Abstract
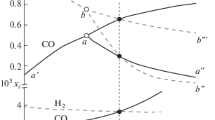
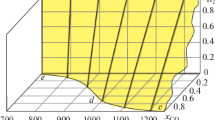
The thermochemical data of iron oxide redox reactions in various textbooks and handbooks are not consistent. To clarify such confusions, the elementary thermodynamic data of various iron oxides, carbon oxides, hydrogen and water vapor are used to calculate the changes of thermodynamic quantities such as enthalpy, entropy and Gibbs free energy of the redox reactions. The predominance area diagrams are then reconstructed according to the newly calculated Gibbs free energy changes. In order to fit the precise Gibbs free energy data, the constrained optimization method is adopted based on the mathematical modeling software Lingo 11. The reduction experiments are successfully carried out to verify the calculated eutectoid temperature. It is concluded with sixteen empirical thermodynamic equilibrium formulas and eight enthalpy values at 25°C for iron oxides reduced by CO and H2, and the eutectoid temperature of the three iron oxide phases is 576°C.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
References
Bogdandy, L. V.; Engell, H. J. The Reduction of Iron Ores; Springer Verlag: Berlin, 1971.
Liang, L. K.; Che, Y. C.; Yang, H.; Li, X. W. Metallurgical Thermodynamics and Kinetics; Northeastern University of Technology Press: Shenyang, 1990; pp 86–90.
Kirkaldy, J. S.; Ward, R. G. Aspects of Modern Ferrous Metallurgy; University of Toronto Press: Toronto, 1964; pp 102–105.
Rao Y. K. Stoichiometry and Thermodynamics of Metallurgical Processes; Cambridge University Press: London, 1985; pp 726–773.
Han, Q. Y. Metallurgical Kinetics; Metallurgical Industry Press: Beijing, 1983; pp 156–157.
John, F. E. The Physical Chemistry of Steelmaking; The Technology Press of MIT, John Wiley & Sons, Inc. & Chapman & Hall, 1956; pp 159–164.
Coudurier, L.; Hopkins, D. W.; Wilkomirsky, I. Fundamentals of Metallurgical Processes, 2nd ed.; Pergamon Press: New York, 1985; pp 159–162.
Chu, M.; Nogami, H.; Yagi, J. I. ISIJ Int. 2004, 44(3), 510.
Chu, M.; Yagi, J. I. Steel Res. Int. 2010, 81(12), 1043.
Bahgat, M.; Halim, K. S. A.; El-Kelesh, H. A.; Nasr M. I. Steel Res. Int. 2012, 83(7), 686.
Barin, I. Thermochemical Data of Pure Substances, 3rd ed.; Wiley-Vch Verlag: Weinheim, 1995.
NIST-JANAF. Thermochemical Tables, 4th ed.; Journal of Physical and Chemical Reference Data Monograph, No.9, 1998.
Barin, L; Knacke, O. Thermochemical Properties of Inorganic Substances, Springer-Verlag: Berlin, 1973; Supplement, 1977.
Braithwaite. Chem. News 1895, 72, 211.
Baur, L. E.; Glaessner, A. Z. Phys. Chemie 1903, 354(43), 68.
Zhang, W.; Zou, Z. S.; Zhang, J. H.; LI, Q.; Qi, Y. H. J. Phys. Chem. C 2013, submitted.
Schenck; Semiller and Falcke. Chem. Ber. 1907, 40, 1704.
Schenck and Heller. Chem. Ber. 1905, 38, 2132.
Eastman, E. D. J. Am. Chem. Soc. 1922, 44(5), 975.
Eastman, E. D.; Evans, R. M. J. Am. Chem. Soc. 1924, 46(4), 888.
Emmett, P. H.; Shultz, J. F. J. Am. Chem. Soc. 1930, 52, 4268.
Emmett, P. H.; Shultz, J. F. J. Am. Chem. Soc. 1933, 55, 1376.
Chipman, J.; Marshall, S. J. Am. Chem. Soc. 1940, 62, 299.
Darken, L. S.; Gurry, R. W. J Am. Chem. Soc. 1945, 67, 1398.
Darken, L. S.; Gurry, R. W. J Am. Chem. Soc. 1946, 68, 789.
Wang, X. L. Ferrous Metallurgy (Ironmaking Department); Metallurgical Industry Press: Beijing, 2002; pp.82–85.
Li, H. G. Metallurgical Theory; Science Press: Beijing, 2005; pp. 156–206.
Zhu, M. Y. Modern Metallurgy (Ferrous Metallurgy); Metallurgical Industry Press: Beijing, 2005; pp.60–62.
Hara, Y.; Tsuchiya, M.; Kondo, S. I. Tetsu to Hagane 1974, No. 9, 1261.
Murayama, T.; Ono, Y.; Kawai, Y. Tetsu to Hagane 1977, No. 7, 1099.
Ono-Nakazato, H.; Yonezawa, T.; Usui, T. ISIJ Int. 2003, 43(10), 1502.
Takahashi, R.; Takahashi, Y.; Yagi, J. I.; Omori, I. Trans. ISIJ 1986, 26, 765.
Pineau, A.; Kanari, N.; Gaballah, I. Thermochim. Acta 2007, 456, 75.
Conejo, A. N.; Martins, G. P. ISIJ Int. 1997, 37(10), 967.
Hayashi, S.; Iguchi, Y. ISIJ Int. 1998, 38(10), 1053.
El-Geassy, A. A.; Nasr, M. I. ISIJ Int. 1990, 30(6), 417.
Pineau, A.; Kanari, N.; Gaballah, I. Thermochim. Acta 2006, 447, 89.
Wimmers, O. J.; Arnoldy, P.; Moulijn, J. A. J. Phys. Chem. 1986, 90, 1331.
Khader, M. M.; El-Anadouli, B. E.; El-Nagar, E.; Ateya, B. G. J. Solid State Chem. 1991, 93, 283.
Jozwiak, W. K.; Kaczmarek, E.; Maniecki, T. P. Appl. Catal., A-Gen. 2007, 326, 17.
Zhao, P.; Guo, P. M.; Zhang, D. W. Iron and Steel 2006, 41, 12.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 TMS (The Minerals, Metals & Materials Society)
About this paper
Cite this paper
Zhang, W. et al. (2013). Thermodynamic Analyses of Iron Oxides Redox Reactions. In: Marquis, F. (eds) Proceedings of the 8th Pacific Rim International Congress on Advanced Materials and Processing. Springer, Cham. https://doi.org/10.1007/978-3-319-48764-9_96
Download citation
DOI: https://doi.org/10.1007/978-3-319-48764-9_96
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-48586-7
Online ISBN: 978-3-319-48764-9
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)