Abstract
Craniopharyngiomas are rare, histologically benign tumors that arise in the suprasellar space. As noted by Harvey Cushing as early as 1932, these lesions while benign in nature are frequently adherent to adjacent critical structures, which makes complete surgical resection challenging. Because a microscopic gross total resection is rarely obtained, adjuvant radiotherapy plays an important role in the management of these lesions. However, given the proximity of critical normal tissues adjacent to or even within target volumes, radiation therapy may be associated with adverse effects. Both surgical- and radiation-induced adverse effects are compounded by the frequent presence of morbidities arising from the tumor itself. While morbidities are frequent, patients with craniopharyngioma have excellent survival outcomes. Thus, practitioners of all specialties must take great care in treatment planning and survivorship in order to optimize post-treatment outcomes.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
1 Epidemiology
Craniopharyngiomas are uncommon, suprasellar, histologically benign tumors that have an incidence of about 1.3 per million person years without a clear preponderance based on gender or race (Bunin et al. 1998). In the United States, craniopharyngiomas constitute about 1–3% of all brain tumors with an estimated 350 new cases per year (Bunin et al. 1998; Jane and Laws 2006). However, in other parts of the world, craniopharyngiomas may be more common, with the relative incidence reportedly 11.6% in Africa compared to 3.9% in Japan (Izuora et al. 1989; Stiller and Nectoux 1994).
There is a bimodal age distribution for patients with craniopharyngioma. The first peak incidence is in children between 5 and 14 years with craniopharyngiomas representing about 4–6% of all pediatric brain tumors. The second peak is among older adults aged 65–74 years, which represents 1–4% of adult brain tumors (Bunin et al. 1998; Samii and Tatagiba 1997; Moore 2000). Histologic type also differs by age with adamantinomatous craniopharyngiomas most commonly found in the pediatric patients and papillary-squamous in adults (Muller 2014).
2 Predisposing Factors: Etiology and Genetics
Currently, there are no known genetic or environmental risk factors that predispose to the development of craniopharyngiomas. Rather, it is believed craniopharyngiomas arise spontaneously from the squamous-cell remnants of Rathke’s pouch, along a line from the nasopharynx to the diencephalon (path of the primitive craniopharyngeal duct and adenohypophysis) (Sughrue et al. 2010; Petito et al. 1976).
As mentioned, there are two histologic subtypes of craniopharyngioma. Adamantinomatous is the most frequent and is commonly cystic with pockets of dark brown, “crank-case oil-like” fluid. Histologically, this subtype has epithelial lobules and palisading epithelium, which resemble tooth-forming tissues. The cyst fluid is rich in cholesterol that accumulates from membrane lipids, as well as keratin that comes from the top layer of desquamated keratinized squamous cells, which can often calcify. The second type of craniopharyngioma is the papillary-squamous subtype that exhibits well-differentiated, non-keratinizing squamous epithelium with papillary projections; these rarely calcify (Muller 2014; Sughrue et al. 2010). A metaplastic origin to the papillary subtype from the adenohypophyseal cells has been suggested but remains controversial (Sughrue et al. 2010).
Newer data suggests the two histologic subtypes have different molecular aberrations. Activation of the WNT signaling pathway appears to drive the development of adamantinomatous craniopharyngiomas. Buslei and colleagues showed that this subtype contains mutations in CTNNB1, the gene that encodes β-catenin (Buslei et al. 2005). In contrast, the papillary subtype contains mutations in the BRAF oncogene (Brastianos et al. 2014; Sekine et al. 2002; Marucci et al. 2015). Targeted genotyping has identified CTNNB1 mutations in nearly all adamantinomatous (96%) craniopharyngiomas and BRAF mutations in nearly all papillary subtypes (95%) (Brastianos et al. 2014). Such insights will hopefully lead to the development of new therapeutic approaches in coming years.
3 Presenting Symptoms
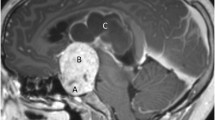
Craniopharyngiomas are slow growing tumors that can reach rather large sizes before diagnosis. Retrospectively, symptoms attributable to the tumor are often apparent at least months, if not years, prior to diagnosis (Muller 2008, 2013). Patients can experience a wide range of symptoms that are largely dependent upon the exact location of the tumor and subsequent compression of nearby critical structures including the optic chiasm, optic nerves or tracts, pituitary stalk, or hypothalamus. However, the initial clinical symptoms may also be commonly related to nonspecific signs of intracranial pressure, including headaches (Fig. 16.1). Moderate to severe daily headaches are found in approximately 50% of patients at presentation, with etiologies related to obstructive hydrocephalus, meningeal irritation, or traction on pain-sensitive structures (Khan et al. 2013).
This example depicts a 16-year-old female who presented with headaches, visual changes, and amenorrhea. On MRI (a), she was found to have a suprasellar mass with radiologic changes consistent with craniopharyngioma. She underwent a subtotal resection as confirmed with postoperative imaging (b). She then received postoperative proton beam radiation therapy to a dose of 50.4 cGE in 28 fractions using a 3-field passive scatter technique (c, d)
Visual symptoms or endocrine abnormalities are more specifically attributable to suprasellar tumors, like craniopharyngiomas. The presentation and severity of symptoms range based on the tumor location. For instance, pressure on the optic chiasm may result in temporal quandrantanopias. However, a variety of visual symptoms are possible based on the particular growth pattern of the tumor and include: diplopia, blurred vision, decreased acuity, or in severe cases sometimes unilateral blindness. Visual symptoms tend to be the presenting sign more common in adults (~80%) than children (~20–60%) (Sughrue et al. 2010; Muller 2013).
Endocrine deficiencies are also common due to disruption of the hypothalamic-pituitary axis. At diagnosis, up to almost 90% of patients present with at least one hormone deficit, most commonly growth hormone (GH) (75%), followed by gonadotropins (40%), adrenocorticotropic hormone (ACTH) (25%), and thyroid-stimulating hormone (TSH) (25%) (Muller 2008; Caldarelli et al. 2005; Hoffman et al. 1992). Each hormone deficit has its own associated symptoms. For instance, GH deficiencies result in growth retardation and delayed bone age. Low gonadotropin levels interfere with pubertal changes, which may be more clinically apparent in adolescents. Low TSH levels result in hypothyroidism leading to fatigue, cold intolerance and weight gain (Rath et al. 2013; Rose et al. 1999).
Overall, there is a relatively specific constellation of symptoms in patients with suprasellar tumors. Therefore, patients presenting with a combination of headaches, visual impairment, and signs of endocrine abnormalities (decreased growth rate, short stature, or polydipsia/polyuria) should be evaluated for a craniopharyngioma (Muller 2010).
4 Radiographic Findings
While plain radiographs are rarely used for diagnosis of this disease, several classic findings can be seen on these films. First, most patients have sellar abnormalities, including enlargement (~65%) or erosion (~44%) (Moore 2000). Second, tumor-associated calcifications can also be seen on plain radiographs, which are more common in children (up to 90%) due to the preponderance of the adamantinomatous histology (Warmuth-Metz et al. 2004).
A computed tomography (CT) scan is commonly the initial imaging obtained for workup of patients with the previously discussed symptomatology. On CT, the tumors are characteristically described as suprasellar and lobular with a central solid component surrounded by multiple, various sized, hypodense cysts. Calcifications are also best demonstrated by CT (Warmuth-Metz et al. 2004). Additional imaging is typically performed using magnetic resonance imaging (MRI); this is particularly useful for providing detailed information related to the anatomic relationship of the tumor to nearby structures. On T1-weighted images, the solid tumor component (appears isointense without contrast) and cyst rims typically enhance with contrast and the cyst fluid may appear hypo-intense. Conversely, on the T2-sequence, the tumor will typically appear hyperintense.
5 Workup
Initial assessment for craniopharyngiomas usually consists of obtaining adequate neuroimaging with CT brain and subsequent MRI, as discussed above. Features such as calcifications and cystic regions often help to narrow the differential diagnosis, making other lesions such as germ cell tumors less likely. Depending on the acuity and need for neurosurgical intervention to manage hydrocephalus, additional workup is warranted.
Ideally, patients with tumors that are suspicious for craniopharyngioma should receive evaluation and management by a multidisciplinary team with subspecialists from neurosurgery, radiation oncology, medical oncology, endocrinology, ophthalmology, and neuropsychology. A baseline visual acuity exam is useful in determining where there is optic pathway compression and for establishing baseline function. Additionally, since most patients have at minimum partial hypopituitarism, a complete endocrine assessment is important, especially in order to identify adrenal or thyroid dysfunction prior to surgery. Finally, despite their relatively unique appearance, imaging alone does not suffice to establish the diagnosis of craniopharyngioma, and histologic confirmation in required (Moore 2000; Sughrue et al. 2010; Muller 2010).
6 Treatment
The optimal therapeutic strategy for craniopharyngiomas remains controversial. The two basic approaches include either an aggressive surgical resection that attempts a gross total resection (GTR) versus a more conservative surgery followed by radiation therapy to treat residual disease. Unfortunately, the published literature and lack of randomized data have not been able to resolve the debate. Notably, biases against either strategy are based primarily on morbidities associated with historical cohorts. For instance, significant advances in neurosurgical techniques have decreased the morbidity of resections and perhaps more aggressive approaches are warranted. Similarly, radiation therapy techniques have evolved rapidly allowing for more accurate, conformal dose deposition while minimizing radiation to nearby critical structures. Therefore, it is imperative that an experienced multidisciplinary team evaluate patients with craniopharyngiomas tumors for personalized and optimal treatment planning.
6.1 Surgery
Surgery is indicated in all patients in order to achieve a histologic diagnosis, allow for cyst or hydrocephalus decompression, and to minimize disease burden (Van Effenterre and Boch 2002). Some surgeons believe that an aggressive GTR is required for cure, while others believe the morbidity associated with that approach is too great and opt for cyst drainage and a subtotal resection (STR) with planned postoperative radiation therapy (Aggarwal et al. 2013; Fahlbusch et al. 1999; Merchant et al. 2002; Sanford 1994; Stripp et al. 2004; Weiner et al. 1994; Yasargil et al. 1990; Clark et al. 2013; Schoenfeld et al. 2012).
Initial surgical intervention is focused on relieving acute symptoms related to these tumors. In patients presenting with hydrocephalus, decompression of the lesion itself is the favored treatment approach to restore CSF flow (Fahlbusch et al. 1999; Choux and Lena 1979). However in severe cases, an external drain may be required to relieve pressure prior to tumor resection.
The approach to surgical resection is dependent on the location and makeup of the tumor. Historically, a common surgical approach included a right frontotemporal incision (Fahlbusch et al. 1999). Also, suprasellar tumors were also resected using a transcranial approach, while prechiasmatic tumors may be best visualized via a supraorbital craniotomy (Moore 2000). For tumors that are primarily intra-sellar, an endoscopic, transsphenoidal approach may provide optimal visualization while remaining minimally invasive (Elliott et al. 2011; Zona and Spaziante 2006). More commonly, newer techniques including microsurgery, endoscopic assistance, and minimally invasive approaches have allowed neurosurgeons to improve the quality of their resections while minimizing morbidity (Sughrue et al. 2010; Muller 2008, 2013). Ultimately, the intraoperative findings will dictate the degree of resection.
6.2 Radiation Therapy
Factors, which most impact the choice of whether to proceed with adjuvant radiotherapy or observation, are most often the presence of residual disease and the age of the patient. For tumors arising in very young patients, a discussion is warranted regarding the appropriateness of observation since very young children are particularly susceptible to adverse radiation effects. However, because incompletely resected lesions have a high rate of local relapse without adjuvant therapy (71–90%), postoperative radiotherapy should be recommended in patients with subtotal resection (Clark et al. 2013; Becker et al. 1999).
Numerous radiation therapy techniques exist and have been utilized with excellent outcomes in terms of disease control for these tumors. It is, however, worth noting that no prospective studies evaluating the benefits of advanced technology exist, in terms of reduced radiation adverse effects, either for photon or proton therapy. In the majority of published literature, photon-based therapies, both traditionally fractionated and hypo-fractionated stereotactic techniques have been employed (Merchant et al. 2002, 2006; Becker et al. 1999; Habrand et al. 1999, 2006; Minniti et al. 2009). Merchant and colleagues conducted a single arm prospective study of reduced margin 3D conformal radiation therapy for pediatric patients with craniopharyngioma (Merchant et al. 2006). In a total of 28 patients, the solid and cystic tumor components along with a 1 cm expansion were targeted to a dose between 54 and 55.8 Gy with acceptable rates of disease control. Greenfield and colleagues have also retrospectively evaluated the use of intensity modulated radiation therapy techniques (Greenfield et al. 2015). These investigators documented high rates of disease control but also noted a high burden of pre-radiotherapy comorbidities including endocrinopathies. More recently, dosimetric studies of volumetric arc therapy (VMAT) have been conducted and indicate that close attention to beam angles is important in order to minimize hippocampal exposure (Uto et al. 2016).
In addition to studies using common 1.8 Gy fractionation schemes, given the relatively well-demarcated tumor boundaries on imaging, radiosurgery, or hypo-fractionated stereotactic radiotherapy have also been explored in the treatment of these tumors. For radiosurgery, the most commonly employed platform has been gamma knife. Patients selected for gamma knife radiotherapy typically have small tumors, measuring less than 3 cm; also, there must be a safe separation (generally 3–5 mm depending on stereotactic delivery technique) between the target and nearby critical structures such as the optic chiasm in order to allow for adequate target coverage while respecting normal tissue constraints. Reported doses delivered in single fractions using gamma knife range from 12 to 14 Gy (Amendola et al. 2003; Kobayashi 2009; Mokry 1999; Chung et al. 2000). In these select patients, reported disease control rates seem acceptable and toxicities minimal (Lee et al. 2014; Park et al. 2011; Xu et al. 2011). However, patient numbers included are small and outcomes for adult and pediatric patients are frequently not separated. Given various differences in patient selection (i.e., tumor volumes, etc.), it is difficult to compare these results to traditional fractionated regimens.
As for many pediatric brain tumors, proton therapy is increasingly used in the treatment of craniopharyngiomas. Boehling and colleagues described in detail the dosimetric advantages of proton therapy including the potential for sparing of vascular structures and the hippocampus, especially with advanced proton therapy delivery modalities, namely intensity modulated proton therapy (IMPT) (Boehling et al. 2012). One of the preliminary reports by Luu and colleagues on the use of proton therapy for patients with craniopharyngiomas showed equivalent local control rates (88%) as compared to previous photon-based studies (Luu et al. 2006). More recently, a multi-institutional study comparing proton therapy to photons, again reported comparable outcomes (Bishop et al. 2014).
7 Radiation Details
Currently, for traditionally fractionated therapies, doses range from 50.4 to 54 Gy delivered at 1.8 per fraction. The commonly used upper limit of 54 Gy likely represents the tolerance for structures such as the optic chiasm, which are frequently immediately adjacent to the target (Merchant et al. 2002, 2006; Bishop et al. 2014; Kiehna and Merchant 2010). Advanced photon-based therapies have improved normal tissue sparing, which includes the use of intensity modulated radiation therapy (IMRT) (Greenfield et al. 2015; Merchant et al. 2013). In a single institution series, 24 patients were treated to a dose of 50.4 Gy using IMRT, and the 5- and 10-year progression-free survival rates were consistent with those seen with 3D conformal or other photon-based therapies (Greenfield et al. 2015). Regarding target delineation, the gross tumor volume (GTV) is typically easily visualized and delineated following fusion of MR imaging with treatment planning CT. While there is some discussion of whether or not all cystic areas should be included in the GTV, tumor cells could be present in the cyst walls, which is the reason to ensure coverage with the prescription isodose line.
Some debate surrounds the appropriate clinical target volume (CTV) expansion to be utilized. Historically, larger expansions of 1–2 cm have been employed given less reliable immobilization and image-guidance. However, more recently, studies have suggested that a smaller target volume expansion of 5 mm may be safely employed (Merchant et al. 2013). This is justifiable based on the fact that craniopharyngiomas are not inherently infiltrative, unlike gliomas or other intrinsic brain tumors. Preliminary studies reporting disease control outcomes utilizing reduced margin treatments have documented good disease control outcomes (Merchant et al. 2013). However, it is important to note that the CTV should be customized for each individual patient; this includes ensuring coverage of areas of adhesion noted intraoperatively. For photon therapy planning target volume (PTV), expansions will depend on institutional practices with common values falling between 3 and 5 mm.
7.1 Cystic Considerations for Radiation Planning
While it has long been known that craniopharyngiomas may contain cystic components, only recently has it been appreciated the dynamic changes that occur within these structures. Investigators at the Massachusetts General Hospital were among the first to describe in detail the potential for rapid cystic changes in these tumors (Winkfield et al. 2009). A group of 24 pediatric patients, 19 with a cystic component, underwent treatment. Seventeen of the 19 patients with cysts received surveillance imaging during the course of radiation therapy. Six of these patients had imaging evidence of cyst enlargement with 4 requiring revision of the radiation plan in order to ensure target coverage. The authors recommended routine imaging during the course of radiation therapy in patients with tumors containing cystic components (Winkfield et al. 2009). Following this initial observation, a subsequent report from the University of Texas MD Anderson Cancer Center confirmed relatively frequent cystic changes during irradiation (40%), with some patients requiring alteration to the treatment plan (20%) (Bishop et al. 2014). Therefore, when small CTV and PTV expansions are used, weekly or biweekly imaging during treatment is recommended in patients with cystic components to their tumors in order to ensure adequate tumor coverage. Additionally, close surveillance imaging is of greater importance in the setting of reduced CTV margins and/or the use of proton therapy.
7.2 Intracavitary Therapies
A less commonly employed treatment approach is the intracavitary injection of either β-emitting radioisotopes or sclerosing substances. The radioisotopes (Schoenfeld et al. 2012) phosphorus or 90yttrium have been used both as a primary and salvage treatment for cystic craniopharyngiomas, in which they are injected into the cysts via a catheter. Once within the cyst, photons are emitted by beta decay allowing for delivery of high doses of radiation to the epithelial lining of the cyst. Several studies have reported cyst regression in 81–88% of patients (Blackburn et al. 1999; Leksell 1952; Pollock et al. 1995). Alternatively, intracystic catheters can be used to instill chemotherapeutic agents (i.e., bleomycin or interferon α) with some efficacy (Cavalheiro et al. 2010; Schubert et al. 2009).
8 Outcomes
There are not evidence-based guidelines or recommendations regarding a follow-up plan for patients with a history of craniopharyngioma. However, given the potential for cystic changes following treatment, a best practices approach would suggest that neuroimaging should be obtained routinely. Our institutional approach is to obtain post-radiation imaging 4–6 weeks following completion of treatment and every 3 months thereafter for 2–3 years, at which point longer intervals may be recommended. In addition to monitoring for disease relapse, a multidisciplinary team is essential to manage disease-related and iatrogenic toxicities. Endocrine function will need to be closely monitored and supplements added as indicated, especially for developing children and adolescents; the importance of close endocrine follow-up cannot be overstated. Periodic visual field testing is also important for early intervention. Other members of a multidisciplinary team (internists/pediatricians, neuropsychologists, teachers, dieticians, etc.) may be required depending on the needs of the patient.
8.1 Outcomes
While disease control rates are high, craniopharyngiomas are associated with decreased overall survival compared to the normal population; the 10-year survival outcomes commonly reported are between 80 and 91% (Schoenfeld et al. 2012; Bishop et al. 2014; Merchant et al. 2013; Karavitaki et al. 2006; Pereira et al. 2005; Fernandez-Miranda et al. 2012). In addition to the deaths attributable to disease progression or surgical mortality, these patients are at higher risk of cardiovascular and cerebrovascular death (Pereira et al. 2005). Notably, based on the current evidence, there does not seem to be a difference in long-term survival between patients treated with GTR versus STR and postoperative radiation therapy. However, a recent SEER study did suggest a short-term survival benefit independently associated with STR (HR 0.39) and RT (HR 0.45) on multivariate analysis (Zacharia et al. 2012).
In terms of surgical resection, GTR rates range widely (between about 30 and 75%), with recurrences most commonly reported to occur in about 8–30% of patients following a GTR (Caldarelli et al. 2005; Fahlbusch et al. 1999; Sanford 1994; Stripp et al. 2004; Shi et al. 2008; Gardner et al. 2008). Conversely, the rate of recurrence following a STR alone is unacceptably high (40–75%) without RT (Fahlbusch et al. 1999; Karavitaki et al. 2005; Villani et al. 1997). However, direct outcome comparisons are difficult given the variable median follow-up and differing definitions of disease progression.
Patients that receive STR and postoperative radiation therapy appear to have similar control rates to GTR with disease relapse occurring in 0–12% of patients (Schoenfeld et al. 2012; Bishop et al. 2014; Merchant et al. 2013). Several studies have also differentiated solid from cystic tumor progression. Immediately following radiotherapy many patients experience transient cyst growth before they regress (Bishop et al. 2014; Shi et al. 2008). Shi and colleagues reported cyst enlargement in 11 of 21 patients following radiation therapy. Similarly, Bishop and colleagues observed a third of patients to have evidence of cyst growth within 3 months of completing treatment. However, in the majority of cases, growth was transient and followed by subsequent shrinkage of the cystic component. Reported 3-year recurrence rates of the solid tumor component were 5% versus 24% for cysts (Bishop et al. 2014). Cyst growth is appreciably more challenging to control, but its biologic significance is undefined.
If tumors recur, management is increasingly difficult primarily because surgical resection of recurrent tumors is more challenging. The recurrent tumors are commonly more adherent to surrounding tissue with poorly defined tissue planes, which is evidenced by the lower rate of resection (13–53%) achieved at the time of recurrence (Fahlbusch et al. 1999; Villani et al. 1997; Duff et al. 2000). For patients that recur, surgery alone is unlikely to provide durable control. In a study by Kalapurakal and colleagues, the 10-year progression-free survival after surgery and radiation therapy for recurrent tumors was 82–83% compared to 0% for surgery alone (Kalapurakal et al. 2000). However, withholding radiation in the definitive treatment setting in order to reserve it for use at the time of salvage is not justified based on available evidence. In fact, a recently published study by Bishop and colleagues showed that recurrent cyst growth was associated with poorer visual outcomes and hypothalamic obesity; furthermore, radiation therapy as salvage therapy negatively affected endocrine function (Bishop et al. 2014). Therefore, reserving radiation for salvage in the setting of STR is not advisable.
8.2 Toxicities
While patients may present with varying degrees of endocrine or visual dysfunction, treatment may impart additional morbidity. The rate of endocrine dysfunction increases significantly after surgical resection given the proximity, and sometimes involvement, of the tumors to the pituitary-hypothalamic axis (Caldarelli et al. 2005; Hoffman et al. 1992; Merchant et al. 2002). Diabetes insipidus is the most commonly reported postoperative endocrine complication. It occurs transiently in nearly all patients (80–100%) and permanently in 40–93% (Caldarelli et al. 2005; Hoffman et al. 1992; Merchant et al. 2002; Poretti et al. 2004). Anterior pituitary function also is often commonly compromised with several reports suggesting panhypopituitarism in up to 75–100% of patients following surgery (Fahlbusch et al. 1999; Kalapurakal et al. 2000; De Vile et al. 1996). Visual outcomes may also be influenced by surgical interventions. While some patients experience improvement in their preoperative visual deficits (41–58%) (Caldarelli et al. 2005; Elliott et al. 2011), others have further deterioration (2–66%) following resection (Sughrue et al. 2010; Fahlbusch et al. 1999; Poretti et al. 2004). Finally, another commonly underreported, yet serious toxicity associated with surgery is hypothalamic dysfunction. Symptoms include disturbed circadian rhythm, behavioral changes, obesity, or temperature/thirst regulation, and it may worsen in up to 65–80% of patients (Muller 2013; Elliott et al. 2011; Poretti et al. 2004). Other less common morbidities associated with surgery include neurologic, cerebrovascular, or cognitive complications. For example, Merchant and colleagues reported a 10-point drop in IQ scores in 15 patients treated with GTR alone (Merchant et al. 2002).
Similar to surgery, it is challenging to distinguish the long-term toxicities of radiation therapy from disease-related sequelae. The most commonly reported late toxicity associated with radiation therapy is worsening endocrine dysfunction, which is observed in 77–95% of patients (Clark et al. 2013; Bishop et al. 2014); panhypopituitarism is reportedly induced in 30–46% of patients (Bishop et al. 2014; Clark et al. 2012). Reduced visual acuity is also a potential long-term toxicity but is unusual using modern radiation techniques if normal tissue constraints are met. Hypothalamic obesity is a particular morbid treatment-related toxicity that requires specialized management; the impact of radiation therapy on this toxicity is uncertain, but the prevalence of obesity following combined modality treatment has been reported in 25–55% of patients (Muller 2008; Hoffman et al. 1992; Elliott et al. 2011; Bishop et al. 2014).
Less common toxicities associated with radiation include neurocognitive decline and cerebrovascular changes. Merchant and colleagues reported a median drop in IQ scores of only 1.25 points in patients treated with surgery and radiation (compared to 10 points in patients receiving aggressive surgery only) (Merchant et al. 2002). Vascular changes and stroke have also been observed in patients treated for craniopharyngiomas. Mueller and colleagues suggested that the incidence of stroke is increased tenfold in patients with childhood cancers,(Mueller et al. 2013) with several studies specific to craniopharyngiomas reporting rates of late vascular accidents or moya-moya in about 10% of patients after long-term follow-up (Bishop et al. 2014; Liu et al. 2009; Lo et al. 2014, 2016). Specifically, one cross-sectional study observed vasculopathies in 32% of patients (n = 6 of 32) (Lo et al. 2016). However, modern radiation techniques may lower the rate of late toxicities compared to those reported in historical cohorts.
Conclusions
Debates regarding the appropriate management of craniopharyngiomas will continue. Debates include deciding upon the appropriate surgical intervention, whether radical resection versus limited decompression is indicated. Increasingly, there is a trend towards limited surgical resection followed by adjuvant radiotherapy. While radiation oncologists will agree there is an established role for adjuvant radiation therapy if residual disease is present following surgery, there is still debate regarding the appropriate treatment modality and/or techniques (photons vs. protons vs. stereotactic, etc.) as there is no prospective data documenting differing disease control or toxicity outcomes. Beyond neurosurgeons and radiation oncologists, other providers, particularly endocrinologists, should routinely be involved in the upfront and continued management of these patients. Given the excellent survival outcomes for craniopharyngioma patients, all practitioners should focus on minimizing and managing post-treatment morbidities, thereby improving quality of life for survivors.
References
Aggarwal A, Fersht N, Brada M (2013) Radiotherapy for craniopharyngioma. Pituitary 16:26–33
Amendola BE, Wolf A, Coy SR et al (2003) Role of radiosurgery in craniopharyngiomas: a preliminary report. Med Pediatr Oncol 41:123–127
Becker G, Kortmann RD, Skalej M et al (1999) The role of radiotherapy in the treatment of craniopharyngioma–indications, results, side effects. Front Radiat Ther Oncol 33:100–113
Bishop AJ, Greenfield B, Mahajan A et al (2014) Proton beam therapy versus conformal photon radiation therapy for childhood craniopharyngioma: multi-institutional analysis of outcomes, cyst dynamics, and toxicity. Int J Radiat Oncol Biol Phys 90:354–361
Blackburn TP, Doughty D, Plowman PN (1999) Stereotactic intracavitary therapy of recurrent cystic craniopharyngioma by instillation of 90yttrium. Br J Neurosurg 13:359–365
Boehling NS, Grosshans DR, Bluett JB et al (2012) Dosimetric comparison of three-dimensional conformal proton radiotherapy, intensity-modulated proton therapy, and intensity-modulated radiotherapy for treatment of pediatric craniopharyngiomas. Int J Radiat Oncol Biol Phys 82:643–652. Epub 2011 Jan 27
Brastianos PK, Taylor-Weiner A, Manley PE et al (2014) Exome sequencing identifies BRAF mutations in papillary craniopharyngiomas. Nat Genet 46:161–165
Bunin GR, Surawicz TS, Witman PA et al (1998) The descriptive epidemiology of craniopharyngioma. J Neurosurg 89:547–551
Buslei R, Nolde M, Hofmann B et al (2005) Common mutations of beta-catenin in adamantinomatous craniopharyngiomas but not in other tumours originating from the sellar region. Acta Neuropathol 109:589–597
Caldarelli M, Massimi L, Tamburrini G et al (2005) Long-term results of the surgical treatment of craniopharyngioma: the experience at the Policlinico Gemelli, Catholic University, Rome. Childs Nerv Syst 21:747–757
Cavalheiro S, Di Rocco C, Valenzuela S et al (2010) Craniopharyngiomas: intratumoral chemotherapy with interferon-alpha: a multicenter preliminary study with 60 cases. Neurosurg Focus 28:E12
Choux M, Lena G (1979) Bases of surgical management of craniopharyngioma in children [proceedings]. Acta Neurochir Suppl (Wien) 28:348
Chung WY, Pan DH, Shiau CY et al (2000) Gamma knife radiosurgery for craniopharyngiomas. J Neurosurg 93(suppl 3):47–56
Clark AJ, Cage TA, Aranda D et al (2012) Treatment-related morbidity and the management of pediatric craniopharyngioma: a systematic review. J Neurosurg Pediatr 10:293–301
Clark AJ, Cage TA, Aranda D et al (2013) A systematic review of the results of surgery and radiotherapy on tumor control for pediatric craniopharyngioma. Childs Nerv Syst 29:231–238
De Vile CJ, Grant DB, Kendall BE et al (1996) Management of childhood craniopharyngioma: can the morbidity of radical surgery be predicted? J Neurosurg 85:73–81
Duff J, Meyer FB, Ilstrup DM et al (2000) Long-term outcomes for surgically resected craniopharyngiomas. Neurosurgery 46:291–302; discussion 302–305
Elliott RE, Jane JA Jr, Wisoff JH (2011) Surgical management of craniopharyngiomas in children: meta-analysis and comparison of transcranial and transsphenoidal approaches. Neurosurgery 69:630–643; discussion 643
Fahlbusch R, Honegger J, Paulus W et al (1999) Surgical treatment of craniopharyngiomas: experience with 168 patients. J Neurosurg 90:237–250
Fernandez-Miranda JC, Gardner PA, Snyderman CH et al (2012) Craniopharyngioma: a pathologic, clinical, and surgical review. Head Neck 34:1036–1044
Gardner PA, Kassam AB, Snyderman CH et al (2008) Outcomes following endoscopic, expanded endonasal resection of suprasellar craniopharyngiomas: a case series. J Neurosurg 109:6–16
Greenfield BJ, Okcu MF, Baxter PA et al (2015) Long-term disease control and toxicity outcomes following surgery and intensity modulated radiation therapy (IMRT) in pediatric craniopharyngioma. Radiother Oncol 114:224–229. doi:10.1016/j.radonc.2014.11.035 . Epub 2014 Dec 23
Habrand JL, Ganry O, Couanet D et al (1999) The role of radiation therapy in the management of craniopharyngioma: a 25-year experience and review of the literature. Int J Radiat Oncol Biol Phys 44:255–263
Habrand JL, Saran F, Alapetite C et al (2006) Radiation therapy in the management of craniopharyngioma: current concepts and future developments. J Pediatr Endocrinol Metab 19(suppl 1):389–394
Hoffman HJ, De Silva M, Humphreys RP et al (1992) Aggressive surgical management of craniopharyngiomas in children. J Neurosurg 76:47–52
Izuora GI, Ikerionwu S, Saddeqi N et al (1989) Childhood intracranial neoplasms Enugu, Nigeria. West Afr J Med 8:171–174
Jane JA Jr, Laws ER (2006) Craniopharyngioma. Pituitary 9:323–326
Kalapurakal JA, Goldman S, Hsieh YC et al (2000) Clinical outcome in children with recurrent craniopharyngioma after primary surgery. Cancer J 6:388–393
Karavitaki N, Brufani C, Warner JT et al (2005) Craniopharyngiomas in children and adults: systematic analysis of 121 cases with long-term follow-up. Clin Endocrinol 62:397–409
Karavitaki N, Cudlip S, Adams CB et al (2006) Craniopharyngiomas. Endocr Rev 27:371–397
Khan RB, Merchant TE, Boop FA et al (2013) Headaches in children with craniopharyngioma. J Child Neurol 28:1622–1625
Kiehna EN, Merchant TE (2010) Radiation therapy for pediatric craniopharyngioma. Neurosurg Focus 28:E10
Kobayashi T (2009) Long-term results of gamma knife radiosurgery for 100 consecutive cases of craniopharyngioma and a treatment strategy. Prog Neurol Surg 22:63–76
Lee CC, Yang HC, Chen CJ et al (2014) Gamma knife surgery for craniopharyngioma: report on a 20-year experience. J Neurosurg 121:167–178. doi:10.3171/2014.8.GKS141411
Leksell L (1952) LK: a therapeutic trial with radioactive isotope in cystic brain tumor: radioisotope technieques I. Med Physiol Appl 1:1–4
Liu AK, Bagrosky B, Fenton LZ et al (2009) Vascular abnormalities in pediatric craniopharyngioma patients treated with radiation therapy. Pediatr Blood Cancer 52:227–230
Lo AC, Howard AF, Nichol A et al (2014) Long-term outcomes and complications in patients with craniopharyngioma: the British Columbia Cancer Agency experience. Int J Radiat Oncol Biol Phys 88:1011–1018
Lo AC, Howard AF, Nichol A et al (2016) A cross-sectional cohort study of cerebrovascular disease and late effects after radiation therapy for Craniopharyngioma. Pediatr Blood Cancer 63:786–793
Luu QT, Loredo LN, Archambeau JO et al (2006) Fractionated proton radiation treatment for pediatric craniopharyngioma: preliminary report. Cancer J 12:155–159
Marucci G, de Biase D, Zoli M et al (2015) Targeted BRAF and CTNNB1 next-generation sequencing allows proper classification of nonadenomatous lesions of the sellar region in samples with limiting amounts of lesional cells. Pituitary 18:905–911. doi:10.1007/s11102-015-0669-y
Merchant TE, Kiehna EN, Sanford RA et al (2002) Craniopharyngioma: the St. Jude Children’s Research Hospital experience 1984-2001. Int J Radiat Oncol Biol Phys 53:533–542
Merchant TE, Kiehna EN, Kun LE et al (2006) Phase II trial of conformal radiation therapy for pediatric patients with craniopharyngioma and correlation of surgical factors and radiation dosimetry with change in cognitive function. J Neurosurg 104:94–102
Merchant TE, Kun LE, Hua CH et al (2013) Disease control after reduced volume conformal and intensity modulated radiation therapy for childhood craniopharyngioma. Int J Radiat Oncol Biol Phys 85:e187–e192
Minniti G, Esposito V, Amichetti M et al (2009) The role of fractionated radiotherapy and radiosurgery in the management of patients with craniopharyngioma. Neurosurg Rev 32:125–132; discussion 132
Mokry M (1999) Craniopharyngiomas: a six year experience with gamma knife radiosurgery. Stereotact Funct Neurosurg 72(suppl 1):140–149
Moore KCW (2000) Craniopharyngioma. In: Bernstein M, Berger M (eds) Neuro-oncology: the essentials. Thieme, New York
Mueller S, Fullerton HJ, Stratton K et al (2013) Radiation, atherosclerotic risk factors, and stroke risk in survivors of pediatric cancer: a report from the Childhood Cancer Survivor Study. Int J Radiat Oncol Biol Phys 86:649–655
Muller HL (2008) Childhood craniopharyngioma. Recent advances in diagnosis, treatment and follow-up. Horm Res 69:193–202
Muller HL (2010) Childhood craniopharyngioma: current controversies on management in diagnostics, treatment and follow-up. Expert Rev Neurother 10:515–524
Muller HL (2013) Childhood craniopharyngioma. Pituitary 16:56–67
Muller HL (2014) Craniopharyngioma. In: Eliers EKM, Romijn JA (eds) Handbook of clinical neurology. Elsevier, Cambridge
Park YS, Chang JH, Park YG et al (2011) Recurrence rates after neuroendoscopic fenestration and Gamma Knife surgery in comparison with subtotal resection and Gamma Knife surgery for the treatment of cystic craniopharyngiomas. J Neurosurg 114:1360–1368. doi:10.3171/2009.9.JNS09301 . Epub 2009 Oct 30
Pereira AM, Schmid EM, Schutte PJ et al (2005) High prevalence of long-term cardiovascular, neurological and psychosocial morbidity after treatment for craniopharyngioma. Clin Endocrinol 62:197–204
Petito CK, DeGirolami U, Earle KM (1976) Craniopharyngiomas: a clinical and pathological review. Cancer 37:1944–1952
Pollock BE, Lunsford LD, Kondziolka D et al (1995) Phosphorus-32 intracavitary irradiation of cystic craniopharyngiomas: current technique and long-term results. Int J Radiat Oncol Biol Phys 33:437–446
Poretti A, Grotzer MA, Ribi K et al (2004) Outcome of craniopharyngioma in children: long-term complications and quality of life. Dev Med Child Neurol 46:220–229
Rath SR, Lee S, Kotecha RS et al (2013) Childhood craniopharyngioma: 20-year institutional experience in Western Australia. J Paediatr Child Health 49:403–408
Rose SR, Lustig RH, Pitukcheewanont P et al (1999) Diagnosis of hidden central hypothyroidism in survivors of childhood cancer. J Clin Endocrinol Metab 84:4472–4479
Samii M, Tatagiba M (1997) Surgical management of craniopharyngiomas: a review. Neurol Med Chir (Tokyo) 37:141–149
Sanford RA (1994) Craniopharyngioma: results of survey of the American Society of Pediatric Neurosurgery. Pediatr Neurosurg 21(suppl 1):39–43
Schoenfeld A, Pekmezci M, Barnes MJ et al (2012) The superiority of conservative resection and adjuvant radiation for craniopharyngiomas. J Neuro-Oncol 108:133–139
Schubert T, Trippel M, Tacke U et al (2009) Neurosurgical treatment strategies in childhood craniopharyngiomas: is less more? Childs Nerv Syst 25:1419–1427
Sekine S, Shibata T, Kokubu A et al (2002) Craniopharyngiomas of adamantinomatous type harbor beta-catenin gene mutations. Am J Pathol 161:1997–2001
Shi XE, Wu B, Fan T et al (2008) Craniopharyngioma: surgical experience of 309 cases in China. Clin Neurol Neurosurg 110:151–159
Stiller CA, Nectoux J (1994) International incidence of childhood brain and spinal tumours. Int J Epidemiol 23:458–464
Stripp DC, Maity A, Janss AJ et al (2004) Surgery with or without radiation therapy in the management of craniopharyngiomas in children and young adults. Int J Radiat Oncol Biol Phys 58:714–720
Sughrue M, Lustig RH, Fisch B et al (2010) Craniopharyngioma. In: Gupta N, Banerjee A, Haas-Kogan D (eds) Pediatric CNS tumors (2nd edn). Springer, Berlin, pp xxv, 357
Uto M, Mizowaki T, Ogura K et al (2016) Non-coplanar volumetric-modulated arc therapy (VMAT) for craniopharyngiomas reduces radiation doses to the bilateral hippocampus: a planning study comparing dynamic conformal arc therapy, coplanar VMAT, and non-coplanar VMAT. Radiat Oncol 11:86. doi:10.1186/s13014–016-0659-x
Van Effenterre R, Boch AL (2002) Craniopharyngioma in adults and children: a study of 122 surgical cases. J Neurosurg 97:3–11
Villani RM, Tomei G, Bello L et al (1997) Long-term results of treatment for craniopharyngioma in children. Childs Nerv Syst 13:397–405
Warmuth-Metz M, Gnekow AK, Muller H et al (2004) Differential diagnosis of suprasellar tumors in children. Klin Padiatr 216:323–330
Weiner HL, Wisoff JH, Rosenberg ME et al (1994) Craniopharyngiomas: a clinicopathological analysis of factors predictive of recurrence and functional outcome. Neurosurgery 35:1001–1010; discussion 1010–1011
Winkfield KM, Linsenmeier C, Yock TI et al (2009) Surveillance of craniopharyngioma cyst growth in children treated with proton radiotherapy. Int J Radiat Oncol Biol Phys 73:716–721
Xu Z, Yen CP, Schlesinger D et al (2011) Outcomes of Gamma Knife surgery for craniopharyngiomas. J Neurooncol 104:305–313. doi:10.1007/s11060–010–0494-0. Epub 2010 Dec 14
Yasargil MG, Curcic M, Kis M et al (1990) Total removal of craniopharyngiomas. Approaches and long-term results in 144 patients. J Neurosurg 73:3–11
Zacharia BE, Bruce SS, Goldstein H et al (2012) Incidence, treatment and survival of patients with craniopharyngioma in the surveillance, epidemiology and end results program. Neuro-Oncology 14:1070–1078
Zona G, Spaziante R (2006) Management of cystic craniopharyngiomas in childhood by a transsphenoidal approach. J Pediatr Endocrinol Metab 19(suppl 1):381–388
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG
About this chapter
Cite this chapter
Bishop, A.J., Grosshans, D.R. (2018). Craniopharyngioma. In: Mahajan, A., Paulino, A. (eds) Radiation Oncology for Pediatric CNS Tumors. Springer, Cham. https://doi.org/10.1007/978-3-319-55430-3_16
Download citation
DOI: https://doi.org/10.1007/978-3-319-55430-3_16
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-55428-0
Online ISBN: 978-3-319-55430-3
eBook Packages: MedicineMedicine (R0)