Abstract
The presence of functional TR, either isolated or in combination with left heart disease is associated with unfavorable natural history. In addition, mortality for isolated tricuspid valve interventions remain higher than for any other single valve surgery. Finally, as more left sided valve disease is treated with transcatheter therapies the need for transcatheter solutions to functional tricuspid regurgitation. Transesophageal echocardiographic (TOE) imaging of the tricuspid valve has become an important intra-procedural tool for assessing the morphology of the valve apparatus and severity of disease, guiding transcatheter solutions and assessing the results of interventions. The following chapter reviews the tricuspid valve anatomy and essential TOE views to define this anatomy and valvular function.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
The most recent American Society of Echocardiography (ASE) guideline for performing a comprehensive TOE examination [1] includes addition imaging, many of which were intended to improve imaging of the tricuspid valve. A comprehensive TOE examination of the tricuspid valve should include imaging from several depths and multiplane angles. Additional views specific to imaging the tricuspid valve which are not included in the guidelines will be discussed below [2]. Multiple guidelines [1, 3] also describe the technique of 3D imaging of the tricuspid valve. Standardization of the image acquisition and display is particularly important when imaging is performed for transcatheter devices, improving communication with the interventionalist. This technology has significantly improved the accuracy of imaging and identification of the tricuspid leaflets and associated anatomic components of the tricuspid valve complex and has already been shown to be integral to tricuspid valve interventions [4].
TOE Imaging for Tricuspid Valve Imaging
Given the position of the heart in relation to the esophagus and stomach, there are four levels which may bring the probe closest to the tricuspid valve for both 2D and 3D imaging: mid-esophageal (ME), low esophageal or gastro-esophageal (LE) junction, shallow transgastric (TG) as well as deep transgastric (DTG) views (Fig. 6.1). A few important anatomic clues to the location of the leaflets can be used. First, the septal leaflet is the shortest in the radial direction and the least mobile due to direct chordal attachment to the septum. The tricuspid leaflet associated with the interventricular or interatrial septae is the septal leaflet. The commissure between the septal and anterior leaflet is typically adjacent to the most posterior border of the aortic noncoronary sinus of Valsalva. A portion of the anterior leaflet is also adjacent to the aorta (noncoronary and right coronary sinuses of Valsalva) but extends further anterior and lateral over the associated walls of the right ventricle, and under the right atrial appendage. The posterior leaflet which is often scalloped and may not be clearly distinguishable from the anterior leaflet, extends from the lateral to posterior walls of the right ventricle. The commissure between the septal and posterior leaflet is typically near the inflow of the coronary sinus. It is important at each level to rotate through multiple planes to comprehensively evaluate the tricuspid valve and to use the simultaneous multiplane modality to help with identifying leaflets and appreciating adjacent anatomy.
Esophageal views of the tricuspid valve. Panel (a) is a simultaneous multiplane image from the mid-esophageal (ME) level. The 4-chamber view is seen in the 0 degree plane with septal (yellow line) and typically the anterior leaflet (blue line) is seen. The orthogonal plane with the aortic valve (AV) in view, the anterior leaflet and posterior leaflet (green line) are seen. Panel (b) is a simultaneous multiplane image from the lowesophageal level with no left atrium (LA) but the coronary sinus (CS) in view
Mid-esophageal level : From the ME four-chamber view, rotating the probe clockwise to center the tricuspid valve in the imaging plane, permits visualization of the septal leaflet (arising from the septum) and typically the anterior leaflet (adjacent to the right atrial appendage); simultaneous biplane imaging may help clarify which leaflet is imaged since the anterior leaflet is typically seen adjacent to the aorta (Fig. 6.1a). Because the lower right heart border is close to the diaphragm, slow insertion brings the TOE probe to the distal esophagus, brings the probe closer to the tricuspid annulus; frequently there is no left atrium seen, and only the right atrium and coronary sinus with the orthogonal view imaging the right ventricular outflow tract (Fig. 6.1b). This view may also align the Doppler beam with the regurgitant jet and allow a comprehensive evaluation of tricuspid valve function, including an assessment of tricuspid regurgitation severity. Acquiring 3D volumes of the tricuspid valve from this view, may allow live-3D imaging of the surgical view (Fig. 6.2).
Three-dimensional imaging of the tricuspid valve. From the distal esophageal views, a user-defined volume is obtained, and rotated to image the valve from the atrial side (panel a). The surgical view is then obtain by rotating this view with the interatrial septum (IAS) in the far field (panel b), which places the anterior leaflet (A) in the near field and to the left, with the posterior leaflet (P) in the near field and to the right. The coronary sinus (CS) is then at the 7-o’clock position
Transgastric level : Advancing the TEE probe into the stomach results in the transgastric views. Using a right flexion and rotating the probe to center the tricuspid valve in the imaging plane, results in the inflow-outflow view of the right heart (Fig. 6.3a). The orthogonal view shows all three tricuspid valve leaflets which can also be imaged using a single plane view between 60 and 90 degrees (Fig. 6.3b). This view may be particularly useful intra-procedurally to identify the leaflets and commissures. And using this view as the primary image, a sweep of the entire tricuspid valve orifice could be imaged using simultaneous multiplane imaging. Advancing the TOE probe further into the stomach along with rightward anterior flexion produces a deep transgastric view of the tricuspid valve (Fig. 6.3c), which may also align the insonation beam with the flow across the tricuspid valve, and should be used to assess tricuspid valve function.
Simultaneous biplane imaging from transgastric views with right and anteflexion, the inflow-outflow view of the right heart (panel a) images the anterior (blue line) and posterior leaflet (green line) at 0 degrees. The orthogonal view shows all three tricuspid valve leaflets. This short-axis view of the tricuspid valve can also be imaged between 60 and 90 degrees (panel b) with the simultaneous multiplane imaging plane used to image all three leaflet tips. Deep transgastric views of the tricuspid valve (panel c), may align the insonation beam with the flow across the tricuspid valve for Doppler assessment
Three-dimensional (3D) Echocardiography : 3D echocardiography has significantly improved the accuracy of imaging and identification of the tricuspid leaflets and associated anatomic components of the tricuspid valve complex and obviates the need for mental reconstruction of multiple 2D planes [5]. Lang et al. [6] has suggested standardized imaging display (Fig. 6.2) for the en face view of the tricuspid valve with the interatrial septum placed inferiorly (at the 6 o’clock position) regardless of the atrial or ventricular orientation. The current 3D systems have different resolution for each of the 3 dimensions with axial resolution (~0.5 mm) better than lateral (~2.5 mm) and elevational resolution (~3 mm) [5]. Similar to 2D imaging however, images in the far field may be subject to beam widening and attenuation. When creating 3D images, keeping these current equipment limitations in mind will help determine the best imaging plane for imaging a specific abnormality. The best imaging plane for the tricuspid valve leaflets in systole (closed leaflets) may be the esophageal views since the closed leaflets are perpendicular to the insonation beam, however the diastolic (open) leaflets may be poorly imaged. Conversely, transgastric views may allow imaging of the diastolic leaflets since they will be perpendicular to the insonation beam, but leaflet definition may not be optimal in systole. Obtaining multiple 3D volumes from different views may still be necessary to fully characterize the valve and annulus. Finally, because of the complex nature of the valve, the volume acquired may need to have adjacent structures to help identify leaflet anatomy; the aortic valve/aorta to identify the anterior leaflet, and the interatrial septum/mitral valve to identify the septal leaflet.
Grading Severity of Tricuspid Regurgitation
Grading of the severity of the tricuspid regurgitation (TR) has been well-described by the ASE guidelines [7] as well as the European Association of Echocardiography guidelines and focuses on assessment by transthoracic imaging [8]. Nonetheless, TOE imaging can be used to assess many of the parameters, although validation of cut-offs is lacking. Table 6.1
summarizes the parameters most commonly used for this assessment. Importantly, many of the studies validating the use of these parameters have significant limitations with a lack of a “gold standard” for comparison or support from outcomes data. It is thus essential to use a multi-parametric method for determining severity since no single parameter has adequate specificity. Future studies are needed to determine the validity and prognostic utility of these parameters.
A recent study evaluated the utility of an algorithm for assessing the severity of TR [9]. Severe TR was present in the presence of a suggestive color Doppler jet and if any one or more of the following combinations of criteria were present: (1) IVC diameter > 2.5 cm AND RA area > 18 cm2 (in the absence of ASD or pulmonic valvular disease); (2) jet area > 10 cm2 AND vena contracta width > 7 mm; (3) systolic flow reversal in the hepatic veins in the absence of AV dissociation, ventricular pacing, or atrial arrhythmia; (4) triangular continuous wave Doppler signal with density equal to or greater than that of tricuspid inflow. These parameters correlated best with expert-reads and magnetic resonance imaging determined regurgitant volume and fraction (using >48% to define severe) and improved inter-observer agreement. The jet area:RA area ratio (in %) is not included in current guidelines, but was previously validated by both thermodilution techniques [10] and open surgical techniques [11].
Quantitation of the severity TR can be performed by a number of methods but also lack validation against imaging modalities such as cardiac magnetic resonance imaging. Although the proximal isovelocity surface area (PISA) method is simple and easy to perform [12], the shape of the tricuspid regurgitant orifice is often elliptical [13] or stellate which may result in significant underestimation of the ROA by this method. More recently, 3D PISA has been used to quantify TR [14]. This method uses a vendor-specific software package to analyze the largest convergence zone, and a specific software which measures the 3D PISA. Regurgitant orifice area is then calculated as 3D PISA × Valiasing)/peak TR velocity where Valiasing is the aliasing velocity. In this study, 3D PISA-derived ROA correlated well with 3D planimetered vena contracta (r = 0.97).
Few studies have used quantitation of TR by relative stroke volumes [12, 15, 16]. In these studies, a single plane tricuspid annular diameter was measured from the 4-chamber view and the tricuspid annular area calculated using a circular formula to calculate the tricuspid annular area. The sample volume for measuring the velocity-time-integral was placed at the tips of the leaflets, unlike the recommended position of the sample volume for mitral quantitation, which is at the level of annulus. Despite these limitations, there was a high correlation with catheterization-derived data. This method has not been validated in patients with pathologic tricuspid regurgitation which may be associated with asymmetric dilatation of the annulus. The Early Feasibility of the Mitralign Percutaneous Tricuspid Valve Annuloplasty System (PTVAS) (SCOUT) trial (ClinicalTrials.gov Identifier: NCT02574650) using the Trialign system (Mitralign Inc., Tewksbury, Massachusetts) has recently been reported. (Hahn RT, Meduri CU, Davidson CJ, Lim S, Nazif TM, Ricciardi MJ, Rajagopal V, Ailawadi G, Vannan MA, Thomas JD, Fowler D, Rich S, Martin R, Ong G, Groothuis A, Kodali S. Early Feasibility Study of a Transcatheter Tricuspid Valve Annuloplasty: SCOUT Trial 30-Day Results. J Am Coll Cardiol. 2017 Apr 11;69(14):1795-1806). Tricuspid regurgitation was assessed using multiple quantitative methods including PISA and relative stroke volumes. For the later, forward stroke volume was measured using with the LVOT or RVOT forward flow. Diastolic stroke volume across the tricuspid valve was calculated using orthogonal plane annular diameters in early diastole (one frame after initial valve opening) in an ellipse formula to calculate annular area, and then multiplying this by the pulsed wave Doppler velocity time integral with sample volume at the tricuspid annular plane. PISA underestimated the EROA by relative stroke volume by >50%. Importantly, a reduction in tricuspid EROA (by either method) following the transcatheter annuloplasty was associated with an increase in left ventricular outflow tract stroke volume, and significant improvement in functional status.
The quantitative EROA inclusion criteria for the patients in the SCOUT trial was </= 1.2 cm2. This is 3 times the criteria for severe. To better characterize the severity of regurgitation in this population of symptomatic patients, a new grading scheme has been proposed, including grades of massive and torrential. (Hahn RT and Zamorano, J, EurHtJ, in press).
Three-dimensional methods may help improve the accuracy of quantitative Doppler methods. Using either 3D planimetered diastolic annular area, or two orthogonal diastolic annular diameters and a pulsed sample volume at the annulus to measure the velocity time integral, a diastolic stroke volume can be measured. Subtracting the forward stroke volume (either from the left ventricular outflow tract or right ventricular outflow tract) results in the regurgitant volume. These methods require validation.
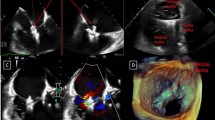
A number of studies have shown the utility of 3D color Doppler to quantify tricuspid regurgitation (Fig. 6.4) [13, 14, 17, 18]. Velayudhan et al. was one of the first to correlate standard Doppler methods of quantifying TR with planimetry of the 3D vena contracta area (VCA). Using the validated measure of regurgitant jet area/right atrial area > 34% [11] and regurgitant jet area > 10 cm2 to define severe TR [19], a 3D TTE planimetered VCA of >0.75 cm2 was the most sensitive cutoff (Sens. 85.2%, Spec. 82.1%). This higher cutoff has also been shown by Chen et al., with severe TR by 2D criteria associated with a 3D VCA of >0.6 ± 0.4 cm2 and non-severe TR by 2D methods with a 3D VCA of ≤0.3 ± 0.1 cm2. However, receiver-operator curve demonstrated that a 3D VCA of 0.36 cm2 was the best cutoff value for severe TR, with sensitivity of 89% and specificity of 84% in predicting severe TR defined by 2D echocardiographic integrative criteria.
Multi-planar reconstruction of a 3D color Doppler volume. Panel a shows the multiplanar reconstruction of a 3D color Doppler volume, where the green and red planes are used to align the blue plane in the vena contracta of the jet. The vena contracta area is then planimetered in the blue plane (panel b)
Key Points
-
Comprehensive tricuspid valve imaging on TOE involves the use of multiple probe levels as well as multi-plane and multi-angle views.
-
Use of three-dimensional imaging of the tricuspid valve can be optimized to assess leaflets and annulus.
-
Assessment of tricuspid valve regurgitation should use a multi-parametric method which includes 2D and 3D measurements, as well as Doppler semi-quantitative and quantitative measurements, although the latter requires further study.
References
Hahn RT, Abraham T, Adams MS, et al. Guidelines for performing a comprehensive transesophageal echocardiographic examination: recommendations from the American society of echocardiography and the society of cardiovascular anesthesiologists. J Am Soc Echocardiogr. 2013;26:921–64.
Hahn RT. State-of-the-Art Review of Echocardiographic Imaging in the Evaluation and Treatment of Functional Tricuspid Regurgitation. Circ Cardiovasc Imaging. 2016;9(12). pii: e005332.
Lang RM, Badano LP, Tsang W, et al. EAE/ASE recommendations for image acquisition and display using three-dimensional echocardiography. Eur Heart J Cardiovasc Imaging. 2012;13:1–46.
Schofer J, Bijuklic K, Tiburtius C, Hansen L, Groothuis A, Hahn RT. First-in-Human transcatheter tricuspid valve repair in a patient with severely regurgitant tricuspid valve. J Am Coll Cardiol. 2015;65(12):1190–5.
Badano LP, Agricola E, de Isla LP, Gianfagna P, Zamorano JL. Evaluation of the tricuspid valve morphology and function by transthoracic real-time three-dimensional echocardiography. Eur J Echocardiogr. 2009;10:477–84.
Lang RM, Badano LP, Tsang W, et al. EAE/ASE recommendations for image acquisition and display using three-dimensional echocardiography. J Am Soc Echocardiogr. 2012;25:3–46.
Zoghbi WA, Adams D, Bonow RO, Enriquez-Sarano M, Foster E, Grayburn PA, Hahn RT, Han Y, Hung J, Lang RM, Little SH, Shah DJ, Shernan S, Thavendiranathan P, Thomas JD, Weissman NJ. Recommendations for Noninvasive Evaluation of Native Valvular Regurgitation: A Report from the American Society of Echocardiography Developed in Collaboration with the Society for Cardiovascular Magnetic Resonance. J Am Soc Echocardiogr. 2017;30(4):303–71.
Lancellotti P, Moura L, Pierard LA, et al. European Association of Echocardiography recommendations for the assessment of valvular regurgitation. Part 2: mitral and tricuspid regurgitation (native valve disease). Eur J Echocardiogr. 2010;11:307–32.
Grant AD, Thavendiranathan P, Rodriguez LL, Kwon D, Marwick TH. Development of a consensus algorithm to improve interobserver agreement and accuracy in the determination of tricuspid regurgitation severity. J Am Soc Echocardiogr. 2014;27:277–84.
Mugge A, Daniel WG, Herrmann G, Simon R, Lichtlen PR. Quantification of tricuspid regurgitation by Doppler color flow mapping after cardiac transplantation. Am J Cardiol. 1990;66:884–7.
Chopra HK, Nanda NC, Fan P, et al. Can two-dimensional echocardiography and Doppler color flow mapping identify the need for tricuspid valve repair? J Am Coll Cardiol. 1989;14:1266–74.
Rivera JM, Mele D, Vandervoort PM, Morris E, Weyman AE, Thomas JD. Effective regurgitant orifice area in tricuspid regurgitation: clinical implementation and follow-up study. Am Heart J. 1994;128:927–33.
Sugeng L, Weinert L, Lang RM. Real-time 3-dimensional color Doppler flow of mitral and tricuspid regurgitation: feasibility and initial quantitative comparison with 2-dimensional methods. J Am Soc Echocardiogr. 2007;20:1050–7.
de Agustin JA, Viliani D, Vieira C, et al. Proximal isovelocity surface area by single-beat three-dimensional color Doppler echocardiography applied for tricuspid regurgitation quantification. J Am Soc Echocardiogr. 2013;26:1063–72.
Loeber CP, Goldberg SJ, Allen HD. Doppler echocardiographic comparison of flows distal to the four cardiac valves. J Am Coll Cardiol. 1984;4:268–72.
Meijboom EJ, Horowitz S, Valdes-Cruz LM, Sahn DJ, Larson DF, Oliveira Lima C. A Doppler echocardiographic method for calculating volume flow across the tricuspid valve: correlative laboratory and clinical studies. Circulation. 1985;71:551–6.
Velayudhan DE, Brown TM, Nanda NC, et al. Quantification of tricuspid regurgitation by live three-dimensional transthoracic echocardiographic measurements of vena contracta area. Echocardiography. 2006;23:793–800.
Chen TE, Kwon SH, Enriquez-Sarano M, Wong BF, Mankad SV. Three-dimensional color Doppler echocardiographic quantification of tricuspid regurgitation orifice area: comparison with conventional two-dimensional measures. J Am Soc Echocardiogr. 2013;26:1143–52.
Gonzalez-Vilchez F, Zarauza J, Vazquez de Prada JA, et al. Assessment of tricuspid regurgitation by Doppler color flow imaging: angiographic correlation. Int J Cardiol. 1994;44:275–83.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Review Questions
Review Questions
Select the Single Best Sentence
-
42.
Which leaflet is seen adjacent to the aortic valve?
-
(a)
Septal leaflet
-
(b)
Anterior leaflet
-
(c)
Posterior leaflet
-
(d)
Anterior or posterior leaflets
-
(e)
Posterior or septal leaflets
-
(a)
-
43.
Which of the following probe locations and angles allows imaging of all three tricuspid leaflets in a single view?
-
(a)
Mid-esophageal view, 0 degrees.
-
(b)
Distal esophageal view, 90 degrees.
-
(c)
Transgastric view, 0 degrees.
-
(d)
Transgastric view, 60 degrees.
-
(e)
Deep transgastric view, 60 degrees.
-
(a)
-
44.
Which of the following statements is most accurate regarding the assessment of tricuspid regurgitation severity?
-
(a)
Well-validated criteria have been established to assess the severity of tricuspid regurgitation.
-
(b)
Color Doppler parameters alone can be reliably used to assess severity.
-
(c)
Quantitative Doppler methods have been validated against cardiac magnetic resonance imaging and are the recommended method of assessing severity.
-
(d)
A multi-parametric method should be used to assess tricuspid regurgitation severity since no single measurement has adequate sensitivity and specificity.
-
(e)
Three-dimensional planimetry of the vena contracta has been validated against outcomes data.
-
(a)
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG
About this chapter
Cite this chapter
Hahn, R.T. (2018). Imaging of the Tricuspid Valve: Transoesophageal Echocardiography. In: Soliman, O.I., ten Cate, F.J. (eds) Practical Manual of Tricuspid Valve Diseases. Springer, Cham. https://doi.org/10.1007/978-3-319-58229-0_6
Download citation
DOI: https://doi.org/10.1007/978-3-319-58229-0_6
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-58228-3
Online ISBN: 978-3-319-58229-0
eBook Packages: MedicineMedicine (R0)