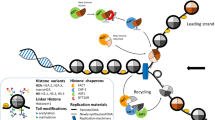
Abstract
The coordination of DNA replication with histone synthesis is of utmost importance as any imbalance between the two processes results in genomic instability and may even cause lethality. Hence, to maintain genome stability, histone synthesis is regulated at multiple levels—transcriptionally, posttranscriptionally and by modulating protein stability. This tight regulation facilitates the creation of a very transient histone pool for replication-coupled chromatin assembly and ensures that histone synthesis is downregulated when DNA replication is completed or stalled due to replication inhibition. As illustrated in this chapter, the bulk of histone synthesis during S phase is activated by the same cell cycle signals that initiate DNA replication and downregulated by the same DNA damage response pathways that arrest the DNA replication machinery upon DNA damage. Conversely, the availability of histone proteins and their chaperones that help package the newly replicated DNA into chromatin in turn regulate replication fork progression. Further, in senescent cells, the histone chaperone Histone Regulatory Homolog A (HIRA), a co-repressor of histone gene transcription, plays an important role in the formation of transcriptionally silent heterochromatin that incorporates replication-dependent histone genes as well as many genes needed for DNA replication to concomitantly shut down both histone and DNA synthesis. This chapter discusses the current state of knowledge on the coregulation of histone and DNA synthesis during S phase.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Histone
- Histone gene regulation
- Chromatin assembly
- Histone chaperone
- Replication-dependent histones
- MCM
- HIRA
- Cyclin
- Cdk
- NPAT
- SLBP
- DDR
- SAHF
- Hir
Introduction
The genomic DNA in eukaryotic cells exists in the form of nucleoprotein filaments collectively known as chromatin. The basic repeating unit of chromatin comprises the nucleosome in which 147 bp of DNA is wrapped around an octameric protein core consisting of two molecules each of the core histone proteins H2A, H2B, H3, and H4 [1]. The binding of linker histone H1 to the nucleosomes helps fold the chromatin further by forming more condensed higher order structures [2]. Histone chaperones help package the DNA into nucleosomes rapidly behind the replication fork as soon as a sufficient amount (~200 bp) of newly synthesized DNA emerges from the replication fork [3]. DNA replication and histone synthesis are strictly coordinated and it has been known for decades that when DNA replication is inhibited upon exposure to genotoxic agents, histone synthesis is rapidly downregulated in an evolutionary conserved response [4–7]. Conversely, a deficiency of histones or histone chaperones also has a strong inhibitory effect on both DNA replication initiation and elongation in higher eukaryotes [8–12], demonstrating that histone and DNA synthesis are intimately interlinked processes. This chapter discusses our current understanding of how histone and DNA synthesis are coordinately regulated during S phase of the cell cycle in budding yeast and mammalian cells.
Coordination of Histone Synthesis and DNA Replication in Budding Yeast
The coupling of histone gene expression and DNA replication in the budding yeast Saccharomyces cerevisiae is believed to be mainly achieved through transcriptional control as histone transcripts have a relatively short half-life [13]. There are 11 histone genes in budding yeast: single genes encoding H2A.Z (HTZ1) variant, centromeric H3 variant (CSE4), the linker histone H1 (HHO1) and two genes for each of the core histone proteins H2A, H2B, H3 and H4. The core histone genes occur in pairs, with the genes for H3 (Histone H Three 1 and 2, HHT1 and HHT2) being paired with H4 (Histone H Four 1 and 2, HHF1 and HHF2) and that of H2A (Histone Two A 1 and 2, HTA1 and HTA2) with H2B (Histone Two B 1 and 2, HTB1 and HTB2), which presumably helps maintain the right stoichiometry of individual core histone proteins relative to each other [14].
Each core histone gene pair is transcribed divergently from a common promoter at the G1/S boundary and repressed outside of S phase. This cell cycle regulation is conferred by activating and repressing elements present in the replication-dependent core histone (H2A, H2B, H3, and H4) promoters (Fig. 11.1a). Common to all the four histone promoters are Upstream Activating Sequence (UAS) elements that are bound by the Suppressor of Ty 10 (Spt10), the only histone promoter specific transcriptional co-activator known so far in the budding yeast. It has been suggested that Spt10 dimerizes and binds to pairs of histone UAS elements in late G1 [15]. Spt10 is bound to the histone promoters in alpha factor arrested G1 cells before histone gene expression is activated. Therefore, histone genes are likely to be turned on through the interaction of Spt10 with another known histone gene regulator Spt21 which is present at all four core histone promoters and its binding peaks in S phase [16].
Regulation of histone mRNA levels in budding yeast and mammals. Although the actual regulatory factors involved are different, in principle similar mechanisms govern histone gene transcription as well as histone mRNA levels in yeast and mammals. (a) Yeast histone genes are repressed outside of S phase and upon DNA damage in S phase by the Hir complex (Hir1–3, Hpc2) with help from the chaperones Asf1 and Rtt106. These factors mediate their repressive effects via the negative regulatory element (NEG). In S phase, SCFCdc4 promotes the activation and accumulation of the mitotic cyclins Clb1/2. We speculate that Clb1/2 may prompt Cdk1 mediated Hir complex phosphorylation, thereby relieving the repression of histone genes. Wee1 kinase phosphorylates and inhibits Cdk1 as part of the G2/M cell cycle checkpoint and the DNA Damage Response (DDR). Additionally, Wee1 phosphorylation of H2B tyrosine 37 has been shown to facilitate Hir complex binding to repress histone gene transcription. The Upstream Activating Sequences (UAS) in the histone gene promoters are bound by coactivators Spt10 and SBF (a heterodimer of Swi4 and Swi6), in a mutually exclusive manner to activate histone gene transcription in early S phase. Polyadenylation of histone mRNAs by Trf4/5 may contribute to their posttranscriptional regulation by modulating their half-lives. Histone mRNAs are degraded by the nuclear exosome in a process possibly mediated by Nrd1. The Lsm1–7-Pat1 complex may also degrade histone mRNAs based on the length of their polyA tails. (b) Mammalian histone genes require NPAT for transcription which is recruited to histone promoters by SSRE binding proteins (SSRBPs). Numerous transcription factors (HiNFs) confer tissue specificity or transcriptional control for different replication-dependent core histone subtypes. Mammalian replication-dependent core histone mRNAs do not possess poly-A tails but a stem loop in their 3′ UTR which is recognized by Stem Loop Binding Protein (SLBP) for posttranscriptional and translational processing. Mature histone mRNAs are stabilized and targeted to the cytoplasm by SLBP. At the end of S phase, the cell cycle machinery represses histone synthesis by inhibiting SLBP and E2F1, whilst activating the yeast Hir1/2 protein homolog HIRA. As in yeast, Wee1 phosphorylation of H2B tyrosine 37 also facilitates HIRA binding and termination of histone synthesis at the end of S phase. In addition, upon DNA damage, Wee1 inhibits Cdk1 activity through specific residues phosphorylation; this conserved mechanism prevents mitotic entry in presence of DNA lesions. In a complex signaling network involving checkpoint kinases, the DDR triggers both replication arrest and downregulation of histone synthesis through inhibition of histone gene transcription via NPAT inactivation and histone mRNA degradation via the LSM1–7 complex. Question marks indicate speculative pathways that currently lack direct experimental evidence. P phosphate. HLBs histone locus bodies
Recently, the Andrews laboratory has shown that Spt21 abundance is restricted to S phase by the Anaphase-Promoting Complex Cdc20 homologue 1 (APCCdh1) (Fig. 11.1a) [17]. This suggests that Spt21 levels oscillate and may contribute to the cell cycle regulation of histone genes. However, the effect of SPT21 deletion on histone gene expression is only partial as the deletion mutants show a strong decrease in HTA2-HTB2 and HHT2-HHF2 histone transcript levels but the expression of the other histone gene pairs HHT1-HHF1 and HTA1-HTB1 are only modestly reduced [17]. In contrast, all histone mRNA levels are greatly reduced in spt10 deletion mutant cells released from alpha factor mediated G1 arrest [18]. Additionally, spt10 deletion mutants exhibit a severe growth defect that can be rescued by a high copy plasmid carrying histone gene pairs. Overall, these observations demonstrate the importance of Spt10 for the activation of multiple histone gene pairs, while Spt21 may be more selective for the activation of the HHT2-HHF2 and HTA2-HTB2 gene pairs (Fig. 11.1a).
Histone promoters have putative binding motifs for the transcription factor Swi4 and genome-wide Chromatin Immunoprecipitation (ChIP) analysis shows that they are bound by SBF and possibly MBF transcription factors [19–22]. MBF and SBF are heterodimers containing Swi6 (a regulatory factor) and a sequence specific DNA binding factor Mbp1 (in MBF) and Swi4 (in SBF) respectively [23]. SBF and Spt10 bind to the same UAS elements (UAS2 and UAS3) at the HTA1-HTB1 promoter and their binding is mutually exclusive in vitro (Fig. 11.1a). SBF binding peaks in early S phase and concomitantly a SBF mediated small peak in histone gene expression is detectable. By the end of S phase, the binding of SBF and Spt10 at the histone promoters is completely abolished [15]. Overall, histone gene expression is only modestly reduced in swi4 or mbp1 deletion strains with some histone genes showing a small but significant reduction [16]. Spt10 and SBF may together control the expression from the HTA1-HTB1 promoter in G1/S as HTA1-HTB1 expression is completely abolished when all four UAS elements are removed from the promoter [15].
Inappropriate expression of histone genes outside of S phase or when DNA replication is arrested is associated with toxicity. To prevent this, histone genes are also subjected to negative regulation [8]. A group of four functionally related Histone Regulators (Hir1, Hir2, and Hir3) and the Histone Promoter Control 2 (Hpc2) proteins are shown to act in a complex as transcriptional co-repressors for three of the four histone gene pairs (Fig. 11.1a), HTA1-HTB1, HHT1-HHF1, and HHT2-HHF2, both outside of S phase and in response to replication arrest [14, 24]. A fifth protein, Asf1 (Anti-Silencing Function 1 ) also co-purifies with Hir proteins and asf1 mutants are partially defective in the repression of transcription from HTA1-HTB1, HHT1-HHF1, and HHT2-HHF2 gene pairs upon replication arrest [25]. Asf1/Hir-mediated repression of transcription relies on a specific DNA sequence, the negative (NEG) regulatory element which is present on the promoters of the three Hir-regulated gene pairs (Fig. 11.1a), but is absent from that of the HTA2-HTB2 promoter [4]. The HTA2-HTB2 gene pair also undergoes cell cycle regulation and its transcripts disappear outside of S phase as well as in response to replication arrest, but in a Hir-independent manner [26]. Asf1 and Hir are also chromatin assembly factors that bind to histones and help assemble and disassemble chromatin [27]. Asf1 stimulates the replication-independent nucleosome assembly activity of the Hir complex in vitro [28]. More recently, the H3/H4 chromatin assembly factor Rtt106 was identified as an additional regulator of histone gene expression [29]. Rtt106 functions downstream of Asf1 and the Hir complex and localizes to the histone gene promoters in a Hir- and Asf1-dependent manner (Fig. 11.1a) [29]. Further, the chromatin remodeling complex Rsc is also recruited to the histone gene promoters in a Hir and Rtt106-dependent manner (Fig. 11.1a) [30]. Rsc recruitment is mediated by Rtt106 and its recruitment to the histone gene promoters is compromised in rtt106 mutant cells, although the Hir complex and Asf1 remain bound [30].
Histone promoters are devoid of nucleosomes in either hir or asf1 or rtt106 mutant cells, suggesting that these histone chaperones form a repressive chromatin structure at the histone gene promoters to shut down transcription [29]. However, the defect in histone gene repression in an rtt106 or asf1 mutant is relatively mild compared to the nearly complete derepression of histone genes observed in hir/hpc mutants [29]. This raises the intriguing question whether a different pathway/factor exists through which the Hir complex exerts its repressive role at the histone gene promoters. It is also possible that part of the repressive activity of the Hir complex is mediated via Asf1 and Rtt106 such as under conditions of replication stress in S phase, while the remainder is carried out directly by the Hir complex itself in G2, M, and G1 in an Asf1/Rtt106 independent manner [31].
Previous work from the Osley and Johnston laboratories has demonstrated the requirement of a functional Cell Division Cycle 4 (Cdc4) protein for the transcription of the HTA1 gene in late G1 [32, 33] (Fig. 11.2). Cdc4 is critical for DNA replication initiation at G1/S and for meiotic nuclear division at G2/M [34, 35]. Cdc4, Cdc34, Cdc53, and Skp1 proteins form the Skp, Cullin, F-box containing SCF-Cdc4 complex which is required for the ubiquitin-dependent proteolysis of a number of genes that regulate cell cycle progression such as Sic1, Cdc6, Clb6, Cln3 [36–41]. Based on this, Amin et al tested if Cdc4 coordinates DNA replication with histone synthesis in G1/S. Using cdc4 ts (temperature sensitive) mutants they demonstrated that the activation of the HTA1-HTB1 gene pair is dependent on Cdc4 [31]. Since the major role of SCF-Cdc4 complex at the G1/S transition is the activation of all six Clb/Cdk1 complexes, this raises the question whether any of the six Clb/Cdk1 complexes are involved in activating histone gene expression [31]. Unexpectedly, S phase cyclins Clb5 and Clb6 which are involved in DNA initiation did not have any major impact on histone gene activation, although their activation was slightly delayed. Instead, the activation of histone genes HTA1-HTB1 and HTA2 showed a strong dependence on the mitotic Clb1 and Clb2 cyclins [31]. Further, Clb1/2 cyclin dependency can be bypassed by the disruption of the Hir complex which negatively regulates histone gene expression [31]. This suggests that the Cdks may negatively regulate the Hir-complex and as a result histone genes are activated in G1/S (Fig. 11.1a). Histone Regulatory homolog A (HIRA) , the mammalian homolog of Hir1 and Hir2 in yeast is a direct Cdk target and as such it is entirely possible that the Hir-complex is also regulated by Cdk phosphorylation [42]. Indeed, Hpc2 was identified as a direct target for Cdk1 in a global screen to identify new Clb2/Cdk1 substrates [43]. Further, Clb2 associates with the Hir complex in vivo [31] and the Clb1/Clb2-associated Cdk1 kinase is active at the G1/S transition when histone gene transcription is activated, although its kinase activity is lower than that observed in G2 [44–46].
Cross talk between the regulation of DNA and histone synthesis. Regulation of histone levels and DNA replication are tightly coupled. Several components of the cell cycle machinery cooperate with common factors involved in regulating both DNA and histone synthesis to ensure that enough histones are synthetized to satisfy chromatin assembly requirements following DNA replication, since both free histones and naked DNA are toxic for the cell. Some of these regulatory factors for DNA/histone synthesis are evolutionarily conserved while others are unique to either yeast or mammalian cells
Histone genes in yeast are also subjected to posttranscriptional regulation and this mechanism contributes to the cell cycle regulation of histone genes. Unlike their mammalian counterparts that end in a stem loop, yeast histone mRNAs are polyadenylated (Fig. 11.1a). The region that confers cell cycle regulation even when histone genes are expressed from a constitutive heterologous promoter has been mapped and lies in the 3′ Untranslated Region (UTR) . The 3′ UTR of the HTB1 gene contains a Distal Downstream Element (DDE) that lies about 100 nt downstream of the 3′-end cleavage site [47]. In addition, the DDE contains a transcription termination site and mutations in the DDE prevent the degradation of a reporter mRNA [48]. Work from the Bond and Campbell laboratories has demonstrated the involvement of the nuclear exosome (a multi-subunit complex of 3′ to 5′ exonucleases) and the 3′-end processing machinery consisting of the Trf4/Air2/Mtr4p Polyadenylation (TRAMP) complex in the cell cycle regulation of histone mRNAs [48, 49]. The nuclear exosome processes many RNAs in the nucleus that need to be first polyadenylated by the TRAMP complex comprising the PolyA polymerases Trf4 and Trf5, a putative ATP-dependent RNA helicase Mtr4p/Dob1p and the zinc knuckle-binding proteins Air1 and Air2 (TRAMP) [50–52]. Deletion of the nuclear exosome component Rrp6 as well as the TRAMP components Trf4 and Trf5 result in high levels of histone mRNAs [49]. It is thought that Trf4 and Trf5 components of the TRAMP complex add short polyA tails and tag the mRNA to be degraded by the nuclear exosome [50, 53]. In the case of histone mRNA regulation, the Air1 and Air2 RNA-binding proteins of the TRAMP complex are not needed and therefore another RNA-binding protein such as Nrd1 may support the recruitment of the nuclear exosome via interaction with Rrp6. Indeed, Nrd1 has been shown to interact with the TRAMP complex and stimulate the 3′-end processing of RNA Pol II transcripts by the exosome [54]. The transcriptional termination site which lies in the DDE of the HTB1 gene seems to be critical for the interaction with TRAMP and the nuclear exosome (Table 11.1).
The Role of Transcriptional Regulation in Coordinating Histone Synthesis and DNA Replication in S Phase in Mammalian Cells
Transcriptional Regulation of Core Histones in Mammalian Cells
The tremendous increase in histone protein levels during S phase to meet the demand for new chromatin assembly is a direct result of the accumulation of histone mRNAs in S phase, which in turn is controlled at the level of transcription, pre-mRNA processing and mRNA stability in mammalian cells [55–57]. We will first consider the contribution of histone gene transcription in ensuring an adequate supply of histones during DNA replication. There are multiple copies of replication-dependent or canonical histone H2A, H2B, H3, and H4 genes, the majority of which are clustered in a small number of chromosomal loci in mammals and are expressed only during S phase in a replication-dependent manner [57, 58]. The remainder of the histones genes is mostly single copy replication-independent histone genes scattered across the genome and these express primary sequence variants of the canonical histone genes throughout the cell cycle and appear to possess unique functions [57]. Replication-dependent histone gene promoters generally contain a core promoter for initiation by RNA polymerase II, a Subtype Specific Consensus Element (SSCE) and a distal activation domain. In the case of histone H4, whose regulation is the best studied among mammalian histone genes, cell cycle dependency comes from the SSCE element. The regulation of each of the mammalian histone genes involves different cis-acting elements and multiple transcription factors. Over the years, studies have uncovered many of these transcription factors and named them HiNFs (Histone Nuclear Factors), although these mostly correspond to known general mammalian transcription factors. For example, histone H4 genes contain two major protein–DNA interaction sites in vivo [59]. One of these is an enhancer element for activating histone H4 transcription via interactions with a variety of general transcription factors such as SP1 (Specificity Protein 1)/HiNF-C, Yin Yang 1 (YY1)/HiNF-I, High Mobility Group-I (HMG-I)/HiNF-A) [60–62]. Cell cycle control in G1/S is conferred by the second regulatory site via interactions with IRF2 (Interferon Regulatory Factor 2)/HiNF-M, CDP (CCAAT Displacement Protein)/HiNF-D and a H4 specific transcription factor HiNF-P [63, 64], all of which contribute to the cell cycle regulation of histone H4 expression [65].
Transcriptional control plays an important role in regulating the expression of the replication-dependent histone genes at the G1/S boundary. As cells enter S phase, histone mRNA transcription increases three- to fivefold compared to basal G1-levels [66, 67]. This cell cycle-dependent S phase expression of histone genes is conferred by the action of Cyclin E/Cdk2, the key regulator of S phase entry which also initiates DNA replication (Fig. 11.1b). The activity of Cyclin E/Cdk2 is required for phosphorylation of Cdc6 which protects it from degradation by the Anaphase-Promoting Complex (APC), which in turn allows the licensing of origins in late G1 [68, 69]. Another essential substrate for Cyclin E/Cdk2 is the Nuclear Protein Ataxia–Telangiectasia locus (NPAT) which is localized in a cell cycle-dependent manner in subnuclear structures known as the Histone Locus Bodies (HLBs) that contain the clustered histone genes [70–73]. The histone locus bodies also contain many factors required for histone mRNA synthesis and processing [74]. NPAT is an essential protein for normal mammalian development and enhances histone gene transcription as overexpression of NPAT activates promoters of multiple histone genes via the SSREs within the promoters [72]. The suppression of NPAT expression through RNA interference or conditional knockout impedes expression of all replication-dependent core histone genes. The promoter DNA sequences of different core histone subtypes are quite divergent, and direct DNA binding by NPAT has not been detected. Therefore, it has been proposed that coordination of the transcription of multiple core histone subtypes by NPAT occurs through the interaction of NPAT with factors that regulate transcription of the individual core histone subtypes. Indeed, after phosphorylation by Cyclin E/Cdk2, NPAT co-activates the transcription of histone genes by interacting with the H4 promoter specific transcription factor HiNF-P, a conserved Zn finger protein that binds to a histone H4 promoter regulatory element [75]. Although a large number of histone gene transcription factors have been characterized, HiNF-P is unique because it is the only known histone H4 promoter-specific factor that interacts directly with NPAT and they both reside in HLBs. Although the mechanism of HiNF-P action is not fully understood, it is clear that the HiNF-P/NPAT complex mediates a unique cell cycle regulatory mechanism that controls the G1/S phase transition.
NPAT also interacts with transcription factor Oct1 (Octamer Binding Factor 1) and its coactivator complex OCA-S (Oct-1 Co-Activator in S phase) that bind to the octamer binding element in the H2B promoter SSRE to activate H2B transcription [76]. Remarkably, components of OCA-S include nuclear p38/glyceraldehyde-3-phosphate dehydrogenase (GAPDH) and lactate dehydrogenase. Further, the activity of OCA-S is regulated by NAD (Nicotinamide Adenine Dinucleotide) and NADH, suggesting a link between the histone gene transcription and the cellular metabolic state/redox status [76]. The molecular mechanism underlying the regulation of H2B transcription by Oct1/OCA-S in accordance with the cellular metabolic/redox states is not yet clear. Nevertheless, the pattern of Oct-1 phosphorylation changes throughout the cell cycle, and may be CDK mediated, thereby potentially regulating Oct1 function at histone promoters [77].
Studies aimed at further elucidating the molecular mechanisms by which NPAT regulates histone gene activation at G1/S revealed an interaction of NPAT with components of the Tip60 Histone Acetyltransferase (HAT) complex [78]. The association of the Tip60-TRRAP (Transformation/Transcription Domain-Associated Protein) complex increases histone H4 acetylation at the H4 promoter during G1/S transition in a NPAT-dependent manner (Fig. 11.1b). Another factor, FLASH (FADD-like IL-1β-converting enzyme associated huge protein), has been implicated in the NPAT-mediated regulation of histone gene transcription [79]. Initially, FLASH was shown to associate with NPAT as well as histone gene promoters and was required for histone transcription and S phase progression [80]. Subsequently, FLASH was shown to be required for 3′-end processing of histone pre-mRNAs [81–83].
A number of transcription factors such as YY1 (Yin Yang 1) and SMAD (Small and Mothers Against Decapentaplegic) that have not been shown to interact with NPAT so far also contribute to the transcription of histone genes, although the detailed molecular mechanisms involved are largely unknown. YY1 is a ubiquitous transcription factor involved in activating histone genes upon binding specific sequences both in the coding sequences [84] and promoter regions [62] of replication-dependent histone genes . A recent study suggests that YY1 may be primarily contributing to tissue specific activation of histone genes upon differentiation [85]. The same study also reported that in embryonic stem cells , histone genes were the major targets for repression by SMAD1 and SMAD2, which are effectors of extracellular signaling by BMP (Bone Morphogenetic Protein) and TGF-β (Transforming Growth Factor-β). Most surprisingly, this study also finds E2F1 and E2F4 transcription factors to be highly enriched on nearly all replication-dependent histone genes, which is contrary to the general belief that most histone genes are not regulated by the E2F family of proteins [86]. Additional studies are required to determine the molecular mechanisms involved in the regulation of histone genes by these transcription regulators.
Wee1 is a conserved nuclear tyrosine kinase and known to negatively regulate the activity of Cdk1 (Cdc28 in yeast) in the Cyclin B/Cdk1 complex by phosphorylating tyrosine 15 of Cdk1 and inhibiting it throughout S phase, thereby preventing cells from entering mitosis until DNA replication is completed [87, 88]. Recently, Wee1 has been shown to be playing an important role in shutting off histone gene transcription at the end of S phase [89]. Wee1 phosphorylates histone H2B at tyrosine 37 in the nucleosomes found upstream of the major histone gene cluster HIST1 and suppresses histone transcription in late S phase [89]. Inhibition of the WEE1 kinase results in the loss of H2B tyrosine 37 phosphorylation (pH2B Y37) and an increase in histone transcript levels. The nucleosomes upstream of the HIST1 cluster containing pH2B Y37 may hinder the recruitment of NPAT and thereby prevent the activation of histone gene transcription (Fig. 11.1a) [89]. Additionally pH2B Y37 has been shown to recruit HIRA to the histone gene cluster, where it may help to enforce a repressive chromatin structure [89]. This mode of transcriptional repression seems to be conserved as pH2B Y37 phosphorylation is enriched on the yeast histone promoters containing the negative regulatory (NEG) element as well (Fig. 11.1a) [89]. Thus, Wee1 couples the completion of DNA replication with the termination of histone synthesis at the end of S phase, thereby ensuring a proper histone–DNA stoichiometry before the cell proceeds to mitosis (Fig. 11.1b).
Taken together, all the available evidence suggests that NPAT functions as a key global regulator of coordinated transcriptional activation of multiple core histone subtypes during the G1/S phase transition and links the regulation of histone gene expression to the cell cycle machinery . In addition to histone gene expression, NPAT has been shown to play a critical role in S phase entry [70, 90, 91]. NPAT deletion cells do not enter S phase despite high CDK activity, demonstrating that it has a CDK-independent role in cell cycle progression. Therefore, while Cyclin E/Cdk2 is at the top of the cascade which leads to both the initiation of DNA replication and histone synthesis in mammalian cells, NPAT may serve as the crucial link that couples histone gene activation and DNA replication. On the other hand, the CDK regulator Wee1 coordinates the shutdown of histone gene transcription with the completion of replication at the end of S phase.
Linker Histone H1 and DNA Replication
There are at least three major groups of H1-encoding genes in vertebrates—cleavage stage H1, replication-dependent H1, and differentiation-specific H1. The promoter of each group consists of a specific combination of regulatory elements that are evolutionarily well conserved.
Among invertebrates, the best-studied species is the sea urchin, where these different types of H1-encoding genes have been described and their promoter functionally dissected [92–94]. The replication-dependent histone promoter contains a CAAT box upstream from the TATA box, and is followed by a GC-rich region and the characteristic H1 box (also named AC box). In addition, there is a near perfect inverted repeat of the H1/AC box named UCE (Upstream Conserved Element) that was first found in the differentiation-associated H10 promoter in Xenopus. This element was also found later in vertebrate H1 promoters and named the TG box [95]. The role of these regulatory elements in the expression of replication-dependent histone H1 has been extensively studied [94]. The first clue regarding the link between the cell cycle and the expression of H1s came from the work of van Wijnen and colleagues showing the involvement of the HiNF-D complex in the control of the expression of several S phase histones, including H1 [96]. It is likely that H1 and core histone genes become activated by the same S phase signaling initiated by the CyclinE/Cdk2 complex [32]. However, H1 is transcribed throughout S phase and core histones are only transcribed in a short pulse during early S phase. Indeed, work from the Tijan laboratory using single-cell imaging in Drosophila suggests that alternative Preinitiation Complex (PIC) subunits are used for H1 transcription initiation [97].
Phosphorylation is an important cell cycle dependent modification for histone H1. A low level of H1 phosphorylation can be detected in G1 which increases steadily through S phase until it reaches its maximum in mitotic cells. S phase histone H1 exist in both unphosphorylated and hypo-phosphorylated forms and there is evidence that H1 phosphorylation is important for the process of DNA replication [98–100]. Replicating DNA and phosphorylated H1 colocalize in vivo [101], suggesting that this H1 phosphorylation may promote DNA decondensation to facilitate DNA replication. The effects of histone H1 on DNA replication has been studied directly using Xenopus egg extracts. Lu et al showed that DNA replication is inhibited to equal extent in extracts supplemented with either of the somatic variants H1c or H10 [102]. Additionally, somatic H1s inhibit replication initiation by limiting the assembly of pre-replication complexes on sperm chromatin [103]. Together, these studies suggest that on one hand the interaction of preexisting histone H1 may be weakened via phosphorylation to facilitate replication fork progression, while on the other hand the deposition of histone H1 on newly synthesized DNA may contribute to block replication licensing and prevent re-replication.
The Role of Posttranscriptional Regulation in Coordinating Histone Synthesis and DNA Replication during S Phase in Mammalian Cells
Since the half-life of the mammalian histone transcript is generally much greater compared to that in yeast, it is important to consider the contribution of posttranscriptional regulation in coordinating histone and DNA synthesis. Mammalian histone mRNAs are unique as they do not end in a polyA tail but instead have a conserved stem loop at their 3′ end that is bound by the Stem Loop Binding Protein (SLBP) [104]. SLPB is involved in the histone pre-mRNA processing and its degradation at the end of S phase (Fig. 11.1b). SLBP is subjected to cell cycle regulation and is stable only in S phase. Accumulation of SLBP before the beginning of S phase is crucial to allow the accumulation of histone mRNAs necessary for histone protein synthesis. At the end of S phase, SLBP needs to be degraded to shut off histone synthesis (Fig. 11.1b). The degradation of SLBP is initiated by Cyclin A/Cdk1 mediated phosphorylation of SLBP on Threonine 61 (T61) which is followed by Casein Kinase 2 (CK2) mediated phosphorylation of T60 [105]. Both sites need to be phosphorylated for subsequent SLBP degradation via the ubiquitin–proteasome pathway, although the ubiquitin ligase responsible for its degradation is not known.
Recent work from the Marzluff laboratory suggests that the major S phase transcription factor, E2F1 is regulated in parallel with SLBP, possibly by Cyclin A/Cdk1 (Fig. 11.1b) [106]. In HeLa cells, E2F1 is cell cycle regulated and the level of E2F1 protein increases as cells enter S phase and decreases at the end of S phase just like SLBP. Further, deletion of the amino terminus of E2F1 that removes the Cyclin A binding site results in the stabilization of E2F1 protein at the end of S phase. A mutant E2F1 carrying a deletion of the C-terminal residues 300–379 was also not degraded at the end of S phase, suggesting that two regions are needed for degradation of E2F1 protein. E2F1 is a critical transcription factor that regulates the transcription of many genes involved in DNA replication including the MCM (Mini Chromosome Maintenance) and ORC (Origin Recognition Complex) proteins, components of the replication apparatus such as RFC (Replication Factor C), PCNA (Proliferating Cell Nuclear Antigen), and RPA (Replication Protein A) to name a few [107]. Further, genes encoding enzymes involved in deoxyribonucleotide metabolism also require the E2F1 transcription factor. At the beginning of S phase, members of the retinoblastoma tumor suppressor pRb family (pRb, p130, and p107) are phosphorylated by Cyclin E/Cdk2 and this results in their dissociation from the transcription factor E2F1 and consequently in the upregulation of the genes involved in DNA replication [108]. At the end of S phase, E2F1 is very likely to be inactivated along with SLBP by Cyclin A/Cdk1, which also phosphorylates and inactivates proteins required for DNA replication such as ORC1, FEN1, and the CDP (CCAAT Displacement Protein) transcription factor that is needed for the expression of the DNA polymerase alpha gene [109, 110]. Thus, Cyclin A/Cdk1 may signal the end of S phase by both inactivating pre-replication complexes to prevent re-replication and simultaneously inhibiting histone synthesis by degrading SLBP.
Coupling of DNA Synthesis and Histone Synthesis upon Replication Inhibition or DNA Damage in S Phase
The previous sections have focused on how the activation of DNA and histone synthesis by Cyclin/Cdk2 activity ensures the coordination between them. This coupling is maintained throughout S phase and as such the amount of free histones is very small. This is not surprising given the known effects of histone imbalance on the fidelity of diverse nuclear processes and genomic stability [8, 9, 111, 112]. The close coordination of histone and DNA synthesis is exemplified in the rapid and concerted downregulation of histone mRNAs in response to DNA replication inhibiting drugs such as hydroxyurea (HU) [14]. This evolutionarily conserved response reflects the existence of S phase controls which ensure that the rate of histone production exactly matches the rate of their incorporation into chromatin. In mammalian cells, the DNA damage response (DDR) kinase ATM (Ataxia Telangiectasia Mutated) is directly involved in the activation of mechanisms that ultimately lead to the repression of histone expression in ionizing radiation (IR) treated cells [113]. The DDR activates a checkpoint when a cell senses DNA damage and the checkpoint response leads to a sequential activation of kinases that slow down the cell cycle to allow time for repair [114]. Following activation of ATM, its downstream targets p53 and p21 are consequently upregulated. p21 blocks the activation of the key regulator Cyclin E/Cdk2 complex which leads the dephosphorylation of NPAT. Unphosphorylated NPAT can no longer localize to the histone clusters in the HLB to activate histone gene transcription [113]. In fact, both NPAT and FLASH are degraded upon DNA damage mediated by UV-C irradiation [115]. This leads to the disruption of HLBs and the subsequent drop in histone transcription may contribute to the cell cycle arrest. Histone mRNAs are also rapidly degraded when DNA replication is inhibited with drugs such as hydroxyurea during S phase. The degradation is induced by the addition of untemplated uridines at the 3′ end of histone transcripts by terminal uridyl transferases [5, 116]. Such 3′ oligouridylated transcripts are preferentially recognized and bound by the hetero-heptameric LSM1–7 complex. The LSM1–7 complex then triggers the degradation of histone mRNAs through the interaction with Eri1, a 3′–5′ exoribonuclease and the RNA helicase Upf1. They all contribute to the subsequent histone mRNA degradation through the 3′ stem loop [117]. Although the mechanism of histone mRNA degradation is now known in significant detail, the important question of how the stalling of DNA replication in the nucleus is signaled to the cytoplasmic histone mRNA degradation machinery is still not clear. An attractive hypothesis is that changes in Upf1 protein phosphorylation in response to DNA replication stress modulate histone mRNA stability. Upf1 is a direct target of the phosphatidylinositol-3 (PI-3) kinases ATR (ATM and Rad3 related) and hSMG-1 [118].
In S. cerevisiae, the Hir complex plays a major role in the DNA damage induced transcriptional repression of histone genes. In addition, other histone chaperones such as Asf1 and Rtt106 and the chromatin remodeler Rsc are also implicated in the formation of a repressive chromatin structure at the histone promoters to block transcription [29]. Our laboratory has obtained evidence suggesting that the DDR kinases trigger the downregulation of histone mRNAs by targeting the Hir-complex (Paik et al., unpublished data). Interestingly, the yeast DDR kinase Rad53 (homolog of the human tumor suppressor CHK2) targets excess histone proteins for degradation via the ubiquitin–proteasome pathway both at the end of a normal S phase, as well as upon DNA damage or replication arrest in S phase, thereby contributing to genome stability [8, 111].
Recent evidence suggests that histone mRNAs in S. cerevisiae are also posttranscriptionally regulated and Herrero and Moreno have reported the importance of Lsm1 (as part of the yeast Lsm1–7-Pat1 complex) in histone mRNA degradation [119]. Lsm1 mutants accumulate high levels of histone mRNAs and are unable to degrade them following HU treatment. How the Lsm1–7 complex recognizes the histone mRNAs for degradation is not clear as yeast histone transcripts are not found to be uridylated. The Lsm1–7-Pat1 complex has been found to bind preferentially to U-tracts carrying mRNAs in humans and to oligoadenylated over polyadenylated mRNAs in yeast [116]. It has been reported that the average length of the poly A tail of yeast histone H2B mRNA is quite short compared to other transcripts. Further, the H2B poly A tail length varies during the cell cycle and shortens as cells progress from G1 to S phase [120]. Hence, it is attractive to speculate that the Lsm1–7-Pat1 complex may recognize histone mRNAs based on the length of their poly A tails and selectively degrade them. Future research should unravel the relative contributions and the molecular details of how the Lsm1–7-Pat1 complex and the TRAMP complex regulate histone mRNAs in yeast.
Is MCM2 a Sensor of Histone Supply and Demand at the Replication Fork?
To maintain a proper histone–DNA stoichiometry, the amount of histones assembled onto chromatin is exactly matched with the rate of ongoing DNA replication. The assembly of chromatin is facilitated by the interaction of certain components of the DNA replication machinery with chromatin assembly factors or histone chaperones which mediate the deposition of histones onto the newly replicated DNA. Accordingly, the pace of DNA replication is tightly coupled to chromatin assembly. This is illustrated by the fact that depletion of chromatin assembly factors such as ASF1 and CAF-1 in mammalian cells [121], as well as diminished histone supply due to knockdown of SLBP result in inhibition of replication [80, 122]. A study from the Groth laboratory suggests that replication fork progression may be coupled to nucleosome assembly through a feedback mechanism from CAF-1 to the MCM replicative helicase and/or the unloading of PCNA from newly synthesized DNA upon nucleosome assembly [123].
There is evidence that MCMs might play a role in sensing histone supply and demand at the replication fork. The amino terminus of MCM2 has been shown to interact with several chromatin proteins [124–126]. Histone H3 binds to the amino-terminal domain of MCM2 with high affinity and can likewise interact with the replicative helicase large T antigen encoded by the SV40 DNA tumor virus. In human cells, the histone chaperone ASF1 forms a complex with MCM2–7 [127], and a fraction of ASF1 colocalizes with MCM2 on chromatin. This interaction is bridged by an H3–H4 dimer, indicating that MCM2–7 loaded onto chromatin can bind non-nucleosomal histone H3–H4 dimers [126]. Histones in complex with ASF1 carry modifications that are typical of new histones, but chromatin-specific marks that would be present on parental histones can also be detected in association with MCM2 [126]. These interactions suggest that MCM2 may be ideally placed to sense the rate of replication fork progression, and in response to replication inhibition, it can potentially detect the accumulation of histones and transduce this signal to the histone synthesis machinery which can then be downregulated accordingly.
Do Senescent Cells Undergo Coordinated Loss of Replicative Potential and Histone Synthesis?
Replicative senescence is defined as an irreversible cell cycle arrest typically in G1 phase of the cell cycle. It is triggered by a variety of endogenous and exogenous cellular insults including eroded telomeres, DNA damage, oxidative stress, and/or oncogene activation [128]. Senescent cells show striking changes in gene expression—two cell cycle inhibitors p21 and p16 are usually upregulated, whereas genes that encode proteins to facilitate cell cycle progression (replication-dependent histones, c-FOS, Cyclin A, Cyclin B, and PCNA) are incorporated into transcriptionally silent heterochromatin [129, 130]. The molecular basis of the G1 arrest is thought to be the result of accumulation of the Cdk inhibitors p21 and p16 that block the inactivating phosphorylation of pRb. In the absence of inactivating Cdk phosphorylation, the Rb family of proteins (Rb, p107, and p130) binds E2F transcription factors and blocks S-phase entry [131, 132]. Many of the senescence associated Rb-specific targets are DNA replication factors as well as Cyclin E1 that induces Cdk2 activity and further phosphorylates and inactivates Rb family members, among others [133]. Interestingly, Cyclin E1 is also believed to stimulate the formation of the pre-replication complexes through the recruitment of MCMs to DNA replication origins that are Cdk-independent [134]. Thus, in senescent cells Rb may inhibit replication by repressing the genes associated with DNA replication—including Cyclin E1 which participates in pre-RC formation.
An additional consequence of Cyclin E1 inhibition in senescent cells is that the synthesis of replication-dependent histones will be inhibited as the transcriptional co-activator NPAT is not phosphorylated and fails to localize to the HLBs where histone gene transcription takes place. The Karlseder laboratory has shown that chronic DNA damage signals caused by telomere shortening decrease new histone synthesis and reduce expression of SLBP and histone chaperones ASF1 and CAF1 during replicative aging [135]. This downregulation of histone synthesis is independent of p53 and pRb, suggesting that the histone synthesis is posttranscriptionally regulated.
Senescence is often characterized by domains of facultative heterochromatin, called Senescence-Associated Heterochromatin Foci (SAHF) , which repress expression of proliferation-promoting genes such as E2F target genes. The formation of these SAHF is dependent on the p16-pRb pathway and requires several days to develop [136]. During this time, a complex of histone chaperones, comprising HIRA/UBN1/CABIN and ASF1a and HP1 transiently interacts and drives the formation of the histone variant macroH2A-containing SAHF and senescence-associated cell cycle exit. At the end of this process, each SAHF contains portions of a single condensed chromosome in which the linker histone H1 is replaced by HP1 and enriched for macroH2A [137, 138]. MacroH2A containing chromatin is resistant to ATP-dependent chromatin remodeling proteins and binding of transcription factors [139].
HIRA/UBN1/CABIN is a functional homolog of the yeast Hir complex and its function in human cells is mostly associated with chromatin assembly of replication-independent histones. However, several studies suggest that this complex may also play an important role in histone regulation. Overexpression of HIRA is able to repress histone gene transcription in human cells and block S-phase progression [42]. Cyclin E/Cdk2 and Cyclin A-Cdk2 can phosphorylate HIRA and this phosphorylation in inhibited by the cyclin inhibitor p21 [121, 140]. Thus, HIRA could act as a repressor for histone gene expression and at the same time as a repressor of genes needed for DNA replication in cells undergoing senescence.
References
Richmond TJ, Davey CA. The structure of DNA in the nucleosome core. Nature. 2003;423(6936):145–50.
Thoma F, Koller T, Klug A. Involvement of histone H1 in the organization of the nucleosome and of the salt-dependent superstructures of chromatin. J Cell Biol. 1979;83(2 Pt 1):403–27. Pubmed Central PMCID: 2111545.
Sogo JM, Stahl H, Koller T, Knippers R. Structure of replicating simian virus 40 minichromosomes. The replication fork, core histone segregation and terminal structures. J Mol Biol. 1986;189(1):189–204.
Lycan DE, Osley MA, Hereford LM. Role of transcriptional and posttranscriptional regulation in expression of histone genes in Saccharomyces cerevisiae. Mol Cell Biol. 1987;7(2):614–21. Pubmed Central PMCID: 365116.
Kaygun H, Marzluff WF. Regulated degradation of replication-dependent histone mRNAs requires both ATR and Upf1. Nat Struct Mol Biol. 2005;12(9):794–800.
Dominski Z, Yang XC, Kaygun H, Dadlez M, Marzluff WF. A 3′ exonuclease that specifically interacts with the 3′ end of histone mRNA. Mol Cell. 2003;12(2):295–305.
Sittman DB, Graves RA, Marzluff WF. Histone mRNA concentrations are regulated at the level of transcription and mRNA degradation. Proc Natl Acad Sci U S A. 1983;80(7):1849–53. Pubmed Central PMCID: 393707.
Gunjan A, Verreault A. A Rad53 kinase-dependent surveillance mechanism that regulates histone protein levels in S. cerevisiae. Cell. 2003;115(5):537–49.
Meeks-Wagner D, Hartwell LH. Normal stoichiometry of histone dimer sets is necessary for high fidelity of mitotic chromosome transmission. Cell. 1986;44(1):43–52.
Prado F, Aguilera A. Impairment of replication fork progression mediates RNA polII transcription-associated recombination. EMBO J. 2005;24(6):1267–76. Pubmed Central PMCID: 556405.
Prado F, Cortes-Ledesma F, Aguilera A. The absence of the yeast chromatin assembly factor Asf1 increases genomic instability and sister chromatid exchange. EMBO Rep. 2004;5(5):497–502. Pubmed Central PMCID: 1299049.
Myung K, Pennaneach V, Kats ES, Kolodner RD. Saccharomyces cerevisiae chromatin-assembly factors that act during DNA replication function in the maintenance of genome stability. Proc Natl Acad Sci U S A. 2003;100(11):6640–5. Pubmed Central PMCID: 164500.
Herrick D, Parker R, Jacobson A. Identification and comparison of stable and unstable mRNAs in Saccharomyces cerevisiae. Mol Cell Biol. 1990;10(5):2269–84. Pubmed Central PMCID: 360574.
Osley MA. The regulation of histone synthesis in the cell cycle. Annu Rev Biochem. 1991;60:827–61.
Eriksson PR, Ganguli D, Nagarajavel V, Clark DJ. Regulation of histone gene expression in budding yeast. Genetics. 2012;191(1):7–20. Pubmed Central PMCID: 3338271.
Hess D, Liu B, Roan NR, Sternglanz R, Winston F. Spt10-dependent transcriptional activation in Saccharomyces cerevisiae requires both the Spt10 acetyltransferase domain and Spt21. Mol Cell Biol. 2004;24(1):135–43. Pubmed Central PMCID: 303362.
Kurat CF, Lambert JP, Petschnigg J, Friesen H, Pawson T, Rosebrock A, et al. Cell cycle-regulated oscillator coordinates core histone gene transcription through histone acetylation. Proc Natl Acad Sci U S A. 2014;111(39):14124–9. Pubmed Central PMCID: 4191790.
Xu F, Zhang K, Grunstein M. Acetylation in histone H3 globular domain regulates gene expression in yeast. Cell. 2005;121(3):375–85.
Iyer VR, Horak CE, Scafe CS, Botstein D, Snyder M, Brown PO. Genomic binding sites of the yeast cell-cycle transcription factors SBF and MBF. Nature. 2001;409(6819):533–8.
Simon I, Barnett J, Hannett N, Harbison CT, Rinaldi NJ, Volkert TL, et al. Serial regulation of transcriptional regulators in the yeast cell cycle. Cell. 2001;106(6):697–708.
Ng HH, Robert F, Young RA, Struhl K. Genome-wide location and regulated recruitment of the RSC nucleosome-remodeling complex. Genes Dev. 2002;16(7):806–19. Pubmed Central PMCID: 186327.
Osley MA, Gould J, Kim S, Kane MY, Hereford L. Identification of sequences in a yeast histone promoter involved in periodic transcription. Cell. 1986;45(4):537–44.
Koch C, Moll T, Neuberg M, Ahorn H, Nasmyth K. A role for the transcription factors Mbp1 and Swi4 in progression from G1 to S phase. Science. 1993;261(5128):1551–7.
Spector MS, Raff A, DeSilva H, Lee K, Osley MA. Hir1p and Hir2p function as transcriptional corepressors to regulate histone gene transcription in the Saccharomyces cerevisiae cell cycle. Mol Cell Biol. 1997;17(2):545–52. Pubmed Central PMCID: 231779.
Sutton A, Bucaria J, Osley MA, Sternglanz R. Yeast ASF1 protein is required for cell cycle regulation of histone gene transcription. Genetics. 2001;158(2):587–96. Pubmed Central PMCID: 1461693.
Xu H, Kim UJ, Schuster T, Grunstein M. Identification of a new set of cell cycle-regulatory genes that regulate S-phase transcription of histone genes in Saccharomyces cerevisiae. Mol Cell Biol. 1992;12(11):5249–59. Pubmed Central PMCID: 360458.
De Koning L, Corpet A, Haber JE, Almouzni G. Histone chaperones: an escort network regulating histone traffic. Nat Struct Mol Biol. 2007;14(11):997–1007.
Green EM, Antczak AJ, Bailey AO, Franco AA, Wu KJ, Yates 3rd JR, et al. Replication-independent histone deposition by the HIR complex and Asf1. Curr Biol. 2005;15(22):2044–9. Pubmed Central PMCID: 2819815.
Fillingham J, Kainth P, Lambert JP, van Bakel H, Tsui K, Pena-Castillo L, et al. Two-color cell array screen reveals interdependent roles for histone chaperones and a chromatin boundary regulator in histone gene repression. Mol Cell. 2009;35(3):340–51.
Ferreira ME, Flaherty K, Prochasson P. The Saccharomyces cerevisiae histone chaperone Rtt106 mediates the cell cycle recruitment of SWI/SNF and RSC to the HIR-dependent histone genes. PLoS One. 2011;6(6), e21113. Pubmed Central PMCID: 3115976.
Amin AD, Dimova DK, Ferreira ME, Vishnoi N, Hancock LC, Osley MA, et al. The mitotic Clb cyclins are required to alleviate HIR-mediated repression of the yeast histone genes at the G1/S transition. Biochim Biophys Acta. 2012;1819(1):16–27. Pubmed Central PMCID: 3249481.
White JH, Green SR, Barker DG, Dumas LB, Johnston LH. The CDC8 transcript is cell cycle regulated in yeast and is expressed coordinately with CDC9 and CDC21 at a point preceding histone transcription. Exp Cell Res. 1987;171(1):223–31.
Sherwood PW, Tsang SV, Osley MA. Characterization of HIR1 and HIR2, two genes required for regulation of histone gene transcription in Saccharomyces cerevisiae. Mol Cell Biol. 1993;13(1):28–38. Pubmed Central PMCID: 358881.
Goh PY, Surana U. Cdc4, a protein required for the onset of S phase, serves an essential function during G(2)/M transition in Saccharomyces cerevisiae. Mol Cell Biol. 1999;19(8):5512–22. Pubmed Central PMCID: 84393.
Drury LS, Perkins G, Diffley JF. The Cdc4/34/53 pathway targets Cdc6p for proteolysis in budding yeast. EMBO J. 1997;16(19):5966–76. Pubmed Central PMCID: 1170227.
Feldman RM, Correll CC, Kaplan KB, Deshaies RJ. A complex of Cdc4p, Skp1p, and Cdc53p/cullin catalyzes ubiquitination of the phosphorylated CDK inhibitor Sic1p. Cell. 1997;91(2):221–30.
Mathias N, Johnson SL, Winey M, Adams AE, Goetsch L, Pringle JR, et al. Cdc53p acts in concert with Cdc4p and Cdc34p to control the G1-to-S-phase transition and identifies a conserved family of proteins. Mol Cell Biol. 1996;16(12):6634–43. Pubmed Central PMCID: 231665.
Schneider BL, Yang QH, Futcher AB. Linkage of replication to start by the Cdk inhibitor Sic1. Science. 1996;272(5261):560–2.
Schwob E, Bohm T, Mendenhall MD, Nasmyth K. The B-type cyclin kinase inhibitor p40SIC1 controls the G1 to S transition in S. cerevisiae. Cell. 1994;79(2):233–44.
Skowyra D, Craig KL, Tyers M, Elledge SJ, Harper JW. F-box proteins are receptors that recruit phosphorylated substrates to the SCF ubiquitin-ligase complex. Cell. 1997;91(2):209–19.
DeSalle LM, Pagano M. Regulation of the G1 to S transition by the ubiquitin pathway. FEBS Lett. 2001;490(3):179–89.
Hall C, Nelson DM, Ye X, Baker K, DeCaprio JA, Seeholzer S, et al. HIRA, the human homologue of yeast Hir1p and Hir2p, is a novel cyclin-cdk2 substrate whose expression blocks S-phase progression. Mol Cell Biol. 2001;21(5):1854–65. Pubmed Central PMCID: 86753.
Ubersax JA, Woodbury EL, Quang PN, Paraz M, Blethrow JD, Shah K, et al. Targets of the cyclin-dependent kinase Cdk1. Nature. 2003;425(6960):859–64.
Amon A, Surana U, Muroff I, Nasmyth K. Regulation of p34CDC28 tyrosine phosphorylation is not required for entry into mitosis in S. cerevisiae. Nature. 1992;355(6358):368–71.
Kellogg DR, Murray AW. NAP1 acts with Clb1 to perform mitotic functions and to suppress polar bud growth in budding yeast. J Cell Biol. 1995;130(3):675–85. Pubmed Central PMCID: 2120541.
Hereford L, Bromley S, Osley MA. Periodic transcription of yeast histone genes. Cell. 1982;30(1):305–10.
Campbell SG, Li Del Olmo M, Beglan P, Bond U. A sequence element downstream of the yeast HTB1 gene contributes to mRNA 3′ processing and cell cycle regulation. Mol Cell Biol. 2002;22(24):8415–25. Pubmed Central PMCID: 139887.
Canavan R, Bond U. Deletion of the nuclear exosome component RRP6 leads to continued accumulation of the histone mRNA HTB1 in S-phase of the cell cycle in Saccharomyces cerevisiae. Nucleic Acids Res. 2007;35(18):6268–79. Pubmed Central PMCID: 2094057.
Reis CC, Campbell JL. Contribution of Trf4/5 and the nuclear exosome to genome stability through regulation of histone mRNA levels in Saccharomyces cerevisiae. Genetics. 2007;175(3):993–1010. Pubmed Central PMCID: 1840065.
LaCava J, Houseley J, Saveanu C, Petfalski E, Thompson E, Jacquier A, et al. RNA degradation by the exosome is promoted by a nuclear polyadenylation complex. Cell. 2005;121(5):713–24.
Vanacova S, Wolf J, Martin G, Blank D, Dettwiler S, Friedlein A, et al. A new yeast poly(A) polymerase complex involved in RNA quality control. PLoS Biol. 2005;3(6), e189. Pubmed Central PMCID: 1079787.
Wyers F, Rougemaille M, Badis G, Rousselle JC, Dufour ME, Boulay J, et al. Cryptic pol II transcripts are degraded by a nuclear quality control pathway involving a new poly(A) polymerase. Cell. 2005;121(5):725–37.
Arigo JT, Eyler DE, Carroll KL, Corden JL. Termination of cryptic unstable transcripts is directed by yeast RNA-binding proteins Nrd1 and Nab3. Mol Cell. 2006;23(6):841–51.
Vasiljeva L, Buratowski S. Nrd1 interacts with the nuclear exosome for 3′ processing of RNA polymerase II transcripts. Mol Cell. 2006;21(2):239–48.
Harris ME, Bohni R, Schneiderman MH, Ramamurthy L, Schumperli D, Marzluff WF. Regulation of histone mRNA in the unperturbed cell cycle: evidence suggesting control at two posttranscriptional steps. Mol Cell Biol. 1991;11(5):2416–24. Pubmed Central PMCID: 359999.
Schumperli D. Cell-cycle regulation of histone gene expression. Cell. 1986;45(4):471–2.
Heintz N. The regulation of histone gene expression during the cell cycle. Biochim Biophys Acta. 1991;1088(3):327–39.
Albig W, Doenecke D. The human histone gene cluster at the D6S105 locus. Hum Genet. 1997;101(3):284–94.
Pauli U, Chrysogelos S, Stein G, Stein J, Nick H. Protein-DNA interactions in vivo upstream of a cell cycle-regulated human H4 histone gene. Science. 1987;236(4806):1308–11.
Birnbaum MJ, Wright KL, van Wijnen AJ, Ramsey-Ewing AL, Bourke MT, Last TJ, et al. Functional role for Sp1 in the transcriptional amplification of a cell cycle regulated histone H4 gene. Biochemistry. 1995;34(23):7648–58.
Guo B, Stein JL, van Wijnen AJ, Stein GS. ATF1 and CREB trans-activate a cell cycle regulated histone H4 gene at a distal nuclear matrix associated promoter element. Biochemistry. 1997;36(47):14447–55.
Last TJ, van Wijnen AJ, Birnbaum MJ, Stein GS, Stein JL. Multiple interactions of the transcription factor YY1 with human histone H4 gene regulatory elements. J Cell Biochem. 1999;72(4):507–16.
van Wijnen AJ, van Gurp MF, de Ridder MC, Tufarelli C, Last TJ, Birnbaum M, et al. CDP/cut is the DNA-binding subunit of histone gene transcription factor HiNF-D: a mechanism for gene regulation at the G1/S phase cell cycle transition point independent of transcription factor E2F. Proc Natl Acad Sci U S A. 1996;93(21):11516–21. Pubmed Central PMCID: 38089.
Vaughan PS, van der Meijden CM, Aziz F, Harada H, Taniguchi T, van Wijnen AJ, et al. Cell cycle regulation of histone H4 gene transcription requires the oncogenic factor IRF-2. J Biol Chem. 1998;273(1):194–9.
Aziz F, van Wijnen AJ, Stein JL, Stein GS. HiNF-D (CDP-cut/CDC2/cyclin A/pRB-complex) influences the timing of IRF-2-dependent cell cycle activation of human histone H4 gene transcription at the G1/S phase transition. J Cell Physiol. 1998;177(3):453–64.
Kelly TJ, Brown GW. Regulation of chromosome replication. Annu Rev Biochem. 2000;69:829–80.
Marzluff WF, Pandey NB. Multiple regulatory steps control histone mRNA concentrations. Trends Biochem Sci. 1988;13(2):49–52.
Mailand N, Diffley JF. CDKs promote DNA replication origin licensing in human cells by protecting Cdc6 from APC/C-dependent proteolysis. Cell. 2005;122(6):915–26.
Duursma A, Agami R. p53-Dependent regulation of Cdc6 protein stability controls cellular proliferation. Mol Cell Biol. 2005;25(16):6937–47. Pubmed Central PMCID: 1190229.
Zhao J, Dynlacht B, Imai T, Hori T, Harlow E. Expression of NPAT, a novel substrate of cyclin E-CDK2, promotes S-phase entry. Genes Dev. 1998;12(4):456–61. Pubmed Central PMCID: 316526.
Ma T, Van Tine BA, Wei Y, Garrett MD, Nelson D, Adams PD, et al. Cell cycle-regulated phosphorylation of p220(NPAT) by cyclin E/Cdk2 in Cajal bodies promotes histone gene transcription. Genes Dev. 2000;14(18):2298–313. Pubmed Central PMCID: 316935.
Zhao J, Kennedy BK, Lawrence BD, Barbie DA, Matera AG, Fletcher JA, et al. NPAT links cyclin E-Cdk2 to the regulation of replication-dependent histone gene transcription. Genes Dev. 2000;14(18):2283–97. Pubmed Central PMCID: 316937.
Ghule PN, Dominski Z, Yang XC, Marzluff WF, Becker KA, Harper JW, et al. Staged assembly of histone gene expression machinery at subnuclear foci in the abbreviated cell cycle of human embryonic stem cells. Proc Natl Acad Sci U S A. 2008;105(44):16964–9. Pubmed Central PMCID: 2579361.
Liu JL, Buszczak M, Gall JG. Nuclear bodies in the Drosophila germinal vesicle. Chromosome Res. 2006;14(4):465–75.
Miele A, Braastad CD, Holmes WF, Mitra P, Medina R, Xie R, et al. HiNF-P directly links the cyclin E/CDK2/p220NPAT pathway to histone H4 gene regulation at the G1/S phase cell cycle transition. Mol Cell Biol. 2005;25(14):6140–53. Pubmed Central PMCID: 1168814.
Zheng L, Roeder RG, Luo Y. S phase activation of the histone H2B promoter by OCA-S, a coactivator complex that contains GAPDH as a key component. Cell. 2003;114(2):255–66.
Roberts SB, Segil N, Heintz N. Differential phosphorylation of the transcription factor Oct1 during the cell cycle. Science. 1991;253(5023):1022–6.
DeRan M, Pulvino M, Greene E, Su C, Zhao J. Transcriptional activation of histone genes requires NPAT-dependent recruitment of TRRAP-Tip60 complex to histone promoters during the G1/S phase transition. Mol Cell Biol. 2008;28(1):435–47. Pubmed Central PMCID: 2223310.
Yang XC, Sabath I, Kunduru L, van Wijnen AJ, Marzluff WF, Dominski Z. A conserved interaction that is essential for the biogenesis of histone locus bodies. J Biol Chem. 2014;289(49):33767–82. Pubmed Central PMCID: 4256312.
Barcaroli D, Bongiorno-Borbone L, Terrinoni A, Hofmann TG, Rossi M, Knight RA, et al. FLASH is required for histone transcription and S-phase progression. Proc Natl Acad Sci U S A. 2006;103(40):14808–12. Pubmed Central PMCID: 1578501.
Burch BD, Godfrey AC, Gasdaska PY, Salzler HR, Duronio RJ, Marzluff WF, et al. Interaction between FLASH and Lsm11 is essential for histone pre-mRNA processing in vivo in Drosophila. RNA. 2011;17(6):1132–47. Pubmed Central PMCID: 3096045.
Yang XC, Xu B, Sabath I, Kunduru L, Burch BD, Marzluff WF, et al. FLASH is required for the endonucleolytic cleavage of histone pre-mRNAs but is dispensable for the 5' exonucleolytic degradation of the downstream cleavage product. Mol Cell Biol. 2011;31(7):1492–502. Pubmed Central PMCID: 3135297.
Sabath I, Skrajna A, Yang XC, Dadlez M, Marzluff WF, Dominski Z. 3′-End processing of histone pre-mRNAs in Drosophila: U7 snRNP is associated with FLASH and polyadenylation factors. RNA. 2013;19(12):1726–44. Pubmed Central PMCID: 3884669.
Eliassen KA, Baldwin A, Sikorski EM, Hurt MM. Role for a YY1-binding element in replication-dependent mouse histone gene expression. Mol Cell Biol. 1998;18(12):7106–18. Pubmed Central PMCID: 109292.
Gokhman D, Livyatan I, Sailaja BS, Melcer S, Meshorer E. Multilayered chromatin analysis reveals E2f, Smad and Zfx as transcriptional regulators of histones. Nat Struct Mol Biol. 2013;20(1):119–26.
Oswald F, Dobner T, Lipp M. The E2F transcription factor activates a replication-dependent human H2A gene in early S phase of the cell cycle. Mol Cell Biol. 1996;16(5):1889–95. Pubmed Central PMCID: 231176.
Gould KL, Nurse P. Tyrosine phosphorylation of the fission yeast cdc2+ protein kinase regulates entry into mitosis. Nature. 1989;342(6245):39–45.
Watanabe N, Broome M, Hunter T. Regulation of the human WEE1Hu CDK tyrosine 15-kinase during the cell cycle. EMBO J. 1995;14(9):1878–91. Pubmed Central PMCID: 398287.
Mahajan K, Fang B, Koomen JM, Mahajan NP. H2B Tyr37 phosphorylation suppresses expression of replication-dependent core histone genes. Nat Struct Mol Biol. 2012;19(9):930–7.
Gao G, Bracken AP, Burkard K, Pasini D, Classon M, Attwooll C, et al. NPAT expression is regulated by E2F and is essential for cell cycle progression. Mol Cell Biol. 2003;23(8):2821–33. Pubmed Central PMCID: 152552.
Ye X, Wei Y, Nalepa G, Harper JW. The cyclin E/Cdk2 substrate p220(NPAT) is required for S-phase entry, histone gene expression, and Cajal body maintenance in human somatic cells. Mol Cell Biol. 2003;23(23):8586–600. Pubmed Central PMCID: 262656.
Watanabe N, Arai H, Iwasaki J, Shiina M, Ogata K, Hunter T, et al. Cyclin-dependent kinase (CDK) phosphorylation destabilizes somatic Wee1 via multiple pathways. Proc Natl Acad Sci U S A. 2005;102(33):11663–8. Pubmed Central PMCID: 1187955.
Lai ZC, Maxson R, Childs G. Both basal and ontogenic promoter elements affect the timing and level of expression of a sea urchin H1 gene during early embryogenesis. Genes Dev. 1988;2(2):173–83.
Khochbin S, Wolffe AP. Developmentally regulated expression of linker-histone variants in vertebrates. Eur J Biochem. 1994;225(2):501–10.
Duncliffe KN, Rondahl ME, Wells JR. A H1 histone gene-specific AC-box-related element influences transcription from a major chicken H1 promoter. Gene. 1995;163(2):227–32.
van Wijnen AJ, Aziz F, Grana X, De Luca A, Desai RK, Jaarsveld K, et al. Transcription of histone H4, H3, and H1 cell cycle genes: promoter factor HiNF-D contains CDC2, cyclin A, and an RB-related protein. Proc Natl Acad Sci U S A. 1994;91(26):12882–6. Pubmed Central PMCID: 45544.
Guglielmi B, La Rochelle N, Tjian R. Gene-specific transcriptional mechanisms at the histone gene cluster revealed by single-cell imaging. Mol Cell. 2013;51(4):480–92.
Gurley LR, Walters RA, Tobey RA. Sequential phsophorylation of histone subfractions in the Chinese hamster cell cycle. J Biol Chem. 1975;250(10):3936–44.
Yasuda H, Matsumoto Y, Mita S, Marunouchi T, Yamada M. A mouse temperature-sensitive mutant defective in H1 histone phosphorylation is defective in deoxyribonucleic acid synthesis and chromosome condensation. Biochemistry. 1981;20(15):4414–9.
Halmer L, Gruss C. Effects of cell cycle dependent histone H1 phosphorylation on chromatin structure and chromatin replication. Nucleic Acids Res. 1996;24(8):1420–7. Pubmed Central PMCID: 145815.
Alexandrow MG, Hamlin JL. Chromatin decondensation in S-phase involves recruitment of Cdk2 by Cdc45 and histone H1 phosphorylation. J Cell Biol. 2005;168(6):875–86. Pubmed Central PMCID: 2171796.
Lu ZH, Sittman DB, Brown DT, Munshi R, Leno GH. Histone H1 modulates DNA replication through multiple pathways in Xenopus egg extract. J Cell Sci. 1997;110(Pt 21):2745–58.
Lu ZH, Sittman DB, Romanowski P, Leno GH. Histone H1 reduces the frequency of initiation in Xenopus egg extract by limiting the assembly of prereplication complexes on sperm chromatin. Mol Biol Cell. 1998;9(5):1163–76. Pubmed Central PMCID: 25338.
Marzluff WF, Wagner EJ, Duronio RJ. Metabolism and regulation of canonical histone mRNAs: life without a poly(A) tail. Nat Rev Genet. 2008;9(11):843–54. Pubmed Central PMCID: 2715827.
Koseoglu MM, Graves LM, Marzluff WF. Phosphorylation of threonine 61 by cyclin a/Cdk1 triggers degradation of stem-loop binding protein at the end of S phase. Mol Cell Biol. 2008;28(14):4469–79. Pubmed Central PMCID: 2447125.
Koseoglu MM, Dong J, Marzluff WF. Coordinate regulation of histone mRNA metabolism and DNA replication: cyclin A/cdk1 is involved in inactivation of histone mRNA metabolism and DNA replication at the end of S phase. Cell Cycle. 2010;9(19):3857–63. Pubmed Central PMCID: 3047749.
Bracken AP, Ciro M, Cocito A, Helin K. E2F target genes: unraveling the biology. Trends Biochem Sci. 2004;29(8):409–17.
Harbour JW, Luo RX, Dei Santi A, Postigo AA, Dean DC. Cdk phosphorylation triggers sequential intramolecular interactions that progressively block Rb functions as cells move through G1. Cell. 1999;98(6):859–69.
Truscott M, Raynal L, Premdas P, Goulet B, Leduy L, Berube G, et al. CDP/Cux stimulates transcription from the DNA polymerase alpha gene promoter. Mol Cell Biol. 2003;23(8):3013–28. Pubmed Central PMCID: 152546.
Henneke G, Koundrioukoff S, Hubscher U. Phosphorylation of human Fen1 by cyclin-dependent kinase modulates its role in replication fork regulation. Oncogene. 2003;22(28):4301–13.
Singh RK, Kabbaj MH, Paik J, Gunjan A. Histone levels are regulated by phosphorylation and ubiquitylation-dependent proteolysis. Nat Cell Biol. 2009;11(8):925–33. Pubmed Central PMCID: 2720428.
Singh RK, Liang D, Gajjalaiahvari UR, Kabbaj MH, Paik J, Gunjan A. Excess histone levels mediate cytotoxicity via multiple mechanisms. Cell Cycle. 2010;9(20):4236–44. Pubmed Central PMCID: 3055206.
Su C, Gao G, Schneider S, Helt C, Weiss C, O'Reilly MA, et al. DNA damage induces downregulation of histone gene expression through the G1 checkpoint pathway. EMBO J. 2004;23(5):1133–43. Pubmed Central PMCID: 380976.
Weinert TA, Kiser GL, Hartwell LH. Mitotic checkpoint genes in budding yeast and the dependence of mitosis on DNA replication and repair. Genes Dev. 1994;8(6):652–65.
Bongiorno-Borbone L, De Cola A, Barcaroli D, Knight RA, Di Ilio C, Melino G, et al. FLASH degradation in response to UV-C results in histone locus bodies disruption and cell-cycle arrest. Oncogene. 2010;29(6):802–10.
Mullen TE, Marzluff WF. Degradation of histone mRNA requires oligouridylation followed by decapping and simultaneous degradation of the mRNA both 5′ to 3′ and 3′ to 5′. Genes Dev. 2008;22(1):50–65. Pubmed Central PMCID: 2151014.
Lyons SM, Ricciardi AS, Guo AY, Kambach C, Marzluff WF. The C-terminal extension of Lsm4 interacts directly with the 3′ end of the histone mRNP and is required for efficient histone mRNA degradation. RNA. 2014;20(1):88–102. Pubmed Central PMCID: 3866647.
Azzalin CM, Lingner J. The human RNA surveillance factor UPF1 is required for S phase progression and genome stability. Curr Biol. 2006;16(4):433–9.
Herrero AB, Moreno S. Lsm1 promotes genomic stability by controlling histone mRNA decay. EMBO J. 2011;30(10):2008–18. Pubmed Central PMCID: 3098488.
Beggs S, James TC, Bond U. The PolyA tail length of yeast histone mRNAs varies during the cell cycle and is influenced by Sen1p and Rrp6p. Nucleic Acids Res. 2012;40(6):2700–11. Pubmed Central PMCID: 3315300.
Alabert C, Groth A. Chromatin replication and epigenome maintenance. Nat Rev Mol Cell Biol. 2012;13(3):153–67.
Zhao X, McKillop-Smith S, Muller B. The human histone gene expression regulator HBP/SLBP is required for histone and DNA synthesis, cell cycle progression and cell proliferation in mitotic cells. J Cell Sci. 2004;117(Pt 25):6043–51.
Mejlvang J, Feng Y, Alabert C, Neelsen KJ, Jasencakova Z, Zhao X, et al. New histone supply regulates replication fork speed and PCNA unloading. J Cell Biol. 2014;204(1):29–43. Pubmed Central PMCID: 3882791.
Holland L, Gauthier L, Bell-Rogers P, Yankulov K. Distinct parts of minichromosome maintenance protein 2 associate with histone H3/H4 and RNA polymerase II holoenzyme. Eur J Biochem. 2002;269(21):5192–202.
Latreille D, Bluy L, Benkirane M, Kiernan RE. Identification of histone 3 variant 2 interacting factors. Nucleic Acids Res. 2014;42(6):3542–50. Pubmed Central PMCID: 3973350.
Richet N, Liu D, Legrand P, Velours C, Corpet A, Gaubert A, et al. Structural insight into how the human helicase subunit MCM2 may act as a histone chaperone together with ASF1 at the replication fork. Nucleic Acids Res. 2015;43(3):1905–17. Pubmed Central PMCID: 4330383.
Groth A, Corpet A, Cook AJ, Roche D, Bartek J, Lukas J, et al. Regulation of replication fork progression through histone supply and demand. Science. 2007;318(5858):1928–31.
Campisi J. d'Adda di Fagagna F. Cellular senescence: when bad things happen to good cells. Nat Rev Mol Cell Biol. 2007;8(9):729–40.
Campisi J. Cellular senescence as a tumor-suppressor mechanism. Trends Cell Biol. 2001;11(11):S27–31.
Braig M, Schmitt CA. Oncogene-induced senescence: putting the brakes on tumor development. Cancer Res. 2006;66(6):2881–4.
Pang JH, Chen KY. Global change of gene expression at late G1/S boundary may occur in human IMR-90 diploid fibroblasts during senescence. J Cell Physiol. 1994;160(3):531–8.
Narita M, Nunez S, Heard E, Narita M, Lin AW, Hearn SA, et al. Rb-mediated heterochromatin formation and silencing of E2F target genes during cellular senescence. Cell. 2003;113(6):703–16.
Sherr CJ, McCormick F. The RB and p53 pathways in cancer. Cancer Cell. 2002;2(2):103–12.
Geng Y, Lee YM, Welcker M, Swanger J, Zagozdzon A, Winer JD, et al. Kinase-independent function of cyclin E. Mol Cell. 2007;25(1):127–39.
O'Sullivan RJ, Kubicek S, Schreiber SL, Karlseder J. Reduced histone biosynthesis and chromatin changes arising from a damage signal at telomeres. Nat Struct Mol Biol. 2010;17(10):1218–25. Pubmed Central PMCID: 2951278.
Zhang R, Poustovoitov MV, Ye X, Santos HA, Chen W, Daganzo SM, et al. Formation of MacroH2A-containing senescence-associated heterochromatin foci and senescence driven by ASF1a and HIRA. Dev Cell. 2005;8(1):19–30.
Zhang R, Chen W, Adams PD. Molecular dissection of formation of senescence-associated heterochromatin foci. Mol Cell Biol. 2007;27(6):2343–58. Pubmed Central PMCID: 1820509.
Funayama R, Saito M, Tanobe H, Ishikawa F. Loss of linker histone H1 in cellular senescence. J Cell Biol. 2006;175(6):869–80. Pubmed Central PMCID: 2064697.
Angelov D, Molla A, Perche PY, Hans F, Cote J, Khochbin S, et al. The histone variant macroH2A interferes with transcription factor binding and SWI/SNF nucleosome remodeling. Mol Cell. 2003;11(4):1033–41.
Dollard C, Ricupero-Hovasse SL, Natsoulis G, Boeke JD, Winston F. SPT10 and SPT21 are required for transcription of particular histone genes in Saccharomyces cerevisiae. Mol Cell Biol. 1994;14(8):5223–8. Pubmed Central PMCID: 359041.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Paik, J., Giovinazzi, S., Gunjan, A. (2016). Coordination of DNA Replication and Histone Synthesis During S Phase. In: Kaplan, D. (eds) The Initiation of DNA Replication in Eukaryotes. Springer, Cham. https://doi.org/10.1007/978-3-319-24696-3_11
Download citation
DOI: https://doi.org/10.1007/978-3-319-24696-3_11
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-24694-9
Online ISBN: 978-3-319-24696-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)