Abstract
Ventricular remodeling is a complex process that results from interactions between the initial myocardial injury and neurohormonal factors that are capable of modifying the cardiomyocyte phenotype and of inducing changes in the extracellular matrix. At the cellular level, myocyte hypertrophy, necrosis, apoptosis, interstitial fibrosis, and degradation of collagen are the major features of myocardial remodeling. Each of these components of the remodeling process contributes importantly to the development and progression of heart failure (HF). At the level of the ventricular chamber, remodeling is associated with progressive ventricular hypertrophy, enlargement, and cavity distortion that is directly related to deterioration in ventricular performance and to long-term adverse clinical outcomes.
The mechanisms responsible for ventricular remodeling and deterioration of ventricular structure and function are not entirely clear, but are related to neurohormonal and cytokine activation. These factors in combination with increased wall stress and mechanical stretch of the myocytes upregulate a large number of signaling pathways. This leads to structural and functional changes in the myocyte and nonmyocyte compartments, contributing to ventricular dysfunction and the progression of HF.
Although, initially, it may be compensatory in certain pressure and volume-overload conditions, progressive ventricular remodeling is ultimately a maladaptive process. After myocardial infarction, however, progressive hypertrophy and remodeling of noninfarcted myocardium may be harmful from the start.
Ventricular remodeling has emerged as an important therapeutic target and a credible surrogate end point in HF. Agents that have beneficial effects in HF also generally attenuate or reverse ventricular remodeling, whereas agents that fail to improve clinical outcomes either have no effect on remodeling or have been associated with adverse remodeling. An important goal in the treatment of HF is to slow or reverse remodeling to improve long-term outcome.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
- Heart failure
- Mechanisms
- Ventricular remodeling
- Reverse remodeling
- Myocyte hypertrophy
- Extracellular matrix
Heart failure (HF) progresses through a process of structural remodeling of the heart to which neurohormonal and cytokine activation makes an important contribution. The term ventricular remodeling refers to deviation in ventricular architecture from normal, with changes in volume, wall thickness, and/or shape. This term was initially applied to the pathologic changes related to myocardial hypertrophy, fibrosis, and associated chamber dilation seen following a large myocardial infarction (MI) [1–3]. The term has also been used in other conditions associated with ventricular dilation and eccentric myocardial hypertrophy, referred to as dilated cardiomyopathy, and to conditions associated with concentric left ventricular (LV) hypertrophy with a normal or reduced chamber volume, as is seen in hypertensive heart disease. A large body of evidence now indicates that these forms of pathologic ventricular remodeling are independently associated with adverse clinical outcomes and, more importantly, that interventions that attenuate or reverse these changes are usually associated with improved clinical outcomes [4].
Remodeling Concept of Heart Failure
It is well known that the heart can enlarge or shrink in response to hemodynamic demands (◘ Fig. 17.1) [5]. Critical to our understanding of HF are observations that HF is related to progressive alterations in the heart’s structure and function. The earliest reference to the role of cardiac structure in development of HF dates back to the nineteenth century [6]. In The Principles and Practice of Medicine, William Osler pointed to hypertrophy as a step in the development of HF, since it is followed by a “period of broken compensation … that commonly takes place slowly and results from degeneration and weakening of the heart muscle” [7]. However, in the modern era, Linzbach has been credited for being the first to recognize that alterations in cardiac structure are the primary determinants of HF and that LV weight of about 200 g was critical in the natural history of the disorder [8].
Conditions leading to remodeling of the heart and resulting in atrophy or hypertrophy. Depending on the circumstances, remodeling can be normal or pathologic. Pathologic remodeling is associated with a propensity toward decompensation, ventricular dilatation, systolic dysfunction, and electrophysiologic changes leading to malignant ventricular arrhythmia. Source: Hill JA, Olson EN. Cardiac plasticity. N Engl J Med. 2008;358:1370–80
In the 1960s, a different view of LV hypertrophy and enlargement began to emerge. In accordance with Laplace’s law, which dictates that afterload-induced increases in systolic wall stress are offset by increases in wall thickness, hypertrophic growth of the heart was seen as “compensatory” and, hence, beneficial [9, 10]. Animal models of pressure overload led Meerson to suggest that cardiac growth induced by biomechanical stress plays a protective role, at least in the short term [11]. Moreover, in the 1970s and 1980s, hemodynamic measurements in patients with valvular heart disease provided support for the concept of adaptive hypertrophic growth which, when “inadequate,” could lead to systolic dysfunction [12–14].
Recent clinical studies have called into question the idea that structural changes of the ventricle are adaptive and protective. Progressive LV hypertrophy, enlargement, and cavity distortion over time have consistently been shown to be directly related to the deterioration of LV performance and an increase in mortality and morbidity [15–19], irrespective of the etiology of HF [20].
Current concepts of ventricular remodeling are largely derived from studies on patients and animal models of MI and hypertension [21–24]. Studies by Chanutin and Barsdale [25] on an experimental model of arterial hypertension demonstrated that LV weight and myocyte fiber diameter increased in relation to the severity of hypertension. Janice Pfeffer and her colleagues [26, 27] studied the relationship between LV mass and function over time, in the spontaneously hypertensive rat model. They demonstrated that despite continuous and marked LV wall thickening, the LV eventually dilates and then fails. At this stage, the stimulus for LV hypertrophy is not only elevated arterial pressure but also chamber dilatation that further aggravates the hemodynamic load by increasing wall stress. This seminal finding laid the foundation for the concept that regardless of the initial insult, ventricular dilatation may become a self-sustaining process of deterioration in LV structure and function.
A consensus statement helped define remodeling as the “genomic expression resulting in molecular, cellular and interstitial changes that are manifested clinically as changes in size, shape and function of the heart after cardiac injury” [28]. Remodeling process is regulated by mechanical, genetic, and neurohormonal factors [29]. The importance of ventricular remodeling has increased with the observation that agents such as inhibitors of the sympathetic and renin-angiotensin-aldosterone systems that have beneficial effects in HF also generally attenuate or reverse ventricular remodeling [30–34], whereas agents that fail to improve clinical outcomes either have no effect on remodeling or have been associated with adverse remodeling [4]. Ventricular remodeling has therefore emerged as a credible surrogate end point and an important therapeutic target in HF [35, 36].
Mechanisms of Left Ventricular Remodeling
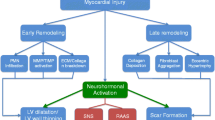
Although ventricular remodeling may occur following any form of myocardial injury [20], most of our knowledge has been acquired from the study of remodeling following MI. Acute coronary occlusion in the clinical setting or in the experimental animal leads to loss of myocardial tissue, depression of myocardial function, and hypotension. This causes baroreceptor-mediated activation of a number of neurohormones that help stabilize the hemodynamics through an increase in heart rate, contractility, and fluid retention. However, continuous activation of these mechanisms, designed for short-term support of blood pressure [37], may lead to progressive LV remodeling and dysfunction. Two distinct phases have been identified following MI: early postinfarct LV remodeling and late progressive LV remodeling.
Early Postinfarct Left Ventricular Remodeling
Loss of regional wall function after acute MI results in an abrupt increase in loading conditions of the ventricle that brings on a unique pattern of remodeling involving the infarct area, the border zone, and the remote noninfarcted myocardium. Thinning and stretching of the acutely infarcted myocardium lead to infarct expansion, the first feature of LV remodeling [21, 22]. Although later thinning of the LV wall also occurs in the noninfarcted myocardium, the cellular mechanisms are different in the two regions. In the infarcted myocardium, wall thinning is pronounced and is a result of loss of myocytes, collapse of the intercellular space, and stretching of surviving myocytes [21–23, 38, 39]. This may lead to bulging of the infarct zone that can result in ventricular rupture, aneurysm, mitral insufficiency, and ventricular tachyarrhythmias. In the noninfarcted regions, myocardium thins because of a decrease in the number of myocytes across the wall [38, 39]. Two mechanisms—myocyte slippage [38] and myocyte loss from necrosis [40–42] and apoptosis [41, 43]—have been proposed to explain this decrease.
It has been suggested that “myocyte slippage” plays a major role in progressive chamber dilation leading to failure [8, 38, 44], although much of the literature mentioning this phenomenon is rather vague. This concept usually refers to slippage of myocytes past one another transversely or linear slippage of individual myofibrils within myocytes [44, 45].
Increased myocardial collagenase activity (see discussion under Extracellular Matrix Remodeling below) is believed to disrupt intermyocyte collagen struts, leading to side-to-side slippage of myocytes [46]. Such a process could reduce wall thickness and increase the volume of the ventricle. Linzbach [44] and others [38] have noted reduced numbers of myocytes across the wall as evidence of myocyte slippage. However, this explanation may be too simplistic. For a meaningful discussion of the slippage concept, we need to consider the three-dimensional nature of myocyte-to-myocyte interconnections. Each myocyte is connected to an average of 5–10 neighboring myocytes via end-to-end and side-to-side intercalated disks (◘ Fig. 17.2) [47]. Slippage implies disruption of intercalated disks. Once the disks are disrupted, they may be unable to reconnect, resulting in poorly coordinated contractions.
Scanning electron micrograph (top) and a drawing (bottom) of the cardiac myocardial fibers. The cardiocyte Ci connects with five neighboring cardiocytes (A 1, A 2, C 1, C 2, and C 3). Source: Yamamoto S, James TN, Sawada K, Okabe M, Kawamura K. Generation of new intercellular junctions between cardiocytes. A possible mechanism compensating for mechanical overload in the hypertrophied human adult myocardium. Circ Res. 1996;78:362–70
Factors Affecting the Magnitude of Remodeling after Myocardial Infarction
The magnitude of infarct expansion and development of LV remodeling largely depends on the extent of myocardial damage and the loading conditions of the ventricle. In the rat infarct model, increase in LV diastolic volume is related to the size of the infarct and correlated with the extent of impaired systolic performance [48, 49]. A critical transmural infarct size of about 20 % of LV myocardium was necessary for significant infarct expansion [22]. It was observed more frequently in patients with a large anterior transmural infarction compared to an infarction in other regions of the LV [50–52]. Distortion of the ventricular contour leading to aneurysm formation is frequent in patients with infarct expansion and is associated with a much higher 1-year mortality than for patients with anterior infarction and comparably reduced ejection fraction (EF) but without aneurysm [53].
The loading conditions of the ventricle are also important in ventricular remodeling. Both early transient increase in afterload after an MI and sustained increase in afterload with aortic banding increased infarct expansion in animal models [54, 55]. Patients with hypertension and LV hypertrophy have increased morbidity and mortality after MI [56], and careful afterload reduction early in the course of MI may have important effects on LV remodeling by reducing infarct expansion and limiting infarct size [57].
Early establishment of patency of the infarct-related coronary artery and restoration of antegrade flow may also confer a beneficial effect on ventricular remodeling and long-term survival in patients with acute MI, whether accomplished pharmacologically [58] or mechanically [59]. However, the open-artery hypothesis that the restoration of antegrade flow in the infarct-related artery days, weeks, or even several months after MI would improve survival with or without improvement of LV function was not proved in the Occluded Artery Trial [60] and the Total Occlusion Study of Canada (TOSCA)–2 Trial [61].
Late Progressive Postinfarct Left Ventricular Remodeling
Early infarct expansion after MI may be followed by progressive ventricular dilatation and dysfunction over subsequent months and years, involving predominantly the noninfarcted segments. The mechanisms responsible for this inexorable deterioration of LV structure and function are not entirely clear but are related to continued activation of neurohormones and cytokines such as norepinephrine, angiotensin II, aldosterone, endothelin, and tumor necrosis factor. These factors, in combination with increased wall stress and mechanical stretch of the myocytes, upregulate a large number of signaling pathways, leading to structural and functional changes in the myocyte and nonmyocyte compartments that underlay a reduction in LV function and the progression of HF. In the discussion that follows, changes in these individual components will be described and their implications discussed .
Alterations in the Myocyte Compartment
The remodeling process results in important changes in the cardiac myocytes. These include myocyte hypertrophy, myocyte loss by necrosis [41, 42, 62] and apoptosis [41, 63–66], and changes in the structural proteins with downregulation of contractile and sarcomeric skeleton proteins and upregulation of cytoskeletal and membrane-associated proteins [67]. In addition, loss of myofilaments, nuclear enlargement, development of multiple small mitochondria, decrease in the T-tubular system, and sarcoplasmic reticulum are common histological features of the failing myocardium [68].
Myocyte Hypertrophy
Grossman and coworkers proposed that alterations in myocyte shape and size determine the type of cardiac hypertrophy [69]. In conditions with pressure overload such as aortic stenosis or hypertension, parallel addition of sarcomere causes an increase in myocyte cross-sectional area with no increase in myocyte length (◘ Fig. 17.3) [70–72]. This leads to an increase in wall thickness and concentric LV hypertrophy (increase in ratio of wall thickness to chamber dimension) [69, 73]. In conditions with volume overload such as aortic and mitral regurgitation, ventricular volume and wall thickness increase proportionally, and this is associated with a corresponding proportional increase in both myocyte length and cross-sectional area (addition of sarcomeres both in parallel and series) [74] (◘ Fig. 17.3). It appears that during the compensated stage of concentric LV hypertrophy, wall stress does not increase.
Schematic representation of myocyte change in left ventricular concentric and eccentric hypertension. In pressure-overload hypertrophy, myocyte cross-sectional area (CSA) increases and the ventricular wall becomes thicker during the compensatory phase. In volume-overload hypertrophy, ventricular volume and wall thickness increase proportionally, and this is associated with a corresponding proportional increase in both myocyte length and CSA. CSA and L, cross-sectional area and length. Source: Gerdes AM. The use of isolated myocytes to evaluate myocardial remodeling. Trends Cardiovasc Med. 1992;2(4):152–5
After a large MI, progressive LV dilatation is due to an increase in myocyte size which occurs predominantly by laying of sarcomeres in series, resulting in an increase in myocyte length, with only mild increase in width and cross-sectional area [75–78] (◘ Fig. 17.4). This further increases cavity volume with no change or a decrease in wall thickness. Myocyte length is the major determinant of changes in LV size, and most of the increase in LV volume can be explained by an increase in myocyte length [75, 77–79]. Although LV mass increases, the increase in LV volume is proportionately greater, so that mass-to-volume ratio, an important determinant of wall stress, is reduced. The development of myocardial hypertrophy after MI, therefore, results in eccentric hypertrophy (cavity dilation with a decrease in wall thickness to chamber dimension ratio) that increases wall stress.
Cardiac myocyte remodeling in the rat infarct model. Myocyte length and width from rats at 2, 4, and 6 weeks after myocardial infarction are compared to those from a sham-operated animal. Note the predominant increase in myocyte length as the major determinant of the increase in ventricular volume. MI myocardial infarction. Source: Anand IS, Liu D, Chugh SS, Prahash AJ, Gupta S, John R, Popescu F, Chandrashekhar Y. Isolated myocyte contractile function is normal in postinfarct remodeled rat heart with systolic dysfunction. Circulation. 1997;96(11):3974–84
In volume-overload conditions such as mitral and aortic regurgitation, ventricular hypertrophy remains appropriate and helps to maintain normal wall stress for variable periods of time. Transition from a compensated to a decompensated state is associated with further increase in chamber volume but no increase in wall thickness. This results in a decrease in mass-to-volume ratio and increase in wall stress. The cellular mechanisms responsible for this are not entirely clear, but they could be related to an arrest in growth of the myocytes in the transverse diameter, resulting in myocyte lengthening without further change in myocyte cross-sectional area. Studies of mitral regurgitation in the dog, and in patients at the time of mitral valve surgery, also show a decrease in myocardial myosin content proportional to the degree of LV dysfunction [74, 80]. Thus, reduced contractility in mitral regurgitation could, in part, be due to loss of contractile elements. Although aortic and mitral regurgitation are often considered together as volume-overload conditions, the two have their specific pathophysiologic features .
In aortic regurgitation, the sum of the regurgitant and forward stroke volume is ejected into the aorta in systole, resulting in a wide pulse pressure and systolic hypertension. Therefore, aortic regurgitation creates both volume and pressure overload on the left ventricle. Systolic wall stress is always higher in aortic regurgitation than in mitral regurgitation [81] and is often as high as in aortic stenosis (the classic pressure-overload condition) [82]. These different loading conditions in mitral and aortic regurgitation create two different types of ventricular geometry. In mitral regurgitation, there is an enlarged thin-walled left ventricle in which the mass-to-volume ratio is less than 1.0 [83]. In contrast, in aortic regurgitation, the mass-to-volume ratio is normal at 1.0 [84]. Whether the cellular hypertrophy at the onset of failure is different in these two conditions remains to be determined.
In pressure-overload conditions, concentric ventricular hypertrophy (thick wall, normal chamber volume, and high mass-to-volume ratio) helps to keep wall stress normal despite high ventricular pressure. Because systolic stress (afterload) is a major determinant of ejection performance, the normalization of systolic stress helps to maintain a normal stroke volume despite the need to generate high levels of systolic pressure [12]. Transition to failure is accompanied by progressive cavity enlargement and decline in the mass-to-volume ratio, resulting in eccentric ventricular hypertrophy. In spontaneously hypertensive rats, transition to failure is preceded by myocyte lengthening without an increase in myocyte cross-sectional area [70, 71].
Myocyte Death
Cell death is an important determinant of progressive cardiac remodeling and LV wall thinning. A reduction of contractile material is a prominent feature in HF, and myocyte loss may occur either by necrosis or apoptosis [41].
Myocyte necrosis : Necrosis generally occurs in the setting of catastrophic events such as MI or inflammation and is characterized by severe membrane alterations, release of cell breakdown products, and polymorphonuclear infiltration. However, slow myocyte loss by necrosis is also a common feature of chronic HF [40–42, 85]. During the progression of HF, activation of several neurohormones occurs, including norepinephrine, angiotensin II, and endothelin. These neurohormones are directly toxic to the myocardium and have been shown to cause myocyte necrosis in various animal models [86, 87]. Moreover, in patients with severe HF, circulating levels of troponin are often increased, suggesting ongoing myocyte necrosis [88, 89]. Myocyte loss through necrosis probably contributes to progressive LV dilatation and wall thinning. Even very low plasma concentrations of troponin are predictive of adverse outcomes in patients with chronic HF [88].
Myocyte apoptosis : Apoptosis or programmed cell death is an evolutionarily conserved process of cell death, wherein cells die without provoking significant inflammatory response. Evidence shows that apoptosis contributes to the progression of HF. Apoptosis occurs through a cascade of subcellular events including cytochrome c release into the cytoplasm and activation of proteolytic caspases [90]. Activated caspases lead to fragmentation of cytoplasmic proteins, including contractile apparatus [91]. Caspase-3 (the final executioner in the apoptotic cascade) overexpression or activation has been shown to directly reduce the contractile performance of the LV [92]. The degree of myosin cleavage with caspases correlated with the contractile performance of the heart [93]. It has been proposed that the release of cytochrome c from mitochondria and contractile protein loss in living heart muscle cells contributes to systolic dysfunction [90]. Apoptosis is involved at multiple points in the natural history of HF. This includes initial events like ischemia, infarction, and inflammation as well as those events occurring later in established LV dysfunction. Several of the factors implicated in the pathogenesis of HF such as myocardial stretch [94], norepinephrine [95], angiotensin II [96, 97], tumor necrosis factor-α (TNF-α), and oxidative stress [98, 99] may provoke apoptosis.
While the presence of myocardial apoptosis has been confirmed in end-stage human HF [65, 66] and in several animal models [41, 43, 63, 64], questions remain whether apoptosis is a cause or a consequence of HF. Myocyte apoptosis may be a factor in the transition from compensated to uncompensated HF [91]. This has been shown in several animal models of experimentally induced LV hypertrophy and HF [100–102]. Several studies have demonstrated the presence of apoptosis late after MI [103–105].
Alterations in Myocyte Structural Proteins
The complexity of events involved in the pathogenesis of ventricular remodeling cannot be solely attributed to myocyte hypertrophy and cell loss. The hypertrophied myocytes in the remodeled failing heart also show alterations in most of the structural proteins (◘ Table 17.1) [67]. Following is a brief description of the structural protein (◘ Fig. 17.5), alterations that occur in HF proteins, and their functional consequences.
Contractile Proteins
The contractile apparatus includes thick filament myosin and thin filament complexes composed of α-actin, α-tropomyosin, and troponins C, I, and T. Ventricular remodeling involves transcriptional and translational downregulation of these proteins [67]. One of the earliest changes is a decrease in α-myosin heavy chain and an increase in β-myosin heavy chain [106].
Sarcomeric Skeleton Proteins
The contractile apparatus is kept in register by different proteins localized in the Z-disk, M-band of the sarcomere, and the giant filament molecule titin, which spans the entire half-sarcomere from the Z-disk to the M-line. The Z-disk is a region of overlapping tails of actin microfilaments cross-linked by α-actinin. The M-line is the region where the myosin tails are linked and organized by the M-line proteins—myomesin, M-line protein, and myosin-binding protein-C. Titin is anchored with its N-terminus at the Z-disk and reaches the M-line region with its C-terminal head portion where it interacts with M-line protein and with myomesin [107]. It spans the Z-disk of the sarcomere [108] and overlaps in the M-line region of the sarcomere [109], thus functioning as a molecular spring and a source of elastic properties of the cardiomyocyte (◘ Fig. 17.5). The interplay between titin and actomyosin suggests a possible role for titin in the Frank-Starling mechanism of the heart [107]. Several studies have reported that the amount of titin is reduced in myocardium of patients with dilated cardiomyopathy, and this could be responsible for the altered ventricular compliance in this condition [110, 111]. Because titin is required for sarcomere formation, lack of titin may also contribute to contractile dysfunction of failing hearts [112].
Cytoskeletal Proteins
The cytoskeleton is a complex network of microtubules (primarily tubulin), nonsarcomeric actin, and intermediate filaments (primarily desmin). Tubulin is the protein of microtubules, which are hollow tubes formed from α- and β-tubulin surrounding the nucleus and spreading mostly in a longitudinal direction throughout the entire cell. The multifunctional roles of microtubules include mitosis, intracellular transport, organization of organelles, cell motility, determination of cell shape, receptor modulation, and signaling [113]. Desmin surrounds the Z-disks and connects the sarcomeres so that they are kept in register during contraction. Desmin filaments also link myofibrils to one another, to the sarcolemma, and to the nuclear envelope [114]. The desmin network plays a role in the underlying structural integrity of the myocyte, as well as participating in the signaling processes needed for integration of cellular responses to external and internal stimuli [114].
In failing human myocardium, both tubulin and desmin are increased [115]. The increase in these proteins mainly occurs in cells that lack myofilaments and could, therefore, help maintain cellular stability. Tubulin accumulation plays a role in certain models of pressure-overload hypertrophy [116]. In feline right ventricular hypertrophy resulting from pulmonary artery banding, isolated myocytes show contractile dysfunction and loss of compliance. These changes are accompanied by an increase in total and polymerized tubulin [117–119].
Desmin-related cardiomyopathies that have, as a hallmark, abnormal deposits of desmin aggregates are increasingly reported. A progressive increase of desmin protein and filaments was shown to accompany the transition from hypertrophy to HF [120]. Overexpression and altered distribution of desmin were also observed in dilated cardiomyopathy [115]. The absence of an intact desmin filament system may also be involved in cardiomyocyte hypertrophy and cardiac dilation with compromised systolic function [121]. Whether alteration in desmin quantity is a cause or a consequence of HF is not yet clear .
Membrane-Associated Proteins
Membrane-associated proteins include dystrophin, vinculin, talin, spectrin, and integrins, which are involved in fixation of sarcomeres to the lateral sarcolemma and stabilization of the T-tubular system [67, 122, 123]. Mutations of these proteins have been shown to cause dilated cardiomyopathy [124–126]. Dystrophin connects intracellular actin and extracellular laminin independent of integrin binding [127] and plays an important role in promoting the action of the cytoskeleton as a stabilizing force and as a mechanotransductor [128].
Intercalated Disk Proteins
The intercalated disk consists of three different types of specialized membranes: fascia adherens, desmosomes, and gap junctions [129]. Fascia adherens establish the longitudinal connections with the contractile filaments. The desmosomes are connected to intracellular desmin via desmoplakins. Connexins are four-pass transmembrane proteins that are assembled in groups of six to form hemichannels, or connexons, and two hemichannels combined to form a gap junction. Gap junctions are responsible for the orderly spread of electrical excitation from one myocyte to the next in the heart. Remodeling of gap junction and connexin expression is a conspicuous feature of human congestive HF and other cardiac conditions with a dysrhythmic tendency. Remodeling of gap junctions and reduced connexin43 levels may contribute to slowing of conduction [130, 131]. Evidence from experimental animals strengthens the case that gap junction remodeling is a key determinant of arrhythmias in the diseased heart [132, 133].
Alterations in the Nonmyocyte Compartment
Apart from the myocyte compartment, the chronically failing heart is characterized by iterations in the extracellular matrix (ECM), particularly by fibrous tissue formation [62]. Such an adverse accumulation of ECM raises myocardial stiffness and impairs contractile behavior [134].
Extracellular Matrix Remodeling
The extracellular matrix of the heart is made up of a number of structural proteins including fibrillar collagen, smaller amounts of elastin, laminin, fibronectin, and signaling peptides. The complex collagen three-dimensional weave, mainly consisting of type I collagen, interconnects individual myocytes through a collagen-integrin-cytoskeletal-myofibril arrangement. This network supports cardiac myocytes during contraction and relaxation and also provides a mechanism for translating individual myocyte shortening and force generation into ventricular contraction. It is also responsible for much of the ventricle’s passive diastolic stiffness [135]. In both human and animal studies, progressive LV remodeling and dysfunction are associated with significant changes in the ECM [136–139]. The specific changes in serological markers of collagen turnover occurring in HF with preserved versus reduced systolic function need to be clarified [140].
The structural hallmark of prolonged pressure-overload hypertrophy is increased collagen accumulation between individual myocytes and myocyte fascicles (◘ Fig. 17.6) [141, 142]. Thus, the highly organized architecture of the ECM undergoes significant alterations in collagen structure, composition, and geometry caused by increased collagen synthesis, postsynthetic processing, posttranslational modification, and decreased degradation and turnover. This “reactive” collagen deposition is characterized by both perivascular and interstitial fibrosis [135, 143, 144]. The changes in collagen homeostasis that occur during the development of chronic pressure-overload hypertrophy are directly associated with increased myocardial diastolic stiffness properties, which in turn cause abnormal diastolic filling [142, 145, 146]. Indeed, clinical evidence suggests that progressive ECM accumulation and diastolic dysfunction are important underlying pathophysiological mechanisms for HF in patients with pressure-overload hypertrophy [147, 148].
Scanning electron micrographs taken from normal nonhuman primate left ventricular myocardium and following the induction of pressure-overload hypertrophy (POH). These microscopic studies demonstrate thickening of the collagen weave and overall increased relative content between myocytes with POH. Source: Abrahams C, Janicki JS, Weber KT. Myocardial hypertrophy in Macaca fascicularis. Structural remodeling of the collagen matrix. Lab Invest. 1987;56(6):676–83
Because of the persistently elevated preload in volume-overload hypertrophy, a much different pattern of ECM remodeling occurs. In large-animal models of volume-overload hypertrophy caused by chronic mitral regurgitation, the LV remodeling process is accompanied by increased degradation of collagen fibrils surrounding individual myocytes [80]. These changes in ECM support are associated with changes in isolated LV myocyte geometry where the cardiac cells increase in length. Representative scanning electron micrographs taken from a model of canine mitral regurgitation [149] are shown in ◘ Fig. 17.7 and show the profound differences in ECM structure and composition compared with normal myocardium. Increased ECM proteolytic activity likely contributes to the reduced ECM content and support and, thereby, facilitates the overall LV remodeling process [150].
Scanning electron micrographs taken from normal canine left ventricular myocardium following chronic mitral regurgitation that causes a volume-overload hypertrophy (VOH). In this model of VOH, a loss of normal ECM architecture was demonstrated between individual myocytes (arrows), and the collagen supporting network is poorly organized. Source: Spinale FG. Myocardial matrix remodeling and the matrix metalloproteinases: influence on cardiac form and function. Physiol Rev. 2007;87(4): 1285–342
Although the mechanisms by which increased degradation of collagen promotes LV dilatation and global LV dysfunction are not entirely clear, dissolution of the collagen weave may lead to increased elasticity and contribute to muscle fiber slippage and, therefore, an increase in chamber size [145]. Loss of collagen struts connecting individual myocytes could prevent transduction of individual myocyte contractions into myocardial force development, resulting in reduced myocardial systolic performance.
The ECM and, particularly, collagen are under dynamic control of two sets of proteins: those that favor degradation and those that tend to inhibit it. The dissolution or degradation of collagen is predominantly related to the activation of matrix metalloproteinases (MMPs ), a family of zinc-containing proteins that includes collagenases, gelatinases, stromelysins, and membrane-type MMPs [150]. A critical control point for MMP activity is through the inhibition of the activated enzyme by the action of a group of specific MMP inhibitors termed tissue inhibitors of metalloproteinases (TIMPs ) [150]. The TIMPs are low-molecular-weight proteins that can combine noncovalently to active MMPs, inhibiting their activity [151, 152].
While the contributory mechanisms for the changes in plasma MMP levels remain speculative, an association between changes in plasma MMP levels to adverse LV remodeling has emerged. A Framingham Heart substudy showed that increased plasma MMP-9 levels were associated with LV dilation [153]. Elevated TIMP-1 plasma levels have been associated with major cardiovascular risk factors and with the presence of LV hypertrophy [153]. Furthermore, changes in plasma TIMP-1 levels have been associated with increased mortality [154]. However, it is likely that the changes in plasma MMP and TIMP levels observed in these studies will be influenced by the underlying etiology of the cardiovascular disease process and, therefore, that future studies will be needed. Furthermore, these studies only measured MMP and TIMP plasma levels at one point in time, so the temporal relation to the natural history of the LV remodeling process and progression to HF remains to be established.
Myocardial Fibrosis
Fibrosis in HF is an ongoing, active process of increasing collagen concentration and not simply a response to myocyte injury [134]. There are two types of fibrosis: reparative and reactive. Reparative fibrosis occurs in response to a loss of myocardial cells and is mainly interstitial. In contrast, reactive fibrosis is observed in the absence of cell loss as a reaction to changes in myocardial load or inflammation and is primarily perivascular. During ventricular remodeling, reactive and reparative fibrosis usually coexist. After MI, reparative fibrosis is organized as a scar and is surrounded by reactive fibrosis and myocyte hypertrophy [135].
The mechanisms responsible for fibrosis are still controversial. Fibrosis is not directly induced by myocardial stretch or mechanical overload. Chronic volume overload due to exercise training, atrial septal defect, or aortic insufficiency is not accompanied by ventricular fibrosis [155, 156]. In contrast, pressure overload is frequently associated with fibrosis. It has been proposed that ventricular fibrosis seen in arterial hypertension is caused by associated factors linked to this condition, such as ischemia [157] and neurohormones [134]. Humoral factors, particularly those of the renin-angiotensin-aldosterone system, are believed to be responsible for fibrosis. Angiotensin II and aldosterone have been implicated in the process as they stimulate collagen synthesis in cultured cardiac fibroblasts, and angiotensin II inhibits collagen degradation [158, 159].
Myocardial fibrosis has a number of deleterious effects on cardiac function. A two- to threefold increase in myocardial collagen content alters ventricular filling properties particularly by increasing diastolic stiffness; a fourfold or greater increase in fibrosis also affects systolic function [160]. Fibrosis contributes to ventricular arrhythmias because disproportionate collagen accumulation creates myocardial electrical heterogeneity. Fibrosis is therefore one of the major biological determinants of fatal issues in cardiac remodeling, including congestive HF, severe arrhythmias, and sudden death .
Changes in Global Structure and Function
The mechanical effects of LV remodeling set in motion several self-sustaining deleterious consequences. As the ventricle enlarges, LV geometry alters from a normal prolate ellipse to a mechanically disadvantageous spherical or globular shape. The result is an increase in meridional wall stress [161], abnormal distribution of fiber shortening, increase in oxygen consumption [161, 162], and abnormal myocardial bioenergetics [163]. The spherical shape of the LV leads to dilatation of the atrioventricular ring and stretching of the papillary muscles, resulting in functional mitral regurgitation [164], which contributes to a further decrease in forward cardiac output. Moreover, the high LV end-diastolic volume and pressure promote subendocardial ischemia that aggravates LV dysfunction and neurohormonal activation, decreases exercise capacity [165], and increases the risk of ventricular arrhythmias [166].
Compensatory Versus Maladaptive Remodeling
A fundamental question that must be addressed before embarking on a strategy to reverse hypertrophic and structural myocardial remodeling is whether remodeling is good or bad. Distinction is often made between a compensatory (adaptive) and a maladaptive process. An adaptive component enables the heart to maintain function in response to pressure or volume overloading in the acute phase of cardiac injury [167]. Acute distension of the viable myocardium and the operation of the Frank-Starling mechanism through an increase in sarcomere length are, therefore, entirely appropriate beneficial responses. Likewise, augmentation of chronotropic and inotropic activity through adrenergic receptor stimulation that tends to maintain pump function during the abrupt loss of contractile tissue can be considered compensatory.
Progressive LV dilatation after MI can also help to maintain stroke volume in the face of reduced contractile function and has been considered an adaptive and compensatory response [3, 168]. Under these circumstances, however, increased LV volume is not due to sarcomere stretch, but because of the addition of new sarcomeres in series [78]. Therefore, it is not a mechanism of enhancing contractility on the basis of Frank-Starling mechanism. Such a progressive remodeling and LV dilation does not normalize, but increases wall stress and is associated with a poor prognosis [19, 168].
Moreover, the prevention of very early LV dilation with the use of angiotensin-converting enzyme (ACE) inhibitors and beta-blockers does not have any deleterious hemodynamic consequence [169–171]. Indeed, Sharpe et al. [169] have demonstrated that the attenuation of LV remodeling with early initiation of ACE inhibitors is associated with a greater increase in stroke volume as compared with a placebo. Furthermore, the prevention of remodeling by early initiation of an ACE inhibitor or beta-blocker, after MI, in selected populations with LV dysfunction [31, 34] and even in unselected populations [172] is associated with significant reduction in mortality and morbidity [34, 170, 173, 174]. Hence, ventricular remodeling and dilation after MI may be maladaptive from the very start and should be a target for aggressive antiremodeling therapy .
In contrast, an increase in ventricular mass that helps to normalize wall stress in aortic stenosis and hypertension may be an appropriate compensatory response. Because systolic stress (afterload) is a major determinant of ejection performance, normalization of systolic stress helps to maintain a normal EF while generating high levels of systolic pressure [12]. However, LV hypertrophy has been shown to be an important independent risk factor for mortality and morbidity [17]. Similarly, a proportional increase in chamber volume, wall thickness, and mass in mitral and aortic regurgitation normalizes wall stress and is an obligatory response to maintain a large stroke volume that is necessitated by the regurgitant volume. Until the initial volume and pressure overload is matched by adequate hypertrophy, the process may be considered adaptive and compensatory. Eventually, a mismatch occurs with progressive dilation and the process becomes maladaptive and decompensatory, and HF becomes clinically manifest [175, 176]. There is no data to indicate when the transition from possible adaptive to maladaptive remodeling occurs; such a transition and its time course can vary greatly. However, once established beyond a certain phase, remodeling likely contributes to progression of HF. Thus, whether remodeling is beneficial or deleterious cannot be viewed as a stereotypical process. Today’s challenge is taking advantage of the adaptive features of the hypertrophic response while eliminating or at least minimizing the maladaptive consequences.
Reverse Remodeling
“Reverse remodeling” is a concept, where progressive LV dilatation and deterioration in contractile function are not simply arrested, but partially reversed. Two important questions related to reverse remodeling are: “Do myocytes have the ability to remove sarcomeres?” and “Is there any time line beyond which reverse remodeling cannot be achieved?”
Surgical and pharmacological experiments have confirmed that the regression of myocyte hypertrophy with removal of sarcomeres is possible. But insufficient data exist to address the second question. Remodeling is believed to be reversible early in the natural transition from hypertrophy to failure, whereas later, with the development of extensive fibrosis, accumulation of cytoskeletal proteins, and loss of the contractile filaments, an irreversible process sets in [111]. Several therapeutic approaches for HF have been shown to halt or even reverse the remodeling process.
Pharmacological Approaches
Numerous experimental studies have shown that modulating neurohormonal activation improves cardiac remodeling [177, 178]. McDonald et al. [179] showed that ACE inhibition and beta-adrenoreceptor blockade can reverse established ventricular remodeling in a canine model of discrete myocardial damage [179]. A significant reduction of LV mass and a trend in reduction of end-diastolic volume were found in both captopril- and beta-blocker-treated groups compared with the control group [179]. Tamura et al. [180] reported that the administration of angiotensin II type 1 receptor blockers produced significant reduction in myocyte volume, length, and cross-sectional area in rats with spontaneously hypertensive HF—below pretreatment values, suggesting true reverse remodeling, rather than simply arrested progression of myocyte hypertrophy [180]. Xu et al. [181] studied the effect of angiotensin II receptor blocker losartan combined with exercise training in a postinfarction rat model and demonstrated that exercise training after MI provides a beneficial effect on cardiac function and LV remodeling by altering the gene and protein expressions that regulate myocardial fibrosis. In contrast, such effects were only slightly improved by combining exercise and losartan [181].
ACE Inhibitors
The first class of medications shown to beneficially affect remodeling and clinical outcomes in patients with HF was the ACE inhibitors. In several trials performed in both asymptomatic and symptomatic patients with reduced EF, ACE inhibitors attenuated the progressive increase in end-diastolic and end-systolic volume compared with placebo-treated groups [32, 169, 182–184].
Beta-Blockers
In contrast to ACE inhibitors that attenuate LV remodeling, the use of beta-blockers has been associated with significant reduction in ventricular volumes and improvement in global LV function (reverse remodeling) [30, 31, 33, 185]. Beta-blockers were shown to reduce myocardial apoptosis which, at least in part, could explain their favorable effect on ventricular remodeling [186].
Aldosterone Receptor Blockers
Aldosterone receptor blockers have been shown to reverse LV remodeling following MI and in patients with HF [187, 188]. The 4E–Left Ventricular Hypertrophy Study [189] used cardiac magnetic resonance imaging (MRI) to compare LV mass regression by the selective aldosterone blocker eplerenone to the ACE inhibitor enalapril and the combination of eplerenone/enalapril in hypertensive patients with LV hypertrophy. Eplerenone was as effective as enalapril in regression of LV hypertrophy and control of blood pressure. The combination of eplerenone and enalapril was more effective in reducing LV mass and systolic blood pressure than eplerenone alone [189]. In a single-site clinical trial, Chan and colleagues [190] demonstrated with serial cardiac magnetic resonance (CMR) that the addition of spironolactone to candesartan has significant beneficial effects on LV reverse remodeling in patients with mild to moderate chronic systolic HF .
Angiotensin Receptor Blockers
Several trials demonstrated the beneficial effect of angiotensin receptor blockers (ARBs) on LV remodeling. In the Valsartan Heart Failure Trial (Val-HeFT) [191, 192], valsartan therapy attenuated LV remodeling [193]. Stratification by baseline severity of remodeling showed that patients with worse LV enlargement and systolic function are at highest risk for an event, yet appear to gain the most antiremodeling effect and clinical benefit with valsartan treatment [194]. The Losartan Intervention For Endpoint (LIFE) study [195] showed that reduction in LV mass by angiotensin II blockade was independent of blood pressure reduction, indicating that the inhibition of the renin-angiotensin-aldosterone system has added benefits beyond blood pressure control [195].
Isosorbide Dinitrate -Hydralazine Combination
In the first Vasodilator-Heart Failure Trial (V-HeFT-I) [196], isosorbide dinitrate combined with hydralazine therapy compared with placebo in patients with HF treated only with digoxin and diuretic resulted in a sustained increase in LV EF that was associated with improved survival [196]. The African American Heart Failure Trial (A-HeFT) confirmed these findings, on top of ACE inhibitors and beta-blockers [197, 198].
Role of Cell Transplantation and Surgical Approaches in Heart Failure
Since about 2000 there has been an explosion of activity in the field of cell transplantation and of advanced surgical approaches in HF. These specialized areas are discussed in detail elsewhere in this book.
Cardiac Resynchronization Approach
Beneficial effects of cardiac resynchronization therapy (CRT ) on survival, New York Heart Association (NYHA) functional class, exercise capacity, and quality of life are associated with significant improvement in LV remodeling as early as 1 month after device implantation [199–201] and with further, progressive reduction in LV volumes beyond 1 year in selective patients [202, 203]. The Cardiac Resynchronization-Heart Failure (CARE-HF) study demonstrated an early and sustained reduction in NT-pro-BNP with CRT that correlated with improvement in LV dimension and EF and mitral regurgitation [204].
Cardiac Constraint Devices
Preclinical studies have shown that passive ventricular containment with cardiac constraint devices halts progressive ventricular remodeling [205–207] and improves myocyte function and structure, as characterized by enhanced myocyte contraction and relaxation, decreased myocyte hypertrophy, and decreased interstitial fibrosis [205, 207]. Ventricular restraint prevents infarct expansion, improves borderzone function, and favorably modifies LV geometry and myocardial structure after MI [208–210]. Limited clinical experience with the Acorn CorCap Cardiac Support Device and the Paracor HeartNet Device has shown amelioration of symptoms and improvement in LV chamber dimensions and EF. However, the implantation of these devices was not associated with improved survival [211–213].
Conclusions
Ventricular remodeling is a complex process. It results from interactions between the initial myocardial injury and alteration in loading conditions and multiple mechanical and neurohormonal factors that are capable of modifying the cardiomyocyte phenotype and inducing changes in the extracellular matrix. Myocyte hypertrophy, cellular necrosis and apoptosis, interstitial fibrosis, and degradation of collagen are the major features of myocardial remodeling. Each of these components of the remodeling process contributes importantly to the development and progression of HF. At the level of the ventricular chamber, remodeling refers to changes in ventricular geometry, volume, and mass. Although, initially, it may be compensatory in certain pressure and volume-overload conditions, progressive ventricular remodeling is ultimately a maladaptive process contributing to progression of symptomatic HF and to an adverse outcome. After acute MI, however, progressive hypertrophy and remodeling of noninfarcted myocardium may be harmful from the start.
Ventricular remodeling had emerged as an important therapeutic target in HF. Treatment with the goal of slowing or reversing remodeling has been shown to improve long-term outcome. Additional research is needed to identify the molecular processes responsible for remodeling and to improve ways to inhibit this maladaptive growth response.
Future Directions
Enormous effort has been directed to identifying new therapeutic strategies with long-term efficacy in HF. The path is littered with successes and failures [4], yet advances in myocardial biology, stem cell research, pharmacologic developments, and mechanical devices hold promise for future treatments. A comprehensive understanding of ventricular remodeling is obligatory, since it reflects the basic mechanisms of HF development and progression.
Although multiple studies have documented that interventions with beneficial effects on HF also generally attenuate or reverse ventricular remodeling and that those failing to improve clinical outcomes either have no effect on remodeling or have been associated with adverse remodeling, few studies have examined the mechanism by which LV reverse remodeling is mediated. Questions remain as to whether the reversal of myocyte structural remodeling is accompanied by normalization of the biology of the failing myocyte and what the mechanisms of changes are at the myocyte level. Further research should focus on the molecular and cellular mechanisms involved in adverse and reverse remodeling, on optimizing therapies to prevent remodeling, and on identifying appropriate patient groups to target.
References
Eaton LW, Weiss JL, Bulkley BH, Garrison JB, Weisfeldt ML. Regional cardiac dilatation after acute myocardial infarction: recognition by two-dimensional echocardiography. N Engl J Med. 1979;300(2):57–62.
Erlebacher JA, Weiss JL, Eaton LW, Kallman C, Weisfeldt ML, Burkley BH. Late effects of acute infarct dilation on heart size: a two dimensional echocardiographic study. Am J Cardiol. 1982;49(5):1120–6.
McKay RG, Pfeffer MA, Pasternak RC, Markis JE, Come PC, Nakao S, Alderman JD, Ferguson JJ, Safian RD, Grossman W. Left ventricular remodeling after myocardial infarction: a corollary to infarct expansion. Circulation. 1986;74(4):693–702.
Anand IS, Florea VG. Traditional and novel approaches to management of heart failure: successes and failures. Cardiol Clin. 2008;26(1):59–72.
Hill JA, Olson EN. Cardiac plasticity. N Engl J Med. 2008;358(13):1370–80.
Flint A. Diseases of the heart. 2nd ed. Philadelphia, PA: HC Lea; 1870.
Osler W. The principles and practice of medicine. New York, NY: Appleton; 1892.
Linzbach AJ. Heart failure from the point of view of quantitative anatomy. Am J Cardiol. 1960;5:370–82.
Hood Jr WP, Rackley CE, Rolett EL. Wall stress in the normal and hypertrophied human left ventricle. Am J Cardiol. 1968;22(4):550–8.
Sandler H, Dodge HT. Left ventricular tension and stress in man. Circ Res. 1963;13:91–104.
Meerson FZ. On the mechanism of compensatory hyperfunction and insufficiency of the heart. Cor Vasa. 1961;3:161–77.
Gunther S, Grossman W. Determinants of ventricular function in pressure-overload hypertrophy in man. Circulation. 1979;59(4):679–88.
Huber D, Grimm J, Koch R, Krayenbuehl HP. Determinants of ejection performance in aortic stenosis. Circulation. 1981;64(1):126–34.
Krayenbuehl HP, Hess OM, Ritter M, Monrad ES, Hoppeler H. Left ventricular systolic function in aortic stenosis. Eur Heart J. 1988;9(Suppl E):19–23.
Hammermeister KE, DeRouen TA, Dodge HT. Variables predictive of survival in patients with coronary disease. Selection by univariate and multivariate analyses from the clinical, electrocardiographic, exercise, arteriographic, and quantitative angiographic evaluations. Circulation. 1979;59(3):421–30.
Koren MJ, Devereux RB, Casale PN, Savage DD, Laragh JH. Relation of left ventricular mass and geometry to morbidity and mortality in uncomplicated essential hypertension. Ann Intern Med. 1991;114(5):345–52.
Levy D, Garrison RJ, Savage DD, Kannel WB, Castelli WP. Prognostic implications of echocardiographically determined left ventricular mass in the Framingham Heart Study. N Engl J Med. 1990;322(22):1561–6.
Vasan RS, Larson MG, Benjamin EJ, Evans JC, Levy D. Left ventricular dilatation and the risk of congestive heart failure in people without myocardial infarction. N Engl J Med. 1997;336(19):1350–5.
White HD, Norris RM, Brown MA, Brandt PW, Whitlock RM, Wild CJ. Left ventricular end-systolic volume as the major determinant of survival after recovery from myocardial infarction. Circulation. 1987;76(1):44–51.
Florea VG, Mareyev VY, Samko AN, Orlova IA, Coats AJ, Belenkov YN. Left ventricular remodelling: common process in patients with different primary myocardial disorders. Int J Cardiol. 1999;68(3):281–7.
Hutchins GM, Bulkley BH. Infarct expansion versus extension: two different complications of acute myocardial infarction. Am J Cardiol. 1978;41(7):1127–32.
Hochman JS, Bulkley BH. Expansion of acute myocardial infarction: an experimental study. Circulation. 1982;65(7):1446–50.
Weisman HF, Bush DE, Mannisi JA, Bulkley BH. Global cardiac remodeling after acute myocardial infarction: a study in the rat model. J Am Coll Cardiol. 1985;5(6):1355–62.
Erlebacher JA. Ventricular remodeling in myocardial infarction--the rat and the human. Am J Cardiol. 1985;56(13):910.
Chanutin A, Ludewig S. Experimental renal insufficiency produced by partial nephrectomy. Arch Intern Med. 1939;64(3):513–25.
Pfeffer JM, Pfeffer MA, Fishbein MC, Frohlich ED. Cardiac function and morphology with aging in the spontaneously hypertensive rat. Am J Physiol. 1979;237(4):H461–8.
Pfeffer J, Pfeffer M, Fletcher P, Braunwald E. Alterations of cardiac performance in rats with established spontaneous hypertension. Am J Cardiol. 1979;44(5):994–8.
Cohn JN, Ferrari R, Sharpe N. Cardiac remodeling--concepts and clinical implications: a consensus paper from an international forum on cardiac remodeling. J Am Coll Cardiol. 2000;35(3):569–82.
Sutton MG, Sharpe N. Left ventricular remodeling after myocardial infarction: pathophysiology and therapy. Circulation. 2000;101(25):2981–8.
Doughty RN, Whalley GA, Gamble G, MacMahon S, Sharpe N. Left ventricular remodeling with carvedilol in patients with congestive heart failure due to ischemic heart disease. Australia-New Zealand Heart Failure Research Collaborative Group. J Am Coll Cardiol. 1997;29(5):1060–6.
Doughty RN, Whalley GA, Walsh HA, Gamble GD, Lopez-Sendon J, Sharpe N, CAPRICORN Echo Substudy Investigators. Effects of carvedilol on left ventricular remodeling after acute myocardial infarction: the CAPRICORN Echo Substudy. Circulation. 2004;109(2):201–6.
Greenberg B, Quinones MA, Koilpillai C, Limacher M, Shindler D, Benedict C, Shelton B. Effects of long-term enalapril therapy on cardiac structure and function in patients with left ventricular dysfunction. Results of the SOLVD echocardiography substudy. Circulation. 1995;91(10):2573–81.
Groenning BA, Nilsson JC, Sondergaard L, Fritz-Hansen T, Larsson HB, Hildebrandt PR. Antiremodeling effects on the left ventricle during beta-blockade with metoprolol in the treatment of chronic heart failure. J Am Coll Cardiol. 2000;36(7):2072–80.
St. John Sutton M, Pfeffer MA, Moye L, Plappert T, Rouleau JL, Lamas G, Rouleau J, Parker JO, Arnold MO, Sussex B, Braunwald E. Cardiovascular death and left ventricular remodeling two years after myocardial infarction: baseline predictors and impact of long-term use of captopril: information from the Survival and Ventricular Enlargement (SAVE) trial. Circulation. 1997;96(10):3294–9.
Konstam MA, Udelson JE, Anand IS, Cohn JN. Ventricular remodeling in heart failure: a credible surrogate endpoint. J Card Fail. 2003;9(5):350–3.
Anand IS, Florea VG, Fisher L. Surrogate end points in heart failure. J Am Coll Cardiol. 2002;39(9):1414–21.
Harris P. Evolution and the cardiac patient. Cardiovasc Res. 1983;17(6):313–9. 373-8, 437-45.
Olivetti G, Capasso JM, Sonnenblick EH, Anversa P. Side-to-side slippage of myocytes participates in ventricular wall remodeling acutely after myocardial infarction in rats. Circ Res. 1990;67(1):23–34.
Weisman HF, Bush DE, Mannisi JA, Weisfeldt ML, Healy B. Cellular mechanisms of myocardial infarct expansion. Circulation. 1988;78(1):186–201.
Beltrami CA, Finato N, Rocco M, Feruglio GA, Puricelli C, Cigola E, Sonnenblick EH, Olivetti G, Anversa P. The cellular basis of dilated cardiomyopathy in humans. J Mol Cell Cardiol. 1995;27(1):291–305.
Anversa P, Kajstura J, Olivetti G. Myocyte death in heart failure. Curr Opin Cardiol. 1996;11(3):245–51.
Kajstura J, Cheng W, Reiss K, Clark WA, Sonnenblick EH, Krajewski S, Reed JC, Olivetti G, Anversa P. Apoptotic and necrotic myocyte cell deaths are independent contributing variables of infarct size in rats. Lab Invest. 1996;74(1):86–107.
Gupta S, Prahash AJ, Anand IS. Myocyte contractile function is intact in the post-infarct remodeled rat heart despite molecular alterations. Cardiovasc Res. 2000;48(1):77–88.
Linzbach A. Hypertrophy, hyperplasia and structural dilation of the human heart. Adv Cardiol. 1976;18:1–14.
Komamura K, Shannon RP, Ihara T, Shen YT, Mirsky I, Bishop SP, Vatner SF. Exhaustion of Frank-Starling mechanism in conscious dogs with heart failure. Am J Physiol. 1993;265(4 Pt 2):H1119–31.
Zhao MJ, Zhang H, Robinson TF, Factor SM, Sonnenblick EH, Eng C. Profound structural alterations of the extracellular collagen matrix in postischemic dysfunctional (“stunned”) but viable myocardium. J Am Coll Cardiol. 1987;10(6):1322–34.
Yamamoto S, James TN, Sawada K, Okabe M, Kawamura K. Generation of new intercellular junctions between cardiocytes. A possible mechanism compensating for mechanical overload in the hypertrophied human adult myocardium. Circ Res. 1996;78(3):362–70.
Pfeffer MA, Pfeffer JM, Fishbein MC, Fletcher PJ, Spadaro J, Kloner RA, Braunwald E. Myocardial infarct size and ventricular function in rats. Circ Res. 1979;44(4):503–12.
Fletcher PJ, Pfeffer JM, Pfeffer MA, Braunwald E. Left ventricular diastolic pressure-volume relations in rats with healed myocardial infarction. Effects on systolic function. Circ Res. 1981;49(3):618–26.
Picard MH, Wilkins GT, Gillam LD, Thomas JD, Weyman AE. Immediate regional endocardial surface expansion following coronary occlusion in the canine left ventricle: disproportionate effects of anterior versus inferior ischemia. Am Heart J. 1991;121(3 Pt 1):753–62.
Pirolo JS, Hutchins GM, Moore GW. Infarct expansion: pathologic analysis of 204 patients with a single myocardial infarct. J Am Coll Cardiol. 1986;7(2):349–54.
Weisman HF, Healy B. Myocardial infarct expansion, infarct extension, and reinfarction: pathophysiologic concepts. Prog Cardiovasc Dis. 1987;30(2):73–110.
Meizlish JL, Berger HJ, Plankey M, Errico D, Levy W, Zaret BL. Functional left ventricular aneurysm formation after acute anterior transmural myocardial infarction. Incidence, natural history, and prognostic implications. N Engl J Med. 1984;311(16):1001–6.
Hammerman H, Kloner RA, Alker KJ, Schoen FJ, Braunwald E. Effects of transient increased afterload during experimentally induced acute myocardial infarction in dogs. Am J Cardiol. 1985;55(5):566–70.
Nolan SE, Mannisi JA, Bush DE, Healy B, Weisman HF. Increased afterload aggravates infarct expansion after acute myocardial infarction. J Am Coll Cardiol. 1988;12(5):1318–25.
Rabkin SW, Mathewson FA, Tate RB. Prognosis after acute myocardial infarction: relation to blood pressure values before infarction in a prospective cardiovascular study. Am J Cardiol. 1977;40(4):604–10.
Jugdutt BI, Khan MI. Effect of prolonged nitrate therapy on left ventricular remodeling after canine acute myocardial infarction. Circulation. 1994;89(5):2297–307.
Effectiveness of intravenous thrombolytic treatment in acute myocardial infarction. Gruppo Italiano per lo Studio della Streptochinasi nell’Infarto Miocardico (GISSI). Lancet. 1986;1(8478):397–402.
Grines CL, Browne KF, Marco J, Rothbaum D, Stone GW, O’Keefe J, Overlie P, Donohue B, Chelliah N, Timmis GC, et al. A comparison of immediate angioplasty with thrombolytic therapy for acute myocardial infarction. The Primary Angioplasty in Myocardial Infarction Study Group. N Engl J Med. 1993;328(10):673–9.
Hochman JS, Lamas GA, Buller CE, Dzavik V, Reynolds HR, Abramsky SJ, Forman S, Ruzyllo W, Maggioni AP, White H, Sadowski Z, Carvalho AC, Rankin JM, Renkin JP, Steg PG, Mascette AM, Sopko G, Pfisterer ME, Leor J, Fridrich V, Mark DB, Knatterud GL, Occluded Artery Trial Investigators. Coronary intervention for persistent occlusion after myocardial infarction. N Engl J Med. 2006;355(23):2395–407.
Dzavik V, Buller CE, Lamas GA. Randomized trial of percutaneous coronary intervention for subacute infarct-related coronary artery occlusion to achieve long-term patency and improve ventricular function: the Total Occlusion Study of Canada (TOSCA)-2 trial. Circulation. 2006;114(23):2449–57.
Beltrami CA, Finato N, Rocco M, Feruglio GA, Puricelli C, Cigola E, Quaini F, Sonnenblick EH, Olivetti G, Anversa P. Structural basis of end-stage failure in ischemic cardiomyopathy in humans. Circulation. 1994;89(1):151–63.
Sharov VG, Sabbah HN, Shimoyama H, Goussev AV, Lesch M, Goldstein S. Evidence of cardiocyte apoptosis in myocardium of dogs with chronic heart failure. Am J Pathol. 1996;148(1):141–9.
Teiger E, Than VD, Richard L, Wisnewsky C, Tea BS, Gaboury L, Tremblay J, Schwartz K, Hamet P. Apoptosis in pressure overload-induced heart hypertrophy in the rat. J Clin Invest. 1996;97(12):2891–7.
Olivetti G, Abbi R, Quaini F, Kajstura J, Cheng W, Nitahara JA, Quaini E, Di Loreto C, Beltrami CA, Krajewski S, Reed JC, Anversa P. Apoptosis in the failing human heart. N Engl J Med. 1997;336(16):1131–41.
Narula J, Haider N, Virmani R, DiSalvo TG, Kolodgie FD, Hajjar RJ, Schmidt U, Semigran MJ, Dec GW, Khaw BA. Apoptosis in myocytes in end-stage heart failure. N Engl J Med. 1996;335(16):1182–9.
Kostin S, Heling A, Hein S, Scholz D, Klovekorn W-P, Schaper J. The protein composition of the normal and diseased cardiac myocyte. Heart Fail Rev. 1998;2:245–60.
Schaper J, Froede R, Hein S, Buck A, Hashizume H, Speiser B, Friedl A, Bleese N. Impairment of the myocardial ultrastructure and changes of the cytoskeleton in dilated cardiomyopathy. Circulation. 1991;83(2):504–14.
Grossman W, Jones D, McLaurin LP. Wall stress and patterns of hypertrophy in the human left ventricle. J Clin Invest. 1975;56(1):56–64.
Gerdes AM, Onodera T, Wang X, McCune SA. Myocyte remodeling during the progression to failure in rats with hypertension. Hypertension. 1996;28(4):609–14.
Onodera T, Tamura T, Said S, McCune SA, Gerdes AM. Maladaptive remodeling of cardiac myocyte shape begins long before failure in hypertension. Hypertension. 1998;32(4):753–7.
Gerdes AM. The use of isolated myocytes to evaluate myocardial remodeling. Trends Cardiovasc Med. 1992;2(4):152–5.
Lorell BH, Carabello BA. Left ventricular hypertrophy: pathogenesis, detection, and prognosis. Circulation. 2000;102(4):470–9.
Urabe Y, Mann DL, Kent RL, Nakano K, Tomanek RJ, Carabello BA, Cooper 4th G. Cellular and ventricular contractile dysfunction in experimental canine mitral regurgitation. Circ Res. 1992;70(1):131–47.
Olivetti G, Capasso JM, Meggs LG, Sonnenblick EH, Anversa P. Cellular basis of chronic ventricular remodeling after myocardial infarction in rats. Circ Res. 1991;68(3):856–69.
Zimmer HG, Gerdes AM, Lortet S, Mall G. Changes in heart function and cardiac cell size in rats with chronic myocardial infarction. J Mol Cell Cardiol. 1990;22(11):1231–43.
Gerdes AM, Kellerman SE, Moore JA, Muffly KE, Clark LC, Reaves PY, Malec KB, McKeown PP, Schocken DD. Structural remodeling of cardiac myocytes in patients with ischemic cardiomyopathy. Circulation. 1992;86(2):426–30.
Anand IS, Liu D, Chugh SS, Prahash AJ, Gupta S, John R, Popescu F, Chandrashekhar Y. Isolated myocyte contractile function is normal in postinfarct remodeled rat heart with systolic dysfunction. Circulation. 1997;96(11):3974–84.
Tamura T, Onodera T, Said S, Gerdes AM. Correlation of myocyte lengthening to chamber dilation in the spontaneously hypertensive heart failure (SHHF) rat. J Mol Cell Cardiol. 1998;30(11):2175–81.
Spinale FG, Ishihra K, Zile M, DeFryte G, Crawford FA, Carabello BA. Structural basis for changes in left ventricular function and geometry because of chronic mitral regurgitation and after correction of volume overload. J Thorac Cardiovasc Surg. 1993;106(6):1147–57.
Wisenbaugh T, Spann JF, Carabello BA. Differences in myocardial performance and load between patients with similar amounts of chronic aortic versus chronic mitral regurgitation. J Am Coll Cardiol. 1984;3(4):916–23.
Sutton M, Plappert T, Spiegel A, Raichlen J, Douglas P, Reichek N, Edmunds L. Early postoperative changes in left ventricular chamber size, architecture, and function in aortic stenosis and aortic regurgitation and their relation to intraoperative changes in afterload: a prospective two-dimensional echocardiographic study. Circulation. 1987;76(1):77–89.
Carabello BA. The relationship of left ventricular geometry and hypertrophy to left ventricular function in valvular heart disease. J Heart Valve Dis. 1995;4 Suppl 2:S132–8. discussion S138-9.
Feiring AJ, Rumberger JA. Ultrafast computed tomography analysis of regional radius-to-wall thickness ratios in normal and volume-overloaded human left ventricle. Circulation. 1992;85(4):1423–32.
Bing OH, Brooks WW, Robinson KG, Slawsky MT, Hayes JA, Litwin SE, Sen S, Conrad CH. The spontaneously hypertensive rat as a model of the transition from compensated left ventricular hypertrophy to failure. J Mol Cell Cardiol. 1995;27(1):383–96.
Mann DL, Kent RL, Parsons B, Cooper 4th G. Adrenergic effects on the biology of the adult mammalian cardiocyte. Circulation. 1992;85(2):790–804.
Tan LB, Jalil JE, Pick R, Janicki JS, Weber KT. Cardiac myocyte necrosis induced by angiotensin II. Circ Res. 1991;69(5):1185–95.
Latini R, Masson S, Anand IS, Missov E, Carlson M, Vago T, Angelici L, Barlera S, Parrinello G, Maggioni AP, Tognoni G, Cohn JN, Val-HeFT Investigators. Prognostic value of very low plasma concentrations of troponin T in patients with stable chronic heart failure. Circulation. 2007;116(11):1242–9.
Missov E, Calzolari C, Pau B. Circulating cardiac troponin I in severe congestive heart failure. Circulation. 1997;96(9):2953–8.
Narula J, Haider N, Arbustini E, Chandrashekhar Y. Mechanisms of disease: apoptosis in heart failure--seeing hope in death. Nat Clin Pract Cardiovasc Med. 2006;3(12):681–8.
Garg S, Narula J, Chandrashekhar Y. Apoptosis and heart failure: clinical relevance and therapeutic target. J Mol Cell Cardiol. 2005;38(1):73–9.
Laugwitz KL, Moretti A, Weig HJ, Gillitzer A, Pinkernell K, Ott T, Pragst I, Stadele C, Seyfarth M, Schomig A, Ungerer M. Blocking caspase-activated apoptosis improves contractility in failing myocardium. Hum Gene Ther. 2001;12(17):2051–63.
Moretti A, Weig HJ, Ott T, Seyfarth M, Holthoff HP, Grewe D, Gillitzer A, Bott-Flügel L, Schömig A, Ungerer M, Laugwitz KL. Essential myosin light chain as a target for caspase-3 in failing myocardium. Proc Natl Acad Sci U S A. 2002;99(18):11860–5.
Cheng W, Li B, Kajstura J, Li P, Wolin MS, Sonnenblick EH, Hintze TH, Olivetti G, Anversa P. Stretch-induced programmed myocyte cell death. J Clin Invest. 1995;96(5):2247–59.
Colucci WS, Sawyer DB, Singh K, Communal C. Adrenergic overload and apoptosis in heart failure: implications for therapy. J Card Fail. 2000;6(2 Suppl 1):1–7.
Cigola E, Kajstura J, Li B, Meggs LG, Anversa P. Angiotensin II activates programmed myocyte cell death in vitro. Exp Cell Res. 1997;231(2):363–71.
Kajstura J, Cigola E, Malhotra A, Li P, Cheng W, Meggs LG, Anversa P. Angiotensin II induces apoptosis of adult ventricular myocytes in vitro. J Mol Cell Cardiol. 1997;29(3):859–70.
Ferrari R, Agnoletti L, Comini L, Gaia G, Bachetti T, Cargnoni A, Ceconi C, Curello S, Visioli O. Oxidative stress during myocardial ischaemia and heart failure. Eur Heart J. 1998;19(Suppl B):B2–11.
DeLong MJ. Apoptosis: a modulator of cellular homeostasis and disease states. Ann N Y Acad Sci. 1998;842:82–90.
Condorelli G, Morisco C, Stassi G, Notte A, Farina F, Sgaramella G, de Rienzo A, Roncarati R, Trimarco B, Lembo G. Increased cardiomyocyte apoptosis and changes in proapoptotic and antiapoptotic genes bax and bcl-2 during left ventricular adaptations to chronic pressure overload in the rat. Circulation. 1999;99(23):3071–8.
Li Z, Bing OH, Long X, Robinson KG, Lakatta EG. Increased cardiomyocyte apoptosis during the transition to heart failure in the spontaneously hypertensive rat. Am J Physiol. 1997;272(5 Pt 2):H2313–9.
Matturri L, Milei J, Grana DR, Lavezzi AM. Characterization of myocardial hypertrophy by DNA content, PCNA expression and apoptotic index. Int J Cardiol. 2002;82(1):33–9.
Baldi A, Abbate A, Bussani R, Patti G, Melfi R, Angelini A, Dobrina A, Rossiello R, Silvestri F, Baldi F, Di Sciascio G. Apoptosis and post-infarction left ventricular remodeling. J Mol Cell Cardiol. 2002;34(2):165–74.
Palojoki E, Saraste A, Eriksson A, Pulkki K, Kallajoki M, Voipio-Pulkki LM, Tikkanen I. Cardiomyocyte apoptosis and ventricular remodeling after myocardial infarction in rats. Am J Physiol Heart Circ Physiol. 2001;280(6):H2726–31.
Sam F, Sawyer DB, Chang DL, Eberli FR, Ngoy S, Jain M, Amin J, Apstein CS, Colucci WS. Progressive left ventricular remodeling and apoptosis late after myocardial infarction in mouse heart. Am J Physiol Heart Circ Physiol. 2000;279(1):H422–8.
Lowes BD, Minobe W, Abraham WT, Rizeq MN, Bohlmeyer TJ, Quaife RA, Roden RL, Dutcher DL, Robertson AD, Voelkel NF, Badesch DB, Groves BM, Gilbert EM, Bristow MR. Changes in gene expression in the intact human heart. Downregulation of alpha-myosin heavy chain in hypertrophied, failing ventricular myocardium. J Clin Invest. 1997;100(9):2315–23.
Labeit S, Kolmerer B. Titins: giant proteins in charge of muscle ultrastructure and elasticity. Science. 1995;270(5234):293–6.
Gregorio CC, Trombitas K, Centner T, Kolmerer B, Stier G, Kunke K, Suzuki K, Obermayr F, Herrmann B, Granzier H, Sorimachi H, Labeit S. The NH2 terminus of titin spans the Z-disc: its interaction with a novel 19-kD ligand (T-cap) is required for sarcomeric integrity. J Cell Biol. 1998;143(4):1013–27.
Obermann WM, Gautel M, Weber K, Fürst DO. Molecular structure of the sarcomeric M band: mapping of titin and myosin binding domains in myomesin and the identification of a potential regulatory phosphorylation site in myomesin. EMBO J. 1997;16(2):211–20.
Hein S, Scholz D, Fujitani N, Rennollet H, Brand T, Friedl A, Schaper J. Altered expression of titin and contractile proteins in failing human myocardium. J Mol Cell Cardiol. 1994;26(10):1291–306.
Hein S, Kostin S, Heling A, Maeno Y, Schaper J. The role of the cytoskeleton in heart failure. Cardiovasc Res. 2000;45(2):273–8.
Gregorio CC, Granzier H, Sorimachi H, Labeit S. Muscle assembly: a titanic achievement? Curr Opin Cell Biol. 1999;11(1):18–25.
Gelfand VI, Bershadsky AD. Microtubule dynamics: mechanism, regulation, and function. Annu Rev Cell Biol. 1991;7:93–116.
Lockard VG, Bloom S. Trans-cellular desmin-lamin B intermediate filament network in cardiac myocytes. J Mol Cell Cardiol. 1993;25(3):303–9.
Heling A, Zimmermann R, Kostin S, Maeno Y, Hein S, Devaux B, Bauer E, Klövekorn WP, Schlepper M, Schaper W, Schaper J. Increased expression of cytoskeletal, linkage, and extracellular proteins in failing human myocardium. Circ Res. 2000;86(8):846–53.
Rappaport L, Samuel JL. Microtubules in cardiac myocytes. Int Rev Cytol. 1988;113:101–43.
Tsutsui H, Ishihara K, Cooper 4th G. Cytoskeletal role in the contractile dysfunction of hypertrophied myocardium. Science. 1993;260(5108):682–7.
Tsutsui H, Tagawa H, Kent RL, McCollam PL, Ishihara K, Nagatsu M, Cooper 4th G. Role of microtubules in contractile dysfunction of hypertrophied cardiocytes. Circulation. 1994;90(1):533–55.
Tagawa H, Wang N, Narishige T, Ingber DE, Zile MR, Cooper 4th G. Cytoskeletal mechanics in pressure-overload cardiac hypertrophy. Circ Res. 1997;80(2):281–9.
Wang X, Li F, Campbell SE, Gerdes AM. Chronic pressure overload cardiac hypertrophy and failure in guinea pigs: II. Cytoskeletal remodeling. J Mol Cell Cardiol. 1999;31(2):319–31.
Milner DJ, Taffet GE, Wang X, Pham T, Tamura T, Hartley C, Gerdes AM, Capetanaki Y. The absence of desmin leads to cardiomyocyte hypertrophy and cardiac dilation with compromised systolic function. J Mol Cell Cardiol. 1999;31(11):2063–76.
Kostin S, Scholz D, Shimada T, Maeno Y, Mollnau H, Hein S, Schaper J. The internal and external protein scaffold of the T-tubular system in cardiomyocytes. Cell Tissue Res. 1998;294(3):449–60.
Ohlendieck K. Towards an understanding of the dystrophin-glycoprotein complex: linkage between the extracellular matrix and the membrane cytoskeleton in muscle fibers. Eur J Cell Biol. 1996;69(1):1–10.
Ortiz-Lopez R, Li H, Su J, Goytia V, Towbin JA. Evidence for a dystrophin missense mutation as a cause of X-linked dilated cardiomyopathy. Circulation. 1997;95(10):2434–40.
Towbin JA. The role of cytoskeletal proteins in cardiomyopathies. Curr Opin Cell Biol. 1998;10(1):131–9.
Towbin JA, Bowles KR, Bowles NE. Etiologies of cardiomyopathy and heart failure. Nat Med. 1999;5(3):266–7.
Klietsch R, Ervasti JM, Arnold W, Campbell KP, Jorgensen AO. Dystrophin-glycoprotein complex and laminin colocalize to the sarcolemma and transverse tubules of cardiac muscle. Circ Res. 1993;72(2):349–60.
Kaprielian RR, Stevenson S, Rothery SM, Cullen MJ, Severs NJ. Distinct patterns of dystrophin organization in myocyte sarcolemma and transverse tubules of normal and diseased human myocardium. Circulation. 2000;101(22):2586–94.
Severs NJ. The cardiac gap junction and intercalated disc. Int J Cardiol. 1990;26(2):137–73.
Smith JH, Green CR, Peters NS, Rothery S, Severs NJ. Altered patterns of gap junction distribution in ischemic heart disease. An immunohistochemical study of human myocardium using laser scanning confocal microscopy. Am J Pathol. 1991;139(4):801–21.
Emdad L, Uzzaman M, Takagishi Y, Honjo H, Uchida T, Severs NJ, Kodama I, Murata Y. Gap junction remodeling in hypertrophied left ventricles of aortic-banded rats: prevention by angiotensin II type 1 receptor blockade. J Mol Cell Cardiol. 2001;33(2):219–31.
Gutstein DE, Morley GE, Tamaddon H, Vaidya D, Schneider MD, Chen J, Chien KR, Stuhlmann H, Fishman GI. Conduction slowing and sudden arrhythmic death in mice with cardiac-restricted inactivation of connexin43. Circ Res. 2001;88(3):333–9.
Lerner DL, Yamada KA, Schuessler RB, Saffitz JE. Accelerated onset and increased incidence of ventricular arrhythmias induced by ischemia in Cx43-deficient mice. Circulation. 2000;101(5):547–52.
Weber KT, Brilla CG, Janicki JS. Myocardial fibrosis: functional significance and regulatory factors. Cardiovasc Res. 1993;27(3):341–8.
Weber KT, Sun Y, Tyagi SC, Cleutjens JP. Collagen network of the myocardium: function, structural remodeling and regulatory mechanisms. J Mol Cell Cardiol. 1994;26(3):279–92.
Rossi MA, Abreu MA, Santoro LB. Images in cardiovascular medicine. Connective tissue skeleton of the human heart: a demonstration by cell-maceration scanning electron microscope method. Circulation. 1998;97(9):934–5.
Weber KT, Pick R, Janicki JS, Gadodia G, Lakier JB. Inadequate collagen tethers in dilated cardiopathy. Am Heart J. 1988;116(6 Pt 1):1641–6.
Gunja-Smith Z, Morales AR, Romanelli R, Woessner Jr JF. Remodeling of human myocardial collagen in idiopathic dilated cardiomyopathy. Role of metalloproteinases and pyridinoline cross-links. Am J Pathol. 1996;148(5):1639–48.
Spinale FG, Tomita M, Zellner JL, Cook JC, Crawford FA, Zile MR. Collagen remodeling and changes in LV function during development and recovery from supraventricular tachycardia. Am J Physiol. 1991;261(2 Pt 2):H308–18.
Florea VG, Anand IS. Troponin T and plasma collagen peptides in heart failure. Circ Heart Fail. 2012;5(4):394–7.
Abrahams C, Janicki JS, Weber KT. Myocardial hypertrophy in Macaca fascicularis. Structural remodeling of the collagen matrix. Lab Invest. 1987;56(6):676–83.
Weber KT, Janicki JS, Shroff SG, Pick R, Chen RM, Bashey RI. Collagen remodeling of the pressure-overloaded, hypertrophied nonhuman primate myocardium. Circ Res. 1988;62(4):757–65.
Weber KT, Brilla CG. Pathological hypertrophy and cardiac interstitium. Fibrosis and renin-angiotensin-aldosterone system. Circulation. 1991;83(6):1849–65.
Schaper J, Speiser B. The extracellular matrix in the failing human heart. Basic Res Cardiol. 1992;87 Suppl 1:303–9.
Kato S, Spinale FG, Tanaka R, Johnson W, Cooper 4th G, Zile MR. Inhibition of collagen cross-linking: effects on fibrillar collagen and ventricular diastolic function. Am J Physiol. 1995;269(3 Pt 2):H863–8.
Stroud JD, Baicu CF, Barnes MA, Spinale FG, Zile MR. Viscoelastic properties of pressure overload hypertrophied myocardium: effect of serine protease treatment. Am J Physiol Heart Circ Physiol. 2002;282(6):H2324–35.
Katz AM, Zile MR. New molecular mechanism in diastolic heart failure. Circulation. 2006;113(16):1922–5.
Zile MR, Baicu CF, Gaasch WH. Diastolic heart failure--abnormalities in active relaxation and passive stiffness of the left ventricle. N Engl J Med. 2004;350(19):1953–9.
Dell’italia LJ, Balcells E, Meng QC, Su X, Schultz D, Bishop SP, Machida N, Straeter-Knowlen IM, Hankes GH, Dillon R, Cartee RE, Oparil S. Volume-overload cardiac hypertrophy is unaffected by ACE inhibitor treatment in dogs. Am J Physiol. 1997;273(2 Pt 2):H961–70.
Spinale FG. Myocardial matrix remodeling and the matrix metalloproteinases: influence on cardiac form and function. Physiol Rev. 2007;87(4):1285–342.
Brew K, Dinakarpandian D, Nagase H. Tissue inhibitors of metalloproteinases: evolution, structure and function. Biochim Biophys Acta. 2000;1477(1-2):267–83.
Nagase H, Visse R, Murphy G. Structure and function of matrix metalloproteinases and TIMPs. Cardiovasc Res. 2006;69(3):562–73.
Sundstrom J, Evans JC, Benjamin EJ, Levy D, Larson MG, Sawyer DB, Siwik DA, Colucci WS, Wilson PW, Vasan RS. Relations of plasma total TIMP-1 levels to cardiovascular risk factors and echocardiographic measures: the Framingham heart study. Eur Heart J. 2004;25(17):1509–16.
Cavusoglu E, Ruwende C, Chopra V, Yanamadala S, Eng C, Clark LT, Pinsky DJ, Marmur JD. Tissue inhibitor of metalloproteinase-1 (TIMP-1) is an independent predictor of all-cause mortality, cardiac mortality, and myocardial infarction. Am Heart J. 2006;151(5):1101.e1–8.
Marino TA, Kent RL, Uboh CE, Fernandez E, Thompson EW, Cooper 4th G. Structural analysis of pressure versus volume overload hypertrophy of cat right ventricle. Am J Physiol. 1985;249(2 Pt 2):H371–9.
Apstein CS, Lecarpentier Y, Mercadier JJ, Martin JL, Pontet F, Wisnewsky C, Schwartz K, Swynghedauw B. Changes in LV papillary muscle performance and myosin composition with aortic insufficiency in rats. Am J Physiol. 1987;253(5 Pt 2):H1005–11.
Silver MA, Pick R, Brilla CG, Jalil JE, Janicki JS, Weber KT. Reactive and reparative fibrillar collagen remodelling in the hypertrophied rat left ventricle: two experimental models of myocardial fibrosis. Cardiovasc Res. 1990;24(9):741–7.
Brilla CG, Maisch B. Regulation of the structural remodelling of the myocardium: from hypertrophy to heart failure. Eur Heart J. 1994;15(Suppl D):45–52.
Brilla CG, Matsubara LS, Weber KT. Anti-aldosterone treatment and the prevention of myocardial fibrosis in primary and secondary hyperaldosteronism. J Mol Cell Cardiol. 1993;25(5):563–75.
Weber KT, Sun Y, Campbell SE. Structural remodelling of the heart by fibrous tissue: role of circulating hormones and locally produced peptides. Eur Heart J. 1995;16(Suppl N):12–8.
Sabbah HN, Goldstein S. Ventricular remodelling: consequences and therapy. Eur Heart J. 1993;14(Suppl C):24–9.
Douglas PS, Morrow R, Ioli A, Reichek N. Left ventricular shape, afterload and survival in idiopathic dilated cardiomyopathy. J Am Coll Cardiol. 1989;13(2):311–5.
Saks VA, Belikova YO, Kuznetsov AV, Khuchua ZA, Branishte TH, Semenovsky ML, Naumov VG. Phosphocreatine pathway for energy transport: ADP diffusion and cardiomyopathy. Am J Physiol. 1991;261(4 Suppl):30–8.
Kono T, Sabbah HN, Rosman H, Alam M, Jafri S, Goldstein S. Left ventricular shape is the primary determinant of functional mitral regurgitation in heart failure. J Am Coll Cardiol. 1992;20(7):1594–8.
Florea VG, Henein MY, Anker SD, Francis DP, Gibson DG, Coats AJ. Relation of changes over time in ventricular size and function to those in exercise capacity in patients with chronic heart failure. Am Heart J. 2000;139(5):913–7.
Cohn JN, Johnson GR, Shabetai R, Loeb H, Tristani F, Rector T, Smith R, Fletcher R. Ejection fraction, peak exercise oxygen consumption, cardiothoracic ratio, ventricular arrhythmias, and plasma norepinephrine as determinants of prognosis in heart failure. Circulation. 1993;87(6 Suppl):VI5–16.
Meerson FZ. Compensatory hyperfunction of the heart and cardiac insufficiency. Circ Res. 1962;10:250–8.
Gaudron P, Eilles C, Kugler I, Ertl G. Progressive left ventricular dysfunction and remodeling after myocardial infarction. Potential mechanisms and early predictors. Circulation. 1993;87(3):755–63.
Sharpe N, Smith H, Murphy J, Greaves S, Hart H, Gamble G. Early prevention of left ventricular dysfunction after myocardial infarction with angiotensin-converting-enzyme inhibition. Lancet. 1991;337(8746):872–6.
Dargie HJ. Effect of carvedilol on outcome after myocardial infarction in patients with left-ventricular dysfunction: the CAPRICORN randomised trial. Lancet. 2001;357(9266):1385–90.
Doughty RN, Whalley GA, Walsh H, et al. Effects of carvedilol on left ventricular remodeling in patients following acute myocardial infarction: the CAPRICORN echo substudy. Circulation. 2001;104 (Suppl.)(17):II-517. Abstract.
The ACE Inhibitor Myocardial Infarction Collaborative Group. Indications for ACE inhibitors in the early treatment of acute myocardial infarction: systematic overview of individual data from 100,000 patients in randomized trials. Circulation. 1998;97(22):2202–12.
Effect of ramipril on mortality and morbidity of survivors of acute myocardial infarction with clinical evidence of heart failure. The Acute Infarction Ramipril Efficacy (AIRE) Study Investigators. Lancet. 1993;342(8875):821–8.
Buch P, Rasmussen S, Abildstrom SZ, Kober L, Carlsen J, Torp-Pedersen C, TRACE investigators. The long-term impact of the angiotensin-converting enzyme inhibitor trandolapril on mortality and hospital admissions in patients with left ventricular dysfunction after a myocardial infarction: follow-up to 12 years. Eur Heart J. 2005;26(2):145–52.
Grossman W. Cardiac hypertrophy: useful adaptation or pathologic process? Am J Med. 1980;69(4):576–84.
Katz AM. Cardiomyopathy of overload. A major determinant of prognosis in congestive heart failure. N Engl J Med. 1990;322(2):100–10.
Raya TE, Fonken SJ, Lee RW, Daugherty S, Goldman S, Wong PC, Timmermans PB, Morkin E. Hemodynamic effects of direct angiotensin II blockade compared to converting enzyme inhibition in rat model of heart failure. Am J Hypertens. 1991;4(4 Pt 2):334S–40.
Schieffer B, Wirger A, Meybrunn M, Seitz S, Holtz J, Riede UN, Drexler H. Comparative effects of chronic angiotensin-converting enzyme inhibition and angiotensin II type 1 receptor blockade on cardiac remodeling after myocardial infarction in the rat. Circulation. 1994;89(5):2273–82.
McDonald KM, Rector T, Carlyle PF, Francis GS, Cohn JN. Angiotensin-converting enzyme inhibition and beta-adrenoceptor blockade regress established ventricular remodeling in a canine model of discrete myocardial damage. J Am Coll Cardiol. 1994;24(7):1762–8.
Tamura T, Said S, Harris J, Lu W, Gerdes AM. Reverse remodeling of cardiac myocyte hypertrophy in hypertension and failure by targeting of the renin-angiotensin system. Circulation. 2000;102(2):253–9.
Xu X, Wan W, Ji L, Lao S, Powers AS, Zhao W, Erikson JM, Zhang JQ. Exercise training combined with angiotensin II receptor blockade limits post-infarct ventricular remodelling in rats. Cardiovasc Res. 2008;78(3):523–32.
Konstam MA, Kronenberg MW, Rousseau MF, Udelson JE, Melin J, Stewart D, Dolan N, Edens TR, Ahn S, Kinan D, Howe DM, Kilcoyne L, Metherall J, Benedict C, Yusuf S, Pouleur H; for the SOLVD Investigators. Effects of the angiotensin converting enzyme inhibitor enalapril on the long-term progression of left ventricular dilatation in patients with asymptomatic systolic dysfunction. SOLVD (Studies of Left Ventricular Dysfunction) Investigators. Circulation. 1993;88(5 Pt 1):2277–83.
Konstam MA, Rousseau MF, Kronenberg MW, Udelson JE, Melin J, Stewart D, Dolan N, Edens TR, Ahn S, Kinan D, et al. Effects of the angiotensin converting enzyme inhibitor enalapril on the long-term progression of left ventricular dysfunction in patients with heart failure. Circulation. 1992;86(2):431–8.
St. John Sutton M, Pfeffer MA, Plappert T, Rouleau JL, Moyé LA, Dagenais GR, Lamas GA, Klein M, Sussex B, Goldman S, et al. Quantitative two-dimensional echocardiographic measurements are major predictors of adverse cardiovascular events after acute myocardial infarction. The protective effects of captopril. Circulation. 1994;89(1):68–75.
Hall SA, Cigarroa CG, Marcoux L, Risser RC, Grayburn PA, Eichhorn EJ. Time course of improvement in left ventricular function, mass and geometry in patients with congestive heart failure treated with beta-adrenergic blockade. J Am Coll Cardiol. 1995;25(5):1154–61.
Sabbah HN, Sharov VG, Gupta RC, Todor A, Singh V, Goldstein S. Chronic therapy with metoprolol attenuates cardiomyocyte apoptosis in dogs with heart failure. J Am Coll Cardiol. 2000;36(5):1698–705.
Hayashi M, Tsutamoto T, Wada A, Tsutsui T, Ishii C, Ohno K, Fujii M, Taniguchi A, Hamatani T, Nozato Y, Kataoka K, Morigami N, Ohnishi M, Kinoshita M, Horie M. Immediate administration of mineralocorticoid receptor antagonist spironolactone prevents post-infarct left ventricular remodeling associated with suppression of a marker of myocardial collagen synthesis in patients with first anterior acute myocardial infarction. Circulation. 2003;107(20):2559–65.
Tsutamoto T, Wada A, Maeda K, Mabuchi N, Hayashi M, Tsutsui T, Ohnishi M, Sawaki M, Fujii M, Matsumoto T, Matsui T, Kinoshita M. Effect of spironolactone on plasma brain natriuretic peptide and left ventricular remodeling in patients with congestive heart failure. J Am Coll Cardiol. 2001;37(5):1228–33.
Pitt B, Reichek N, Willenbrock R, Zannad F, Phillips RA, Roniker B, Kleiman J, Krause S, Burns D, Williams GH. Effects of eplerenone, enalapril, and eplerenone/enalapril in patients with essential hypertension and left ventricular hypertrophy: the 4E-left ventricular hypertrophy study. Circulation. 2003;108(15):1831–8.
Chan AK, Sanderson JE, Wang T, Lam W, Yip G, Wang M, Lam YY, Zhang Y, Yeung L, Wu EB, Chan WW, Wong JT, So N, Yu CM. Aldosterone receptor antagonism induces reverse remodeling when added to angiotensin receptor blockade in chronic heart failure. J Am Coll Cardiol. 2007;50(7):591–6.
Cohn JN, Tognoni G, Valsartan Heart Failure Trial Investigators. A randomized trial of the angiotensin-receptor blocker valsartan in chronic heart failure. N Engl J Med. 2001;345(23):1667–75.
Maggioni AP, Anand I, Gottlieb SO, Latini R, Tognoni G, Cohn JN, Val-HeFT Investigators (Valsartan Heart FailureTrial). Effects of valsartan on morbidity and mortality in patients with heart failure not receiving angiotensin-converting enzyme inhibitors. J Am Coll Cardiol. 2002;40(8):1414–21.
Wong M, Staszewsky L, Latini R, Barlera S, Volpi A, Chiang YT, Benza RL, Gottlieb SO, Kleemann TD, Rosconi F, Vandervoort PM, Cohn JN, Val-HeFT Heart Failure Trial Investigators. Valsartan benefits left ventricular structure and function in heart failure: Val-HeFT echocardiographic study. J Am Coll Cardiol. 2002;40(5):970–5.
Wong M, Staszewsky L, Latini R, Barlera S, Glazer R, Aknay N, Hester A, Anand I, Cohn JN. Severity of left ventricular remodeling defines outcomes and response to therapy in heart failure: Valsartan heart failure trial (Val-HeFT) echocardiographic data. J Am Coll Cardiol. 2004;43(11):2022–7.
Lindholm LH, Ibsen H, Dahlof B, Devereux RB, Beevers G, de Faire U, Fyhrquist F, Julius S, Kjeldsen SE, Kristiansson K, Lederballe-Pedersen O, Nieminen MS, Omvik P, Oparil S, Wedel H, Aurup P, Edelman J, Snapinn S, LIFE Study Group. Cardiovascular morbidity and mortality in patients with diabetes in the Losartan Intervention For Endpoint reduction in hypertension study (LIFE): a randomised trial against atenolol. Lancet. 2002;359(9311):1004–10.
Cintron G, Johnson G, Francis G, Cobb F, Cohn JN. Prognostic significance of serial changes in left ventricular ejection fraction in patients with congestive heart failure. The V-HeFT VA Cooperative Studies Group. Circulation. 1993;87(6 Suppl):VI17–23.
Cohn JN, Tam SW, Anand IS, Taylor AL, Sabolinski ML, Worcel M, A-HeFT Investigators. Isosorbide dinitrate and hydralazine in a fixed-dose combination produces further regression of left ventricular remodeling in a well-treated black population with heart failure: results from A-HeFT. J Card Fail. 2007;13(5):331–9.
Taylor AL, Ziesche S, Yancy C, Carson P, D’Agostino Jr R, Ferdinand K, Taylor M, Adams K, Sabolinski M, Worcel M, Cohn JN, African-American Heart Failure Trial Investigators. Combination of isosorbide dinitrate and hydralazine in blacks with heart failure. N Engl J Med. 2004;351(20):2049–57.
Cleland JG, Daubert JC, Erdmann E, Freemantle N, Gras D, Kappenberger L, Tavazzi L, Cardiac Resynchronization-Heart Failure (CARE-HF) Study Investigators. The effect of cardiac resynchronization on morbidity and mortality in heart failure. N Engl J Med. 2005;352(15):1539–49.
Linde C, Leclercq C, Rex S, Garrigue S, Lavergne T, Cazeau S, McKenna W, Fitzgerald M, Deharo JC, Alonso C, Walker S, Braunschweig F, Bailleul C, Daubert JC. Long-term benefits of biventricular pacing in congestive heart failure: results from the MUltisite STimulation in cardiomyopathy (MUSTIC) study. J Am Coll Cardiol. 2002;40(1):111–8.
Molhoek SG, Bax JJ, van Erven L, Bootsma M, Boersma E, Steendijk P, van der Wall EE, Schalij MJ. Comparison of benefits from cardiac resynchronization therapy in patients with ischemic cardiomyopathy versus idiopathic dilated cardiomyopathy. Am J Cardiol. 2004;93(7):860–3.
Sutton MG, Plappert T, Hilpisch KE, Abraham WT, Hayes DL, Chinchoy E. Sustained reverse left ventricular structural remodeling with cardiac resynchronization at one year is a function of etiology: quantitative Doppler echocardiographic evidence from the Multicenter InSync Randomized Clinical Evaluation (MIRACLE). Circulation. 2006;113(2):266–72.
Sutton MS, Keane MG. Reverse remodelling in heart failure with cardiac resynchronisation therapy. Heart. 2007;93(2):167–71.
Fruhwald FM, Fahrleitner-Pammer A, Berger R, Leyva F, Freemantle N, Erdmann E, Gras D, Kappenberger L, Tavazzi L, Daubert J-C, Cleland JGF. Early and sustained effects of cardiac resynchronization therapy on N-terminal pro-B-type natriuretic peptide in patients with moderate to severe heart failure and cardiac dyssynchrony. Eur Heart J. 2007;28(13):1592–7.
Chaudhry PA, Mishima T, Sharov VG, Hawkins J, Alferness C, Paone G, Sabbah HN. Passive epicardial containment prevents ventricular remodeling in heart failure. Ann Thorac Surg. 2000;70(4):1275–80.
Saavedra WF, Tunin RS, Paolocci N, Mishima T, Suzuki G, Emala CW, Chaudhry PA, Anagnostopoulos P, Gupta RC, Sabbah HN, Kass DA. Reverse remodeling and enhanced adrenergic reserve from passive external support in experimental dilated heart failure. J Am Coll Cardiol. 2002;39(12):2069–76.
Sabbah HN, Sharov VG, Gupta RC, Mishra S, Rastogi S, Undrovinas AI, Chaudhry PA, Todor A, Mishima T, Tanhehco EJ, Suzuki G. Reversal of chronic molecular and cellular abnormalities due to heart failure by passive mechanical ventricular containment. Circ Res. 2003;93(11):1095–101.
Blom AS, Mukherjee R, Pilla JJ, Lowry AS, Yarbrough WM, Mingoia JT, Hendrick JW, Stroud RE, McLean JE, Affuso J, Gorman RC, Gorman 3rd JH, Acker MA, Spinale FG. Cardiac support device modifies left ventricular geometry and myocardial structure after myocardial infarction. Circulation. 2005;112(9):1274–83.
Blom AS, Pilla JJ, Arkles J, Dougherty L, Ryan LP, Gorman 3rd JH, Acker MA, Gorman RC. Ventricular restraint prevents infarct expansion and improves borderzone function after myocardial infarction: a study using magnetic resonance imaging, three-dimensional surface modeling, and myocardial tagging. Ann Thorac Surg. 2007;84(6):2004–10.
Pilla JJ, Blom AS, Gorman 3rd JH, Brockman DJ, Affuso J, Parish LM, Sakamoto H, Jackson BM, Acker MA, Gorman RC. Early postinfarction ventricular restraint improves borderzone wall thickening dynamics during remodeling. Ann Thorac Surg. 2005;80(6):2257–62.
Mann DL, Acker MA, Jessup M, Sabbah HN, Starling RC, Kubo SH, Acorn Trial Principal Investigators and Study Coordinators. Clinical evaluation of the CorCap Cardiac Support Device in patients with dilated cardiomyopathy. Ann Thorac Surg. 2007;84(4):1226–35.
Starling RC, Jessup M, Oh JK, Sabbath HN, Acker MA, Mann DL, Kubo SH. Sustained benefits of the CorCap Cardiac Support Device on left ventricular remodeling: three year follow-up results from the Acorn clinical trial. Ann Thorac Surg. 2007;84(4):1236–42.
Klodell Jr CT, Aranda Jr JM, McGiffin DC, Rayburn BK, Sun B, Abraham WT, Pae Jr WE, Boehmer JP, Klein H, Huth C. Worldwide surgical experience with the Paracor HeartNet cardiac restraint device. J Thorac Cardiovasc Surg. 2008;135(1):188–95.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing AG
About this chapter
Cite this chapter
Anand, I.S., Florea, V.G. (2017). Structural Remodeling in the Development of Chronic Systolic Heart Failure: Implication for Treatment. In: Garry, D., Wilson, R., Vlodaver, Z. (eds) Congestive Heart Failure and Cardiac Transplantation. Springer, Cham. https://doi.org/10.1007/978-3-319-44577-9_17
Download citation
DOI: https://doi.org/10.1007/978-3-319-44577-9_17
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-44575-5
Online ISBN: 978-3-319-44577-9
eBook Packages: MedicineMedicine (R0)