Abstract
Choline, the precursor of acetylcholine (ACh), is an essential amino acid that is associated with healthy brain development and neuroprotective functions. Choline supplementation can enhance ACh in the brain, which is important for learning and memory functions. Several studies investigated the cognitive enhancing effects of choline in animal models. However, reports on the effectiveness of choline supplementation in healthy humans vary considerably, with some studies showing positive effects, whereas others report no significant changes. In this chapter, we review the available cognitive and behavioral studies on choline, to clarify if and under which conditions choline supplementation might enhance cognition in heathy humans. A long-term diet rich in choline seems to enhance cognitive functioning across the life span. Short-term choline supplementation seems to have the potential to enhance various cognitive processes, especially in poor performers. Although more research is needed to fully understand the effects choline exerts on cognition, we conclude that choline is a promising tool for enhancing in particular memory functions in low-performing individuals.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
- Choline
- Acetylcholine
- Cognitive enhancer
- Cognition
- Memory
- Life span
- Amino acid supplement
- Individual differences
1 Introduction

The amino acid choline is a biochemical precursor of phospholipids and sphingomyelin, which are essential components of all membranes, especially necessary for the structural integrity and signaling functions of cell membranes (Howe et al. 2004; Zeisel et al. 1991). It is also a precursor of acetylcholine (ACh) (Zeisel et al. 1991). ACh is a major neurochemical of the central and peripheral nervous system involved in learning and memory processes (Hasselmo 2006). Choline supplementation increases choline blood plasma concentration and subsequently inter-neuronal choline brain concentration. This increase may accelerate ACh synthesis (Batool et al. 2016; Cohen and Wurtman 1975). Given that choline can enhance choline metabolites in the human brain (Babb et al. 2004), several studies investigated the effectiveness of choline supplementation as a tool to promote cognitive abilities, especially in the clinical domain (for reviews see Leermakers et al. 2015; Parnetti et al. 2007). Unfortunately, reports on the effects of choline supplementation on cognition in healthy humans are quite sparse and inconsistent. The aim of this chapter is to provide a summary of the available studies on the effects of choline supplementation on cognition in healthy humans in order to better understand under which circumstances choline has a positive effect and to clarify whether choline can act as a useful cognitive enhancer
.
In this chapter, we will first describe how choline supplementation affects ACh function. Second, we will outline the available studies investigating a long-term diet rich in choline across the life span. Third, we will review studies exploring the short-term effect of choline supplementation on various cognitive processes. The studies indicate that choline has the potential to enhance in particular memory processes in low-performing individuals, suggesting an inverted u-shaped pattern of effectivity.
2 Mechanism of Action
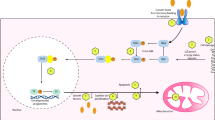
Acetylcholine is synthesized from choline and acetyl coenzyme A in a reaction that is catalyzed by the enzyme choline acetyltransferase (Wu and Hersh 1994; Zeisel and Blusztajn 1994). The administration of choline can cause sequential increases in choline and ACh concentrations in rats. Choline uptake peaks approximately 30 min after ingestion and ACh levels in the brain significantly rise after about 40 min and remain high for around 90 min (Cohen and Wurtman 1975; Haubrich et al. 1975). This increase in ACh concentration is dose dependent between 15 and 60 mg/kg. Higher doses of choline do not lead to higher increments in ACh concentration (Cohen and Wurtman 1975). These outcomes indicate that precursor availability may affect brain ACh synthesis similar to how brain tyrosine and tryptophan levels have been previously found to regulate the syntheses of brain dopamine and serotonin (see Chapters “Tyrosine” and “Tryptophan” on tyrosine and tryptophan). Hence, an increase in plasma choline is not a sufficient condition for increased ACh synthesis. It seems that increased plasma choline enhances cholinergic function to the extent that cholinergic neurons are activated simultaneously with choline treatment or as a consequence of ACh depletion (Jope 1982; Trommer et al. 1982). After ingestion, choline is transported across the blood–brain barrier by a bidirectional and unsaturated carrier mechanism (Cornford et al. 1978; Lockman and Allen 2002). This mechanism transports choline at a rate proportional to serum choline concentration. That is, when the blood choline concentration increases so does the brain choline concentration (Lockman and Allen 2002). Effects of choline on ACh synthesis mainly occur because of the enzyme choline acetyltransferase, which is involved in the first step of the synthesis of choline into ACh and responsible for regulating at which rate choline is converted into ACh (Ueland 2011), see Fig. 1. The activity of the enzyme choline acetyltransferase is affected by several factors like precursor availability, feedback inhibition by its end-products, and the individual variability in the activity of high-affinity uptake of choline (Zeisel 1981).
Schematic representation of the effect of acute choline supplementation on choline to acetylcholine conversion . Once it has passed the blood–brain barrier, the acetyl coenzyme A (AcCoA) and the enzyme choline acetyltransferase convert choline into acetylcholine leading to an elevation in acetylcholine level. In another series of reactions choline, via several steps, is synthesized to phosphatidylcholine, a phospholipid that incorporates choline as a headgroup
Given that choline is an essential amino acid, the body cannot synthesize it in a sufficient amount by itself (Zeisel and Blusztajn 1994). Accordingly, it is crucial to ingest food that contains a certain amount of choline (Zeisel 1981). The Institute of Medicine and National Academy of Sciences USA recommended an adequate intake for choline of 550 mg/day for men and 425 mg/day for women, with a higher recommended intake during pregnancy and lactation (Yates et al. 1998).Choline and esters of choline can be ingested through protein-rich food. The highest total choline concentrations (mg/100 g) are contained in beef liver (418 mg), chicken liver (290 mg), and eggs (251 mg). But also fish, milk, as well as several plant foods like certain beans or nuts are a good source of choline (Howe et al. 2004; Zeisel et al. 2003). The established recommended upper intake level for adults is 3.5 g (Food and nutrition board of the US—Institute of Medicine). The ingestion of high choline doses (e.g., >5 g/day) can produce side effects such as sweating, nausea, vomiting, bloating, and diarrhea.
3 Choline at Long-Term: Enhancement Across the Life Span
Across the life span a diet rich in choline seems to exert neuroprotective functions and to enhance cognition. In this section, we will give an overview about recent findings involving long-term studies on this topic. Several animal studies suggest that a diet rich in choline leads to positive outcomes in the offspring. For instance, the offspring of rat dams showed better memory function when they were exposed to higher levels of choline in utero (Williams et al. 1998). Boeke et al. (2013) demonstrated that choline intake during pregnancy also affects visuospatial memory in humans. In this study the researchers assessed the amount of choline intake of 1896 pregnant women with high socioeconomic backgrounds by means of the 130-item semi-quantitative food frequency questionnaire as well as a 33-item detailed interview about the usage of nutritional supplements. At age 7, children were approached and asked to perform cognitive tests to assess visuospatialmemory (Wide Range Assessment of Memory and Learning, Design and Picture Memory subtests; WRAML2), and verbal and nonverbal intelligence (Kaufman Brief Intelligence Test, Second Edition; KBIT-2). Results showed that a diet rich in choline during the second trimester, and to a smaller extent also in the first trimester, was linked to better visuospatial memory at age 7, as measured by the WRAML2 Design and Picture Memory subtests. Moreover, a positive correlation between second-trimester choline intake and nonverbal intelligence has been found. The researchers suggested that the beneficial effects of maternal gestational choline ingestion on the children were due to enhanced brain function via the modulation of cholinergic transmission or epigenetics changes in the myelination of neurons. On the other hand, another study could not replicate this outcome. A randomized, double-blind, placebo-controlled trial did not show an enhancement of infant cognitive functions when pregnant women, who were consuming moderate-choline diets, were supplemented with phosphatidylcholine (a class of phospholipids that incorporate choline as a headgroup) (Cheatham et al. 2012). The discrepancy in the results of these two studies may be due to the different samples in terms of socioeconomic status of women investigated. It may be that the lower socioeconomic status and associated unfavorable environmental conditions overshadowed the efficacy of choline supplementation. This idea would also be in line with another study in which participants from a disadvantaged background (Signore et al. 2008) showed no association between serum choline and child IQ score. Unfortunately, it is plausible to assume that a diet rich in choline cannot compensate for unfavorable environmental factors. However, a study by Jiang et al. (2012) underlines the beneficial effect of ingesting choline during pregnancy. Twenty-four women in the third trimester of pregnancy either consumed 480 or 930 mg choline per day. Researchers collected maternal and placental blood samples as well as samples of placental tissue. Women who were administered more choline, had lower levels of cortisol in the placental cord and changes in cortisol-regulatinggenes in both the placental and fetal tissue. The authors concluded that choline supplementation during pregnancy might counteract the adverse effects of prenatal stress. This finding may be particularly relevant for women who experience considerable amount of stress during their pregnancy and might want to enhance the neuroendocrine and metabolic development of their offspring.
The intake ofcholine seems not only to be beneficial in prenatal and postnatal cognitive enhancement in young children , but also to compensate for cognitive decline in healthy elderly. Given that during the course of normal aging a reduction in phospholipid phosphatidylcholine takes place (Roth et al. 1995; Rouser and Yamamoto 1968), Babb et al. (2002) investigated with a proton decoupled P-MRS technique whether chronic citicoline (intermediate product of phosphatidylcholine) intake could increase phospholipid phosphatidylcholine and further enhance cognition. Elderly volunteers were scanned and neurologically assessed before and after taking 500 mg citicoline every day over the course of 6 weeks. In addition to the measurable increases in phospholipid phosphatidylcholine , a correlation between this increase and improved verbal memory was established. The authors conclude that choline supplementation might have neuroprotective properties in aging adults, preserving learning, and memory abilities (Babb et al. 2002). Consistent with the aforementioned result, Spiers et al. (1996) explored in a randomized, double-blind, placebo-controlled study the effects of citicoline on verbal memory in elderly mentally healthy adults. The volunteers received a placebo or citicoline, 1000 mg/day, for 3 months. For the crossover study, the low-performing individuals were tested again. This time the subjects took both placebo and citicoline, 2000 mg/day, each for 2 months. It was found that citicoline therapy promoted delayed recall on logical memory only for the subjects with relatively low-functioning memories. The higher dosage of citicoline was linked to improved immediate and delayed logical memory. Hence the authors suggested that citicoline therapy improves verbal memory functioning in healthy older individuals with relatively low-functioning memory. Furthermore, Poly et al. (2011) conducted in a community-based population of 1391 healthy, nondemented individuals between 36 and 83 years of age, a longitudinal study to explore the association between choline intake and cognitive outcome measures. At two time points (1991–1995 and 1998–2001), daily choline intake was measured by means of the Harvard Food Frequency Questionnaire . At the second test point, structural imaging measures and four neuropsychologicalfactors were assessed in addition to the food questionnaire: verbal memory , visual memory , verbal learning , and executive function . Higher concurrent choline intake was associated with better verbal memory and visual memory performance, whereas higher remote choline intake, on the other hand, was linked to smaller white matter hyperintensity. The fact that choline intake is related to white matter hyperintensity suggests that choline intake at midlife may be neuroprotective. Likewise, in an extensive cross-sectional study by Nurk et al. (2013) the association between plasma free choline, betaine, and cognitive functioning in a sample of 2195 healthy elderly participants was examined. A low plasma concentration of free choline was associated with poor performance in global cognition, sensorimotor speed, perceptual speed, and executive function (Nurk et al. 2013). However, Eussen et al. (2007) did not find an association of plasma choline with cognitive function and argued that plasma choline might be a poor marker for the effects choline exerts on the brain (Eussen et al. 2007).
In sum, the long-term intake of choline seems to show promising potential to enhance certain memory functions across the life span: to achieve prenatal and postnatal cognitive enhancement in young children and to compensate for cognitive decline in aging.
4 Choline at Short-Term: Enhancement in Healthy Adults
In this section, the few studies investigating the effect of short-term choline supplementation on several cognitive functions will be reviewed.
In a placebo-controlled study, Naber et al. (2015) reported improved visuomotor performance in healthy adults after the ingestion of 3 g choline bitartrate administered 70–90 min before testing. The authors suggested that choline administration is able to shift the speed-accuracy trade-off of actions toward accuracy, and thus leads to an improved spatial coordination. Moreover, a decrement in pupil size was found, indicating possible changes in the autonomous nervous system induced by the cholinergic modulation of attention (Naber et al. 2015). Further, in a study by Deuster (2002), cognitive and physical performance was assessed after participants received either two doses of 25 mg/kg choline supplementation or a placebo 30 min before and 60 min after an extensive load carrying exercise task. Whereas Naber et al. (2015) found changes in spatial coordination and attention, an increase in plasma choline did not lead to changes in physical performance nor in reaction time, logical reasoning, vigilance, spatial memory, or working memory (Deuster 2002). The discrepancy between the two studies in physical performance may be explained by the fact that choline supplementation is more likely to affect fine-motor control rather than ballistic movements (Nag and Berger-Sweeney 2007). The null-findings in the cognitive domain by Deuster (2002) may be due by failing to take into account individual differences (i.e., low vs. high performers). Notably, in another study enhancing effects on processing speed, working memory, verbal learning, verbal memory, and executive function were only observable in low-performing individuals (Knott et al. 2015a). In a randomized, double-blind, crossover design by Knott et al. (2015a) participants were given a placebo, a single low (500 mg) or moderate (1000 mg) dose of CDP-choline. Four hours after ingestion they were asked to complete the CogState Schizophrenia Battery, used to detect cognitive changes (Falleti et al. 2006), in each of the three test sessions. CDP-choline supplementation was found to enhance cognitive performance of the low achieving individuals but impair performance in moderately efficient and high cognitive task performers. Likewise, in a double-blind counterbalanced study by Ladd et al. (1993) a trend to greater enhancement in slow learners was observed. Healthy subjects, after having received either a dose of 25, 10 g phosphatidylcholine or a placebo, had to complete a serial learning task 60 or 90 min after ingestion. After intake of 25 g phosphatidylcholine a significant enhancement of explicit memory after 90 min and a moderate enhancement after 60 min was found, but not for remote, semantic, or implicit memory. Along the same line, Sitaram et al. (1978) showed that the supplementation of choline significantly enhanced serial learning in healthy adults and that “poor” performers were more receptive to the enhancing effects after the administration. This series of outcomes suggest that compared to high, low-performing individuals might profit the most from choline supplementation. An explanation for this pattern of results might be that choline supplementation follows an inverted u-shaped pattern of effectivity (Bentley et al. 2011). That is, for high performing individuals who already possess an optimal cholinergic state, choline supplementation might lead to a hyper-cholinergic supraoptimal state, decreasing cognitive performance. In contrast, for low performing individuals who instead possess a suboptimal cholinergic state, choline supplementation, by filling the cholinergic supply, might lead to improved cholinergic stimulation and enhanced cognitive performance. Accordingly, choline may exert protective and enhancing properties only in individuals associated to a low cholinergic baseline levels (Knott et al. 2015a).
All in all, these results suggest that short-term choline supplementation can enhance cognition in healthy adults. However, the positive effects are restricted to certain cognitive domains, like visual and verbal episodic memory and visuomotor abilities. Furthermore, individuals with low choline baseline levels in the brain are likely to profit the most from the supplementation.
5 Conclusion
As the biochemical precursor of ACh , choline has the potential to enhance cholinergic metabolism in the brain. Although more research is needed to explain contradictory results and to fully understand the effects choline exerts on cognition in healthy humans, a long-term diet rich in choline seems to enhance cognitive functioning across the life span. Besides leading to prenatal and postnatal cognitive enhancement in young child , choline might exert neuroprotective functions in normal aging given that a diet rich in choline was associated with lower white matter hyperintensities, which is associated with decreased risk of stroke, dementia, and premature death (Poly et al. 2011).
At short-term, choline supplementation seems to have the potential to enhance various cognitive processes (in particular memory), especially in poor performers. Low performing individuals might profit the most from choline supplementation, because of an inverted u-shaped pattern of choline effectivity on cognitive performance. Accordingly, future studies investigating the effect of choline on cognition should mandatorily take into account individual difference in performance (low vs. high performers) and genetic markers associated with ACh availability in the brain. Indeed, genetic carriers associated with low ACh baseline levels are the ones expected to profit the most from the choline intervention. Hence, we suggest that genetic predisposition might modulate the effect ofcholine in its role as cognitive enhancer.
Future studies need to shed light on the neurological underpinnings underlying the cognitive enhancement effects induced by choline. Notably, Knott et al. (2015b) showed dose-dependent effects of choline supplementation on resting state brain oscillations. Spectral analysis exhibited dose-dependent declines in delta and increments in alpha oscillations , which were also accompanied by decrements in beta and gamma oscillatory activity. As pointed out by the authors, these outcomes sustain the idea that choline-induced cognitive enhancement implicates multiple mechanisms including facilitated nicotinic cholinergic action.
In sum, even if more research is necessary, we can conclude that choline is a promising tool for enhancing in particular memory functions in low-performing individuals and might exert neuroprotective functions across the life span.
References
Babb, S. M., Ke, Y., Lange, N., Kaufman, M. J., Renshaw, P. F., & Cohen, B. M. (2004). Oral choline increases choline metabolites in human brain. Psychiatry Research Neuroimaging, 130(1), 1–9.
Babb, S., Wald, L., Cohen, B., Villafuerte, R., Gruber, S., Yurgelun-Todd, D., et al. (2002). Chronic citicoline increases phosphodiesters in the brains of healthy older subjects: An in vivo phosphorus magnetic resonance spectroscopy study. Psychopharmacology (Berl), 161(3), 248–254.
Batool, Z., Sadir, S., Liaquat, L., Tabassum, S., Madiha, S., Rafiq, S., … Naqvi, F. (2016). Repeated administration of almonds increases brain acetylcholine levels and enhances memory function in healthy rats while attenuates memory deficits in animal model of amnesia. Brain Research Bulletin, 120, 63–74.
Bentley, P., Driver, J., & Dolan, R. J. (2011). Cholinergic modulation of cognition: Insights from human pharmacological functional neuroimaging. Progress in Neurobiology, 94(4), 360–388.
Boeke, C. E., Gillman, M. W., Hughes, M. D., Rifas-Shiman, S. L., Villamor, E., & Oken, E. (2013). Choline intake during pregnancy and child cognition at age 7 years. American Journal of Epidemiology, 177, 1338–1347.
Cheatham, C. L., Goldman, B. D., Fischer, L. M., da Costa, K. A., Reznick, J. S., & Zeisel, S. H. (2012). Phosphatidylcholine supplementation in pregnant women consuming moderate choline diets does not enhance infant cognitive function: A randomized, double-blind, placebo-controlled trial. The American Journal of Clinical Nutrition, 96(6), 1465–1472.
Cohen, E. L., & Wurtman, R. J. (1975). Brain acetylcholine: Increase after systematic choline administration. Life Sciences, 16(7), 1095–1102.
Cornford, E. M., Braun, L. D., & Oldendorf, W. H. (1978). Carrier mediated blood–brain barrier transport of choline and certain choline analogs. Journal of Neurochemistry, 30(2), 299–308.
Deuster, P. A. (2002). Choline ingestion does not modify physical or cognitive performance. Military Medicine, 167(12), 1020.
Eussen, S. J., Ueland, P. M., Clarke, R., Blom, H. J., Hoefnagels, W. H., Van Staveren, W. A., et al. (2007). The association of betaine, homocysteine and related metabolites with cognitive function in Dutch elderly people. British Journal of Nutrition, 98(05), 960–968.
Falleti, M. G., Maruff, P., Collie, A., & Darby, D. G. (2006). Practice effects associated with the repeated assessment of cognitive function using the CogState battery at 10-minute, one week and one month test-retest intervals. Journal of Clinical and Experimental Neuropsychology, 28(7), 1095–1112.
Hasselmo, M. E. (2006). The role of acetylcholine in learning and memory. Current Opinion in Neurobiology, 16(6), 710–715.
Haubrich, D. R., Wang, P. F., Clody, D. E., & Wedeking, P. W. (1975). Increase in rat brain acetylcholine induced by choline or deanol. Life Science, 17, 975–980.
Howe, J. C., Williams, J. R., Holden, J. M., Zeisel, S. H., & Mar, M. (2004). USDA database for the choline content of common foods. US Department of Agriculture, Agricultural Research Service, Beltsville Human Nutrition Research Center, Nutrient Data Laboratory.
Jiang, X., Yan, J., West, A. A., Perry, C. A., Malysheva, O. V., Devapatla, S., … Caudill, M. A. (2012). Maternal choline intake alters the epigenetic state of fetal cortisol-regulating genes in humans. The FASEB Journal, 26(8), 3563–3574.
Jope, R. S. (1982). Effects of phosphatidylcholine administration to rats on choline in blood and choline and acetylcholine in brain. Journal of Pharmacology and Experimental Therapeutics, 220, 322–328.
Knott, V., de la Salle, S., Choueiry, J., Impey, D., Smith, D., Smith, M., et al. (2015a). Neurocognitive effects of acute choline supplementation in low, medium and high performer healthy volunteers. Pharmacology Biochemistry and Behavior, 131, 119–129.
Knott, V., de la Salle, S., Smith, D., Choueiry, J., Impey, D., Smith, M., … Labelle, A. (2015b). Effects of acute CDP-choline treatment on resting state brain oscillations in healthy volunteers. Neuroscience Letters, 591, 121–125.
Ladd, S. L., Sommer, S. A., LaBerge, S., & Toscano, W. (1993). Brief report: Effect of phosphatidylcholine on explicit memory. Clinical Neuropharmacology, 16(6), 540–549.
Leermakers, E. T., Moreira, E. M., Kiefte-de Jong, J. C., Darweesh, S. K., Visser, T., Voortman, T., … Franco, O. H. (2015). Effects of choline on health across the life course: A systematic review. Nutrition Reviews, 73(8), 500–522.
Lockman, P., & Allen, D. (2002). The transport of choline. Drug Development and Industrial Pharmacy, 28(7), 749–771.
Naber, M., Hommel, B., & Colzato, L. S. (2015). Improved human visuomotor performance and pupil constriction after choline supplementation in a placebo-controlled double-blind study. Scientific Reports, 5, 13188.
Nag, N., & Berger-Sweeney, J. E. (2007). Postnatal dietary choline supplementation alters behavior in a mouse model of Rett syndrome. Neurobiology of Disease, 26(2), 473–480.
Nurk, E., Refsum, H., Bjelland, I., Drevon, C. A., Tell, G. S., Ueland, P. M., … Smith, D. A. (2013). Plasma free choline, betaine and cognitive performance: The Hordaland health study. British Journal of Nutrition, 109(03), 511–519.
Parnetti, L., Mignini, F., Tomassoni, D., Traini, E., & Amenta, F. (2007). Cholinergic precursors in the treatment of cognitive impairment of vascular origin: Ineffective approaches or need for re-evaluation? Journal of the Neurological Sciences, 257(1), 264–269.
Poly, C., Massaro, J. M., Seshadri, S., Wolf, P. A., Cho, E., Krall, E., … Au, R. (2011). The relation of dietary choline to cognitive performance and white-matter hyperintensity in the framingham offspring cohort. The American Journal of Clinical Nutrition, 94(6), 1584–1591.
Roth, G. S., Joseph, J. A., & Mason, R. P. (1995). Membrane alterations as causes of impaired signal transduction in Alzheimer’s disease and aging. Trends in Neurosciences, 18(5), 203–206.
Rouser, G., & Yamamoto, A. (1968). Curvilinear regression course of human brain lipid composition changes with age. Lipids, 3(3), 284–287.
Signore, C., Ueland, P. M., Troendle, J., & Mills, J. L. (2008). Choline concentrations in human maternal and cord blood and intelligence at 5 y of age. The American journal of clinical nutrition, 87(4), 896–902.
Sitaram, N. (1978). Human serial learning: Enhancement with arecholine and choline and impairment with scopolamine. Science, 201, 1978.
Spiers, P. A., Myers, D., Hochanadel, G. S., Lieberman, H. R., & Wurtman, R. J. (1996). Citicoline improves verbal memory in aging. Archives of Neurology, 53(5), 441–448.
Trommer, B. A., Schmidt, D. E., & Wecker, L. (1982). Exogenous choline enhances the synthesis of acetylcholine only under conditions of increased cholinergic neuronal activity. Journal of Neurochemistry, 39, 1704–1709.
Ueland, P. M. (2011). Choline and betaine in health and disease. Journal of Inherited Metabolic Disease, 34(1), 3–15.
Williams, C. L., Meck, W. H., Heyer, D. D., & Loy, R. (1998). Hypertrophy of basal forebrain neurons and enhanced visuospatial memory in perinatally choline-supplemented rats. Brain Research, 794(2), 225–238.
Wu, D., & Hersh, L. B. (1994). Choline acetyltransferase: Celebrating its fiftieth year. Journal of Neurochemistry, 62, 1653–1663.
Yates, A. A., Schlicker, S. A., & Suitor, C. W. (1998). Dietary reference intakes: The new basis for recommendations for calcium and related nutrients, B vitamins, and choline. Journal of the American Dietetic Association, 98(6), 699–706.
Zeisel, S. H. (1981). Dietary choline: Biochemistry, physiology, and pharmacology. Annual Review of Nutrition, 1(1), 95–121.
Zeisel, S. H., & Blusztajn, J. K. (1994). Choline and human nutrition. Annual Review of Nutrition, 14(1), 269–296.
Zeisel, S. H., Da Costa, K. A., Franklin, P. D., Alexander, E. A., Lamont, J. T., Sheard, N. F., et al. (1991). Choline, an essential nutrient for humans. FASEB Journal: Official Publication of the Federation of American Societies for Experimental Biology, 5(7), 2093–2098.
Zeisel, S. H., Mar, M. H., Howe, J. C., & Holden, J. M. (2003). Concentrations of choline-containing compounds and betaine in common foods. The Journal of Nutrition, 133(5), 1302–1307.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Copyright information
© 2017 Springer International Publishing AG
About this chapter
Cite this chapter
Colzato, L.S., Schlauch, K., Kullmann, J.K., Wild, T. (2017). Choline. In: Theory-Driven Approaches to Cognitive Enhancement. Springer, Cham. https://doi.org/10.1007/978-3-319-57505-6_3
Download citation
DOI: https://doi.org/10.1007/978-3-319-57505-6_3
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-57504-9
Online ISBN: 978-3-319-57505-6
eBook Packages: Behavioral Science and PsychologyBehavioral Science and Psychology (R0)