Abstract
Functional dyspepsia (FD) is a functional gastrointestinal disorder characterized by pain or discomfort in the upper abdomen. Symptoms of dyspepsia are frequently, but not always, associated with the ingestion of a meal. Common symptoms associated with FD also include fullness, early satiety, and nausea. FD may coexist with other disorders such as IBS, gastroparesis, and gastroesophageal reflux disease which sometimes complicates its diagnosis. The recognition of this common overlap is important in order to limit unnecessary diagnostic testing and initiate directed therapy. The diagnosis of FD is mainly clinical and based on the Rome IV criteria. Treatment of FD relies on the biopsychosocial model and may consist of education about the disorder as well as pharmacologic, psychologic, and complementary therapies.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
- Functional dyspepsia
- Rome IV criteria
- Dyspepsia, functional
- Functional abdominal pain disorder
- Irritable bowel syndrome
- Chronic upper abdominal pain
The understanding of pathophysiology and clinical characteristics of functional dyspepsia (FD ) have greatly evolved over the last several years. FD is a functional abdominal pain disorder (FAPD) characterized by pain or discomfort in the upper abdomen. Symptoms of dyspepsia are frequently, but not always, associated with the ingestion of a meal. Common symptoms associated with FD also include fullness, early satiety, and nausea [1]. A distinct characteristic that differentiates the bothersome symptoms of FD from irritable bowel syndrome (IBS) is the lack of association between the child’s pain/discomfort with changes in bowel movement frequency or consistency. An important consideration is that the symptoms of dyspepsia may coexist with other disorders such as IBS, gastroparesis, and gastroesophageal reflux disease (GERD ) which sometimes complicates its diagnosis. The recognition of this common overlap is important at the time of diagnosis in order to limit unnecessary diagnostic testing and initiate directed therapy. The diagnosis of FD is mainly clinical and based on the Rome IV criteria.
Epidemiology/Impact
Chronic abdominal pain is common in children with a subgroup of these children meeting criteria for a FAPD. Recent epidemiologic studies have shown a high prevalence of FAPDs in children of South, Central, and North America, Europe, Asia, and Africa [2–9]. Prevalence of FAPDs varies among countries, but generally falls in the range of 10–20 %. A school-based study conducted in Colombia found a prevalence of FD of 7 % [2] while another study conducted in Nigeria found that only 0.4 % of school children had FD [8]. Most children who do not meet strict criteria for a FAPD fail to meet the minimum frequency of symptoms established by the Rome III criteria (more than once monthly but less than once weekly) [10]. A US epidemiological study found that 1.4 % of children had pain or discomfort in the upper abdomen weekly, but only 0.2 % met the Rome III criteria for the diagnosis of FD [11]. FD is typically identified with lower prevalence than IBS, with overlap of the two disorders frequently occurring in adults [12] and children [13].
Chronic abdominal pain is likely under-recognized or underreported, with only 2–3 % of children having weekly pain seeking medical care [2, 14]. Despite the low ratio of consultation, FAPDs are among the most common causes of consultation to pediatric gastroenterologists. A study of new patients presenting to a single pediatric gastroenterology clinic found that functional gastrointestinal disorders (FGIDs) were the most common reason for consultation. Seven percent of children and adolescents who consulted for FGIDs were diagnosed with FD [15].
Disability related to FAPDs is substantial, with reduced quality of life for affected children. Quality of life of children with FAPDs is even lower than in children with organic gastrointestinal disease including inflammatory bowel disease (IBD) [16]. Children with FAPDs frequently miss school and have increased anxiety, depression, and worry [14, 17] as well as sleep problems [18].
The management of FAPDs has a substantial impact on health care expenses. Total annual costs associated with management of some of the most common FAPDs in children in the Netherlands are estimated to be €2512 per patient [19]. The diagnostic workup for FGIDs has been estimated to be approximately $6000 per child [20]. Cost of care of children with FGIDs has greatly increased over the last decade and is likely to continue to increase. The health care costs associated with the management of FGIDs tripled from 1997 to 2009 [21]. These enormous costs do not account for indirect costs such as work absenteeism and over the counter medications.
Etiology
FD should be considered in the context of the biopsychosocial model that proposes that the development, persistence, and exacerbation of symptoms result from the interplay of multiple factors including early life events. The triggering event of FD is only occasionally uncovered, however it is not uncommon for a child to associate the onset of symptoms following an acute gastrointestinal infection [22]. The fact that not all children who suffer an infection develop symptoms of dyspepsia and that children with a history of gastrointestinal infection may present with different intensity of symptoms, disability, and comorbidities illustrates the importance of nonorganic factors such as the psychosocial milieu and the subject’s and parents’ coping skills.
The presence of a genetic predisposition may help explain the phenotypic variation in symptoms of dyspepsia. Many genetic polymorphisms have been postulated as risk factors for FD [23, 24]. However, these potential hereditary factors are not yet well understood and their role and importance is still undefined [25].
Early life events increase the risk of FAPDs including FD. Early adverse events that have been associated with an increased risk of FAPDs in children include surgical procedures [26, 27], inflammatory intestinal disease [28], cow’s milk protein allergy [29], infections [22, 30], child abuse [31], and war exposure [32]. Children may be more susceptible to the effects of gastroenteritis due to their immaturity of the immune response to pathogens, intestinal barrier, and enteric nervous system. Gastrointestinal and non-gastrointestinal infections in adulthood (respiratory infection, cellulitis, urinary tract infection) [33] have also been associated with an increased risk of FAPDs. The effect of infections in children transcends the pediatric period. Children who had a Salmonella spp . infection have a greater likelihood of developing FAPDs as adults [34]. Parasitic infections (e.g., Giardia lamblia) [35] and the use of antibiotics have also been associated with the development of FAPDs [36]. However, this is not the case of every infection as pathogens such as H. pylori [37] and D. fragilis [38] do not increase the risk of developing FAPDs.
The study of the microbiome and the post-inflammatory effects of infection are emerging as an exciting and evolving field. The mechanisms that link gastrointestinal infections to the development of FD are incompletely understood. Alterations of intestinal microbes [39] may play a role in the pathogenesis of post-infectious FAPDs and post-antibiotic-use associated FAPDs. There is a complex cross-talk between the brain, gut, and microbiome. Anxiety, depression, somatization, and catastrophizing frequently precede or coexist with FAPD symptoms (including FD) and are often associated with symptom severity and treatment outcomes [40–43]. Acute stress has been shown to result in changes in microbiome, reduced number of CD4(+) T lymphocytes, and increased mast cell degranulation [44, 45]. Psychological stress affects visceral sensation and intestinal immune reaction [46, 47]. Studies have shown a bidirectional relation between the gut flora and the central nervous system (brain–gut–microbiota axis). Stress affects the intestinal microbiome composition [48] which in turn influences intestinal inflammation and permeability [49]. Stress activates the hypothalamic–pituitary–adrenal (HPA) axis and sympathetic nervous system. This results in increased intestinal permeability and a greater influx of antigens across the intestinal epithelial barrier that activates an immune response affecting the microbiome [50]. Animal studies have also shown that alterations in gut microbiota influence social and emotional behaviors [48, 51].
FAPDs frequently overlap with other functional or organic disease and the overlap is increasingly recognized in adults and children. Children with FD are prone to have poor sleep, symptoms of orthostatic dysregulation, and headache [52]. The presence of comorbid functional disorders in a child with organic disease may confound the diagnosis or activity state of the organic disease, and affect treatment and overall disability. Children with active IBD frequently consult for gastrointestinal symptoms that may mimic a FAPD. Some of the medications used in the treatment of IBD may also lead to dyspeptic symptoms [53]. Eight to thirteen percent of children with IBD in remission also have gastrointestinal symptoms of functional origin [54, 55]. Children with IBD and FAPDs have increased rates of depression compared to children with IBD in remission who do not have FAPDs [55]. Other organic conditions have also been associated with increased rates of FAPDs in children. Some pediatric reports have associated celiac disease with FAPDs [56]. Nevertheless, the association between celiac disease and FAPDs remains controversial with one study showing no change in risk of IBS in children with celiac disease [57] while another study found a fourfold higher risk [56]. The long-term effect of celiac disease in the development of FGIDs in children is also a matter of controversy. While a study has shown that children with celiac disease on gluten-free diet are not at increased risk of developing FAPDs [58], another pediatric study found an increased risk of FGIDs with preponderance of functional constipation [59].
Weight excess has been associated with an increased prevalence of FGIDs [60] and poor treatment outcomes in children [61]. Forty-seven percent of obese/overweight children have FAPDs, nearly twice the rate of normal-weight controls [62]. Studies in adults have found that a high visceral adiposity and a high ratio of visceral to subcutaneous adiposity were associated with FD [63].
In adults the relation between BMI and the presence of FD is influenced by gender with females who are underweight or obese having a greater likelihood of FD [64]. Obese children with FAPDs also have more severe pain and associated disability than normal weight children [61].
Pathophysiology
Intake of solid foods is associated with changes in gastric motor activity including fundic accommodation and antral grinding. Under normal circumstances these processes do not result in noxious symptoms. However, alterations in the peripheral or central nervous system or abnormal motor activity can result in the perception of pain or discomfort. Multiple pathophysiologic mechanisms have been implicated in the development of symptoms in patients with FD, including visceral hypersensitivity, electromechanical dysfunction, and eosinophilic mucosal inflammation. Understanding the mechanisms behind each patient’s symptoms would be instrumental to help direct treatment. However, alterations in several mechanisms frequently occur in a single patient, complicating the testing and understanding of FD pathophysiology [65].
Abnormal visceral perception may result in abdominal pain, postprandial fullness, early satiety, or nausea. Patients with visceral hypersensitivity are prone to develop early satiety and abdominal pain with lower volumes of gastric distension. There are numerous techniques with different degrees of invasiveness used to assess upper gastrointestinal visceral hypersensitivity in the research setting, some of which are too invasive or cumbersome to be used in daily clinical care. One of these techniques includes the use of the barostat, an infinitely compliant bag that allows measuring organ pressures and compliance, and relates these values to symptom perception. The technique requires that a balloon connected to a computer be positioned in the proximal stomach allowing the recording of intraballoon volume at a fixed pressure to measure fundic tone. Evaluation of FD using gastric and rectal barostats identified predominant gastric hypersensitivity in FD, compared to predominant rectal hypersensitivity in IBS [66], although this specificity was not replicated in all studies. Barostat studies in children with FD have shown an organ-specific heightened perception to gastric distension [67] with onset of discomfort at lower distension pressures than healthy controls [68]. Although the use of a barostat is a helpful technique to understand pathophysiologic mechanisms of FD, the use of the barostat in children is cumbersome and invasive. Thus, alternative techniques have been developed such as drinking tests that are more suitable to be used in pediatric patients. A practical example of the drinking test is the water load test. Two different techniques have been frequently used for the water load test in children; children are either asked to drink water at a fixed rate until the child reports feeling full, or children are invited to drink water ad libitum for a fixed amount of time (3–5 min) with the volume being then measured. Studies with the water load test in adults reproduce dyspepsia symptoms and have shown lower maximum tolerated volumes [69, 70]. Few studies have investigated its use in children. Sood et al. established normal values for the water load test in healthy children that seemed to pave the way for use of this test to differentiate healthy children from those with FD [71]. In this protocol, children were instructed to drink as much water as possible for 3 min (rapid drinking) or until full. A later study validated the water load provocation test by correlating symptom questionnaires with the results of the test in healthy school children and children with functional abdominal pain [72]. However, a subsequent study put into question its use by suggesting that the test had poor sensitivity (62 %) and specificity (40 %) to correctly diagnose FD in children [73]. Still, the study found that children with chronic abdominal pain ingested a lower volume of water than healthy controls, a finding that is in line with the results of barostat studies. A matter of criticism of the water load test is that in the absence of feedback inhibition, as is likely to occur with the fixed time/3 min protocol, rapid gastric emptying of water could influence the results by eliminating the filling of the proximal stomach (although according to the authors this should for an error of less than 10 % of the ingested volume). Lack of filling would also make it an inaccurate test to measure gastric accommodation. In fact in adults, the results of the measurement of accommodation with the rapid drinking water test do not correlate with results obtained with the barostat [70] which is considered the gold standard to investigate proximal stomach function.
Other techniques such as the assessment of chemosensitivity to intestinal infusion have been used to assess hypersensitivity in adults with FD, but not in children. Studies using this technique have shown that duodenal and gastric acid infusion result in increased symptoms in FD patients [74, 75] while lipid infusion increases sensitivity to gastric distension and increases nausea compared to healthy controls [76].
Electromechanical Dysfunction
Visceral sensation and gastric motor function are interdependent, but this and other reviews consider them as independent factors in order to present a more clear description and understanding of the pathophysiology of dyspepsia. Normal gastric motor function serves to accommodate the large volume of a meal without resulting in discomfort (fundic accommodation) and to prepare food for digestion and initiate passage of chyme through the small intestine. Alteration of the gastric function including impaired meal induced relaxation of the fundus [77], delayed gastric emptying, altered antroduodenal motility, and gastric electrical rhythm disturbances have been implicated in the pathogenesis of dyspeptic symptoms [65].
As previously mentioned, gastric barostat testing is a reliable but invasive and stressful instrument for the assessment of gastric accommodation. Nutrient drinking tests are a noninvasive method to estimate meal-induced gastric accommodation. The test adequately correlates with barostat studies of gastric accommodation in adults [78]. In this slow drinking isocaloric satiety test, children are given a liquid meal that will provoke meal-induced gastric accommodation. Children are instructed to drink the nutrient meal (i.e., isocaloric liquid test meal—Nutridrink, Nutricia, Bornem, Belgium) at a set rate and score their satiety on a scale. In the Hoffman et al. protocol, children were asked to grade satiety from 0 to 5 and to stop drinking when they reach a score of 5 [79]. A study using this protocol found that 93 % of children with FD had a decreased nutrient drinking capacity compared with healthy age matched controls. The impaired nutrient drinking capacity is thought to be due to poor accommodation although this has not yet been definitively proven. Children with FD had a higher satiety score than healthy controls who in turn had a higher satiety score than children who were obese. Normal values for the satiety drinking test have been published by the same group for children 5–15 years [80]. A recent study in adults comparing the drinking test with an objective measure of accommodation (Single Photon Emission Computed Tomography, SPECT ) in community subjects with FD puts into question the reliability of the test. The study showed that the maximum tolerated volume of the nutrient test did not reflect gastric volume measurements in healthy controls or FD subjects [81].
Other tests have also been used in the evaluation of accommodation in pediatric patients. A study of children with chronic abdominal pain utilized ultrasound to demonstrate impairments in antral relaxation, proximal filling, and gastric liquid distribution [82]. SPECT assesses fundic accommodation using intravenous injection of radiolabelled 99mTc pertechnetate that accumulates in the gastric mucosa and allows the visualization of the stomach and changes in postprandial volume. However, this technique requires radiation and it is considered less ideal than the gastric barostat for assessing changes in gastric tone [83]. Using SPECT and MRI , 40–50 % of adults with FD demonstrated abnormal postprandial gastric volume and impaired accommodation [84, 85]. A study with SPECT showed that children with FD had decreased postprandial gastric volume change [86]. However, the correlation between accommodation and symptoms is inconsistent.
A subset of children with FD has delayed gastric emptying. Specific mechanisms of delayed gastric emptying in FD may include altered ghrelin physiology [87, 88], antral mast cell density [89], or other sources of immune activation [90]. Constipation, through the putative colo-gastric brake, was also implicated in delayed gastric emptying in FD patients [91]. Gastric emptying rate in pediatric FD has been evaluated by several modalities. Four-hour gastric scintigraphy, 13C-s platensis and 13C-octanoic breath tests, and ultrasound evaluation have demonstrated delayed emptying in a subset of pediatric FD patients [79, 86, 92, 93]. Friesen et al., using an isotope labeled solid meal, found that 47 % of children with FD had slow GE in the first hour of the meal [94]. Hoffman et al. measured gastric emptying with a breath hydrogen test and found that as a group, children with FD had slower gastric emptying [79]. However, only 26 % had an abnormally delayed gastric emptying rate. As in adult studies, there is not consistent correlation of emptying rate with reported symptom severity or satiety in children [92, 94].
Gastric myoelectrical activity was found to be altered in some children with FD. Up to 50 % of children with FD have abnormal electrogastrography (EGG ) with symptoms that correlate with the abnormalities found [94, 95]. Specific abnormalities include decreased slow waves and less rhythmic activity time in both fasting and fed states [96]. Adult FD patients have similar EGG findings and also demonstrate good symptom correlation [97, 98]. Abnormal EGGs seem independent of chronic gastritis, but are associated with antral eosinophil and mast cell density in children [89, 99]. The stress response may also alter gastric myoelectrical activity [100] through HPA axis mediated pathways. A problem of the EGG technique is the low reliability of the test and the number of artifacts that are seen during the study.
In summary, the study of alterations in the electrical and motor characteristics of children with FD is important to advance the understanding of the pathophysiology of FD, but a common problem with most of the techniques is inconsistent association between symptoms and findings. A possible explanation for some of the poor correlation between symptoms and findings is the multifactorial nature of FD. Prandial state, nutrient type, mucosal inflammation, autonomic, enteric, and central neural input, distal intestinal motor function, presence of constipation, and regulatory hormones all have the potential to affect filling and emptying of the stomach as well as perception of associated symptoms. Abnormalities of any of the various motor functions of the stomach can manifest with different severity of symptoms depending on the level of visceral hypersensitivity, anxiety, or hyperawareness.
Intestinal Inflammation
Intestinal inflammation is increasingly recognized as a potential pathophysiologic mechanism in FD [101–103]. The mechanism by which these inflammatory cells relate to dyspeptic symptoms remains unclear. A possible link between inflammation and symptoms is through motor or visceral sensory alterations. Although still preliminary, some data suggests that impaired intestinal mucosal barrier function may be related to low-level inflammation identified in patients with FD [104]. Intestinal mast cell and eosinophils are increased in some adults with IBS and FD [105, 106]. Children with FD were shown to have increased antral mast cell degranulation, and antral mast cell density was correlated with gastric dysrhythmias and delayed gastric emptying [89]. Normative data for mucosal eosinophil numbers is controversial, but location, number, and extent of degranulation are important considerations. Pediatric FD patients were found to have moderate to extensive duodenal eosinophil degranulation on electron microscopy in a small study [107]. Similar results were found in adults with FD [106]. Increased numbers of duodenal mucosal eosinophils are also demonstrated in several pediatric studies of both FD and unspecified chronic abdominal pain [108, 109].
Altered Peripheral and Central Nervous System
Alterations in peripheral and central nervous system function may also contribute to FD. Adult FD patients were shown to have functional and structural abnormalities in duodenal submucosal ganglia using live imaging techniques [110]. It is possible that localized inflammation is responsible for these neuronal alterations [111]. Central nervous alterations in FD and other chronic pain conditions are increasingly identified using advanced imaging techniques [112]. Examples of differences between FD adults and healthy controls include altered structure in multiple brain regions (posterior insula most significantly) by MR evaluation [113], and positron emission tomography showing upregulation of serotonin transporter (SERT) in the midbrain and thalamus [114] as well as increased cannabinoid-1 receptor availability [115].
Evaluation and Diagnosis
The symptoms of FD cannot be explained by structural, metabolic, or inflammatory disease. Pain or an uncomfortable sensation in the upper abdomen is a hallmark of functional dyspepsia, but multiple other symptoms may coexist or predominate. Nausea, early satiety, fullness, and bloating are frequently reported, and many patients will have more than one symptom. Hoffman et al., using the nutrient drink test, found that the most common dyspeptic symptoms in children with FD were early satiety (96 %), postprandial fullness (89 %), epigastric pain (79 %), and nausea (50 %) [79]. Vomiting and belching were present in only few children. Another study in a small group of children who met Rome III criteria for FD found that in addition to epigastric pain, 65 % of children manifested nausea, 63 % early satiety, and 59 % had bloating and postprandial fullness [11]. In agreement with these findings, Kovacic et al. have found that a large proportion of children with FD have nausea [1]. Pain is the only symptom among those reported in these studies that is part of the Rome III pediatric criteria diagnosis of FD. The new edition (Rome IV) of the Rome criteria addresses some of these shortcomings (Table 36.1) [116].
The pediatric FD Rome IV criteria now include two subtypes, postprandial distress (PDS ) and epigastric pain syndrome (EPS ). These FD subtypes previously described only in adults may be identified in pediatrics [117]. Turco et al. found that 29 % of children with FD met criteria for the adult Rome III diagnosis of postprandial distress syndrome, 24 % met the criteria or the diagnosis of epigastric pain syndrome, and 26 % met the criteria for both conditions. Only 21 % of children did not meet any of the diagnoses. PDS and EPS are shown to associate with variable outcomes and response to treatment in the adult population.
Given the variety of upper abdominal symptoms that can encompass FD and their possible overlap with other conditions, the consideration of a broad differential diagnosis appropriate to the clinical history and exam is warranted (Table 36.2). Delayed gastric emptying may exist in a subset of children with FD, and relatively common disorders such as lactose intolerance or GERD may cause upper intestinal symptoms similar to FD. Children with small bowel intestinal overgrowth may have loss of appetite and belching that may also be seen in patients with FD [118]. Multiple comorbid symptoms are sometimes recognized in association with FD and other FAPDs including joint hypermobility, postural orthostatic tachycardia syndrome, and headaches [1, 119].
The severity of symptoms is highly variable and associated disability (such as school absence, and physical activity interference), anxiety, and depression are common. Patients presenting to pediatric gastroenterology practice may have increased severity compared to the population as it is known that most with chronic abdominal pain never seek medical attention [10].
Evaluation of pediatric FD and other FAPDs in the clinical setting can be limited to history and physical exam, although screening tests are frequently used. Evaluations for intestinal inflammation or obstruction are sometimes performed to rule out other potential disease with similar chronic symptoms. Screening tests including newer markers of intestinal inflammation such as fecal calprotectin have an increasingly important role in the exclusion of organic disease when FAPDs are suspected [120]. The presence of “red flags” (Table 36.3) indicates to the clinician to consider further evaluation for organic disease, although their utility has been questioned [121]. Traditional red flags include night time pain and joint pain, but a recent study found equal incidence of these symptoms in children with functional disorders and organic disease [122]. The same study identified that unexplained anemia, weight loss, and hematochezia strongly suggested inflammatory bowel disease, indicating that the presence of one or more of these red flags may indicate the need for additional diagnostic testing. The differential diagnosis for functional dyspepsia includes disorders frequently treated by pediatric gastroenterologists including GERD, eosinophilic esophagitis, celiac disease, and Helicobacter pylori gastritis. These disorders may present with similar symptoms to FD and if clinically suspected then evaluation may include directed diagnostic testing including pH probe, serum screening tests, or upper endoscopy with biopsy.
The diagnosis and treatment of FD can generally be completed by a primary care provider [123]. Pediatric gastroenterologists are valuable consultants when organic intestinal disease needs to be evaluated, or when initial therapy attempts are ineffective. Cost of diagnostic evaluation is up to 5-times higher in pediatric FGID patients when performed by a gastroenterologist compared to a primary care provider [124]. Upper endoscopy with biopsy (EGD) may provide vital information in specific cases. EGD can identify mucosal inflammatory disease including eosinophilic esophagitis, H. pylori gastritis, celiac disease, and reflux esophagitis, among other disorders. Identification of mild histologic inflammation does not preclude the diagnosis of FD or other FAPDs [101, 125]. Generally EGD in pediatric patients with functional abdominal pain has low yield and does not result in significant therapeutic change [20, 126]. Thus, EGD is not routinely recommended for all children with FD but on a case-by-case basis. Other diagnostic tests (including ultrasound, computed tomography, and X-ray) are not recommended if FD is suspected and no red flags are present [127].
Management
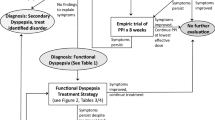
The primary step in management is positive identification of FD and education of the patient and family. Once the diagnosis is made, further testing can be eliminated or minimized and therapeutic options discussed. Reassurance that FD is a known diagnosis with specific therapies and favorable prognosis can alleviate the patient and parents’ concerns that the symptoms are caused by a rare disease and that an extensive evaluation may be needed to uncover the cause of the child’s complaints. Treatment should be framed using the biopsychosocial model, explaining the biologic and psychological components of symptom generation or maintenance. Explaining the bidirectional interaction of the brain–gut axis will help address the child’s disability and the effect of stressors that may negatively impact symptoms and treatment outcomes. In addition to treatment of GI symptoms, attention should be paid to helping the child with sleep and school problems, if those exist, and providing the patient with treatment that adapts to the patient’s needs and family beliefs. Medications are often used in conjunction with complementary therapies with a goal of symptom reduction (not necessarily elimination) and return of daily function. There is little evidence to guide the treatment of FD in children. Most pediatric clinical trials were conducted on children with FAPDs in general, which in some cases included children with FD [128, 129] with only one randomized controlled trial conducted exclusively in children with FD [130]. Thus, most available data on the treatment of dyspepsia derives from expert opinion, retrospective pediatric studies [131], and literature reviews [65, 101, 132–134].
Pharmacological Therapy
In daily clinical practice, proton pump inhibitors (PPIs ) and prokinetics are usually used as initial medications. Acid reduction therapy is commonly used as first-line medication mainly in cases of FD with epigastric pain while prokinetics are usually prescribed in cases of early satiety and postprandial fullness. A 3 weeks long randomized placebo controlled trial of the histamine-2 receptor antagonist (H2RA) famotidine conducted on a small group of children with FD has shown global improvement of symptoms but no beneficial effect on pain [130]. A pediatric trial compared four antisecretory agents (omeprazole, famotidine, ranitidine, cimetidine) for a period of 4 weeks in 169 children (age 2–16) diagnosed with functional dyspepsia [135]. The study showed that omeprazole was the most effective in achieving complete resolution of symptoms. Similar results were obtained in a study on adults with FD. One study showed that proton pump inhibitor (PPI) therapy was more effective than H2RA for FD [136]. A review of clinical trials in adult patients reported significant improvement in symptoms with PPI therapy [137]. These data suggests that empiric acid reduction for a limited period of time may be recommended in pediatric patients with FD.
Medications affecting visceral sensation are sometimes used to treat pediatric FD. Tricyclic antidepressants (TCAs) reduce nausea, abdominal pain, and delay gastric emptying. Amitriptyline is one of the more commonly used TCAs despite mixed results of efficacy in pediatric studies and the adverse effects related to anticholinergic and antihistaminic activity. An RCT of low-dose amitriptyline in children and adolescents with FAPDs identified high placebo effect (and high treatment effect) which made interpretation of medication efficacy difficult [138]. An earlier trial in pediatric IBS showed improved quality of life, but no clear improvement in abdominal pain [139]. A multicenter placebo controlled trial of amitriptyline in adults with FD showed modest, but significant improvement in symptoms [140]. The beneficial effect was similar to Saps et al. pediatric trial if calculated with intention to treat analysis (53 %), but the placebo effect was lower (40 %) than in the pediatric study. The study showed greater benefit in patients with pain predominant symptoms. Selective serotonin reuptake inhibitors (SSRI) were studied in a RCT of pediatric FAPDs, but did not show significant differences in symptom resolution [141]. The Talley et al. study of adult subjects with FD included an SSRI arm (escitalopram), and also did not show a benefit over placebo [140]. Prior studies of SSRIs in adults with FAPDs have shown efficacy equivalent to TCAs [142]. Given reports of cardiac dysrhythmia associated with TCA and SSRI use, it is reasonable to obtain baseline EKG to assess QT interval prior to initiation of therapy. While the use of TCAs and SSRIs is directed towards altered visceral sensitivity, their influence on gastrointestinal motility may affect symptom resolution. Two controlled studies on TCAs (nortriptyline, amitriptyline) found no benefit of these drugs in FD with delayed emptying [143]. Mirtazapine, an antidepressant with antagonism of H1, α2, 5HT2c, and 5HT3 receptors, was studied in a pilot RCT of adults with FD and weight loss [144]. Use of mirtazapine reduced dyspepsia symptom severity and improved quality of life, nutrient tolerance, and weight. An alternative strategy of targeting the specific nociceptor TRPV1 was attempted in adults with FD. A double blind, placebo controlled trial of red pepper powder in adults with FD showed initial, transient discomfort, but overall reduction on symptoms of pain and fullness [145].
Prokinetics as a class have shown mixed results in the treatment of FD in adults. A meta-analysis of prokinetics in 1844 adult FD patients found reduction in symptoms [146], but a separate study found no correlation between symptom improvement and gastric emptying rate [147]. Specific prokinetics may target 5HT, dopamine, or motilin receptors. Cisapride and other 5HT4 receptor agonists improve gastric emptying and accommodation, and potentially alter visceral sensitivity. However, cisapride was withdrawn from the USA and European markets due to concern for fatal cardiac arrhythmias (although not clearly a concern in healthy children [148]). In adult trials, mosapride (5HT4 agonist and 5HT3 antagonist) demonstrated FD symptom improvement [149] although a meta-analysis could not support the effects of mosapride, possibly due to heterogeneity among study definitions and outcomes [150]. Cinitapride (5HT4 receptor agonist, dopamine-2 receptor antagonist) reduced symptom severity as well as domperidone [151]. Domperidone and metoclopramide are dopamine antagonists used as prokinetic agents. Although metoclopramide is effective in FD, the potential for irreversible extrapyramidal adverse effects limits its use in pediatrics. Domperidone also improves FD symptoms in adult trials [152] and does not have similar risk of extrapyramidal adverse effects. However, due to its potential for adverse cardiac effects, in the USA it is only available for compassionate use as an investigational drug through the FDA. Erythromycin activates antral and small intestinal motilin receptors and decreases bloating with improved gastric emptying in adult FD patients, but does not alter postprandial symptoms [153]. Other motilin receptor agonists including camicinal are promising and improve gastric emptying, but efficacy to reduce FD symptoms is not yet established [154, 155]. Another prokinetic agent, acotiamide, improves gastric accommodation through acetylcholinesterase inhibition and shows promise in treatment of adult FD in ongoing trials [156–158]. Other novel prokinetics studied in gastroparesis (e.g., relamorelin, a ghrelin receptor agonist [159]) may in the future be studied in FD patients given the overlapping spectrum of symptoms and electromechanical dysfunction of the two disorders. Botulinum toxin A endoscopically injected in the pylorus safely reduced symptoms in pediatric patients with gastroparesis refractory to conventional therapy [160], and may be considered in a similar subgroup of refractory FD patients.
Gastric accommodation is an additional target of electromechanical dysfunction in FD. Cyproheptadine antagonizes 5HT2a and 5HT2b, histamine-1, and muscarinic receptors whose putative mechanism is improved gastric accommodation through fundic relaxation, although it may also have an effect by decreasing gastric hypersensitivity. It was found to be effective in a RCT of children with FAPDs [161], and provided symptom improvement in pediatric FD patients in an open-label trial, with few and mild adverse effects [131]. Buspirone, a 5HT1a receptor agonist, improved accommodation in adults with FD and decreased symptom severity [162]. Other medications targeting the 5HT1a receptor to improve accommodation include tandospirone and sumatriptan, but these have not as clearly reduced symptoms [163, 164]. Tegaserod, a 5-HT4 agonist that was taken off of the market, was found to enhance gastric accommodation in adult patients with normal gastric emptying [165]. Ondansetron, a 5HT3 antagonist, improved accommodation and reduced nausea in adults with FD, but mechanical and symptom effects were not seemingly associated [166].
Neuromodulation with gastric electrical stimulation significantly improved nausea and vomiting, and improved tolerance of nutritional intake in children with dyspepsia [167]. Gastric electric stimulation was also shown to improve quality of life and global health [168] in children with excellent long-term tolerance and few adverse effects [169]. Although the technology is currently in use only in few pediatric centers, it is a promising therapy for patients with refractory symptoms.
Pediatric FD patients with duodenal eosinophilia were demonstrated to have reduction of symptoms with histamine1/2 antagonism or cromolyn, a mast cell stabilizer [170]. Of 21 patients who did not initially respond to ranitidine and hydroxyzine combination therapy, two were lost to follow-up and 17 clinically responded (complete or nearly complete resolution of pain) with the addition of cromolyn. Treatment of a similar cohort in a placebo-controlled crossover trial showed that children receiving montelukast, a leukotriene receptor antagonist, had a greater reduction in global pain than those on placebo without having any adverse effects [171]. However, symptom improvement does not seem to correlate to mucosal eosinophil density or activation, and the mechanisms of action are not yet determined [108].
Nonpharmacologic Therapy
There are several herbal preparations that have been purported to improve chronic abdominal pain. A multicenter placebo controlled trial of STW 5 (iberogast), an herbal compound with a mechanism of action that is not yet clearly understood, showed improvement of adult FD symptoms [172, 173]. An open label trial of iberogast also showed beneficial effects in pediatric patients with FAPDs [174]. Peppermint oil seems to affect various mechanisms involved with the pathophysiology of IBS. Two studies on adult volunteers have investigated the gastric sensorimotor aspects of peppermint oil. Papathanasopoulos et al. found that peppermint oil reduces intragastric pressure and proximal phasic contractility without affecting gastric tone, accommodation, visceral sensitivity, epigastric pain, or early satiety [175] while Inamori et al. found that peppermint oil enhances gastric emptying [176]. Studies in adults and children with IBS have shown a beneficial effect of peppermint oil on IBS symptoms [177]. Nigella sativa (black cumin) seed oil mixed with honey, added to standard treatment with famotidine, showed reduction of FD symptoms in an 8 week randomized controlled trial in adults [178]. Similarly, Pimpinella anisum (anise) supplementation reduced pain in adults with FD compared to controls [179]. Rikkunshito, a Japanese herbal preparation, was studied in a multicenter randomized clinical trial of FD adults and was shown to improve pain, accelerate gastric emptying, and improve accommodation possibly through serotonergic or ghrelin mediated pathways [180]. Although ginger is used to treat nausea and enhances gastric emptying, it did not improve symptoms in an adult FD trial [181]. Various phytotherapy compounds and plant extracts such as curcumin [182] have been used with different degrees of efficacy and evidence.
Complementary Therapy
Therapies aimed at modifying psychosocial stress, catastrophizing behavior, and anxiety indirectly target visceral hypersensitivity and electromechanical dysfunction given the cross-talk between the gut, brain, and the environment. Importantly these therapies may be more effective in conjunction with medical therapy rather than in isolation [183]. Although sometimes difficult for patients and medical providers to access due to local absence of therapists or financial constraints, complementary therapies should be considered whenever possible. Over the last few years there have been several studies of gut directed hypnotherapy [184, 185] and cognitive behavioral therapy [186, 187] showing beneficial effect in children with FAPDs. Other complementary therapies with potential beneficial effects in the treatment of children with FAPDs are biofeedback assisted relaxation therapy [183], yoga [188], and acupuncture [189].
Prognosis and Future Directions
The results of studies assessing prognosis show mixed results. A study on children with FD demonstrated significant improvement in 70 % of patients at 2 years [190], while another study showed that most patients with FAPDs improve within 12 months of presentation [191]. However, another study followed a group of children (8–16 years) evaluated for dyspepsia and 5–15 years later found more chronic dyspeptic symptoms, higher frequency of anxiety disorder, and reduced quality of life compared to controls [192]. Novel pharmacologic and nonpharmacologic therapies continue to be investigated for the treatment of dyspepsia. Ongoing efforts are needed to identify most effective treatment options and determine how best to personalize these options to individual patients. Evidence-based care strategies tailored to the needs of the individual may help optimize results by minimizing symptom severity and duration and reducing adverse medication effects. The consistent use of clinically meaningful patient-reported outcome measures across studies of FAPDs will enhance research by allowing comparisons of therapeutic trials using different interventions [193, 194].
References
Kovacic K, Williams S, Li BU, Chelimsky G, Miranda A. High prevalence of nausea in children with pain-associated functional gastrointestinal disorders: are Rome criteria applicable? J Pediatr Gastroenterol Nutr. 2013;57(3):311–5.
Saps M, Nichols-Vinueza DX, Rosen JM, Velasco-Benitez CA. Prevalence of functional gastrointestinal disorders in Colombian school children. J Pediatr. 2014;164(3):542–5 e1.
Zablah R, Velasco-Benitez CA, Merlos I, Bonilla S, Saps M. Prevalence of functional gastrointestinal disorders in school-aged children in El Salvador. Rev Gastroenterol Mex. 2015;80(3):186–91.
Devanarayana NM, Mettananda S, Liyanarachchi C, Nanayakkara N, Mendis N, Perera N, et al. Abdominal pain-predominant functional gastrointestinal diseases in children and adolescents: prevalence, symptomatology, and association with emotional stress. J Pediatr Gastroenterol Nutr. 2011;53(6):659–65.
Sagawa T, Okamura S, Kakizaki S, Zhang Y, Morita K, Mori M. Functional gastrointestinal disorders in adolescents and quality of school life. J Gastroenterol Hepatol. 2013;28(2):285–90.
Son YJ, Jun EY, Park JH. Prevalence and risk factors of irritable bowel syndrome in Korean adolescent girls: a school-based study. Int J Nurs Stud. 2009;46(1):76–84.
Dong L, Dingguo L, Xiaoxing X, Hanming L. An epidemiologic study of irritable bowel syndrome in adolescents and children in China: a school-based study. Pediatrics. 2005;116(3):e393–6.
Udoh E, Devanarayana NM, Rajindrajith S, Meremikwu M, Benninga MA. Abdominal pain predominant functional gastrointestinal disorders in adolescent Nigerians. J Pediatr Gastroenterol Nutr. 2015;62:588–93.
Jativa E, Velasco-Benitez CA, Koppen IJ, Cabezas ZJ, Saps M. Prevalence of functional gastrointestinal disorders in school children in Ecuador. J Pediatr Gastroenterol Nutr. 2016;63(1):25–8.
Saps M, Adams P, Bonilla S, Chogle A, Nichols-Vinueza D. Parental report of abdominal pain and abdominal pain-related functional gastrointestinal disorders from a community survey. J Pediatr Gastroenterol Nutr. 2012;55(6):707–10.
Van Tilburg MA, Walker L, Palsson OS, Kim SM, Spiegel BM, Spiller R, et al. Prevalence of child/adolescent functional gastrointestinal disorders in a National U.S. Community sample. Gastroenterology. 2014;146(5):S143–4. Abstract.
Ford AC. Overlap among the functional gastrointestinal disorders. Am J Gastroenterol. 2010;105(11):2512.
Schurman JV, Friesen CA, Danda CE, Andre L, Welchert E, Lavenbarg T, et al. Diagnosing functional abdominal pain with the Rome II criteria: parent, child, and clinician agreement. J Pediatr Gastroenterol Nutr. 2005;41(3):291–5.
Saps M, Seshadri R, Sztainberg M, Schaffer G, Marshall BM, Di Lorenzo C. A prospective school-based study of abdominal pain and other common somatic complaints in children. J Pediatr. 2009;154(3):322–6.
Rouster AS, Karpinski AC, Silver D, Monagas J, Hyman PE. Functional gastrointestinal disorders dominate pediatric gastroenterology outpatient practice. J Pediatr Gastroenterol Nutr. 2015;62:847–51.
Varni JW, Bendo CB, Nurko S, Shulman RJ, Self MM, Franciosi JP, et al. Health-related quality of life in pediatric patients with functional and organic gastrointestinal diseases. J Pediatr. 2015;166(1):85–90.
Varni JW, Bendo CB, Denham J, Shulman RJ, Self MM, Neigut DA, et al. PedsQL Gastrointestinal Symptoms Scales and Gastrointestinal Worry Scales in pediatric patients with functional and organic gastrointestinal diseases in comparison to healthy controls. Qual Life Res. 2015;24(2):363–78.
Huntley ED, Campo JV, Dahl RE, Lewin DS. Sleep characteristics of youth with functional abdominal pain and a healthy comparison group. J Pediatr Psychol. 2007;32(8):938–49.
Hoekman DR, Rutten JM, Vlieger AM, Benninga MA, Dijkgraaf MG. Annual costs of care for pediatric irritable bowel syndrome, functional abdominal pain, and functional abdominal pain syndrome. J Pediatr. 2015;167(5):1103–8 e2.
Dhroove G, Chogle A, Saps M. A million-dollar work-up for abdominal pain: is it worth it? J Pediatr Gastroenterol Nutr. 2010;51(5):579–83.
Park R, Mikami S, LeClair J, Bollom A, Lembo C, Sethi S, et al. Inpatient burden of childhood functional GI disorders in the USA: an analysis of national trends in the USA from 1997 to 2009. Neurogastroenterol Motil. 2015;27(5):684–92.
Pensabene L, Talarico V, Concolino D, Ciliberto D, Campanozzi A, Gentile T, et al. Postinfectious functional gastrointestinal disorders in children: a multicenter prospective study. J Pediatr. 2015;166(4):903–7 e1.
Singh R, Mittal B, Ghoshal UC. Functional dyspepsia is associated with GNbeta3 C825T and CCK-AR T/C polymorphism. Eur J Gastroenterol Hepatol. 2016;28(2):226–32.
Dai F, Liu Y, Shi H, Ge S, Song J, Dong L, et al. Association of genetic variants in GNbeta3 with functional dyspepsia: a meta-analysis. Dig Dis Sci. 2014;59(8):1823–30.
Kourikou A, Karamanolis GP, Dimitriadis GD, Triantafyllou K. Gene polymorphisms associated with functional dyspepsia. World J Gastroenterol. 2015;21(25):7672–82.
Rosen JM, Adams PN, Saps M. Umbilical hernia repair increases the rate of functional gastrointestinal disorders in children. J Pediatr. 2013;163(4):1065–8.
Saps M, Bonilla S. Early life events: infants with pyloric stenosis have a higher risk of developing chronic abdominal pain in childhood. J Pediatr. 2011;159(4):551–4 e1.
Saps M, Dhroove G, Chogle A. Henoch-Schonlein purpura leads to functional gastrointestinal disorders. Dig Dis Sci. 2011;56(6):1789–93.
Saps M, Lu P, Bonilla S. Cow’s-milk allergy is a risk factor for the development of FGIDs in children. J Pediatr Gastroenterol Nutr. 2011;52(2):166–9.
Rosen JM, Kriegermeier A, Adams PN, Klumpp DJ, Saps M. Urinary tract infection in infancy is a risk factor for chronic abdominal pain in childhood. J Pediatr Gastroenterol Nutr. 2015;60(2):214–6.
Bradford K, Shih W, Videlock EJ, Presson AP, Naliboff BD, Mayer EA, et al. Association between early adverse life events and irritable bowel syndrome. Clin Gastroenterol Hepatol. 2012;10(4):385–90 e1–3.
Klooker TK, Braak B, Painter RC, de Rooij SR, van Elburg RM, van den Wijngaard RM, et al. Exposure to severe wartime conditions in early life is associated with an increased risk of irritable bowel syndrome: a population-based cohort study. Am J Gastroenterol. 2009;104(9):2250–6.
McKeown ES, Parry SD, Stansfield R, Barton JR, Welfare MR. Postinfectious irritable bowel syndrome may occur after non-gastrointestinal and intestinal infection. Neurogastroenterol Motil. 2006;18(9):839–43.
Cremon C, Stanghellini V, Pallotti F, Fogacci E, Bellacosa L, Morselli-Labate AM, et al. Salmonella gastroenteritis during childhood is a risk factor for irritable bowel syndrome in adulthood. Gastroenterology. 2014;147(1):69–77.
Hanevik K, Dizdar V, Langeland N, Hausken T. Development of functional gastrointestinal disorders after Giardia lamblia infection. BMC Gastroenterol. 2009;9:27.
Paula H, Grover M, Halder SL, Locke 3rd GR, Schleck CD, Zinsmeister AR, et al. Non-enteric infections, antibiotic use, and risk of development of functional gastrointestinal disorders. Neurogastroenterol Motil. 2015;27(11):1580–6.
Spee LA, Madderom MB, Pijpers M, van Leeuwen Y, Berger MY. Association between helicobacter pylori and gastrointestinal symptoms in children. Pediatrics. 2010;125(3):e651–69.
de Jong MJ, Korterink JJ, Benninga MA, Hilbink M, Widdershoven J, Deckers-Kocken JM. Dientamoeba fragilis and chronic abdominal pain in children: a case-control study. Arch Dis Child. 2014;99(12):1109–13.
Simren M, Barbara G, Flint HJ, Spiegel BM, Spiller RC, Vanner S, et al. Intestinal microbiota in functional bowel disorders: a Rome foundation report. Gut. 2013;62(1):159–76.
Kindt S, Van Oudenhove L, Mispelon L, Caenepeel P, Arts J, Tack J. Longitudinal and cross-sectional factors associated with long-term clinical course in functional dyspepsia: a 5-year follow-up study. Am J Gastroenterol. 2011;106(2):340–8.
Vu J, Kushnir V, Cassell B, Gyawali CP, Sayuk GS. The impact of psychiatric and extraintestinal comorbidity on quality of life and bowel symptom burden in functional GI disorders. Neurogastroenterol Motil. 2014;26(9):1323–32.
Pinto-Sanchez MI, Ford AC, Avila CA, Verdu EF, Collins SM, Morgan D, et al. Anxiety and depression increase in a stepwise manner in parallel with multiple FGIDs and symptom severity and frequency. Am J Gastroenterol. 2015;110(7):1038–48.
Schurman JV, Danda CE, Friesen CA, Hyman PE, Simon SD, Cocjin JT. Variations in psychological profile among children with recurrent abdominal pain. J Clin Psychol Med Settings. 2008;15(3):241–51.
Lee HS, Kim DK, Kim YB, Lee KJ. Effect of acute stress on immune cell counts and the expression of tight junction proteins in the duodenal mucosa of rats. Gut Liver. 2013;7(2):190–6.
Demaude J, Salvador-Cartier C, Fioramonti J, Ferrier L, Bueno L. Phenotypic changes in colonocytes following acute stress or activation of mast cells in mice: implications for delayed epithelial barrier dysfunction. Gut. 2006;55(5):655–61.
Wouters MM, Boeckxstaens GE. Is there a causal link between psychological disorders and functional gastrointestinal disorders? Expert Rev Gastroenterol Hepatol. 2016;10(1):5–8.
Monnikes H, Tebbe JJ, Hildebrandt M, Arck P, Osmanoglou E, Rose M, et al. Role of stress in functional gastrointestinal disorders. Evidence for stress-induced alterations in gastrointestinal motility and sensitivity. Dig Dis. 2001;19(3):201–11.
Cong X, Henderson WA, Graf J, McGrath JM. Early life experience and gut microbiome: the brain-gut-microbiota signaling system. Adv Neonatal Care. 2015;15(5):314–23; quiz E1–2.
Shulman RJ, Eakin MN, Czyzewski DI, Jarrett M, Ou CN. Increased gastrointestinal permeability and gut inflammation in children with functional abdominal pain and irritable bowel syndrome. J Pediatr. 2008;153(5):646–50.
Collins SM, Surette M, Bercik P. The interplay between the intestinal microbiota and the brain. Nat Rev Microbiol. 2012;10(11):735–42.
Keightley PC, Koloski NA, Talley NJ. Pathways in gut-brain communication: evidence for distinct gut-to-brain and brain-to-gut syndromes. Aust N Z J Psychiatry. 2015;49(3):207–14.
Kumagai H, Yokoyama K, Imagawa T, Yamagata T. Functional dyspepsia & irritable bowel syndrome in teenagers: an internet survey. Pediatr Int. 2015.
Chande N, Wang Y, MacDonald JK, McDonald JW. Methotrexate for induction of remission in ulcerative colitis. Cochrane Database Syst Rev. 2014;8:CD006618.
Diederen K, Hoekman DR, Koot B, Tabbers MM, Kindermann A, Benninga M. The prevalence of irritable-bowel like symptoms in pediatric inflammatory bowel disease. Gastroenterology. 2015;148(4):S-378. Abstract.
Zimmerman LA, Srinath AI, Goyal A, Bousvaros A, Ducharme P, Szigethy E, et al. The overlap of functional abdominal pain in pediatric Crohn’s disease. Inflamm Bowel Dis. 2013;19(4):826–31.
Cristofori F, Fontana C, Magista A, Capriati T, Indrio F, Castellaneta S, et al. Increased prevalence of celiac disease among pediatric patients with irritable bowel syndrome: a 6-year prospective cohort study. JAMA Pediatr. 2014;168(6):555–60.
Kansu A, Kuloglu Z, Demir A, Yaman A, Turkish Celiac Study Group. Yield of coeliac screening in abdominal pain-associated functional gastrointestinal system disorders. J Paediatr Child Health. 2015;51(11):1066–70.
Saps M, Adams P, Bonilla S, Nichols-Vinueza D. Abdominal pain and functional gastrointestinal disorders in children with celiac disease. J Pediatr. 2013;162(3):505–9.
Turco R, Boccia G, Miele E, Giannetti E, Buonavolonta R, Quitadamo P, et al. The association of coeliac disease in childhood with functional gastrointestinal disorders: a prospective study in patients fulfilling Rome III criteria. Aliment Pharmacol Ther. 2011;34(7):783–9.
Teitelbaum JE, Sinha P, Micale M, Yeung S, Jaeger J. Obesity is related to multiple functional abdominal diseases. J Pediatr. 2009;154(3):444–6.
Bonilla S, Wang D, Saps M. Obesity predicts persistence of pain in children with functional gastrointestinal disorders. Int J Obes (Lond). 2011;35(4):517–21.
Phatak UP, Pashankar DS. Prevalence of functional gastrointestinal disorders in obese and overweight children. Int J Obes (Lond). 2014;38(10):1324–7.
Jung JG, Yang JN, Lee CG, Choi SH, Kwack WG, Lee JH, et al. Visceral adiposity is associated with an increased risk of functional dyspepsia. J Gastroenterol Hepatol. 2016;31(3):567–74.
Le Pluart D, Sabate JM, Bouchoucha M, Hercberg S, Benamouzig R, Julia C. Functional gastrointestinal disorders in 35,447 adults and their association with body mass index. Aliment Pharmacol Ther. 2015;41(8):758–67.
Rosen JM, Cocjin JT, Schurman JV, Colombo JM, Friesen CA. Visceral hypersensitivity and electromechanical dysfunction as therapeutic targets in pediatric functional dyspepsia. World J Gastrointest Pharmacol Ther. 2014;5(3):122–38.
Bouin M, Lupien F, Riberdy M, Boivin M, Plourde V, Poitras P. Intolerance to visceral distension in functional dyspepsia or irritable bowel syndrome: an organ specific defect or a pan intestinal dysregulation? Neurogastroenterol Motil. 2004;16(3):311–4.
Di Lorenzo C, Youssef NN, Sigurdsson L, Scharff L, Griffiths J, Wald A. Visceral hyperalgesia in children with functional abdominal pain. J Pediatr. 2001;139(6):838–43.
Hoffman I, Vos R, Tack J. Assessment of gastric sensorimotor function in paediatric patients with unexplained dyspeptic symptoms and poor weight gain. Neurogastroenterol Motil. 2007;19(3):173–9.
Montano-Loza A, Schmulson M, Zepeda-Gomez S, Remes-Troche JM, Valdovinos-Diaz MA. Maximum tolerated volume in drinking tests with water and a nutritional beverage for the diagnosis of functional dyspepsia. World J Gastroenterol. 2005;11(20):3122–6.
Boeckxstaens GE, Hirsch DP, van den Elzen BD, Heisterkamp SH, Tytgat GN. Impaired drinking capacity in patients with functional dyspepsia: relationship with proximal stomach function. Gastroenterology. 2001;121(5):1054–63.
Sood MR, Schwankovsky LM, Rowhani A, Zangen T, Ziring D, Furtado T, et al. Water load test in children. J Pediatr Gastroenterol Nutr. 2002;35(2):199–201.
Walker LS, Williams SE, Smith CA, Garber J, Van Slyke DA, Lipani T, et al. Validation of a symptom provocation test for laboratory studies of abdominal pain and discomfort in children and adolescents. J Pediatr Psychol. 2006;31(7):703–13.
Schurman JV, Friesen CA, Andre L, Welchert E, Lavenbarg T, Danda CE, et al. Diagnostic utility of the water load test in children with chronic abdominal pain. J Pediatr Gastroenterol Nutr. 2007;44(1):51–7.
Miwa H, Nakajima K, Yamaguchi K, Fujimoto K, Veldhuyzen VANZSJ, Kinoshita Y, et al. Generation of dyspeptic symptoms by direct acid infusion into the stomach of healthy Japanese subjects. Aliment Pharmacol Ther. 2007;26(2):257–64.
Samsom M, Verhagen MA, vanBerge Henegouwen GP, Smout AJ. Abnormal clearance of exogenous acid and increased acid sensitivity of the proximal duodenum in dyspeptic patients. Gastroenterology. 1999;116(3):515–20.
Fried M, Feinle C. The role of fat and cholecystokinin in functional dyspepsia. Gut. 2002;51 Suppl 1:i54–7.
Bisschops R, Tack J. Dysaccommodation of the stomach: therapeutic nirvana? Neurogastroenterol Motil. 2007;19(2):85–93.
Tack J, Caenepeel P, Piessevaux H, Cuomo R, Janssens J. Assessment of meal induced gastric accommodation by a satiety drinking test in health and in severe functional dyspepsia. Gut. 2003;52(9):1271–7.
Hoffman I, Tack J. Assessment of gastric motor function in childhood functional dyspepsia and obesity. Neurogastroenterol Motil. 2012;24(2):108–12 e81.
Hoffman I, Vos R, Tack J. Normal values for the satiety drinking test in healthy children between 5 and 15 years. Neurogastroenterol Motil. 2009;21(5):517–20 e6.
Gonenne J, Castillo EJ, Camilleri M, Burton D, Thomforde GM, Baxter KL, et al. Does the nutrient drink test accurately predict postprandial gastric volume in health and community dyspepsia? Neurogastroenterol Motil. 2005;17(1):44–50.
Olafsdottir E, Gilja OH, Tefera S, Fluge G, Berstad A. Intragastric maldistribution of a liquid meal in children with recurrent abdominal pain assessed by three-dimensional ultrasonography. Scand J Gastroenterol. 2003;38(8):819–25.
van den Elzen BD, Bennink RJ, Wieringa RE, Tytgat GN, Boeckxstaens GE. Fundic accommodation assessed by SPECT scanning: comparison with the gastric barostat. Gut. 2003;52(11):1548–54.
Tack J, Piessevaux H, Coulie B, Caenepeel P, Janssens J. Role of impaired gastric accommodation to a meal in functional dyspepsia. Gastroenterology. 1998;115(6):1346–52.
Bredenoord AJ, Chial HJ, Camilleri M, Mullan BP, Murray JA. Gastric accommodation and emptying in evaluation of patients with upper gastrointestinal symptoms. Clin Gastroenterol Hepatol. 2003;1(4):264–72.
Chitkara DK, Camilleri M, Zinsmeister AR, Burton D, El-Youssef M, Freese D, et al. Gastric sensory and motor dysfunction in adolescents with functional dyspepsia. J Pediatr. 2005;146(4):500–5.
Hijaz NM, Friesen CA, Schurman JV, Pearce RE, Abdel-Rahman SM. Plasma ghrelin and liquid gastric emptying in children with functional dyspepsia consistent with post-prandial distress syndrome. Neurogastroenterol Motil. 2015;27(8):1120–6.
Kazemi M, Eshraghian A, Hamidpour L, Taghavi S. Changes in serum ghrelin level in relation to meal-time in patients with functional dyspepsia. United European Gastroenterol J. 2015;3(1):11–6.
Friesen CA, Lin Z, Singh M, Singh V, Schurman JV, Burchell N, et al. Antral inflammatory cells, gastric emptying, and electrogastrography in pediatric functional dyspepsia. Dig Dis Sci. 2008;53(10):2634–40.
Liebregts T, Adam B, Bredack C, Gururatsakul M, Pilkington KR, Brierley SM, et al. Small bowel homing T cells are associated with symptoms and delayed gastric emptying in functional dyspepsia. Am J Gastroenterol. 2011;106(6):1089–98.
Boccia G, Buonavolonta R, Coccorullo P, Manguso F, Fuiano L, Staiano A. Dyspeptic symptoms in children: the result of a constipation-induced cologastric brake? Clin Gastroenterol Hepatol. 2008;6(5):556–60.
Chitkara DK, Delgado-Aros S, Bredenoord AJ, Cremonini F, El-Youssef M, Freese D, et al. Functional dyspepsia, upper gastrointestinal symptoms, and transit in children. J Pediatr. 2003;143(5):609–13.
Devanarayana NM, Rajindrajith S, Perera MS, Nishanthanie SW, Benninga MA. Gastric emptying and antral motility parameters in children with functional dyspepsia: association with symptom severity. J Gastroenterol Hepatol. 2013;28(7):1161–6.
Friesen CA, Lin Z, Hyman PE, Andre L, Welchert E, Schurman JV, et al. Electrogastrography in pediatric functional dyspepsia: relationship to gastric emptying and symptom severity. J Pediatr Gastroenterol Nutr. 2006;42(3):265–9.
Riezzo G, Chiloiro M, Guerra V, Borrelli O, Salvia G, Cucchiara S. Comparison of gastric electrical activity and gastric emptying in healthy and dyspeptic children. Dig Dis Sci. 2000;45(3):517–24.
Chen JD, Lin X, Zhang M, Torres-Pinedo RB, Orr WC. Gastric myoelectrical activity in healthy children and children with functional dyspepsia. Dig Dis Sci. 1998;43(11):2384–91.
Sha W, Pasricha PJ, Chen JD. Rhythmic and spatial abnormalities of gastric slow waves in patients with functional dyspepsia. J Clin Gastroenterol. 2009;43(2):123–9.
Koch KL, Hong SP, Xu L. Reproducibility of gastric myoelectrical activity and the water load test in patients with dysmotility-like dyspepsia symptoms and in control subjects. J Clin Gastroenterol. 2000;31(2):125–9.
Friesen CA, Lin Z, Garola R, Andre L, Burchell N, Moore A, et al. Chronic gastritis is not associated with gastric dysrhythmia or delayed solid emptying in children with dyspepsia. Dig Dis Sci. 2005;50(6):1012–8.
Yin J, Levanon D, Chen JD. Inhibitory effects of stress on postprandial gastric myoelectrical activity and vagal tone in healthy subjects. Neurogastroenterol Motil. 2004;16(6):737–44.
Friesen CA, Schurman JV, Colombo JM, Abdel-Rahman SM. Eosinophils and mast cells as therapeutic targets in pediatric functional dyspepsia. World J Gastrointest Pharmacol Ther. 2013;4(4):86–96.
Keely S, Walker MM, Marks E, Talley NJ. Immune dysregulation in the functional gastrointestinal disorders. Eur J Clin Invest. 2015;45(12):1350–9.
Wouters MM, Vicario M, Santos J. The role of mast cells in functional GI disorders. Gut. 2016;65(1):155–68.
Vanheel H, Vicario M, Vanuytsel T, Van Oudenhove L, Martinez C, Keita AV, et al. Impaired duodenal mucosal integrity and low-grade inflammation in functional dyspepsia. Gut. 2014;63(2):262–71.
O’Sullivan M, Clayton N, Breslin NP, Harman I, Bountra C, McLaren A, et al. Increased mast cells in the irritable bowel syndrome. Neurogastroenterol Motil. 2000;12(5):449–57.
Walker MM, Salehian SS, Murray CE, Rajendran A, Hoare JM, Negus R, et al. Implications of eosinophilia in the normal duodenal biopsy—an association with allergy and functional dyspepsia. Aliment Pharmacol Ther. 2010;31(11):1229–36.
Friesen CA, Andre L, Garola R, Hodge C, Roberts C. Activated duodenal mucosal eosinophils in children with dyspepsia: a pilot transmission electron microscopic study. J Pediatr Gastroenterol Nutr. 2002;35(3):329–33.
Friesen CA, Neilan NA, Schurman JV, Taylor DL, Kearns GL, Abdel-Rahman SM. Montelukast in the treatment of duodenal eosinophilia in children with dyspepsia: effect on eosinophil density and activation in relation to pharmacokinetics. BMC Gastroenterol. 2009;9:32.
Kokkonen J, Ruuska T, Karttunen TJ, Niinimaki A. Mucosal pathology of the foregut associated with food allergy and recurrent abdominal pains in children. Acta Paediatr. 2001;90(1):16–21.
Cirillo C, Bessissow T, Desmet AS, Vanheel H, Tack J, Vanden BP. Evidence for neuronal and structural changes in submucous ganglia of patients with functional dyspepsia. Am J Gastroenterol. 2015;110(8):1205–15.
Muller PA, Koscso B, Rajani GM, Stevanovic K, Berres ML, Hashimoto D, et al. Crosstalk between muscularis macrophages and enteric neurons regulates gastrointestinal motility. Cell. 2014;158(2):300–13.
Mayer EA, Gupta A, Kilpatrick LA, Hong JY. Imaging brain mechanisms in chronic visceral pain. Pain. 2015;156 Suppl 1:S50–63.
Nan J, Liu J, Mu J, Zhang Y, Zhang M, Tian J, et al. Anatomically related gray and white matter alterations in the brains of functional dyspepsia patients. Neurogastroenterol Motil. 2015;27(6):856–64.
Tominaga K, Tsumoto C, Ataka S, Mizuno K, Takahashi K, Yamagami H, et al. Regional brain disorders of serotonin neurotransmission are associated with functional dyspepsia. Life Sci. 2015;137:150–7.
Ly HG, Ceccarini J, Weltens N, Bormans G, Van Laere K, Tack J, et al. Increased cerebral cannabinoid-1 receptor availability is a stable feature of functional dyspepsia: a [F]MK-9470 PET study. Psychother Psychosom. 2015;84(3):149–58.
Hyams JS, Di Lorenzo C, Saps M, Shulman RJ, Staiano A, van Tilburg M. Childhood functional gastrointestinal disorders: child/adolescent. Gastroenterology. 2016;150(6):1456–68.e2.
Turco R, Russo M, Martinelli M, Castiello R, Coppola V, Miele E, et al. Do distinct functional dyspepsia subtypes exist in children? J Pediatr Gastroenterol Nutr. 2016;62(3):387–92.
Korterink JJ, Benninga MA, van Wering HM, Deckers-Kocken JM. Glucose hydrogen breath test for small intestinal bacterial overgrowth in children with abdominal pain-related functional gastrointestinal disorders. J Pediatr Gastroenterol Nutr. 2015;60(4):498–502.
Kovacic K, Chelimsky TC, Sood MR, Simpson P, Nugent M, Chelimsky G. Joint hypermobility: a common association with complex functional gastrointestinal disorders. J Pediatr. 2014;165(5):973–8.
Ezri J, Nydegger A. Pediatrics. Fecal calprotectin in children: use and interpretation. Rev Med Suisse. 2011;7(277):69–70.
Gijsbers CF, Benninga MA, Schweizer JJ, Kneepkens CM, Vergouwe Y, Buller HA. Validation of the Rome III criteria and alarm symptoms for recurrent abdominal pain in children. J Pediatr Gastroenterol Nutr. 2014;58(6):779–85.
El-Chammas K, Majeskie A, Simpson P, Sood M, Miranda A. Red flags in children with chronic abdominal pain and Crohn’s disease-a single center experience. J Pediatr. 2013;162(4):783–7.
American Academy of Pediatrics Subcommittee on Chronic Abdominal Pain, North American Society for Pediatric Gastroenterology Hepatology, and Nutrition. Chronic abdominal pain in children. Pediatrics. 2005;115(3):e370–81.
Lane MM, Weidler EM, Czyzewski DI, Shulman RJ. Pain symptoms and stooling patterns do not drive diagnostic costs for children with functional abdominal pain and irritable bowel syndrome in primary or tertiary care. Pediatrics. 2009;123(3):758–64.
Di Nardo G, Barbara G, Cucchiara S, Cremon C, Shulman RJ, Isoldi S, et al. Neuroimmune interactions at different intestinal sites are related to abdominal pain symptoms in children with IBS. Neurogastroenterol Motil. 2014;26(2):196–204.
Thakkar K, Dorsey F, Gilger MA. Impact of endoscopy on management of chronic abdominal pain in children. Dig Dis Sci. 2011;56(2):488–93.
Di Lorenzo C, Colletti RB, Lehmann HP, Boyle JT, Gerson WT, Hyams JS, et al. Chronic abdominal pain in children: a clinical report of the American Academy of Pediatrics and the North American Society for Pediatric Gastroenterology, Hepatology and Nutrition. J Pediatr Gastroenterol Nutr. 2005;40(3):245–8.
Saps M, Biring HS, Pusatcioglu CK, Mintjens S, Rzeznikiewiz D. A comprehensive review of randomized placebo-controlled pharmacological clinical trials in children with functional abdominal pain disorders. J Pediatr Gastroenterol Nutr. 2015;60(5):645–53.
Francavilla R, Miniello V, Magista AM, De Canio A, Bucci N, Gagliardi F, et al. A randomized controlled trial of Lactobacillus GG in children with functional abdominal pain. Pediatrics. 2010;126(6):e1445–52.
See MC, Birnbaum AH, Schechter CB, Goldenberg MM, Benkov KJ. Double-blind, placebo-controlled trial of famotidine in children with abdominal pain and dyspepsia: global and quantitative assessment. Dig Dis Sci. 2001;46(5):985–92.
Rodriguez L, Diaz J, Nurko S. Safety and efficacy of cyproheptadine for treating dyspeptic symptoms in children. J Pediatr. 2013;163(1):261–7.
Rutten JM, Korterink JJ, Venmans LM, Benninga MA, Tabbers MM. Nonpharmacologic treatment of functional abdominal pain disorders: a systematic review. Pediatrics. 2015;135(3):522–35.
Hussain SZ, Hyman PE. Psychotropic medications for pediatric functional gastrointestinal disorders. J Pediatr Gastroenterol Nutr. 2014;59(3):280–7.
Korterink JJ, Rutten JM, Venmans L, Benninga MA, Tabbers MM. Pharmacologic treatment in pediatric functional abdominal pain disorders: a systematic review. J Pediatr. 2015;166(2):424–31 e6.
Dehghani SM, Imanieh MH, Oboodi R, Haghighat M. The comparative study of the effectiveness of cimetidine, ranitidine, famotidine, and omeprazole in treatment of children with dyspepsia. ISRN Pediatr. 2011;2011:219287.
Armstrong D, van Zanten SJ V, Barkun AN, Chiba N, Thomson AB, Smyth S, et al. Heartburn-dominant, uninvestigated dyspepsia: a comparison of ‘PPI-start’ and ‘H2-RA-start’ management strategies in primary care—the CADET-HR Study. Aliment Pharmacol Ther. 2005;21(10):1189–202.
Talley NJ, Lauritsen K. The potential role of acid suppression in functional dyspepsia: the BOND, OPERA, PILOT, and ENCORE studies. Gut. 2002;50 Suppl 4:iv36–41.
Saps M, Youssef N, Miranda A, Nurko S, Hyman P, Cocjin J, et al. Multicenter, randomized, placebo-controlled trial of amitriptyline in children with functional gastrointestinal disorders. Gastroenterology. 2009;137(4):1261–9.
Bahar RJ, Collins BS, Steinmetz B, Ament ME. Double-blind placebo-controlled trial of amitriptyline for the treatment of irritable bowel syndrome in adolescents. J Pediatr. 2008;152(5):685–9.
Talley NJ, Locke GR, Saito YA, Almazar AE, Bouras EP, Howden CW, et al. Effect of amitriptyline and escitalopram on functional dyspepsia: a multicenter randomized controlled study. Gastroenterology. 2015;149(2):340–9 e2.
Roohafza H, Pourmoghaddas Z, Saneian H, Gholamrezaei A. Citalopram for pediatric functional abdominal pain: a randomized, placebo-controlled trial. Neurogastroenterol Motil. 2014;26(11):1642–50.
Tack J, Broekaert D, Fischler B, Van Oudenhove L, Gevers AM, Janssens J. A controlled crossover study of the selective serotonin reuptake inhibitor citalopram in irritable bowel syndrome. Gut. 2006;55(8):1095–103.
Tack J, Carbone F, Rotondo A. Gastroparesis. Curr Opin Gastroenterol. 2015;31(6):499–505.
Tack J, Ly HG, Carbone F, Vanheel H, Vanuytsel T, Holvoet L, et al. Efficacy of mirtazapine in patients with functional dyspepsia and weight loss. Clin Gastroenterol Hepatol. 2016;14(3):385–92.
Bortolotti M, Coccia G, Grossi G, Miglioli M. The treatment of functional dyspepsia with red pepper. Aliment Pharmacol Ther. 2002;16(6):1075–82.
Hiyama T, Yoshihara M, Matsuo K, Kusunoki H, Kamada T, Ito M, et al. Meta-analysis of the effects of prokinetic agents in patients with functional dyspepsia. J Gastroenterol Hepatol. 2007;22(3):304–10.
Janssen P, Harris MS, Jones M, Masaoka T, Farre R, Tornblom H, et al. The relation between symptom improvement and gastric emptying in the treatment of diabetic and idiopathic gastroparesis. Am J Gastroenterol. 2013;108(9):1382–91.
Levy J, Hayes C, Kern J, Harris J, Flores A, Hyams J, et al. Does cisapride influence cardiac rhythm? Results of a United States multicenter, double-blind, placebo-controlled pediatric study. J Pediatr Gastroenterol Nutr. 2001;32(4):458–63.
Kinoshita Y, Hashimoto T, Kawamura A, Yuki M, Amano K, Sato H, et al. Effects of famotidine, mosapride and tandospirone for treatment of functional dyspepsia. Aliment Pharmacol Ther. 2005;21 Suppl 2:37–41.
Bang CS, Kim JH, Baik GH, Kim HS, Park SH, Kim EJ, et al. Mosapride treatment for functional dyspepsia: a meta-analysis. J Gastroenterol Hepatol. 2015;30(1):28–42.
Du Y, Su T, Song X, Gao J, Zou D, Zuo C, et al. Efficacy and safety of cinitapride in the treatment of mild to moderate postprandial distress syndrome-predominant functional dyspepsia. J Clin Gastroenterol. 2014;48(4):328–35.
Veldhuyzen van Zanten SJ, Jones MJ, Verlinden M, Talley NJ. Efficacy of cisapride and domperidone in functional (nonulcer) dyspepsia: a meta-analysis. Am J Gastroenterol. 2001;96(3):689–96.
Arts J, Caenepeel P, Verbeke K, Tack J. Influence of erythromycin on gastric emptying and meal related symptoms in functional dyspepsia with delayed gastric emptying. Gut. 2005;54(4):455–60.
Barshop K, Kuo B. The investigational drug camicinal for the treatment of gastroparesis. Expert Opin Investig Drugs. 2015;24(1):133–40.
Sanger GJ, Furness JB. Ghrelin and motilin receptors as drug targets for gastrointestinal disorders. Nat Rev Gastroenterol Hepatol. 2016;13(1):38–48.
Kusunoki H, Haruma K, Manabe N, Imamura H, Kamada T, Shiotani A, et al. Therapeutic efficacy of acotiamide in patients with functional dyspepsia based on enhanced postprandial gastric accommodation and emptying: randomized controlled study evaluation by real-time ultrasonography. Neurogastroenterol Motil. 2012;24(6):540–5. e250–1.
Matsueda K, Hongo M, Tack J, Saito Y, Kato H. A placebo-controlled trial of acotiamide for meal-related symptoms of functional dyspepsia. Gut. 2012;61(6):821–8.
Matsushita M, Masaoka T, Suzuki H. Emerging treatments in neurogastroenterology: Acotiamade, a novel treatment option for functional dyspepsia. Neurogastroenterol Motil. 2016;28(5):631–8.
Camilleri M, Acosta A. Emerging treatments in neurogastroenterology: relamorelin: a novel gastrocolokinetic synthetic ghrelin agonist. Neurogastroenterol Motil. 2015;27(3):324–32.
Rodriguez L, Rosen R, Manfredi M, Nurko S. Endoscopic intrapyloric injection of botulinum toxin A in the treatment of children with gastroparesis: a retrospective, open-label study. Gastrointest Endosc. 2012;75(2):302–9.
Sadeghian M, Farahmand F, Fallahi GH, Abbasi A. Cyproheptadine for the treatment of functional abdominal pain in childhood: a double-blinded randomized placebo-controlled trial. Minerva Pediatr. 2008;60(6):1367–74.
Tack J, Janssen P, Masaoka T, Farre R, Van Oudenhove L. Efficacy of buspirone, a fundus-relaxing drug, in patients with functional dyspepsia. Clin Gastroenterol Hepatol. 2012;10(11):1239–45.
Miwa H, Nagahara A, Tominaga K, Yokoyama T, Sawada Y, Inoue K, et al. Efficacy of the 5-HT1A agonist tandospirone citrate in improving symptoms of patients with functional dyspepsia: a randomized controlled trial. Am J Gastroenterol. 2009;104(11):2779–87.
Malatesta MG, Fascetti E, Ciccaglione AF, Cappello G, Grossi L, Ferri A, et al. 5-HT1-receptor agonist sumatriptan modifies gastric size after 500 ml of water in dyspeptic patients and normal subjects. Dig Dis Sci. 2002;47(11):2591–5.
Tack J, Janssen P, Bisschops R, Vos R, Phillips T, Tougas G. Influence of tegaserod on proximal gastric tone and on the perception of gastric distention in functional dyspepsia. Neurogastroenterol Motil. 2011;23(2):e32–9.
Marzio L, Cappello G, Grossi L, Manzoli L. Effect of the 5-HT3 receptor antagonist, ondansetron, on gastric size in dyspeptic patients with impaired gastric accommodation. Dig Liver Dis. 2008;40(3):188–93.
Teich S, Mousa HM, Punati J, Di Lorenzo C. Efficacy of permanent gastric electrical stimulation for the treatment of gastroparesis and functional dyspepsia in children and adolescents. J Pediatr Surg. 2013;48(1):178–83.
Lu PL, Teich S, Di Lorenzo C, Skaggs B, Alhajj M, Mousa HM. Improvement of quality of life and symptoms after gastric electrical stimulation in children with functional dyspepsia. Neurogastroenterol Motil. 2013;25(7):567–e456.
Islam S, McLaughlin J, Pierson J, Jolley C, Kedar A, Abell T. Long-term outcomes of gastric electrical stimulation in children with gastroparesis. J Pediatr Surg. 2016;51(1):67–71.
Friesen CA, Sandridge L, Andre L, Roberts CC, Abdel-Rahman SM. Mucosal eosinophilia and response to H1/H2 antagonist and cromolyn therapy in pediatric dyspepsia. Clin Pediatr (Phila). 2006;45(2):143–7.
Friesen CA, Kearns GL, Andre L, Neustrom M, Roberts CC, Abdel-Rahman SM. Clinical efficacy and pharmacokinetics of montelukast in dyspeptic children with duodenal eosinophilia. J Pediatr Gastroenterol Nutr. 2004;38(3):343–51.
von Arnim U, Peitz U, Vinson B, Gundermann KJ, Malfertheiner P. STW 5, a phytopharmacon for patients with functional dyspepsia: results of a multicenter, placebo-controlled double-blind study. Am J Gastroenterol. 2007;102(6):1268–75.
Braden B, Caspary W, Borner N, Vinson B, Schneider AR. Clinical effects of STW 5 (Iberogast) are not based on acceleration of gastric emptying in patients with functional dyspepsia and gastroparesis. Neurogastroenterol Motil. 2009;21(6):632–8 e25.
Vinson BR, Radke M. The herbal preparation STW 5 for the treatment of functional gastrointestinal diseases in children aged 3–14 years—a prospective non interventional study. Gastroenterology. 2011;140(5 Supplement 1):S-102.
Papathanasopoulos A, Rotondo A, Janssen P, Boesmans W, Farre R, Vanden Berghe P, et al. Effect of acute peppermint oil administration on gastric sensorimotor function and nutrient tolerance in health. Neurogastroenterol Motil. 2013;25(4):e263–71.
Inamori M, Akiyama T, Akimoto K, Fujita K, Takahashi H, Yoneda M, et al. Early effects of peppermint oil on gastric emptying: a crossover study using a continuous real-time 13C breath test (BreathID system). J Gastroenterol. 2007;42(7):539–42.
Kline RM, Kline JJ, Di Palma J, Barbero GJ. Enteric-coated, pH-dependent peppermint oil capsules for the treatment of irritable bowel syndrome in children. J Pediatr. 2001;138(1):125–8.
Mohtashami R, Fallah Huseini H, Heydari M, Amini M, Sadeqhi Z, Ghaznavi H, et al. Efficacy and safety of honey based formulation of Nigella sativa seed oil in functional dyspepsia: a double blind randomized controlled clinical trial. J Ethnopharmacol. 2015;175:147–52.
Ghoshegir SA, Mazaheri M, Ghannadi A, Feizi A, Babaeian M, Tanhaee M, et al. Pimpinella anisum in the treatment of functional dyspepsia: a double-blind, randomized clinical trial. Journal Res Med Sci. 2015;20(1):13–21.
Suzuki H, Matsuzaki J, Fukushima Y, Suzaki F, Kasugai K, Nishizawa T, et al. Randomized clinical trial: rikkunshito in the treatment of functional dyspepsia—a multicenter, double-blind, randomized, placebo-controlled study. Neurogastroenterol Motil. 2014;26(7):950–61.
Hu ML, Rayner CK, Wu KL, Chuah SK, Tai WC, Chou YP, et al. Effect of ginger on gastric motility and symptoms of functional dyspepsia. World J Gastroenterol. 2011;17(1):105–10.
Asher GN, Spelman K. Clinical utility of curcumin extract. Altern Ther Health Med. 2013;19(2):20–2.
Schurman JV, Wu YP, Grayson P, Friesen CA. A pilot study to assess the efficacy of biofeedback-assisted relaxation training as an adjunct treatment for pediatric functional dyspepsia associated with duodenal eosinophilia. J Pediatr Psychol. 2010;35(8):837–47.
Vlieger AM, Rutten JM, Govers AM, Frankenhuis C, Benninga MA. Long-term follow-up of gut-directed hypnotherapy vs. standard care in children with functional abdominal pain or irritable bowel syndrome. Am J Gastroenterol. 2012;107(4):627–31.
van Tilburg MA, Chitkara DK, Palsson OS, Turner M, Blois-Martin N, Ulshen M, et al. Audio-recorded guided imagery treatment reduces functional abdominal pain in children: a pilot study. Pediatrics. 2009;124(5):e890–7.
Levy RL, Langer SL, Walker LS, Romano JM, Christie DL, Youssef N, et al. Twelve-month follow-up of cognitive behavioral therapy for children with functional abdominal pain. JAMA Pediatr. 2013;167(2):178–84.
Warschburger P, Calvano C, Becker S, Friedt M, Hudert C, Posovszky C, et al. Stop the pain: study protocol for a randomized-controlled trial. Trials. 2014;15:357.
Kuttner L, Chambers CT, Hardial J, Israel DM, Jacobson K, Evans K. A randomized trial of yoga for adolescents with irritable bowel syndrome. Pain Res Manag. 2006;11(4):217–23.
Kim KN, Chung SY, Cho SH. Efficacy of acupuncture treatment for functional dyspepsia: a systematic review and meta-analysis. Complement Ther Med. 2015;23(6):759–66.
Hyams JS, Davis P, Sylvester FA, Zeiter DK, Justinich CJ, Lerer T. Dyspepsia in children and adolescents: a prospective study. J Pediatr Gastroenterol Nutr. 2000;30(4):413–8.
Miele E, Simeone D, Marino A, Greco L, Auricchio R, Novek SJ, et al. Functional gastrointestinal disorders in children: an Italian prospective survey. Pediatrics. 2004;114(1):73–8.
Rippel SW, Acra S, Correa H, Vaezi M, Di Lorenzo C, Walker LS. Pediatric patients with dyspepsia have chronic symptoms, anxiety, and lower quality of life as adolescents and adults. Gastroenterology. 2012;142(4):754–61.
Mohammad S, Di Lorenzo C, Youssef NN, Miranda A, Nurko S, Hyman P, et al. Assessment of abdominal pain through global outcomes and recent FDA recommendations in children: are we ready for change? J Pediatr Gastroenterol Nutr. 2014;58(1):46–50.
Rashid AN, Taminiau JA, Benninga MA, Saps M, Tabbers MM. Definitions and outcome measures in pediatric functional upper gastrointestinal tract disorders: a systematic review. J Pediatr Gastroenterol Nutr. 2016;62(4):581–7.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Rosen, J.M., Saps, M. (2017). Functional Dyspepsia. In: Faure, C., Thapar, N., Di Lorenzo, C. (eds) Pediatric Neurogastroenterology. Springer, Cham. https://doi.org/10.1007/978-3-319-43268-7_36
Download citation
DOI: https://doi.org/10.1007/978-3-319-43268-7_36
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-43266-3
Online ISBN: 978-3-319-43268-7
eBook Packages: MedicineMedicine (R0)