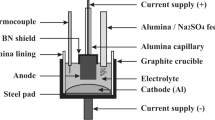
Abstract
The effects of anode current density, electrolysis temperature and KF addition in electrolyte on the consumption of prebaked anode during aluminum electrolysis was investigated in a closed laboratory cell. The current density varied from 0.75 to 1.1 A/cm2, the electrolysis temperature varied from 930 to 960 °C with modified electrolyte composition. KF content varied from 0 to 4 wt% in the electrolyte of the industrial composition. Electrolysis durations was 2 h, the distance between the electrodes was 35 mm. The total consumption of anode, the amount of carbon dust and the amount of carbon gasification were determined under the operating conditions similar to the industrial application. It is established that the increased current density and the KF addition reduces the anode consumption, but the consumption increases again when the current density is more than 0.9 A/cm2. The anode consumption increases with increasing electrolysis temperature.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
References
H.F. Xu, et al., “Baked anode quality improvement through optimization of green anode processing,” Light Metals 2012, 1169–1172.
A. Zarouni, et al., “DX+, an Optimized Version of DX Technology,” Light Metals 2012, 697–702.
Z. Kuang, et al., “Effect of Backing Temperature and Anode Carrent Density on Anode Carbon Consumption,” Metallurgical and Materials Transactions B, 27B (1996), 177–183.
W.K. Fischer, F. Keller, R. Perruchoud, “Interdependence between Anode Net Consumption and Pot Design, Pot Operating Parameters and Anode Properties”, Light Metals 1991, 681–686.
Cutshall E R, “Influence of Anode Baking Temperature and Current Density upon Carbon Sloughing,” Light Metals 1986, 629–635.
S.M. Hume, et al., “The Influence of Low Current Densities on Anode Performance,” Light Metals 1992, 687–692.
A.N. Smordinov, M.A. Korobov, “Relationship between Anodic Paste Consumption and Geometric Dimension of Aluminum Electrolysis Cells,” Soviet Journal of Non-ferrous Metals, 13 (9)(1970), 27–32.
A. Revazyan, “Anode Carbon Consumption of Industrial Aluminum Electrolyzer,” Soviet Journal of Non-ferrous Metals, 3 (8)(1960), 27–32.
P.J. Rhedey, “Carbon Reactivity and Aluminum Reduction Cell Anodes,” Light Metals 1982, 713–721.
A.M. Vinogradov, et al., “An Investigation of The Effect of The Electrolyte Composition on The Consumption of Fired Anodes during Electrolysis Aluminum Production,” Russian Journal of Non-Ferrous Metals, 5 (49)(2008), 346–351.
J. Rafael, et al., “Boron Salt Inhibitors of Air Reactivity of Prebaked Carbon Anodes-Literature Review and Laboratory Studies,” Light Metals 2009, 1173–1176.
A. Silny´, et al., “Carbon Consumption during Laboratory Aluminum Electrolysis,” Canadian Metallurgical Quarterly, 45 (3)(2006), 275–282.
X. Li, J. Xue, and T. Chen, “Characterization of porous structure and its correlation to sodium expansion of graphite cathode materials using image analysis,” Light Metals 2013, 1263–1267.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 TMS (The Minerals, Metals & Materials Society)
About this chapter
Cite this chapter
Feng, L. et al. (2015). Effects of Current Density and Temperature on Anode Carbon Consumption in Aluminum Electrolysis. In: Hyland, M. (eds) Light Metals 2015. Springer, Cham. https://doi.org/10.1007/978-3-319-48248-4_194
Download citation
DOI: https://doi.org/10.1007/978-3-319-48248-4_194
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-48610-9
Online ISBN: 978-3-319-48248-4
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)