Abstract
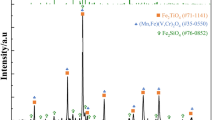
In order to seek an effective extraction process for vanadium, the recovery of vanadium from a high Ca/V ratio vanadium slag was studied by sodium roasting and ammonia leaching. In the present paper, the oxidation and leaching process of vanadium slag was investigated by X-ray diffraction (XRD), scanning electron microscopy and energy dispersive X-ray spectrometry (SEM/EDS) techniques. The effects of ammonium carbonate concentration, leaching temperature and leaching time on the leaching ratio of vanadium were discussed. As indicated in the experimental result, the optimal (NH4)2CO3 concentration was 120g/L, leaching temperature was 60°C and leaching time was 20 min. Approximately 92% of the vanadium was recovered under the optimal conditions. Furthermore, by means of X-ray diffraction analysis, the phase transformations of the vanadium slag during roasting and leaching processes were analyzed and discussed.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
References
R.R. Moskalyk and A.M. Alfantazi. Processing of vanadium: a review[J]. Minerals Engineering, 2003, 16(9): 793–805.
P.R. Taylor, et al. Extractive metallurgy of vanadium-containing titaniferous magnetite ores: A review[J]. Minerals and Metallurgical Processing, 2006, 23(2): 80–86.
A. Parasuraman, et al. Review of material research and development for vanadium redox flow battery applications [J]. Electrochimica Acta, (0).
W. You. The Resources of Vanadium and Its Applications, Development and Outlook[J]. Rare metals and cemented carbides, 1996, 01: 51–55. (in Chinese)
B. Raja. Vanadium market in the world[J]. Steelworld, 2007, 13(2): 19–22.
B.C. Jena, W. Dresler, and I.G. Reilly. Extraction of Titanium, Vanadium and Iron from Titanomagnetite Deposits at Pipestone Lake, Manitoba, Canada[J]. Minerals Engineering, 1995, 8(1–2): 159–168.
D. Chen, et al. A novel process for recovery of iron, titanium, and vanadium from titanomagnetite concentrates: NaOH molten salt roasting and water leaching processes[J]. Journal of Hazardous Materials, 2013, 244–245(0): 588–595.
H. Daoxin. Vanadium Extraction and Steelmaking[M]. Beijing, China: Metallurgical Industry Press, 2000. (in Chinese)
A. Mahdavian, et al. Recovery of Vanadium from Esfahan Steel Company Steel Slag; Optimizing of Roasting and Leaching Parameters [J]. International Journal of Iron and Steel Society of Iran, 2006.
L. Shiming. The metallurgy of vanadium abroad[M]. Beijing, China: Metallurgical Industry Press, 1985. (in Chinese)
H.S. Chen. Extraction of vanadium pentoxide from vanadium slag by lime calcination[J]. Iron Steel Vanadium Titanium, 1992, 13(6): 1–9. (in Chinese)
Y. JingLing. A new way of recovering vanadium from iron/vanadium slag[J]. Journal of Beijing University of Chemical Technology, 2007, 34(3): 254–257. (in Chinese)
Z.L. Li, et al. Acid Leaching of Calcined Vanadium Titanomagnetite with Calcium Compounds for Extraction of Vanadium [J]. The Chinese Journal of Process Engineering, 2011, 11(4): 573–578.
X. Zhang, et al. Nucleation and growth kinetics of spinel crystals in vanadium slag[J]. Ironmaking & Steelmaking, 2012, 39(2): 147–154.
H.A. Barolin. Oxidation of Vanadium Slag[M]. Beijing, China: Metallurgical Industry Press, 1982.
Song Xu, et al. Study on Roasting Parameters for Extraction of Vanadium from a High Ca/V Ratio Vanadium Slag[J]. Metalurgia International, 2013, 18(8): 38–40.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 TMS (The Minerals, Metals & Materials Society)
About this paper
Cite this paper
Xu, S., Long, M., Chen, D., Fan, H., Chen, Y., Sun, X. (2014). Recovery of Vanadium from a High Ca/V Ratio Vanadium Slag Using Sodium Roasting and Ammonia Leaching. In: Mackey, P.J., Grimsey, E.J., Jones, R.T., Brooks, G.A. (eds) Celebrating the Megascale. Springer, Cham. https://doi.org/10.1007/978-3-319-48234-7_61
Download citation
DOI: https://doi.org/10.1007/978-3-319-48234-7_61
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-48591-1
Online ISBN: 978-3-319-48234-7
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)