Abstract
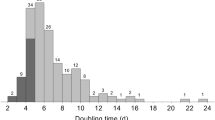
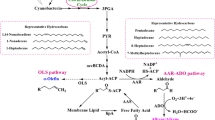
Hydrocarbons are detected in species of all algal phyla, but their contents are generally below 2% of algal dry weight skewed toward odd-carbon number, typically at C15, C17, or C21. Botryococcus braunii, a green colonial species (300–500 μm), contains exceptionally high hydrocarbons. Among the three races of B. braunii, race A contains C25–C31 n-alkadienes/trienes up to 61% dry weight and race B contains C31–C37 botryococcenes (triterpenes) up to 86% of dry weight. Race L contains lycopadienes (tetraterpene) C40H78 up to 8% dry weight. Cultures with 0.3% CO2-enriched air could shorten mass doubling time by 3.6 times. Nitrogen deficiency favors lipid accumulation, but nitrogen required for growth should be above 0.2 mg L−1. The optimal temperature for B. braunii is 20–25 °C with a light intensity of 60–100 Wm−2. Slow growth is the major hurdle retarding the production of hydrocarbon at a large scale. The combined approach of molecular biology, genetic engineering and ecology is recommended to escalate the algal growth and hydrocarbon production to yield a commercially competitive alternative for renewable biofuels from algae.
Similar content being viewed by others
References
Antia NJ, Lee RF, Nevenzel JC, Cheng JY (1974) Wax ester production by the marine cryptomonad Chroomonas salina grown photoheterotrophically on glycerol. J Protozool 21:768–771
Banerjee A, Sharma R, Chisti Y, Banerjee U (2002) Botryococcus braunii: a renewable source of hydrocarbons and other chemicals. Crit Rev Biotechnol 22:245–279
Ben-Amotz A, Torbene TG, Thomas WH (1985) Chemical profile of selected species of microalgae with emphasis on lipids. J Phycol 21:72–78
Blumer M, Mullin MM, Guillard RRL (1970) A polyunsaturated hydrocarbon (3, 6, 9, 12, 15, 18-heneicosahexaene) in the marine food web. Mar Biol 6:226–235
Blumer M, Guillard RRL, Chase T (1971) Hydrocarbons of marine phytoplankton. Mar Biol 8:183–189
Brown AC, Knights BA (1969) Hydrocarbon content and its relationship to physiological state in the green alga Botryococcus braunii. Phytochemistry 8:543–547
Cane RF (1969) Coorongite and the genesis of oil shale. Geochim Cosmochim Acta 33:569–577
Casadevall E, Largeau C, Metzger P, Chirac C, Berkaloff C, Coute A (1983) Hydrocarbon production by unicellular microalga Botryococcus braunii. Biosciences 2:129–138
Chisti Y (2007) Biodiesel from microalgae. Biotechnol Adv 25:294–306
Cox RE, Burlingame AI, Wilson DM, Eglinton GJ (1973) Botryococcene- a tetramethylated acyclic triterpenoid of algal origin. Chem Commun
Douglas AG, Eglinton G, Maxwell JR (1969) The hydrocarbons of coorongite. Geochim Cosmochim Acta 33:569–577
Drew KM, Ross R (1964) Some generic names in Bangiophycidae. Taxon 14:93–98
Fehler SWG, Light RJ (1970) Biosynthesis of hydrocarbons in Anabaena variabilis. Incorportion of [methyl- 14C]- and [methyl- 2H 3]-methionine. Biochemistry 9:418–428
Gelpi E, Schneider H, Mann J, Oro J (1970) Hydrocarbons of geochemical significance in microscopic algae. Phytochemistry 9:603–608
Gschwend PM, Macfarlane JK, Newman KA (1985) Volatile halogenated organic compounds released to seawater from temperate marine macroalgae. Science 227:1033–1035
Huang Z, Poulter CD (1989) Tetramethylsqualene, a triterpene from Botryococcus braunii var. Showa. Phytochemistry 28:1467–1470
Komárek J, Marvan P (1992) Morphological differences in natural populations of the genus Botryococcus (Chlorophyceae). Arch Protistenkd 141:65–100
Ladygina N, Dedyukhina EG, Vainshtein MB (2006) A review on microbial synthesis of hydrocarbons. Process Biochem 41:1001–1014
Largeau C, Casadevall E, Berkaloff C, Dhamliencourt P (1980) Sites of accumulation and composition of hydrocarbons in Botryococcus braunii. Phytochemistry 19:1043–1048
Lee RF, Loeblich AR (1971) Distribution of 21: 6 hydrocarbon and its relationship to 22: 6 fatty acid in algae. Phytochemistry 10:593–598
Li Y, Qin JG (2005) Comparison of growth and lipid content in three Botryococcus braunii strains. J Appl Physiol 17:551–556
Lupi FM, Fernandes HML, Tomme MM, Sa Correia I, Novais JM (1994) Influence of nitrogen source and photoperiod on exopolysaccharide synthesis by the microalga Botryococcus braunii. Enzym Microb Technol 6:546–558
Maxwell JR, Douglas AG, Eglinton G, McCormick A (1968) The Botryococcenes-hydrocarbons of novel structure from the alga Botryococcus braunii, Kützing. Phytochemistry 7:2157–2171
McKirdy DM, Cox RE, Volkman JK, Howell VJ (1986) Botryococcane in a new class of Australian non-marine crude oils. Nature 320:57–59
McMurry J (2000) Organic chemistry. Brooks/Cole, Pacific Grove
Metzger P, Casadevall E (1987) Lycopadiene, a tetraterpenoid hydrocarbon from new strains of the green-alga Botryococcus braunii. Tetrahedron Lett 28:3931–3934
Metzger P, Largeau C (1999) Chemicals of Botryococcus braunii. In: Cohen Z (ed) Chemicals from microalgae. Taylor & Francis, London, pp 205–260
Metzger P, Largeau C (2005) Botryococcus braunii: a rich source for hydrocarbons and related ether lipids. Appl Microbiol Biotechnol 66:486–496
Metzger P, Berkaloff C, Couté A, Casadevall E (1985) Alkadieneand botryococcene-producing races of wild strains of Botryococcus braunii. Phytochemistry 24:2305–2312
Metzger P, Casadevall E, Coute A (1988) Botryococcene distribution in strains of green alga Botryococcus braunii. Phytochemistry 27:1383–1988
Metzger P, Allard B, Casadevall E, Berkaloff C, Coute A (1990) Structure and chemistry of a new chemical race of Botryococcus braunii (Chlorophyceae) that produces lycopadiene, a tetraterpenoid hydrocarbon. J Phycol 26:258–266
Metzger P, Villarrealrosales E, Casadevall E (1991) Methyl-branched fatty aldehydes and fatty-acids in Botryococcus braunii. Phytochemistry 30:185–191
Nevenzel JC (1989) Biogenic hydrocarbons of marine organisms. In: Ackman RG (ed) Marine biogenic lipids, fats, and oils. CRC Press, Boca Baton, pp 3–71
Nishimoto S (1974) Chemotaxonomic study of n-alkanes in aquatic plants. J Sci Hiroshima Univ Ser A Phys Chem 38:159–168
Okada S, Devarenne TP, Chappell J (2000) Molecular characterization of squalene synthase from the green microalga Botryococcus braunii, race B. Arch Biochem Biophys 373:307–317
Patterson GW (1967) The effect of culture conditions on the hydrocarbon content of Chlorella vulgaris. J Phycol 3:22–28
Perry GJ, Gillan FT, Johns RB (1978) Lipid composition of a prochlorophyte. J Phycol 14:369–371
Qin JG (2005) Bio-hydrocarbons from algae: impacts of temperature, light and salinity on algae growth. Rural Industries Research and Development Corporation
Qin JG, Li Y (2006) Optimization of the growth environment of Botryococcus braunii strain CHN 357. J Freshw Ecol 21:169–176
Rezanka T, Zahradnik J, Podojil M (1977) Hydrocarbons in green and blue-green algae. Folia Microbiol (Prague) 27:450–454
Senousy HH, Beakes GW, Hack E (2004) Phylogenetic placement of Botryococcus braunii (Trebouxiophyceae) and Botryococcus sudeticus isolate UTEX 2629 (Chlorophyceae). J Phycol 40:412–423
Smith GM (1950) The fresh-water algae of the United States. McGraw-Hill, New York
Wake LV, Hillen LW (1981) Nature and hydrocarbon content of blooms of the alga Botryococcus braunii occurring in Australian freshwater lakes. Aust J Mar Freshwat Res 32:353–367
Wolf FR, Nanomura AM, Bassham JA (1985) Growth and branched hydrocarbon production in a strain of Botryococcus braunii. J Phycol 21:388–398
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer International Publishing AG
About this entry
Cite this entry
Qin, J.G. (2016). Hydrocarbons from Algae. In: Lee, S. (eds) Consequences of Microbial Interactions with Hydrocarbons, Oils, and Lipids: Production of Fuels and Chemicals. Handbook of Hydrocarbon and Lipid Microbiology . Springer, Cham. https://doi.org/10.1007/978-3-319-31421-1_209-1
Download citation
DOI: https://doi.org/10.1007/978-3-319-31421-1_209-1
Received:
Accepted:
Published:
Publisher Name: Springer, Cham
Online ISBN: 978-3-319-31421-1
eBook Packages: Springer Reference Biomedicine and Life SciencesReference Module Biomedical and Life Sciences