Abstract
People in modern, affluent societies are living longer but also becoming increasingly overweight. With increased life expectancy comes increased risk of developing age-related cognitive decline and neurodegenerative diseases, such that an increasing proportion of life may be lived with cognitive impairment as age increases. Obesity is associated with poorer cognitive function in elderly subjects, and often leads to ill-health arising from various complications such as metabolic syndrome and type-2 diabetes mellitus. This chapter provides an overview of the effects of administering pan-phosphodiesterase-4 (PDE4) inhibitors to animal models of cognitive ageing, Alzheimer’s disease, frontotemporal dementia, fragile X syndrome, obesity and diabetes. Inhibition of the PDE4B subtype specifically is discussed as an approach to avoid the emetic side effects of pan-PDE4 inhibitors, whilst retaining their therapeutic effects. Finally, the findings of rodent studies that employ genetic and pharmacological approaches to specifically target PDE4B are discussed in relation to the potential utility of PDE4B-selective inhibitors for the treatment of cognitive impairment and obesity-related metabolic diseases.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
- Phosphodiesterase-4
- PDE4B
- cAMP signalling
- Rolipram
- Cognitive enhancement
- Alzheimer’s disease
- Frontotemporal dementia
- Fragile X
- Obesity
- Diabetes
- Traumatic brain injury
1 Introduction
Cyclic adenosine monophosphate (cAMP) is a second messenger used for intracellular signal transduction to regulate a wide range of biological processes, such as the cellular response to neurotransmitters in the central nervous system (CNS) (Houslay et al. 2007). Intracellular concentrations of cAMP are regulated by its synthetase, adenylyl cyclase, and its hydrolase, the cAMP-specific phosphodiesterases. Phosphodiesterase-4 (PDE4) is the predominant enzyme that selectively hydrolyses and inactivates cAMP. Thus, inhibitors of PDE4 provide a pharmacological strategy to increase prevailing cAMP levels in relevant target tissues. The PDE4 family comprises four subtypes (A–D), each encoded by a separate gene, which have clearly distinct expression patterns at the regional and cellular levels (Richter et al. 2013). These enzymes are expressed throughout the body, with the highest levels of activity and protein found in the brain (Richter et al. 2013).
PDE4B is expressed in various isoforms across a wide range of human tissues, including adipose tissue, whole blood, liver and brain (Bunnage et al. 2015). All five PDE4B isoforms identified in mammals contain the catalytic domain (Zhang 2009). The long isoforms PDE4B1 (736 a.a.), PDE4B3 (721 a.a.) and PDE4B4 (659 a.a.) contain both Upstream Conserved Region 1 (UCR1) and Upstream Conserved Region 2 (UCR2); the short isoform PDE4B2 (564 a.a.) lacks UCR1; and the super-short isoform PDE4B5 (484 a.a.) lacks UCR1 and has a truncated UCR2 (Shepherd et al. 2003; Cheung et al. 2007; Fatemi et al. 2008). Long isoforms are activated by protein kinase A (PKA)-mediated phosphorylation of a single serine residue in UCR1 (MacKenzie et al. 2002), and are inhibited by extracellular signal-related kinase-2 (ERK2)-mediated phosphorylation of a single serine residue located within the C-terminal portion of the catalytic domain (Baillie et al. 2000). Conversely, the short isoform PDE4B2 is activated by ERK2 phosphorylation (Baillie et al. 2000). PDE4B2 is the predominant and often only isoform present in tissues outside the CNS, while the CNS contains the long (PDE4B1, 4B3, 4B4), short (PDE4B2) and super-short (PDE4B5) isoforms (Bunnage et al. 2015). This chapter discusses the potential utility of PDE4 inhibitors, especially those selective for PDE4B, in the treatment of cognitive impairment and obesity-related metabolic diseases.
2 Effects of Pan-PDE4 Inhibitors on Cognitive Impairment
Long-term memory formation following new protein synthesis relies on the expression of genes upregulated by cAMP response element binding protein (CREB) (Guzowski and McGaugh 1997). A transcription factor, CREB is phosphorylated and activated by PKA via an increase in cAMP, and regulates gene transcription by binding to the cAMP response element on target genes (Habener 1990). As a consequence, PDE4 enzymes, as regulators of cAMP gradients and ultimately CREB, are promising targets for the development of cognition-enhancing agents (Randt et al. 1982; Ghavami et al. 2006; Richter et al. 2013). PDE4 inhibitors are drugs used to block the degradative action of PDE4 on cAMP. The prototypical PDE4 inhibitor is rolipram (CAS: 61413-54-5), a non-subtype-selective or pan-PDE4 inhibitor (targeting all of four subtypes) with a half-life of 3 h that readily passes through the blood-brain barrier, is rapidly cleared by the kidneys, and does not accumulate in the tissues (Schneider 1984; Krause and Kuhne 1988). Rolipram engages the N-terminal UCR2 domain (truncated in super-short isoform PDE4B5), thereby closing UCR2 across the PDE4 active site and preventing access of cAMP (Burgin et al. 2010). It has an inhibitory concentration 50% (IC50) value of 225 nM against PDE4B and 228 nM against PDE4D (Burgin et al. 2010; Fox et al. 2014). PDE4B knockout (KO; Pde4b tm1Mct) mice (see Sects. 5.3.1 and 5.5.1) show a partial (~50%) reduction in sensitivity to rolipram (3.2 mg/kg) in a conditioned avoidance response task (Siuciak et al. 2007), suggesting that the effect of rolipram in this test is mediated, in part, through PDE4B.
The primary effect of inhibition of PDE4 is the elevation in intracellular levels of cAMP (Scheider 1984), which is assumed to regulate transcription of various target genes by increasing cAMP-dependent PKA activity. Pan-PDE4 inhibitors, such as rolipram, have shown therapeutic benefit in preclinical models of psychiatric and neurological conditions including memory and cognition impairments (Ghavami et al. 2006; Richter et al. 2013). It has thus been postulated that PDE4 inhibition may promote improved synaptic function or ‘synaptic resilience’ (Bales et al. 2010). This section gives an overview of inhibition of PDE4 as a potential therapeutic strategy for cognitive impairment, focussing on evidence from animal model studies of cognitive ageing, Alzheimer’s disease, frontotemporal dementia, and fragile X syndrome (Table 5.1).
2.1 Age-Associated Cognitive Decline
Cognitive ageing is a lifelong process of gradual, ongoing, yet highly variable changes in cognitive function that occurs as people get older. Mental capabilities that decline from middle age onwards include aspects of memory, executive functions, processing speed and reasoning. All of these so-called ‘fluid’ mental abilities are important for carrying out everyday activities, living independently and leading a fulfilling life (Deary et al. 2000). Individuals may, for example, have difficulty driving a car, making financial decisions, or following directions for prescription medicines (Blazer et al. 2015). Life expectancy in developed countries has increased substantially (Hicks and Allen 1999), such that an increasing proportion of life may be lived with cognitive impairment as age increases.
In a Barnes maze, aged (18-month-old) C57BL/6 mice given rolipram (0.05 μM/day, i.p.) on days 15–40 of training showed a reduced number of errors, and an increased percentage reached a learning criterion, compared with controls (Bach et al. 1999). In contextual fear conditioning, rolipram (0.1 μmol/kg, s.c.) given 30 min before training increased contextual freezing after 24 h in both young (12–16-week-old) and aged (18 month-old) C57BL/6 mice compared with controls (Barad et al. 1998). The effective dose increased cAMP signalling without affecting basal levels of cAMP (Barad et al. 1998). In a novel object recognition test, aged (23-month-old) rats given rolipram (0.1 mg/kg) immediately after training showed enhanced memory after 24 h, compared with controls (de Lima et al. 2008). The efficacy of this post-training treatment ruled out the possibility that the rolipram effects were due to drug-induced behavioural or sensory alterations at training. In a Morris water maze, aged (20-month-old) C57BL/6NTac mice given another pan-PDE4 inhibitor, HT-0712 (0.15 mg/kg/day, i.p.), 20 min before training over 15 days demonstrated enhanced memory of the location of an escape platform after 10 and 15 days of training compared with controls (Peters et al. 2014). In both contextual and trace fear conditioning, aged (20–24-month-old) C57BL/6NTac mice given HT-0712 (0.1 mg/kg, i.p.) 20 min before training showed increased freezing after 24 h compared with controls (Peters et al. 2014).
Adult rats of an unspecified age were given four different doses of rolipram (0.01, 0.03, 0.1, and 0.3 mg/kg) 3 h after the last training trial on each of 4 days in the Morris water maze (Hosseini-Sharifabad et al. 2012). Rats given 0.03 mg/kg rolipram showed a shorter escape latency and distance swum on the second day of training. In a probe trial at the end of training, with the escape platform removed, rats treated with 0.03 mg/kg rolipram spent more time at the target location, indicating enhanced spatial reference memory. Other doses of rolipram did not show any differences compared with controls. Thus, rolipram enhanced spatial memory consolidation in an inverted U-shaped dose-response curve, such that higher doses of rolipram were not as efficacious as lower doses in improving hippocampus-dependent learning (Hosseini-Sharifabad et al. 2012). This might be explained by sedative effects, which have been observed in mice following the administration of rolipram at higher doses (≥0.12 mg/kg), demonstrated by dose-dependent hypoactivity in the open field task (Griebel et al. 1991; Barad et al. 1998; Hu et al. 2011).
Presynaptic ChAT activity was found to be reduced in the frontal cortex and the hippocampus of aged (24-month-old) rats, but was restored to young-adult control levels by rolipram treatment (0.1 mg/kg/day) for 14 days (Asanuma et al. 1993). Since the 5′ upstream region of the rat ChAT gene contains a CREB binding site (Bejanin et al. 1992), it is possible that the function of rolipram in improving memory in aged rodents may be partly implemented through the enhancement of ChAT activity via phosphorylation of CREB (pCREB).
By contrast with the rodent findings, aged (18–30 years) macaque monkeys given rolipram 1 h prior to training in a spatial delayed response task showed no difference in working memory (number of trials correct) at lower doses (0.01–1 μg/kg, i.m.), and performed significantly worse than vehicle-treated controls following the highest dose (0.01 mg/kg, i.m.) (Ramos et al. 2003). Another pan-PDE4 inhibitor, etazolate (delivered near the recorded neurones by iontophoresis), was found to reduce the memory-related firing rate of prefrontal cortical neurones in a single aged (17-year-old) male macaque as it performed the oculomotor delayed response spatial working memory task (Wang et al. 2011a). The apparent discrepancy between the aged rodent and monkey findings may exist because only hippocampus-dependent reference memory was assessed in the rodent studies, whereas only prefrontal cortex-dependent working memory was assessed in the monkey studies. The impairment of prefrontal cortical function by PDE4 inhibition in aged monkeys may be related, in part, to possible decreases in cortical PDE4 subtype expression that occur with age. In aged (25-month-old) rats, PDE4D mRNA expression is reduced in the cortex and cerebellum, and PDE4A mRNA expression is reduced in the striatum, whereas PDE4B mRNA expression in the hippocampus, cortex, striatum and cerebellum shows no change compared with young (5-month-old) rats (Kelly et al. 2014).
2.2 Alzheimer’s Disease
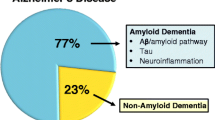
Alzheimer’s disease (AD) is a chronic neurodegenerative condition characterised by progressive cognitive decline, memory loss and personality changes, eventually leading to death. The prevalence of AD increases with life expectancy, being rare before the age of 60 but affecting more than one-third of people over the age of 90 (Querfurth and LaFerla 2010). Absolute confirmation of diagnosis can only be made by a post-mortem examination of the brain to demonstrate the neuropathological changes first described by Alois Alzheimer, namely plaques and tangles (Alzheimer 1907). The extracellular plaques, formed by fibrillary aggregates of amyloid-β (Aβ) peptides, together with the intraneuronal neurofibrillary tangles formed by hyperphosphorylated tau protein, are thought to be the causative agents in AD (Hardy 2009).
There is a growing body of evidence suggesting that neuroinflammation can contribute to AD pathogenesis (Heppner et al. 2015). High levels of the pro-inflammatory cytokine tumour necrosis factor-alpha (TNFα) have been detected in AD patients (Fillit et al. 1991; Tarkowski et al. 1999). A recent study used PET imaging to measure TSPO, a biomarker for inflammation, in amyloid-positive AD patients and amyloid-negative controls (Kreisl et al. 2013). AD was found to be associated with increased inflammation in the brain, mostly in temporal and parietal regions known to be affected by amyloid plaque pathology. In these same regions, increased inflammation correlated with amyloid burden and with the degree of cognitive impairment. Early-onset AD patients showed greater inflammation than late-onset patients (Kreisl et al. 2013), which might explain the more precipitous disease course typically seen in early-onset patients. The immune system may thus provide novel routes for the treatment of AD.
Cyclic AMP signalling is involved in the regulation of postsynaptic, protein synthesis-dependent long-term potentiation (LTP) in the hippocampus (Kandel 2001), a form of synaptic strengthening thought to underlie the formation and persistence of memory (Malenka and Bear 2004). Rolipram has been shown to potentiate LTP in hippocampal slice preparations (Barad et al. 1998). Aβ peptides strongly inhibit LTP in hippocampal slices (Cullen et al. 1997; Itoh et al. 1999; Vitolo et al. 2002), but Aβ-induced inhibition of LTP is reversed by rolipram (Vitolo et al. 2002), suggesting a direct effect of Aβ on the cAMP signalling pathway. Similarly, Aβ-induced reduction in the spine density of pyramidal hippocampal neurones is attenuated by rolipram (Smith et al. 2009). Treatment with rolipram (0.5 mg/kg/day, i.p.) beginning 12–13 prior to the onset of training reversed the impairments in Morris water maze and passive avoidance tests of rats infused with aggregated Aβ peptides into the hippocampal CA1 (Cheng et al. 2010; Wang et al. 2012). Rolipram also reversed the LTP deficit of hippocampal slices from 3-month-old APP/PSEN1 double-transgenic mice, which overexpress both mutant APP-K670N,M671L under the control of the hamster PRNP promoter (Tg(APPSWE)2576Kha) and PSEN1-M146L under the control of the human PDGFB promoter (Tg(PDGFB-PSEN1M146V)3Jhd) (Gong et al. 2004). Acute rolipram treatment (0.03 mg/kg, s.c.) 30 min before training reversed the deficit of APP/PSEN1 mice in contextual fear conditioning but not in a radial-arm water maze. Chronic rolipram treatment (0.03 mg/kg/day, s.c.) for 3 weeks, followed by a 3-month hiatus, showed persistent effects by reversing the deficits of 6-month-old APP/PSEN1 mice in hippocampal LTP, contextual fear conditioning, a radial arm water maze and a Morris water maze, but had no effect on Aβ load (Gong et al. 2004). Pyramidal hippocampal neurones from APP/PSEN1 mice given rolipram (0.03 mg/kg) daily for 3 weeks from either 3 or 15 months of age showed increased spine density, dendrite area and diameter, and spine head diameter compared with neurones from vehicle-treated APP/PSEN1 mice (Smith et al. 2009). Rolipram (0.1 mg/kg, i.p.) given 30 min before training also reversed the deficit of 9-month-old mutant APP-K670N,M671L transgenic mice in contextual fear conditioning, but had no effect on contextual conditioning in wild-type (WT) mice (Comery et al. 2005).
Several observations suggest that the beneficial effects of rolipram in rodent models of AD are mediated, at least in part, through PDE4B. The transcription of PDE4B is upregulated in area CA1 of the hippocampus after LTP induction (Ahmed and Frey 2005), suggesting a specific role for PDE4B in this form of synaptic plasticity. PDE4B transcription is also upregulated in microglial cells exposed to Aβ peptides, resulting in an increased production of TNFα, but rolipram markedly reduces the release of TNFα from Aβ-stimulated microglia cells (Sebastiani et al. 2006). In inflammatory cells from PDE4B, but not PDE4A or PDE4D, KO mice, TNFα production in response to an inflammatory stimulus (lipopolysaccharide, LPS) is reduced by over 50% (Jin and Conti 2002; Jin et al. 2005), while PDE4B-selective inhibitors (see Sect. 5.5.1) similarly blunt monocytic production of TNFα in vitro and in vivo (Naganuma et al. 2009; Suzuki et al. 2013). PDE4B mRNA expression across the brain remains stable throughout the ageing process in rats, whereas aged (25-month-old) rats show reduced expression of PDE4A and PDE4D mRNA in some brain regions (Kelly et al. 2014). Finally, PDE4B is the only subtype of PDE4 expressed in the locus coeruleus (Cherry and Davis 1999), a brain region affected by neurofibrillary degeneration early in the course of events leading to AD (Grudzien et al. 2007).
2.3 Frontotemporal Dementia
Mutations in the MAPT gene encoding the tau protein cause frontotemporal dementia with parkinsonism-17 (FTDP-17), or Pick’s disease, a rare neurodegenerative disorder that affects the frontal and temporal lobes of the brain. Clinical manifestations of FTDP-17, including behavioural and personality changes, cognitive impairment, and motor symptoms, usually become noticeable in a person’s forties or fifties (Hutton et al. 1998). FTDP-17 is a tauopathy, characterized by an abnormal build-up of tau proteins in neurones, accumulating into spherical aggregations known as ‘Pick bodies’ (Piguet et al. 2011). Expression of human tau containing the most common FTDP-17 mutation, P301L (Hutton et al. 1998), in forebrain neurones under the control of the Camk2a promoter results in progressive age-related neurofibrillary tangles, loss of neurones and generalized forebrain atrophy, and spatial reference memory impairments in rTg(tauP301L)4510 transgenic mice (Santacruz et al. 2005). At 3–4 months, these mice model early-stage disease; by 8 months they resemble a more severe stage of the human disease.
Tau can be degraded by both lysosomal processes, such as autophagy, and the ubiquitin-proteasome system (Lee et al. 2013). Cyclic AMP-dependent PKA phosphorylation of the proteasome enhances proteolytic activity (Asai et al. 2009). Exposure of cortical brain slices from rTg(tauP301L)4510 mice aged 3–4 months to rolipram for 8 h reduced the amounts of total and insoluble tau (Myeku et al. 2016). To assess the effect of rolipram on tauopathy-associated cognitive impairment, 3–4 month-old rTg(tauP301L)4510 mice were given rolipram (0.03 mg/kg, i.p., twice daily) for 21 days and then tested for spatial learning in the Morris water maze 6 h after the final administration. Untreated rTg(tauP301L)4510 mice showed an increased escape latency on the fourth (last) day of training, whereas the learning curve of rolipram-treated rTg(tauP301L)4510 mice was indistinguishable from that of WT mice (Myeku et al. 2016). Unfortunately, a probe trial to assess spatial memory retention at the end of learning was not undertaken. When experimental animals have deficits during probe trials, this dissociates memory from performance because measures recorded on probe trials are insensitive to swimming speed (Vorhees and Williams 2006).
PKA is known to phosphorylate tau at Ser214 (pS214) (Zhu et al. 2010). Brain extracts from rolipram-treated rTg(tauP301L)4510 mice showed a reduced total level of tau, but an increased level of pS214 tau, indicating that PKA activity was enhanced (Myeku et al. 2016). To assess proteome function in vivo, rTg(tauP301L)4510 mice were crossed with mice overexpressing a transgenic reporter for proteasome activity (ubiquitinated fragment fused to GFP). Normally, the GFP fusion protein is hydrolyzed rapidly by the proteasome, such that GFP is undetectable in normal cells (Peth et al. 2013). However, the transgenic rTg(tauP301L)4510-GFP mice showed accumulation of GFP puncta, indicating proteasome dysfunction, which increased with worsening tauopathy from 5 to 8 months. However, rolipram treatment reduced the levels of GFP in the hippocampus and cortex of rTg(tauP301L)4510-GFP mice (Myeku et al. 2016), indicating that rolipram improved proteasome activity in mice with tauopathy. This reinforces the concept that altered cAMP-PKA signalling is an important determinant of proteasome activity in vivo.
In 8–10-month-old rTg(tauP301L)4510 mice with more advanced tauopathy, rolipram treatment for 3 weeks had no significant benefits on proteasome activity, tau levels or cognition (Myeku et al. 2016). Nonetheless, given the ability of rolipram to promote proteasome activity, reduce tau and ameliorate spatial learning defects in mice during early-stage disease, it would be interesting to test whether enhancement of proteasomal function by rolipram treatment would enhance the clearance of pathogenic protein aggregates in animal models of other cerebral proteopathies, particularly Alzheimer’s disease, which is considered a secondary tauopathy.
2.4 Fragile X Syndrome
Inhibition of PDE4 has also shown promise as a therapeutic strategy for cognitive impairment in a neurodevelopmental disorder. Fragile X syndrome (FXS) is the most common monogenic cause of intellectual disability and autism (Bagni and Oostra 2013; McCary and Roberts 2013). The FMR1 gene contains a CGG repeat present in the 5′-untranslated region which can be unstable upon transmission to the next generation. The repeat is up to 55 CGGs long in the normal population. In patients with FXS, a repeat length exceeding 200 CGGs generally leads to methylation of the repeat and the promoter region, which is accompanied by silencing of the FMR1 gene. The disease is a result of lack of expression of the fragile X mental retardation protein (FMRP) (Verkerk et al. 1991), leading to severe symptoms, including intellectual disability, autistic behaviours, hyperactivity and childhood seizures. FMRP is an RNA binding protein whose function is incompletely understood, but is believed to be involved in translational regulation (Bagni and Oostra 2013). This is of particular interest because new protein synthesis is required for LTP (Malenka and Bear 2004).
A key to understanding FMRP function is to identify its RNA targets. High-throughput sequencing of RNAs isolated by crosslinking immunoprecipitation has been used to capture FMRP-mRNA interactions present in WT mouse brain, but not in Fmr1 KO mouse brain, at postnatal days 11–25 (Darnell et al. 2011). This approach identified a robust set of 842 FMRP target transcripts that includes ADCY1 and ADCY5, encoding adenylyl cyclases, and PDE4B, but not the other PDE4 subtypes. Thus, FMRP regulates the expression of proteins involved in the regulation of cAMP levels. Consistent with this finding, overexpression of FMRP was found to increase the production of cAMP experimentally induced by the adenylyl cyclase activator forskolin or prostaglandin E1 in neural cells (Berry-Kravis and Ciurlionis 1998).
It has been postulated that the ability of FMRP to repress translation of target mRNAs suggests that FXS may result from the overexpression of specific dosage-sensitive genes through loss of translational suppression (Darnell and Klann 2013). The expression levels of ADCY1, ADCY5 and PDE4B in brain from Fmr1 KO mice or FXS patients are unknown, but it might be expected that upregulation of ADCY1/5 would increase cAMP synthesis, whereas upregulation of PDE4B would increase cAMP hydrolysis. Platelets, lymphoblastoid cells and neural cells from FXS patients have shown unaltered basal levels of cAMP, but decreased levels of cAMP production (Berry-Kravis and Huttenlocher 1992; Berry-Kravis and Sklena 1993; Berry-Kravis et al. 1995; Kelley et al. 2007). Consistent with these findings, platelets and cortex from Fmr1 KO mice and heads from fmr1 null Drosophila fruit flies showed unaltered basal cAMP levels, but decreased levels of cAMP production (Kelley et al. 2007), indicating that a robust defect in cAMP production in FXS is conserved across species. These observations have led to the ‘cAMP theory of FXS’, which posits that alterations in the cAMP pathway may result, in part, from dysregulated expression of proteins involved in the cAMP cascade (Kelley et al. 2008).
The human and Drosophila FMR1 genes are reported to contain CREB binding sites and to be regulated by CREB-mediated gene transcription (Hwu et al. 1997; Smith et al. 2006; Kanellopoulos et al. 2012), such that elevating cAMP levels might be expected to upregulate FMRP expression, via activation of PKA and CREB. Consistent with this, the provision of rolipram (200 μM) in the diet for 5–6 h both restored fmr1 mRNA expression to WT levels and eliminated a robust olfactory learning deficit in heterozygous fmr1 null flies (Kanellopoulos et al. 2012). Rolipram also abrogated a long-term (24-h) olfactory memory deficit in heterozygous fmr1 null flies, but only when fed both before and after training (Kanellopoulos et al. 2012). In another fly study, chronic (9-day) dietary administration of rolipram at both low (50 μM) and high (500 μM) doses rescued immediate-recall (<2 min) and short-term (1 h) memory deficits of fmr1 null flies in a conditioned courtship-associative memory test, but only the high dose rescued developmental malformation of the mushroom bodies (Choi et al. 2015), important structures for olfactory learning and memory in Drosophila. This finding suggests that the cognitive phenotype is not irreversibly determined by pathogenic developmental circuitry. Acute (12-h) dietary administration of rolipram at 50 μM also rescued the immediate-recall of fmr1 null flies in the olfactory conditioning test (Choi et al. 2015).
Fmr1 KO mice have exhibited impaired novel object recognition in several studies (Ventura et al. 2004; Restivo et al. 2005; Pacey et al. 2011), but the effect of rolipram on this cognitive phenotype has not been tested. However, administration of rolipram (0.03 mg/kg/day, s.c.) for 8 weeks, followed by a 3–5 week hiatus, has been shown to abrogate a robust neurophysiological phenotype, enhanced long-term depression (LTD), in Fmr1 null mice (Huber et al. 2002; Choi et al. 2015).
3 Metabolic Effects of PDE4 Inhibition
The combined effects of less physical activity and the consumption of calorie-dense diets have led to unprecedented levels of overweight and obesity in affluent, sedentary societies (Baker 2015). As obesity rates rise among adults, metabolic complications of obesity, such as metabolic syndrome, are becoming more common. The main sign of metabolic syndrome is abdominal obesity, along with elevated blood pressure, elevated fasting plasma glucose, high serum triglycerides, and low high-density lipoprotein levels (Alberti et al. 2005). People with metabolic syndrome have a fivefold greater risk of developing type-2 diabetes (Stern et al. 2004) (see Sect. 5.3.2). Obesity can also exacerbate the age-related decline in physical and cognitive function in older people, and increases the risk for chronic diseases that result in poor quality of life, such as hypertension, coronary heart disease and stroke, osteoarthritis and cancer (Kopelman 2007; Wolf et al. 2007). Neuroimaging studies show increased brain atrophy in overweight and obese elderly subjects (Raji et al. 2010; Cherbuin et al. 2015). Obesity-related ill-health thus poses a serious, established and growing burden with consequences for individuals, healthcare services and wider society (Wang et al. 2011b). This section gives an overview of inhibition of PDE4 as a potential therapeutic approach for obesity and diabetes.
3.1 Obesity
The C57BL/6J and C57BL/6N mouse strains both develop obesity when allowed ad libitum access to a high-fat diet, but remain lean and physically normal when restricted to a standard diet (Collins et al. 2004; Nicholson et al. 2010; Podrini et al. 2013). Obesity in mice fed a high-fat diet leads to upregulation of pro-inflammatory processes (Pistell et al. 2010), increased production of reactive oxygen species (ROS) (Zhang et al. 2005), increased apoptosis and neurodegeneration, and decreased neurogenesis (Cai 2013), which together lead to decreased brain volumes, particularly of the hippocampus, and impaired memory and other cognitive deficits (Jeon et al. 2012).
When high-fat diet-fed C57BL/6J mice were treated with rolipram (2 mg/kg/day, oral gavage) for 12–14 weeks, they showed resistance to weight gain and had less body fat, even though their food intake was unaltered. These mice also showed an increased basal metabolic rate, with higher oxygen consumption and higher body temperatures in the fasting state, but unaltered physical activity levels. Furthermore, rolipram-treated mice had lower ROS levels in white adipose tissue (WAT), increased mitochondrial biogenesis in skeletal muscle, improved exercise tolerance on a treadmill, and increased glucose tolerance (Park et al. 2012). The dose of rolipram used (2 mg/kg) is greater than a dose (0.12 mg/kg) that showed sedative effects in mice in another study (Griebel et al. 1991). Consistent with the metabolic effects of rolipram, chronic obstructive pulmonary disease (COPD) patients treated with another pan-PDE4 inhibitor, roflumilast, showed reductions in body weight, with greater weight loss observed in individuals having a higher body mass index (BMI) (Gupta 2012). Roflumilast also shown an anti-obesity effect in women with polycystic ovary syndrome (PCOS), a disorder associated with insulin resistance and obesity. All of the women had been pretreated with metformin, the most widely used medication for type-2 diabetes (see Sect. 5.3.3), and were randomized to either continue on metformin alone, or in combination with roflumilast. Over 12 weeks, patients on metformin alone gained weight and showed increases in BMI and visceral adipose tissue (VAT) area, whereas those on metformin + roflumilast lost weight and showed reductions in BMI and VAT area (Jensterle et al. 2014).
Evidence that PDE4B may be a mediator of some of the metabolic effects of rolipram is provided by PDE4B KO mice, which are leaner, with lower fat pad weights, smaller adipocytes, and decreased serum leptin levels, when fed either a standard diet or a high-fat diet (Zhang et al. 2009), indicating that PDE4B deficiency can reduce adiposity. In WT mice, the high-fat diet decreased locomotor activity, but PDE4B deficiency almost completely blunted this reduction. PDE4B KO mice on a high-fat diet also showed lower levels of TNFα mRNA and macrophage infiltration in WAT, indicating that PDE4B deficiency can also suppress some important obesity-induced inflammatory changes. Although the basal level of cAMP was unaltered in adipocytes from PDE4B KO mice, PDE4B-deficient adipocytes showed a greater accumulation of cAMP when experimentally exposed to the adenylyl cyclase activator isoproterenol, suggesting that PDE4B is involved in cAMP regulation in adipocytes. Nevertheless, insulin sensitivity was not improved in PDE4B KO mice because no differences from WT mice were observed in levels of fasting serum glucose and insulin or in glucose tolerance and insulin tolerance tests (Zhang et al. 2009). Cross-species support for a role for PDE4B in the regulation of adiposity comes from the finding that genetic variation in PDE4B affects subcutaneous fat thickness in both pigs and humans (Lee et al. 2011). These findings suggest that PDE4B inhibitors could have utility in the treatment of obesity and associated metabolic complications.
3.2 Alcoholic Fatty Liver
Excessive alcohol consumption is associated with greater risk of obesity and large waist circumference (Arif and Rohrer 2005). In the liver, the first response to excessive alcohol consumption is the development of large fatty globules (hepatic steatosis). Although alcoholic fatty liver is reversible and typically has no associated symptoms, steatosis is the first stage of alcoholic liver disease, which can progress to alcoholic hepatitis and eventually cirrhosis (O’Shea et al. 2010). In the most widely used model for alcoholic liver injury, male C57BL/6J mice are fed the Lieber-DeCarli liquid diet containing ethanol [5% (v/v)] ad libitum for 4 weeks (Lieber et al. 1989). C57BL/6J is a strain of mice that voluntarily drink a large amount of ethanol (Yoneyama et al. 2008). In this alcoholic liver injury model, the mRNA levels for all PDE4 subtypes, PDE4A-D, are significantly upregulated 1–2 weeks after starting 5% ethanol compared with controls on an isocaloric liquid diet. This increase in PDE4 expression is accompanied by elevated PDE4 enzymatic activity and a decrease in cAMP and pCREB levels in the liver (Avila et al. 2016).
To examine the role of PDE4 in alcohol-induced hepatic steatosis, Avila et al. (2016) employed both pharmacological (rolipram; 5 mg/kg, 3 times/week, 4 weeks) and genetic (PDE4B KO) interventions to inhibit the activity of PDE4 and prevent the degradation of cAMP in the alcoholic liver injury model. Both rolipram and PDE4B KO led to significant reductions in fat accumulation and free fatty acid levels in the liver, and prevented the ethanol-mediated decrease in hepatic levels of cAMP and pCREB (Avila et al. 2016). The comparable effectiveness of both pan-PDE4 (rolipram) and specific PDE4B (PDE4B KO) inhibition suggests a predominant pathogenic role for PDE4B (among the PDE4 subtypes) in the development of alcohol-induced hepatic steatosis. PDE4B thus could serve as a therapeutic target in the treatment of alcoholic fatty liver disease. Although neither intervention for inhibiting PDE4 affected food consumption or ethanol metabolism (Avila et al. 2016), it is notable that lower doses of rolipram (0.5–1 mg/kg) were previously shown to reduce ethanol intake in male C57BL/6J mice in 24-h two-bottle choice tests (Hu et al. 2011; Blednov et al. 2014).
3.3 Diabetes
Diabetes mellitus in humans is a genetically and clinically heterogeneous group of glucose intolerance syndromes. Type-2 (non-insulin-dependent) diabetes is the more prevalent clinical form, in which obesity associated with progressively more severe insulin resistance is a common predictor of the pre-diabetic state. In patients with newly diagnosed type-2 diabetes, without COPD, roflumilast treatment led to reductions in glycated haemoglobin and fasting plasma glucose levels (Wouters et al. 2012). Consistent with this improvement in glycaemic variables, an animal model of type-2 diabetes, the Lepr db/db mouse (Hummel et al. 1966), showed reduced food and water consumption, reduced glycated haemoglobin levels, reduced blood glucose levels, increased fasted serum insulin levels, preserved pancreatic islet morphology and insulin production in pancreatic islet β cells, and minimal islet atrophy in response to 28-day treatment with roflumilast-N-oxide (3 mg/kg/day), the active metabolite of roflumilast (Vollert et al. 2012). Rolipram, roflumilast and roflumilast-N-oxide all increase serum levels of glucagon-like peptide-1 (GLP-1) (Park et al. 2012; Vollert et al. 2012; Ong et al. 2009), a cAMP-regulated gut hormone that increases insulin secretion from β cells (Gevrey et al. 2002; Holz and Habener 1992), and promotes satiety and suppresses energy intake in humans (Flint et al. 1998).
Type-1 (insulin-dependent) diabetes usually has an autoimmune T cell-mediated aetiology in which the pre-diabetic state is characterized by development of autoantibodies against certain proteins expressed by β cells, including insulin. The risk for development of type-1 diabetes is increased by obesity and may occur at an earlier age among obese individuals with a predisposition. Obesity also increases the risk for comorbidities among individuals with type-1 diabetes, especially metabolic syndrome, and microvascular and macrovascular diseases (Polsky and Ellis 2015). In an animal model of type-1 diabetes, the non-obese diabetic (NOD) mouse (Makino et al. 1980), treatment with rolipram (14 mg/kg, twice daily) for 4 weeks, from 12 to 16 weeks of age, had a lasting protective effect, associated with a significant reduction in the severity of insulitis, an inflammation of the islets of Langerhans (Liang et al. 1998). By 27 weeks of age, 80% of untreated NOD mice had diabetes, whereas only 20% of rolipram-treated mice were hyperglycaemic. Thus, 11 weeks after withdrawing drug therapy, the incidence of disease was still three to four times lower (Liang et al. 1998).
The inability of insulin to suppress hepatic glucose output is a major aetiological factor in the hyperglycaemia of type-2 and type-1 diabetic patients (DeFronzo et al. 1982). Biguanides such as metformin have an antihyperglycaemic action primarily by inhibiting hepatic glucose output via inhibition of glucagon-induced cAMP/PKA signalling (Miller et al. 2013). Although biguanides are known to phosphorylate AMP-activated protein kinase (AMPK), it is unclear how their glucose-lowering effect is related to AMPK activation. However, it was recently demonstrated using a small-molecule AMPK activator (compound 991) that AMPK activation antagonizes glucagon signalling by S304 phosphorylation and activation of PDE4B, thereby lowering cAMP levels and decreasing PKA activation in intact hepatocytes. In hepatocytes from mice bearing a liver-specific deletion of the two AMPK catalytic subunits, these effects of compound 991 treatment were lost (Johanns et al. 2016). PDE4B activation could thus explain the reduction in glucagon-stimulated cAMP levels by biguanides (Miller et al. 2013).
4 Adverse Effects of PDE4 Inhibition
4.1 Role of PDE4D in Nausea and Emesis
In principle, PDE4 inhibitors have considerable therapeutic potential. In practice, however, their clinical utility has been compromised by mechanism-associated side effects that limit maximally tolerated doses. Severe dose-limiting emesis following the administration of pan-PDE4 inhibitors has been observed humans and various animal species endowed with a vomiting reflex (Heaslip and Evans 1995; Robichaud et al. 2001). For example, in two separate macaque studies, the maximum tolerated doses of rolipram were 0.01 and 0.03 mg/kg, respectively, but the next highest dose of 0.05 mg/kg was not tolerated due to emesis (Ramos et al. 2003; Rutten et al. 2008). Although both PDE4B and PDE4D mRNAs are expressed in the area postrema, the chemosensitive trigger zone in the brain stem (Mori et al. 2010), PDE4D appears to be of particular importance in emesis.
As mice are unable to vomit, reduction in the duration of xylazine/ketamine-induced anaesthesia is used as a behavioural surrogate measure of emesis. Application of this procedure to PDE4B KO and PDE4D KO mice provided the following observations giving credence to the notion that PDE4D inhibition is responsible for the emetic side effect of pan-PDE4 inhibitors: (1) the duration of xylazine/ketamine-induced anaesthesia in PDE4D KO mice was significantly shorter compared with WT mice, whereas the anaesthetic affected PDE4B KO and WT mice to the same degree; (2) a PDE4 inhibitor significantly reduced the duration of anaesthesia in WT mice but not in PDE4D KO mice; (3) PDE4 activity in the brainstem of PDE4D KO mice was markedly lower compared with WT and PDE4B KO mice, indicating that PDE4D is the principle regulator of cAMP metabolism in the brainstem (Robichaud et al. 2002).
PDE4D-selective compounds that fully inhibit PDE4D activity have been found to potently reduce anaesthesia in the xylazine/ketamine test at doses (0.01 or 0.03 mg/kg) that showed maximal cognitive benefit in the novel object recognition test, thus corroborating the association of PDE4D inhibition with emesis. However, other PDE4D-selective compounds that only partially inhibit PDE4D had little or no effect on anaesthesia duration, even at a dose (3 mg/kg) 1000× that which showed maximal cognitive benefit (0.003 mg/kg) (Burgin et al. 2010), suggesting that partial inhibitors of PDE4D are likely to have better tolerability with regard to emesis.
4.2 Vascular Injury
In addition to the gastric adverse effects, a worrisome vascular toxicity has been observed in various organs during preclinical toxicology testing of pan-PDE4 inhibitors in laboratory animals including rats, dogs and monkeys (Giembycz 2005). Rats are particularly sensitive to the development of vascular injury following treatment with PDE4 inhibitors. In one study, rolipram was administered orally at a range of doses to rats for up 14 days, after which the animals were necropsied (Larson et al. 1996). No lesions were seen at a dose of 10 mg/kg, whereas inflammation and a necrotizing vasculitis in the mesentery and interstitial areas of the liver were clearly evident at a dose of 30 mg/kg. At the highest dose (100 mg/kg), rolipram was lethal (Larson et al. 1996). These findings demonstrate that repeated exposure to rolipram can cause vascular injury and death in rats, but it is noteworthy that the doses administered (≥30 mg/kg/day) are at least 60 times higher than the daily doses that have shown cognition-enhancing effects in rodent models of age-associated cognitive impairment (0.1 mg/kg; de Lima et al. 2008), Alzheimer’s disease (0.03–0.5 mg/kg; Gong et al. 2004; Comery et al. 2005; Cheng et al. 2010; Wang et al. 2012) and frontotemporal dementia (0.06 mg/kg; Myeku et al. 2016) (see Sect. 5.2). Vascular toxicity has not been reported in humans, although mesenteric vasculopathy is difficult to monitor in human clinical trials (Burgin et al. 2010). To date, little is known about the role of the four individual PDE4 subtypes (A–D) in the development of vascular injury, but mesenteric vasculopathy has not been observed in initial studies of rats, mice and dogs treated with PDE4D inhibitors that partially inhibit enzyme activity (Burgin et al. 2010).
5 Selective Inhibition of PDE4B
5.1 Specific Inhibition of PDE4B in Mice
Evidence supporting the notion that subtype-selective pharmacological inhibition of PDE4B might have pro-cognitive effects is provided by a study of mice with a missense mutation (Y358C) in the catalytic domain of PDE4B (McGirr et al. 2016). At the cAMP binding site, there is an interaction between the central phosphate group of cAMP and H406 in PDE4B1. Though the Y358 residue is located within the catalytic domain, it is neither at the site of cAMP binding nor rolipram binding (Richter et al. 2001). The Y358C mutation severely disrupts the docking position of cAMP, as the side chain of K282 bisects the binding site, resulting in partial (27%) inhibition of enzymatic activity. This magnitude of PDE4 inhibition is proportionate with the physiological 33% inhibition of cAMP hydrolysis when PDE4B1 is phosphorylated by ERK2 (Baillie et al. 2000). This contrasts with the ≥70% inhibition of PDE4D7 elicited by D159687, a PDE4D partial inhibitor that has shown pro-cognitive effects in mice and rats (Burgin et al. 2010). Partial inhibition of PDE4B is likely to maintain spatial and temporal aspects of cAMP signalling. Indeed, hippocampal slices from homozygous PDE4BY358C (Pde4b enu1H) mice have similar basal levels of cAMP to WT mice, but show a greater accumulation of cAMP when experimentally exposed to forskolin, either alone or in combination with rolipram (McGirr et al. 2016).
Young adult (12-week-old) PDE4BY358C mice show enhancements in spatial working memory in the Y-maze, in spatial memory acquisition, retention and reversal in the Morris water maze, and in object location recognition. The enhancement in object location recognition was greater when mice were tested in an aversive environment, suggesting that PDE4BY358C mice were less anxious. Indeed, PDE4BY358C mice exhibited lower anxiety and greater exploratory behaviour in the elevated plus maze, open field, light-dark box and hole-board tests. Additionally, PDE4BY358C mice show enhanced LTP, but unaltered basal synaptic transmission and LTD, in hippocampal slices, as well as increased pCREB levels and dendritic spine density in both the hippocampus and amygdala, and enhanced neurogenesis in the adult dentate gyrus (McGirr et al. 2016).
Notwithstanding the obvious differences between a genetic manipulation from conception versus a pharmacological manipulation in adulthood, these results suggest potential outcome measures to investigate in mice treated with compound A, A-33 or other PDE4B-selective inhibitors. It remains to be established whether the cognitive enhancement exhibited by 12-week-old PDE4BY358C mice persists into old age or when challenged with MK-801 (dizocilpine), an N-methyl-D-aspartate (NMDA) receptor antagonist that is used to model symptoms of schizophrenia (Zhang et al. 2000). It would also be interesting to challenge PDE4BY358C mice with a high-fat diet, and to determine whether the Y358C mutation has cognitive benefits in mouse models of AD or FXS.
PDE4B KO mice (see Sect. 5.3.1), by contrast, show enhanced basal synaptic transmission and LTD, but unaltered LTP, in hippocampal slices (Rutten et al. 2011), and unaltered spatial memory acquisition and retention (Rutten et al. 2011; Siuciak et al. 2008; Zhang et al. 2008), but impaired reversal learning (Rutten et al. 2011), in the Morris water maze. PDE4B KO mice also exhibit a moderately anxiogenic behavioural profile with decreased exploratory activity in the hole-board and light-dark box tests (Zhang et al. 2008), decreased locomotor activity in some open field tests (Rutten et al. 2011; Siuciak et al. 2008; Zhang et al. 2008), but unaltered performance in the elevated plus maze (Siuciak et al. 2008). Like PDE4BY358C mice, PDE4B KO mice have enhanced adult neurogenesis in the dentate gyrus (Zhang et al. 2008).
The neuronal signalling network is set up to maintain an optimal range for cAMP signalling in the brain, such that both the excessive activation and deactivation of the cAMP-PKA signalling pathway may induce the impairment of learning and memory (Sato et al. 2004). Thus, the divergent behavioural phenotypes of the PDE4BY358C and PDE4B KO mice may be due to the different magnitudes of PDE4B inhibition elicited by the KO mutation (full inhibition) versus the Y358C mutation (partial inhibition).
5.2 PDE4B-Selective Inhibitors
Clinical studies of rolipram were limited by side effects including nausea and emesis that are thought to arise from inhibition of the PDE4D subtype. Various strategies have been employed to develop compounds with a reduced side effect profile (Maurice et al. 2014). The development of PDE4D partial inhibitors has been an effective strategy to achieve efficacy while reducing the side effect liabilities in preclinical species (Burgin et al. 2010). A complementary approach to overcome the tolerability issues of pan-PDE4 inhibitors is the development of PDE4B-selective inhibitors that capture therapeutic effects while avoiding PDE4D that mediates emesis and nausea (Srivani et al. 2008). In recent years, a number of compounds that selectively inhibit PDE4B over PDE4D have been developed for their use as therapeutic agents, as reviewed extensively elsewhere (Azam and Tripuraneni 2014). However, to date, in vivo tests have been reported for only two PDE4B-selective inhibitors.
Compound A (2-[4-[[2-(3-fluoro-4-methoxy-phenyl)-7,8-dihydro-6H-thiopyrano[3,2-d]pyrimidin-4-yl] amino]phenyl]acetic acid) has an IC50 of 5.5 and 26 nM against human and mouse PDE4B2, respectively, and is selective for PDE4B2 over both human and mouse PDE4D2 (80- and 29-fold, respectively) (Suzuki et al. 2013). In mice, oral administration of compound A (3–100 mg/kg) had a dose-dependent anti-inflammatory effect by inhibiting the elevation of the plasma concentration of TNFα induced by injection of LPS. However, compound A was less effective against neutrophil accumulation in the lung induced by inhalation of LPS, showing a reduction only at the highest dose tested (300 mg/kg) (Suzuki et al. 2013).
Another compound, A-33 (2-(4-{[2-(5-chlorothiophen-2-yl)-5-ethyl-6-118 methylpyrimidin-4-yl]amino}phenyl)acetic acid), is reported to have IC50 values of 19 and 32 nM against human PDE4B1 and 27 nM against PDE4B3 measured in vitro, and to be 113-fold and 49-fold more selective towards PDE4B1 as compared to PDE4D3 and PDE4D7, respectively (Naganuma et al. 2009; Fox et al. 2014; Titus et al. 2016). A-33 binds to a C-terminal regulatory helix termed CR3 (Control Region 3) that is present in all PDE4B isoforms, thereby locking the enzyme in an inactive ‘closed’ conformation (Fox et al. 2014). In mice, oral administration of A-33 (14 mg/kg) reduced LPS-induced TNFα production (Naganuma et al. 2009), while a median effective dose (ED50) of 0.1 mg/kg had antidepressant effects in the forced swim test (Titus et al. 2016). The half-life of A33 in the mouse brain is 3.8–4.5 h (Titus et al. 2016). In ferrets, oral administration of A-33 (12.5 mg/kg) reduced LPS inhalation-induced neutrophil accumulation in the lungs by 44%, while a much higher dose of 100 mg/kg was tolerated without emesis (Naganuma et al. 2009). In a rat model of traumatic brain injury, A-33 was recently shown to have pro-cognitive effects (Titus et al. 2016; see Sect. 5.5.3).
An alternative approach to activating the cAMP-PKA signalling pathway in the mammalian brain may eventually be provided by photoactivated adenylyl cyclase (PAC), an emerging optogenetic tool that has been shown to increase intracellular cAMP levels and alter behaviour in Drosophila flies and Caenorhabditis elegans nematodes upon stimulation by blue light (Schröder-Lang et al. 2007; Weissenberger et al. 2011).
5.3 Traumatic Brain Injury
The majority of survivors of moderate and severe traumatic brain injury (TBI) have chronic neurobehavioural sequelae, including impairment in cognitive domains such as frontal executive functions, attention, short-term memory and learning, speed of information processing, and speech and language functions (Arciniegas et al. 2002). The high prevalence of cognitive impairment is due, in part, to the vulnerability of the hippocampus, which exhibits progressive bilateral atrophy even when not initially damaged (Bigler et al. 2002). There are currently no effective treatments to improve TBI-induced cognitive impairment (Wheaton et al. 2009), but potential therapeutic approaches have been evaluated in animal models of TBI.
In one of the oldest and most commonly used models, TBI is surgically induced in young adult male Sprague-Dawley rats by lateral fluid-percussion injury (Kabadi et al. 2010). In this rat lateral fluid-percussion model, hippocampal levels of cAMP show an acute decrease from 15 min to 4 h post-surgery (Atkins et al. 2007), associated with an elevated level of PDE4B2 (short isoform), but not PDE4B1/3/4 (long isoforms), between 1 and 24 h post-surgery (Wilson et al. 2016). Levels of TNFα, a pro-inflammatory cytokine mediated by PDE4B (Jin and Conti 2002), are enhanced maximally within 3–8 h post-surgery and return to non-injured levels by 24 h post-surgery (Titus et al. 2016). At 3 months post-surgery, the chronically injured hippocampus shows decreased levels of basal pCREB (Atkins et al. 2009), but PDE4B isoforms are not upregulated and TNFα is not detectable (Titus et al. 2016).
The potential utility of the PDE4B inhibitor A-33 (see Sect. 5.5.2) for cognitive manifestations of TBI has been evaluated in the rat lateral fluid-percussion model by Titus et al. (2016). Ex vivo assessment of synaptic plasticity in ipsilateral hippocampal slices from rats at 3 months post-surgery revealed a significant reduction in basal synaptic transmission and impaired expression of LTP. Bath application of A-33 (300 nM) in artificial CSF to hippocampal slices significantly reduced the deficits in basal synaptic transmission and rescued LTP expression (Titus et al. 2016).
To determine whether PDE4B inhibition by A-33 would restore cognitive function in vivo during the chronic TBI recovery period, rats received A-33 (0.3 mg/kg, i.p.) 30 min before cue and contextual fear conditioning, water maze training and a delayed match-to-place task in the water maze between 3–4 months post-surgery. Retention of learned behaviour or of learning was measured 1 day and 1 month after training. Treatment with A-33 significantly reversed the TBI-induced deficits in cue and contextual fear conditioning and water maze retention, but did not enhance the performance of non-injured rats (Titus et al. 2016).
When the rats were killed at 5 months post-surgery, after behavioural testing, the intermittent A-33 treatment (eight doses over 4 weeks) was found to have had no effect on the significant atrophy and microglia accumulation (quantified using Iba1 immunostaining) observed in the cortex and hippocampus (Titus et al. 2016). A-33 administered 3–4 months post-surgery thus appears to have pro-cognitive benefits in injured rats, without reversing the pathology caused by TBI. Similarly, rolipram (0.03 mg/kg, i.p.) was previously shown to rescue the cognitive deficits of TBI rats in the fear conditioning and water maze tests, but had no effect on hippocampal atrophy in TBI rats at 8 weeks post-surgery (Titus et al. 2013). As the administration of A-33 (0.3 mg/kg, i.p.) at 5 h post-surgery was shown to reduce TNFα levels at 6 h post-surgery (Titus et al. 2016), it would be interesting to test whether the administration of A-33 within 24 h post-surgery, when TNFα levels are higher, would reduce the TBI-related neural damage.
To evaluate the feasibility of A-33 for inhibiting PDE4B in the injured brain, brain concentrations of A-33 at the dose used for behaviour assessment (0.3 mg/kg) were measured. Rats received A-33 at 3 months post-surgery and were decapitated 30 min later for the collection of trunk blood and brain tissue. Quantification of A-33 levels in plasma and brain tissue (cortex and hippocampus combined) by mass spectrometry revealed that A-33 has low brain distribution, with a similar B/P (brain/plasma) ratio of 3.2% in TBI rats and 2.4% in non-injured controls (Titus et al. 2016). Despite its low brain penetration, brain levels of A-33 were four to fivefold higher than the IC50 against PDE4B measured in vitro (see Sect. 5.5.2), suggesting that A-33 can attain relevant concentrations in the brain against the PDE4B target. A caveat is that the rats were not perfused before decapitation, so the brain tissue analysed contained cerebral blood. The ability of systemically administered A-33 to cross the blood-brain barrier and reach its site of action thus requires further investigation.
A-33 treatment was found to rescue the decrease in pCREB levels in the hippocampus of TBI rats (Titus et al. 2016), suggesting that a signalling pathway in the brain known to be regulated by PDE4B is inhibited with A-33. This finding provides support for the notion that A-33 improved learning and memory in TBI rats by altering cAMP signalling rather than the pathology caused by TBI. However, because inhibition of PDE4B in the brain after A-33 treatment was not directly measured by Titus et al. (2016), it is currently unknown whether brain-specific inhibition of PDE4B was the underlying mechanism for the improvements in learning and memory.
6 Concluding Remarks
Affluent, sedentary societies are becoming increasingly elderly and overweight, thus presenting a major challenge to prolong health in an ageing population. Indeed, ‘Adding life to years as well as years to life’ has become something of a mantra in health policy circles (World Health Organization 2002). Inhibition of PDE4 in preclinical rodent models has shown promise as a therapeutic strategy for age-related neurodegenerative conditions and obesity-related metabolic diseases, as well as for FXS, the most common inherited cause of intellectual disability. But the clinical application of pan-PDE4 inhibitors has been hampered by adverse side effects that limit maximally tolerated doses. The findings of cognitive enhancement in PDE4BY358C mice (McGirr et al. 2016), pro-cognitive effects of a non-emetic PDE4B-selective inhibitor (A-33) in a rat model of TBI (Naganuma et al. 2009; Titus et al. 2016), and resistance to obesity in PDE4B KO mice (Zhang et al. 2009) highlight the PDE4B subtype as a promising target for the development of therapeutic interventions in both neurological and metabolic disorders.
Reference
Ahmed T, Frey JU. Phosphodiesterase 4B (PDE4B) and cAMP-level regulation within different tissue fractions of rat hippocampal slices during long-term potentiation in vitro. Brain Res. 2005;1041:212–22.
Alberti KG, Zimmet P, Shaw J, Epidemiology Task IDF. Force consensus group. The metabolic syndrome – a new worldwide definition. Lancet. 2005;366:1059–62.
Alzheimer A. Uber eine eigenartige Erkrankung der Hirnrinde. Allg Z Psychiat Psych-Gericht Med. 1907;64:146–8.
Arciniegas DB, Held K, Wagner P. Cognitive impairment following traumatic brain injury. Curr Treat Options Neurol. 2002;4:43–57.
Arif AA, Rohrer JE. Patterns of alcohol drinking and its association with obesity: data from the Third National Health and Nutrition Examination Survey, 1988-1994. BMC Public Health. 2005;5:126.
Asai M, Tsukamoto O, Minamino T, Asanuma H, Fujita M, et al. PKA rapidly enhances proteasome assembly and activity in in vivo canine hearts. J Mol Cell Cardiol. 2009;46:452–62.
Asanuma M, Ogawa N, Kondo Y, Hirata H, Mori A. Effects of repeated administration of rolipram, a cAMP-specific phosphodiesterase inhibitor, on acetylcholinergic indices in the aged rat brain. Arch Gerontol Geriatr. 1993;16:191–8.
Atkins CM, Oliva AA Jr, Alonso OF, Pearse DD, Bramlett HM, et al. Modulation of the cAMP signaling pathway after traumatic brain injury. Exp Neurol. 2007;208:145–58.
Atkins CM, Falo MC, Alonso OF, Bramlett HM, Dietrich WD. Deficits in ERK and CREB activation in the hippocampus after traumatic brain injury. Neurosci Lett. 2009;59:52–6.
Avila DV, Barker DF, Zhang J, McClain CJ, Barve S, et al. Dysregulation of hepatic cAMP levels via altered Pde4b expression plays a critical role in alcohol-induced steatosis. J Pathol. 2016;240:96–107.
Azam MA, Tripuraneni NS. Selective phosphodiesterase 4B inhibitors: a review. Sci Pharm. 2014;82:453–81.
Bach ME, Barad M, Son H, Zhuo M, Lu YF, et al. Age-related defects in spatial memory are correlated with defects in the late phase of hippocampal long-term potentiation in vitro and are attenuated by drugs that enhance the cAMP signaling pathway. Proc Natl Acad Sci U S A. 1999;96:5280–5.
Bagni C, Oostra BA. Fragile X syndrome: from protein function to therapy. Am J Med Genet A. 2013;161A:2809–21.
Baillie GS, MacKenzie SJ, McPhee I, Houslay MD. Sub-family selective actions in the ability of Erk2 MAP kinase to phosphorylate and regulate the activity of PDE4 cyclic AMP-specific phosphodiesterases. Br J Pharmacol. 2000;131:811–9.
Baker C. Obesity statistics. Briefing paper 3336. London: House of Commons Library; 2015.
Bales KR, Plath N, Svenstrup N, Menniti FS. Phosphodiesterase inhibition to target the synaptic dysfunction in Alzheimer’s disease. Top Med Chem. 2010;6:57–90.
Barad M, Bourtchouladze R, Winder DG, Golan H, Kandel E. Rolipram, a type IV-specific phosphodiesterase inhibitor, facilitates the establishment of long-lasting long-term potentiation and improves memory. Proc Natl Acad Sci U S A. 1998;95:15020–5.
Bejanin S, Habert E, Berrard S, Edwards JB, Loeffler JP, et al. Promoter elements of the rat choline acetyltransferase gene allowing nerve growth factor inducibility in transfected primary cultured cells. J Neurochem. 1992;58:1580–3.
Berry-Kravis E, Ciurlionis R. Overexpression of fragile X gene (FMR-1) transcripts increases cAMP production in neural cells. J Neurosci Res. 1998;51:41–8.
Berry-Kravis E, Huttenlocher PR. Cyclic AMP metabolism in fragile X syndrome. Ann Neurol. 1992;31:22–6.
Berry-Kravis E, Sklena P. Demonstration of abnormal cyclic AMP production in platelets from patients with fragile X syndrome. Am J Med Genet. 1993;45:81–7.
Berry-Kravis E, Hicar M, Ciurlionis R. Reduced cyclic AMP production in fragile X syndrome: cytogenetic and molecular correlations. Pediatr Res. 1995;38:638–43.
Bigler ED, Anderson CV, Blatter DD. Temporal lobe morphology in normal aging and traumatic brain injury. AJNR Am J Neuroradiol. 2002;23:255–66.
Blazer DG, Yaffe K, Karlawish J. Cognitive aging: a report from the Institute of Medicine. JAMA. 2015;313:2121–2.
Blednov YA, Benavidez JM, Black M, Harris RA. Inhibition of phosphodiesterase 4 reduces ethanol intake and preference in C57BL/6J mice. Front Neurosci. 2014;8:129.
Bunnage ME, Gilbert AM, Jones LH, Hett EC. Know your target, know your molecule. Nat Chem Biol. 2015;11:368–72.
Burgin AB, Magnusson OT, Singh J, Witte P, Staker BL, et al. Design of phosphodiesterase 4D (PDE4D) allosteric modulators for enhancing cognition with improved safety. Nat Biotechnol. 2010;28:63–70.
Cai D. Neuroinflammation and neurodegeneration in overnutrition-induced diseases. Trends Endocrinol Metab. 2013;24:40–7.
Cheng YF, Wang C, Lin HB, Li YF, Huang Y, et al. Inhibition of phosphodiesterase-4 reverses memory deficits produced by Aβ25-35 or Aβ1-40 peptide in rats. Psychopharmacology (Berl). 2010;212:181–91.
Cherbuin N, Sargent-Cox K, Fraser M, Sachdev P, Anstey KJ. Being overweight is associated with hippocampal atrophy: the PATH Through Life Study. Int J Obes (Lond). 2015;39:1509–14.
Cherry JA, Davis RL. Cyclic AMP phosphodiesterases are localized in regions of the mouse brain associated with reinforcement, movement, and affect. J Comp Neurol. 1999;407:287–301.
Cheung YF, Kan Z, Garrett-Engele P, Gall I, Murdoch H, et al. PDE4B5, a novel, super-short, brain-specific cAMP phosphodiesterase-4 variant whose isoform-specifying N-terminal region is identical to that of cAMP phosphodiesterase-4D6 (PDE4D6). J Pharmacol Exp Ther. 2007;322:600–9.
Choi CH, Schoenfeld BP, Weisz ED, Bell AJ, Chambers DB, et al. PDE-4 inhibition rescues aberrant synaptic plasticity in Drosophila and mouse models of fragile X syndrome. J Neurosci. 2015;35:396–408.
Collins S, Martin TL, Surwit RS, Robidoux J. Genetic vulnerability to diet-induced obesity in the C57BL/6J mouse: physiological and molecular characteristics. Physiol Behav. 2004;81:243–8.
Comery TA, Martone RL, Aschmies S, Atchison KP, Diamantidis G, et al. Acute gamma-secretase inhibition improves contextual fear conditioning in the Tg2576 mouse model of Alzheimer's disease. J Neurosci. 2005;25:8898–902.
Cullen WK, Suh YH, Anwyl R, Rowan MJ. Block of LTP in rat hippocampus in vivo by beta-amyloid precursor protein fragments. Neuroreport. 1997;8:3213–7.
Darnell JC, Van Driesche SJ, Zhang C, Hung KY, Mele A, et al. FMRP stalls ribosomal translocation on mRNAs linked to synaptic function and autism. Cell. 2011;146:247–61.
Darnell JC, Klann E. The translation of translational control by FMRP: therapeutic targets for FXS. Nat Neurosci. 2013;16:1530–6.
Deary IJ, Whalley LJ, Lemmon H, Crawford JR, Starr JM. The stability of individual differences in mental ability from childhood to old age: follow-up of the 1932 Scottish Mental Survey. Intelligence. 2000;28:49–55.
DeFronzo RA, Simonson D, Ferrannini E. Hepatic and peripheral insulin resistance: a common feature of type 2 (non-insulin-dependent) and type 1 (insulin-dependent) diabetes mellitus. Diabetologia. 1982;23:313–9.
Fatemi SH, King DP, Reutiman TJ, Folsom TD, Laurence JA, et al. PDE4B polymorphisms and decreased PDE4B expression are associated with schizophrenia. Schizophr Res. 2008;101:36–49.
Fillit H, Ding WH, Buee L, Kalman J, Altstiel L, et al. Elevated circulating tumor necrosis factor levels in Alzheimer's disease. Neurosci Lett. 1991;129:318–20.
Flint A, Raben A, Astrup A, Holst JJ. Glucagon-like peptide 1 promotes satiety and suppresses energy intake in humans. J Clin Invest. 1998;101:515–20.
Fox D, Burgin AB, Gurney ME. Structural basis for the design of selective phosphodiesterase 4B inhibitors. Cell Signal. 2014;26:657–63.
Gevrey JC, Cordier-Bussat M, Némoz-Gaillard E, Chayvialle JA, Abello J. Co-requirement of cyclic AMP- and calcium-dependent protein kinases for transcriptional activation of cholecystokinin gene by protein hydrolysates. J Biol Chem. 2002;277:22407–13.
Ghavami A, Hirst WD, Novak TJ. Selective phosphodiesterase (PDE)-4 inhibitors: a novel approach to treating memory deficit? Drugs R D. 2006;7:63–71.
Giembycz MA. Life after PDE4: overcoming adverse events with dual-specificity phosphodiesterase inhibitors. Curr Opin Pharmacol. 2005;5:238–44.
Gong B, Vitolo OV, Trinchese F, Liu S, Shelanski M, et al. Persistent improvement in synaptic and cognitive functions in an Alzheimer mouse model after rolipram treatment. J Clin Invest. 2004;114:1624–34.
Griebel G, Misslin R, Vogel E, Bourguignon JJ. Behavioral effects of rolipram and structurally related compounds in mice: behavioral sedation of cAMP phosphodiesterase inhibitors. Pharmacol Biochem Behav. 1991;39:321–3.
Grudzien A, Shaw P, Weintraub S, Bigio E, Mash DC, et al. Locus coeruleus neurofibrillary degeneration in aging, mild cognitive impairment and early Alzheimer’s disease. Neurobiol Aging. 2007;28:327–35.
Gupta S. Side-effects of roflumilast. Lancet. 2012;379:710–1.
Guzowski JF, McGaugh JL. Antisense oligodeoxynucleotide-mediated disruption of hippocampal cAMP response element binding protein levels impairs consolidation of memory for water maze training. Proc Natl Acad Sci U S A. 1997;94:2693–8.
Habener JF. Cyclic AMP response element binding proteins: a cornucopia of transcription factors. Mol Endocrinol. 1990;4:1087–94.
Hardy J. The amyloid hypothesis for Alzheimer’s disease: a critical reappraisal. J Neurochem. 2009;110:1129–34.
Heaslip RJ, Evans DY. Emetic, central nervous system, and pulmonary activities of rolipram in the dog. Eur J Pharmacol. 1995;286:281–90.
Heppner FL, Ransohoff RM, Becher B. Immune attack: the role of inflammation in Alzheimer disease. Nat Rev Neurosci. 2015;16:358–72.
Hicks J, Allen G. A century of change: trends in UK statistics since 1900. Research paper 99/111. London: House of Commons Library; 1999. p. 8.
Holz GG, Habener JF. Signal transduction crosstalk in the endocrine system: pancreatic beta-cells and the glucose competence concept. Trends Biochem Sci. 1992;17:388–93.
Hosseini-Sharifabad A, Ghahremani MH, Sabzevari O, Naghdi N, Abdollahi M, et al. Effects of protein kinase A and G inhibitors on hippocampal cholinergic markers expressions in rolipram- and sildenafil-induced spatial memory improvement. Pharmacol Biochem Behav. 2012;101:311–9.
Houslay MD, Baillie GS, Maurice DH. cAMP-specific phosphodiesterase-4 enzymes in the cardiovascular system: a molecular toolbox for generating compartmentalized cAMP signaling. Circ Res. 2007;100:950–66.
Hu W, Lu T, Chen A, Huang Y, Hansen R, et al. Inhibition of phosphodiesterase-4 decreases ethanol intake in mice. Psychopharmacology (Berl). 2011;218:331–9.
Huber KM, Gallagher SM, Warren ST, Bear MF. Altered synaptic plasticity in a mouse model of fragile X mental retardation. Proc Natl Acad Sci U S A. 2002;99:7746–50.
Hummel KP, Dickie MM, Coleman DL. Diabetes, a new mutation in the mouse. Science. 1966;153:1127–8.
Hutton M, Lendon CL, Rizzu P, Baker M, Froelich S, et al. Association of missense and 5’-splice-site mutations in tau with the inherited dementia FTDP-17. Nature. 1998;393:702–5.
Hwu WL, Wang TR, Lee YM. FMR1 enhancer is regulated by cAMP through a cAMP-responsive element. DNA Cell Biol. 1997;16:449–53.
Itoh A, Akaike T, Sokabe M, Nitta A, Iida R, et al. Impairments of long-term potentiation in hippocampal slices of beta-amyloid-infused rats. Eur J Pharmacol. 1999;382:167–75.
Jensterle M, Kocjan T, Janez A. Phosphodiesterase 4 inhibition as a potential new therapeutic target in obese women with polycystic ovary syndrome. J Clin Endocrinol Metab. 2014;99:E1476–81.
Jeon BT, Jeong EA, Shin HJ, Lee Y, Lee DH, et al. Resveratrol attenuates obesity-associated peripheral and central inflammation and improves memory deficit in mice fed a high-fat diet. Diabetes. 2012;61:1444–54.
Jin SL, Conti M. Induction of the cyclic nucleotide phosphodiesterase PDE4B is essential for LPS-activated TNF-alpha responses. Proc Natl Acad Sci U S A. 2002;99:7628–33.
Jin SL, Lan L, Zoudilova M, Conti M. Specific role of phosphodiesterase 4B in lipopolysaccharide-induced signaling in mouse macrophages. J Immunol. 2005;175:1523–31.
Johanns M, Lai YC, Hsu MF, Jacobs R, Vertommen D, et al. AMPK antagonizes hepatic glucagon-stimulated cyclic AMP signalling via phosphorylation-induced activation of cyclic nucleotide phosphodiesterase 4B. Nat Commun. 2016;7:10856.
Kabadi SV, Hilton GD, Stoica BA, Zapple DN, Faden AI. Fluid-percussion-induced traumatic brain injury model in rats. Nat Protoc. 2010;5:1552–63.
Kandel ER. The molecular biology of memory storage: a dialogue between genes and synapses. Science. 2001;294:1030–8.
Kanellopoulos AK, Semelidou O, Kotini AG, Anezaki M, Skoulakis EM. Learning and memory deficits consequent to reduction of the fragile X mental retardation protein result from metabotropic glutamate receptor-mediated inhibition of cAMP signaling in Drosophila. J Neurosci. 2012;32:13111–24.
Kelley DJ, Davidson RJ, Elliott JL, Lahvis GP, Yin JC, et al. The cyclic AMP cascade is altered in the fragile X nervous system. PLoS One. 2007;2:e931.
Kelley DJ, Bhattacharyya A, Lahvis GP, Yin JC, Malter J, et al. The cyclic AMP phenotype of fragile X and autism. Neurosci Biobehav Rev. 2008;32:1533–43.
Kelly MP, Adamowicz W, Bove S, Hartman AJ, Mariga A, et al. Select 3’,5’-cyclic nucleotide phosphodiesterases exhibit altered expression in the aged rodent brain. Cell Signal. 2014;26:383–97.
Kopelman P. Health risks associated with overweight and obesity. Obes Rev. 2007;8(Suppl 1):13–7.
Krause W, Kuhne G. Pharmacokinetics of rolipram in the rhesus and cynomolgus monkeys, the rat and the rabbit. Studies on species differences. Xenobiotica. 1988;18:561–71.
Kreisl WC, Lyoo CH, McGwier M, Snow J, Jenko KJ, et al. In vivo radioligand binding to translocator protein correlates with severity of Alzheimer’s disease. Brain. 2013;136:2228–38.
Larson JL, Pino MV, Geiger LE, Simeone CR. The toxicity of repeated exposures to rolipram, a type IV phosphodiesterase inhibitor, in rats. Pharmacol Toxicol. 1996;78:44–9.
Lee KT, Byun MJ, Kang KS, Park EW, Lee SH, et al. Neuronal genes for subcutaneous fat thickness in human and pig are identified by local genomic sequencing and combined SNP association study. PLoS One. 2011;6:e16356.
Lee MJ, Lee JH, Rubinsztein DC. Tau degradation: the ubiquitin-proteasome system versus the autophagy-lysosome system. Prog Neurobiol. 2013;105:49–59.
Liang L, Beshay E, Prud’homme GJ. The phosphodiesterase inhibitors pentoxifylline and rolipram prevent diabetes in NOD mice. Diabetes. 1998;47:570–5.
Lieber CS, DeCarli LM, Sorrell MF. Experimental methods of ethanol administration. Hepatology. 1989;10:501–10.
de Lima MN, Presti-Torres J, Garcia VA, Guimarães MR, Scalco FS, et al. Amelioration of recognition memory impairment associated with iron loading or aging by the type 4-specific phosphodiesterase inhibitor rolipram in rats. Neuropharmacology. 2008;55:788–92.
MacKenzie SJ, Baillie GS, McPhee I, MacKenzie C, Seamons R, et al. Long PDE4 cAMP specific phosphodiesterases are activated by protein kinase A-mediated phosphorylation of a single serine residue in Upstream Conserved Region 1 (UCR1). Br J Pharmacol. 2002;136:421–33.
Makino S, Kunimoto K, Muraoka Y, Mizushima Y, Katagiri K, et al. Breeding of a non-obese, diabetic strain of mice. Jikken Dobutsu. 1980;29:1–13.
Malenka RC, Bear MF. LTP and LTD: an embarrassment of riches. Neuron. 2004;44:5–21.
Maurice DH, Ke H, Ahmad F, Wang Y, Chung J, et al. Advances in targeting cyclic nucleotide phosphodiesterases. Nat Rev Drug Discov. 2014;13:290–314.
McCary LM, Roberts JE. Early identification of autism in fragile X syndrome: a review. J Intellect Disabil Res. 2013;57:803–14.
McGirr A, Lipina TV, Mun HS, Georgiou J, Al-Amri AH, et al. Specific inhibition of phosphodiesterase-4B results in anxiolysis and facilitates memory acquisition. Neuropsychopharmacology. 2016;41:1080–92.
Miller RA, Chu Q, Xie J, Foretz M, Viollet B, et al. Biguanides suppress hepatic glucagon signalling by decreasing production of cyclic AMP. Nature. 2013;494:256–60.
Mori F, Pérez-Torres S, De Caro R, Porzionato A, Macchi V, et al. The human area postrema and other nuclei related to the emetic reflex express cAMP phosphodiesterases 4B and 4D. J Chem Neuroanat. 2010;40:36–42.
Myeku N, Clelland CL, Emrani S, Kukushkin NV, WH Y, et al. Tau-driven 26S proteasome impairment and cognitive dysfunction can be prevented early in disease by activating cAMP-PKA signaling. Nat Med. 2016;22:46–53.
Naganuma K, Omura A, Maekawara N, Saitoh M, Ohkawa N, et al. Discovery of selective PDE4B inhibitors. Bioorg Med Chem Lett. 2009;19:3174–6.
Nicholson A, Reifsnyder PC, Malcolm RD, Lucas CA, MacGregor GR, et al. Diet-induced obesity in two C57BL/6 substrains with intact or mutant nicotinamide nucleotide transhydrogenase (Nnt) gene. Obesity (Silver Spring). 2010;18:1902–5.
O’Shea RS, Dasarathy S, McCullough AJ. Alcoholic liver disease. Hepatology. 2010;51:307–28.
Ong WK, Gribble FM, Reimann F, Lynch MJ, Houslay MD, et al. The role of the PDE4D cAMP phosphodiesterase in the regulation of glucagon-like peptide-1 release. Br J Pharmacol. 2009;157:633–44.
Pacey LK, Doss L, Cifelli C, van der Kooy D, Heximer SP, et al. Genetic deletion of regulator of G-protein signaling 4 (RGS4) rescues a subset of fragile X related phenotypes in the FMR1 knockout mouse. Mol Cell Neurosci. 2011;46:563–72.
Park SJ, Ahmad F, Philp A, Baar K, Williams T, et al. Resveratrol ameliorates aging-related metabolic phenotypes by inhibiting cAMP phosphodiesterases. Cell. 2012;148:421–33.
Peters M, Bletsch M, Stanley J, Wheeler D, Scott R, Tully T. The PDE4 inhibitor HT-0712 improves hippocampus-dependent memory in aged mice. Neuropsychopharmacology. 2014;39:2938–48.
Peth A, Kukushkin N, Bossé M, Goldberg AL. Ubiquitinated proteins activate the proteasomal ATPases by binding to Usp14 or Uch37 homologs. J Biol Chem. 2013;288:7781–90.
Piguet O, Halliday GM, Reid WG, Casey B, Carman R, et al. Clinical phenotypes in autopsy-confirmed Pick disease. Neurology. 2011;76:253–9.
Pistell PJ, Morrison CD, Gupta S, Knight AG, Keller JN, et al. Cognitive impairment following high fat diet consumption is associated with brain inflammation. J Neuroimmunol. 2010;219:25–32.
Podrini C, Cambridge EL, Lelliott CJ, Carragher DM, Estabel J, et al. High-fat feeding rapidly induces obesity and lipid derangements in C57BL/6N mice. Mamm Genome. 2013;24:240–51.
Polsky S, Ellis SL. Obesity, insulin resistance, and type 1 diabetes mellitus. Curr Opin Endocrinol Diabetes Obes. 2015;22:277–82.
Querfurth HW, LaFerla FM. Alzheimer’s disease. N Engl J Med. 2010;362:329–44.
Raji CA, Ho AJ, Parikshak NN, Becker JT, Lopez OL, et al. Brain structure and obesity. Hum Brain Mapp. 2010;31:353–64.
Ramos BP, Birnbaum SG, Lindenmayer I, Newton SS, Duman RS, et al. Dysregulation of protein kinase A signaling in the aged prefrontal cortex: new strategy for treating age-related cognitive decline. Neuron. 2003;40:835–45.
Randt CT, Judge ME, Bonnet KA, Quartermain D. Brain cyclic AMP and memory in mice. Pharmacol Biochem Behav. 1982;17:677–80.
Restivo L, Ferrari F, Passino E, Sgobio C, Bock J, et al. Enriched environment promotes behavioral and morphological recovery in a mouse model for the fragile X syndrome. Proc Natl Acad Sci U S A. 2005;102:11557–62.
Richter W, Unciuleac L, Hermsdorf T, Kronbach T, Dettmer D. Identification of inhibitor binding sites of the cAMP-specific phosphodiesterase 4. Cell Signal. 2001;13:287–97.
Richter W, Menniti FS, Zhang HT, Conti M. PDE4 as a target for cognition enhancement. Expert Opin Ther Targets. 2013;17:1011–27.
Robichaud A, Savoie C, Stamatiou PB, Tattersall FD, Chan CC. PDE4 inhibitors induce emesis in ferrets via a noradrenergic pathway. Neuropharmacology. 2001;40:262–9.
Robichaud A, Stamatiou PB, Jin SL, Lachance N, MacDonald D, et al. Deletion of phosphodiesterase 4D in mice shortens alpha(2)-adrenoceptor-mediated anesthesia, a behavioral correlate of emesis. J Clin Invest. 2002;110:1045–52.
Rutten K, Basile JL, Prickaerts J, Blokland A, Vivian JA. Selective PDE inhibitors rolipram and sildenafil improve object retrieval performance in adult cynomolgus macaques. Psychopharmacology (Berl). 2008;196:643–8.
Rutten K, Wallace TL, Works M, Prickaerts J, Blokland A, et al. Enhanced long-term depression and impaired reversal learning in phosphodiesterase 4B-knockout (PDE4B-/-) mice. Neuropharmacology. 2011;61:138–47.
Santacruz K, Lewis J, Spires T, Paulson J, Kotilinek L, et al. Tau suppression in a neurodegenerative mouse model improves memory function. Science. 2005;309:476–81.
Sato T, Tanaka K, Ohnishi Y, Teramoto T, Irifune M, et al. Inhibitory effects of group II mGluR-related drugs on memory performance in mice. Physiol Behav. 2004;80:747–58.
Schneider HH. Brain cAMP response to phosphodiesterase inhibitors in rats killed by microwave irradiation or decapitation. Biochem Pharmacol. 1984;33:1690–3.
Schröder-Lang S, Schwärzel M, Seifert R, Strünker T, Kateriya S, et al. Fast manipulation of cellular cAMP level by light in vivo. Nat Methods. 2007;4:39–42.
Sebastiani G, Morissette C, Lagacé C, Boulé M, Ouellette MJ, et al. The cAMP-specific phosphodiesterase 4B mediates Abeta-induced microglial activation. Neurobiol Aging. 2006;27:691–701.
Shepherd M, McSorley T, Olsen AE, Johnston LA, Thomson NC, et al. Molecular cloning and subcellular distribution of the novel PDE4B4 cAMP-specific phosphodiesterase isoform. Biochem J. 2003;370:429–38.
Siuciak JA, Chapin DS, McCarthy SA, Martin AN. Antipsychotic profile of rolipram: efficacy in rats and reduced sensitivity in mice deficient in the phosphodiesterase-4B (PDE4B) enzyme. Psychopharmacology (Berl). 2007;192:415–24.
Siuciak JA, McCarthy SA, Chapin DS, Martin AN. Behavioral and neurochemical characterization of mice deficient in the phosphodiesterase-4B (PDE4B) enzyme. Psychopharmacology (Berl). 2008;197:115–26.
Smith KT, Nicholls RD, Reines D. The gene encoding the fragile X RNA-binding protein is controlled by nuclear respiratory factor 2 and the CREB family of transcription factors. Nucleic Acids Res. 2006;34:1205–15.
Smith DL, Pozueta J, Gong B, Arancio O, Shelanski M. Reversal of long-term dendritic spine alterations in Alzheimer disease models. Proc Natl Acad Sci U S A. 2009;106:16877–82.
Srivani P, Usharani D, Jemmis ED, Sastry GN. Subtype selectivity in phosphodiesterase 4 (PDE4): a bottleneck in rational drug design. Curr Pharm Des. 2008;14:3854–72.
Stern MP, Williams K, González-Villalpando C, Hunt KJ, Haffner SM. Does the metabolic syndrome improve identification of individuals at risk of type 2 diabetes and/or cardiovascular disease? Diabetes Care. 2004;27:2676–81.
Suzuki O, Mizukami K, Etori M, Sogawa Y, Takagi N, et al. Evaluation of the therapeutic index of a novel phosphodiesterase 4B-selective inhibitor over phosphodiesterase 4D in mice. J Pharmacol Sci. 2013;123:219–26.
Tarkowski E, Blennow K, Wallin A, Tarkowski A. Intracerebral production of tumor necrosis factor-alpha, a local neuroprotective agent, in Alzheimer disease and vascular dementia. J Clin Immunol. 1999;19:223–30.
Titus DJ, Sakurai A, Kang Y, Furones C, Jergova S, et al. Phosphodiesterase inhibition rescues chronic cognitive deficits induced by traumatic brain injury. J Neurosci. 2013;33:5216–26.
Titus DJ, Wilson NM, Freund JE, Carballosa MM, Sikah KE, et al. Chronic cognitive dysfunction after traumatic brain injury is improved with a phosphodiesterase 4B inhibitor. J Neurosci. 2016;36:7095–108.
Ventura R, Pascucci T, Catania MV, Musumeci SA, Puglisi-Allegra S. Object recognition impairment in Fmr1 knockout mice is reversed by amphetamine: involvement of dopamine in the medial prefrontal cortex. Behav Pharmacol. 2004;15:433–42.
Verkerk AJ, Pieretti M, Sutcliffe JS, YH F, Kuhl DP, et al. Identification of a gene (FMR-1) containing a CGG repeat coincident with a breakpoint cluster region exhibiting length variation in fragile X syndrome. Cell. 1991;65:905–14.
Vitolo OV, Sant’Angelo A, Costanzo V, Battaglia F, Arancio O, et al. Amyloid beta-peptide inhibition of the PKA/CREB pathway and long-term potentiation: reversibility by drugs that enhance cAMP signaling. Proc Natl Acad Sci U S A. 2002;99:13217–21.
Vollert S, Kaessner N, Heuser A, Hanauer G, Dieckmann A, et al. The glucose-lowering effects of the PDE4 inhibitors roflumilast and roflumilast-N-oxide in db/db mice. Diabetologia. 2012;55:2779–88.
Vorhees CV, Williams MT. Morris water maze: procedures for assessing spatial and related forms of learning and memory. Nat Protoc. 2006;1:848–58.
Wang M, Gamo NJ, Yang Y, Jin LE, Wang XJ, et al. Neuronal basis of age-related working memory decline. Nature. 2011a;476:210–3.
Wang YC, McPherson K, Marsh T, Gortmaker SL, Brown M. Health and economic burden of the projected obesity trends in the USA and the UK. Lancet. 2011b;378:815–25.
Wang C, Yang XM, Zhuo YY, Zhou H, Lin HB, et al. The phosphodiesterase-4 inhibitor rolipram reverses Aβ-induced cognitive impairment and neuroinflammatory and apoptotic responses in rats. Int J Neuropsychopharmacol. 2012;15:749–66.
Weissenberger S, Schultheis C, Liewald JF, Erbguth K, Nagel G, et al. PACα – an optogenetic tool for in vivo manipulation of cellular cAMP levels, neurotransmitter release, and behavior in Caenorhabditis elegans. J Neurochem. 2011;116:616–25.
Wheaton P, Mathias JL, Vink R. Impact of early pharmacological treatment on cognitive and behavioral outcome after traumatic brain injury in adults: a meta-analysis. J Clin Psychopharmacol. 2009;29:468–77.
Wilson NM, Titus DJ, Oliva AA Jr, Furones C, Atkins CM. Traumatic brain injury upregulates phosphodiesterase expression in the hippocampus. Front Syst Neurosci. 2016;10:5.
Wolf PA, Beiser A, Elias MF, Au R, Vasan RS, et al. Relation of obesity to cognitive function: importance of central obesity and synergistic influence of concomitant hypertension. The Framingham Heart Study. Curr Alzheimer Res. 2007;4:111–6.
World Health Organization. Active ageing: a policy framework. Aging Male. 2002;5:1–37.
Wouters EF, Bredenbröker D, Teichmann P, Brose M, Rabe KF, et al. Effect of the phosphodiesterase 4 inhibitor roflumilast on glucose metabolism in patients with treatment-naive, newly diagnosed type 2 diabetes mellitus. J Clin Endocrinol Metab. 2012;97:E1720–5.
Yoneyama N, Crabbe JC, Ford MM, Murillo A, Finn DA. Voluntary ethanol consumption in 22 inbred mouse strains. Alcohol. 2008;42:149–60.
Zhang HT. Cyclic AMP-specific phosphodiesterase-4 as a target for the development of antidepressant drugs. Curr Pharm Des. 2009;15:1688–98.
Zhang HT, Crissman AM, Dorairaj NR, Chandler LJ, O’Donnell JM. Inhibition of cyclic AMP phosphodiesterase (PDE4) reverses memory deficits associated with NMDA receptor antagonism. Neuropsychopharmacology. 2000;23:198–204.
Zhang X, Dong F, Ren J, Driscoll MJ, Culver B. High dietary fat induces NADPH oxidase-associated oxidative stress and inflammation in rat cerebral cortex. Exp Neurol. 2005;191:318–25.
Zhang HT, Huang Y, Masood A, Stolinski LR, Li Y, et al. Anxiogenic-like behavioral phenotype of mice deficient in phosphodiesterase 4B (PDE4B). Neuropsychopharmacology. 2008;33:1611–23.
Zhang R, Maratos-Flier E, Flier JS. Reduced adiposity and high-fat diet-induced adipose inflammation in mice deficient for phosphodiesterase 4B. Endocrinology. 2009;150:3076–82.
Zhu B, Zhang L, Creighton J, Alexeyev M, Strada SJ, et al. Protein kinase A phosphorylation of tau-serine 214 reorganizes microtubules and disrupts the endothelial cell barrier. Am J Physiol Lung Cell Mol Physiol. 2010;299:L493–501.
Conflict of Interest
The authors declare that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing AG
About this chapter
Cite this chapter
Clapcote, S.J. (2017). Phosphodiesterase-4B as a Therapeutic Target for Cognitive Impairment and Obesity-Related Metabolic Diseases. In: Zhang, HT., Xu, Y., O'Donnell, J. (eds) Phosphodiesterases: CNS Functions and Diseases. Advances in Neurobiology, vol 17. Springer, Cham. https://doi.org/10.1007/978-3-319-58811-7_5
Download citation
DOI: https://doi.org/10.1007/978-3-319-58811-7_5
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-58809-4
Online ISBN: 978-3-319-58811-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)