Abstract
Historically, the majority of plant virology focused on agricultural systems. Recent efforts have expanded our knowledge of the true diversity of plant viruses by studying those viruses that infect wild, undomesticated plants. Those efforts have provided answers to basic ecological questions regarding viruses in the wild, and insights into evolutionary questions, regarding the origins of viruses. While much work has been done, we have merely scratched the surface of the diversity that is estimated to exist. In this chapter we discuss the state of our knowledge of virus diversity, both in agricultural systems as well as in native wild systems, the border between these two systems and how viruses adapt and move across this border into an artificial, domesticated environment. We look at how this diversity has affected our outlook on viruses as a whole, shifting our past view of viruses as purely antagonistic entities of destruction to one where viruses are in a mutually beneficial relationship with their hosts. Additionally, we discuss the current work that plant virology has put forth regarding the evolutionary mechanisms, the life histories, and the deep evolution of viruses.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
8.1 Introduction
Until recent years, our knowledge of the breadth of plant virus diversity was limited. The field of plant virology traditionally has focused on agricultural systems, with little study of viruses found in wild plants. In the past decade, several efforts have begun to fill these gaps in the form of biodiversity surveys. These surveys have given us a new view into the true diversity of plant viruses, as well as their distribution.
One of the most powerful advances in microbe discovery has been massively parallel sequencing, or Next Generation Sequencing (NGS). Previously the most common methods for virus detection were protein based immunological tests such as ELISA, or nucleotide specific PCR assays. Neither of these are sensitive enough to detect low titers of virus in wild plants or general enough to detect novel viruses, or even related strains or species. NGS technology has boosted our ability to fully sequence whole genomes, and advanced the field of metagenomics, the study of all the genetic information from a given environment. When the requirement for culture is removed, the ability to sequence and identify fastidious or unculturable microbes becomes possible.
NGS has become the gold standard for metagenomics. Metagenomics can be used to identify novel virus species, using various techniques to enrich viral nucleic acids such as isolating specific forms of RNA (dsRNA, siRNA, ssRNA) or virus particle isolation. Each method has positive and negative aspects (Stobbe and Roossinck 2014; Roossinck et al. 2015). While NGS can be used for virus discovery, it also has been applied to plant virus diagnostics (Stobbe et al. 2013; Massart et al. 2014). NGS also has been used to look further into the population diversity of individual virus strains. Using this deep sequencing, one is able to determine all of the minor variants found in a given infection (Simmons et al. 2012). NGS has applications in many evolutionary questions regarding systemic movement, vectoring and epidemiology.
In this chapter we look into the recent work looking into the diversity of plant viruses, not only in species diversity but also diversity within the species or quasispecies. This variation comes from many sources, including high mutation rates of RNA viruses, recombination and reassortment. The variation we see within a single plant host has profound effects on the how the virus responds to selective pressures associated with new hosts, and factors such as the bottleneck events associated with cell-to-cell movement or vectoring. Additionally, with our ever increasing knowledge of the breadth of virus diversity, as well as advances in technology, questions of the deep evolutionary history of viruses and their relationship to their hosts are beginning to be answered. While there has been a large body of work on algae-infecting viruses (VanEtten and Dunigan 2012), here we only consider the viruses of vascular plants.
8.2 Viruses Within Agricultural Systems
Agriculture has been an important aspect of virology from the beginning of the field (Beijerinck 1898), and has been the focus of most work in plant virology throughout its 120 year history. Much of the early work characterizing and describing viruses was done with viruses of crop plants. While it is understandable that so much work has been put into a few specific plant species, this has left out a lot of information about viruses in natural settings. In modern agriculture the use of vast areas of monoculture, extended growing seasons, irrigation, and artificial soil amendments have each impacted plant pathogen prevalence, including viruses.
8.2.1 Human Effect on Virus Diversity
Agriculture is a human invention, and the cultivation of crops has propelled the human race to increasing cultural and technological advances, but with this advancement we have disturbed many natural systems throughout our history. Domestication of the earliest crops probably began about 10,000 to 12,000 years ago (Balter 2007), presumably with their viruses experiencing a shift in selective pressure as well (Stukenbrock and McDonald 2008). Densely spaced, monoculture crops have been increasingly favored due to the ease of production, but these conditions also are excellent for the spread and infection of viruses and other deleterious microbes within the crop (Thresh 1982; Power and Mitchell 2004). To combat the yield loss associated with virus–induced disease, breeders have focused efforts on engineering disease resistant cultivars of crops. However, several forms of virus variation, such as the high mutation rates of RNA and some DNA viruses, recombination, and reassortment lead to resistance breaking (Duffy and Holmes 2008; McDonald and Linde 2002; Harrison 2002). Although breeding of resistant cultivars has had some success, other methods such as increasing the plant species diversity in a given area, breaking the spatial and temporal components of the disease cycle, have been suggested (Ratnadass et al. 2012). For example, genetic diversity (heterosis) induced tolerance to Turnip mosaic virus in wild cress (Lepidium sp.) hybrids, while plants that were selfed were more susceptable to disease, suggesting that small populations with low genetic diversity could lead to increased disease symptoms, and infection rates (Houliston et al. 2015). Intercropping cowpea with cassava or plantains has reduced the incidence of viruses in Central America (Valverde et al. 1982). These practices suggest that increases in plant diversity, either within a species or with diverse species, could lower the incidence or pathology of viruses (see Sect. 8.2.3).
With the globalization of today’s society, it is not surprising to find that humans are playing a role in the movement of plant viruses. Human movement of both the plants and vectors associated with pathogens has facilitated the spread of viruses. The effects of climate change in the form of CO2 and ozone may change the impacts of viruses on their plant hosts (Trebicki et al. 2015). While many pathogens move closer to the poles as climate change occurs, there is some evidence that viruses and nematodes are moving closer to the equator (Bebber et al. 2013). This may be an analytical artifact as viral symptoms are often misdiagnosed. Increases in the range of insect vectors of viruses due to numerous factors, including climate change, predicts increased virus spread (Fereres 2015).
8.2.2 Vectors
A majority of crop viruses are insect vectored, and relationships between plants, viruses and insects are complex (Roossinck 2015b). Using insect vectors as targets for virus discovery is an attractive opportunity. Vector-enabled metagenomics is a recent method for virus discovery that allows one to discover and characterize viruses that are in the area that the vector occupies, including both cultivated and wild plants. In a recent study using vector-enabled metagenomics 79 % of the sequences obtained were related to known viruses, suggesting that many vector transmitted viruses are known (Ng et al. 2011). This number was much higher than the number of identifiable virus sequences found in wild plants, where as many as 60 % of sequences from virus-enriched pools have no similarity to sequences in GenBank (Roossinck et al. 2015).
The “Viral Manipulation Hypothesis” states that by modifying the production of certain volatiles, the plant host will be more attractive to the virus’ vectors (Gutiérrez et al. 2013). Vector transmission mechanisms of plant viruses influence the effects the virus has on the plant host, with persistently transmitted viruses tending to either improve the host quality for the vector or mimic high quality, and nonpersistantly transmitted viruses lowering quality to facilitate the rapid dispersal of the viruliforous insect to neighboring plants (Mauck et al. 2012). Host manipulation is seen in unrelated families of plant viruses, implying convergent evolution (Wu et al. 2014). Barley yellow mosaic virus (BYDV) is persistently transmitted by the aphid Rhopalosiphum padi. Virus-free aphids have a feeding preference for BYDV-infected plants, while the reverse is true for BYDV-carrying aphids (Ingwell et al. 2012). Cucumber mosaic virus (CMV), a member of the Bromoviridae family, increases the release of volatiles that mimic healthy plants, attracting vectors despite the low quality of the plant for the aphids (Mauck et al. 2015b). Additionally, CMV effects other non-vectoring insects, repelling some while attracting others, in the absence of aphids (Mauck et al. 2015a). In mixed infections, competition can favor one microbe over another, as seen with the potyvirus, Zucchini yellow mosaic virus (ZYMV) out-competing another potyvirus, Watermelon mosaic virus (WMV). ZYMV and WMV are very similar viruses, in terms of genetics, hosts, and vectors. These similarities places them in direct competition with each other. One important difference is in vector manipulation; ZYMV manipulates the host-aphid relationship, while WMV does not. When co-infecting a plant, ZYMV will maintain its typical level of replication, while the replication of WMV is reduced. Despite this, WMV is still transmitted from a mixed infection, taking advantage of ZYMV host manipulation that attracts the aphid vectors (Salvaudon et al. 2013). In an analysis of genetic turnover during transmission, several clones containing the same mutation leading to a premature stop codon was found within a plant. Further transmissions using this experimental isolate lost this mutation, but this suggests that ZYMV has the ability to complement defective ZYMV genomes in the aphid vector (Simmons et al. 2011).
Viruses not only manipulate their hosts, they also respond to the presence of a vector feeding. Cauliflower mosaic virus (CaMV), a double-stranded DNA virus in the Caulimoviridae family, is acquired up by its aphid vector packaged into transmission bodies. The transmission bodies change their morphology in different contexts, such as in response to C02 levels or host wounding. In addition, the transmission bodies change to a morphology that favors transmission when in proximity to the saliva from aphid feeding (Martinière et al. 2013).
8.2.3 The Agro-Eco Border: Spill Over and Movement
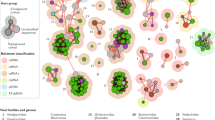
Obviously there are many differences between agricultural systems, and ecosystems of wild undisturbed plants. We have already touched on the use of monoculture, and the effect of plant biodiversity on the diversity of viruses, in this section we look into how nearby systems can influence viruses. The intersection of wild and agricultural systems has been described as the agro-eco border (Roossinck and García-Arenal 2015). This border may be the source of new pathogenic plant viruses that can impact crops in spillover events (Fig. 8.1).
Spillover of viruses from wild plants. Viruses are abundant and often inapparent in wild plants. At the agro-eco boundary viruses may move into crop plants from nearby wild plants. In most cases these infections will be dead-end: either the virus is not competent for further transmission in the new host, it may not establish sufficient virus titer to allow transmission, or it may rapidly kill the host. Rarely, a spillover virus may develop the ability to be further transmitted to more similar hosts, resulting in an emerging virus infection
Due to the relatively recent introduction of modern agriculture to Australia (in the past 200 years), considerable work has been done on this continent to look at the effect of agriculture across this border. Many of the native viruses in Australia have not been influence by agriculture. Three different new encounter events of legume-infecting potyviruses have been described in Australia. The interspecies genetic diversity of each virus differed, with the native viruses having greater diversity than the exotic viruses (Webster et al. 2007).
Viruses that infect crop plants often find reservoirs within nearby wild plants or in volunteer plants from the previous crop. These viruses, that cause disease in crops, may be asymptomatic in other hosts. For example, Peanut stunt virus causes disease in peanuts, but is asymptomatic in clover (Sherwood 1997). The presence of highly susceptible hosts in a plant community can increase the incidence of the virus across all susceptible species in the community (Power 2008). Several scenarios can be seen where the spread of virus moves between an asymptomatic native host to cultivated plants (Jones 2014). Emergent viral diseases may come from “silent” infections within a nearby wild population, and are driven by anthropogenic factors, such as food production or the introduction of vectors (Anderson et al. 2004). In Africa, there have been several emergent viruses in agricultural systems. While many changes in the pathogens themselves have promoted the emergence of disease, changes in agricultural practices have also promoted the introductions. Rice yellow mottle virus (RYMV, in the genus Sobemovirus) infects Oryza sp. in both wild and agricultural systems. RYMV was first described in 1966 in Kenya and is currently an economically important plant virus. The rise of rice production in Kenya is thought to be the main driver of RYMV spread, as epidemics of the virus were not seen until after the intensification of rice production in the 1960s (Fargette et al. 2006). Despite many examples of host jumps leading to disease, there have been examples of viruses that have switched hosts several times with no apparent increase in pathology (Thresh 2006).
The level of biodiversity on the wild side of the agro-eco border effects the emergence of viral movement across the border. A lowered incidence of two begomoviruses was seen in wild peppers with decreasing levels of cultivation or management, suggesting a dilution effect with higher levels of biodiversity (Pagán et al. 2012). This correlation with biodiversity appears to hold true with CMV in wild plants, but not with CMV in crops (Sacristán et al. 2004). The loss of biodiversity appears to increase the movement of a virus across the agro-eco border, but a high degree of biodiversity can lead to a large number of viral species, which may serve as a reservoir for new infections (Keesing et al. 2010). Opportunistic viruses quickly move into susceptible crops, decrease, then recover in the susceptible population, causing a rapid cycles of epidemics and decline (Harrison 1981; Thresh 1981). In France, where ZYMV and WMV are both present, only WMV has significant natural reserviours, which explains the fragmented nature of ZYMV incidence across France (Lecoq et al. 2014). In Spain, two strains of the potexvirus Pepino mosaic virus (PepMV-EU and PepMV-CH2) co-circulate among tomato crops, with the CH2 strain being the dominate strain. PepMV-EU primarily exists in coinfections with PepMV-CH2, and these coinfections allow for an extended host range of PepMV, thus extending the potential number of reservoirs. This has implications for coinfection effecting the emergence of PepMV in tomato plants (Gómez et al. 2009).
8.3 Viruses Within Natural Systems
Early attempts to explore virus prevalence within wild plants was hampered by a lack of sensitive detection methods (Cooper and Jones 2006). In recent years, there have been a number of plant virus diversity surveys, in which plant tissue was sampled without targeting symptomatic plants (Wren et al. 2006; Roossinck 2013). These tissue samples were then enriched for viral nucleic acid, and sequenced using NGS. The prevalence and distribution of viruses in these studies varies, but inevitably evidence for many novel virus is found, and the variation of viruses in wild systems is much greater than what is seen in crops (Roossinck et al. 2015).
8.3.1 Impacts of Viruses on Wild Plants
The enemy-release hypothesis states that plants invading a new territory may have an advantage because they have left behind their pathogens (Power 2008; Rúa et al. 2011). However, in the invasive grasses of the Pacific coastal region of North America, a non-native plant uses its own adapted Barley/Cereal yellow dwarf virus (B/CYDV) to gain an advantage over the native grasses (Malmstrom et al. 2005). The reverse was seen in another related system; Venetanata dubai (African wiregrass), an invasive non-native grass that is not adapted to B/CYDV was slowed in its movement across the northwest grasslands of America (Ingwell and Bosque-Pérez 2014).
The extended phenotype of viruses can change based on many contexts, including the genotype of the host (vanMölken and Stuefer 2011), and biotic and abiotic conditions. These phenotypes vary from the classic disease symptoms to host benefitting-qualities such as drought or cold tolerance (Roossinck 2015b). The context of the plant hosts can effect the spread and diversity of plant viruses more than the composition of the plant host species. Competition between BYDV and CYDV was altered by changing the nutrition resources in the form of nitrogen and phosphorous for their hosts (Lacroix et al. 2014). In a B/CYDV survey in North American Pacific coast grasslands targeting three different host species (Avena fatua, Elymus glaucus, and Bromus hordeaceus) virus prevalence was determined not only by host species identity, with A. fatua having the highest prevalence and B. hordeaceus having the lowest, but also by biotic and abiotic factors, including an increase of virus prevalence with a decrease in precipitation and increase in soil nitrogen (Seabloom et al. 2010).
Many viruses that are found in wild plants have either mild symptoms or are completely asymptomatic (Prendeville et al. 2012; Jones 2014; Davis et al. 2015). Many wild plants host multiple viruses, in some cases up to seven different viruses were found co-existing in a single plant (Roossinck et al. 2010). In two studies in the United States and Costa Rica, over 50 % of the virus sequences found in wild plants belonged to three virus families: Partitiviridae, Chrysoviridae, and Totiviridae (Roossinck 2012b). These virus families, along with the Endornavirdae, have been described as persistent plant viruses (Roossinck 2010, 2015a). Most of the persistent plant viruses are double stranded RNA viruses, although the Endornaviridae are likely single-stranded RNA viruses that are isolated as replicative intermediates (Roossinck et al. 2011). Most interestingly, the persistent viruses are wholly transmitted vertically, with no known form of horizontal transmission. In fact, there is no cell-to-cell movement of persistent viruses, spreading throughout the host via cell division (Roossinck 2010, 2012a). The Partitiviridae, Endornaviridae and Chrysoviridae infect both plants and fungi, while the Totiviridae also infect fungi and protozoa. While there is no observable effect of these viruses on their hosts, there have been multiple instances of integration of some persistent viral genomes into plant and fungal genomes (Liu et al. 2010; Chiba et al. 2011). This should not be surprising given the intimate symbiotic nature of the relationship. Currently the persistent viruses are understudied and many aspects of their nature are unknown (Roossinck 2015a).
In a study looking at the effect of both CMV and ZYMV in wild populations of Cucurbita pepo, the context of the host population, either adjacent to a road, within a managed peanut field, or within an unmanaged pasture, seemed to be the dominate factor in whether ZYMV was detrimental, beneficial or neutral, respectively (Prendeville et al. 2014). While latent viruses are common in wild plants, there are of course pathogenic viruses found in the wild as well (Cooper and Jones 2006).
8.3.2 Difficulties of Virus Discovery
It is not surprising that with an in depth look into the viral biodiversity of plants one would find novel viruses related to known viruses, nor is it surprising that sequences with little or no similarity to anything in a curated database would be found. Often, even when there are related viruses within a database, the curation is not in a state to be useful. There are few centralized databanks to store metadata collected during large biodiversity surveys, though attempts have been made. Metavir, a website service offering basic analysis of viromes, allows for viromes to be made public, and at the time of writing houses 368 different viromes from 67 different projects (Roux et al. 2014). It is unclear how these potential viruses should be treated. In a recent virus survey in Costa Rica, 60 % of the sequence reads received no hit when searched against the GenBank database (Roossinck et al. 2010). For viruses with no known relative, a cluster analysis can give structure to a population of unknown microbes, including viruses (Labonté and Suttle 2013). While having a sequence identity for these viruses can offer some answers, the viruses ultimately need to be characterized experimentally.
8.4 Variation Within Virus Isolates
Evidence for genetic variation of plant viruses was reported as early as 1926 (Kunkel 1947). Numerous studies have looked at variation both within individual virus isolates and among isolates of the same virus species. Variation provides the basis for evolution of traits through natural selection, and has resulted in adaptation of plant viruses to new hosts, to new vectors, and to overcoming host resistance, including natural resistance, resistance introgressed through breeding, or genetically engineered resistance.
8.4.1 Quasispecies
The high levels of mutation generated by viral polymerases leads to high levels of variation within a single infection, known as a quasispecies. The term quasispecies refers to a single replicating population, and is an “individual” that selection acts upon (Holland and Domingo 1998). Do to the many different selective pressures an RNA virus experiences (different hosts, cell tropisms, vectors, etc.), a population that is more genetically robust, having a high degree of evolvability may have a selective advantage. This means in a fitness landscape, quasispecies that have a narrow fitness peak (less robust) experience a sharp decrease in fitness due to a single mutation, and are less likely to adapt rapidly to a new environment. Conversely, those with a wide fitness peak (more robust) will experience a small change in fitness, allowing for multiple mutations to accumulate for selection to act upon. This is known as the survival of the flattest (Wilke 2005).
It was been thought for some time that the lack of error correction within the RNA dependent RNA polymerase (RdRp) of RNA viruses is responsible for the size of the quasispecies (Steinhauer et al. 1992). However, in coronaviruses it is clear that error correction can occur (Denison et al. 2011). The quasispecies is effected by not only the mutation rate of the RdRp, but also by the mode of replication, logrithmic or stamping machine (Safari and Roossinck 2014). While double-stranded RNA viruses replicate predominantly by the stamping machine method, the mode of replication of other RNA viruses is not clear. Although the mutation rate of RNA viruses are high, the level of variation within the quasispecies may be lower than expected (García-Arenal et al. 2003). This is due to both positive and negative selection; however, defective genomes are often carried in the population and can provide extended function in some cases. While there are significant genetic bottleneck during systemic infection (Li and Roossinck 2004), as well as vector transmission (Ali and Roossinck 2010), viruses probably recover their diversity rapidly.
The size of the quasispecies, or level of variation of a virus within a host, is dependent on factors in both the virus and the host. When comparing three related Sindbis-like viruses, CMV, Tobacco mosaic virus (TMV), and Cowpea chlorotic mottle virus each had significantly different levels of variation within the same host background (Schneider and Roossinck 2000). In both TMV and CMV, the level of variation changed based on the host background (Schneider and Roossinck 2001). Different strains of CMV, Fny and LS, display different levels of diversity in tobacco and pepper plants, which maps to both the 1a and 2a proteins (Pita and Roossinck 2013b). By using a non-coding satellite RNA the indel fidelity of the CMV RdRp was analyzed in planta. While insertion mutations were rare, deletion mutations were more abundant and their rates differed based on the host background and the sequence context (Pita et al. 2007).
NGS is being used to identify minor variants within quasispecies. With a level of coverage of 2500x, the full range of variation can be uncovered (Simmons et al. 2012). This type of analysis can lead to answers to previously difficult questions of quasispecies dynamics in nature. Mutations within the ZYMV quasispecies were maintained through the aphid vector transmission, as well as seen throughout the plant, suggesting that the bottleneck of vector transmission and movement throughout the plant may be lower than previously thought (Simmons et al. 2015). There is evidence that some variants within a quasispecies are necessary for specific functions. Several ZYMV variants were found in different seed transmitted lines, suggesting that these variants have a role to play in seed transmission (Simmons et al. 2015).
Randomly generated point mutations in Tobacco etch virus (TEV) were used to determine the effect the mutations had on the virulence and fitness of TEV. The majority of the mutations were lethal, with the majority of non-lethal mutations leading to a significant reduction of fitness. (Carrasco et al. 2007). The lab strain of TEV is adapted to tobacco, but when TEV was adapted to pepper, virulence increased, but was found to decrease in the tobacco host, suggesting a tradeoff in becoming more specialized. No tradeoff was found for becoming more of a generalist (Bedhomme et al. 2012; Elena et al. 2008). Furthermore, pepper-adapted TEV acquires mutations that have a wide range of effects both positive and negative, implying pleiotropic effects. Interestingly, the fitness of mutants in the tobacco host does not predict the fitness in other non-native hosts (Lalic et al. 2011).
By passaging Plum pox virus (PPV; M strain) though several different host species for six years and analyzing the fixed mutations after host adaptation, it was found that peach yielded the lowest number of fixed mutations (two fold lower than other hosts). This suggests that PPV-M is highly adapted to peach (Vozárová et al. 2013). Passaging Pepino mosaic virus (Alphaflexiviridae) through several tomato cultivars with varying degrees of tolerance, convergently leads to isolates with higher pathogenicity. These passages also have an increase in the genetic diversity, with genetic diversity being a good indicator of pathogenicity (Minicka et al. 2015).
Previously it was thought that the high levels of variation within the begomoviruses (circular ssDNA) was due to high levels of recombination (Lima et al. 2013), but begomovirues have substitution rates much higher than other DNA viruses, on the order of 10−4 substitutions/site/year, in line with rates seen in RNA viruses (Duffy and Holmes 2008). Macroptilium yellow spot virus (MaYSV) and Tomato severe rugose virus (ToSRV), both begomoviruses, were analyzed for their variability. Interestingly, MaYSV, which primarily infects wild weeds, but does occasionally infect Phaseolus vulgaris (the common bean), had a greater diversity than ToSRV, which primarily infects tomato, and has a low incidence in wild plants. Several recombination events were detected for MaYSV, which drove the majority of the variability in the species.
8.4.2 Recombination
Recombination is not only an important part of population variation, but also can be used as a repair mechanism, balancing the high mutation rate of RNA viruses (Nagy and Simon 1997). Recombination is a frequent occurrence in CaMV, with over 50 % of isolates being recombinants after only 21 days of infection (Froissart et al. 2005). RNA 3 of bromoviruses may contain recombination hotspots (Bruyere et al. 2000). While recombination is an important part of increasing the variation of a species, recombination events that lead to hybrid proteins are most likely less fit than recombinants between whole genes or protein domains (Bonnet et al. 2005). Recombination offers a path for the adaptation of viruses to a new environment as seen with the introduction of TYLCV into Spain (García-Andrés et al. 2007). Interestingly, it appears the eukaryotic hosts may have adapted a method for modulating or regulating the degree of recombination of their infecting viruses. In a yeast model system modified to allow infection by Tomato bushy stunt virus, XRN1, a host exoribonuclease, was found to degrade 5′ truncated viral RNA. These truncated RNAs are substrates for recombination (Cheng et al. 2006). Recombination is commonly found in the ssDNA geminiviruses, both within and between different species (Padidam et al. 1999; Pagán and Holmes 2010). Citrus tristeza virus (CTV, Closteroviridae) is interesting in that many strains of the virus are commonly found within a single host plant. These strains have been phylogenetically analyzed to elucidate their evolutionary history, and it can be inferred that the current diversity of CTV was influenced by the original ancestral diversification, selection pressure of genes between and within strains, and significant recombination among strains (Harper 2013).
A number of studies have looked at recombination frequencies in experimental systems (Bujarski 2013; Sztuba-Solinska et al. 2011), especially in the Bromoviridae. In general, recombination frequencies are high in RNA viruses, and hot spots for recombination have been identified that result in exchanges between related RNA molecules, or in deletions leading to defective RNAs. In the cucumoviruses experimental infections with interspecific reassortants have frequently led to recombinants in RNA 3, where the 3′ end is exchanged with that of RNA 2, presumably to establish a minus strand promoter that is cognizant with the replicase (Aaziz and Tepfer 1999; deWispelaer et al. 2005; Pita and Roossinck 2013a). In a recent study different strains of CMV had very different frequencies of recombination. Interestingly, the high recombination strain was the same strain that had low mutation frequency, and the 2a protein that encodes the RdRp was responsible for both phenotypes (Pita et al. 2015).
8.5 Plant Virus Deep Evolution
The deep evolutionary history of viruses is a matter for considerable speculation. Since no virus fossils are available, studies have relied on comparisons of extant sequences; however, the recent explosion of complete genome information for plants and many other hosts has led to the development of a new field of virology known as paleovirology, which considers the viral sequences integrated into genomes as molecular fossils (Katzourakis 2012).
8.5.1 Origins of Plant Viruses
The majority of known plant viruses are RNA viruses. The origins of RNA viruses are thought come directly from the ancient RNA world, a time before cellular life, where RNA replicated itself without a DNA phase (Bernhardt 2012). It was proposed three decades ago that animal and plant viruses have a common ancestor, likely an insect virus (Goldbach 1986). Specific motifs in virus hallmark genes (genes which are unique to viruses and are found across many families of viruses) such as the RdRp were analyzed for similarity across a wide range of animal and plant RNA viruses, with positive, negative, and double-stranded genomes. Motifs in both the positive and double-stranded RNA viruses suggests that these groups are monophyletic, with the negative strand RNA viruses less likely to be monophyletic (Koonin et al. 2015). Virus hallmark genes are shared with other selfish genetic elements, such as plasmids and transposons, suggesting that viruses have a deep lineage, which some suggest dates to a pre-cellular time (Koonin and Dolja 2014). One can think of viruses as both an organism and a mobile genetic element, much as light can be thought of as both a particle and a wave. These entities evolve within their environment, and then move as genetic elements through higher organisms (Forterre and Prangishvili 2013). Indeed examples of viral elements being incorporated into their host’s genome can be found across all of the tree of life (Katzourakis 2012).
A high degree of recombination and/or reassortment of genetic material allows for modular evolution, where genes, protein motifs, or separate RNA molecules will evolve independently from each other. Within the luteoviruses, recombination is often seen near the gene borders of the RdRp and the coat protein (CP) genes but rarely within genes. This suggests that the genetic histories of these genes are independent of each other (Pagán and Holmes 2010). Phylogenetic analysis of CMV genes implies that each of the viruses three RNA segments have unique histories (Roossinck 2002). The extent of movement of genetic material across viruses can be seen with the recently named Amalgaviridae, a double-stranded RNA monopartite virus with 2 open reading frames, an RdRp and another gene. The RdRp most closely resembles that of another double-stranded RNA virus family, the Partitiviridae, while the other gene resembles that of the nucleoprotein of negative-stranded RNA viruses of the Bunyaviridae family (Krupovic et al. 2015). The iconic movement protein common and unique to plant viruses, may have been originally derived from the structural proteins used in the formation of the plasmodesmata (Lucas and Wolf 1993).
While there is a lot of movement of genetic material between viruses and their eukaryotic hosts, this movement is vastly biased towards viral genes being moved to their hosts; hence viruses have a major role in the evolutionary history of higher organisms (Forterre and Prangishvili 2013). It is extremely difficult to extract preserved viral nucleic acid from more than a few decades ago, though it is possible (Roossinck, unpublished results). Because of this difficulty, these rare integration events can be used to elucidate the life histories of the virus. Long before so many virus sequences were found in genomes, geminivirus sequences were found within the Nicotiana genome (Bejarano et al. 1996). Begomoviruses have been described as being Old world or New world, with several distinct qualities associated with each group. Most notably, the New world begomoviruses are monopartite while the Old world are bipartite. Using the intergration events within Nicotiana, an estimate of 20–30 MYA was found for the Old/New world split, suggesting that this virus crossed the Beringian land bridge (Lefeuvre et al. 2011). Cooperation of viruses in mixed infections may be the initial step towards multipartiate viruses. Two different monopartite viruses are known to cooperate, using each other’s proteins for their own function. If the relationship of the two viruses becomes too dependent, essential gene loss can occur in one or both viruses that may remain viable due to the complementary gene of the other virus. This will eventually lead to merging the two species into a single species (Shirogane et al. 2013).
8.5.2 Early Speciation
A recent phylogenetic analysis of the potyvirus genus suggests that the genus diverged approximately 7250 years ago, in monocots from the Southern Eurasia or Northern African regions (Gibbs et al. 2010). Some ZYMV lineages have been shown to be no older than 800 years old, suggesting that humans had a role to play in there movement and diversification (Simmons et al. 2008). Lueteoviridae diversification happened in three stages; The luteo/polerovirus split estimated at 2000 years ago, diversification of each genus estimated at 1000–5000 years ago, and the diversification of the species within the past 300 years (Pagán and Holmes 2010). The knowledge gained from extensive sampling including wild samples give us more insight into the life histories of viruses (Wylie et al. 2008). Potyviruses have a large amount of diversity within and between their species. Yam mosaic virus (YMV) is thought to have been originated in Africa. High levels of recombination are found within natural populations of YMV (Bousalem et al. 2000). The endogenization of Caulimoviruses distantly related to Rice tungro bacciliform virus into the rice genome have given us insight into the family’s evolutionary history. The edongenization events occured before the divergence of the domestic rice progenitor Oryza rufipogon, placing this event at about 160,000 years ago (Chen et al. 2014).
8.6 Conclusions
The diversity of plant viruses is still largely unknown, but what we have learned is that the virus diversity of agriculture systems is vastly different than that of natural ecosystems. While the majority of viruses infecting crops cause disease, it appears that viruses in natural areas are neutral or may provide some small benefit to their hosts. This paradigm shift opens the door to many future applications, as well as exciting implications to the field of virology as a whole. Understanding the mechanisms and consequences of movement across the agro-eco boundary, as well as an increased understanding of the mechanisms underlying virus evolution, may provide us with methods of predicting future epidemics, or attenuating the outbreak of new crop pathogens. The modern era of genomics is revealing new and exciting areas of research into virus evolution, and studies on the origins of viruses will likely lead to an understanding of the very origins of life on earth.
References
Aaziz R, Tepfer M (1999) Recombination between genomic RNAs of two cucumoviruses under conditions of minimal selection pressure. Virology 263:282–289
Ali A, Roossinck MJ (2010) Genetic bottlenecks during systemic movement of Cucumber mosaic virus vary in different host plants. Virology 404:279–283
Anderson PK, Cunningham AA, Patel NG, Morales FJ, Epstein PR, Daszak P (2004) Emerging infectious diseases of plants: pathogen pollution, climate change and agrotechnology. Trends Ecol Evol 19(10):535–544
Balter M (2007) Seeking agricultures ancient roots. Science 316:1830–1835
Bebber DP, Ramotowski MAT, Gurr SJ (2013) Crop pests and pathogens move polewards in a warming world. Nat Clim Chang 3:985–988
Bedhomme S, Lafforgue G, Elena SF (2012) Multihost experimental evolution of a plant RNA virus reveals local adaptation and host-specific mutations. Mol Biol Evol 29(5):1481–1492
Beijerinck MW (1898) Concerning a contagium vivum fluidum as cause of the spot disease of tobacco leaves. In: Johnson J (ed) Phylopathological classics, no. 7. American Phytopathological Society, St. Paul, pp 33–52
Bejarano ER, Khashoggi A, Witty M, Lichenstein C (1996) Integration of multiple repeats of geminiviral DNA into nuclear genome of tobacco during evolution. Proc Nat Acad Sci USA 93:759–764
Bernhardt HS (2012) The RNA world hypothesis: the worst hypothesis of early evolution of life (except for all the others). Biol Direct 7:23
Bonnet J, Fraile A, Sacristán S, Malpica JM, García-Arenal F (2005) Role of recombination in the evolution of natural populations of Cucumber mosaic virus, a tripartite RNA plant virus. Virology 332:359–368
Bousalem M, Douzery EJP, Fargette D (2000) High genetic diversity, distant phylogenetic relationships and intraspecies recombination events among natural populations of Yam mosaic virus: a contribution to understanding potyvirus evolution. J Gen Virol 81:243–255
Bruyere A, Wantroba M, Flasinski S, Dzianott A, Bujarski JJ (2000) Frequent homologous recombination events between molecules of one RNA component in a multipartite RNA virus. J Virol 74:4214–4219
Bujarski JJ (2013) Genetic recombination in plant-infecting messenger-sense RNA viruses: overview and research perspectives. Front Plant Sci 4:9. doi:10.3389/fpls.2013.00068
Carrasco P, Daròs JA, Agudelo-Romero P, Elena SF (2007) A real-time RT-PCR assay for quantifying the fitness of tobacco etch virus in competition experiments. J Virol Methods 139:181–188
Chen S, Liu R, Koyanagi KO, Kishima Y (2014) Rice genomes recorded ancient pararetrovirus activities: virus genealogy and multiple origins of endogenization during rice speciation. Virology 473:141–152
Cheng C-P, Serviene E, Nagy PD (2006) Suppression of viral RNA recombination by a host exoribonuclease. J Virol 80(6):2631–2640
Chiba S, Kondo H, Tani A, Saisho D, Sakamoto W, Kanematsu S, Suzuki N (2011) Widespread endogenization of genome sequences of non-retroviral RNA viruses into plant genomes. PLoS Pathog 7(7), e1002146
Cooper I, Jones RAC (2006) Wild plants and viruses: underinvestigated ecosystems. Adv Virus Res 67:1–47
Davis TS, Pérez NAB, Foote NE, Magney T, Eigenbrode SD (2015) Environmentally dependent host-pathogen and vector-pathogen interactions in the Barley yellow dwarf virus pathosystem. Journal of Applied Ecology 52:1392–1401
Denison MR, Graham RL, Donaldson EF, Eckerle LD, Baric RS (2011) An RNA proofreading machine regulates replication fidelity and diversity. RNA Biol 8(2):270–279
deWispelaer M, Gaubert S, Trouilloud S, Belin C, Tepfer M (2005) A map of the diversity of RNA3 recombinants appearing in plants infected with Cucumber mosaic virus and Tomato aspermy virus. Virology 331:117–127
Duffy S, Holmes EC (2008) Phylogenetic evidence for rapid rates of molecular evolution in the single-stranded DNA begomovirus Tomato yellow leaf curl virus. J Virol 82(2):957–965
Elena SF, Agudelo-Romero P, Carrasco P, Codoñer FM, Martín S, Torres-Barceló C, Sanjuán R (2008) Experimental evolution of plant RNA viruses. Heredity 100:478–483
Fargette D, Konaté G, Fauquet C, Muller E, Peterschmitt M, Thresh JM (2006) Molecular ecology and emergence of tropical plant viruses. Annu Rev Phytopathol 44:235–260
Fereres A (2015) Insect vectors as drivers of plant virus emergence. Curr Opin Virol 10:42–46
Forterre P, Prangishvili D (2013) The major role of viruses in cellular evolution: facts and hypotheses. Curr Opin Virol 3:558–565
Froissart R, Roze D, Uzest M, Galibert L, Blanc S, Michalakis Y (2005) Recombination every day: abundant recombination in a virus during a single multi-cellular host infection. PLoS Biol 3(3):389–395
García-Andrés S, Accotto GP, Navas-Castillo J, Moriones E (2007) Founder effect, plant host, and recombination shape the emergence populations of begomoviruses that cause tomato yellow leaf curl disease in the Mediterranean basis. Virology 359:302–312
García-Arenal F, Fraile A, Malpica J (2003) Variation and evolution of plant virus populations. Int Microbiol 6:225–232
Gibbs AJ, Fargette D, García-Arenal F, Gibbs MJ (2010) Time--the emerging dimension of plant virus studies. J Gen Virol 91:13–22
Goldbach RW (1986) Molecular evolution of plant RNA viruses. Ann Rev Phytopathol 24:289–310
Gómez P, Sempere RN, Elena SF, Aranda MA (2009) Mixed infections of Pepino mosaic virus strains modulate the evolutionary dynamics of this emergent virus. J Virol 83(23):12378–12387
Gutiérrez S, Michalakis Y, VanMunster M, Blanc S (2013) Plant feeding by insect vectors can affect ife cycle, population genetics and evolution of plant viruses. Funct Ecol 27:610–622
Harper SJ (2013) Citrus tristeza virus: evolution of complex and varied genotypic groups. Frontiers in Microbiology 4:article 93
Harrison BD (1981) Plant virus ecology: ingredients, interactions and environmental influences. Ann Appl Biol 99:195–209
Harrison BD (2002) Virus variation in relation to resistance-breaking in plants. Euphytica 124:181–192
Holland J, Domingo E (1998) Origin and evolution of viruses. Virus Gene 16:13–21
Houliston GJ, Fletcher JD, Heenan PB, Chapman HM (2015) Consequences of interspecies hybridization and virus infection on the growth and fecundity of three threatened coastal Lepidium (Brassicaceae) species from New Zealand. Aust Ecol
Ingwell LL, Bosque-Pérez NA (2014) The invasive weed Ventenata dubia is a host of Barley yellow dwarf virus with implications for an endangered grassland habitat. Weed Res 55:62–70
Ingwell LL, Eigenbrode SD, Bosque-Pérez NA (2012) Plant viruses alter insect behavior to enhance their spread. Sci Rep 2:578
Jones RAC (2014) Plant virus ecology and epidemiology: historical perspectives, recent progress and future prospects. Ann Appl Biol 164:320–347
Katzourakis A (2012) Paleovirology: inferring viral evolution from host genome sequence data. Philos Trans R Soc B 369:4
Keesing F, Belden LK, Daszak P, Dobson A, Harvell CD, Holt RD, Hudson P, Jolles A, Jones KE, Mitchell CE, Myers SS, Bogich T, Ostfeld RS (2010) Impacts of biodiversity on the emergence and transmission of infectious diseases. Nature 468:647–652
Koonin EV, Dolja VV (2014) Virus world as an evolutionary network of viruses and capsidless selfish elements. Microbiol Mol Biol Rev 78(2):278–303
Koonin EV, Dolja VV, Krupovic M (2015) Origins and evolution of viruses of eukaryotes: the ultimate modularity. Virology 479–480:2–25
Krupovic M, Dolja VV, Koonin EV (2015) Plant viruses of the Almagaviridae family evolved via recombination between virusese with double-stranded and negative-strand RNA genomes. Biol Direct 10:12. doi:10.1186/s13062-015-0047-8
Kunkel LO (1947) Variation in phytopathogenic viruses. Annual Rev Microb 1:85–100
Labonté JM, Suttle CA (2013) Previously unknown and highly divergent viruses populate the oceans. Int Soc Microbial Ecol J 7:2169–2177
Lacroix C, Seabloom EW, Borer ET (2014) Environmental nutrient supply alters prevalence and weakens competitive interactions among coinfecting viruses. New Phytol 204:424–433
Lalic J, Cuevas JM, Elena SF (2011) Effect of host species on the distribution of mutational fitness effects for an RNA virus. PLoS Genet 7(11), e1002378
Lecoq H, Wipf-Scheibel C, Nozeran K, Millot P, Desbiez C (2014) Comparative molecular epidemiology provides new insights into Zucchini yellow mosaic virus occurrence in France. Virus Res 186:135–143
Lefeuvre P, Harkins GW, Lett J-M, Briddon RW, Chase MW, Moury B, Martin DP (2011) Evolutionary time-scale of the begomoviruses: evidence from integrated sequences in the Nicotiana genome. PLoS One 6(5), e19193
Li H, Roossinck MJ (2004) Genetic bottlenecks reduce population variation in an experimental RNA virus population. J Virol 78(19):10582–10587
Lima ATM, Sobrinho RR, González-Aguilera J, Rocha CS, Silva SJC, Xavier CAD, Silva FN, Duffy S, Zervini FM (2013) Synonymous site variation due to recombination explains higher genetic variability in begomovirus populations infecting non-cultivated hosts. J Gen Virol 94:418–431
Liu H, Fu Y, Jiang D, Li G, Xie J, Cheng J, Pend Y, Ghabriel SA, Yi X (2010) Widespread horizontal gene transfer from double-stranded RNA viruses to eukaryotic nuclear genomes. J Virol 84(22):11879–11887
Lucas WJ, Wolf S (1993) Plasmodesmata: the intercellular organelles of green plants. Trends Cell Biol 3:308–315
Malmstrom CM, Hughes CC, Newton LA, Stoner CJ (2005) Virus infection in remnant native bunchgrasses from invaded California grasslands. New Phytol 168(1):217–230
Martinière A, Bak A, Marcia J-L, Lautredou N, Gargani D, Doumayrou J, Garzo E, Moreno A, Fereres A, Blanc S, Drucker M (2013) A virus responds instantly to the presence of the vector on the host and forms transmission morphs. eLIFE 2, e00183
Massart S, Olmos A, Jijakli H, Candresse T (2014) Current impact and future directions of high throughput sequencing in plant virus diagnostics. Virus Res 188:90–96
Mauck K, Bosque-Pérez NA, Eigenbrode SD, DeMoraes CM, Mescher MC (2012) Transmission mechanisms shape pathogen effects on host-vector interactions: evidence from plant viruses. Funct Ecol 26:1162–1175
Mauck KE, DeMoraes CM, Mescher MC (2015a) Infection of host plants by Cucumber mosaic virus increases the susceptibility of Myzus persicae aphis to the parasitoid Aphidius colemani. Sci Rep 5:10963
Mauck KE, Smyers E, DeMoraes CM, Mescher MC (2015b) Virus infection influences host plant interactions with non-vector herbivores and predators. Funct Ecol 29:662
McDonald BA, Linde C (2002) Pathogen population genetics, evolutionary potential, and durable resistance. Annu Rev Phytopathol 40:349–379
Minicka J, Rymelska N, Elena SF, Czerwoniec A, Hasiów-Jaroszewska B (2015) Molecular evolution of Pepino mosaic virus during long-term passaging in different hosts and its impact on virus evolution. Ann Appl Biol 166:389–401
Nagy PD, Simon AE (1997) New insights into the mechanisms of RNA recombination. Virology 235:1–9
Ng TFF, Duffy S, Polston JE, Bixby E, Vallad GE, Breitbart M (2011) Exploring the diversity of plant DNA viruses and their satellites using vector-enabled metagenomics on whiteflies. PLoS One 6(4):8
Padidam M, Sawyer S, Fauquet CM (1999) Possible emergence of new geminiviruses by frequent recombination. Virology 265:218–225
Pagán I, Holmes EC (2010) Long-term evolution of the Luteoviridae: time scale and mode of virus speciation. J Virol 84(12):6177–6187
Pagán I, González-Jara P, Moreno-Letelier A, Rodelo-Urrego M, Fraile A, Piñero D, García-Arenal F (2012) Effect of biodiversity changes in disease risk: exploring disease emergence in a plant-virus system. PLoS Pathog 8(7):12
Pita JS, Roossinck MJ (2013a) Fixation of emerging interviral recombinants in Cucumber mosaic virus populations. J Virol 87(2):1264–1269
Pita JS, Roossinck MJ (2013b) Mapping viral functional domains for genetic diversity in plants. J Virol 87(2):790–797
Pita JS, deMiranda JR, Schneider WL, Roossinck MJ (2007) Environment determines fidelity for an RNA virus replicase. J Virol 81(17):9072–9077
Pita JS, Morris V, Roossinck MJ (2015) Mutation and recombination frequencies reveal a biological contrast within strains of Cucumber mosaic virus. J Virol 89(13):6817–6823
Power AG (2008) Community ecology of plant viruses. In: Roossinck MJ (ed) Plant virus evolution. Springer, Heidelberg, pp 15–26
Power AG, Mitchell CE (2004) Pathogen spillover in disease epidemics. Am Nat 164(Suppl):S79–S89
Prendeville HR, Ye X, Morris TJ, Pilson D (2012) Virus infections in wild plant populations are both frequent and often unapparent. Am J Bot 99(6):1033–1042
Prendeville HR, Tenhumberg B, Pilson D (2014) Effects of virus on plant fecundity and populations dynamics. New Phytol 202:1346–1356
Ratnadass A, Fernandes P, Avelino J, Habib R (2012) Plant species diversity for sustainable management of pests and diseases in agroecosystems: a review. Agron Sustain Dev 32:273–303
Roossinck MJ (2002) Evolutionary history of Cucumber mosaic virus deduced by phylogenetic analyses. J Virol 76(7):3382–3387
Roossinck MJ (2010) Lifestyles of plant viruses. Philos Trans R Soc B 365:1899–1905
Roossinck MJ (2012a) Persistent plant viruses: molecular hitchhikers or epigenetic elements? In: Witzany G (ed) Viruses: essential agents of life. Springer, Dordrecht, pp 177–186
Roossinck MJ (2012b) Plant virus metagenomics: biodiversity and ecology. Ann Rev Genet 46:357–367
Roossinck MJ (2013) Plant virus ecology. PLoS Pathogens 9(5):e1003304
Roossinck MJ (2015a) Evolution of persistent viruses in plants. In: Weaver S, Denison M, Roossinck MJ, Vignuzzi M (eds) Virus evolution: current research and future directions. Caister Academic Press/Horizon Scientific Press, pp 261–270
Roossinck MJ (2015b) Plants, viruses and the environment: ecology and mutualism. Virology 479–480:271–277
Roossinck MJ, García-Arenal F (2015) Ecosystem simplification, biodiversity loss and plant virus emergence. Curr Opin Virol 10:56–62
Roossinck MJ, Saha P, Wiley GB, Quan J, White JD, Lai H, Chavarría F, Shen G, Roe BA (2010) Ecogenomics: using massively parallel pyrosequencing to understand virus ecology. Mol Ecol 19(S1):81–88
Roossinck MJ, Sabanadzovic S, Okada R, Valverde RA (2011) The remarkable evolutionary history of endornaviruses. J Gen Virol 92:2674–2678
Roossinck MJ, Martin DP, Roumagnac P (2015) Plant virus metagenomics: advances in virus discovery. Phytopathology 105:716–727
Roux S, Tournayre J, Mahul A, Debroas D, Enault F (2014) Metavir 2: new tools for viral metagnome comparison and assembled virome analysis. BMC Bioinform 15:76
Rúa MA, Pollina EC, Power AG, Mitchell CE (2011) The role of viruses in biological invasions: friend or foe? Curr Opin Virol 1(1):68–72
Sacristán S, Fraile A, García-Arenal F (2004) Population dynamics of Cucumber mosaic virus in melon crops and in weeds in central Spain. Phytopahtology 94:992–998
Safari M, Roossinck MJ (2014) How does the genome structure and lifestyle of a virus affect its population variation? Curr Opin Virol 9:39–44
Salvaudon L, DeMoraes CM, Mescher MC (2013) Outcomes of co-infection by two potyviruses: implications for the evolution of manipulative strategies. Proc R Soc B 280:20122959
Schneider WL, Roossinck MJ (2000) Evolutionarily related sindbis-like plant viruses maintain different levels of population diversity in a common host. J Virol 74(7):3130–3134
Schneider WL, Roossinck MJ (2001) Genetic diversity in RNA viral quasispecies is controlled by host-virus interactions. J Virol 75(14):6566–6571
Seabloom EW, Borer ET, Mitchell CE, Power AG (2010) Viral diversity and prevalence gradients in North American Pacific coast grasslands. Ecology 91(3):721–732
Sherwood RT (1997) Viruses of white clover in pastures of Pennsylvania, New York, and Vermont. Plant Dis 81:817–820
Shirogane Y, Watanabe S, Yanagi Y (2013) Cooperation: another mechanism of viral evolution. Trends Microbiol 21(7):320–324
Simmons HE, Holmes EC, Stephenson AG (2008) Rapid evolutionary dynamics of zucchini yellow mosaic virus. J Gen Virol 89:1081–1085
Simmons HE, Holmes EC, Stephenson AG (2011) Rapid turnover of intra-host genetic diversity in Zucchini yellow mosaic virus. Virus Res 155:389–396
Simmons HE, Dunham JP, Stack JC, Dickins BJA, Pagán I, Holmes EC, Stephenson AG (2012) Deep sequencing reveals persistence of intra- and inter-host genetic diversity in natural and greenhouse populations of zucchini yello mosaic virus. J Gen Virol 93:1831–1840
Simmons HE, Prendeville HR, Dunham JP, Ferrari MJ, Earnest JD, Pilson D, Munkvold GP, Holmes EC, Stephenson AG (2015) Transgenic virus-resistance in crop-wild Cucurbita pepo does not prevent transmission of Zucchini yellow mosaic virus. Plant Disease 99:1616–1621
Steinhauer DA, Domingo E, Holland JJ (1992) Lack of evidence for proofreading mechanisms associated with an RNA virus polymerase. Gene 122:281–288
Stobbe AH, Roossinck MJ (2014) Plant virus metagenomics: what we know and why we need to know more. Front Plant Sci 5:150. doi:10.3389/fpls.2014.00150
Stobbe AH, Daniels J, Espindola AS, Verma R, Melcher U, Ochoa-Corona F, Garzon C, Fletcher J, Schneider W (2013) E-probe diagnostic nucleic acid analysis (EDNA): a theoretical approach for handling of next generation sequencing data for diagnostics. J Microbiol Methods 94:356–366
Stukenbrock EH, McDonald BA (2008) The origins of plant pathogens in agro-ecosystems. Ann Rev Phytopathol 46:75–100
Sztuba-Solinska J, Urbanowicz A, Figlerowicz M, Bujarski JJ (2011) RNA-RNA recombination in plant virus replication and evolution. Annu Rev Phytopathol 49:415–443
Thresh JM (1981) The role of weeds and wild plants in the epidemiology of plant virus diseases. In: Thresh JM (ed) Pests, pathogens and vegetation. Pitman, London, pp 53–70
Thresh JM (1982) Cropping practices and virus spread. Ann Rev Phytopathol 20:193–218
Thresh JM (2006) Plant virus epidemiology: the concept of host genetic vulnerability. Adv Virus Res 67:89–125
Trebicki P, Nancarrow N, Cole E, Bosque-Pérez NA, Constable FE, Freeman AJ, Rodoni BL, Yen A, Luck JE, Fitzgerald GJ (2015) Virus disease in wheat is predicted to increase with changing climate. Glob Chang Biol 21:3511–3519
Valverde RA, Moreno R, Gamez R (1982) Incidence and some ecological aspects of Cowpea severe mosaic virus in two cropping systems in Costa Rica. Turrialba 32(1):29–32
VanEtten JL, Dunigan DD (2012) Chloroviruses: not your everyday plant virus. Trends Plant Sci 17(1):1–8
vanMölken T, Stuefer J (2011) The potential of plant viruses to promote genotypic diversity via genotype X environment interactions. Ann Bot 107:1391–1397
Vozárová Z, Kamencayová M, Glasa M, Subr Z (2013) Plum pox virus accumulates mutations in different genome parts during a long-term maintenance in Prunus host plants and passage in Nicotiana benthamiana. Acta Virol 57:369–372
Webster CG, Coutts BA, Jones RAC, Jones MGK, Wylie SJ (2007) Virus impact at the interface of an ancient ecosystem and a recent agroecosystem: studies on three legume-infecting potyviruses in the southwest Australian floristic region. Plant Path 56:729–742
Wilke CO (2005) Quasispecies theory in the context of populations genetics. BioMedCentral Evolut Biol 5:44
Wren JD, Roossinck MJ, Nelson RS, Sheets K, Palmer MW, Melcher U (2006) Plant virus biodiversity and ecology. PLoS Biol 4(e80):1–2
Wu Y, Davis TS, Eigenbrode SD (2014) Aphid behavioral responses to virus-infected plants are similar despite divergent fitness effects. Entolologia Experimentalis et Applicata 153:246–255
Wylie SJ, Couts BA, Jones MGK, Jones RAC (2008) Phylogenetic analysis of Bean yellow mosaic virus isolates from four continents: relationship between the seven groups found and their hosts and origins. Plant Dis 92(12):1596–1603
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Stobbe, A., Roossinck, M.J. (2016). Plant Virus Diversity and Evolution. In: Wang, A., Zhou, X. (eds) Current Research Topics in Plant Virology. Springer, Cham. https://doi.org/10.1007/978-3-319-32919-2_8
Download citation
DOI: https://doi.org/10.1007/978-3-319-32919-2_8
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-32917-8
Online ISBN: 978-3-319-32919-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)