Abstract
The sweetpotato whitefly, Bemisia tabaci, is a devastating cosmopolitan insect pest that inflicts serious damage by direct feeding on plants, secreting honeydew, and vectoring more than 100 plant viruses that belong to different virus genera. The interactions between the whitefly and plant viruses, plants, and environmental factors have been extensively studied. In recent years more than 100,000 expressed sequences tags (ESTs) from the whitefly have been made available to the scientific community by several mass sequencing projects, and a genome sequencing project is underway. Tools for functional analysis of gene expression are being developed for studies in the whitefly. Combining EST and genomic sequences with functional analysis will pave the way for addressing urgent issues in whitefly research and developing better strategies for whitefly control.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Plant Virus
- Tomato Yellow Leaf Curl Virus
- Amplify Fragment Length Polymorphism
- Tomato Yellow Leaf
- African Cassava Mosaic Virus
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
2.1 Introduction
For decades, agriculture has been the main source of food and other useful products for humans and livestock. The world population is expected to grow by one-third from the present by 2050 (www.fao.org) leading to an increase in demand for agricultural products to feed growing populations worldwide. Production of agricultural crops is at risk due to the incidence of herbivores and disease-causing pathogens. Herbivores, mainly insect pests, are undoubtedly the most important competitors for food, fiber, and other natural products. Beyond their economic importance, they have a direct impact on agricultural food production by chewing, sucking, and boring plant parts and, most importantly, by spreading devastating plant pathogens, particularly plant viruses. Many important plant viruses depend on insect vectors which mainly belong to the orders Hemiptera, Thysanoptera, and Coleoptera, which transmit a great diversity of plant viruses across the tropical and subtropical regions of the world. Among them, phloem-feeding insects (aphids, whiteflies, plant, and leafhoppers) are the most common insect vectors that transmit numerous viruses and cause significant losses to food crops of socioeconomic importance.
In the last 20 years of the twentieth century, vector biology research switched from an understanding of the general biology of virus-vector relationships to a more deep understanding of the viral and vector proteins and components involved in the transmission process. However, the insect vector component was largely ignored due to a lack of genetic and genomic resources. Developments in mass sequencing projects such as expressed sequence tags (ESTs), together with genome annotation and functional gene discovery, are greatly advancing our understanding of the transmission process of viruses by their vectors and the components required to facilitate virus transmission. The emergence of massive sequencing technologies such as 454 pyrosequencing and the Illumina sequencing platform has been widely used to understand the underlying molecular mechanisms [1] involved in virus transmission, mating behavior, and genomes of many hemipterans and the whitefly, Bemisia tabaci, its endosymbionts, and the interactions of the insect with the environment. Functional genomic projects of the whitefly will facilitate an understanding of not only its interactions with viruses but also its resistance to insecticides, its developmental patterns, and its interactions with plants and other microorganisms. This chapter reviews the recent advances in the genomics of B. tabaci and the relevance of these advancements to our understanding of its interactions with various factors.
2.2 Status of B. tabaci as a Global Agricultural Pest
The whitefly, B. tabaci (Gennadius) (Hemiptera: Aleyrodidae), is a phloem-feeding insect that inflicts a serious worldwide threat by direct feeding on plants and by the transmission of plant viruses [2–4] (Fig. 2.1). B. tabaci was first described in 1889 as a tobacco pest in Greece and named Aleyrodes tabaci [5]; later on it was described as A. inconspicua Quaintance in 1900 [6] and B. tabaci in 1957 [7]. Comprehensive recent reviews about the history of B. tabaci [8] and its cryptic species status [9–13] are available.
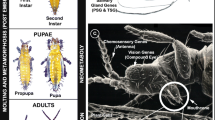
Direct and indirect plant damage caused by Bemisia tabaci. (a) B. tabaci adult. (b) Representative damage caused by B. tabaci feeding showing an infested basil leaf with B. tabaci nymphs. (c) A basil leaf showing sooty mold growth after B. tabaci infestation and massive secretion of honeydew. (d) An uninfested watermelon field. (e) Another watermelon field that was infested by two B. tabaci-transmitted begomoviruses squash leaf curl virus (SLCV) and watermelon chlorotic stunt virus (WmCSV)
B. tabaci adults are about 1–2 mm long and have opaque-shaped wings covered with a whitish powder or wax, leading to the common name of whiteflies. B. tabaci was first reported to be a serious insect pest in the late 1920s in Northern India [14, 15] and is now globally distributed, in the USA, Africa, the Middle East, Europe, the Orient, Russia, Southeast Asia, and South America [3], except in Antarctica [16]. It is a highly polyphagous insect that feeds on over 700 plant species from 86 families, including a large number of agriculturally and industrially important crops [9, 17, 18]. Whiteflies damage plants in various ways. The delicate mouthparts of the whitefly adults and nymphal stages cause direct feeding damage by weakening plants (Fig. 2.1). They also cause indirect damage due to the secretion of honeydew that causes sooty mold growth which interferes with photosynthesis and makes the fruits become unsightly.
The whitefly’s major impact on agriculture is due to the fact that this insect serves as a vector of more than 110 plant viruses [9, 16–19], many of which have great impact on plants and agricultural production. Estimates of their destructive reach extend to 20 million hectares of crops and 15 million farmers [20]. Next to aphids, whiteflies are of greater importance as vectors of the most devastating plant virus groups, including Begomovirus, Crinivirus, Closterovirus, Carlavirus, and Torradovirus [17, 21]. Among whitefly-transmitted viruses, 90 % belong to the Begomovirus genera [17], which include approximately 200 species, and have emerged as the most threatening group of plant viruses globally during the past two decades as reported from dicotyledonous host-causing diseases of economic importance.
B. tabaci is considered as a cryptic species complex [11]. Individual cryptic species within the complex differ in their adaptability to hosts, in their resistance to chemicals, and importantly in their ability to transmit begomoviruses [22–26]. Exotic whitefly biotypes or cryptic species transmit Old and New World begomoviruses in many places of the world resulting from changes in environmental conditions and agricultural practices. Studies on whitefly biotypes/cryptic species and the begomoviruses they transmit worldwide reveal that grouping of begomoviruses based on CP (coat protein) sequences parallels grouping of biotypes/genotypes so that both begomoviruses and vector are grouped in similar patterns according to their geographic origin. As different members of the species complex are morphologically indistinguishable, various molecular methods have been applied over the past two decades to delimit the members of the B. tabaci species complex.
2.3 Biotypes or Cryptic Species of B. tabaci: Definitions and Distribution
B. tabaci has numerous biotypes which transmit a large number of viruses that infect many important agricultural plants, causing major economic impacts [27]. As early as the 1950s, biotypes were proposed to characterize the morphologically indistinguishable populations of B. tabaci on the basis of host range, host plant adaptability, and plant virus transmission capabilities [28–30]. During the 1980s, the A biotype became a serious problem in cotton and cucurbits in the Southwestern USA and Mexico [3]. In 1991, the introduced B biotype displaced the A biotype in the Southwestern USA [3]. In the same year, Perring et al. [31] suggested that A and B biotypes were separate species. A newly evolved B biotype, commonly referred to as the silver leaf whitefly or poinsettia strain, has a very wide host range, which has contributed to the spread of geminiviruses to new hosts [32] and has been documented as a separate species named B. argentifolii [33]. Using allozymes and the random amplified polymorphic cDNA-polymerase chain reaction (RAPD-PCR), Perring et al. [34] and Gawel and Bartlett [35] further showed consistent differences between A and B biotypes.
As the different members of the B. tabaci species complex are morphologically indistinguishable, various molecular methods have been applied over the past two decades to delimit the members of this species complex. The most popular techniques and the types of DNA markers used to study B. tabaci are sequence characterized amplified regions (SCAR) [36–41], cleaved amplified polymorphic sequences or restriction fragment length polymorphisms (CAPS/RFLP) [37, 42], amplified fragment length polymorphisms (AFLP) [43], 16S ribosomal RNA [44], mitochondrial cytochrome oxidase 1 (mtCO1) [10–12, 45–49], nuclear ribosomal internal transcribe spacer 1 (ITS1) [50–54], and microsatellites [55–65]. Among them, mtCOI has several advantages over other approaches and has been extensively used [11].
Using CO1-based Bayesian phylogenetic analysis and sequence divergence, Dinsdale et al. [10] and De Barro et al. [11] proposed a speciation system on the basis of a demarcation criterion of a 3.5 % divergence threshold. Recently, Lee et al. [49] observed that a 4.0 % genetic boundary was more realistic than 3.5 % in distinguishing B. tabaci species. Following the above criteria, 37 morphologically indistinguishable species (Africa, Asia I, Asia II 1, Asia II 2, Asia II 3, Asia II 4, Asia II 5, Asia II 6, Asia II 7, Asia II 8, Asia II 9, Asia II 10, Asia II 11, Asia II 12, Asia III, Asia IV, Australia, Australia/Indonesia, China 1, China 2, China 3, China 4, Indian Ocean, Middle East-Asia Minor (MEAM) I, MEAM II, Mediterranean (MED), New World 1, New World 2, Japan 1, Japan 2, Uganda, Italy 1, Sub-Saharan Africa 1, Sub-Saharan Africa 2, Sub-Saharan Africa 3, Sub-Saharan Africa 4, Sub-Saharan Africa 5) have been currently delimited at the global level [10–13, 46–49, 66]. Among the B. tabaci species complex, the most important biotypes worldwide are B and Q, recently termed as the MEAM1 and MED species, respectively [3, 11, 44, 67].
2.4 Genomic and Postgenomic Resources Developed for B. tabaci and What Is Missing
As indicated, B. tabaci is a complex of biotypes that differ in their behavior, plant host, ability to induce disorders in plants, ability to transmit plant viruses, the bacterial endosymbionts they harbor, and genetic makeup [68]. In addition to the endogenous species, new invasive and better fit biotypes such as B and Q have invaded crop systems, exacerbating damage [69]. Very little is known about the virus transmission specificity, mating behavior, or genomes of either the whitefly or its bacterial endosymbionts. Collective functional genome projects of the whitefly will help to understand the whitefly genetic makeup and its interactions with environmental factors.
In 2005, the first attempt to measure nuclear DNA content of B. tabaci complex males and females was estimated using flow cytometry. This was the first step toward the exploration of the whitefly genome [70], and yielded values of 1.04 and 2.06 pg, respectively. Conversion between DNA content and genome size (1 pg DNA = 980 Mbp) indicated that the haploid genome size of B. tabaci is 1020 Mbp, which is approximately five times the size of the genome of the fruit fly, Drosophila melanogaster [70].
The first large-scale sequencing of ESTs from B. tabaci was initiated by Leshkowitz et al. in 2006 [71]. To address whitefly genomic sequence information, they constructed three cDNA libraries from non-viruliferous whiteflies (eggs, immature instars, and adults) and two libraries from adult insects with tomato yellow leaf curl virus (TYLCV) and tomato mottle virus (ToMoV). In total, 9110 sequences from the libraries were found to be involved in cellular and developmental processes. In addition, approximately 1000 bases were aligned with the genome of the B. tabaci primary endosymbiotic bacterium, Candidatus Portiera aleyrodidarum, originating primarily from the egg and immature instar libraries [71]. Apart from the mitochondrial sequences, abundant sequences in the libraries encoded vitellogenin gene sequences, indicating that much of the gene expression in B. tabaci is directed toward the production of eggs [71]. Studies thereafter were aimed to understand the mechanisms and the function of genes involved in various aspects of the whitefly interaction with its environment, and the first case was its resistance to insecticides. For the first time a DNA microarray containing 6000 unique ESTs from the whitefly was developed in 2007, and deep analysis revealed ESTs involved in insecticide resistance and xenobiotic detoxification such as those transcribed from P450 monooxygenases and oxidative stress genes, genes associated with protein, lipid and carbohydrate metabolism, and others related to juvenile hormone-associated processes in insects such as oocyte and egg development [72].
Following these pioneering studies and the development of new deep sequencing technologies and bioinformatic tools, the first de novo characterization of a whitefly transcriptome without prior genome annotation was sequenced by Wang et al. in 2010 [73], using the Illumina sequencing platform. This study obtained 168,900 unique sequences and a large number of genes associated with specific developmental stages and insecticide resistance. In 2011, using the same technology, Luan et al. [74] investigated the transcriptional response of the invasive B. tabaci MEAM1 species to tomato yellow leaf curl China virus (TYLCCNV). Results showed that 1606 genes involved in 157 biochemical pathways were differentially expressed in the viruliferous whiteflies. This indicates that TYLCCNV can perturb the cell cycle and primary metabolism, which explains the negative effect of this virus on the longevity and fecundity of B. tabaci [75]. Further results showed that TYLCCNV can activate whitefly immune responses, such as autophagy and antimicrobial peptide production, which might lead to a gradual decrease of viral particles within the body of the infected insect. Furthermore, PCR results showed that TYLCCNV can invade ovary and fat body, and LysoTracker and Western blot analyses revealed that the invasion of TYLCCNV induced autophagy in both types of tissue. Surprisingly, TYLCCNV also suppressed whitefly immune responses by downregulating the expression of genes involved in Toll-like signaling and mitogen-activated protein kinase (MAPK) pathways [74].
Using Illumina sequencing, further research investigated the transcriptome of the primary salivary glands of the MED species of the B. tabaci complex [76]. This study obtained 13,615 unigenes involved in metabolism and transport. Further analysis revealed genes related to processing of secretory proteins, secretion, and virus transmission [76]. Following this sequencing, Wang et al. [77, 78] sequenced an indigenous species, Asia II 3, and compared its genetic divergence with the transcriptomes of two invasive whitefly species, MEAM1 and MED. This study revealed a conserved group of 3203 protein families among the Asia II 3, MEAM1, and MED species which might be responsible for core cellular and physiological functions of the B. tabaci complex. Further results identifying hundreds of highly diverged genes and compiling sequence annotation data into functional groups found the most divergent gene classes to be the cytochrome P450 monooxygenases, glutathione metabolism, and oxidative phosphorylation. Moreover, many of these genes are predicted to be involved in protein metabolism such as peptide deformylase, cathepsin, cysteine proteinase, and metalloendopeptidase, which might be the driving force of the MEAM1-MED divergence and MEAM1- Asia II 3 differentiation [78]. A recent pyrosequencing study of the transcriptome of an invasive B biotype from China revealed a highly diverse bacterial community and robust system for insecticide resistance [79]. As mentioned above, de novo assembly generated 178,669 unigenes including 30,980 from the insects and 17,881 from the bacteria. Further in-depth transcriptome analysis revealed additional genes potentially involved in insecticide resistance and nutrient digestion [79].
Recently, Wang et al. [80] obtained the complete mitochondrial genome of the invasive Mediterranean species and identified 37 genes, including 13 protein-coding genes (PCGs), 2 ribosomal RNAs, and 22 transfer RNAs (tRNA). Comparative analyses of the genomes from MED and New World species revealed that there are no gene arrangements. This study also revealed that atp6 and atp8, nd4 and nd4l, and nd6 and cytb were on the same cistronic transcripts, whereas the other mature mitochondrial transcripts were monocistronic.
Recent transcriptome profiling of B. tabaci revealed stage-specific gene expression signatures for thiamethoxam resistance [81, 82]. Furthermore, analysis of gut transcriptomes of two invasive whitefly species in the B. tabaci complex, MEAM1 and MED, demonstrated the important role of the gut in the metabolism of insecticides and secondary plant chemicals [83]. Interestingly, using a microarray from B. tabaci that contained about 6000 genes, it was shown that parasitization by the wasp, Eretmocerus mundus, induced genes related to immune responses and revealed a role for the bacterial symbiont, Rickettsia, in these responses [84].
The foregoing transcriptomic studies reveal that a number of highly expressed genes that belong to different and conserved metabolic pathways with other insects are involved in virus transmission, insecticide resistance, and immune responses to parasitoids. Taken together, the transcriptomic results collected so far provide not only a roadmap for further functional genomic studies and extensive whitefly research in general but also a large collection of gene and EST sequences for future genome sequencing and annotation efforts.
2.5 B. tabaci Bacterial Symbionts
Bacterial symbionts in insects have drawn the attention of many research projects around the world because of their potential for developing friendly pest control methods by intervention with the insect-symbiont interactions. Recent surveys of facultative endosymbionts in whiteflies and aphids have used hundreds of insect populations collected around the world [85–90]. B. tabaci exhibits the highest diversity of secondary endosymbionts that infect one insect, and populations were reported to harbor seven different facultative (secondary) symbionts including the Alphaproteobacteria Rickettsia (Rickettsiales), Orentia-like organism (Rickettsiales), Wolbachia (Rickettsiales), the Gammaproteobacteria Arsenophonus (Enterobacteriales), Hamiltonella (Enterobacteriales), Cardinium (Bacteroidetes), and Fritschea (Chlamydiales) [91–97] (Fig. 2.2). All whitefly species including B. tabaci biotypes harbor the primary symbiont Portiera; however, they differ in their secondary symbiont composition [91, 98]. A study from Israel showed that B and Q biotypes differ in their infection with secondary symbionts, and while the B biotype was infected with Hamiltonella, the Q biotype was infected with Arsenophonus and Wolbachia. Both biotypes were infected with Rickettsia and none was infected with Fritschea and Cardinium [85]. A different study surveyed the distribution of secondary symbionts in whitefly species collected in the Balkan region and detected only the Q biotype of B. tabaci, which harbored Hamiltonella, Rickettsia, Wolbachia, and Cardinium, while Arsenophonus and Fritschea were not detected in any B. tabaci populations [86]. Recent studies focused on the role that these symbionts might play in the biology of their whitefly hosts. Two studies showed that Wolbachia [99] and Hamiltonella [100, 101] increase the fitness and enhance B. tabaci MED biotype performance. Other studies have shown that Rickettsia increases B. tabaci tolerance to heat [102] and influences the whitefly response to insecticides [103, 104]. A study conducted in the USA showed that Rickettsia increases the fitness of the B biotype of B. tabaci and enhances the proportion of females in the populations [105].
Fluorescence in situ hybridization (FISH) analysis on representative B. tabaci developmental stages and dissected organs for the localization of representative symbiotic bacteria. (a) An adult whitefly showing the primary symbiont Portiera in red and the secondary symbiont Hamiltonella in green. (b) A second-stage nymph showing the primary symbiont Portiera in red and the secondary symbiont Rickettsia in blue. (c) A fourth-stage nymph showing the primary symbiont Portiera in red and the secondary symbiont Hamiltonella in green. (d) A pupa showing the primary symbiont Portiera in red and the secondary symbiont Arsenophonus in yellow. (e) A dissected ovary showing oocytes in different developmental stages, the primary symbiont Portiera inside bacteriocyte cells in red and the secondary symbiont Rickettsia in green. (f) A portion of dissected midgut showing DAPI staining of the nuclei in blue and the secondary symbiont Rickettsia in red. Green represents FISH for localizing heat shock protein 70
Bacterial symbionts in B. tabaci have been shown to play an important role in begomovirus transmission [106, 107]. Gottlieb et al. [108] showed that only a GroEL protein from Hamiltonella interacted with TYLCV CP and protected the virus while circulating in the whitefly hemolymph for transmission, whereas other GroEL proteins from other symbionts did not interact with TYLCV CP. It was further shown that release of virions protected by GroEL occurs adjacent to the primary salivary gland. However, CP of abutilon mosaic virus (AbMV), a begomovirus non-transmissible by B. tabaci, was also found to bind to GroEL. The midgut was shown to serve as the barrier for AbMV transmission and not interactions with GroEL [107]. A study from India showed that GroEL protein of Arsenophonus interacted with the cotton leaf curl virus (CLCuV) CP, suggesting the involvement of Arsenophonus in the transmission of CLCuV in the Asia II genetic group of B. tabaci [109]. Additionally, a recent study has demonstrated that Rickettsia plays an important role in TYLCV transmission. Rickettsia-infected females transmitted TYLCV at twice the rate of Rickettsia-uninfected females and retained the virus longer [110]. Interestingly, an antagonistic relationship was discovered between Rickettsia and TYLCV in this study, in which high levels of the bacterium in the midgut resulted in higher virus concentrations in the filter chamber, a favored site for virus translocation along the transmission pathway, whereas low levels of Rickettsia in the midgut resulted in an even distribution of the virus.
Several recent projects have sequenced the genomes of some B. tabaci symbionts, including the primary symbiont, Portiera [111–113], and secondary symbionts, Rickettsia and Hamiltonella [100, 114], and Cardinium [115]. These projects have provided unique insights into the genome reduction of these symbionts, which has made them completely dependent on their hosts and, therefore, not culturable. Additionally, the genome sequences of these symbionts have shown the dependence of the insect host on amino acid biosynthetic pathways lacking in the insect genome, providing further insights on the mutual symbiosis between the insect and its symbiotic bacteria. This is more evident when examining the genome of Portiera, the primary symbiont, which has an extremely reduced genome, yet maintains biosynthetic pathways essential for the survival of the symbiont as well as the insect.
Several additional recent B. tabaci EST sequencing projects identified a total of 17,766 bacterial unigenes which were classified into 322 genera [(Proteobacteria (92 %), Betaproteobacteria (59 %), Burkholderiales (58 %), Comamonadaceae (50 %), and Delftia (43 %)] and revealed a highly diverse bacterial community which represents a high and rapid coevolution of insects and their symbionts [79]. Additional population surveys from Brazil using rRNA gene sequencing showed that Hamiltonella and Rickettsia are highly prevalent in all MEAM1 populations tested, while Cardinium was close to fixation in only three populations. Surprisingly, some MEAM1 individuals and one New World 2 population were infected with Fritschea [90]. Guo et al. [116] observed more females than males harboring Hamiltonella, and the results suggested that both the female-biased symbiont infections and female-biased TYLCV infections promote the rapid spread of TYLCV in China. Recently, Zchori-Fein et al. [97] modeled the variations in the symbiotic communities of about 2000 sweetpotato whitefly individuals and demonstrated facultative endosymbiotic combinations which were positively correlated with both distance from the equator and specificity of the genetic code of the insect host.
The reports cited here show that begomovirus translocation in the vector involves not only hypothesized insect proteins and receptors but also bacterial proteins, which could be acting directly or indirectly to influence the virus transmission. Future whitefly genomics research is expected to gain better understanding of the novel genes and mechanisms involved in virus-vector-symbiont relationships.
2.6 Studying the Interaction Between B. tabaci and Begomoviruses
B. tabaci is known to transmit a great diversity of plant viruses; among them, begomoviruses are the most devastating and cause serious viral diseases in agricultural cropping systems worldwide. The whitefly-begomovirus interactions depend on the virus, the biotype of the whitefly vector, and the endosymbionts harbored in the specific vector [68]. More than 80 % of the mono- and bipartite begomoviruses (both New and Old worlds) are transmitted by whiteflies in and around tropical and subtropical regions. Whitefly-transmitted begomoviruses are known to cause leaf curling, yellow mosaic, and yellow vein mosaic in several important crops and weeds. Selection pressure exerted by vectors of a particular geographical location is a crucial factor in the evolution of begomoviruses. Excellent reviews are available on the whitefly-transmitted viruses in New World and Old World and their interaction with insect vectors [21, 68, 117–122].
Begomoviruses are vectored by B. tabaci in a persistent and circulative manner (Fig. 2.3). The stylet of B. tabaci penetrates the plant epidermis and moves intracellularly through the parenchyma to reach the phloem, which is required for both virus acquisition and transmission. Whiteflies acquire virions during feeding from the phloem of an infected plant. The virions move through the alimentary canal into the whitefly midgut (Fig. 2.4), where they enter the hemolymph and transit to the salivary glands for transmission during the next feeding cycle [123] (Fig. 2.3).
Schematic representation of the circulative route of tomato yellow leaf curl virus (TYLCV) inside B. tabaci. The virus (red particles) is acquired from the plant phloem (p) through the stylet (s). The virus moves along the esophagus (e) and reaches the filter chamber (fc) in the midgut (mg) where the majority of the virus particles are translocated into the hemolymph. This translocation is aided, in part, by the heat shock protein 70 (black particles). In the hemolymph, Hamiltonella, a bacterial symbiont harbored within bacteriocyte cells (yellow organs), produces a GroEL protein (blue particles) that interacts with TYLCV and aids in its safe arrival to the primary salivary glands (psg). Once inside the salivary glands, the virus moves along the salivary duct to be injected into a new plant while the insect is feeding. hg hindgut
Immunolocalization of TYLCV in the midgut of B. tabaci following acquisition of the virus from infected plants for several days. First antibody was used against the virus CP and secondary antibody attached to Cy3 dye (red). (a) Shows the virus in red, scattered along the midgut and concentrating in the filter chamber (fc). (b) The same midgut as in the left panel showing DAPI staining of the nuclei (blue). am ascending midgut, dm descending midgut, ca ceca, CP coat protein
The capsid protein plays a crucial role in the transmission of begomoviruses by B. tabaci. Evidence for a CP role in virus transmission emerged from following observations: (a) exchange of an African cassava mosaic virus (ACMV) CP gene with that of beet curly top virus (BCTV) altered the insect specificity of ACMV from whiteflies to leafhoppers [124]; (b) whiteflies have been shown to be unable to acquire coat protein mutants of geminiviruses that did not form capsids [125, 126]; and (c) replacing the CP of the non-transmissible begomovirus, Abutilon mosaic virus (AbMV), by that of a transmissible isolate of Sida golden mosaic virus (SiGMV), restored transmission by B. tabaci [127].
Site-directed mutagenesis in the CP domain was used to map the functional regions in tomato yellow leaf curl Sardinia virus (TYLCSV) which are involved in the transmission of the virus by B. tabaci [128, 129]. Briefly, a region between amino acids 129 and 152, including Q129, Q134, and D152, was found to be relevant for virion assembly, systemic infection, and transmission by the vector. In addition, it was shown that crossing the salivary gland barrier may not be sufficient for transmission. These results were confirmed in other bipartite begomoviruses, namely, watermelon chlorotic stunt virus (WmCSV) [130] and AbMV [131]. Using immunogold labeling studies, the CP mutants PNHD and PNHE were detected in B. tabaci salivary glands similar to the wild-type virus, but the mutant QDQD CP was not detected in salivary gland cells. The most interesting result was that although the mutants PNHD and PNHE were present in primary salivary gland, transmission did not occur, suggesting that their presence in salivary gland does not ensure transmission. Thus, molecular interactions with saliva might be necessary to maintain infectivity, which, perhaps, are absent from these mutants [129].
Further studies to unravel the importance of TYLCV CP in virus transmission showed its interaction with a member of the small heat shock protein family (BtHSP16), which was identified using a yeast two-hybrid system screen against TYLCSV CP [132]. Another study recently demonstrated that another heat shock protein, HSP70, interacts with the TYLCV CP in vivo and in vitro, and membrane feeding with anti-HSP70 antibodies resulted in an increase in TYLCV transmission. This result suggested that under normal conditions HSP70 restricts virus activity, thereby protecting the insect from deleterious effects of the circulating virus [133]. Interactions with these proteins may be necessary for refolding of the virion particle and facilitating its crossing the barrier between the midgut and the hemolymph or hemolymph and the salivary glands.
2.7 Interaction of B. tabaci with Plants Using Genomics Tools
Many plants have developed various chemical defenses to deter or combat insect pests. To date, some genomic studies were conducted for understanding the plant responses to B. tabaci. This has limited our understanding of the plant response to hemipteran species in general. Hemipterans cause minimal cellular wounding, and although the duration of feeding can be very long, the molecular plant responses can be minimal; however, they can also be dramatic [134]. Salicylic acid (SA) and jasmonic acid (JA) are two important components in the signal transduction cascades that regulate plant defense responses against biotic stresses. JA affects plant resistance to necrotrophic pathogens and tissue-damaging insects as well as some phloem-feeding insects [135]. Other plant defense proteins such as proteinase inhibitors (PI) [136, 137], PI II [138–140], LOX genes [140], and others are inducible during insect feeding.
To understand the major plant defenses in responding to feeding by the whitefly, studies were conducted with the model plant Arabidopsis thaliana [141, 142]. Zarate et al. [141] reported that the whitefly induces SA defenses and suppresses effectual JA defenses. Supporting this study, using the Affymetrix ATH1 GeneChip to monitor the A. thaliana transcriptome, 700 transcripts were found to be upregulated and 556 downregulated by feeding of the silverleaf whitefly nymphs [142]. Closer examination of the regulation of secondary metabolite (glucosinolate) and defense pathway genes after feeding showed that responses were qualitatively and quantitatively different from chewing insects and aphids. Furthermore, the JA genes PDF1.2, PR4, and CORI3 were repressed and SA-regulated genes were induced after whitefly feeding. Cytological staining of whitefly-infected tissue showed that pathogen defenses, such as localized cell death and hydrogen accumulation, were not observed [142].
Few studies have shown that virus proteins can act as suppressors for plant defense systems which are directed to block or reduce defenses against insect feeding. Coinfection of the begomovirus TYLCCNV and its tomato yellow leaf curl China virus betasatellite (TYLCCNB) could repress the JA response but not the SA response [143]. Microarray analysis of the A. thaliana transcriptome in response to cabbage leaf curl virus (CabLCV) infection uncovered 5365 differentially expressed genes; data mining revealed that CabLCV triggers a pathogen response via the SA pathway and induces expression of genes involved in programmed cell death, genotoxic stress, and DNA repair [144]. Further, coinfection of plants by TYLCCNV and TYLCCNB increased expression of the whitefly vitellogenin gene [145]. Recently, Shi et al. [146] demonstrated that SA content was always higher in leaves infested with the viruliferous B biotype compared with the viruliferous Q biotype. Additional recent studies by Shi et al. [147, 148] showed that B. tabaci Q carrying TYLCV strongly suppressed host plant defenses.
2.8 Interactions of B. tabaci with Parasitoids and Pathogens Using Genomic Tools
Parasitoids that specifically attack B. tabaci have been used commercially for the biocontrol of whiteflies. Most whitefly parasitoids kill the insect by piercing the body contents and feeding on host fluids. Parasitoids from the genera Encarsia, Eretmocerus (Aphelinidae), and Amitus (Platygastridae) can attack whiteflies [149, 150]. Recent studies identified Encarsia sophia and Eretmocerus hayati (Hymenoptera: Aphelinidae) as two key parasitoids of B. tabaci (MEAM1) [151, 152], and En. formosa [153], for which it was suggested that begomoviruses transmitted by the whitefly can manipulate the host suitability of a parasitoid and hence the parasitoid-host interactions.
Mahadav et al. [84] investigated the response of B. tabaci to parasitization by the wasp, E. mundus, using a whitefly cDNA-based microarray. The results clearly indicated that genes known to be part of defense pathways described in other insects are also involved in the response of B. tabaci to parasitization by E. mundus. Some of these responses include repression of a serine protease inhibitor (serpin) and induction of a melanization cascade. Quantitative real-time RT-PCR (qRT-PCR) and fluorescence in situ hybridization (FISH) analyses showed that the proliferation of Rickettsia, a facultative secondary symbiont, was strongly induced upon initiation of the parasitization process [84].
Recently, using RNA-seq technology, Zhang et al. [154] conducted a comprehensive investigation of the whitefly defense response to infection by Pseudomonas aeruginosa. Compared to uninfected whiteflies, 6- and 24-h post-infected whiteflies showed 1348 upregulated and 1888 downregulated genes that were differentially expressed. Functional analysis of these genes revealed that the mitogen-associated protein kinase (MAPK) pathway was activated after P. aeruginosa infection. Additional research included investigating the response of B. tabaci to the entomopathogenic fungus, Beauveria bassiana, using next-generation sequencing, which showed that conserved pathways such as the DNA repair and MAPK pathways are responsive to attack by this fungus [155].
2.9 Concluding Remarks and Future Perspective
B. tabaci is undoubtedly one of the major worldwide insect pests in agriculture. Despite many years of research, conventional chemical control methods for reducing the damage caused by this pest are still dominating. The past two decades have witnessed a dramatic increase in the research aimed toward understanding the interactions of B. tabaci with its environment and other organisms, including those that are part of its damage cycle such as plant viruses. However, those interactions also include plants, parasitoids, symbionts, and abiotic factors such as heat. The use of newly developed molecular and genomic tools will facilitate our understanding of the genetic makeup of the whitefly and will enable further development of novel tools for better means of controlling this insect pest.
Abbreviations
- AbMV:
-
Abutilon mosaic virus
- CabLCV:
-
Cabbage leaf curl virus
- CP:
-
Coat protein
- EST:
-
Expressed sequence tag
- FISH:
-
Fluorescence in situ hybridization
- HSP:
-
Heat shock protein
- ITS1:
-
Ribosomal internal transcribed spacer 1
- JA:
-
Jasmonic acid
- MEAM1:
-
Middle East-Asia Minor 1
- MED:
-
Mediterranean
- mtCO1:
-
Mitochondrial cytochrome oxidase 1
- qRT-PCR:
-
Quantitative real-time RT-PCR
- RAPD-PCR:
-
Random amplified polymorphic cDNA-polymerase chain reaction
- RFLP:
-
Restriction fragment length polymorphism
- SA:
-
Salicylic acid
- SCAR:
-
Sequence-characterized amplified regions
- ToMoV:
-
Tomato mottle virus
- TYLCCNB:
-
Tomato yellow leaf curl China virus betasatellite
- TYLCCNV:
-
Tomato yellow leaf curl China virus
- TYLCSV:
-
Tomato yellow leaf curl Sardinia virus
- TYLCV:
-
Tomato yellow leaf curl virus
- WmCSV:
-
Watermelon chlorotic stunt virus
References
Severson DW, Behura SK (2012) Mosquito genomics: progress and challenges. Annu Rev Entomol 57:143–166
Brown JK (1994) Current status of Bemisia tabaci as a plant pest and virus vector in agro-ecosystems worldwide. FAO Plant Prot Bull 42:3–32
Brown JK, Frohlich DR, Rosell RC (1995) The sweetpotato or silverleaf whiteflies: biotypes of Bemisia tabaci or a species complex? Annu Rev Entomol 40:511–534
Costa HS, Brown JK, Sivasupramaniam S, Bird J (1993) Regional distribution, insecticide resistance, and reciprocal crosses between the ‘A’ and ‘B’ biotypes of Bemisia tabaci. Insect Sci Appl 14:255–266
Gennadius P (1889) Disease of tobacco plantations in the Trikonia. The aleyrodid of tobacco. Ellenike Georgia 5:1–3
Quaintance AL (1900) Contribution towards a monograph of the American aleurodidae. US Department of Agriculture. Technica series. Bur Entomol 8:9–64
Russell LM (1957) Synonyms of Bemisia tabaci (Gennadius) (Homoptera, Aleyrodidae). Bull Brooklyn Entomol Soc 52:122–123
Perring TM (2001) The Bemisia tabaci species complex. Crop Prot 20:725–737
Oliveira MRV, Henneberry TJ, Anderson P (2001) History, current status, and collaborative research projects for Bemisia tabaci. Crop Prot 20:709–723
Dinsdale A, Cook L, Riginos C, Buckley YM, De Barro P (2010) Refined global analysis of Bemisia tabaci (Hemiptera: Sternorrhyncha: Aleyrodidae: Aleyrodidae) mitochondrial cytochrome oxidase 1 to identify species level genetic boundaries. Ann Entomol Soc Am 103:196–208
De Barro PJ, Liu SS, Boykin L, Dinsdale A (2011) Bemisia tabaci: a statement of species status. Annu Rev Entomol 56:1–19
Firdaus S, Vosman B, Hidayati N, Supena E, Visser RGF, van Heusden AW (2013) The Bemisia tabaci species complex: additions from different parts of the world. Insect Sci 20:723–733
Boykin LM, Bell CD, Evans G, Small I, De Barro PJ (2013) Is agriculture driving the diversification of the Bemisia tabaci species complex (Hemiptera: Sternorrhyncha: Aleyrodidae)?: dating, diversification and biogeographic evidence revealed. BMC Evol Biol 13:228
Misra CS, Lamba SK (1929) The cotton whitefly (Bemisia gossypiperda n. sp.). Bull Agric Res Inst Pusa 196:1–7
Husain MA, Trehan KN (1933) Observations on the life history, bionomics and control of the whitefly of cotton (Bemisia gossypierda, n. sp.). Bull Agric Res Inst Pusa 196:7
Martin JH, Mifsud D, Rapisarda C (2000) The whiteflies (Hemiptera: Aleyrodidae) of Europe and the Mediterranean Basin. Bull Entomol Res 90:407–448
Jones DR (2003) Plant viruses transmitted by whiteflies. Eur J Plant Pathol 109:195–219
Mugiira RB, Liu SS, Zhou X (2008) Tomato yellow leaf curl virus and Tomato leaf curl Taiwan virus invade south-east coast of China. J Phytopathol 156:217–221
Riley DG, Palumbo JC (1995) Interaction of silverleaf whitefly (Homoptera: Aleyrodidae) with cantaloupe yield. J Econ Entomol 88:1726–1732
Anderson PK, Morales FJ (2005) Whitefly and whitefly-borne viruses in the tropics: building a knowledge base for global action. CIAT, Cali, p 351
Navas-Castillo J, Fiallo-Olive E, Sanchez-Campos S (2011) Emerging virus diseases transmitted by whiteflies. Ann Rev Phytopathol 49:219–248
Brown JK (2000) Molecular markers for the identification and global tracking of whitefly vector-Begomovirus complexes. Virus Res 71:233–260
Liu SS, De Barro PJ, Xu J, Luan JB, Zang LS, Ruan YM, Wan FH (2007) Asymmetric mating interactions drive widespread invasion and displacement in a whitefly. Science 318:1769–1772
Jiu M, Zhou X-P, Tong L, Xu J, Yang X, Wan FH, Liu SS (2007) Vector-virus mutualism accelerates population increase of an invasive whitefly. PLoS One 2:e182
Crowder DW, Horowitz AR, De Barro PJ (2010) Mating behaviour, life history and adaptation to insecticides determine species exclusion between whiteflies. J Anim Ecol 79:563–570
Gorman K, Slater R, Blande J, Clarke A, Wren J, McCaffery A, Denholm I (2010) Cross-resistance relationships between neonicotinoids and pymetrozine in Bemisia tabaci (Hemiptera: Aleyrodidae). Pest Manag Sci 66:1186–1190
Brown JK, Czosnek H (2002) Whitefly transmission of plant viruses. In: Plumb RT (ed) Advances in botanical research, vol 36. Academic, New York
Bird J (1957) A whitefly transmitted mosaic of Jatropha gossypifolia. Technical paper. University of Puerto Rico, Agricultural Experiment Station 22:1–35
Bird J, Maramorosch K (1978) Viruses and virus diseases associated with whiteflies. Adv Virus Res 22:55–110
Costa HS, Russel M (1975) Failure of Bemisia tabaci to breed on cassava plants in Brazil (Homoptera, Aleyrodidae). Cienia Cult 27:388–390
Perring TM, Cooper A, Kazmer DJ, Shields C, Sheilds J (1991) New strain of sweet potato whitefly invades California vegetables. Calif Agric 45:10–12
Bedford ID, Briddon RW, Brown JK, Rossel RC, Markham PG (1994) Geminivirus transmission and biological characterisation of Bemisia tabaci (Genn) biotypes from different geographic regions. Ann Appl Biol 125:311–325
Bellows TS, Perring TM, Gill RJ, Headrich DH (1994) Description of a species of Bemisia tabaci (Homoptera: Aleyrodidae). Ann Entomol Soc Am 87:195–206
Perring TM, Cooper AD, Rodrigues RJ, Farrar CA, Bellows TSJ (1993) Identification of a whitefly species by genomic and behavioural studies. Science 259:74–77
Gawel NJ, Bartlett AC (1993) Characterization of differences between whiteflies using RAPD-PCR. Insect Mol Biol 2:33–38
Chu D, Zhang YJ, Cong B, Xu BY, Wu QJ (2004) Developing sequence characterized amplified regions (SCARs) to identify Bemisia tabaci and Trialeurodes vaporariorum. Plant Prot 30:27–30
Khasdan V, Levin I, Rosner A, Morin S, Kontsedalov S et al (2005) DNA markers for identifying biotypes B and Q of Bemisia tabaci (Hemiptera: Aleyrodidae) and studying population dynamics. Bull Entomol Res 95:605–613
Zang LS, Chen WQ, Liu SS (2006) Comparison of performance on different host plants between the B biotype and a non-B biotype of Bemisia tabaci from Zhejiang, China. Entomol Exp Appl 121:221–227
Boukhatem N, Jdaini S, Mukovski Y, Jacquemin JM, Bouali A (2007) Identification of Bemisia tabaci (Gennadius) (Homoptera: Aleyrodidae) based on RAPD and design of two SCAR markers. J Biol Res (Thessaloniki) 8:167–176
Ko CC, Hung YC, Wang CH (2007) Sequence characterized amplified region markers for identifying biotypes of Bemisia tabaci (Hem., Aleyrodidae). J Appl Entomol 131:542–547
Shatters RG Jr, Powell CA, Boykin LM, He LS, McKenzie CL (2009) Improved DNA barcoding method for Bemisia tabaci and related Aleyrodidae: development of universal and Bemisia tabaci biotype-specific mitochondrial cytochrome c oxidase chain reaction primers. J Econ Entomol 102:750–758
Ma DY, Li XC, Dennehy TJ, Lei CL, Wang M et al (2009) Utility of mtCO1 polymerase chain reaction-restriction fragment length polymorphism in differentiating between Q and B whitefly Bemisia tabaci biotypes. Insect Sci Appl 16:107–114
Cervera MT, Cabezas JA, Simon B, Martinez-Zapater JM, Beitia F, Cenis JL (2000) Genetic relationships among biotypes of Bemisia tabaci Hemiptera, Aleyrodidae based on AFLP analysis. Bull Entomol Res 90:391–396
Frohlich DR, Torres-Jerez I, Bedford ID, Markham PG, Brown JK (1999) A phylogeographical analysis of the Bemisia tabaci species complex based on mitochondrial DNA markers. Mol Ecol 8:1683–1691
Hu J, De Barro P, Zhao H, Wang J, Nardi F (2011) An extensive field survey combined with a phylogenetic analysis reveals rapid and widespread invasion of two alien whiteflies in China. PLoS One 6:e16061
Alemandri V, De Barro PJ, Bejerman N, Argüello Caro EB, Dumón AD, Mattio MF, Rodriguez SM, Truoli G (2012) Species within the Bemisia tabaci (Hemiptera: Aleyrodidae) complex in soybean and bean crops in Argentina. J Econ Entomol 105:48–53
Boykin LM, Armstrong KF, Kubatko L, De Barro PJ (2012) Species delimitation and global biosecurity. Evol Bioinform 8:1–37
Liu SS, Colvin J, De Barro PJ (2012) Species concepts as applied to the whitefly Bemisia tabaci systematics: how many species are there? J Integr Agric 11:176–186
Lee W, Park J, Lee G, Lee S, Akimoto S (2013) Taxonomic status of the Bemisia tabaci complex (Hemiptera: Aleyrodidae) and reassessment of the number of its constituent species. PLoS One 8:e63817
De Barro PJ, Driver F, Trueman JWH, Curran J (2000) Phylogenetic relationship of world populations of Bemisia tabaci (Gennadius) using ribosomal ITS1. Mol Phylogenet Evol 16:29–36
Wu X, Li Z, Hu D, Shen Z (2003) Identification of Chinese populations of Bemisia tabaci Gennadius by analyzing ITS1 sequence. Prog Nat Sci 13:276–281
Abdullahi I, Winter S, Atirim GI, Thottappilly G (2003) Molecular characterization of whitefly, Bemisia tabaci Hemiptera, Aleyrodidae populations infesting cassava. Bull Entomol Res 93:97–106
De Barro PJ (2005) Genetic structure of the whitefly Bemisia tabaci in the Asia-Pacific region revealed using microsatellite markers. Mol Ecol 14:3695–3718
Li ZX, Lin HZ, Guo XP (2007) Prevalence of Wolbachia infection in Bemisia tabaci. Curr Microbiol 54:467–471
De Barro PJ, Scott KD, Graham GC, Lange CL, Schutze MK (2003) Isolation and characterization of microsatellite loci in Bemisia tabaci. Mol Ecol Notes 3:40–43
Tsagkarakou A, Roditakis N (2003) Isolation and characterization of microsatellite loci in Bemisia tabaci (Hemiptera: Aleyrodidae). Mol Ecol Notes 3:196–198
De Barro PJ, Trueman JWH, Frohlich DR (2005) Bemisia argentifolii is a race of B. tabaci (Hemiptera: Aleyrodidae): the molecular genetic differentiation of B. tabaci populations around the world. Bull Entomol Res 95:193–203
De Barro PJ, Hidayat SH, Frohlich D, Subandiyah S, Ueda S (2008) A virus and its vector, pepper yellow leaf curl virus and Bemisia tabaci, two new invaders of Indonesia. Biol Invasions 10:411–433
Delatte H, David P, Granier M, Lett JM, Goldbach R, Peterschmitt M, Reynaud B (2006) Microsatellites reveal extensive geographical, ecological and genetic contacts between invasive and indigenous whitefly biotypes in an insular environment. Genet Res 87:109–124
Delatte H, Reynaud B, Granier M, Thornary L, Lett JM, Goldbach R, Peterschmitt M (2005) A new silverleaf-inducing biotype Ms of Bemisia tabaci (Hemiptera: Aleyrodidae) indigenous to the islands of the southwest Indian Ocean. Bull Entomol Res 95:29–35
Dalmon A, Halkett F, Granier M, Delatte H, Peterschmitt M (2008) Genetic structure of the Invasive pest Bemisia tabaci: evidence of limited but persistent genetic differentiation in glasshouse populations. Heredity 100:316–325
Tsagkarakou A, Tsigenopoulos CS, Gorman K, Lagnel J, Bedford ID (2007) Biotype status and genetic polymorphism of the whitefly Bemisia tabaci (Hemiptera: Aleyrodidae) in Greece: mitochondrial DNA and microsatellites. Bull Entomol Res 97:29–40
Gauthier N, Dalleau-Clouet C, Bouvret M-E (2008) Twelve new polymorphic microsatellite loci and PCR multiplexing in the whitefly, Bemisia tabaci. Mol Ecol Resour 8:1004–1007
Valle G, Lourençao A, Zucchi M, Pinheiro J (2012) Low polymorphism revealed in new microsatellite markers for Bemisia tabaci (Hemiptera: Aleyrodidae). Genet Mol Res 11:3899–3903
Wang HL, Yang J, Boykin LM, Zhao QY, Wang YJ, Liu SS, Wang XW (2014) Developing conversed microsatellite markers and their implications in evolutionary analysis of the Bemisia tabaci complex. Sci Rep 4:6351
Tay WT, Evans GA, Boykin LM, De Barro PJ (2012) Will the real Bemisia tabaci please stand up? PLoS ONE 7:e50550
Brown JK (2007) The Bemisia tabaci complex: genetic and phenotypic variation and relevance to TYLCV-vector interactions. In: Czosnek H (ed) Tomato yellow leaf curl virus disease. Springer, Dordrecht, pp 25–56
Ghanim M (2014) A review of the mechanisms and components that determine the transmission efficiency of Tomato yellow leaf curl virus (Geminiviridae; Begomovirus) by its whitefly vector. Virus Res 186:47–54
Czosnek H, Brown J (2010) The whitefly genome – white paper: proposal to sequence multiple genomes of Bemisia tabaci. In: Stansly PA, Naranjo SE (eds) Bemisia: bionomics and management of a global pest. Springer, Dordrecht, pp 503–532, 540 pages
Brown JK, Lambert GM, Ghanim M, Czosnek H, Galbraith DW (2005) Nuclear DNA content of the whitefly Bemisia tabaci (Genn.) (Aleyrodidae: Homoptera/Hemiptera) estimated by flow cytometry. Bull Entomol Res 95:309–312
Leshkowitz D, Gazit S, Reuveni E, Ghanim M, Czosnek H, McKenzie C, Shatters RG Jr, Brown JK (2006) Whitefly (Bemisia tabaci) genome project: analysis of sequenced clones from egg, instar, and adult (viruliferous and non-viruliferous) cDNA libraries. BMC Genomics 7:79
Ghanim M, Kontsedalov S (2007) Gene expression in pyriproxyfen-resistant Bemisia tabaci Q biotype. Pest Manag Sci 63:776–783
Wang XW, Luan JB, Li JM, Bao YY, Zhang CX, Liu SS (2010) De novo characterization of a whitefly transcriptome and analysis of its gene expression during development. BMC Genomics 11:400
Luan JB, Li JM, Varela N, Wang YL, Li FF, Bao YY, Zhang CX, Liu SS, Wang XW (2011) Global analysis of the transcriptional response of whitefly to tomato yellow leaf curl china virus reveals their relationship of coevolved adaptations. J Virol 85:3330–3340
Rubinstein G, Czosnek H (1997) Long-term association of tomato yellow leaf curl virus with its whitefly vector Bemisia tabaci: effect on the insect transmission capacity, longevity and fecundity. J Gen Virol 78:2683–2689
Su YL, Li JM, Li M, Luan JB, Ye XD, Wang XW, Liu SS (2012) Transcriptomic analysis of the salivary glands of an invasive whitefly. PLoS One 7:e39303
Wang XW, Luan JB, Li JM, Su YL, Xia J, Liu SS (2011) Transcriptome analysis and comparison reveal divergence between two invasive whitefly cryptic species. BMC Genomics 12:458
Wang XW, Zhao QY, Luan JB, Wang YJ, Yan GH, Liu SS (2012) Analysis of a native whitefly transcriptome and its sequence divergence with two invasive whitefly species. BMC Genomics 13:529
Xie W, Meng Q, Wu Q, Wang S, Yang X, Yang N, Li R, Jiao X, Pan H, Liu B, Su Q, Xu B, Hu S, Zhou X, Zhang Y (2012) Pyrosequencing the Bemisia tabaci transcriptome reveals a highly diverse bacterial community and a robust system for insecticide resistance. PLoS One 7:e35181
Wang HL, Yang J, Boykin LM, Zhao QY, Li Q, Wang XW, Liu SS (2013) The characteristics and expression profiles of the mitochondrial genome for the Mediterranean species of the Bemisia tabaci complex. BMC Genomics 14:401
Xie W, Yang X, Wang S, Wu Q, Yang N, Li R, Jiao X, Pan H, Liu B, Feng Y, Xu B, Zhou X, Zhang Y (2012) Gene expression profiling in the thiamethoxam resistant and susceptible B-biotype sweetpotato whitefly, Bemisia tabaci. J Insect Sci 12:46
Yang N, Xie W, Yang X, Wang S, Wu Q, Li R, Pan H, Liu B, Shi X, Fang Y, Xu B, Zhou X, Zhang Y (2014) Transcriptomic and proteomic responses of sweetpotato whitefly, Bemisia tabaci, to Thiamethoxam. PLoS One 8:e61820
Ye XD, Su YL, Zhao QY, Xia WQ, Liu SS, Wang XW (2014) Transcriptomic analyses reveal the adaptive features and biological differences of guts from two invasive whitefly species. BMC Genomics 15:370
Mahadav A, Gerling D, Gottlieb Y, Czosnek H, Ghanim M (2008) Gene expression in the whitefly Bemisia tabaci pupae in response to parasitization by the wasp Eretmocerus mundus. BMC Genomics 9:342
Chiel E, Gottlieb Y, Zchori-Fein E, Mozes-Daube N, Katzir N, Inbar M, Ghanim M (2007) Biotype-dependent secondary symbiont communities in sympatric populations of Bemisia tabaci. Bull Entomol Res 97:407–413
Skaljac M, Zanic K, Goreta-Ban S, Kontsedalov S, Ghanim M (2010) Co-infection and localization of secondary symbionts in two whitefly species. Isr J Plant Sci 58:103–111
Skaljac M, Zanic K, Hrncic S, Radonjic S, Perovic T, Ghanim M (2013) Diversity and localization of bacterial symbionts in three whitefly species (Hemiptera: Aleyrodidae) from the east coast of the Adriatic Sea. Bull Entomol Res 103:48–59
Gueguen G, Vavre F, Gnankine O, Peterschmitt M, Charif D, Chiel E, Gottlieb Y, Ghanim M, Zchori-Fein E, Fleury F (2010) Endosymbiont metacommunities, mtDNA diversity and the evolution of the Bemisia tabaci (Hemiptera: Aleyrodidae) species complex. Mol Ecol 19:4365–4376
Henry LM, Peccoud J, Simon JC, Hadfield JD, Maiden MJ, Ferrari J, Godfray HC (2013) Horizontally transmitted symbionts and host colonization of ecological niches. Curr Biol 23:1713–1717
Marubayashi JM, Kliot A, Yuki VA, Marques-Rezende JA, Krause-Sakate R, Pavan MA, Ghanim M (2014) Diversity and localization of bacterial endosymbionts from whitefly species collected in Brazil. PLoS One 9:e108363
Gottlieb Y, Ghanim M, Gueguen G, Kontsedalov S, Vavre F, Fleury F, Zchori-Fein E (2008) Inherited intracellular ecosystem: symbiotic bacteria share bacteriocytes in whiteflies. FASEB J 22:2591–2599
Chu D, Gao CS, De Barro P, Zhang YJ, Wan FH, Khan IA (2011) Further insights into the strange role of bacterial endosymbionts in whitefly, Bemisia tabaci: comparision of secondary symbionts from biotypes B and Q in China. Bull Entomol Res 101:477–486
Liu S, Chougule NP, Vijayendran D, Bonning BC (2012) Deep sequencing of the transcriptomes of soybean aphid and associated endosymbionts. PLoS One 7:e45161
Bing XL, Ruan YM, Rao Q, Wang XW, Liu SS (2013) Diversity of secondary endosymbionts among different putative species of the whitefly Bemisia tabaci. Insect Sci 20:194–206
Bing XL, Xia WQ, Gui JD, Yan GH, Wang XW, Liu SS (2014) Diversity and evolution of the Wolbachia endosymbionts of Bemisia (Hemiptera: Aleyrodidae) whiteflies. Ecol Evol 4:2714–2737
Helene D, Remy B, Nathalie B, Anne-Laure G, Traore RS, Jean-Michel L, Bernard R (2014) Species and endosymbiont diversity of Bemisia tabaci (Homoptera: Aleyrodidae) on vegetable crops in Senegal. Insect Sci. doi:10.1111/1744-7917.12134
Zchori-Fein E, Lahav T, Freilich S (2014) Variations in the identity and complexity of endosymbiont combinations in whitefly hosts. Front Microbiol 5:310
Thao ML, Baumann P (2004) Evolutionary relationships of primary prokaryotic endosymbionts of whiteflies and their hosts. Appl Environ Microbiol 70:3401–3406
Xue X, Li SJ, Ahmed MZ, De Barro PJ, Ren SX, Qiu BL (2012) Inactivation of Wolbachia reveals its biological roles in whitefly host. PLoS One 7:e48148
Rao Q, Wang S, Su YL, Bing XL, Liu SS, Wang XW (2012) Draft genome sequence of “Candidatus Hamiltonella defensa”, an endosymbiont of the whitefly Bemisia tabaci. J Bacteriol 194:3558
Su Q, Xie W, Wang S, Wu Q, Liu B, Fang Y, Xu B, Zhang Y (2014) The endosymbiont Hamiltonella increases the growth rate of its host Bemisia tabaci during periods of nutritional stress. PLoS One 9:e89002
Brumin M, Kontsedalov S, Ghanim M (2011) Rickettsia influences thermotolerance in the whitefly Bemisia tabaci B biotype. Insect Sci 18:57–66
Kontsedalov S, Zchori-Fein E, Chiel E, Gottlieb Y, Inbar M, Ghanim M (2008) The presence of Rickettsia is associated with increased susceptibility of Bemisia tabaci (Homoptera: Aleyrodidae) to insecticides. Pest Manag Sci 64:789–792
Kontsedalov S, Gottlieb Y, Ishaaya I, Nauen R, Horowitz AR, Ghanim M (2009) Toxicity of spiromesifen to the developmental stages of Bemisia tabaci biotype B. Pest Manag Sci 65:5–13
Himler AG, Adachi-Hagimori T, Bergen JE, Kozuch A, Kelly SE, Tabashnik BE, Chiel E, Duckworth VE, Dennehy TJ, Zchori-Fein E, Hunter MS (2011) Rapid spread of a bacterial symbiont in an invasive whitefly is driven by fitness benefits and female bias. Science 332:254–256
Morin S, Ghanim M, Zeidan M, Czosnek H, Verbeek M, van den Heuvel JF (1999) A GroEL homologue from endosymbiotic bacteria of the whitefly Bemisia tabaci is implicated in the circulative transmission of tomato yellow leaf curl virus. Virology 256:75–84
Morin S, Ghanim M, Sobol I, Czosnek H (2000) The GroEL protein of the whitefly Bemisia tabaci interacts with the coat protein of transmissible and nontransmissible begomoviruses in the yeast two-hybrid system. Virology 276:404–416
Gottlieb Y, Zchori-Fein E, Mozes Daube N, Kontsedalov S, Skaljac M, Brumin M, Sobol I, Czosnek H, Vavre F, Fleury F, Ghanim M (2010) The transmission efficiency of Tomato yellow leaf curl virus by the whitefly Bemisia tabaci is correlated with the presence of a specific symbiotic bacterium species. J Virol 84:9310–9317
Rana VS, Singh ST, Priya NG, Kumar J, Rajagopal R (2012) Arsenophonus GroEL interacts with CLCuV and is localized in midgut and salivary gland of whitefly B. tabaci. PLoS One 7:e42168
Kliot A, Cilia M, Czosnek H, Ghanim M (2014) Implication of the Bacterial Endosymbiont Rickettsia spp. In interactions of the whitefly Bemisia tabaci with Tomato yellow leaf curl virus. Virology 88:5652–5660
Sloan DB, Moran NA (2012) Endosymbiotic bacteria as a source of carotenoids in whiteflies. Biol Lett 8:986–989
Jiang ZF, Xia FF, Johnson KW, Bartom E, Tuteja JH, Stevens R, Grossman RL, Brumin M, White KP, Ghanim M (2012) Genome sequences of the primary endosymbiont Candidatus Portiera aleyrodidarum in the whitefly Bemisia tabaci B and Q biotypes. J Bacteriol 194:6678–6679
Santos-Garcia D, Farnier PA, Beitia F, Zchori-Fein E, Vavre F, Mouton L, Moya A, Latorre A, Silva FJ (2012) Complete genome sequence of “Candidatus Portiera aleyrodidarum” BT-QVLC, an obligate symbiont that supplies amino acids and carotenoids to Bemisia tabaci. J Bacteriol 194:6654–6655
Rao Q, Wang S, Zhu DT, Wang XW, Liu SS (2012) Draft genome sequence of Rickettsia sp. strain MEAM1, isolated from the whitefly Bemisia tabaci. J Bacteriol 194:4741–4742
Santos-Garcia D, Latorre A, Moya A, Gibbs G, Hartung V, Dettner K, Kuechler SM, Silva FJ (2014) Small but powerful, the primary endosymbiont of moss bugs, Candidatus Evansia muelleri, holds a reduced genome with large biosynthetic capabilities. Genome Biol Evol 6:1875–1893
Guo H, Qu Y, Liu X, Zhong W, Fang J (2014) Female-biased symbionts and tomato yellow leaf curl virus infections in Bemisia tabaci. PLoS One 9:e84538
Morales FJ, Anderson PK (2001) The emergence and dissemination of whitefly-transmitted geminiviruses in Lain America. Arch Virol 146:415–441
Mansoor S, Briddon RW, Bull SE, Bedford ID, Bashir A, Hussain M, Saeed Zafar MY, Malik KA, Fauquet C, Markham PG (2003) Cotton leaf curl disease is associated with multiple monopartite begomoviruses supported by a single DNA b. Arch Virol 148:1969–1986
Varma A, Malathi VG (2003) Emerging geminivirus problems: a serious threat to crop production. Ann Appl Biol 142:145–164
Fargette D, Konate G, Fauquet C, Muller E, Peterschmitt M, Thresh JM (2006) Molecular ecology and emergence of tropical plant viruses. Annu Rev Phytopathol 44:235–260
Rojas MR, Gilbertson RL (2008) Emerging plant viruses: a diversity of mechanisms and opportunities. In: Roossinck MJ (ed) Plant virus evolution. Springer, Berlin/Heidelberg, pp 27–52
Hogenhout SA, Ammar ED, Whitfield AE, Redinbaugh MG (2008) Insect vector interactions with persistently transmitted viruses. Annu Rev Phytopathol 46:327–359
Ghanim M, Morin S, Czosnek H (2001) Rate of Tomato yellow leaf curl virus (TYLCV) translocation in the circulative transmission pathway of its vector, the whitefly Bemisia tabaci. Phytopathology 91:188–196
Briddon RW, Pinner MS, Stanley J, Markham PG (1990) Geminivirus coat protein gene replacement alters insect specificity. Virology 177:85–94
Azzam O, Frazer J, De La Rosa D, Beaver JS, Ahlquist P, Maxwell DP (1994) Whitefly transmission and efficient ssDNA accumulation of bean golden mosaic geminivirus require functional coat protein. Virology 204:289–296
Liu S, Bedford ID, Briddon RW, Markham PG (1997) Efficient whitefly transmission of bipartite geminiviruses requires both genomic components. J Gen Virol 78:1791–1794
Höfer P, Bedford ID, Markham PG, Jeske H, Frischmuth T (1997) Coat protein gene replacement results in whitefly transmission of an insect nontransmissible geminivirus isolate. Virology 236:288–295
Noris E, Vaira AM, Caciagli P, Masenga V, Gronenborn B, Accotto GP (1998) Amino acids in the capsid protein of tomato yellow leaf curl virus that are crucial for systemic infection, particle formation, and insect transmission. J Virol 72:10050–10057
Caciagli P, Medina Piles V, Marian D, Vecchiati M, Masenga V, Mason G, Falcioni T, Noris E (2009) Virion stability is important for the circulative transmission of Tomato yellow leaf curl Sardinia virus by Bemisia tabaci, but virion access to salivary glands does not guarantee transmissibility. J Virol 83:5784–5795
Kheyr-Pour A, Bananej K, Dafalla GA, Caciagli P, Noris E, Ahoonmanesh A, Lecoq H, Gronenborn B (2000) Watermelon chlorotic stunt virus from the Sudan and Iran: sequence comparisons and identification of a whitefly-transmission determinant. Phytopathology 90:629–635
Höhnle M, Höfer P, Bedford ID, Briddon RW, Markham PG, Frischmuth T (2001) Exchange of three amino acids in the coat protein results in efficient whitefly transmission of a nontransmissible Abutilon mosaic virus isolate. Virology 290:164–171
Ohnesorge S, Bejarano ER (2009) Begomovirus coat protein interacts with a small heat-shock protein of its transmission vector (Bemisia tabaci). Insect Mol Biol 18:693–703
Gotz M, Popovski S, Kollenberg M, Gorovits R, Brown JK, Cicero J, Czosnek H, Winter S, Ghanim M (2012) Implication of Bemisia tabaci heat shock protein 70 in begomovirus-whitefly interactions. J Virol 86:13241–13252
Walling LL (2000) The myriad plant responses to herbivores. J Plant Growth Regul 19:195–216
Pieterse CMJ, Dicke M (2007) Plant interactions with microbes and insects: from molecular mechanisms to ecology. Trends Plant Sci 12:564–568
Green TR, Ryan CA (1972) Wound-induced proteinase inhibitor in plant leaves: a possible defense mechanism against insects. Science 175:776–777
Thaler JS, Karban R, Ullman DE, Boege K, Bostock RM (2002) Cross-talk between jasmonate and salicylate plant defense pathways: effects on several plant parasites. Oecologia 131:227–235
Ryan CA (1990) Protease inhibitors in plants: genes for improving defenses against insects and pathogens. Annu Rev Phytopathol 28:425–449
Zhang HY, Xie XZ, Xu YZ, Wu NH (2004) Isolation and functional assessment of a tomato proteinase inhibitor II gene. Plant Physiol Biochem 42:437–444
Moran PJ, Thompson GA (2001) Molecular responses to aphid feeding in Arabidopsis in relation to plant defense pathways. Plant Physiol 125:1074–1085
Zarate SI, Kempema LA, Walling LL (2007) Silverleaf whitefly induces salicylic acid defenses and suppresses effectual jasmonic acid defenses. Plant Physiol 143:866–875
Kempema LA, Cui X, Holzer FM, Walling LL (2007) Arabidopsis transcriptome changes in response to phloem-feeding silverleaf whitefly nymphs. Similarities and distinctions in responses to aphids. Plant Physiol 143:849–865
Zhang T, Luan JB, Qi JF, Huang CJ, Li M, Zhou XP, Liu SS (2012) Begomovirus-whitefly mutualism is achieved through repression of plant defenses by a virus pathogenicity factor. Mol Ecol 21:1294–1304
Ascencio-Ibanez JT, Sozzani R, Lee TJ, Chu TM, Wolfinger RD, Cella R, Hanley-Bowdoin L (2008) Global analysis of Arabidopsis gene expression uncovers a complex array of changes impacting pathogen response and cell cycle during geminivirus infection. Plant Physiol 148:436–454
Guo JY, Dong SZ, Yang X, Cheng L, Wan FH et al (2012) Enhanced vitellogenesis in a whitefly via feeding on a begomovirus-infected plant. PLoS One 7:e43567
Shi X, Pan H, Xie W, Wu Q, Wang S, Liu Y, Fang Y, Chen G, Gao X, Zhang Y (2013) Plant virus differentially alters the plant’s defense response to its closely related vectors. PLoS One 8:e83520
Shi X, Pan H, Xie W, Jiao X, Fang Y, Chen G, Yang X, Wu Q, Wang S, Zhang Y (2014) Three-way interactions between the tomato plant, tomato yellow leaf curl virus, and the whitefly Bemisia tabaci facilitate virus spread. J Econ Entomol 107:920–926
Shi X, Pan H, Zhang H, Jiao X, Xie W, Wu Q, Wang S, Fang Y, Chen G, Zhou X, Zhang Y (2014) Bemisia tabaci Q carrying tomato yellow leaf curl virus strongly suppresses host plant defenses. Sci Rep 4:5230
Gerling D, Alomar O, Arno J (2001) Biological control of Bemisia tabaci using predators and parasitoids. Crop Prot 20:779–799
Huang J, Zheng QH, Fu JW, Huang PY, Gu DX (2000) Investigation and identification of the whitefly parasitoids (Hymenoptera: Aphelinidae, Platygasteridae). Entomol J East China 9:29–33
Yang NW, Wan FH (2011) Host suitability of different instars of Bemisia tabaci biotype B for the parasitoid Eretmocerus hayati. Biol Control 2:313–317
Xu HY, Yang NW, Wan FH (2013) Competitive interactions between parasitoids provide new insight into host suppression. PLoS One 8:e82003
Liu X, Xiang W, Jiao X, Zhang Y, Xie W, Wu Q, Zhou X, Wang S (2014) Effects of plant virus and its insect vector on ‑Encarsia formosa, a biocontrol agent of whiteflies. Sci Rep 4:5926
Zhang CR, Zhang S, Xia J, Li FF, Xia WQ, Liu SS, Wang XW (2014) The immune strategy and stress response of the Mediterranean species of the Bemisia tabaci complex to an orally delivered bacterial pathogen. PLoS One 9:e94477
Xia J, Zhang C, Zhang S, Li F, Feng M et al (2013) Analysis of whitefly transcriptional responses to Beauveria bassiana infection reveals new insights into insect-fungus interactions. PLoS One 8:e68185
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Kanakala, S., Ghanim, M. (2015). Advances in the Genomics of the Whitefly Bemisia tabaci: An Insect Pest and a Virus Vector. In: Raman, C., Goldsmith, M., Agunbiade, T. (eds) Short Views on Insect Genomics and Proteomics. Entomology in Focus, vol 3. Springer, Cham. https://doi.org/10.1007/978-3-319-24235-4_2
Download citation
DOI: https://doi.org/10.1007/978-3-319-24235-4_2
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-24233-0
Online ISBN: 978-3-319-24235-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)