Abstract
The concept of oocyte quality represents the oocyte developmental competence, meaning the intrinsic ability of oocytes to undergo meiotic maturation, fertilization, proper embryonic development and successful pregnancy. As follicular growth disturbances have been widely shown in PCOS, especially during controlled ovarian hyperstimulation, the consecutive endocrine and/or paracrine follicular microenvironment modifications could have detrimental effects on the oocyte quality in PCOS. Moreover, the paucity of clinical studies focusing on oocyte quality in PCOS women makes definitive conclusions on this topic difficult and controversial. In this chapter, we will firstly focus on the three main approaches, morphological, genetic and OMICS, which have been proposed to investigate oocytes from PCOS. Secondly, we will look over the clinical relevance of all the compiled results from these studies by examining the final oocyte competence during the IVF process in PCOS patients.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Polycystic ovarian morphology (PCOM) and polycystic ovary syndrome (PCOS) are very common findings in an in vitro fertilization (IVF) centre population, explaining why the concept of oocyte quality is so challenging. Indeed, 18–25% of the infertile couples meet the diagnosis criteria for PCOS ([1]; http://www.ivf-worldwide.com/survey/pcos-results.htm), whereas the prevalence of PCOM has been estimated as high as 33% in asymptomatic patients [2,3,4]. With the advent of highly sensitive ultrasound machines, the PCOM, mainly based on the antral follicle count, has become one of the main diagnosis criteria of PCOS and is now well recognized to be the common base of the wide spectrum of clinical, hormonal and metabolic phenotypes of the PCOS. PCOM is characterized by a significantly enlarged cohort of early growing and recruitable follicles. This excessive follicle number is linked to disturbances in folliculogenesis which are thought to be the consequence of intra-ovarian hyperandrogenism [5,6,7] (see also Chaps. 2 and 8). During controlled ovarian hyperstimulation (COH), the cohort of growing follicles is frequently heterogeneous in size, with mature, intermediary and small follicles. In parallel, the number and quality of mature oocytes has been proposed as being poor [6, 8, 9] leading to lower pregnancy rates and higher abortion rates. Furthermore, recent data suggested that oocyte competence could be impaired in PCOS patients due to an inadequate dialogue between the cumulus cells and the oocyte and an impairment of the follicular microenvironment [10, 11].
Despite these assumptions, the paucity of clinical studies focusing on oocyte quality in PCOS women does not allow to make definitive conclusions. Most of these studies are retrospective, concerning PCOS patients diagnosed with heterogeneous criteria and with various clinical and metabolic phenotypes with a historical control group [12,13,14,15,16,17] (see also Chaps. 2 and 7).
Mostly, oocyte quality was only extrapolated through fecundation, implantation and pregnancy rates according to the number of mature oocytes, i.e. metaphase II oocytes [12,13,14,15,16,17]. Consequently, the results provided by these studies are conflicting, reporting either a better oocyte/embryo quality and pregnancy rates or vice versa.
The concept of oocyte quality represents the oocyte developmental competence, meaning the intrinsic ability of oocytes to undergo meiotic maturation, fertilization, proper embryonic development and successful pregnancy [18]. These competences are progressively acquired during the follicular development through the cross-talk between oocyte and somatic/surrounding granulosa cells (GCs) [19]. As follicular growth disturbances have been widely shown in PCOS, especially during COH, it has been postulated that the consecutive endocrine and/or paracrine follicular microenvironment modifications could have detrimental effects on the oocyte quality.
In this review, we will firstly focus on the main approaches proposed to investigate oocytes from PCOS, and then we will look over the clinical relevance of all the compiled results from these studies by examining the final oocyte competence during the IVF process in PCOS patients.
2 Evaluation of Oocyte Quality
Evaluating the oocyte quality is obviously complicated because of the few number of oocytes retrieved during the IVF process. Hence, there is various ways, invasive or non-invasive, direct or indirect, to investigate the oocyte competence. Three main approaches, morphological, genetic and OMICS, have been proposed to investigate oocytes from PCOS.
2.1 Morphological Approach
After having removed the cumulus-corona cells in preparation for intracytoplasmic injection (ICSI), oocyte evaluation is based on the nuclear maturation status, the morphology of the cytoplasm and on the appearance of the extracytoplasmic structures. A higher number but a same rate of metaphase II oocytes between PCOS patients and controls were highlighted in various retrospective studies [12,13,14], as well as a higher number but a same rate of top-quality embryos [13, 14]. These results were confirmed in the meta-analysis of Heijnen et al. and in a recent prospective study comparing PCOM patients versus non-PCOM [20, 21]. Despite being the main morphological indicator, nuclear maturity examination alone is not enough to determine the quality of an oocyte: the nucleus and the cytoplasm have to mature in synergy in order to reach the conditions for an optimal fertilization rate. In addition, it is now well recognized that some specific morphological oocyte abnormalities, such as the presence of a wide perivitelline space (PVS) or a granular cytoplasm, must be given attention since it has been reported that they are associated with a significant decrease in the chance of fertilization [22, 23]. Only three studies are interested in oocyte morphology in PCOS patients. Sahu et al. [13] reported similar oocyte morphology in isolated PCOM, PCOS and age-matched controls. Sigala et al. [21] performed a prospective comparative study with a systematic examination of oocyte morphology in PCOM versus non-PCOM patients. Nuclear maturation, extra- and intracytoplasmic oocyte abnormalities were assessed, i.e. fragmented or abnormal first polar body (IPB), abnormal zona pellucida (ZP), presence of an enlarge perivitelline space (PVS) or material in the PVS and an abnormal shape of the oocyte [21]. No specific morphological abnormalities as well as no difference regarding the incidence of these abnormalities were observed in the PCOM versus non-PCOM patients. In this study, PCOM was defined according to the threshold of 19 follicles per ovary. Among the PCOM group, there were 52.5% of PCOS and 47.5% of PCOM only. No difference was observed between oocytes from PCOS and PCOM-only patients, but it has to be mentioned that obese PCOM patients were excluded [21]. Piomboni et al. [24] compared the oocyte quality based on the precited morphologic criteria in three groups of nonobese PCOS patients: PCOS treated by d-chiro-inositol, PCOS treated by metformin and non-treated PCOS. They showed a significantly higher number of good-quality oocytes in the groups treated by insulin-sensitizing agents, as well as they highlighted in parallel a significant reduction of reactive oxygen species production in the follicular fluid [24]. Data on the effect of inositol on oocyte quality are extensively reviewed in Chap. 16.
A few studies using polarized light microscopy (PLM) have shown, by highlighting meiotic spindle abnormalities, that some of metaphase II oocytes may still be immature [18, 25]. Indeed, meiotic spindle, when detectable with polscope microscope, is not always aligned with the first polar body (PB1) in fresh metaphase II oocytes, which may adversely affect the fertilizing ability and in vitro/in vivo developmental competence [25]. In addition, it has recently been shown that oocytes with normal spindle morphology are significantly more likely to produce euploid embryos [26]. Only two studies are interested in the spindle and chromosome configurations in oocytes from PCOS patients. Li et al. [27] compared the incidence of abnormality in spindle and chromosomal configurations in both in vitro and in vivo-matured oocytes. A higher rate of abnormalities was found in the group of in vitro-matured oocytes [27]. Vieira et al. compared in vitro-matured oocytes from PCOS versus non-PCOS patients and didn’t find any difference [28]. To the point of view of the authors, it is more likely that the in vitro conditions of oocyte maturation might explain these meiotic abnormalities rather than the PCOS itself.
2.2 Genetic Approach
The decrease of fertilization rate and the increase in pregnancy loss in certain subgroups of PCOS patients have led to the hypothesis that oocyte and embryos could be of poorer quality due to a higher aneuploidy rate. Morphological examination is insufficient to detect genetic abnormalities such as aneuploidy. Currently, the only method yielding a definitive evaluation of oocytes/embryos is aneuploidy diagnosis, which provides information of normal or abnormal chromosomal constitution. Moreover, aneuploidy detection requires breach of the zona pellucida and biopsy of the polar bodies from the oocyte that is obviously invasive. Because of the difficulty to directly evaluate the ploidy of the oocytes, some studies investigated indirectly the genetic potential of the PCOM gamete. Sengoku et al. [29] performed cytogenetic analysis on the unfertilized oocytes with normal morphology from PCOS and control patients but didn’t find any difference in the incidence of aneuploidy or diploidy [29]. Weghofer et al. [30] examined the association between PCOS and embryonic aneuploidy. They compared the results of preimplantation genetic diagnosis (PGD) between properly documented PCOS patients and controls. Despite a statistically higher absolute number of euploid embryos in PCOS group due to the higher number of metaphase II oocytes retrieved, there was no difference regarding the rate of aneuploidy [30]. Wang et al. [31] conducted a prospective cohort study by performing genetic analysis on abortuses from PCOS and non-PCOS patients who conceived after IVF. The aneuploidy rate was not significantly higher in the PCOS group but significantly lower in comparison with the non-PCOS patients [31].
2.3 OMICS Approach
The emergence of the—OMICS technologies, i.e. epigenomics, genomics, transcriptomics, proteomics and metabolomics, provides a huge amount of new information regarding the biological processes involved in the reproductive field. Although using an invasive technology, microarray-based transcription profiles of oocytes at various stage of growth and maturation have led to a better understanding of the genes expressed during oocyte development: disruption of transcription within an oocyte or any modification of their adequate transcriptomes could compromise its growth and development, as well as the resulting embryo, since oocyte mRNAs pool is strongly correlated with the ability to develop into the blastocyst stage [32].
Recent cluster of analysis revealed differences in global gene expression profile between normal and PCOS tissues and oocytes [33]. Wood et al. [34] identified 374 genes with different mRNA transcript levels when analyzing metaphase II oocytes from normal responders and PCOS patients. A subset of these genes found to be differentially expressed in PCOS is involved in spindle dynamics, homologous recombination/chromosome alignment, cell cycle checkpoint and centrosome function during mitosis and/or meiosis [34]. Furthermore, some of the other differentially expressed genes contain putative androgen receptors and/or peroxisome proliferating Y binding sites [34]. The authors make the assumption that these observations could be related to a lower oocyte quality.
Cai et al. [35] interested in the in vitro effects of overexpression of Hsp27 on oocyte maturation and development derived from PCOS patients as emerging evidence indicates this heat-shock protein has strong antiapoptotic properties and has been shown to be mainly expressed in human oocytes. Interestingly, the same team has previously shown that Hsp27 was downregulated in ovarian tissue and in oocytes isolated from women with PCOS [36]. The results of upregulation of Hsp27 expression were a lowered oocyte maturation rate, similar fertilization but high-quality embryo blastocyst formation rates leading the authors to postulate that Hsp27 could be involved in the apoptotic imbalance of the oocytes via growth and differentiation factor 9 (GDF9) and bone morphogenic protein (BMP15) [36].
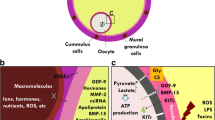
It is now recognized that there is a continuous bidirectional cross-talk between oocytes and somatic cells during folliculogenesis through gap junctions and paracrine signalling [19]. If it is sure that oocyte is nurtured and supported by the closely associated somatic cells, especially those of the cumulus, the fact remains that oocyte itself plays an active role via secretion of paracrine signalling factors, such as GDF9 and BMP15, which maintain an appropriate microenvironment for a proper follicular growth [19, 33]. It is now possible to identify the transcriptome of GCs with the microarray technology, and evidence supporting GCs genes markers as valuable and non-invasive predictors of oocyte competence is rapidly emerging [37].
As folliculogenesis disturbances frequently occur in PCO patients, it seemed logical to investigate this oocyte/granulosa cells dialogue. Ouandaogo et al. [38] compared the transcriptome profiles of cumulus cells (CCs) isolated from in vivo and IVM cumulus-oocyte complexes (COCs) at different nuclear maturation stages from normal responders and PCOS patients undergoing ICSI following in vivo or IVM. In the PCOS subgroup, the authors found a strong alteration of the expression profile of the CCs derived from IVM metaphase II oocytes in comparison to in vivo metaphase II oocytes [37]. The expression profile also differed significantly between normal and PCOS patients, but the authors conclude that these significant differences were related to the culture condition, not to the PCOS per se [37]. The same team focused few months later on the gene expression profile in CCs of in vivo metaphase II oocytes from PCOS and non-PCOS patients using the DNA microarray technology [39]. There were significant differences between groups in the gene expression profile. In addition, CCs from PCOS patients were characterized by abnormal expression of many growth factors, including members of the epidermal growth factor-like and IGF-like families that are known to play a role in oocyte competence [39]. Likewise, mRNA transcripts of factors involved in steroid metabolism seem to be deregulated in PCOS CCs [39].
miRNAs are small, non-coding RNAs detected in biological fluids that are able to regulate gene expression at the post-transcriptional level and which may be involved in reproductive function [40]. A limited number of studies have aimed to extensively profile circulating microRNAs (miRNAs) expression and function in the follicular fluid within a PCOS study population, and the results are at times not yet conclusive [40, 41]. Both highlight different miRNA expression between PCOS and non-PCOS patients but with no clear correlation with oocyte maturation or fertilization competence [40, 41].
3 Clinical Data
Research on oocyte is extremely complex, especially in PCOS patients undergoing IVF. Firstly, the fact that PCOS patients need IVF, independently of tubal or sperm alterations, introduces some recruitment bias. Secondly, the high ovarian response under COH in these patients may have detrimental effects on the oocyte quality through vascular and inflammatory factors. Likewise, the in vitro/in vivo culture conditions may play a major role in the oocyte quality. PCOS underlies endocrine, ovulatory and/or metabolic dysfunction. These three components are solely or synergistically strong confounders regarding the interpretation of the studies on oocyte quality. Indeed, the source of oocytes is highly variable according to the different PCOS phenotypes, from the lean PCOM-only patient to the obese PCOS patient. In addition, the question as to whether asymptomatic women with PCOM constitute a heterogeneous population in terms of ovarian dysfunction ranging from entirely healthy ovulatory women to women with mild occult PCOS [2] or, alternatively, constitute a homogenous population representing the milder end of the PCOS spectrum remains debatable. More data about PCOS phenotypes are discussed in Chaps. 2 and 7.
Undoubtedly, oocytes from PCOM in IVF condition exhibit molecular specificities in comparison with oocytes from non-PCOM patients. But surely, the key point is to have sufficient oocytes of quality to give substantial chances of pregnancy. One has to recognize that the larger prospective studies regarding the IVF results in PCOS failed to highlight poor results in terms of pregnancy chances, at least in nonobese patients.
Heijnen et al. [20] reported in a meta-analysis the comparative IVF outcome of PCOS patients defined by the Rotterdam criteria to the one of matched non-PCOS controls. Except the higher number of oocytes in PCOS patients, they failed to find any difference between fertilization, pregnancy and take baby home rates between groups [20]. Likewise, Sigala et al. [21] in a large prospective comparative study have shown the same rate of metaphase II and morphologically normal oocytes in nonobese PCOM versus nonobese non-PCOM patients. The rate of top-quality embryo was equivalent in the two groups while the implantation and clinical pregnancy rates were even better in the PCOM group [21]. These results were also confirmed after having divided the PCOM group in PCOS and “sonographic-only” PCOM. Engmann et al. [15] previously reported same results in PCOM patients. Hence, the bad prognosis frequently argued regarding pregnancy rates and/or abortion risk could be more related to the metabolic profile than to PCOM per se. It is well known that high body mass index (BMI) and hyperinsulinaemia are main contributors to the follicular microenvironment disturbances [9]. Indeed, studies in follicular fluids from PCOS patients highlight high levels of interrelated endocrino-metabolic factors such as androgens, VEGF, AMH, insulin and IGF, all of them playing an active role in the oocyte-CCs dialogue [9]. Moreover, numerous studies highlighted benefits from taking insulin-sensitizing agents like metformin as a co-treatment before and during the IVF process [42, 43]. The use of insulin-sensitizing drugs in PCOS is reviewed in Chap. 11.
From another point of view, as mentioned above, it seems that the meiotic/mitotic cell cycle pathway is altered in PCOS oocytes [34], but no more aneuploid oocytes were detected in PCOS patients [29] as well as no more aneuploidy embryos were observed [30]. It is therefore difficult to make the connection between such molecular specificities and the reality of the clinical experience.
Conclusion
It remains difficult to conclude about the oocyte competence in PCOS. Molecular specificities have been properly highlighted in PCOS oocytes, but it doesn’t mean that there are abnormalities in their reproductive competence. Too few studies have taken into account the very wide spectrum of PCOS phenotypes and the potential influence of comorbidity factors such as obesity and insulin resistance in their analysis. However, the largest prospective studies performed in nonobese PCOM patients so far are in favour to good pregnancy chances due to a high number of good-quality oocytes. The coexistence of a metabolic syndrome to PCOM seems to impair the oocyte competence and the pregnancy rates. Prospective studies with consensual international diagnosis criteria are needed to allow in better understanding of the exact molecular mechanisms involved in the oocyte competence according to each phenotype of PCOS and would elucidate if the PCOS oocyte deserve its bad reputation.
References
Hull MG, Glazener CM, Kelly NJ, Conway DI, Foster PA, Hinton RA, et al. Population study of causes, treatment, and outcome of infertility. Br Med J. 1985;291:1693–7.
Mortensen M, Ehrmann DA, Littlejohn E, Rosenfield RL. Asymptomatic volunteers with a polycystic ovary are a functionally distinct but heterogeneous population. J Clin Endocrinol Metab. 2009;94:1579–86.
Kim YJ, Ku S-Y, Jee BC, Suh CS, Kim SH, Choi YM, et al. A comparative study on the outcomes of in vitro fertilization between women with polycystic ovary syndrome and those with sonographic polycystic ovary-only in GnRH antagonist cycles. Arch Gynecol Obstet. 2010;282:199–205.
March WA, Moore VM, Willson KJ, Phillips DIW, Norman RJ, Davies MJ. The prevalence of polycystic ovary syndrome in a community sample assessed under contrasting diagnostic criteria. Hum Reprod. 2010;25:544–51.
Jonard S. The follicular excess in polycystic ovaries, due to intra-ovarian hyperandrogenism, may be the main culprit for the follicular arrest. Hum Reprod Update. 2004;10:107–17.
Franks S, Stark J, Hardy K. Follicle dynamics and anovulation in polycystic ovary syndrome. Hum Reprod Update. 2008;14:367–78.
Homburg R. Androgen circle of polycystic ovary syndrome. Hum Reprod. 2009;24:1548–55.
Dumesic D, Abbott D. Implications of polycystic ovary syndrome on oocyte development. Semin Reprod Med. 2008;26:53–61.
Qiao J, Feng HL. Extra- and intra-ovarian factors in polycystic ovary syndrome: impact on oocyte maturation and embryo developmental competence. Hum Reprod Update. 2010;17:17–33.
Kenigsberg S, Bentov Y, Chalifa-Caspi V, Potashnik G, Ofir R, Birk OS. Gene expression microarray profiles of cumulus cells in lean and overweight-obese polycystic ovary syndrome patients. Mol Hum Reprod. 2008;15:89–103.
Kwon H, Choi D-H, Bae J-H, Kim J-H, Kim Y-S. mRNA expression pattern of insulin-like growth factor components of granulosa cells and cumulus cells in women with and without polycystic ovary syndrome according to oocyte maturity. Fertil Steril. 2010;94:2417–20.
Ludwig M, Finas DF, Al-Hasani S, Diedrich K, Ortmann O. Oocyte quality and treatment outcome in intracytoplasmic sperm injection cycles of polycystic ovarian syndrome patients. Hum Reprod. 1999;14:354–8.
Sahu B, Ozturk O, Ranierri M, Serhal P. Comparison of oocyte quality and intracytoplasmic sperm injection outcome in women with isolated polycystic ovaries or polycystic ovarian syndrome. Arch Gynecol Obstet. 2008;277:239–44.
Esinler I, Bayar U, Bozdag G, Yarali H. Outcome of intracytoplasmic sperm injection in patients with polycystic ovary syndrome or isolated polycystic ovaries. Fertil Steril. 2005;84:932–7.
Engmann L, Maconochie N, Sladkevicius P, Bekir J, Campbell S, Tan SL. The outcome of in-vitro fertilization treatment in women with sonographic evidence of polycystic ovarian morphology. Hum Reprod. 1999;14:167–71.
Esmailzadeh S, Faramarzi M, Jorsarai G. Comparison of in vitro fertilization outcome in women with and without sonographic evidence of polycystic ovarian morphology. Eur J Obstet Gynecol Reprod Biol. 2005;121:67–70.
Zhong Y-P, Ying Y, Wu H-T, Zhou C-Q, Xu Y-W, Wang Q, et al. Comparison of endocrine profile and in vitro fertilization outcome in patients with PCOS, ovulatory PCO, or normal ovaries. Int J Endocrinol. 2012;2012:1–6.
Rienzi L, Balaban B, Ebner T, Mandelbaum J. The oocyte. Hum Reprod. 2012;27:2–21.
Li Q, McKenzie LJ, Matzuk MM. Revisiting oocyte-somatic cell interactions: in search of novel intrafollicular predictors and regulators of oocyte developmental competence. Mol Hum Reprod. 2008;14:673–8.
Heijnen EM, Eijkemans MJ, Hughes EG, Laven JS, Macklon NS, Fauser BC. A meta-analysis of outcomes of conventional IVF in women with polycystic ovary syndrome. Hum Reprod Update. 2006;12:13–21.
Sigala J, Sifer C, Dewailly D, Robin G, Bruyneel A, Ramdane N, Lefebvre-Khalil V, Mitchell V, Decanter C. Is polycystic ovarian morphology related to a poor oocyte quality after controlled ovarian hyperstimulation for intracytoplasmic sperm injection? Results from a prospective, comparative study. Fertil Steril. 2015;103:112–8.
Rienzi L, Vajta G, Ubaldi F. Predictive value of oocyte morphology in human IVF: a systematic review of the literature. Hum Reprod Update. 2011;17:34–45.
Setti AS, Figueira RCS, Braga DPAF, Colturato SS, Iaconelli A, Borges E. Relationship between oocyte abnormal morphology and intracytoplasmic sperm injection outcomes: a meta-analysis. Eur J Obstet Gynecol Reprod Biol. 2011;159:364–70.
Piomboni P, Focarelli R, Capaldo A, Stendardi A, Cappelli V, Cianci A, La Marca A, Luddi A, De Leo V. Protein modification as oxidative stress marker in follicular fluid from women with polycystic ovary syndrome: the effect of inositol and metformin. J Assist Reprod Genet. 2014;31:1269–76.
Rienzi L, Ubaldi F, Martinez F, Iacobelli M, Minasi MG, Ferrero S, Tesarik J, Greco E. Relationship between meiotic spindle location with regard to the polar body position and oocyte developmental potential after ICSI. Hum Reprod. 2003;18:1289–93.
Tilia L, Venetis C, Kilani S, Cooke S, Chapman M. Is oocyte meiotic spindle morphology associated with embryo ploidy? A prospective cohort study. Fertil Steril. 2016;105:1085–92.
Li Y, Feng HL, Cao YJ, Zheng GJ, Yang Y, Mullen S, Critser JK, Chen ZJ. Confocal microscopic analysis of the spindle and chromosome configurations of human oocytes matured in vitro. Fertil Steril. 2006;85:827–32.
Vieira RC, Barcelos ID, Ferreira EM, Martins WP, Ferriani RA, Navarro PA. Spindle and chromosome configurations of in vitro-matured oocytes from polycystic ovary syndrome and ovulatory infertile women: a pilot study. J Assist Reprod Genet. 2011;28:15–21.
Sengoku K, Tamate K, Takuma N, Yoshida T, Goishi K, Ishikawa M. The chromosomal normality of unfertilized oocytes from patients with polycystic ovarian syndrome. Hum Reprod. 1997;12:474–7.
Weghofer A, Munne S, Chen S, Barad D, Gleicher N. Lack of association between polycystic ovary syndrome and embryonic aneuploidy. Fertil Steril. 2007;88:900–5.
Wang Q, Luo L, Lei Q, Lin MM, Huang X, Chen MH, Zeng YH, Zhou CQ. Low aneuploidy rate in early pregnancy loss abortuses from patients with polycystic ovary syndrome. Reprod Biomed Online. 2016;33:85–92.
Egea RR, Puchalt NG, Escrivá MM, Varghese AC. OMICS: current and future perspectives in reproductive medicine and technology. J Hum Reprod Sci. 2014;7:73–92.
Assou S, Boumela I, Haouzi D, Anahory T, Dechaud H, De Vos J, Hamamah S. Dynamic changes in gene expression during human early embryo development: from fundamental aspects to clinical applications. Hum Reprod Update. 2011;17:272–90.
Wood JR, Dumesic DA, Abbott DH, Strauss JF 3rd. Molecular abnormalities in oocytes from women with polycystic ovary syndrome revealed by microarray analysis. J Clin Endocrinol Metab. 2007;92:705–13.
Cai L, Ma X, Liu S, Liu J, Wang W, Cui Y, Ding W, Mao Y, Chen H, Huang J, Zhou Z, Liu J. Effects of upregulation of Hsp27 expression on oocyte development and maturation derived from polycystic ovary syndrome. PLoS One. 2013;8:e83402.
Ma X, Fan L, Meng Y, Hou Z, Mao YD, Wang W, Ding W, Liu JY. Proteomic analysis of human ovaries from normal and polycystic ovarian syndrome. Mol Hum Reprod. 2007;13:527–35.
Ouandaogo ZG, Frydman N, Hesters L, Assou S, Haouzi D, Dechaud H, Frydman R, Hamamah S. Differences in transcriptomic profiles of human cumulus cells isolated from oocytes at GV, MI and MII stages after in vivo and in vitro oocyte maturation. Hum Reprod. 2012;27:2438–47.
Haouzi D, Assou S, Monzo C, Vincens C, Dechaud H, Hamamah S. Altered gene expression profile in cumulus cells of mature MII oocytes from patients with polycystic ovary syndrome. Hum Reprod. 2012;27:3523–30.
Fragouli E, Lalioti MD, Wells D. The transcriptome of follicular cells: biological insights and clinical implications for the treatment of infertility. Hum Reprod Update. 2014;20:1–11.
Sørensen AE, Wissing ML, Salö S, Englund AL, Dalgaard LT. MicroRNAs related to polycystic ovary syndrome (PCOS). Genes (Basel). 2014;5:684–708.
Scalici E, Traver S, Mullet T, Molinari N, Ferrières A, Brunet C, Belloc S, Hamamah S. Circulating microRNAs in follicular fluid, powerful tools to explore in vitro fertilization process. Sci Rep. 2016;6:24976.
Palomba S, Falbo A, La Sala GB. Effects of metformin in women with polycystic ovary syndrome treated with gonadotrophins for in vitro fertilisation and intracytoplasmic sperm injection cycles: a systematic review and meta-analysis of randomised controlled trials. BJOG. 2013;120:267–76.
Tso LO, Costello MF, Albuquerque LE, Andriolo RB, Macedo CR. Metformin treatment before and during IVF or ICSI in women with polycystic ovary syndrome. Cochrane Database Syst Rev. 2014;11:CD006105.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Decanter, C. (2018). Oocyte Quality in PCOS. In: Palomba, S. (eds) Infertility in Women with Polycystic Ovary Syndrome. Springer, Cham. https://doi.org/10.1007/978-3-319-45534-1_4
Download citation
DOI: https://doi.org/10.1007/978-3-319-45534-1_4
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-45533-4
Online ISBN: 978-3-319-45534-1
eBook Packages: MedicineMedicine (R0)