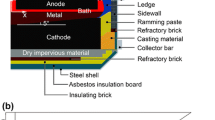
Abstract
The lifetime of an aluminum electrolysis cell is mainly determined by the cathode wear, especially for high amperage cells utilizing graphitized carbon cathodes. The cathode wear mechanisms are, however, complex and still subject to debate. Laboratory tests using inverted cell configurations have previously been used to study commercial cathode materials. Here we report on results obtained in the last 5 years for three different commercial cathode materials. The current density applied in the tests was in the range from 0 to 2 A/cm2 and the rotation speed of the cathode was from 0 to 125 rpm, corresponding to surface velocity 0–19.6 cm/s. The wear mechanisms with respect to current density, limiting current of Al4C3 formation and mass transport are discussed. In addition, similarities and differences between wear observed in the laboratory and in industry cells are discussed.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
References
M. Sørlie, H.A. Øye, Cathodes in Aluminium Electrolysis, 3rd ed., Aluminium-Verlag Marketing & Kommunikation GmbH, Germany, 2010.
E. Skybakmoen, S. Rorvik, A. Solheim, K.R. Holm, P. Tiefenbach, O. Ostrem, “Measurement of Cathode Surface Wear Profiles by Laser Scanning,” Light Metals, 2011, 1061–1066.
P. Reny, S. Wilkening, “Graphite cathode wear study at Alouette,” Light Metals, 2000, 399–404.
D. Lombard, T. Beheregaray, B. Feve, J.M. Jolas, “Aluminium Pechiney experience with graphitized cathode blocks,” Light Metals, 1998, 653–658.
C.W. Bale, P. Chartrand, S.A. Degterov, G. Eriksson, K. Hack, R.B. Mahfoud, J. Melançon, A.D. Pelton, S. Petersen, “FactSage thermochemical software and databases,” Calphad-computer Coupling of Phase Diagrams and Thermochemistry, 26, 2002, 189–228.
A.P. Ratvik, A. Støre, A. Solheim, T. Foosnæs, “I,” Light Metals, 2008, 973–978.
E. Skybakmoen, A.P. Ratvik, A. Solheim, S. Rolseth, H. Gudbrandsen, “Laboratory test methods for determining the cathode wear mechanism in aluminium cells,” Light Metals, 2007, 815–820.
K. Tschöpe, A. Støre, A. Solheim, E. Skybakmoen, T. Grande, A. Ratvik, “Electrochemical Wear of Carbon Cathodes in Electrowinning of Aluminum,” JOM 65, 2013, 1403–1410.
Y. Sato, P. Patel, P. Lavoie, “Erosion Measurements of High Density Cathode Block Samples Through Laboratory Electrolysis with Rotation,” Light Metals, 2010, 817–822.
P. Patel, Y. Sato, P. Lavoie, “Determination of the Effect of Pitch Impregnation on Cathode Erosion Rate,” Light Metals, 2011, 1073–1078.
K. Tschope, A. Store, S. Rorvik, A. Solheim, E. Skybakmoen, T. Grande, A.P. Ratvik, “Investigation of the Cathode Wear Mechanism in a Laboratory Test Cell,” Light Metals 2012, 1349–1354.
K. Tschöpe, E. Skybakmoen, A. Solheim, T. Grande, “Cathode wear in Hall-Heroult cells,” Aluminium, 2013, 40–43.
K. Tschöpe, A. Støre, E. Skybakmoen, A. Solheim, T. Grande, A.P. Ratvik, “Critical Reflections on Laboratory Wear Tests for Ranking Commercial Cathode Materials in Aluminium Cells,” Light Metals, 2013, 1251–1256.
H. Gudbrandsen, Å. Sterten, R. Ødegård, “Cathodic dissolution of carbon in cryolitic melts,” Light Metals, 1992, 521–528.
R. Ødegård, On the solubility and electrochemcial behaviour of aluminium and aluminium carbide in cryolitic melts, Institutt for teknisk elektrokjemi, Universitetet i Trondheim, Norges Tekniske Høgskole, Trondheim, 1985.
P. Rafiei, F. Hiltmann, M. Hyland, B. James, B. Welch, “Electrolytic degradation within cathode materials,” Light Metals, 2001, 747–752.
D.R. Gabe, “Review — the rotating cylinder electrode,” Journal of Applied Electrochemistry, 4, 1974, 91–108.
D.R. Gabe, F.C. Walsh, “The rotating cylinder electrode — a review of development,” Journal of Applied Electrochemistry, 13, 1983, 3–22.
J. Thonstad, P. Fellner, G.M. Haarberg, J. Hives, H. Kvande, Å. Sterten, Aluminium Electrolysis, 3rd ed., Luminium-Verlag -Marketing & Kommunikation GmbH, Düseldorf, 2001.
J. Thonstad, S. Rolseth, “On the cathodic overvoltage on aluminium in cryolite-alumina melts -II;” Electrochimica Acta, 23, 1978, 233–241.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 TMS (The Minerals, Metals & Materials Society)
About this chapter
Cite this chapter
Wang, Z. et al. (2016). Cathode Wear in Electrowinning of Aluminum Investigated by a Laboratory Test Cell. In: Williams, E. (eds) Light Metals 2016. Springer, Cham. https://doi.org/10.1007/978-3-319-48251-4_151
Download citation
DOI: https://doi.org/10.1007/978-3-319-48251-4_151
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-48615-4
Online ISBN: 978-3-319-48251-4
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)