Abstract
Ficus deltoidea is one of the widely used traditional herbal medicines in Malaysia. However, scientific data for this herb are very limited, particularly its phytochemical profile. The complexity of the phytochemical profile has hindered the development of this herb to wider application. Recent advancement of hybrid technology in analytical tools has eased the work of phytochemical profiling by simultaneous separation and identification using liquid chromatography integrated with photo diode array and tandem mass spectrometer (LC-PDA-MS/MS ). This hyphenated technique was successfully used in the present study. A wide range of phytochemicals including amino acids, organic acids, alkaloids, terpenoids, polyphenols, and their derivatives have been identified. The marker compound, isovitexin (apigenin) was identified at the retention time 17.5 min in the negative ion mode of mass spectrometry. Besides flavone (apigenin), this non-targeted mass screening detected isoflavones (genistein), flavanone (naringenin), flavonol (kaempferol), and flavanol (catechin) in the leaves extract of the plant. Several organic acids and phenolic acids which are very important for plant physiology and defense mechanism were also found in this study. The identification was carried out by matching the fragmentation pattern of the compound to the literature data. The presence of product ions which are the characteristic fragments for a particular compound was used for phytochemical identification. In other words, the fragmentation pattern of phytochemical is similar to the fingerprint of human beings.
Access provided by Autonomous University of Puebla. Download conference paper PDF
Similar content being viewed by others
Keywords
4.1 Introduction
The genus Ficus belongs to the Moraceae family which comprises about 750 species [1]. Most of the Ficus species can be found in the tropical countries as an evergreen shrub tree. According to Harrison [2], the rainforests of Borneo contain more than 160 Ficus species. The common species in Malaysia are Ficus deltoidea var. kunstleri, var. trengganuensis, and var. angustifolia [3]. The leaves of F. deltoidea have traditionally been used as herbal drink by people from South East Asia to treat numerous ailments such as diabetes, diarrhea, high blood pressure, skin infection, as well as to strengthen uterus of women after childbirth since ancient time [4, 5].
However, literature scientific data for this plant is very limited. Because of the wide application and usefulness of this herbal plant, many researchers have started their efforts to generate technical data for this herb, in terms of the pharmacological activities and its phytochemical profile recently. It was found that most researchers used methanolic extract of the plant in their studies. This is because the solvent system containing methanol would enhance the extraction of secondary metabolites from plant samples. Secondary metabolites are produced by plants through a number of defense mechanisms against both biotic and abiotic stresses for survival. In particular, flavonoids and terpenoids are considered as the major secondary metabolites attributed to the reported pharmacological properties.
Till date, few studies have been conducted on this herb, particularly on the pharmacological activities such as anti-nociceptivity [5], anti-inflammatory [6], and anti-diabetic [7, 8] by Malaysian researchers. The acute toxicity at the lethal dose of 50 % on Sprague Dawley rats was found to be greater than 5 g/kg [9]. There were also no significant adverse effects on the body weight, mortality, hematology, and histopathology of the rats from the subchronic toxicity. Furthermore, the toxicity effect was also evaluated based on the content of the characteristic phytochemicals in F. deltoidea, namely vitexin and isovitexin (C-glycosyl flavonoid). The result showed that there was no significant toxicity effect on normoglycemic mice and induced diabetic rats treated with the highest dose of 2 g vitexin and isovitexin per kilogram of body weight [10]. Another research team from France also investigated the volatile compounds, mostly from the chemical group of terpenoids and other oxygenated hydrocarbon compounds for 13–20 Ficus species [1, 11]. To the best of our knowledge, limited data are available for the phytochemicals in F. deltoidea. Therefore, the present study was carried out to identify the phytochemicals from the plant using a high sensitivity and mass accuracy hyphenated technique, LC-PDA-MS/MS .
4.2 Methodology
4.2.1 Plant Material and Chemicals
The plant materials of F. deltoidea Jack var. trengganuensis Corner used in this study were harvested from the garden of Institute of Bioproduct Development, Universiti Teknologi Malaysia (Johor bahru, Malaysia). The species was authenticated and deposited in the Universiti Kebangsaan Malaysia Herbarium (Bangi, Selangor, Malaysia) with voucher specimen 40213. HPLC grade of methanol and acetonitrile were purchased from Merck (Darmstadt, Germany). Nanopure water (18.2 MΩ cm) was produced from Barnstead NANOpure Diamond water purification system (Thermo, Waltham, MA).
4.2.2 Herbal Extraction
Fifty gram of dried and ground leaves were extracted with 650 ml of 50 % methanol in a reflux extraction system for 60 min. The crude extract solution was filtered and dried in vacuo for the determination of extraction yield (9.13 %w/w). Consequently, 1 g/ml of the crude extract was prepared for LC-PDA-MS/MS analysis.
4.2.3 LC-PDA-MS/MS
A capillary liquid chromatography (Dionex Corporation Ultimate 3000; Sunnyvale, CA) system was integrated with a diode array detector (Dionex Ultimate 3000) and a quadrupole and time-of-flight, QTOF mass spectrometer (AB SCIEX QSTAR Elite; Foster City, CA). A C18 reversed phase XSelect HSS T3 column (2.1 × 100 mm, 2.5 µm) with a flow rate of 150 µl/min was used for separation. Samples were scanned for a wide range of wavelengths from 200 to 500 nm. The binary gradient system consists of solvent A (water with 0.1 % formic acid) and solvent B (acetonitrile). The LC gradient was: 0–30 min, 2–30 % B; 30–40 min, 30–98 % B; 98 % B hold for 0.1 min and 40.1–60 min, 98–8 % B. The injection volume was 5 µl. All samples were filtered with 0.2-µm nylon membrane filter prior to injection.
The QTOF mass spectrometer was used for the small metabolite screening from m/z 100–1000. It was calibrated using 1 pmol of reserpine (m/z 609.2807) before use to ensure mass accuracy. A single Information Dependent Acquisition (IDA) method was created to acquire both TOF MS and three dependent runs of product ion scan with rolling collision energy. Nitrogen gas was used for nebulizing (40 psi) and as curtain gas (25 psi). Collision gas was set at 3, the accumulation time was 1 s for TOF MS and 2 s for each product ion scan. The voltage of ion spray was 5500 V for positive ion mode and −4500 V for negative ion mode. The declustering potential was 40 V and the focusing potential was set at 200 V.
4.3 Results and Discussion
The plant crude extract was separated by decreasing the polarity of mobile phases through a C18 column for compound elution. The most polar phytochemicals would be eluted first from the non-polar column, followed by less polar phytochemicals. Therefore, the phytochemicals would be eluted accordingly based on their polarity and scanned at a wide range of wavelengths by the photo diode array detector. The chromatograms at the four wavelengths with the most intense peaks such as 254, 282, 310, and 340 nm are presented in Fig. 4.1. Mostly, plant secondary metabolites could be detected at these wavelengths. The compounds which were eluted from the column consequently ionized at the ion source before fragmentation and detection by mass spectrometer.
Significantly, the number of peaks detected in the chromatograms (Fig. 4.1) was less than compounds ionized in mass analyzer as presented in the mass spectra (Fig. 4.2).
The lower number of compounds detected by the chromatographic method compared to the mass spectrometric technique can be explained by the weak absorbance of many compounds under UV and visible range of light. Therefore, this hybrid system appears to be more effective for phytochemical profiling compared to single technique of chromatographic and/or mass spectrometric approach. Moreover, the combination of quadrupole and time-of-flight technology in a mass analyzer is known to be a powerful analytical tool for unknown compound elucidation because of its high resolution and mass accuracy.
Based on the mass screening, a number of secondary metabolites were identified from both positive and negative ion modes. The identification was carried out by matching with the literature data. The compounds included amino acids, organic acids, alkaloids, terpenoids, flavonoids, and their glycosides (Table 4.1). The table shows that the majority of the phytochemicals , particularly organic acids, phenolic acids, flavonoids, and other hydrocarbons were detected at the negative ion mode. Several organic acids were detected in this study and they are intermediates in the plant carbon metabolism. Previous experimental evidence has proven organic acids as key components in various plant mechanisms to cope with nutrient deficiencies, metal tolerance and plant–microbe interactions [12]. The activation of the plant defense mechanism also produces phenolic acids and flavonoids as by-products from the phenylpropanoid pathway. These secondary metabolites are important to regulate plant growth hormones and to protect plants from ultraviolet radiation.
Flavonoids could be detected at both positive and negative ion modes depending upon the capability of the compound to be protonated or deprotonated under high voltage condition of ion source. Interestingly, isoflavones were detected at the positive ion mode, whereas the other classes of flavonoids such as flavone (apigenin), flavanone (naringenin), and flavonol (kaempferol) were detected at the negative ion mode. Isovitexin (apigenin-6-C-glycoside), which is the characteristic phytochemical in F. deltoidea, was detected in the negative ion modes of mass screening. According to Sanchez-Rabaneda et al. [13], isovitexin could be differentiated from its isomer, vitexin based on the presence of the only diagnostic ion, m/z 353 as the product ion in the negative ion. Kaempferol C-glycoside was also detected in the plant extract with its aglycone at m/z 285. Kaempferol could easily be differentiated from its counterpart, luteolin (flavone) which has similar aglycone ion at m/z 285, by the absence of neutral loss 42 in the flavonol [14].
In the positive ion mode, the C-glycosyl of isoflavone, namely genistein-8-C-glucoside (m/z 433) and its diglycoside (m/z 579) were detected in the plant extract. The fragmentation of this compound matched with the data reported by Rauter et al. [15] for 5,7,4′-trihydroxyisoflavone-8-C-glucoside (genistein-8-C-glucoside) which was found in the anti-hyperglycemic flavonoid extract of Genista tenera for diabetic treatment. This is because isoflavones could be aldose reductase inhibitors blocking the sorbitol pathway that is linked to many problems associated with diabetes [16]. In addition, genistein (isoflavone) was found to have anticancer activity [17] and estrogen-like activity against estrogen deficiency for women [18, 19]. The product ions of m/z 415, 397, and 379 were due to the loss of one, two and three water molecules, respectively from the parent ion, m/z 433. Besides that, the detection of product ions such as m/z 367, 337, and 295 was contributed by the further fragmentation of the protonated ions 2,3X+—loss of two water molecules, 0,4X+—loss of two water molecules and 0,2X+—loss of one water molecule, respectively from the sugar moiety. The cleavage of carbon bond 0,1X+ at the sugar moiety also produced m/z 283 in the spectrum. Previously, Li et al. [20] reported that flavonoid-C-glycosides appeared to be cleaved at the cross-ring of sugar moiety and caused loss of water molecules in the fragmentation. On the other hand, flavonoids-O-glycosides were likely to be fragmented at the glycosidic bond at low collision energy [21].
The flavanols of catechin and epicatechin are commonly found in tea. Interestingly, these compounds were also detected in the leaves extract of F. deltoidea. The detection of the characteristic ions for flavanol such as m/z 137 and 109 confirmed the presence of catechin and epicatechin in the plant extract [14]. The characteristic ions were found to result from the mechanism of ring contraction as previously reported by Fabre et al. [22]. Based on the findings of Dou et al. [23], epicatechin would elute after catechin using a C18 column in LC-MS/MS analysis. Since catechin is more polar than epicatechin stereogeometrically, the two negative ion peaks of m/z 289 with similar fragmentation pattern will be assigned as catechin and epicatechin at 11.59 and 16.2 min, respectively. However, only epicatechin (Rt 16.2 min) was detected at the positive ion mode. This might be due to the low concentration of catechin for protonation, which was approximately 10 times lower than epicatechin in the plant extract (data not shown).
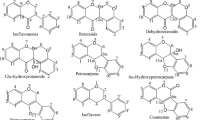
This study also detected metabolites resulting from the cleavage of seven-membered unsaturated ring C of a triterpenoid of duboscic acid (m/z 501) such as m/z 263, 245, 217, and 199 by the loss of formic acid and water molecule. Based on the observation of Musharraf et al. [24], the fragmentation at the seven-membered ring C became more prominent at high collision energy (35 eV) which was similar to the collision energy applied in this study. Another terpenoid detected in the positive ion mode was yuexiandujisu E or D which was also found in the roots of Euphorbia ebracteolata [25, 26]. On the other hand, genipinoside was detected at the negative ion mode.
4.4 Conclusions
LC-PDA-MS/MS is a fast and reliable hyphenated analytical technique for phytochemical profiling. Approximately, 50 phytochemicals have been identified from the plant extract of F. deltoidea in this study. The identified phytochemicals are from a broad range chemical classification ranging from amino acid, organic acid, polyphenol, alkaloid, and terpenoid. This high throughput identification is unlikely to be carried out using single chromatographic and/or spectrometric technique. The combination between good separation of chromatographic technique and excellent identification power of mass spectrometric technique appears to be suitable for highly complex samples including herbal plant materials.
References
Grison-Pige L, Hossaert-McKey M, Greeff JM, Bessiere JM (2002) Fig volatile compounds-a first comparative study. Phytochemistry 61:61–71
Harrison RD (2005) Figs and the diversity of tropical rainforests. Bioscience 55:1053–1064
Hakiman M, Syed MA, Ahmad S, Maziah M (2012) Flavonoid content in leaf extracts of different varieties of Ficus deltoidea Jack using high performance liquid chromatography (HPLC). In: UMT 11th International annual symposium on sustainability science and management, Terengganu, Malaysia, pp 470–473, 9–11 July 2012
Hakiman M, Maziah M (2009) Non-enzymatic and enzymatic antioxidant activities in aqueous extract of different Ficus deltoidea accessions. J Med Plants Res 3:120–131
Sulaiman MR, Hussain MK, Zakaria ZA, Somchit MN, Moin S, Mohamad AS, Israf DA (2008) Evaluation of the antinociceptive activity of Ficus deltoidea aqueous extract. Fitoterapia 79:557–561
Abdullah Z, Hussain K, Ismail Z, Ali RM (2009) Anti-inflammatory activity of standardized extracts of leaves of three varieties of Ficus deltoidea. Int J Pharm Clin Res 1:100–105
Adam Z, Khamis S, Ismail A, Hamid M (2012) Ficus deltoidea: a potential alternative medicine for diabetes mellitus. Evid-Based Complement Altern Med. Article ID 632763:12p
Farsi E, Shafaei A, Hor SY, Ahmad MBK, Yam MF, Attitalla IH, Asmawi MZ, Ismail Z (2011) Correlation between enzymes inhibitory effects and antioxidant activities of standardized fractions of methanolic extract obtained from Ficus deltoidea leaves. Afr J Biotechnol 10:15184–15194
Farsi E, Shafaei A, Hor SY, Ahmed MBK, Yam MF, Asmawi MZ, Ismail Z (2013) Genotoxicity and acute and subchronic toxicity studies of a standardized methanolic extract of Ficus deltoidea leaves. Clinics 68:865–875
Choo CY, Sulong NY, Wong TW (2012) Vitexin and isovitexin from the leaves of Ficus deltoidea with in-vivo α-glucosidase inhibition. J Ethnopharmacol 142:776–781
Grison L, Edwards AA, Hossaert-McKey M (1999) Interspecies variation in floral fragrances emitted by tropical Ficus species. Phytochemistry 52:1293–1299
Lopez-Bucio J, Nieto-Jacobo MF, Ramirez-Rodrıiguez V, Herrera-Estrella L (2000) Organic acid metabolism in plants: from adaptive physiology to transgenic varieties for cultivation in extreme soils. Plant Sci 160:1–13
Sanchez-Rabaneda F, Jauregui O, Casals I, Andres-Lacueva C, Izquierdo-Pulido M, Lamuela-Raventos RM (2003) Liquid chromatographic/electrospray ionization tandem mass spectrometric study of the phenolic composition of cocoa (Theobroma cacao). J Mass Spectrom 38:35–42
Gates PJ, Lopes NP (2012) Characterisation of flavonoid aglycones by negative ion chip-based nanospray tandem mass spectrometry. Int J Anal Chem. Article ID 259217:7p
Rauter AP, Martins A, Borges C, Ferreira J, Justino J, Bronze MR, Coelho AV, Choi YH, Verpoorte R (2005) Liquid chromatography-diode array detection-electrospray ionization mass spectrometry/nuclear magnetic resonance analyses of the anti-hyperglycemic flavonoid extract of Genista tenera: structure elucidation of a flavonoid-C-glycoside. J Chromatogr A 1089:59–64
Kawanishi K, Ueda H, Moriyasu M (2003) Aldose reductase inhibitors from the nature. Curr Med Chem 10:1353–1374
Ye F, Wu J, Dunn T, Yi J, Tong XD, Zhang D (2004) Inhibition of cyclooxygenase-2 activity in head and neck cancer cells by genistein. Cancer Lett 211:39–46
Harris RM, Wood DM, Bottomley L, Blagg S, Owen K, Hughes PJ, Waring RH, Kirk CJ (2004) Phytoestrogens are potent inhibitors of estrogen sulfation: implications for breast cancer risk and treatment. J Clin Endocrinol Metab 89:1779–1787
Vaya J, Tamir S (2004) The relation between the chemical structure of flavonoids and their estrogen-like activities. Curr Med Chem 11:1333–1343
Li QM, Den Heuvel V, Dillen L, Claeys M (1992) Differentiation of 6-C- and 8-C-glycosidic flavonoids by positive ion fast atom bombardment and tandem mass spectrometry. Biol Mass Spectrom 21:213–221
Cuyckens F, Claeys M (2004) Mass spectrometry in the structural analysis of flavonoids. J Mass Spectrom 39:1–15
Fabre N, Rustan I, de Hoffmann E, Quetin-Leclercq J (2001) Determination of flavone, flavonol, and flavanone aglycones by negative ion liquid chromatography electrospray ion trap mass spectrometry. J Am Soc Mass Spectrom 12:707–715
Dou J, Lee VS, Tzen JT, Lee MR (2007) Identification and comparison of phenolic compounds in the preparation of oolong tea manufactured by semi-fermentation and drying processes. J Agric Food Chem 55:7462–7468
Musharraf SG, Goher M, Wafo P, Kamdem RST (2012) Electrospray tandem mass spectrometric analysis of duboscic acid, exploring the structural features of a new class of triterpenoids, dubosane. Int J Mass Spectrom 310:77–80
Shi HM, Williams ID, Sung HH, Zhu HX, Ip NY, Min ZD (2005) Cytotoxic diterpenoids from the roots of Euphorbia ebracteolata. Planta Med 71:349–354
Wang WX, Ding XB (1998) Studies on diterpenoids from the roots of Euphorbia ebracteolata. Acta Pharmacol Sin 33:128–131
Gogichaeva NV, Williams T, Alterman MA (2007) MALDI TOF/TOF tandem mass spectrometry as a new tool for amino acid analysis. J Am Soc Mass Spectrom 18:279–284
Yamada K, Miyazaki T, Shibata T, Hara N, Tsuchiya M (2008) Simultaneous measurement of tryptophan and related compounds by liquid chromatography/electrospray ionization tandem mass spectrometry. J Chromatogr B 867:57–61
Allan RD, Duke RK, Hambley TW, Johnston GAR, Mewett KN, Quickert N, Tran HW (1996) Synthesis of unsaturated analogues of glutamic acid: amination of trianions from unsaturated dicarboxylic acids with chloramine. Aust J Chem 49:785–791
Wu ZJ, Ma XL, Fang DM, Qi HY, Ren WJ, Zhang GL (2009) Analysis of caffeic acid derivatives from Osmanthus yunnanensis using electrospray ionization quadrupole time-of-flight mass spectrometry. Eur J Mass Spectrom 15:415–429
Atoui AK, Mansouri A, Boskou G, Kefalas P (2005) Tea and herbal infusions: their antioxidant activity and phenolic profile. Food Chem 89:27–36
Shaw LH, Lin LC, Tsai TH (2012) HPLC–MS/MS analysis of a traditional Chinese medical formulation of Bu-Yang-Huan-Wu-Tang and its pharmacokinetics after oral administration to rats. PLoS ONE 7:e43848
Shaw LH, Chen WM, Tsai TH (2013) Identification of multiple ingredients for a traditional Chinese medicine preparation (Bu-yang-huan-wu-tang) by liquid chromatography coupled with tandem mass spectrometry. Molecules 18:11281–11298
Miao XS, Song P, Savage RE, Zhong C, Yang RY, Kizer D, Wu H, Volckova E, Ashwell MA, Supko JG, He X, Chan TCK (2008) Identification of the in vitro metabolites of 3,4-dihydro-2,2-dimethyl-2H-naphthol[1,2-β]pyran-5,6-dione (ARQ 501;β-Lapachone) in whole blood. Drug Metab Dispos 36:641–648
Mass Bank, High quality mass spectral database from Nara Institute of Science and Technology (NAIST), Japan. http://www.massbank.jp/jsp/Dispatcher.jsp?type=disp&id=BML01208&site=23
Pellati F, Orlandini G, Pinetti D, Benvenuti S (2011) HPLC-DAD and HPLC-ESI-MS/MS methods for metabolite profiling of propolis extracts. J Pharm Biomed Anal 55:934–948
Application Note 285 (2011) Dionex, Part of Thermo Fisher Scientific 1–6. http://www.thermoscientific.com/dionex
Rasche F, Scheubert K, Hufsky F, Zichner T, Kai M, Svatos A, Bocker S (2012) Identifying the unknowns by aligning fragmentation trees. Anal Chem 84:3417–3426
de la Cadiz-Gurrea ML, Fernandez-Arroyo S, Joven J, Segura-Carretero A (2013) Comprehensive characterization by UHPLC-ESI-Q-TOF-MS from an Eryngium bourgatii extract and their antioxidant and anti-inflammatory activities. Food Res Int 50:197–204
Wishart DS, Knox C, Guo AC, Eisner R, Young N, Gautam B, Hau DD, Psychogios N, Dong E, Bouatra S, Mandal R, Sinelnikov I, Xia J, Jia L, Cruz JA, Lim E, Sobsey CA, Shrivastava S, Huang P, Liu P, Fang L, Peng J, Fradette R, Cheng D, Tzur D, Clements M, Lewis A, De Souza A, Zuniga A, Dawe M, Xiong Y, Clive D, Greiner R, Nazyrova A, Shaykhutdinov R, Li L, Vogel HJ, Forsythe I (2009) HMDB: a knowledge base for the human metabolome. Nucleic Acids Res 37:D603–D610. http://www.hmdb.ca/spectra/spectra/ms_ms/873
Gouveia S, Castilho PC (2011) Characterisation of phenolic acid derivatives and flavonoids from different morphological parts of Helichrysum obconicum by a RP-HPLC–DAD–ESI-MSn method. Food Chem 129:333–344
Bystrom LM, Lewis BA, Brown DL, Rodriguez E, Obendorf RL (2008) Characterisation of phenolics by LC–UV/Vis, LC–MS/MS and sugars by GC in Melicoccus bijugatus Jacq. ‘Montgomery’ fruits. Food Chem 111:1017–1024
Schuhegger R, Nafisi M, Mansourova M, Petersen BL, Olsen CE, Svatos A, Halkier BA, Glawischnig E (2006) CYP71B15 (PAD3) catalyzes the final step in camalexin biosynthesis. Plant Physiol 141:1248–1254
Stalmach A, Mullen W, Barron D, Uchida K, Yokota T, Cavin C, Steiling H, Williamson G, Crozier A (2009) Metabolite profiling of hydroxycinnamate derivatives in plasma and urine after the ingestion of coffee by humans: Identification of biomarkers of coffee consumption. Drug Metab Dispos 37:1749–1758
Harbaum B, Hubbermann EM, Wolff C, Herges R, Zhu Z, Schwarz K (2007) Identification of flavonoids and hydroxycinnamic acids in pak choi varieties (Brassica campestris L. ssp. chinensis var. communis) by HPLC-ESI-MSn and NMR and their quantification by HPLC-DAD. J Agric Food Chem 55:8251–8260
Tala VR, Candida da Silva V, Rodrigues CM, Nkengfack AE, dos Santos LC, Vilegas W (2013) Characterization of proanthocyanidins from Parkia biglobosa (Jacq.) G. Don. (Fabaceae) by flow injection analysis-electrospray ionization ion trap tandem mass spectrometry and liquid chromatography/electrospray ionization mass spectrometry. Molecules 18:2803–2820
Perez-Magarino S, Revilla I, Gonzalez-San Jose ML, Beltran S (1999) Various applications of liquid chromatography–mass spectrometry to the analysis of phenolic compounds. J Chromatogr A 847:75–81
Sanchez-Rabaneda F, Jauregui O, Casals I, Andres-Lacueva C, Izquierdo-Pulido M, Lamuela-Raventos RM (2003) Liquid chromatographic/electrospray ionization tandem mass spectrometric study of the phenolic composition of cocoa (Theobroma cacao). J Mass Spectrom 38:35–42
Aslam SN, Stevenson PC, Phythian SJ, Veitch NC, Hall DR (2006) Synthesis of cicerfuran, an antifungal benzofuran, and some related analogues. Tetrahedron 62:4214–4226
Lavoie JC, Chessex P, Rouleau T, Migneault D, Comte B (2004) Light-induced byproducts of vitamin C in multivitamin solutions. Clin Chem 50:135–140
Ciss L, Tine A, Kabor L, Saba A (2009) Mass spectrometry study of coumarins: correlation between charges of atoms and fragmentation processes. Spectrosc Lett Int J Rapid Commun 42:95–99
Lecture notes University of California, Los Angeles. http://www.chemistry.ucla.edu/
Acknowledgments
The author would like to acknowledge the financial support of Exploratory Research Grant Scheme (ERGS 4L114) from the Ministry of Education, Malaysia. I also appreciate the help of Dr. Manjunatha Devagondanahalli Hadagali who is a post-doctoral researcher in the laboratory preparing the plant crude extract for analysis.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer International Publishing Switzerland
About this paper
Cite this paper
Chua, L.S. (2016). Hyphenated Technique of LC-PDA -MS/MS for Phytochemical Profiling of Ficus deltoidea . In: Ramasami, P., Gupta Bhowon, M., Jhaumeer Laulloo, S., Li Kam Wah, H. (eds) Crystallizing Ideas – The Role of Chemistry. Springer, Cham. https://doi.org/10.1007/978-3-319-31759-5_4
Download citation
DOI: https://doi.org/10.1007/978-3-319-31759-5_4
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-31758-8
Online ISBN: 978-3-319-31759-5
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)