Abstract
Sustained isometric contractions of skeletal muscles produce intramuscular pressures that lead to blood flow restriction. Thus, we have the paradox of rising O2 demand due to muscle activity and at the same time reduced blood flow. The aim was to assess muscle oxygenation during sustained isometric low (30%), moderate (60%) and submaximal [90% of maximal voluntary contraction (MVC)] contraction of the dorsiflexor muscle. Experiments were conducted on the dominant (right) leg of 8 male students (age 19 ± 2 years, weight 75 ± 6 kg). Tissue oxygen saturation (StO2) was recorded from the tibialis anterior using near-infrared spectroscopy. StO2 was higher at 30% compared to both 60% and 90% MVC at all time points after the start of the exercise and higher at 60% than 90%. This indicates that the supply of O2 did not keep up with its consumption. During arterial occlusion the minimal StO2 reached 52%, which is significantly higher than StO2 during 60% and 90% MVC. After each contraction there was a large and immediate hyperemic response, whose resaturation rate continuously increased from 30% to 60% to 90% MVC. The StO2 resaturation rate was positively correlated with the MVC, indicating a vasodilation depending on the intensity of the exercise.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Sustained isometric contraction (SIC) of skeletal muscle s produces intramuscular pressure (IMP) that restricts muscle blood flow (MBF) and limits O2 delivery to tissue [1]. MBF plays a key role in regulating the intensity and type of muscle contractions [2]. A limited MBF due to SIC leads to fatigue due lack of O2 and nutrients. Thus, we have the paradox of rising O2 demand due to muscle activity and at the same time reduced MBF. To clarify this situation much research has been performed mainly on MBF [3,4,5,6,7]. However, during exercise there are few measurements of MBF by Doppler ultrasound. Previous studies indicate that complete occlusion of MBF occurs at 50–60% of maximal voluntary contractions (MVC) during SIC [3, 4, 6]. MBF was not occluded at the level of the conduit artery during any contraction intensity [3]. Thus, our understanding of the oxygenation of the skeletal muscle in response to different intensities of SIC (low, moderate, submaximal MVC) is still limited.
Despite advantages of fMRI, PET and Doppler ultrasound only one paper has been devoted to measure the hemodynamic response of muscles to SIC [3] by near infrared spectroscopy (NIRS) . In the near-infrared (NIR) spectrum (700–900 nm) light penetrates deeply into the tissue and oxyhemoglobin (O2Hb) and deoxyhemoglobin (HHb) are the strongest absorbers, while myoglobin (Mb) absorbs less. NIRS is an established optical technique to monitor concentration changes of O2Hb, HHb, total haemoglobin (tHb) and tissue oxygen saturation (StO2) in a variety of tissues [8, 9]. NIRS instruments are non-invasive, small, and applicable in exercise physiology.
The aim was to assess changes in muscle oxygenation during low, moderate and submaximal SIC of the dorsiflexor muscle.
2 Methods
Eight male students of USIPC were included (age 19 ± 2 years, weight 75 ± 6 kg) and measured on the dominant (right) leg. The foot was placed in an in-house built isometric torso-dynamometer (DC-200, Russia). Tissue O2 saturation (StO2) was recorded from the tibialis anterior by NIRS (SenSmart™ Model X-100, NONIN). The sensor was placed over the belly of the tibialis anterior muscle of each individuals with spacing of optodes of 4 cm.
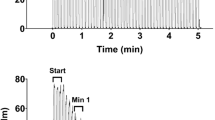
The experiment consisted of pre-exercise (rest) and three successive periods (each ~1 min) of sustained isometric contractions at 30%, 60% and 90% of MVC and separated by rest periods of 3 min (Fig. 4.1). To determine the minimal StO2 in the dorsiflexors an arterial occlusion was performed at the end of the experiment. The dorsiflexor muscle was selected, because both venous outflow and arterial inflow can be occluded by a proximal cuff. Without blood supply, the muscle metabolism depends on the O2 in capillaries and muscle cells. Consequently, the O2Hb and StO2 decrease, while HHb increases and tHb remains constant. After the occlusion a hyperemic response occurs, i.e. a rapid increase in O2Hb, tHb and StO2, while HHb is washed-out. From this procedure, we calculated O2 consumption, reoxygenation rate and the half-recovery times of the signals [10].
The recovery baseline (RB) value is the StO2 value after stabilisation during the rest period following a test period. The performance baseline is the minimum StO2 value reached during SIC. For each individual the NIRS signal was normalized to its maximal value reached during reperfusion after cessation of occlusion. Differences in the StO2, desaturation rate (De) and resaturation rate (Re) between muscle contraction intensities were analysed by Student’s t-test in the statistical software package Statistica for Windows (version 13).
3 Results
Figure 4.1 displays a typical measurement in one subject. The drop in StO2 increased from low to moderate to submaximal SIC (group values: Table 4.1). Figure 4.2 compares the StO2 traces for the three different intensities of SIC in one subject. StO2 was higher at 30% compared with both 60% and 90% MVC and higher at moderate than submaximal MVC (p < 0.05). Desaturation rate (De) increased from slow (30%) to moderate (60%) to submaximal contractions (p < 0.05) (Table 4.1). Trends of De as a function of MVC are shown in Fig. 4.3. In Fig. 4.3 the deoxygenation rate has an S-like shape and its linear approximation is shown as black line. Similarly the linear approximation of the reoxygenation rate in Fig. 4.3 shown as black line. After each contraction there was a large and immediate hyperemic response (Fig. 4.2). The resaturation rate (Re) of StO2 after SIC depends on the intensity of the SIC and reflects the integrity and functionality of vascular system. It corresponds to a blood vessel vasodilation in response to the SIC. Re increased from slow to moderate (p < 0.05), but remained similar for moderate to submaximal SIC (Table 4.1 and Fig. 4.2).
During the arterial occlusion the minimal StO2 was 52% (Table 4.1 and Fig. 4.1), which is significantly higher than StO2 after moderate or submaximal SIC.
4 Discussion
According to the literature [11] an arterial occlusion is applied by inflating the cuff to a pressure of at least 60 mmHg above the systolic pressure. This way, both venous outflow and arterial inflow are blocked. In our case the mean pressure was 88 ± 6 mmHg what correspond to a systolic arterial pressure of about 115–120 mmHg. Blood flow was occluded to the leg by inflating the cuff beyond 180 mmHg. But either this external pressure was not high enough to block blood inflow, or the lower extremity circumference of greater than 75 cm would have required a more efficient tourniquet. Due to these reasons, in dorsiflexor muscle we did have not a full blocking of blood inflow and accordingly we observed during the arterial occlusion a minimal StO2 which was significantly higher than the StO2 after moderate or submaximal SIC.
The muscle deoxygenation rate was more enhanced at 90% than 60% MVC because O2 consumption in exercising muscle is higher at 90% than 60% which we can see as statistically significant increasing of StO2 from 60% to 90% MVC (Fig. 4.3).
5 Conclusion
The sharp decreases in StO2 after the start of moderate and submaximal SIC, indicate that the blood vessels are occluded due to intramuscular pressure. This indeed shows that O2 delivery is impeded and cannot cope with the increased O2 consumption.
References
Degens H, Salmons S, Jarvis JC (1998) Intramuscular pressure, force and blood flow in rabbit tibialis anterior muscles during single and repetitive contractions. Eur J Appl Physiol Occup Physiol 78:13–19
Saltin B, Radegran G, Koskolou MD, Roach RC (1998) Skeletal muscle blood flow in humans and its regulation during exercise. Acta Physiol Scand 162:421–436
Chris J, Mcneil CJ, Matti D, Allen MD, Olympico E, Shoemaker JK, Rice CL (2015) Blood flow and muscle oxygenation during low, moderate, and maximal sustained isometric contractions. Am J Physiol Regul Integr Comp Physiol 309:R475–R481
Sjogaard G, Savard G, Juel C (1988) Muscle blood flow during isometric activity and its relation to muscle fatigue. Eur J Appl Physiol Occup Physiol 57:327–335
Vollestad NK, Wesche J, Sejersted OM (1990) Gradual increase in leg oxygen uptake during repeated submaximal contractions in humans. J Appl Physiol 68:1150–1156
Wesche J (1986) The time course and magnitude of blood flow changes in the human quadriceps muscles following isometric contraction. J Physiol 377:445–462
Hughson RL, Shoemaker JK, Tschakovsky ME, Kowalchuk JM (1996) Dependence of muscle VO2 on blood flow dynamics at onset of forearm exercise. J Appl Physiol 81:1619–1626
Wolf M, Ferrari M, Quaresima V (2007) Progress of near-infrared spectroscopy and topography for brain and muscle clinical applications. J Biomed Opt 12(6):062104
Hamaoka T, McCully KK, Quaresima V, Yamamoto K, Chance B (2007) Near-infrared spectroscopy/imaging for monitoring muscle oxygenation and oxidative metabolism in healthy and diseased humans. J Biomed Opt 12(6):062105 1–12
Gerovasili V, Dimopoulos S, Tzanis G, Anastasiou-Nana M, Nanas S (2010) Utilizing the vascular occlusion technique with NIRS technology. Int J Ind Ergon 40:218–222
Van Beekvelt MCP, Colier WNJM, Wopvers RA, Van Engelen BGM (2001) Performance of infra-red spectroscopy in measuring local O2 consumption and blood flow in skeletal muscle. J Appl Physiol 90(2):511–519
Acknowledgments
The authors gratefully acknowledge funding by the Swiss National Science Foundation (Gr. no. 137423).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing AG
About this chapter
Cite this chapter
Paiziev, A., Wolf, M., Kerimov, F. (2017). Dorsiflexor Muscle Oxygenation During Low, Moderate and Submaximal Sustained Isometric Contraction. In: Halpern, H., LaManna, J., Harrison, D., Epel, B. (eds) Oxygen Transport to Tissue XXXIX. Advances in Experimental Medicine and Biology, vol 977. Springer, Cham. https://doi.org/10.1007/978-3-319-55231-6_4
Download citation
DOI: https://doi.org/10.1007/978-3-319-55231-6_4
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-55229-3
Online ISBN: 978-3-319-55231-6
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)