Abstract
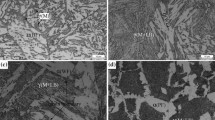
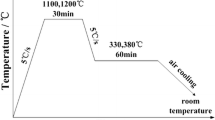
In this work, the isothermal bainite transformation in a low carbon multi-microalloyed steel was investigated using a dilatometer. Two different transformation behaviors were identified according to the measured dilatometric curves. The variation of transformation mechanism was explored based on the microstructural observation and the change of the Avrami exponent with the holding time and isothermal temperature. For example, the solute drag effect is mainly responsible for the transformation stasis phenomenon at the first stage of the abnormal reaction curve. A physically-based model was proposed to describe the normal bainite formation kinetics curves on the basis of the modification of the Johnson-Mehl-Avrami (JMA) equation. The transformation mode is considered as the site saturation of nucleation and interface controlled growth under the large undercooling condition. Comparison between the calculation curves and the experimental data in this experimental steel gives a very good agreement.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
References
S.E. Offerman et al., “Grain Nucleation and Growth during Phase Transformation,” Science, 298 (2002), 1003–1005.
A. Van der Ven and L. Delaey, “Models for Precipitate Growth during the γ→α + γ Transformation in Fe-C and Fe-C-M alloys,” Progress in Materials Science, 40 (1996), 181–264.
J.W. Christian, The Theory of Transformations in Metals and Alloys (Great Britain, GB: Pergamon press of Elsevier Science Ltd, 2002) 16–580.
K. Nishioka and K. Ichikawa, “Progress in Thermomechanical Control of Steel Plates and Their Commercialization,” Science and Technology of Advanced Materials, 13 (2012), 1–21.
F. Liu et al., “Analysis of Solid State Phase Transformation Kinetics: Models and Recipes,” International Materials Reviews, 52 (2007), 193–212.
E.J. Mittemeijer and F. Sommer, “Solid State Phase Transformation Kinetics: Evaluation of the Modular Transformation Model,” International Journal of Materials and Research, 102 (2011), 784–795.
D. Quidort and Y.J.M. Brechet, “A Model of Isothermal and Non Isothermal Transformation Kinetics of Bainite in 0.5% C Steel,” ISIJ International, 42 (2002), 1010–1017.
H. Matsuda and H.K.D.H. Bhadeshia, “Kinetics of the bainite transformation,” Proceeding of the Roral Society A, 460 (2004), 1707–1722.
F. Fazeli and M. Militzer, “Modelling Simultaneous Formation of Bainitic Ferrite and Carbide in TRIP Steels,” ISIJ International, 52 (2012), 650–658.
M.J. Santofimia et al., “New Model for the Overall Transformation Kinetics of Bainite. Part 1: the Model,” Materials Transcations, 47 (2006), 2465–2472.
W.T. Reynolds et al., “The Incomplete Transformation Phenomenon in Fe-C-Mo Alloys,” Metallurgical Transactions A, 21A (1990), 1433–1463.
F. Fazeli and M. Militzer, “Application of Solute Drag Theory to Model Ferrite Formation in Multiphase Steels,” Metallurgical and Materials Transactions A, 36A (2005), 1395–1405.
E. Kozeschnik and E. Gamsjager, “High-Speed Quenching Dilatometer Investigation of the Austenite-to-Ferrite Transformation in a Low to Ultralow Carbon Steel,” Metallurgical and Materials Transactions A, 37A (2006), 1791–1797.
Z.Q. Liu et al., “Direct measurement of carbon enrichment during austenite to ferrite transformation in hypoeutectoid Fe-2Mn-C alloys,” Acta Materialia, 2013, http://dx.doi.org/10.1016/j.actamat.2013.02.003.
M. Hillert, “Diffusion in growth of bainite,” Metallurgical and Materials Transactions A, 1994, 25A: 1957–1966.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 TMS (The Minerals, Metals & Materials Society)
About this paper
Cite this paper
Lan, L., Qiu, C., Zhao, D. (2013). Kinetics Modelling of Isothermal Bainite Transformation in Low Carbon Multi-Microalloyed Steel. In: Marquis, F. (eds) Proceedings of the 8th Pacific Rim International Congress on Advanced Materials and Processing. Springer, Cham. https://doi.org/10.1007/978-3-319-48764-9_339
Download citation
DOI: https://doi.org/10.1007/978-3-319-48764-9_339
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-48586-7
Online ISBN: 978-3-319-48764-9
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)