Abstract
Contact dermatitis is an inflammatory skin disease induced by direct contact of a external agent to the skin. It can be classified into two main types: Irritant contact dermatitis and Allergic contact dermatitis. Irritant contact dermatitis represents a non-specific cutaneous response to the toxic or physical effects of environmental agents, while Allergic contact dermatitis represents a specific type IV hypersensitivity reaction to specific haptens. Both types are characterized by a highly variable clinical presentation that includes erythema, papules, vesicles, bullae, scaling and erosions in acute cases, and papules, plaques, lichenification, hyperkeratosis and fisures in the chronic. Pruritus is a very common symptom most frequently associated with Allergic contact dermatitis but also frequent in Irritant contact dermatitis. Furthermore, occasionally pruritus may be the leading or only symptom that guides the clinician to suspect the diagnosis of Contact dermatitis, as it is in the case of Allergic contact dermatitis of the anogenital region or when the process occurs in the elderly. Although the mechanisms underlying the pathogenicity of the inflammatory cutaneous response in irritant and allergic contact dermatitis has been widely studied, little is known about the mechanisms leading to pruritus. This chapter summarizes the most important aspects of contact dermatitis in these specific situations as well as the last insights into the pathogenicity of pruritus in contact dermatitis.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Contact dermatitis
- Allergic contact dermatitis
- Irritant contact dermatitis
- Pruritus
- Anogenital pruritus
- Pruritus ani
- Pruritus elderly
- Transient receptor potential (TRP) ion channels
Introduction
Contact dermatitis is an inflammatory skin disease induced by direct contact of a external agent to the skin. Contact dermatitis can be classified into two main types: Irritant contact dermatitis and Allergic contact dermatitis. Irritant contact dermatitis is the most common form of contact dermatitis and represents a non-specific cutaneous response to the toxic or physical effects of a wide variety of environmental agents. It is a dose and time dependent process that may occur in all individuals exposed [1]. On the other hand, ACD represents a type IV hypersensitivity reaction mediated by specific T cell-lymphocytes that recognize low molecular weight substances, called haptens. The development of ACD depends on an individual susceptibility and requires prior sensitization to the specific hapten [1].
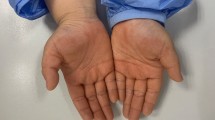
The clinical presentation of ICD and ACD is highly variable and include macular erythema, edema, papules, vesicles, bullae, scaling and erosions in acute cases, and papules, plaques, lichenification, hyperkeratosis and fisures in the chronic. Although the clinical appearance of both types of CD may be similar and patch testing be the only current means of differentiation, several but not conclusive clinical clues may be helpful [2]. Irritant contact dermatitis may be produced after a single enviromental exposure with the onset of symptoms within minutes to several hours after the contact. There is usually a sharp circumspection of the dermatitis, with a lack of tendency for spread (Fig. 20.1). Allergic contact dermatitis, in contrast, requires a previous contact with the allergen and time to develop the sensitization. Dermatitis develops hours to days after the exposure and lesions are usually ill-defined (Fig. 20.2).
In both types of CD the pruritus is a very common symptom, however, in ICD it is usually mild and often replaced by a burning, pain and stinging sensation. Pruritus in ACD can be the most important symptom and is consider, as one of the main aspects strongly associated with the poor quality of life in patients [3]. Its presence probably reflects the allergic pathogenicity of ACD and also plays an important role in its severity and chronicity as it may lead to scratching and further skin damage with the secondary access for more allergens [4, 5]. Furthermore, occasionally pruritus may be the leading or only symptom that guides the clinician to suspect the diagnosis of CD. This is specially true when the process involves certain locations such as the anogenital regions or when the CD occurs in the elderly.
Pruritus and Anogenital Contact Dermatitis
Contact dermatitis of the anogenital region is a common phenomenom. The particular anatomic and physiologic characteristics of this region makes it very susceptible to develop allergic and irritant contact dermatitis. The skin is continually exposed to different secretions as well as the occlussion, friction and sweat characteristic of this region. In addition, several substances and topical medications, are often retained, increasing time exposure and resulting in more frequent and severe reactions. It is therefore not surprising that ICD and ACD are one of the most common causes of vulvar and perianal dermatitis [6–8]. Irritant contact dermatitis is usually produced by lack or excess of hygiene. Poor hygiene leads to prolonged exposure of physiological fluids or depositions that acts as strong irritants, as in the case of patients with urinary or fecal incontinence. On the other hand the excesive hygiene and exposure of detergents and soaps can also damage the skin and leads to ICD. In cases of ACD, the disease can be a primary disorder or a complication of a preexisting condition, including an ICD treated with multiple topical treatments [9]. Up to 57 % of patients with anogenital complaints report to applied different chemicals and medications to this particular area and positive patch test reactions have been found in up to 78 % of patients with anogenital symptoms [10]. The symptoms are usually nonspecific, being pruritus the most commonly reported. Fragances, topical antibiotics, over-the counter-remedies and topical anesthetics are the most common allergens implicated [11–14]. Other allergens such as spices, plants, rubbers and glues have also been reported. Some series report nickel as one of the most common allergen in vulvar pruritus, however, the relevance of nickel has to be assesed carefully as in many cases its relevance its questionable [6, 11, 12, 15, 16]. A single case of chronic anal pruritus was reported due to a systemic contact dermatitis to nickel [17]. Due the high prevalence of ACD in women with vulvar symptoms, patch test to rule out ACD is recommended for all patients with non-specific chronic vulvar symptoms, specially if they have pruritus.
Pruritus and Contact Dermatitis in the Elderly
Contact dermatitis manifested by acute or chronic pruritus is a common complaint among aged persons. The inflamatory reaction is more subtle in this population and dermatitis is therefore less visible, being pruritus the only symptom. The likelihood of developing an ICD and ACD varies with the age and the type of irritant [17, 18]. The irritant response to the contact of a external agent is known to be higher in childhood and lower in the elderly. A decrease irritative response to various compounds such as sodium lauryl sulphate, dimethyl sulphoxide, histamine, ethynil nicotate, “croton oil”, clorophorm-methanol and lactic acid has been shown in several studies. However, the elderly shows also an increase irritant response with other substances such as soaps and detergents which make them more prone to develop an ICD [17, 19–21].
In case of ACD, the prevalence in the elderly population has been reported to be up to 11 %, being more common in women than in men [22, 23].
Pruritus has been the most common complaint, with an overall prevalence of 29 % in subjects ranking in age from 50 to 91 years (mean age, 75 years [24]. In some cases a history of severe pruritus, without any visible sign of dermatitis has been associated with positive patch test results [24].
Patch test results in elderly are varied. Although elderly people present a decrease in their inmune system response with a decline of delayed contact reactions to some patch test allergenes, the abnormalities in permeability of the epidermal barrier and the long time and high level of exposure to new different allergens increase the potential of allergen sensitization [24–27]. A lower frequency of positive patch test reactions to thimerosal, nickel, epoxy resin and cobalt chloride has been reported [28–30]. Instead, other allergens such as primin, diaminiodiphenylmethane, neomycine sulphate, lanolin alcohols, paraben mix, Euxyl 400, quinoline mix and methylisothiazolinone showed higher sensitization rates [25, 31]. The use of topical treatments to treat leg ulcers or xerosis are often the most common cause of sensitization. Patients usually develop pruriginous eccematous reactions on their wounds and the surrounding skin. In addition, the frequent consumption of drugs chemically related to topics sensitizers leads them to develop eczematous rashes which are more extensive and symmetrical, and often associated with much itching. Therfore ICD and ACD should be consider in all elderly patient with acute or chronic pruritus, specially if they have eccema of unknown etiology.
Pathogencity
While the mechanisms underlying the pathogenicity of the inflammatory cutaneous response in irritant and allergic contact dermatitis has been widely studied, little is known about the mechanisms leading to pruritus. Inflammation in ICD is known to be produced by multiple mechanisms including skin barrier disruption and epidermal changes, which leads to inflammatory infiltrates and cytokine release. Exposure to an irritant would disrupt the epidermal barrier inducing the release of proinflammatory cytokines such as interleukin (IL-1), IL-1beta, IL-6 and tumour necrosis factor (TNF) alpha from keratinocytes injured. Several other inflammatory cells, cytokines and intracellular adhesion molecules help to maintain the inflammatory process [32].
In ACD the inflamation results from a T cell-mediated, delayed type hypersensitivity (DTH) reaction. The process can be divided into two phases: The sensitization or aferent phase and the elicitation or eferent phase. The sensitizaton phase involves profesional antigen presenting cells which initiate an adaptative immune response. As a result a clonal expansion of hapten-specific memory/effector T cells is created. This cells can be found in lymph nodes, blood, and the skin of sensitized individuals and are activated upon reexposure with the same antigen in the elicitation phase. The elicitation phase is the responsible for the cutaneous manifestations of the ACD. The offending hapten activates CD8+ T cells which then initiate the inflamatory response.
Pruritus in contact dermatitis is known to be produce by excitation of small sensory nerves by the inflamed skin, however the exact pruritic pathway of activation is not well understood. The fact that antihistaminics usually do not subside pruritus does raise the possibility that pruritus associated with contact dermatitis may be mediated by histamine-independent inflammatory pathways [33, 34]. Several nonhistaminergic mediators such as substance P, Endothelin 1, 5-Hydroxytryptamine (5-HT), chloroquine, BAM8-22 peptide, leukotriene B4 and prostaglandin E2 induced pruritus when injectec to skin [4].
Animal models have shown that some of this mediators may act through downstream activation of transient receptor potential (TRP) cation channel, subfamily A, member 1 (TRPA1) ion channels. Inhibition of TRPA1 or its genetic deletion (TRPA1 −/−) in mice showed diminished chronic dermatitis and reduced scratching behavior. In addition, the Neurokinin-1 receptor (NK1R) may also be involved since its inhibition effectively suppressed dermatitis and pruritus in ACD. Furthermore, the inflammatory process also seems to play an important role in the development and persistence of pruritus. Bradykinin, an algesic chemical, which normally induce pain in healthy skin of humans and mice, evokes pruritus in a skin contact dermatitis [35]. Mediators that are chronically elevated in ACD such as 4-hydroxynonenal (4-HNE) may increase the activity of TRPA1 channels in sensory nerves resulting in pruritus. There is thus a direct relationship between pruritus and inflammation and probably neuronal TRPA1 channels and other receptors serve as major integrator of the neuronal and inflammatory process.
Treatment
The primary therapeutic intervention to treat and prevent irritant and allergic contact dermatitis is withdrawal and avoidance of the causative agent. Treatment of pruritus is also one of the main therapeutic goals as it leads to scratching and secondary access to more irritants, allergens or pathogens. The first line treatment for localized CD are topical corticosteroids [36]. The potency of the corticosteroid is subject to the location and severity of the dermatitis. Topical corticosteroids have shown efficacy in eczema-related itch and relief of pruritus is usually achieved in the first 3 days of treatment [37–42]. Addition of other antipruritic agents such as pramoxine may also increased the anti-itch efficacy [42]. In cases of bacterial superinfection topical or oral antibiotics may be added to the treatment. Systemic treatment with oral corticosteroids is used in cases with great extension (involvement of more than 20 %) or cases of acute dermatitis involving face or genitalia [43, 44].
In chronic localized dermatitis without response, or with partial response to topical corticosteroids, topical calcineurin inhibitors such as tacrolimus or pimecrolimus can be effectively used [45–47].
Systemic treatment with phototherapy or immunosuppressive drugs such as azathioprine, mycophenolate mofetil and cyclosporine may be used in exceptional cases without response to corticosteroid treatment [48, 49].
Regular use of barrier creams and emmolients may also help to mantain the skin barrier function and prevent the development of dermatitis [36, 50].
References
AIe IS, Maibach HI. Irritant contact dermatitis. Rev Environ Health. 2014;29(3):195–206.
Rietschel RL. Clues to an accurate diagnosis of contact dermatitis. Dermatol Ther. 2004;17(3):224–30.
Ayala F, Nino M, Fabbrocini G, Panariello L, Balato N, Foti C, et al. Quality of life and contact dermatitis: a disease-specific questionnaire. Dermatitis. 2010;21(2):84–90.
Pall PS, Hurwitz OE, King BA, LaMotte RH. Psychophysical measurements of itch and nociceptive sensations in an experimental model of allergic contact dermatitis. J Pain. 2015;16(8):741–9.
Bruckner AL, Weston WL. Allergic contact dermatitis in children: a practical approach to management. Skin Therapy Lett. 2002;7(8):3–5.
Haverhoek E, Reid C, Gordon L, Marshman G, Wood J, Selva-Nayagam P. Prospective study of patch testing in patients with vulval pruritus. Australas J Dermatol. 2008;49(2):80–5.
Lambert J. Pruritus in female patients. Biomed Res Int. 2014;2014:541867.
Schlosser BJ. Contact dermatitis of the vulva. Dermatol Clin. 2010;28(4):697–706.
Al-Niaimi F, Felton S, Williams J. Patch testing for vulval symptoms: our experience with 282 patients. Clin Exp Dermatol. 2014;39(4):439–42.
Margesson LJ. Contact dermatitis of the vulva. Dermatol Ther. 2004;17(1):20–7.
Marren P, Wojnarowska F, Powell S. Allergic contact dermatitis and vulvar dermatoses. Br J Dermatol. 1992;126(1):52–6.
Lewis FM, Harrington CI, Gawkrodger DJ. Contact sensitivity in pruritus vulvae: a common and manageable problem. Contact Dermatitis. 1994;31(4):264–5.
Nardelli A, Degreef H. Goossens A Contact allergic reactions of the vulva: a 14-year review. Dermatitis. 2004;15(3):131–6.
Lucke TW, Fleming CJ, McHenry P, Lever R. Patch testing in vulval dermatoses: how relevant is nickel? Contact Dermatitis. 1998;38(2):111–2.
Vermaat H, Smienk F, Rustemeyer T, Bruynzeel DP, Kirtschig G. Anogenital allergic contact dermatitis, the role of spices and flavour allergy. Contact Dermatitis. 2008;59(4):233–7.
Silvestri DL, BArmettler S. Pruritus ani as a manifestation of systemic contact dermatitis: resolution with dietary nickel restriction. Dermatitis. 2001;22(1):50–5.
Nilzen A, Voss-Lagerlund K. Epicutaneous tests with detergents and a number of other common allergens. Dermatologica. 1962;124:42–52.
Patil S, Maibach HI. Effect of age and sex on the elicitation of irritant contact dermatitis. Contact Dermatitis. 1994;30(5):257–64.
Cua AB, Wilhelm KP, Maibach HI. Cutaneous sodium lauryl sulphate irritation potential: age and regional variability. Br J Dermatol. 1990;123(5):607–13.
Coenraads PJ, Bleumink E, Nater JP. Susceptibility to primary irritants: age dependence and relation to contact allergic reactions. Contact Dermatitis. 1975;1(6):377–81.
Skassa-Brociek W, Manderscheid JC, Michel FB, Bousquet J. Skin test reactivity to histamine from infancy to old age. Allergy Clin Immunol. 1987;80(5):711–6.
Fitzpatrick JE. Common inflammatory skin diseases of the elderly. Geriatrics. 1989;44(7):40–6.
Beauregard S, Gilchrest BA. A survey of skin problems and skin care regimens in the elderly. Arch Dermatol. 1987;123(12):1638–43.
Mangelsdorf HC, Fleischer AB, Sherertz EF. Patch testing in an aged population without dermatitis: high prevalence of patch test positivity. Am J Contact Dermat. 1996;7(3):155–7.
Piaserico S, Larese F, Recchia GP, Corradin MT, Scardigli F, Gennaro F, et al. Allergic contact sensitivity in elderly patients. North-East Italy Contact Dermatitis Group. Aging Clin Exp Res. 2004;16(3):221–5.
Elias PM, Ghadially R. The aged epidermal permeability barrier: basis for functional abnormalities. Clin Geriatr Med. 2002;18(1):103–20.
Ghadially R. Aging and the epidermal permeability barrier: implications for contact dermatitis. Am J Contact Dermat. 1998;9(3):162–9.
Wantke F, Hemmer W, Jarisch R, Götz M. Patch test reactions in children, adults and the elderly. A comparative study in patients with suspected allergic contact dermatitis. Contact Dermatitis. 1996;34(5):316–9.
Uter W, Geier J, Pfahlberg A, Effendy I. The spectrum of contact allergy in elderly patients with and without lower leg dermatitis. Dermatology. 2002;204(4):266–72.
Bangha E, Elsner P. Sensitizations to allergens of the European standard series at the Department of Dermatology in Zurich 1990–1994. Dermatology. 1996;193(1):17–21.
Lundov MD, Opstrup MS, Johansen JD. Methylisothiazolinone contact allergy – growing epidemic. Contact Dermatitis. 2013;69(5):271–5.
Smith HR, Basketter DA, McFadden JP. Irritant dermatitis, irritancy and its role in allergic contact dermatitis. Clin Exp Dermatol. 2002;27(2):138–46.
Funk JO, Maibach HI. Horizons in pharmacologic intervention in allergic contact dermatitis. J Am Acad Dermatol. 1994;31(6):999–1014.
Thurmond RL, Gelfand EW, Dunford PJ. The role of histamine H1 and H4 receptors in allergic inflammation: the search for new antihistamines. Nat Rev Drug Discov. 2008;7(1):41–53.
Fu K, Qu L, Shimada SG, Nie H, LaMotte RH. Enhanced scratching elicited by a pruritogen and an algogen in a mouse model of contact hypersensitivity. Neurosci Lett. 2014;579:190–4.
Bourke J, Coulson I, English J. Guidelines for the management of contact dermatitis: an update. British Association of Dermatologists Therapy Guidelines and Audit Subcommittee. Br J Dermatol. 2009;160(5):946–54.
Hoare C, Li Wan Po A, Williams H. Systematic review of treatments for atopic eczema. Health Technol Assess. 2000;4(37):1–191.
Thomas KS, Armstrong S, Avery A, Po AL, O'Neill C, Young S, et al. Randomised controlled trial of short bursts of a potent topical corticosteroid versus prolonged use of a mild preparation for children with mild or moderate atopic eczema. BMJ. 2002;324(7340):768.
Luger TA. Balancing efficacy and safety in the management of atopic dermatitis: the role of methylprednisolone aceponate. J Eur Acad Dermatol Venereol. 2011;25(3):251–8.
Ruzicka T. Methylprednisolone aceponate in eczema and other inflammatory skin disorders – a clinical update. Int J Clin Pract. 2006;60(1):85–92.
Bieber T, Vick K, Fölster-Holst R, Belloni-Fortina A, Städtler G, Worm M, et al. Efficacy and safety of methylprednisolone aceponate ointment 0.1% compared to tacrolimus 0.03% in children and adolescents with an acute flare of severe atopic dermatitis. Allergy. 2007;62(2):184–9.
Curto L, Carnero L, López-Aventin D, Traveria G, Roura G, Giménez-Arnau AM. Fast itch relief in an experimental model for methylprednisolone aceponate topical corticosteroid activity, based on allergic contact eczema to nickel sulphate. J Eur Acad Dermatol Venereol. 2014;28(10):1356–62.
American Academy of Allergy, Asthma and Immunology, American College of Allergy, Asthma and Immunology. Contact dermatitis: a practice parameter. Ann Allergy Asthma Immunol. 2006;97(3 Suppl 2):S1–38.
Meingassner JG, Grassberger M, Fahrngruber H, Moore HD, Schuurman H, Stütz A. A novel anti-inflammatory drug, SDZ ASM 981, for the topical and oral treatment of skin diseases: in vivo pharmacology. Br J Dermatol. 1997;137(4):568–76.
Queille-Roussel C, Graeber M, Thurston M, Lachapelle JM, Decroix J, de Cuyper C, Ortonne JP. SDZ ASM 981 is the first non-steroid that suppresses established nickel contact dermatitis elicited by allergen challenge. Contact Dermatitis. 2000;42(6):349–50.
Belsito D, Wilson DC, Warshaw E, Fowler J, Ehrlich A, Anderson B, et al. A prospective randomized clinical trial of 0.1% tacrolimus ointment in a model of chronic allergic contact dermatitis. J Am Acad Dermatol. 2006;55(1):40–6.
Pacor ML, Di Lorenzo G, Martinelli N, Mansueto P, Friso S, Pellitteri ME, et al. Tacrolimus ointment in nickel sulphate-induced steroid-resistant allergic contact dermatitis. Allergy Asthma Proc. 2006;27(6):527–31.
van Coevorden AM, Kamphof WG, van Sonderen E, Bruynzeel DP, Coenraads PJ. Comparison of oral psoralen-UV-A with a portable tanning unit at home vs hospital-administered bath psoralen-UV-A in patients with chronic hand eczema: an open-label randomized controlled trial of efficacy. Arch Dermatol. 2004;140(12):1463–6.
Sezer E, Etikan I. Local narrowband UVB phototherapy vs. local PUVA in the treatment of chronic hand eczema. Photodermatol Photoimmunol Photomed. 2007;23(1):10–4.
Williams C, Wilkinson SM, McShane P, Lewis J, Pennington D, Pierce S, et al. A double-blind, randomized study to assess the effectiveness of different moisturizers in preventing dermatitis induced by hand washing to simulate healthcare use. Br J Dermatol. 2010;162(5):1088–92.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer-Verlag London
About this chapter
Cite this chapter
Rozas-Muñoz, E., Serra-Baldrich, E. (2016). Allergic and Irritant Contact Dermatitis. In: Misery, L., Ständer, S. (eds) Pruritus. Springer, Cham. https://doi.org/10.1007/978-3-319-33142-3_20
Download citation
DOI: https://doi.org/10.1007/978-3-319-33142-3_20
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-33140-9
Online ISBN: 978-3-319-33142-3
eBook Packages: MedicineMedicine (R0)