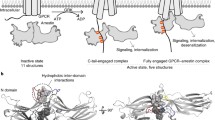
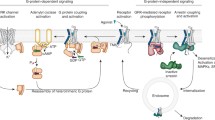
Abstract
G protein-coupled receptors (GPCR ) signal primarily through G proteins and arrestins. Arrestin not only intervenes with G protein-mediated signaling, but also initiates a variety of G protein-independent functions. The functional diversity of arrestins has been attributed to their multiple active conformations after interaction with ligand-activated receptors, which are regulated by both the ligand-specific conformational states and the phosphorylation patterns of the receptors. Here we reviewed the current knowledge of the structural features of arrestins that underlie selective arrestin-mediated signaling. Recent breakthroughs in the functional correlation of the conformational changes in arrestin to downstream effector molecules were also highlighted.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
G protein-coupled receptors (GPCRs ), also known as seven-transmembrane domain receptors (7TMRs), are the largest family of membrane proteins that communicate extracellular stimuli to intracellular signals and play critical roles in mediating most known physiological functions (Pierce et al. 2002). The classical paradigm of GPCR signaling is based on a ligand-induced conformational change of the receptor (Farrens et al. 1996), which is recognized by heterotrimeric G proteins to regulate levels of second messengers. The activated receptor is also phosphorylated by GRKs, which results in subsequent recruitment of arrestins for endocytosis and/or G protein-independent signaling (Walther and Ferguson 2013; Thomsen et al. 2016).
The arrestin family consists of four members. In mammals, arrestin-1 and arrestin-4 are located almost exclusively in photoreceptor cells in the retina and interact with very few visual GPCRs (Wilden et al. 1986; Craft et al. 1994), whereas β-arrestin-1 and β-arrestin-2 (also called arrestin-2 and arrestin-3, respectively) are ubiquitously expressed in most tissues and interact with low specificity with the majority of non-visual GPCRs (Lohse et al. 1990; Attramadal et al. 1992). By binding to GPCRs, these four arrestins desensitize G protein-mediated signaling in many cases and also function as adapters to promote receptor internalization by recruiting endocytic proteins, such as clathrin (Goodman et al. 1996), adaptor protein 2 (AP2) (Laporte et al. 1999) and N-ethylmaleimide-sensitive fusion protein (NSF) (McDonald et al. 1999), linking the receptors to the clathrin-coated pit machinery (Shenoy and Lefkowitz 2003; Lefkowitz and Shenoy 2005). In addition to their roles in GPCR desensitization, internalization and trafficking, mounting evidence has revealed that β-arrestins also act as signaling transducers that interact with a growing list of intracellular effectors that include, but not limited to, signaling proteins such as c-Src, Raf-1, Akt, ERK1/2, JNK3 and IκB (Luttrell et al. 1999; Gao et al. 2004; Xiao et al. 2010; Reiter et al. 2012; Wang et al. 2014; Ning et al. 2015; Smith and Rajagopal 2016). Through interaction with these signaling molecules, β-arrestins are capable of initiating G-protein-independent signaling via MAPK, PI3K/AKT, and NF-κB pathways, connecting the activated receptor to diverse physiological responses. Although some studies indicate that G protein and arrestin signal synergistically (Thomsen et al. 2016), β-arrestin -dependent and G-protein-dependent signaling pathways could be pharmacologically separable. Biased ligands, which selectively activate one of these two signaling pathways have great therapeutic potential and have been extensively studied (Rajagopal et al. 2010; Reiter et al. 2012).
The remarkable functional diversity of arrestins raises the inevitable question of how distinct arrestin functions are precisely regulated. Aiming at the detailed mechanism, numerous biomedical and biophysical studies have been performed over the past decade using multiple novel techniques and approaches, leading to fruitful results. Recent structural studies of GPCR -arrestin complex provided considerable insight into the activation mechanism of arrestin (Kang et al. 2015). In the present chapter, we will review current knowledge of the structural features that underlie selective arrestin signaling.
Arrestin Undergoes Conformational Changes After Interaction with Activated Receptors
Based on the diverse functional capacities of arrestins, especially β-arrestins, it has been hypothesized that arrestins adopt multiple active conformations after activation that dictate their distinct functions (Gurevich and Gurevich 2006). In cells, the conformational change of β-arrestin-2 was detected by BRET-based biosensors after activation of the angiotensin II receptor type 1a (AT1aR), β2 adrenergic receptor (β2AR), and parathyroid hormone 1 receptor (PTH1R) (Shukla et al. 2008). In detail, different conformations of β-arrestin-2 induced by full agonists or β-arrestin-biased agonists in stimulated cells were observed with the BRET sensor. Later, the conformational biosensor of arrestin was improved by the insertion of the FlAsH (intramolecular fluorescein arsenical hairpin) in the specific loops of arrestin. Significant conformational features of arrestins that correlated with their downstream functions were revealed by this new version of probes (Lee et al. 2016). Further, using the FlAsH-decorated arrestins, a rapid activation/deactivation cycle of β-arrestins was disclosed by FRET analysis, which reshapes our thinking of the arrestin signaling (Nuber et al. 2016).
Phosphorylation of the GPCRs plays a key role in the recruitment and activation of arrestins . Since the 1970s, the finding of phosphorylation of rhodopsin and β2AR on their C-termini during desensitization has led to the identification of a seven-member family of G protein-coupled receptor kinases (GRKs) (Weller et al. 1975; Benovic et al. 1986; Pitcher et al. 1998). Although subsequent studies revealed that receptors could be phosphorylated by multiple intracellular kinases other than GRKs, such as PKA (Tran et al. 2004) and PKC (Garcia et al. 1998), it has been suggested that the phosphorylation mediated by GRKs initiates arrestin recruitment and receptor internalization (Tobin et al. 2008; Walther and Ferguson 2013). Whereas GRK1 and 7 are confined to the retina and GRK4 is distributed primarily in the reproductive system, GRKs 2, 3, 5, and 6 are ubiquitously expressed and thus regulate the phosphorylation patterns of most GPCRs (Krupnick and Benovic 1998; Pitcher et al. 1998). Studies of different GPCRs revealed that siRNA-mediated knockdown of specific GRK or combinations of GRKs leads to clearly distinguishable effects on receptor functions, suggesting that different sets of GRKs might phosphorylate distinct sites on the receptors. Many of the regulatory roles of GRKs on receptors are through arrestin-mediated functions, including receptor desensitization, internalization and non-receptor-binding partners activation (Kim et al. 2005; Ren et al. 2005). Studies regarding specific GRK-regulated GPCR functions through arrestin brought up the barcode concept of the phosphorylation-regulated receptor function, of which the phosphorylation pattern of the receptor resembles the barcode of the beer can, transducing specific information to downstream effectors (Tobin et al. 2008; Nobles et al. 2011). Using the prototypical GPCR β2AR as a model, mass spectrometry-based quantitative proteomics and phosphorylation site-specific antibodies were exploited to map the phosphorylation sites of GRK2 and GRK6 at C-terminus and the third intracellular loop of the β2AR expressed in HEK293 cells, whereas RNAi-induced GRK silencing and BRET assay were employed to delineate conformation-specific β-arrestin capabilities imparted by specific phosphorylation patterns induced by GRK2 and GRK6. Results indicated that whereas both GRK2 and GRK6 contribute to desensitization of the β2AR through β-arrestin-2, GRK2 phosphorylation sites (T360, S364, S396, S401, S407, and S411) are primarily responsible for receptor internalization, whereas GRK6 sites (S355 and S356) are required for β-arrestin2-mediated ERK1/2 activation . Notably, phosphorylation of GRK2 sites appears to inhibit phosphorylation of GRK6 sites as well as β2AR signaling to ERK1/2. It was also demonstrated that isoproterenol, the full agonist of β2AR, initiates phosphorylation on both GRK2 and GRK6 sites, whereas carvedilol, a β-arrestin-biased ligand of β2AR (Wisler et al. 2007), induces phosphorylation only at the GRK6 sites. Moreover, the BRET data suggested that distinct phosphorylation patterns on the β2AR induced by either GRK2 or GRK6 result in different β-arrestin-2 conformations. Collectively, these results provide evidence that the receptor phosphorylation patterns induced by different GRKs establish a ‘barcode’ that is related to its functional capabilities. This barcode hypothesis has been strengthened by growing evidence from the studies of many other GPCRs, such as M3 muscarinic receptor (Butcher et al. 2011), cannabinoid 1 receptor (Delgado-Peraza et al. 2016), ghrelin receptor (Bouzo-Lorenzo et al. 2016), and fatty acid receptor GPR120 (Prihandoko et al. 2016).
In vitro, binding of the fully phosphorylated vasopressin 2 receptor (V2R) C-tail induces significant conformational changes of both β-arrestin-1 (Nobles et al. 2007) and β-arrestin-2 (Xiao et al. 2004). Recently, a flute model of how phosphorylation pattern of GPCRs was recognized by arrestin and thereafter transduced to downstream effectors through specific conformational changes were revealed by 19F-NMR and unnatural amino acid incorporation approaches (Yang et al. 2015). Not only the phosphorylation states of the receptor, but the ligand-induced specific receptor conformations also contribute to specific arrestin conformations and thus activation . Studies comparing the binding of both visual and non-visual arrestins to four functional forms (inactive unphosphorylated receptor, inactive phosphoreceptor, active unphosphorylated receptor, and active phosphoreceptor) of rhodopsin, β2AR and M2 muscarinic receptor suggested the existence of an ‘activation sensor’ in arrestin, which is proposed to mediate the interaction of arrestin with the ligand-induced activated conformation of the receptor (Gurevich et al. 1995; Gurevich and Gurevich 2004). Correspondingly, electron paramagnetic resonance (EPR) studies on rhodopsin-arrestin interactions revealed strong intermolecular contacts between the finger loop, a highly flexible loop connecting β-strands V and VI of arrestin, and the transmembrane core of light-activated phosphorylated rhodopsin, but not that of inactive phosphorylated rhodopsin (Hanson et al. 2006). Moreover, the light-activated unphosphorylated rhodopsin has been further demonstrated to independently induce a unique conformational change in arrestin, which is different from that induced by phosphorylated rhodopsin (Zhuang et al. 2013). Similarly, A NAPol-reconstituted V2R was demonstrated to interact with purified β-arrestin-2 after stimulation with full agonist AVP or β-arrestin -biased ligand SR121463, but not with the Gs-biased ligand MCF14 (Rahmeh et al. 2012). Therefore, ligand-specific conformational states of GPCR might not only determine the GRK-mediated phosphorylation pattern but also be directly correlated with the conformation changes of arrestin (Fig. 16.1).
A model of arrestin recruitment and activation by phosphorylated GPCRs. GPCRs are phosphorylated after activation , leading to the recruitment of arrestins. The phosphorylation states of the receptor and ligand-induced receptor conformations collectively determine the active conformations of the arrestins
Taken together, these findings revealed how arrestin is activated and then directs distinct functions downstream of the receptor. Ligand-dependent stabilization of distinct receptor conformations leads to specific phosphorylation patterns at the C-terminus and/or intracellular loops, which, together with ligand-specific conformational states of the receptor, define distinct arrestin conformations that might dictate selective signaling pathways (Fig. 16.1).
Structural Insight into Arrestin Conformational Change by Crystallography
Crystal structures of all four arrestins at inactive or basal state have been determined and they demonstrate high sequence and structural homology (Hirsch et al. 1999; Han et al. 2001; Sutton et al. 2005; Zhan et al. 2011). The arrestins essentially consist of N- and C-domains that are built almost entirely from antiparallel β sheets and linked by a relatively flexible ‘hinge’ region (Fig. 16.2a). The inactive conformation of the arrestin has been suggested to be constrained by two distinct subsets of intramolecular interactions: polar core that consists of five interacting charged residues in the center of arrestin (D26, R169, D290, D297, and R393 in β-arrestin-1) (Vishnivetskiy et al. 1999) and three-element interactions that comprises the β-strand I, α-helix I, and C-terminus of arrestin (Fig. 16.2b, d) (Vishnivetskiy et al. 2000).
Comparison of inactive β-arrestin-1 structure and active β-arrestin-1 structure. a Overall comparison of the inactive β-arrestin -1 structure (PDB accession code 1G4M; gold) and the active β-arrestin-1 structure (PDB accession code 4JQI; purple). The β-arrestin-1 C-terminus and the V2Rpp are highlighted in red and blue, respectively. The three-element interaction in the inactive β-arrestin-1 structure (b) and active β-arrestin-1 structure (c). The β-arrestin-1 C-terminal b-strand lying along the three-element interaction in the inactive conformation is displaced by the C-terminus of V2Rpp upon activation . The polar core in the inactive β-arrestin-1 structure (d) and active β-arrestin-1 structure (e). Upon V2Rpp binding, C-terminal residue R393 of β-arrestin-1is displaced, contributing to the disruption of the polar core
Multiple mutagenesis and biophysical studies suggested that visual arrestin undergoes a conformational change upon binding to light-activated phosphorylated rhodopsin (Gurevich and Gurevich 2003, 2004). Subsequently, direct evidence of a substantial conformational change of both β-arrestins upon activation was obtained using limited tryptic proteolysis and matrix-assisted laser desorption/ionization-time of flight mass spectrometry analysis in the presence of a phosphopeptide derived from the C-terminus of V2R (Xiao et al. 2004; Nobles et al. 2007). Collectively, results from these studies support a model in which both polar core and three-element interactions might be disrupted during the activation of the arrestin (Gurevich and Gurevich 2006; Ostermaier et al. 2014).
Two X-ray crystal structures of activated arrestins , including pre-activated arrestin-1 (Kim et al. 2013) and V2R phosphopeptide (V2Rpp)-bound β-arrestin-1 (Shukla et al. 2013), were determined in 2013. In the latter study, the crystal structure of activated β-arrestin-1 revealed the displacement of β-arrestin-1 C-terminus by V2Rpp that binds to the N-domain as an antiparallel β-strand at a similar location to the β-arrestin-1 C-terminus in inactive structure but with a reversed direction (Fig. 16.2a, c). The replacement of the C-terminus of arrestin with V2Rpp disrupts both 3-element interaction and the polar core that restrain the arrestin in the inactive state (Fig. 16.2c, e). It was observed that two highly conserved residues on the β-stand I, the K10 and K11, which interact with F388 and F391 and are proposed to be a key component of three-element interaction in the inactive structure (Gurevich and Gurevich 2006), make instead charge-charge interactions with phosphates of V2Rpp, which therefore disrupt the 3-element interaction (Fig. 16.2c). Displacement of C-terminus of β-arrestin-1 also removes the R393 of the C-terminus, therefore disables the charge-charge interaction of the R393 with the D26 and D297, and distorts the lariat loop to form the new interaction between the pT360 and K294 (Fig. 16.2e). These changes disrupt the polar core. In the active structure of the V2Rpp/arrestin complex, the disruption of the 3-element interaction and the polar core in arrestin release the N-/C-domain associations and enable 20° twisting of the domains relative to each other. The twisting of these two domains of arrestin not only repositions three loops, including the finger loop, the middle loop and the lariat loop, which may facilitate receptor core interactions; but also exposes substantial regions that might enable downstream effectors recruitment (Sommer et al. 2012; Vishnivetskiy et al. 2013; Zhuo et al. 2014).
A similar twisting between the N-/C-domains of the visual arrestin (arrestin-1 or v-arrestin ) was also observed in the recently solved crystal structure of rhodopsin/v-arrestin-phosphorylation-independent-active-mutant fusion complex obtained by serial femtosecond X-ray laser crystallography (Fig. 16.3) (Kang et al. 2015). Conformational changes of the finger loop, middle loop and the region between the I157-K168 enable the interaction of the rhodopsin with the v-arrestin. In detail, whereas the interaction between the rhodopsin C-terminus and v-arrestin N-domain was not observed, this structure revealed three patches of rhodopsin-v-arrestin interfaces, which include the interaction of the finger loop of v-arrestin with the intracellular loop-1 (ICL1), TM7 and H8 of the rhodopsin, the accommodation of rhodopsin ICL2 by the middle and C-loops of v-arrestin, and the engagement of β-strand following finger loop and the N-terminal β-strand 6 with the TM5, TM6, and ICL3 of rhodopsin. Interestingly, the structure of the v-arrestin bound to rhodopsin and β-arrestin-1 bound to V2Rpp are very similar other than the receptor interaction regions, suggesting activated arrestins assume similar conformations (Fig. 16.3). However, this result does not support the idea that distinct arrestin functions result from different receptor activation and phosphorylation by different GRKs. Therefore, the low-resolution crystal structures lack important detailed information of structural changes in arrestin to explain how it mediates differential signaling.
Conformational Changes in Arrestin and the Correlation with Downstream Functions
Although mounting evidence has indicated that different arrestin conformations are coupled to distinct functional outcomes, the precise correlation between them is not well established. Since the identification of clathrin as the first non-receptor binding partner of arrestins, a variety of trafficking and signaling proteins, such as c-Src, MAPK, and ASK1, have been reported to interact with arrestin (Goodman et al. 1996; Lefkowitz and Shenoy 2005). The binding sites of clathrin and AP2 on arrestin have been well characterized: both are localized in the C-terminus of β-arrestins (Kim and Benovic 2002; Schmid et al. 2006; Kang et al. 2009). In contrast, few binding sites of other non-receptor binding partners have been precisely mapped, although particular residues in β-arrestins are suggested to participate in the binding of MEK1 or Raf1 (Meng et al. 2009; Coffa et al. 2011a, b). It has been demonstrated that most of these non-receptor binding partners except for clathrin and AP2 interact with both N-/C-domains of arrestin (Song et al. 2007, 2009) and therefore, the relative rotation between these two domains and rearrangement of the arrestin conformation, which have been confirmed in the structural studies, might explain the structural preference of certain partners that serve as effector molecules leading to distinct functional outcomes (Gurevich and Gurevich 2003, 2013). However, despite the continuous improvement and strengthening of barcode hypothesis, the detailed mechanism by which arrestin transmits signals encoded in the receptor to downstream effector molecules still remains largely unknown.
To better detect the conformational changes in arrestin and further explore how arrestin recognizes specific phosphorylation patterns in the receptor and translates them into distinct functional outcomes, we developed a series of structural sensors in β-arrestin-1 by incorporating the unnatural amino acid 3, 5-difluorotyrosin (F2Y) at specific locations and detected phospho-interaction patterns and residue-specific structural information in β-arrestin-1 using 19F-NMR spectroscopy (Yang et al. 2015). In addition to V2Rpp, we also synthesized specific GRK2-, GRK6-, or PKA-phosphorylated β2AR C-terminal fragments (GRK2pp, GRK6pp, and PKApp, respectively) and examined their effects on the biochemical properties of β-arrestin-1. We demonstrated that β-arrestin-1 reads phospho-messages in receptor C-tail with its concave surface, which harbors at least ten potential phosphate-binding sites (numbered 1–7 according to the binding mode of V2Rpp to β-arrestin-1 in the V2Rpp/β-arrestin-1 complex (Shukla et al. 2014), with A1–A3 indicating potential additional phosphate-binding sites at the N-terminus). Although all the GRK-phosphopeptides (GRKpps) but not PKApp interact with phosphate-binding site 1 of β-arrestin-1, our results revealed distinct phospho-interaction patterns between different GRKpps and β-arrestin-1 at the other binding sites, which are coupled to selective functional outcomes (Fig. 16.4a). Whereas GRK2pp interacts with β-arrestin-1 through binding sites 1–4–6–7 and promotes clathrin recruitment and receptor endocytosis, GRK6pp interacts with β-arrestin -1 in a different 1–5 pattern and elicits Src signaling (Fig. 16.4a). Mutations of key residues in specific phosphate-binding sites selectively eliminate corresponding β-arresin-1-mediated functions downstream of several GPCRs, including β2AR, cholecystokinin type-A receptor (CCKAR), and somatostatin receptor type 2 (SSTR2). These data suggested that specific phospho-patterns were recognized by arrestins and then translated to different cellular signaling pathways through distinct downstream effectors.
Conformational changes of arrestin induced by different receptor phosphorylation patterns and their correlation with distinct cellular functions. a The flute model for the phospho-decision mechanism of the arrestin signaling. A phospho-barcode of 1–4–6–7 directs clathrin recruitment and endocytic function of arrestin , whereas a phospho-barcode of 1–5 directs Src recruitment and signaling. There are potentially more than 1000 phospho-patterns that a single arrestin could recognize. b Conformationl changes of arrestin correlate to its distinct functions. The conformational states of the Y249, L338 and N375 are recognized by clathrin and correlated to the endocytic function of β-arrestin-1, whereas the conformational state of F277 is correlated to arrestin-mediated Src signaling
Using 7 19F-NMR probes incorporated into the specific arrestin sites other than phospho-interaction regions, we were able to detect different conformational states that were induced by binding of different GRKpps. Binding of the two distinct GRK2pp phospho-peptides induced conformational change at the Y249 in the loop between β-strands XV and XVI, the L338 in the splice loop, and the N375 in the C-terminal region of β-arrestin-1 (Fig. 16.4b). These structural states could be specifically recognized by the subsequent binding of clathrin. In contrast, binding of the GRK6pp to β-arrestin -1 caused a chemical shift at F277 in the end of the lariat loop, whereas binding of the GRK2pps caused no significant conformational change at this site (Fig. 16.4b). Deletion of the partial lariat loop encompassing F277 abolished the GRK6pp-induced recruitment of Src to arrestin. Collectively, these data have clarified the receptor phospho-coding mechanism, by which changes in a GPCR phosphorylation pattern are translated to distinct conformations of arrestin that could be further recognized by different downstream effector molecules.
In addition to β-arrestin-1, the conformational change of β-arrestin-2 have also been investigated downstream of several receptors in cellular systems. A very recent study monitored the conformational changes of β-arrestin-2 using a panel of intramolecular fluorescein arsenical hairpin (FlAsH) BRET reporters in cells (Lee et al. 2016). Studies of six different types of GPCRs demonstrated the existence of β-arresin-2 “conformational signature” that was indicated by the changes in BRET efficacy from multiple vantage points. This β-arresin-2-FlAsH signature is conserved between GPCRs with similar arrestin binding/signaling characteristics and the changes in BRET efficacy at selected positions correlate with distinct arrestin functions, for example, trafficking pattern of GPCR-arrestin complex and arrestin -dependent ERK1/2 activation . Therefore, these data, together with the results of our study, provide considerable insight into the correlation between specific arrestin conformations and selective arrestin functions and pave the way to the future studies of the detailed functional roles of the conformational changes of arrestin.
Although receptor binding and arrestin conformational change are necessary for the engagement of arrestin with multiple downstream effectors, such as ERK1/2 (Luttrell et al. 2001; Coffa et al. 2011a, b), it has been reported that some binding partners could interact with arrestin in both inactive and active conformations (Song et al. 2009; Ahmed et al. 2011; Gurevich and Gurevich 2013), with some proteins, such as MEK1, even binding to arrestins in both conformations equally well (Coffa et al. 2011a, b; Gurevich and Gurevich 2014). This has added a new layer of complexity in the arrestin-dependent conformational signaling. Moreover, the two isoforms of β-arrestin, which share more than 70% sequence identity and high structural similarity, have been demonstrated to be functionally non-redundant in GPCR regulation by accumulating evidence (Srivastava et al. 2015). Whereas both β-arrestins facilitate the signaling in c-Raf1-MEK1-ERK1/2 cascade, only β-arrestin-2 promotes the signaling in ASK1-MKK4-JNK3 cascade (McDonald et al. 2000). Receptor-specific reciprocal regulation of selective signaling pathways by these two β-arrestins has also been reported. For example, siRNA knockdown of β-arrestin-2 attenuates PTH1R-mediated ERK1/2 activation , whereas knockdown of β-arrestin-1 yields opposite effect (Ahn et al. 2004). Therefore, the interaction between arrestin and effector molecules and the arrestin-dependent signal transduction might be much more complicated than expected. To further decipher the molecular mechanism underlying arrestin conformational signaling, more detailed information from the potential structures , using crystallography , electron microscopy or NMR approaches, of GPCR with arrestin and effectors is required.
References
Ahmed MR, Zhan X, Song X, Kook S, Gurevich VV, Gurevich EV (2011) Ubiquitin ligase parkin promotes Mdm2-arrestin interaction but inhibits arrestin ubiquitination. Biochemistry 50(18):3749–3763
Ahn S, Wei H, Garrison TR, Lefkowitz RJ (2004) Reciprocal regulation of angiotensin receptor-activated extracellular signal-regulated kinases by beta-arrestins 1 and 2. J Biol Chem 279(9):7807–7811
Attramadal H, Arriza JL, Aoki C, Dawson TM, Codina J, Kwatra MM, Snyder SH, Caron MG, Lefkowitz RJ (1992) Beta-arrestin2, a novel member of the arrestin/beta-arrestin gene family. J Biol Chem 267(25):17882–17890
Benovic JL, Strasser RH, Caron MG, Lefkowitz RJ (1986) Beta-adrenergic receptor kinase: identification of a novel protein kinase that phosphorylates the agonist-occupied form of the receptor. Proc Natl Acad Sci USA 83(9):2797–2801
Bouzo-Lorenzo M, Santo-Zas I, Lodeiro M, Nogueiras R, Casanueva FF, Castro M, Pazos Y, Tobin AB, Butcher AJ, Camina JP (2016) Distinct phosphorylation sites on the ghrelin receptor, GHSR1a, establish a code that determines the functions of ss-arrestins. Sci Rep 6:22495
Butcher AJ, Prihandoko R, Kong KC, McWilliams P, Edwards JM, Bottrill A, Mistry S, Tobin AB (2011) Differential G-protein-coupled receptor phosphorylation provides evidence for a signaling bar code. J Biol Chem 286(13):11506–11518
Coffa S, Breitman M, Hanson SM, Callaway K, Kook S, Dalby KN, Gurevich VV (2011a) The effect of arrestin conformation on the recruitment of c-Raf1, MEK1, and ERK1/2 activation. PLoS One 6(12):e28723
Coffa S, Breitman M, Spiller BW, Gurevich VV (2011b) A single mutation in arrestin-2 prevents ERK1/2 activation by reducing c-Raf1 binding. Biochemistry 50(32):6951–6958
Craft CM, Whitmore DH, Wiechmann AF (1994) Cone arrestin identified by targeting expression of a functional family. J Biol Chem 269(6):4613–4619
Delgado-Peraza F, Ahn KH, Nogueras-Ortiz C, Mungrue IN, Mackie K, Kendall DA, Yudowski GA (2016) Mechanisms of biased beta-arrestin-mediated signaling downstream from the cannabinoid 1 receptor. Mol Pharmacol 89(6):618–629
Farrens DL, Altenbach C, Yang K, Hubbell WL, Khorana HG (1996) Requirement of rigid-body motion of transmembrane helices for light activation of rhodopsin. Science 274:768–770
Gao H, Sun Y, Wu Y, Luan B, Wang Y, Qu B, Pei G (2004) Identification of beta-arrestin2 as a G protein-coupled receptor-stimulated regulator of NF-kappaB pathways. Mol Cell 14(3):303–317
Garcia DE, Brown S, Hille B, Mackie K (1998) Protein kinase C disrupts cannabinoid actions by phosphorylation of the CB1 cannabinoid receptor. J Neurosci 18(8):2834–2841
Goodman OB Jr, Krupnick JG, Santini F, Gurevich VV, Penn RB, Gagnon AW, Keen JH, Benovic JL (1996) Beta-arrestin acts as a clathrin adaptor in endocytosis of the beta2-adrenergic receptor. Nature 383(6599):447–450
Gurevich VV, Dion SB, Onorato JJ, Ptasienski J, Kim CM, Sterne-Marr R, Hosey MM, Benovic JL (1995) Arrestin interactions with G protein-coupled receptors. Direct binding studies of wild type and mutant arrestins with rhodopsin, beta 2-adrenergic, and m2 muscarinic cholinergic receptors. J Biol Chem 270(2):720–731
Gurevich VV, Gurevich EV (2003) The new face of active receptor bound arrestin attracts new partners. Structure 11(9):1037–1042
Gurevich VV, Gurevich EV (2004) The molecular acrobatics of arrestin activation. Trends Pharmacol Sci 25(2):105–111
Gurevich VV, Gurevich EV (2006) The structural basis of arrestin-mediated regulation of G-protein-coupled receptors. Pharmacol Ther 110(3):465–502
Gurevich VV, Gurevich EV (2013) Structural determinants of arrestin functions. Prog Mol Biol Transl Sci 118:57–92
Gurevich VV, Gurevich EV (2014) Extensive shape shifting underlies functional versatility of arrestins. Curr Opin Cell Biol 27:1–9
Han M, Gurevich VV, Vishnivetskiy SA, Sigler PB, Schubert C (2001) Crystal structure of beta-arrestin at 1.9 A: possible mechanism of receptor binding and membrane translocation. Structure 9(9):869–880
Hanson SM, Francis DJ, Vishnivetskiy SA, Kolobova EA, Hubbell WL, Klug CS, Gurevich VV (2006) Differential interaction of spin-labeled arrestin with inactive and active phosphorhodopsin. Proc Natl Acad Sci U S A 103(13):4900–4905
Hirsch JA, Schubert C, Gurevich VV, Sigler PB (1999) The 2.8 A crystal structure of visual arrestin: a model for arrestin’s regulation. Cell 97(2):257–269
Kang DS, Kern RC, Puthenveedu MA, von Zastrow M, Williams JC, Benovic JL (2009) Structure of an arrestin2-clathrin complex reveals a novel clathrin binding domain that modulates receptor trafficking. J Biol Chem 284(43):29860–29872
Kang Y, Zhou XE, Gao X, He Y, Liu W, Ishchenko A, Barty A, White TA, Yefanov O, Han GW, Xu Q, de Waal PW, Ke J, Tan MH, Zhang C, Moeller A, West GM, Pascal BD, Van Eps N, Caro LN, Vishnivetskiy SA, Lee RJ, Suino-Powell KM, Gu X, Pal K, Ma J, Zhi X, Boutet S, Williams GJ, Messerschmidt M, Gati C, Zatsepin NA, Wang D, James D, Basu S, Roy-Chowdhury S, Conrad CE, Coe J, Liu H, Lisova S, Kupitz C, Grotjohann I, Fromme R, Jiang Y, Tan M, Yang H, Li J, Wang M, Zheng Z, Li D, Howe N, Zhao Y, Standfuss J, Diederichs K, Dong Y, Potter CS, Carragher B, Caffrey M, Jiang H, Chapman HN, Spence JC, Fromme P, Weierstall U, Ernst OP, Katritch V, Gurevich VV, Griffin PR, Hubbell WL, Stevens RC, Cherezov V, Melcher K, Xu HE (2015) Crystal structure of rhodopsin bound to arrestin by femtosecond X-ray laser. Nature 523(7562):561–567
Kim J, Ahn S, Ren XR, Whalen EJ, Reiter E, Wei H, Lefkowitz RJ (2005) Functional antagonism of different G protein-coupled receptor kinases for beta-arrestin-mediated angiotensin II receptor signaling. Proc Natl Acad Sci U S A 102(5):1442–1447
Kim YJ, Hofmann KP, Ernst OP, Scheerer P, Choe HW, Sommer ME (2013) Crystal structure of pre-activated arrestin p44. Nature 497(7447):142–146
Kim YM, Benovic JL (2002) Differential roles of arrestin-2 interaction with clathrin and adaptor protein 2 in G protein-coupled receptor trafficking. J Biol Chem 277(34):30760–30768
Krupnick JG, Benovic JL (1998) The role of receptor kinases and arrestins in G protein-coupled receptor regulation. Annu Rev Pharmacol Toxicol 38:289–319
Laporte SA, Oakley RH, Zhang J, Holt JA, Ferguson SG, Caron MG, Barak LS (1999) The 2-adrenergic receptor/arrestin complex recruits the clathrin adaptor AP-2 during endocytosis. Proc Nat Acad Sci U S A 96:3712–3717
Lee MH, Appleton KM, Strungs EG, Kwon JY, Morinelli TA, Peterson YK, Laporte SA, Luttrell LM (2016) The conformational signature of beta-arrestin2 predicts its trafficking and signalling functions. Nature 531(7596):665–668
Lefkowitz RJ, Shenoy SK (2005) Transduction of receptor signals by beta-arrestins. Science 308(5721):512–517
Lohse MJ, Benovic JL, Codina J, Caron MG, Lefkowitz RJ (1990) beta-Arrestin: a protein that regulates beta-adrenergic receptor function. Science 248(4962):1547–1550
Luttrell LM, Ferguson SS, Daaka Y, Miller WE, Maudsley S, Della Rocca GJ, Lin F, Kawakatsu H, Owada K, Luttrell DK, Caron MG, Lefkowitz RJ (1999) Beta-arrestin-dependent formation of beta2 adrenergic receptor-Src protein kinase complexes. Science 283(5402):655–661
Luttrell LM, Roudabush FL, Choy EW, Miller WE, Field ME, Pierce KL, Lefkowitz RJ (2001) Activation and targeting of extracellular signal-regulated kinases by beta-arrestin scaffolds. Proc Natl Acad Sci U S A 98(5):2449–2454
McDonald PH, Chow CW, Miller WE, Laporte SA, Field ME, Lin FT, Davis RJ, Lefkowitz RJ (2000) Beta-arrestin 2: a receptor-regulated MAPK scaffold for the activation of JNK3. Science 290(5496):1574–1577
McDonald PH, Cote NL, Lin FT, Premont RT, Pitcher JA, Lefkowitz RJ (1999) Identification of NSF as a beta-arrestin1-binding protein. Implications for beta2-adrenergic receptor regulation. J Biol Chem 274:10677–10680
Meng D, Lynch MJ, Huston E, Beyermann M, Eichhorst J, Adams DR, Klussmann E, Houslay MD, Baillie GS (2009) MEK1 binds directly to betaarrestin1, influencing both its phosphorylation by ERK and the timing of its isoprenaline-stimulated internalization. J Biol Chem 284(17):11425–11435
Ning SL, Zheng WS, Su J, Liang N, Li H, Zhang DL, Liu CH, Dong JH, Zhang ZK, Cui M, Hu QX, Chen CC, Liu CH, Wang C, Pang Q, Chen YX, Yu X, Sun JP (2015) Different downstream signalling of CCK1 receptors regulates distinct functions of CCK in pancreatic beta cells. Br J Pharmacol 172(21):5050–5067
Nobles KN, Guan Z, Xiao K, Oas TG, Lefkowitz RJ (2007) The active conformation of beta-arrestin1: direct evidence for the phosphate sensor in the N-domain and conformational differences in the active states of beta-arrestins1 and -2. J Biol Chem 282(29):21370–21381
Nobles KN, Xiao K, Ahn S, Shukla AK, Lam CM, Rajagopal S, Strachan RT, Huang TY, Bressler EA, Hara MR, Shenoy SK, Gygi SP, Lefkowitz RJ (2011) Distinct phosphorylation sites on the beta(2)-adrenergic receptor establish a barcode that encodes differential functions of beta-arrestin. Sci Signal 4(185):ra51
Nuber S, Zabel U, Lorenz K, Nuber A, Milligan G, Tobin AB, Lohse MJ, Hoffmann C (2016) beta-Arrestin biosensors reveal a rapid, receptor-dependent activation/deactivation cycle. Nature 531(7596):661–664
Ostermaier MK, Schertler GF, Standfuss J (2014) Molecular mechanism of phosphorylation-dependent arrestin activation. Curr Opin Struct Biol 29:143–151
Pierce KL, Premont RT, Lefkowitz RJ (2002) Seven-transmembrane receptors. Nat Rev Mol Cell Biol 3(9):639–650
Pitcher JA, Freedman NJ, Lefkowitz RJ (1998) G protein-coupled receptor kinases. Annu Rev Biochem 67:653–692
Prihandoko R, Alvarez-Curto E, Hudson BD, Butcher AJ, Ulven T, Miller AM, Tobin AB, Milligan G (2016) Distinct phosphorylation clusters determine the signaling outcome of free fatty acid receptor 4/G protein-coupled receptor 120. Mol Pharmacol 89(5):505–520
Rahmeh R, Damian M, Cottet M, Orcel H, Mendre C, Durroux T, Sharma KS, Durand G, Pucci B, Trinquet E, Zwier JM, Deupi X, Bron P, Baneres JL, Mouillac B, Granier S (2012) Structural insights into biased G protein-coupled receptor signaling revealed by fluorescence spectroscopy. Proc Natl Acad Sci U S A 109(17):6733–6738
Rajagopal S, Rajagopal K, Lefkowitz RJ (2010) Teaching old receptors new tricks: biasing seven-transmembrane receptors. Nat Rev Drug Discov 9(5):373–386
Reiter E, Ahn S, Shukla AK, Lefkowitz RJ (2012) Molecular mechanism of beta-arrestin-biased agonism at seven-transmembrane receptors. Annu Rev Pharmacol Toxicol 52:179–197
Ren XR, Reiter E, Ahn S, Kim J, Chen W, Lefkowitz RJ (2005) Different G protein-coupled receptor kinases govern G protein and beta-arrestin-mediated signaling of V2 vasopressin receptor. Proc Natl Acad Sci U S A 102(5):1448–1453
Schmid EM, Ford MG, Burtey A, Praefcke GJ, Peak-Chew SY, Mills IG, Benmerah A, McMahon HT (2006) Role of the AP2 beta-appendage hub in recruiting partners for clathrin-coated vesicle assembly. PLoS Biol 4(9):e262
Shenoy SK, Lefkowitz RJ (2003) Multifaceted roles of beta-arrestins in the regulation of seven-membrane-spanning receptor trafficking and signalling. Biochem J 375(Pt 3):503–515
Shukla AK, Manglik A, Kruse AC, Xiao K, Reis RI, Tseng WC, Staus DP, Hilger D, Uysal S, Huang LY, Paduch M, Tripathi-Shukla P, Koide A, Koide S, Weis WI, Kossiakoff AA, Kobilka BK, Lefkowitz RJ (2013) Structure of active beta-arrestin-1 bound to a G-protein-coupled receptor phosphopeptide. Nature 497(7447):137–141
Shukla AK, Violin JD, Whalen EJ, Gesty-Palmer D, Shenoy SK, Lefkowitz RJ (2008) Distinct conformational changes in beta-arrestin report biased agonism at seven-transmembrane receptors. Proc Natl Acad Sci U S A 105(29):9988–9993
Shukla AK, Westfield GH, Xiao K, Reis RI, Huang LY, Tripathi-Shukla P, Qian J, Li S, Blanc A, Oleskie AN, Dosey AM, Su M, Liang CR, Gu LL, Shan JM, Chen X, Hanna R, Choi M, Yao XJ, Klink BU, Kahsai AW, Sidhu SS, Koide S, Penczek PA, Kossiakoff AA, Woods VL Jr, Kobilka BK, Skiniotis G, Lefkowitz RJ (2014) Visualization of arrestin recruitment by a G-protein-coupled receptor. Nature 512(7513):218–222
Smith JS, Rajagopal S (2016) The beta-arrestins: multifunctional regulators of G protein-coupled receptors. J Biol Chem 291(17):8969–8977
Sommer ME, Hofmann KP, Heck M (2012) Distinct loops in arrestin differentially regulate ligand binding within the GPCR opsin. Nat Commun 3:995
Song X, Coffa S, Fu H, Gurevich VV (2009) How does arrestin assemble MAPKs into a signaling complex? J Biol Chem 284(1):685–695
Song X, Gurevich EV, Gurevich VV (2007) Cone arrestin binding to JNK3 and Mdm2: conformational preference and localization of interaction sites. J Neurochem 103(3):1053–1062
Srivastava A, Gupta B, Gupta C, Shukla AK (2015) Emerging functional divergence of beta-arrestin isoforms in GPCR function. Trends Endocrinol Metab 26(11):628–642
Sutton RB, Vishnivetskiy SA, Robert J, Hanson SM, Raman D, Knox BE, Kono M, Navarro J, Gurevich VV (2005) Crystal structure of cone arrestin at 2.3 A: evolution of receptor specificity. J Mol Biol 354(5):1069–1080
Thomsen AR, Plouffe B, Cahill TJ 3rd, Shukla AK, Tarrasch JT, Dosey AM, Kahsai AW, Strachan RT, Pani B, Mahoney JP, Huang L, Breton B, Heydenreich FM, Sunahara RK, Skiniotis G, Bouvier M, Lefkowitz RJ (2016) GPCR-G protein-beta-arrestin super-complex mediates sustained G protein signaling. Cell
Tobin AB, Butcher AJ, Kong KC (2008) Location, location, location…site-specific GPCR phosphorylation offers a mechanism for cell-type-specific signalling. Trends Pharmacol Sci 29(8):413–420
Tran TM, Friedman J, Qunaibi E, Baameur F, Moore RH, Clark RB (2004) Characterization of agonist stimulation of cAMP-dependent protein kinase and G protein-coupled receptor kinase phosphorylation of the beta2-adrenergic receptor using phosphoserine-specific antibodies. Mol Pharmacol 65(1):196–206
Vishnivetskiy SA, Baameur F, Findley KR, Gurevich VV (2013) Critical role of the central 139-loop in stability and binding selectivity of arrestin-1. J Biol Chem 288(17):11741–11750
Vishnivetskiy SA, Paz CL, Schubert C, Hirsch JA, Sigler PB, Gurevich VV (1999) How does arrestin respond to the phosphorylated state of rhodopsin? J Biol Chem 274(17):11451–11454
Vishnivetskiy SA, Schubert C, Climaco GC, Gurevich YV, Velez MG, Gurevich VV (2000) An additional phosphate-binding element in arrestin molecule. Implications for the mechanism of arrestin activation. J Biol Chem 275(52):41049–41057
Walther C, Ferguson SS (2013) Arrestins: role in the desensitization, sequestration, and vesicular trafficking of G protein-coupled receptors. Prog Mol Biol Transl Sci 118:93–113
Wang HM, Dong JH, Li Q, Hu Q, Ning SL, Zheng W, Cui M, Chen TS, Xie X, Sun JP, Yu X (2014) A stress response pathway in mice upregulates somatostatin level and transcription in pancreatic delta cells through Gs and beta-arrestin 1. Diabetologia 57(9):1899–1910
Weller M, Virmaux N, Mandel P (1975) Light-stimulated phosphorylation of rhodopsin in the retina: the presence of a protein kinase that is specific for photobleached rhodopsin. Proc Natl Acad Sci U S A 72(1):381–385
Wilden U, Wust E, Weyand I, Kuhn H (1986) Rapid affinity purification of retinal arrestin (48 kDa protein) via its light-dependent binding to phosphorylated rhodopsin. FEBS Lett 207(2):292–295
Wisler JW, DeWire SM, Whalen EJ, Violin JD, Drake MT, Ahn S, Shenoy SK, Lefkowitz RJ (2007) A unique mechanism of beta-blocker action: carvedilol stimulates beta-arrestin signaling. Proc Natl Acad Sci U S A 104(42):16657–16662
Xiao K, Shenoy SK, Nobles K, Lefkowitz RJ (2004) Activation-dependent conformational changes in {beta}-arrestin 2. J Biol Chem 279(53):55744–55753
Xiao K, Sun J, Kim J, Rajagopal S, Zhai B, Villen J, Haas W, Kovacs JJ, Shukla AK, Hara MR, Hernandez M, Lachmann A, Zhao S, Lin Y, Cheng Y, Mizuno K, Ma’ayan A, Gygi SP, Lefkowitz RJ (2010) Global phosphorylation analysis of beta-arrestin-mediated signaling downstream of a seven transmembrane receptor (7TMR). Proc Natl Acad Sci U S A 107(34):15299–15304
Yang F, Yu X, Liu C, Qu CX, Gong Z, Liu HD, Li FH, Wang HM, He DF, Yi F, Song C, Tian CL, Xiao KH, Wang JY, Sun JP (2015) Phospho-selective mechanisms of arrestin conformations and functions revealed by unnatural amino acid incorporation and (19)F-NMR. Nat Commun 6:8202
Zhan X, Gimenez LE, Gurevich VV, Spiller BW (2011) Crystal structure of arrestin-3 reveals the basis of the difference in receptor binding between two non-visual subtypes. J Mol Biol 406(3):467–478
Zhuang T, Chen Q, Cho MK, Vishnivetskiy SA, Iverson TM, Gurevich VV, Sanders CR (2013) Involvement of distinct arrestin-1 elements in binding to different functional forms of rhodopsin. Proc Natl Acad Sci U S A 110(3):942–947
Zhuo Y, Vishnivetskiy SA, Zhan X, Gurevich VV, Klug CS (2014) Identification of receptor binding-induced conformational changes in non-visual arrestins. J Biol Chem 289(30):20991–21002
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing AG
About this chapter
Cite this chapter
Yang, Z. et al. (2017). The Functional Role of the Conformational Changes in Arrestin Upon Activation. In: Gurevich, V. (eds) The Structural Basis of Arrestin Functions. Springer, Cham. https://doi.org/10.1007/978-3-319-57553-7_16
Download citation
DOI: https://doi.org/10.1007/978-3-319-57553-7_16
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-57552-0
Online ISBN: 978-3-319-57553-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)