Abstract
Active role of food in human health is well documented. In past decade, there is increasing awareness about functional foods and their beneficial effects on human health. Among the functional foods, nondairy preparations have increased demand due to milk cholesterol content and lactose intolerance in human individuals. The microorganisms with probiotic potential significantly add to the nutritional value of various nondairy products. Commercial use of their functionality requires better understanding of their characteristics and their ability to tolerate and survive different techniques of food processing, packaging, and storage. In order to conserve their favorable effects and to develop user-friendly functional foods, with commercially useful shelf life, new technologies are being introduced and/or developed. The present review encompasses probiotics in nondairy functional foods and effects on food qualities. The technological innovations in the food processing and packaging along with recent techniques of storage and preservation that are devoid of any adverse effects on probiotic potential of microorganisms are also discussed.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
1 Introduction
Nowadays concern about one’s health and well-being is growing due to realization of importance of co-relation between diet, nutrition, and healthy lifestyle. Consumers demand for safe and varied food products which will ensure longevity and reduce risk of diseases. This is mainly due to increased incidences of obesity and overweight in men and women, chronic and non-communicable diseases, and mental health problems like depression, poor memory, and loss of memory. Majority of them are the result of increased urbanization, lack of physical exercise, and inclusion of high calorie foods in diet. Hence, foods which play significant role in various disorders or diseases are gaining importance. This has led to commercialization of functional foods. These are the foods that positively affect health and can be defined as foods containing significant levels of biologically active components that provide specific health benefits beyond the traditional nutrients they contain (Drozen and Harrison 1998). Such bioactive components include probiotics, antioxidants, omega-3-fatty acids, or synthetic food ingredients like prebiotics, vitamins, minerals, amino acids, proteins, etc. Another important reason for increasing interest in functional foods is the increase in healthcare cost.
Functional food may help to prevent or reduce risk of developing diseases and enhance human health. Noteworthy benefits of functional foods are in reducing risk of cardiovascular disease, cancer, and osteoporosis. Also, they play an important role in the improvement in general health and mental health. Thus, they have both protective and remedial effects (Stanton et al. 2001). Majority of the immunomodulatory effects of functional foods are due to probiotic microorganisms conventionally carried through milk-based products like yogurt, curd, cheese, etc. However, nowadays there is growing interest in non-dairy functional foods fortified with probiotics although traditionally they have been consumed in various countries for a long time. This chapter focuses on various non-dairy probiotic foods, their nutritional value, technological aspects, and challenges in developing such food types.
2 Global Market
The world market of functional foods is predicted to reach $130 billion after 2015 (http://www.reportlinker.com/ci02036/Functional-Food.html). Among the functional foods most important are probiotic yogurts, plant sterol spreads, functional waters, juices, deserts, and cheeses (Granato et al. 2010; Stanton et al. 2001). Major factors affecting their market potential are government support, consumer demand, consumer confidence in products, and health awareness. Functional foods have to compete with organic foods and foods with low fat, low sugar, and low salt labels. Thus, the prerequisites to increase their market are communication of their health benefits in simple language, good taste, convenience, and affordable price. In addition, brand name, loyalty, advertising and promotion, quality control, competitors, and economics are also important (Euromonitor 2009). In Europe, huge market exists for pro-, pre-, and synbiotics (Bhadoria and Mahapatra 2011).
3 Probiotics
3.1 Health Benefits
Probiotics are defined as “live microorganisms which when administered in adequate amount confer health benefits on the host by improving the properties of indigenous microflora” (Tabbers and Benninga 2007). Earlier research has shown that certain strains of probiotic bacteria have many health benefits. These microorganisms
-
(a)
enhance immune response by (1) improving innate and acquired immunity, (2) changing cytokine profiles, and (3) increasing levels of immunoglobulins.
-
(b)
reduce severity of constipation and improve bowel moment frequency (Ouwehand et al. 2003)
-
(c)
control urogenital infections in women (Dani et al. 2002)
-
(d)
inhibit effect on Helicobacter pylori (Hamilton-Miller et al. 2003)
-
(e)
reduce the risk of bladder cancer (Rafter 2004)
-
(f)
decrease LDL cholesterol levels (Pereira and Gibson 2002)
-
(g)
prevent fungal outgrowth and allergic reactions
-
(h)
produce vital nutrients like vitamin K and act as antioxidants (Crittenden et al. 2005; Anonymous 2010).
3.2 Lactobacilli and Bifidobacteria
The probiotic bacteria used today mainly belong to the genera Lactobacillus and Bifidobacterium. Commonly used strains are L. acidophilus, L. casei, L. rhamnosus, L. johnsonii, etc. Bifidobacteria strains include B. bifidum, B. longum, B. infantis, etc. (Reuter 1997; Holzapfel et al. 1997; Huis in’t Veld and Havenaar 1997; Bonaparte and Reuter 1997). Both lactobacilli and bifidobacteria are important microorganisms in the gastrointestinal tract (GIT) and urogenital tract of humans and higher animals (Sgorbati et al. 1995). Many different environmental factors like pH, O2 availability, specific substrates, and bacterial interactions affect distribution of lactobacilli, while age and diet are the main deciding factors for bifidobacteria. The latter are predominantly present in infants; however, with increasing age their number decreases (Finegold et al. 1983). Thus, they belong to the category of generally regarded as safe (GRAS) microorganisms and act as health promoters.
3.3 Strain Selection
The probiotic strain selection for food application depends on their technologic properties, besides the health benefits. The main selection criteria include (1) their survival during transit through stomach and small intestine, (2) adhesion to human GIT, (3) tolerance to oxygen, acid, bile, and salt, and (4) genetic stability (Karovicova et al. 1994, 1999; Holzapfel 2002; Aukrust et al. 1994; Ausco et al. 1998). Moreover, they should produce final product with good taste and acceptable texture, be produced on a large scale, be nonpathogenic and nontoxic, and be safe for technological uses (Adams and Marteau 1995; Donohue and Salminen 1996). Also, changes in food due to microbial metabolism should not affect their stability and functional properties. In addition, their survival throughout manufacturing process, storage, and distribution also must be regularly controlled and monitored. In order to achieve maximum benefits, the selected strain of microorganisms must be present in high numbers, i.e., 109 cells/daily ingested dose. Moreover, the minimum dose must be indicated on the product to confer specific health benefits (Guarner and Schaafsma 1998). Thus, their functional and technological properties are equally important. Nevertheless, food production process affects properties of probiotics, indicating importance of their interactions with other microorganisms as well as with food components. The latter depends on the time when probiotics are added to it, their physiological state, and treatment of probiotics during and after harvesting (Ross et al. 2005).
Synergism of probiotics with other food microorganisms normally results in increased acidification and increase in the number of organisms (Driessen et al. 1982; Radke-Mitchell and Sadine 1986; Perez et al. 1991; Zourari et al. 1992), while inhibition of other microorganisms results due to (1) competition for available nutrients, (2) decrease in redox potential, (3) organic acid production, (4) decrease in pH, and (5) production of bacteriocins, H2O2, biogenic amines, benzoic acid, etc. Their ability to produce bacteriocins helps to extend shelf life and safety of the product (Kalantzopoulos 1997). These biopreservatives can be destroyed by digestive enzymes which is an advantage over classical antibiotics and chemical preservatives. Such strains can be used along with starter cultures to improve the quality of food (Caplice and Fitzerland 1999). However, their antagonistic activity may hinder the development of probiotic food with starters (Joseph et al. 1998). Thus, in order to produce marketable probiotic products, the most important prerequisites are (1) survival of microorganisms in sufficient number in the product, (2) their physical and genetic stability during storage of the product, and (3) expression of their beneficial health effects after consumption. Table 1 enlist commercial probiotic strains sold by different companies in the world.
4 Preference for Non-dairy Probiotic Food
Usually, health benefits of probiotics are achieved and maintained by consumption of milk-based products or dairy products. However, lactose intolerance, cholesterol content, and allergenic milk proteins are the major limitations of consuming milk-based products (Yoon et al. 2006). Almost 75 % of world population is lactose intolerant (http://www.pcrm.org/health/diets/vegdiets/what-is-lactose-intolerance). It is mainly due to deficiency of one or more enzymes required for lactose digestion. Unfortunately, there is no treatment to improve the enzyme levels. Hence, the symptoms have to be treated by changing the diet containing alternatives to dairy products (Schaafsma 2008). The starter culture in yogurt and cheeses can lessen lactose intolerance in those individuals consuming probiotics through dairy products. These microorganisms produce β-galactosidase in small intestine which assists in lactose digestion (Li et al. 2012). But the effectiveness depends on certain factors like number of cells in the product and amount of lactose produced. In case of milk-sensitive individuals, lactose indigestion results in bloating, cramping, and flatulence that affects the quality of life. Besides, these people are deprived of other health benefits resulting from consumption of probiotics. This emphasizes the need to develop non-dairy products with probiotic benefits. Additionally, worldwide trend of vegetarian diet and traditional and economic reasons in developing countries support the concept of using the substrates other than milk to deliver probiotics.
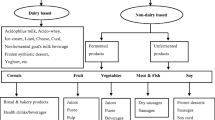
5 Traditional Non-dairy Probiotic Foods
As mentioned above, lactose intolerance and cholesterol content are the two major drawbacks associated with probiotic dairy products (Yoon et al. 2006). Therefore, traditional non-dairy fermented foods are being examined for their nutritional value as well as analyzed microbiologically. Current research is also focused on developing new or innovative non-dairy probiotic products, their health-promoting effects, sensory qualities, and shelf life. Commercialization of research efforts has led to the manufacture of some of these products on industrial scale. The following section discusses traditional as well as new products and challenges in product development.
5.1 Cereals and Legumes
The traditional cereal-based beverages have been commonly consumed in various countries for a long time mainly because cereal grains are nutritionally important due to their content of proteins, carbohydrates, vitamins, minerals, water-soluble fibers, and oligosaccharides (which can act as prebiotics). However, they have only been recently studied for their health benefits. Their microbiological analyses revealed the presence of probiotic microorganisms in these products. Table 2 summarizes traditional probiotic beverages and foods based on cereals and pulses, the probiotic strains identified in them, and their preparation procedure. Besides, wheat, rye, millet, sorghum, and oats have also been used as substrates for fermentation (Angelov et al. 2006). Another widely accepted substrate is soybean. Fermented soymilk and yogurt are popular alternatives to dairy products (Fuchs et al. 2005). Fermentation by probiotic bacteria also helps in preservation of food such as rice wine/beer, rice cakes, fish (Bonaparte and Reuter 1997; Shah et al. 1995), etc., by producing organic acids which control spoilage microorganisms and pathogens as discussed in the previous section. In addition, they produce desirable flavors and improve nutritional value during fermentation of product.
5.2 Fruits and Vegetables
Other than cereals, fruits and vegetables or their juices are promising substrates to produce non-dairy probiotic foods. Fruit juices fortified with probiotics and prebiotics are gaining importance as fruits have additional advantage of being tasty, healthy, and refreshing and are rich in vitamins, mineral, fibers, and antioxidants (Luckow and Delahunty 2004). Traditionally consumed fruit-based products are Yan-Taozih (pickled peaches), Pobuzihi (fermented cummingcordia), etc. (Table 3). In Turkey, the popular traditional fermented beverage is hardaliye based on grape juice. It also contains mustard seeds which add to the flavor of product. Studies on hardaliye by Arici and Coskun (2001) indicated different strains of lactobacilli in this beverage viz. L. casei, L. paracasei, L. brevis, etc. (Table 3). Oranges, pineapples, grapes, and cranberry are also commonly used substrates. Studies on pineapple and cranberry probiotic juices were published by Sheehan et al. (2007). The authors have reported better survival of lactobacillus strains in orange and pineapple as compared to cranberry. However, the organisms were unable to withstand pasteurization (76 °C for 30 s) and high pressure treatment required for preservation.
Traditional vegetable-based fermented foods are Kimchi, Saurekraut Soidon, Gundruk, Dakguadong, etc. (Table 3). Mostly, the strains used are L. casei, L. acidophilus, L. plantarum, and L. delbrueckii. Table 4 summarizes recent studies carried out to produce probiotic non-dairy functional foods using vegetables and fruits as raw material.
6 Commercial Products
The technological advances are moving ahead to manufacture traditionally prepared beverages/foods by industrial processes. However, such foods differ in their sensory qualities as compared to the traditional ones, e.g., conventional orange juice is preferred by consumers over its probiotic-fortified counterpart due to its sensory properties; traditional rice wine has a deep and bounty flavor, while industrial wine has light simple flavor (Henneberg 1926). This difference may be attributed to a number of acid-forming bacteria, types of acids, and other metabolites produced. In spite of this, the advantages of probiotics and vegan diets are becoming more appealing. Hence, new plant-based probiotic products are coming in the market, e.g., grainfields whole grain liquid containing beans, oats, maize, rice, alfalfa seed, pearl, barley, linseed, mung beans, wheat, rye grains, and millet. It is fermented with lactobacilli and yeast (Saarela et al. 2006). Other non-dairy probiotic products include vita Biosa, Proviva, Malted barley, Gefilus fruit drinks, etc. Table 5 describes commercially available non-dairy probiotic products and their manufacturers. Majority of their health claims include improvement in digestive system and immune response. Also, they are claimed to be safe for everyday use.
7 Sensory Qualities
Most of the probiotics containing non-dairy fermented foods prepared from cereals, fruits, or vegetables usually have favorable texture, flavor, and aroma, e.g., salty taste, fresh carbonated sensation, and crispy texture of kimchi (Dodd and Gasson 1994). Its optimum taste and high vitamin C content of the latter are attained when pH decreases (4–4.5) due to acid fermentation (Metchnikoff 1908). Normally, fermentation results in pleasant acid taste and characteristic aroma due to esters produced from organic acids and alcohols which are produced from sugars (e.g., fructose) (Biacs 1986). In acid-fermented vegetables, a rapid decrease in pH at the beginning of fermentation is of great importance for the quality of end product (Viander et al. 2003). In addition, CO2 produced in acid-fermented vegetables by Leuconostoc mesenteroides inhibits growth of unwanted microorganisms, preventing undesirable softening of vegetables. Also, the anaerobic conditions produced by CO2 help in stabilization of ascorbic acid and natural colors of vegetables (Metchnikoff 1908). Moreover, exopolysaccharides produced by LAB also help to improve rheological and textural properties of food products (Leroy et al. 2002).
Fruit juice-based functional beverages fortified with probiotic microorganisms are dairy free, soy free, wheat free, and vegan. Hence, they are preferred by a large portion of the population. However, it is important to get rid of the off flavors in fermented fruit juices and enhance their sensory acceptability (Granato et al. 2010). For this purpose, addition of fruit juices like pineapple, mango, or passion fruit was found to be useful to improve the aroma and flavor of the final product masking the probiotic off flavors (Luckow and Delahunty 2004). On the other hand, undesirable aromas/flavors might gain consumers’ assertion as indicators of probiotic ingredients or their action (Juttlestad 1998). Alternative to adding probiotic microorganisms directly to the foods is by using them in previously immobilized form which is discussed in the following section. Iron-fortified soy yogurt is found to have suitable hedonic scores for creaminess and flavor. It is also observed that selection of probiotic strains for fermentation purpose affects sensory qualities of soy beverages, e.g., fermented soy beverages produced by using bifidobacteria have better acceptability as compared to that of L. casei. Besides, addition of prebiotics like oligofructose and inulin to soy yogurt was found to increase acceptance index more than 70 % (Hauly et al. 2005). Thus, elevating the palatability of probiotic products is one way to make them more appealing to the consumers, while another means can be making people aware of probiotic health benefits of the product.
8 Additional Health Benefits of Non-dairy Probiotic Functional Foods
8.1 Increase in Nutritional Quality
The nutritional value of a particular food depends on its digestibility and its content of essential nutrients. Both are improved by fermentation, since fermentation increases its nutrient density, the amount, and bioavailability of nutrients. The latter may be achieved by degradation of antinutritional factors, by predigestion of certain food components, and by improving the absorption and uptake of nutrients by the mucosa (Svanberg and Lorri 1997). In addition, fermentation results in the (1) increase in protein solubility and availability of scarce amino acids by as much as 50 %, (2) increase in micronutrient availability because of reduction of phytates and reduction in tannins by as much as 50 %, and (3) decrease in oligosaccharides by as much as 90 % (Nout and Ngoddy 1997) concentration and increase in the availability of proteins and vitamins like thiamine, folic acid, riboflavin, etc. Hence, such types of foods have direct curative effects on consumers of such foods (Steinkraus 1997). LAB also help in increase in iron uptake (Venkatesh 1998). Besides, fermentation may reduce the content of non-digestible plant foods like cellulose, hemicelluloses, polygalacturonic, and glucuronic acids. Breakdown of these compounds may lead to increase in bioavailability of minerals and trace elements (Kalantzopoulos 1997).
8.2 Soy-Based Products
Soy-based products flavored with fruit juices have become very popular as alternatives to dairy products (Champagne et al. 2005). Global market for soy-based probiotic yogurts is increasing annually by 10 %. Various health benefits of such products include (1) weight management, (2) decrease in risk of heart diseases and some cancers (Larkin et al. 2007), (3) good protein source, (4) help in immunomodulation, reduction in levels of –CHO causing gas production in intestine, (5) increase in isoflavone levels, and (6) beneficial to bone health. Besides, soy is in itself a source of vitamins and minerals, antioxidants, and some isoflavones. Hence, traditional soy-based products, e.g., miso, natto, tempe, etc., have better antioxidant activity than the non-fermented ones (Esaki et al. 1994).
8.3 Fruit- and Vegetable-Based Products
Probiotic foods prepared using fruits, vegetables, and cereals also have additional health benefits since fruits and vegetables are themselves considered as health foods due to vitamins, minerals, antioxidants, phenolics, and fibers present in them. Moreover, many products contain additional ingredients used to enhance their flavoring and/or taste. These flavoring agents may also increase nutritional benefits of such foods, e.g., antitumor activities of cabbage and garlic used in kimchi, inhibition of aflatoxin B1 due to red pepper extract (Park et al. 1991), etc. Currently, there are more than 21 different commercial vegetable fermentations in Europe along with a large number of fermented vegetable juices and blends. The fermentations of olives, cucumbers, and cabbage are economically most significant (Caplice and Fitzerland 1999). Nonetheless, inhibitory effects of fermentation metabolites such as bacteriocins are well known (Dave and Shah 1997). Several bacteriocin strains have been isolated from kimchi. The inhibitory effects were observed not only in bacteria but also in fungi, e.g., antifungal compound 3,6-bis(2-methylpropyl)-2,5-piperazinedione was identified as being produced by L. plantarum strain from kimchi (Yang and Chang 2010). Bacteriocins along with organic acids (e.g., lactic acid) play a key role in controlling growth of undesirable and pathogenic organisms, thus avoiding costly treatments and packaging. The large amount of raw material is processed in this way in food industry because of nutritional, physiological, and hygienic aspects of the process (Karovicova et al. 1999).
9 Product Development and Challenges
New product development is always a challenge for both basic and applied research. It is an expensive process and requires detailed knowledge of the product, procedure, and consumers. In food sector, many parameters have to be considered such as sensory and physicochemical properties, extended shelf life, stability, and reasonable price. Careful selection of strains used and good monitoring throughout the manufacturing process are required to control the metabolic products and hence the final pH. So, use of mixed probiotic cultures is preferred which help to increase the growth rates, decrease the fermentation time, and eliminate certain sensory and texture defects and above all improve the nutritional value of the product (Gomes and Malcata 1999).
Another means to use probiotic strains is by adding them to beverages directly into the finished product which may help to retain better viability and functionality (Prado et al. 2008). Probiotic soy products or the products prepared from cereals, vegetables, and fruits are excellent substitutes for dairy products as a source of probiotics. Not only probiotic fermented vegetables but also their by-products have commercial value, e.g., during sauerkraut production, pH of final product is 3.5–3.8, at which cabbage or other vegetables can be preserved for a long time. Sauerkraut brine is an important by-product of cabbage fermentation industry and can be used as a substance for production of carotenoids by Rhodotorula rubra or for β-glucosidase production by Candida wickerhamii (Sim and Hang 1996) for commercial applications.
10 Quality Control
It is evident from the above discussion that non-dairy probiotic beverages are recognized as health drinks with a lot of scope in product development. However, use of probiotic organisms depends on many parameters like processing, storage, chemical composition of food, growth phase of organism, pH, water activity and salt content, interaction with starter culture, food matrix, etc. which affect their ability to survive in the product as well as in the consumers’ GI tract. Hence, quality control of probiotic strains is an important issue especially for monitoring their adhesion, gastric stability, and viability during manufacture, storage, and distribution. For these, some recommended practices are (1) appropriate culture maintenance procedures to reduce the number of passages, (2) use of more than one model to examine stability of the strain, (3) attempts for in vivo quantitative extrapolation of in vitro assays, and (4) studies on interspecies variation with respect to functional properties (Lee and Salminen 1995).
If adhesion properties of probiotic strains are altered during industrial processes their technological traits may also alter. Hence, they should be monitored carefully. Adhesion of probiotic bacteria varies in different in vitro models within the same strains and also shows difference between the strains (Lehto and Salminen 1996, 1997; Tuomola and Salminen 1998). This may be due to difference in surface properties required for adhesion to epithelial cells. Commonly used system is Caco-2 cell line and human ileostomy glycoprotein (Lehto and Salminen 1996, 1997; Tuomola and Salminen 1998).
Viability is another important characteristic as the strain should be viable during manufacture, storage, and after consumption. It is essential that fermented products should contain satisfactory number of active cells at the time of consumption (106 CFU/ml) since minimum therapeutic daily dose is 108–109 viable cells which is equivalent to 100 g of food intake containing 106–107 viable cells/ml (Rasic and Kurmann 1983). The organism should survive in sufficient numbers without any adverse effects on sensory properties of the product and should not increase acidification of product during shelf life (Shah et al. 1995; Roy et al. 1997). Such products must be consumed regularly to achieve and maintain the desirable effects on intestinal microflora. Unfavorable water activity (e.g., cereals, honey, marmalade, chocolate, etc.), pH, bile and salt concentration, and oxygen tension lead to death of bacteria (Vasudha and Mishra 2013). Therefore, acquiring the data for long-term stability to acid/bile is essential (Lee and Salminen 1995; Lee and Wong 1998).
Among the health benefits, hypocholesterolemic potential of probiotics is well known. However, there is lack of dosage response studies to determine minimal effective dosage of probiotics and prebiotics to reduce blood cholesterol levels (Kun et al. 2008). A review of previous studies represents that it depends on strains used and clinical characters of patients. It is necessary to establish clinically effective dosage based on human studies (Larkin et al. 2007). It is clear from the above discussion that viability, stability of probiotic strains (Bonaparte and Reuter 1997; Joseph et al. 1998), and determination of effective dosage are main challenges in research and development of probiotic products. In order to overcome the challenges, microencapsulation technologies have been developed and used successfully in various foods, which increase their viability in cereal- and fruit-based matrices.
11 Technological Innovations
The markets of probiotic products and supplements are increasing worldwide (Playne 1997). Today, there are >70 bifidus and acidophilus containing products worldwide (Shah 2000). Probiotic survival in products is affected by a range of factors including pH, hydrogen peroxide production, oxygen toxicity, storage temperatures, stability in dried or frozen form, and compatibility with traditional starter culture during fermentation (Dave and Shah 1997; Kailasapathy and Rybka 1997). Oxygen plays a major role in the poor survival of probiotic bacteria (Brunner et al. 1993). Hence, it is necessary to prevent their exposure to adverse external conditions. Therefore, the research in the past decade was focused on replacing their carrier food or by improving the protection of acid-sensitive strain via microencapsulation with cellulose acetate phthalate (Rao et al. 1989), n-carrageenan (Dinakar and Mistry 1994), or Ca-alginate (Kim et al. 1996). The former appears to be technologically and commercially most feasible since food carrier has buffering/protective effect and helps in survival in gastric juice.
11.1 Microencapsulation
Microencapsulation helps to separate a core material from its environment until it is released. It protects the unstable core from its environment, thereby improving its stability and viability, extends the core’s shelf life, and provides a sustained and controlled release in specific parts of the gut. The size of capsules may vary from submicron to several millimeters and they can be of different shapes (Franjione and Vasishtha 1995). Most commonly applied technologies are emulsification, coacervation, spray drying, spray cooling, freeze drying, fluid bed coating, and extrusion technologies. More expensive techniques are liposome encapsulation and cyclodextrin encapsulation. Sensitivity of the system to mechanical stress, pH, or different microbial enzymes in the gut facilitates targeted and sustained release.
Microencapsulation using the gentle and nontoxic matrices helps to enhance survival of the bacteria in acid and bile as well as heat. For this, alginate is commonly used due to (1) its non-toxicity, (2) its ability to form gentle matrices with calcium chloride to trap probiotic bacteria, (3) the viability of bacteria following encapsulation, and (4) reversibility of immobilization (Shah and Ravula 2000). Microencapsulation techniques are commonly used for probiotics, vitamins, minerals, antioxidants, etc. Further research in this line will help to develop co-encapsulation technique so as to combine two or more bioactive components for collective effect.
Stress response mechanism in LABs for industrial applications has been studied at the molecular level (Prasad et al. 2003). In L. acidophilus, the genes identified are FiFo-ATPase in acid stress response (Kullen and Klaenhammer 1999) and molecular chaperones groESL and dnaK in heat stress (Shah 2002). Thermotolerance was found to be better in heat-adapted strains of L. paracasei NFB338 (Desmond et al. 2001). Also, Lactobacilli were found to acquire cross-stress tolerance to heat when exposed to mild osmotic stress (Desmond et al. 2001). The authors have also demonstrated enhanced viability of salt-adapted culture as compared to the control one when dried under same conditions. In addition to acid and heat, organisms in fermented foods are also exposed to oxygen stress (Shah 2002). Oxygen content in the product and oxygen permeation through package may affect viability of probiotics in fermented products. Hence, it is necessary to measure oxygen tolerance of probiotic bacteria. Talkwalker and kailashpathy (2004) have published modified Relative Bacterial Growth Ratio (RBGR) method for quantitative measurement of oxygen tolerance, which will help in screening more oxygen-tolerant strains (Talkwalker and kailashpathy 2004). Osp protein was found to be upregulated in O2-tolerant Bifidobacterium strain (Ahn et al. 2001).
11.2 Spray Drying and Freeze Drying
Conventionally, freeze drying or spray drying of probiotic strains is used to make them available in the powder form on a large scale (Holzapfel et al. 2001). Spray drying is the most commonly used microencapsulation method in the food industry since it is economical and flexible, easy to scale up, produces a good quality product, and can be operated on continuous basis with simple equipmentation (Dzieazak 1988). However, further research in this area showed that retaining viability is the challenge due to temperature and osmotic extremes used in this process (Silva et al. 2002). This problem can be overcome either by optimizing the drying technology to reduce the harshness of the treatment or by improving the strains by gene manipulation or mutation. The former has been achieved by proper control and monitoring of processing conditions to produce viable encapsulated cultures of desired particle size, e.g., O’Riordan et al. (2001) have found that 100 °C inlet temperature and 45 °C of outlet temperature are suitable to produce microspheres of bifidobacteria with gelatinized modified starch as coating material. Meng et al. (2006) have reviewed that the stress responses in probiotic strains can have a remarkable effect on their ability to survive processing such as freeze drying, spray drying, and during gastric transit. In such cases, overexpressing heat shock proteins like GroEsL or addition of thermoprotectants to drying medium is advantageous (Desmond et al. 2004). Stress induced by temperature changes, phase changes, drying, or a combination tends to damage cell membrane and proteins. To overcome cell injury or death due to heat and dehydration, thermoprotectants have been added to media prior to drying, e.g., trehalose (Conrad et al. 2000), non-fat milk solids (Corcoran et al. 2004), prebiotics (Corcoran et al. 2005), granular starch (Crittenden et al. 2001), gum acacia (Desmond et al. 2004), etc. The addition of gum acacia in the drying medium resulted in 1000-fold increase in stability of dried L. paracasei NF13C 338 during powder storage at 15 °C and 30 °C (Lian et al. 2002). Hundred fold increase in viability was also observed when exposed to porcine gastric juice compared to control spray-dried culture (Desmond et al. 2002). Similarly, addition of cryoprotectants like inulin improves viability during freeze drying. Freeze-dried L. bulgaricus survived better at −20 °C for more than 10 months when grown in the presence of fructose, lactose, or mannose. Incorporation of glucose, fructose, or sorbitol in drying medium also resulted in better survival at low temperature (Carvalho et al. 2004).
11.3 Emulsion and Phase Separation
Most of the literature reported on the encapsulation of probiotic bacteria has used the emulsion technique to produce small amount of capsules. The capsules or beads are formed in a two-step procedure involving dispersion and hardening. The dispersion can be performed either by extrusion or by emulsification (Groboillot et al. 1994). The former involves projecting an emulsion core and coating material through a nozzle at high pressure. If the droplets are formed in a controlled manner, the technique is known as prilling. Beads can be produced on a large scale by using multinozzle systems, rotating disk atomizers, or by the jet cutting technique (Heinzen 2002).
11.4 Other Technologies
Another technology reported is vacuum impregnation to have beneficial effects of probiotics with fruits and vegetables. In this, apple cylinders were impregnated either with commercial apple juice containing Saccharomyces cerevisiae, or with whole milk or apple juice containing 107 or 108 cfu/ml of Lactobacillus casei. Impregnated apple samples were air dried at 40 °C and stored at room temperature for 2 months to increase stability and to assure fruit preservation. The number of L. casei viable cells in dried and stored product was more than 106 CFU/g which is similar to that in commercial dairy products (Vos et al. 2010).
12 Future Perspectives
Non-dairy probiotic products have a huge potential for food industry. These functional foods may be further explored through the development of new ingredients, processes, and technologies in order to improve their nutritional and sensory appeal. The key areas for research and development in non-dairy food products are raw materials used, sensory qualities, microencapsulation technologies, and strain improvement. The studies on novel or different matrices/carrier foods will help to increase the variety of non-dairy probiotic foods to offer a wide choice to consumers. The main reason for this is occurrence of non-dairy allergies linked to soya, gluten, and vegetables. Enhancement in sensory appeal of probiotic foods will ensure its consumption in the quantity and frequency to achieve anticipated health benefits. Also, consumers must be convinced by clear and trustworthy health claims so as to further increase the market of these functional foods.
In order to achieve the claimed health benefits, sustained viability and stability of the culture are significant. Hence, the technology of microencapsulation needs to develop with more precise machinery, capsule, and better delivery systems to protect the strains from external stress. In this regard, nano-encapsulation may get importance in near future to develop designer probiotic bacterial preparations for delivery to certain parts of GIT where they can interact with specific receptors. Such probiotic preparations may act as de novo vaccines and help in immunomodulation. Thus, research and development in this area will improve the delivery and sustained release of viable cells. Moreover, this has to be coupled with in vivo studies using human subjects (Kaliasapathy 2002).
Efforts for strain development are required to obtain mildly acidifying, less fastidious strains with sustained probiotic potential for application as starters or as supplements for food fortification. Besides, the advances in genetic engineering techniques will lead to the designing of more efficient strains and also introduce new functions in same strains. Earlier examples of such studies are expression of S. mutans surface protein Ag in L. lactis (Iwaki et al. 1990), murine interleukin 10 (IL-10) in recombinant L. lactis (Steidler et al. 2000), etc. Research and development in this area will result in live recombinant vaccines (Seegers 2002) using food grade bacteria (GRAS). However, studies on safety and efficacy of engineered strains and original strains are highly desirable. This is particularly necessary for children, pregnant women, elderly people, and immunocompromised people.
In addition to the technological aspects, future efforts should be directed to understand in detail the mechanisms of their health-promoting effects, increase in public awareness, and their applications in human and veterinary foods so as to gain their health benefits as per the recommended dosage, thereby reducing the health care cost.
References
Adams MR, Marteau P (1995) On the safety of lactic acid bacteria from food. Int J Food Microbiol 27:263–264
Ahn JB, Hwang HJ, Park JH (2001) Physiological responses of oxygen-tolerant anaerobic Bifidobacterium longum under oxygen. J Microbiol Biotechnol 11:443–451
Angelov A, Gotcheva V, Kuncheva R, Hristozova T (2006) Development of a new oat-based probiotic drink. Int J Food Microbiol 112:75–80
Anonymous (2010) The benefits of probiotics for your pet. Available at http://www.flintriver.com/ProductInfo.asp?pi=Probiotics-Overview.htm. Accessed 8 Jan 2010
Arici M, Coskun F (2001) Hardaliye: fermented grape juice as a traditional Turkish beverage. Food Microbiol 18:417–421
Aukrust W, Blom H, Sandtorv F, Slinde E (1994) Interaction between starter culture and raw material in lactic acid fermentation of sliced carrot. Lebensm Wiss Technol 27:337–341
Ausco M, Leal V, Baras M, Ruiz-Barba JL, Florianz B, Jimenez R (1998) Bacteriocin production and competitiveness of Lactobacillus plantarum LPC010 in olive juice broth a culture medium obtained from olives. Int J Food Microbiol 43:129–134
Bhadoria PBS, Mahapatra SC (2011) Prospects technological aspects and limitation of probiotics. Eur J food Res Rev 1(2):23–42
Blandino A, Al-Aseeri ME, Pandiella SS, Cantero D, Webb C (2003) Cereal-based fermented foods and beverages. Food Res Int 36(6):527–543
Biacs P (1986) Fermentované potraviny. Bull PV 25:1–13
Bonaparte C, Reuter G (1997) Bifidobacteria in commercial dairy products: which species are used? Microecol Ther 26:181–198
Brunner et al (1993) Abnormal behaviour associated with a point mutation in the structural gene for monamine oxidase A. Science 262(S133):578–580
Caplice E, Fitzerland GF (1999) Food fermentations: role of microorganisms in food production and preservation. Int J Food Microbiol 50:131–149
Carvalho AS, Silva J, Ho P, Teixeira P, Malcata FX, Gibbs P (2004) Effects of various sugars added to growth and drying media upon thermotolerance and survival throughout storage of freeze-dried Lactobacillus delbrueckii ssp. bulgaricus. Biotechnol Prog 20:248–254
Champagne CP, Roy D, Gardner N (2005) Challenges in the addition of probiotic cultures to foods. Crit Rev Food Sci Nutr 45:61–84
Conrad PB, Miller DP, Cielenski PR, de Pablo JJ (2000) Stabilization and preservation of Lactobacillus acidophilus in saccharide matrices. Cryobiology 41:17–24
Corcoran BM, Ross RP, Fitzgerald GF, Stanton C (2004) Comparative survival of probiotic lactobacilli spray-dried in the presence of prebiotic substances. J Appl Microbiol 96:1024–1039
Corcoran BM, Stanton C, Fitzgerald GF, Ross RP (2005) Survival of probiotic lactobacilli in acidic environments is enhanced in the presence of metabolizable sugars. Appl Environ Microbiol 17(6):3060–3067
Crittenden R, Laitila A, Forsell P, Matto J, Saarela M, Matilla-Sandholm T, Myllarinen P (2001) Adhesion of bifidobacteria to granular starch and its implications in probiotic technologies. Appl Environ Microbiol 67:3469–3475
Crittenden R, Bird AR, Gopal P, Henriksson A, Lee YK, Playne MJ (2005) Probiotic research in Australia, New Zealand and the Asia-Pacific region. Curr Pharm Des 11:37–53
Dani C, Biadaioli R, Bertini G, Martelli E, Rubaltelli FF (2002) Probiotics feeding in prevention of urinary tract infection, bacterial sepsis and necrotizing enterocolitis in preterm infants. Neonatology 82(2):103–108
Dave RI, Shah NP (1997) Effect of cysteine on the viability of yogurt and probiotic bacteria in yogurts made with commercial starter cultures. Int Dairy J 7:537–545
Desmond C, Stanton C, Fitzgerald GF, Collins K, Ross RP (2001) Environmental adaptation of probiotic lactobacilli towards improved performance during spray drying. Int Dairy J 11:801–808
Desmond C, Ross RP, O’Callaghan E, Fitzgerald G, Stanton C (2002) Improved survival of Lactobacillus paracasei NFBC 338 in spray-dried powders containing gum acacia. J Appl Microbiol 93:1003–1011
Desmond C, Fitzgerald GF, Stanton C, Ross RP (2004) Improved stress tolerance of GroESL-overproducing Lactococcus lactis and probiotic Lactobacillus paracasei NFBC 338. Appl Environ Microbiol 70(10):5929–5936
Dinakar P, Mistry VV (1994) Growth and viability of Bi®dobacterium bi®dum in Cheddar Cheese. J Dairy Sci 77:2854–2864
Dodd HM, Gasson MJ (1994) Bacteriocins of lactic acid bacteria. In: Gasson MJ, de Vos WM (eds) Genetics and biotechnology of lactic acid bacteria. Blackie Academic and Professional, London, pp 211–252
Donohue D, Salminen S (1996) ‘Safety of probiotic bacteria’ in Asia Paci®c. J Clin Nutr 5:25–28
Driessen FM, Kingma F et al (1982) Evidence that Lactobacillus bulgaricus in yogurt is stimulated by carbon dioxide produced by Streptococcus thermophilus. Neth Milk Dairy 36:135–144
Drozen M, Harrison T (1998) Structure/function claims for functional foods and nutraceuticals. Nutraceut World 1:18
Dziezak JD (1988) Microencapsulation and encapsulated ingredients. Food Technol 42:36–151
Esaki H, Onozaki T, Osawa T (1994) Antioxidative activity of fermented soybean products. In: Huang MT (ed) Food phytochemicals for cancer prevention I, fruits and vegetables. American Chemical Society, Washington, DC, pp 353–360
Euromonitor (2009) Functional foods: a world survey. Euromonitor international, London. Functional Times, Food Business, February, 35, p 6
Finegold SM, Sutter VL et al (1983) Normal indigenous intestinal flora. In: Human intestinal microflora in health and disease. Academic Press, New York, NY, pp 3–31
Franjione J, Vasishtha N (1995) The art and science of microencapsulation. Southwest Research Institute, San Antonio, TX
Fuchs RHB, Borsato D, Bona E, Hauly MCO (2005) “Iogurte” de soja suplementado com oligofrutose e inulina. Cieˆncia e Tecnologia de Alimentos 25:175–181
Giraud E, Grosselin L, Parada JL, Raimbault M (1993) Purification and characterization of an extracellular amylase from Lactobacillus plantarum strain A6. J Appl Bacteriol 75:276–282
Gomes AMP, Malcata FX (1999) Bifidobacterium spp. and Lactobacillus acidophilus: biological, biochemical, technological and therapeutical properties relevant for use as probiotics. Trends Food Sci Technol 10:139–157
Granato D, Branco GF, Nazzaro F, Cruz AG, Faria JAF (2010) Functional foods and non dairy probiotic food development: trends, concepts, and products. Compr Rev Food Sci food Saf 9:292–302
Groboillot A, Boadi DK, Poncelet D, Neufeld RJ (1994) Immobilization of cells for application in the food industry. Crit Rev Biotechnol 14:75–107
Guarner F, Schaafsma GJ (1998) Probiotics. Int J Food Microbiol 39(3):237–238
Hamilton JMT, Miller et al (2003) Probiotics and prebiotics in the elderly. Post Grad Med J 80:47–57
Hauly MCO, Fuchs RHB, Prudencio-Ferreira SH (2005) Soymilk yoghurt supplemented with fructooligosaccharides: probiotic properties and acceptance. Braz J Nutr 18:613–622
Heinzen C (2002) Microencapsulation by prilling and co-extrusion. In: Nutraceuticals and probiotics (Workshop No. 53). Technology Training Centre, Basil, Germany, 26–28 June 2002 (Abstract)
Henneberg W (1926) About Bacillus acidophilus and “acidophilus-milk” (reform-yogurt). Molkerei Zeitung 40:2633–2635 (in German)
Holzapfel WH (2002) Appropriate starter culture technologies for small-scale fermentation in developing countries. Int J Food Microbiol 75:197–212
Holzapfel W-H, Schillinger U et al (1997) Systematics of probiotic lactic acid bacteria with reference to modern phenotypic and genomic methods. Microecol Ther 26:1–10
Holzapfel WH, Haberer P, Geisen R, Bjorkroth J, Schillinger U (2001) Taxonomy and important features of probiotic microorganisms in food and nutrition. Am J Clin Nutr 73:365s–373s
Huis in’t Veld JHJ, Havenaar R (1997) Selections criteria and application of probiotic microorganisms in man and animal. Microecol Ther 26:43–58
Iwaki M, Okahashi N, Takahashi I, Kanamoto T, Sugita-Konishi Y, Aibara K, Koga T (1990) Oral immunization with recombinant Streptococcus lactis carrying the Streptococcus mutans surface protein antigen gene. Infect Immun 58:2929–2934
Joseph PJ, Dave RI, Shah NP (1998) Antagonism between yogurt bacteria and probiotic bacteria isolated from commercial starter cultures, commercial yogurts, and a probiotic capsule. Food Aust 50:20–23
Joshi VK, Sharma S (2009) Lactic acid fermentation of radish for shelf-stability and pickling. Nat Prod Rad 8(1):19–24
Juttlestad A (1998) Crafting appetizing nutraceuticals. Food Prod Des 3:97–106
Kalantzopoulos G (1997) Fermented products with probiotic qualities. Anaerobe 3:185–190
Kaliasapathy K (2002) Microencapsulation of probiotic bacteria: technology and potential applications. Curr Issues Intest Microbiol 3:39–48
Kailasapathy K, Rybka S (1997) L. acidophilus and Bifidobacterium spp. – their therapeutic potential and survival in yoghurt. Aust J Dairy Technol 52:28–35
Karovicova J, Drdak M, Polonsky J (1994) Výber vhodných kmeňov mikroorganizmov pre konzerváciu karotky a papriky mliečnou fermentáciou. Potrav Vědy 12:105–113
Karovicova J, Drdak M, Greif G, Hybenova E (1999) The choice of strains of Lactobacillus species for the lactic acid fermentation of vegetable juices. Eur Food Res Technol 210:53–56
Kim KI, Baek YJ, Yoon YH (1996) Effects of rehydration media and immobilization in Ca-alginate on the survival of Lactobacillus casei and Bi®dobacterium bi®dum. Korean J Dairy Sci 18:193–198
Kullen MJ, Klaenhammer TR (1999) Identification of the pH inducible, proton translocating F1F0ATPase (atpBEFHAGDC) operon of Lactobacillus acidophilus by differential display: gene structure, cloning and characterization. Mol Microbiol 33(6):1152–1161
Kun S, Rezessy-Szabo JM, Nguyen QD, Hoschke A (2008) Changes of microbial population and some components in carrot juice during fermentation with selected Bifidobacterium strains. Process Biochem 43(8):816–821
Larkin TA, Astheimer LB, Price WE (2007) Dietary combination of soy with probiotic or probiotic food significantly reduces total and LDL cholesterol in mildly hypercholesterolaemic subjects. Eur J Clin Nutr 63:238–245
Lee YK, Salminen S (1995) The coming of age of probiotics. Trends Food Sci Technol 6:241–245
Lee YK, Wong S-F (1998) Stability of lactic acid bacteria in fermented milk. In: Salminen S, von Wright A (eds) Lactic acid bacteria, microbiology and functional aspects. Marcel Dekker, New York, pp 103–114
Lehto E, Salminen S (1996) Adhesion of twelve different Lactobacillus strains to Caco-2 cell cultures. Nutr Today 31:49–50
Lehto E, Salminen S (1997) Adhesion of two Lactobacillus strains, one Lactococcus strain and one Propionibacterium strain to cultured human intestinal Caco-2 cell line. Biosci Microflora 16:13–17
Leroy F, Degeest B, De Vuyst L (2002) A novel area of predictive modelling: describing the functionality of beneficial microorganisms in foods. Int J Food Microbiol 73:251–259
Li J, Zhang W, Wang C, Yu Q, Dai R, Pei X (2012) Lactococcus lactis expressing food-grade β-galactosidase alleviates lactose intolerance symptoms in post-weaning Balb/c mice. Appl Microbiol Biotechnol 96(6):1499–1506
Lian WC, Hsiao HC, Chou CC (2002) Survival of bifidobacteria after spray-drying. Int J Food Microbiol 74:79–86
Luckow T, Delahunty C (2004) Which juice is ‘healthier’? A consumer study of probiotic non-dairy juice drinks. Food Qual Prefer 15:751–759
Martensson O, Staaf J, Duenas-Chaso M, Irastorza A, Oste R, Holst O (2002) A fermented, ropy, non-dairy oat product based on the exopolysaccharide producing strain Pediococcus damnosus. Adv Food Sci 24:4–11
Meng XC, Stanton XC, Fitzgerald C, Daly GF, Ross RP (2006) Anhydrobiotics: the challenges of drying probiotic cultures. Food Chem 106:1406–1416
Metchnikoff E (1908) The prolongation of life—optimistic studies. Heinemann, London
Mousavi ZE, Mousavi SM, Razavi SH, Emam-Djomeh Z, Kiani H (2011) Fermentation of pomegranate juice by probiotic lactic acid bacteria. World J Microbiol Biotechnol 27(1):123–128
Muianja CMBK, Narvhus JA, Treimo J, Langsrud T (2003) Isolation, characterisation and identification of lactic acid bacteria from bushera: a Ugandan traditional fermented beverage. Int J Food Microbiol 80:201–210
Nout MJR, Ngoddy PO (1997) Technological aspects of preparing affordable fermented complementary foods. Food Control 8:279–287
O'riordan K, Andrews D, Buckle K, Conway P (2001) Evaluation of microencapsulation of a Bifidobacterium strain with starch as an approach to prolonging viability during storage. J Appl Microbiol 91(6):1059–1066
Ouwehand AC, Salvadori B, Fonden R, Mogensen G, Salminen S, Sellara R (2003) Health effects of probiotics and culture-containing dairy products in humans. Bull Int Dairy Fed 380:4–91
Parada JL, Sambucetti ME, Zuleta A, Rio ME (2003) Lactic acid fermented products as vehicles for probiotics. In: Roussos S et al (eds) New horizons in biotechnology, vol 23. Springer, Berlin, pp 5–12
Park KY, Kim SH, Suh MJ, Chung HY (1991) Inhibitory effect of garlic on the mutagenicity in Salmonella assay system and on the growth of HT-29 human colon carcinoma cells. Korean J Food Sci Technol 23:370–374
Pereira DIA, Gibson GR (2002) Effects of consumption of probiotics and prebiotics on serum lipid levels in humans. Crit Rev Biochem Mol Biol 37(4):259–281
Pereira AF, Maciel TC, Rodrigues S (2011) Probiotic beverage from cashew apple juice fermented with Lactobacillus casei. Food Res Int 44(5):1276–1283
Perez PF, De Antoni GL et al (1991) Formate production Streptococcus thermophilus cultures. J Dairy Sci 74:2850–2854
Playne M (1997) Trends in probiotics in Europe. Aust Dairy Foods Feb:20–21
Prado FC, Parada JL, Pandey A, Soccol CR (2008) Trends in non-dairy probiotic beverages. Food Res Int 41:111–123
Radke-Mitchell LC, Sadine WE (1986) Influence of temperature on associative growth of Streptococcus thermophilus and Lactobacillus bulgaricus. J Dairy Sci 69:2558–2568
Rafter J (2004) The effects of probiotics on colon cancer development. Nutr Res Rev 17:277–284
Rao AV, Shiwnarain N, Maharaj I (1989) Survival of Microencapsulated Bi®dobacterium pseudolongum in Simulated Gastric and Intestinal Juices. Can Inst Food Sci Technol J 22:345–349
Rasic JL, Kurmann JA (1983) Bi®dobacteria and their role. BirkhauÈ ser, Basel
Reuter G (1997) Present and future of probiotics in Germany and in Central Europe. Biosci Microflora 16:43–51
Ross RP, Desmond C, Fitzgerald GF, Stanton C (2005) Overcoming the technological hurdles in the development of probiotic foods. J Appl Microbiol 98(6):1410–1417. doi:10.1111/j.1365-2672.2005.02654.x
Roy D, Mainville I, Mondou F (1997) Bifidobacteria and their role in yogurt-related products. Microecol Ther 26:167–180
Saarela M, Virkajärvi I, Alakomi HL, Sigvart-Mattila P, Mättö J (2006) Stability and functionality of freeze-dried probiotic Bifidobacterium cells during storage in juice and milk. Int Dairy J 16:121477–121482
Santos MCR (2001) Desenvolvimento de bebida e farinha láctea fermentada de ac ̧ão probiótica a base de soro de leite e farinha de mandioca por cultura mista de Lactobacillus plantarumA6, Lactobacillus casei Shirotae, Lactobacillus acidophilus. MSc thesis. UFPR, 106 p
Schaafsma G (2008) Lactose and lactose derivatives as bioactive ingredients in human nutrition. Int Dairy J 18:458–465
Seegers JF (2002) Lactobacilli as live vaccine delivery vectors: progress and prospects. Trends Biotechnol 20:508–515
Sgorbati B, Biavati B et al (1995). The genus Bifidobacterium in the lactic acid bacteria. In: The genera of lactic acid bacteria, vol 2. Blackie Academic, London, pp 279–306 (Chapter 8)
Shah NP (2000) Probiotic bacteria: selective enumeration and survival in dairy foods. J Dairy Sci 83:894–907
Shah N (2002) The exopolysaccharides production by starter cultures and their influence on textural characteristics of fermented milks. In: Symposium on new developments in technology of fermented milks. International Dairy Federation, 3 June 2002, Comwell Scanticon, Kolding, Denmark. Abstract, p 5
Shah NP, Lankaputhra WEV, Britz ML, Kyle WSA (1995) Survival of L. acidophilus and Bifidobacterium bifidum in commercial yogurt during refrigerated storage. Int Dairy J 5:515–521
Shah NP, Ravula RR (2000) Microencapsulation of probiotic bacteria and their survival in frozen fermented dairy desserts. Aust J Dairy Technol 55:139–144
Sheehan VM, Ross P, Fitzgerald GF (2007) Assessing the acid tolerance and the technological robustness of probiotic cultures for fortification in fruit juices. Innov Food Sci Emerg Technol 8:279–284
Silva J, Carvahlo AS, Teixeira P, Gibbs PA (2002) Bacteriocin production by spray dried lactic acid bacteria. Lett Appl Microbiol 34:77–81
Sim SL, Hang YD (1996) Sauerkraut brine: a potential substrate for production of yeast ß-glucosidase. Lebensm Wiss Technol 29:365–367
Stanton C, Gardiner G, Meehan H, Collins K, Fitzgerald G, Lynch PB, Ross RP (2001) Market potential for probiotics. Am J Clin Nutr 73(2S):476S–483S
Steinkraus KH (1997) Classification of fermented foods: worldwide review of household fermentation techniques. Food Control 8:311–317
Steidler L, Hans W, Schotte L, Neirynck S, Obermeier F, Falk W et al (2000) Treatment of murine colitis by Lactococcus lactis secreting interleukin-10. Science 289(5483):1352–1355
Svanberg U, Lorri W (1997) Fermentation and nutrient availability. Food Control 8:319–327
Swain MR, Anandharaj M, Ray RC, Rani RP (2014) Fermented fruits and vegetables of Asia: a potential source of probiotics. Biotechnol Res Int 2014:19, http://dx.doi.org/10.1155/2014/250424
Tabbers MM, Benninga MA (2007) Administration of probiotic Lactobacilli to children with gastrointestinal problems: there is still little evidence. Ned Tijdschr Geneeskd 151(40):2198–2202
Talwalkar A, Kailasapathy K (2004) The role of oxygen in the viability of probiotic bacteria with reference to L. acidophilus and Bifidobacterium spp. Curr Iss Intest Microbiol 5:1–8
Tuomola E, Salminen S (1998) Adhesion of some probiotic and dairy Lactobacillus strains to Caco-2 cell cultures. Int J Food Microbiol 41:45–51
Vasudha S, Mishra HN (2013) Non dairy probiotic beverages. Int Food Res J 20(1):7–15
Venkatesh MG (1998) Designing effective programmes to prevent and control iron deficiency anaemia. Am J Clin Nutr 89:23–26
Viander B, Maki M, Palva A (2003) Impact of low salt concentration, salt quality on natural large-scale sauerkraut fermentation. Food Microbiol 20:391–395
Vos P, Fass MM, Spasojevic M, Sikkema J (2010) Encapsulation for preservation of functionality and targeted delivery of bioactive food components. Int Dairy J 20:292–302
Wacher C, Cañas A, Bárzana E, Lappe P, Ulloa M, Owens JD (2000) Microbiology of Indian and Mestizo pozol fermentation. Food Microbiol 17:251–256
Yang EJ, Chang HC (2010) Purification of a new antifungal compound produced by AF1 isolated from Kimchi. Int J Food Microbiol 139:56–63. doi:10.1016/j.ijfoodmicro.2010.02.012
Yoon KY, Woodams EE, Hang YD (2005) Fermentation of beet juice by beneficial lactic acid bacteria. LWT Food Sci Technol 38(1):73–75
Yoon KY, Woodams EE, Hang YD (2006) Production of probiotic cabbage juice by lactic acid bacteria. Bioresour Technol 97:1427–1430
Zourari A, Accolas JP et al (1992) Metabolism and biochemical characteristics of yogurt bacteria: A review. Lait 72:1–34
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Bhadekar, R., Parhi, P. (2016). Non-dairy Functional Foods: Potential of Probiotics. In: Garg, N., Abdel-Aziz, S., Aeron, A. (eds) Microbes in Food and Health. Springer, Cham. https://doi.org/10.1007/978-3-319-25277-3_1
Download citation
DOI: https://doi.org/10.1007/978-3-319-25277-3_1
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-25275-9
Online ISBN: 978-3-319-25277-3
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)