Varroa destructor resistance in a population of Africanized bees. Apidologie 37(1):67–74
Navajas M, Le Conte Y, Solignac M, Cros-Arteil S, Cornuet JM (2002) The complete sequence of the mitochondrial genome of the honeybee ectoparasite mite Varroa destructor (Acari: Mesostigmata). Mol Biol Evol 19(12):2313–2317
Navajas M, Migeon A, Alaux C et al (2008) Differential gene expression of the honey bee Apis mellifera associated with Varroa destructor infection. BMC Genomics 9
Popova M, Reyes M, Le Conte Y, Bankova V (2014) Propolis Chemical Composition and Honeybee Resistance against Varroa Destructor. Nat Prod Res: 1–7
Rivera-Marchand B, Oskay D, Giray T (2012) Gentle Africanized bees on an oceanic island. Evol Appl 5(7):746–756
Rosenkranz P, Aumeier P, Ziegelmann B (2010) Biology and control of Varroa destructor. J Invertebr Pathol 103:S96–S119
Solignac M, Vautrin D, Pizzo A, Navajas M, Le Conte Y, Cornuet JM (2003) Characterization of microsatellite markers for the apicultural pest Varroa destructor (Acari: Varroidae) and its relatives. Mol Ecol Notes 3(4):556–559
Solignac M, Cornuet JM, Vautrin D, Le Conte Y, Anderson D, Evans J, Cros-Arteil S, Navajas M (2005) The invasive Korea and Japan types of Varroa destructor, ectoparasitic mites of the western honeybee (Apis mellifera), are two partly isolated clones. Proc R Soc B-Biol Sci 272(1561):411–419
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing AG
About this chapter
Cite this chapter
Le Conte, Y., Mondet, F. (2017). Natural Selection of Honeybees Against Varroa destructor . In: Vreeland, R., Sammataro, D. (eds) Beekeeping – From Science to Practice. Springer, Cham. https://doi.org/10.1007/978-3-319-60637-8_12
Download citation
DOI: https://doi.org/10.1007/978-3-319-60637-8_12
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-60635-4
Online ISBN: 978-3-319-60637-8
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)