Abstract
In the most severe forms of acute respiratory distress syndrome, profound gas-exchange abnormalities threatening patients’ lives can occur despite using conventional salvage therapies. In this situation, venovenous extracorporeal membrane oxygenation was developed to rescue these dying patients. ECMOpermit “ultraprotective” mechanical ventilation with further reduction of volume and pressure that might ultimately enhance lung protection and improve clinical outcomes.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
1 Introduction
Mechanical ventilation remains the cornerstone of respiratory support for acute respiratory failure (ARF) patients. However, high pressure and volume associated with tidal ventilation are known to aggravate lung injury in this setting [1]. In the most severe forms of the disease, profound gas-exchange abnormalities threatening patients’ lives can occur despite using the conventional salvage therapies [2, 3]. Venovenous extracorporeal membrane oxygenation (VV-ECMO) was developed more than 40 years ago [4, 5] to rescue these dying patients. ECMO also permit “ultraprotective” mechanical ventilation with further reduction of volume and pressure that might ultimately enhance lung protection and improve clinical outcomes of ECMO [6, 7]. More recently, the successful use of ECMO for the most severe ARDS cases associated with the recent influenza A (H1N1) pandemic who failed on conventional ventilation [8–10] and positive results of the randomized CESAR trial [11] have been associated with a steep increase in the number of VV-ECMO procedures performed in very recent years.
2 How Does It Work?
2.1 Settings
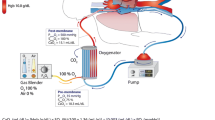
In recent years, major technological advances occurred, and the latest ECMO devices with polymethylpentene hollow-fiber membrane lungs and Mendler-designed centrifugal pumps offer lower resistance to blood flow, have smaller priming volumes, higher effective gas exchange properties, and are coated with more biocompatible materials. The extracorporeal system consisted of polyvinyl chloride tubing, a membrane oxygenator, and a centrifugal pump. An oxygen-air blender is used to ventilate the membrane oxygenator (2–14 l.min-1). Venovenous ECMO provides complete extracorporeal blood oxygenation and decarboxylation using high blood flows (4–6 L/min) and large (20–30 Fr) cannulas [12–15]. Blood is usually drained from the right atrium or the inferior vena cava through a multiperforated cannula inserted percutaneously into the right femoral vein and is returned to the superior vena cava through a cannula inserted percutaneously into the right internal jugular vein (Femorojugular setting) or in the right atrium through a cannula inserted into the femoral vein (Femoral–femoral setting). During the procedure, using transthoracic or transesophageal echocardiography is fostered to properly set the position of the drainage cannula.
2.2 Determinants of Oxygenation on VV-ECMO
The main determinants of oxygen delivery (DO2) to peripheral tissues, which is critical to preserve organ function, are hemoglobin concentration, SaO2, and cardiac index [16]. When DO2 falls below a critical threshold, oxygen consumption becomes dependent on DO2 and lactate concentration may increase, reflecting activation of anaerobic metabolism. To prevent tissue hypoxia, recommended oxygenation objective is to maintain SaO2 ≥88% using high PEEP and high FiO2 in mechanically ventilated ARDS patients [17, 18]. However, when refractory hypoxemia develops, recourse to VV-ECMO is a reasonable therapeutic option [8, 9, 11, 19, 20]. In this circumstance, blood oxygenation may become completely dependent on membrane oxygenator oxygen transfer capability. Factors determining oxygenator oxygen transfer in this setting are blood oxygen saturation in the ECMO drainage cannula, hemoglobin concentration, blood flow in the ECMO circuit, and intrinsic membrane oxygenator properties, which depend on the exchange membrane surface and diffusibility of O2 through hollow microfibers. O2 transfer through recent modern oxygenator is theoretically >400 ml O2/min when blood flow through the ECMO circuit is >6 l/min, while oxygen saturation in the ECMO drainage cannula is 70% and hemoglobin concentration is 15 g/dl [21]. However, since both drainage and return cannulae are positioned within the venous system in VV-ECMO, blood recirculation into the oxygenator occurs, that is, a proportion of returned blood is drained again into the circuit instead of passing through the right heart, thus markedly reducing O2 transfer efficiency [22]. To minimize blood recirculation into the circuit, it can be configured in several ways [19, 20]. In the bifemoral setting, drainage cannula is positioned in the inferior vena cava (IVC), and a femoral return cannula is advanced to the right atrium (see Fig. 3.1a). However, 50% of the patients who received bifemoral VV-ECMO for H1N1-induced ARDS in the ANZICS ICUs also needed a second (jugular) drainage cannula, because of insufficient blood drainage [8]. Alternatively, a single bicaval dual-lumen cannula (Avalon Elite®) can be inserted via the right jugular vein and positioned to allow drainage from the IVC and SVC and oxygenated blood return via a second lumen in the right atrium [23] (see Fig. 3.1b). This setting minimizes blood recirculation, but insertion of the jugular catheter requires an experienced and skilled operator and recourse to fluoroscopy or TEE guidance for its adequate positioning. Lastly, femorojugular setting for VV-ECMO allows minimizing blood recirculation if the tip of the return cannula is positioned away from that of the inflow cannula. To achieve this goal, mean distance between both cannulae should be measured on the chest X-ray. A minimal distance of 12 cm is generally advocated. Additionally, it has been shown in a previous study that, compared to the jugulofemoral configuration, the femorojugular bypass provided higher maximal ECMO flow, higher pulmonary arterial mixed venous oxygen saturation, and required comparatively less flow to maintain an equivalent mixed venous oxygen saturation [24].
Single-site and two-site approaches to venovenous ECMO cannulation (With permission [13] (du NEJM)). (a) A two-site approach to venovenous ECMO cannulation. (b) A single-site approach to venovenous ECMO cannulation.
To improve oxygen blood transfer in the oxygenator and to increase oxygen transport to peripheral organs, a recent study has demonstrated that besides ECMO cannulae configuration, ECMO flow through the ECMO circuit is the major determinant of blood oxygenation. ECMO flow >60% of systemic blood flow permitted adequate peripheral oxygenation [25]. Thus, depending on the patient size, cardiac output, oxygen consumption, and lung shunt, circuit blood flow between 4–7 l/min will typically be required to achieve arterial oxygen saturations >88–90%, while maintaining safe lung ventilation. Therefore, large size (24–30 Fr) and multihole drainage cannula should be preferred to obtain high flows with reasonable negative pressure in the drainage cannula. Indeed, if small cannulae are used with high flows, the suction created by the centrifugal pump can cause excessive depression and cavitation in the inflow line resulting in massive intravascular hemolysis [19, 20]. Physiological in vivo study demonstrates that, for patients who received VV-ECMO for refractory hypoxemia and whose native lung gas exchange function was almost completely abolished, the determining factors of arterial oxygenation are VV-ECMO blood flow and FiO2ECMO . Specifically, using the femorojugular ECMO setting, achieving VV-ECMO flow >60% of systemic blood flow was constantly associated with arterial blood saturation >90%.
The other important parameter that might be manipulated to enhance tissue oxygen delivery and maximize extracorporeal circuit efficiency is blood hemoglobin concentration [16] (Table 3.1). In patients under ECMO support, guidelines from the Extracorporeal Life Support Organization (ELSO) and investigators of the CESAR trial recommend maintaining normal hematocrit (40–45%) and hemoglobin concentrations at 14 g/dl, respectively [11, 26]. However, critically ill patients and specifically those already suffering from diffuse alveolar damage may be at even greater risk of transfusion-related acute lung injury [27–29]. Accordingly, a restrictive transfusion strategy with red-cell transfusion threshold set at 7–8 g/dl in most patients under ECMO is doable. Schmidt et al. demonstrated that despite mean hemoglobin concentration and DO2 at 8.0 g.dl-1 and 679 ml/min, respectively, every patient had adequate SaO2, and no sign of VO2/DO2 mismatch was observed [25]. Lastly, transfusion of blood products increases volemia, which might also complicate the course of ARDS, since a study reported slower lung function improvement and longer mechanical ventilation duration when a liberal strategy of fluid management was used in patients with acute lung injury [30].
2.3 Determinants of Decarboxylation on VV-ECMO
The determining factor of blood decarboxylation is the rate of sweep gas flow ventilating the membrane lung, while PaCO2 is unaffected when ECMO blood flow and FiO2ECMO are reduced to <2.5 l/min and 40%, respectively.
CO2 transfer through the membrane lung also depends on ECMO flow, with maximum transfer being >300 ml/min when ECMO flow is >6 l/min with the Quadrox® oxygenator. However, since CO2 diffuses 20 times faster than O2, large amount of CO2 can be exchanged through the membrane lung even when low flow is applied through the circuit [25]. For instance, recent data showed that PaCO2 remained unchanged when ECMO blood flow was reduced to <2.5 l/min. Indeed, this property is the basis for developing low-flow extracorporeal CO2 removal devices, for which CO2 removal is >70 ml/min at blood flows of only 450 ml/min [31, 32]. Alternatively, sweep gas flow across the oxygenator is the main determinant of CO2 removal by ECMO [25].
3 Main Indications of VV-ECMO for Severe ARDS
Indications are usually based on: (1) severe hypoxemia (e.g., PaO2 to FiO2 ratio <80 mmHg, despite optimization of mechanical ventilation (tidal volume set at 6 ml/kg and trial of PEEP≥10 cm H2O)) for at least 6 h in patients with potentially reversible respiratory failure and possible recourse to adjunctive therapies (NO, prone position, etc.) and/or or (2) uncompensated hypercapnia with acidemia (pH <7.15) despite the best accepted standard of care for management with a ventilator and/or (3) excessively high end-inspiratory plateau pressure (>32 cm of water). However, considering the CESAR trial, the ongoing EOLIA trial, and the recommendations of the Extra Life Support Organization (ELSO), the thresholds of PaO2 to FiO2 ratio, pH, or plateau pressure may vary considerably across studies and guidelines.
Relative contraindications are usually mechanical ventilation for more than 7 days, limited vascular access, and any condition or organ dysfunction that would limit the likelihood of overall benefit from ECMO, such as malignancies with fatal prognosis within 5 years, moribund patients, or those with irreversible neurological pathologies and decisions to limit therapeutic interventions. Contraindication to the use of anticoagulation therapy is mentioned in several reviews or guidelines. However, several publications have stressed that, while using new-coated heparin circuit, anticoagulation on ECMO VV may be safely withheld for days or weeks.
4 Recent Data of ECMO VV in ARDS
The most recent trial (CESAR trial) which was conducted in the UK from 2001 to 2006 evaluated a strategy of transfer to a single center (Glenfield, Leicester) which had ECMO capability, while the patients randomized to the control group were treated conventionally at designated treatment centers [6]. The primary endpoint combining mortality or severe disability 6 months after randomization was lower for the 90 patients randomized to the ECMO group (37% vs. 53%, p = 0.03). However, results of that trial should be analyzed carefully. First, 22 patients randomized to the ECMO arm did not receive ECMO (died before or during transport, improved with conventional management at the referral center, or had a contraindication to heparin). Second, no standardized protocol for lung-protective mechanical ventilation existed in the control group, and the time spent with “protective” mechanical ventilation was significantly higher in the ECMO arm. Third, more patients received corticosteroids in the ECMO group. In the most recent series, patients benefited from the latest ECMO technology, which include a centrifugal pump, a polymethylpentene membrane oxygenator, and tubing with biocompatible surface treatment. Mortality rates ranged from 36 to 56% in the studies performed in the last 15 years and reporting outcomes of >30 ECMO patients (Table 3.1). Interestingly, ECMO was provided through a mobile ECMO rescue team in some of these studies. For example, in a series of 124 patients treated at a Danish center between 1997 and 2011 [33], survival was 71%, and 85% of these patients received ECMO via a mobile unit before being transferred to the referral hospital. Similarly, in the Regensburg cohort, 59/176 received ECMO at another hospital by a mobile unit [34]. In a multicenter French cohort of 140 patients treated between 2008 and 2012, 68% patients were retrieved via a mobile ECMO team, and their prognosis was comparable to those who received VV-ECMO support in their initial center hospital [35]. ECMO support might also cause severe and potentially life-threatening complications, such as bleeding, infections, intravascular hemolysis, thrombocytopenia, or consumption coagulopathy [35–39].
Mortality rates of ECMO for pandemic influenza A (H1N1)-associated ARDS ranged from 14 to 64% in the 16 studies from 11 countries reporting on the experience of ECMO for influenza A (H1N1)-associated ARDS [8–10, 35, 40–50]. The Australia and New Zealand collaborative group (ANZICS) was the first to report its experience [8]. Despite extreme disease severity at the time of ECMO initiation (median PaO2/FiO2 ratio 56 mmHg, median positive end-expiratory pressure [PEEP] at 18 cm H2O, and median lung injury score of 3.8), only 25% of the 68 ECMO patients died. A British collaborative cohort series [9] depicted the outcome of 80 patients transferred into ECMO referral centers in United Kingdom of whom 69 received ECMO. Mortality in this cohort was 27.5%. A propensity-matched analysis comparing survival of patients referred for consideration of ECMO to other ARDS patients showed better outcomes for referred patients. Alternatively, mortality of propensity-matched patients treated conventionally was comparable to that of ECMO patients in French ICUs of the REVA network. However, only 50% of ECMO patients were successfully matched with control ARDS patients, while unmatched ECMO patients were younger, suffered more severe respiratory failure, and had considerably lower mortality [10]. Interestingly, a higher plateau pressure under ECMO was independently associated with mortality, indicating for the first time that an ultraprotective ventilation strategy with reduction of plateau pressure to around 25 cm H2O following ECMO installation might improve outcomes. Lastly, mortality was 29% on a cohort of 49 proven influenza A (H1N1) patients from the 14 ECMO centers of the ECMO-NET Italian collaborative group [51]. In this series, patients ventilated for <7 days before ECMO initiation had a significantly higher survival.
5 Mortality Risk Factors and Predictive Survival Models
Factors associated with poor outcomes after ECMO for acute respiratory failure include older age [34–36, 52–55], a greater number of days of mechanical ventilation before the ECMO establishment [35, 36, 52, 53, 55], a higher number of organ failure [34–36, 52–55], low pre-ECMO respiratory system compliance [55], as well as immunosuppression [35, 55, 56]. Predictive survival models have been recently developed which might help clinicians select appropriate candidates for ECMO [35, 54–57]. For instance, the RESP-score [55] constructed on data extracted from a large multicenter international population (n = 2355) computes 12 simple pre-ECMO parameters to provide a relevant and validated tool predicting survival after ECMO for acute respiratory failure. Cumulative predicted hospital survival were 92, 76, 57, 33, and 18% for five RESP-score risk class I (≥6), II (3 to 5), III (−1 to 2), IV (−5 to −2), and V (≤−6), respectively.
6 Conclusions
Recent technological advances have improved the safety and the simplicity of ECMO use in ARDS. In addition, mobile ECMO team has made this therapy more accessible for all patients. Actual literature has reported that early implementation of VV-ECMO in refractory and severe ARDS can strongly reduce pressures and volumes applied on the alveoli in order to minimize ventilation-induced lung injury. However, strong evidence of its benefit and optimal timing for cannulation are still lacking. Therefore, results of next multicenter randomized trials (i.e EOLIA trial) are needed before wide spreading this promising technology.
References
Dreyfuss D, Soler P, Basset G, Saumon G. High inflation pressure pulmonary edema. Respective effects of high airway pressure, high tidal volume, and positive end-expiratory pressure. Am Rev Respir Dis. 1988;137(5):1159–64.
Guerin C, Reignier J, Richard JC, Beuret P, Gacouin A, Boulain T, et al. Prone positioning in severe acute respiratory distress syndrome. N Engl J Med. 2013;368(23):2159–68.
Papazian L, Forel JM, Gacouin A, Penot-Ragon C, Perrin G, Loundou A, et al. Neuromuscular blockers in early acute respiratory distress syndrome. N Engl J Med. 2010;363(12):1107–16.
Hill JD, O’Brien TG, Murray JJ, Dontigny L, Bramson ML, Osborn JJ, et al. Prolonged extracorporeal oxygenation for acute post-traumatic respiratory failure (shock-lung syndrome). Use of the Bramson membrane lung. N Engl J Med. 1972;286(12):629–34.
Kolobow T, Zapol W, Pierce JE, Keeley AF, Replogle RL, Haller A. Partial extracorporeal gas exchange in alert newborn lambs with a membrane artificial lung perfused via an A-V shunt for periods up to 96 hours. Trans Am Soc Artif Intern Organs. 1968;14:328–34.
Zapol WM, Snider MT, Hill JD, Fallat RJ, Bartlett RH, Edmunds LH, et al. Extracorporeal membrane oxygenation in severe acute respiratory failure. A randomized prospective study. JAMA. 1979;242(20):2193–6.
Morris AH, Wallace CJ, Menlove RL, Clemmer TP, Orme Jr JF, Weaver LK, et al. Randomized clinical trial of pressure-controlled inverse ratio ventilation and extracorporeal CO2 removal for adult respiratory distress syndrome. Am J Respir Crit Care Med. 1994;149(2 Pt 1):295–305.
Davies A, Jones D, Bailey M, Beca J, Bellomo R, Blackwell N, et al. Extracorporeal membrane oxygenation for 2009 influenza A(H1N1) acute respiratory distress syndrome. JAMA. 2009;302(17):1888–95.
Noah MA, Peek GJ, Finney SJ, Griffiths MJ, Harrison DA, Grieve R, et al. Referral to an extracorporeal membrane oxygenation center and mortality among patients with severe 2009 influenza A(H1N1). JAMA. 2011;306(15):1659–68.
Pham T, Combes A, Roze H, Chevret S, Mercat A, Roch A, et al. Extracorporeal membrane oxygenation for pandemic influenza A(H1N1)-induced acute respiratory distress syndrome: a cohort study and propensity-matched analysis. Am J Respir Crit Care Med. 2013;187(3):276–85.
Peek GJ, Mugford M, Tiruvoipati R, Wilson A, Allen E, Thalanany MM, et al. Efficacy and economic assessment of conventional ventilatory support versus extracorporeal membrane oxygenation for severe adult respiratory failure (CESAR): a multicentre randomised controlled trial. Lancet. 2009;374(9698):1351–63.
Combes A, Brechot N, Luyt CE, Schmidt M. What is the niche for extracorporeal membrane oxygenation in severe acute respiratory distress syndrome? Curr Opin Crit Care. 2012;18(5):527–32.
Brodie D, Bacchetta M. Extracorporeal membrane oxygenation for ARDS in adults. N Engl J Med. 2012;365(20):1905–14.
Combes A, Bacchetta M, Brodie D, Muller T, Pellegrino V. Extracorporeal membrane oxygenation for respiratory failure in adults. Curr Opin Crit Care. 2012;18(1):99–104.
MacLaren G, Combes A, Bartlett RH. Contemporary extracorporeal membrane oxygenation for adult respiratory failure: life support in the new era. Intensive Care Med. 2012;38(2):210–20.
Vincent JL, De Backer D. Oxygen transport-the oxygen delivery controversy. Intensive Care Med. 2004;30(11):1990–6.
The. Acute Respiratory Distress Syndrome Network. Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. N Engl J Med. 2000;342(18):1301–8.
Mercat A, Richard JC, Vielle B, Jaber S, Osman D, Diehl JL, et al. Positive end-expiratory pressure setting in adults with acute lung injury and acute respiratory distress syndrome: a randomized controlled trial. JAMA. 2008;299(6):646–55.
Combes A, Bacchetta M, Brodie D, Muller T, Pellegrino V. Extracorporeal membrane oxygenation for respiratory failure in adults. Curr Opin Crit Care. 2011;18(1):99–104.
Maclaren G, Combes A, Bartlett RH. Contemporary extracorporeal membrane oxygenation for adult respiratory failure: life support in the new era. Intensive Care Med. 2011;38(2):210–20.
Jegger D, Tevaearai HT, Mallabiabarrena I, Horisberger J, Seigneul I, von Segesser LK. Comparing oxygen transfer performance between three membrane oxygenators: effect of temperature changes during cardiopulmonary bypass. Artif Organs. 2007;31(4):290–300.
Walker JL, Gelfond J, Zarzabal LA, Darling E. Calculating mixed venous saturation during veno-venous extracorporeal membrane oxygenation. Perfusion. 2009;24(5):333–9.
Bermudez CA, Rocha RV, Sappington PL, Toyoda Y, Murray HN, Boujoukos AJ. Initial experience with single cannulation for venovenous extracorporeal oxygenation in adults. Ann Thorac Surg. 2010;90(3):991–5.
Rich PB, Awad SS, Crotti S, Hirschl RB, Bartlett RH, Schreiner RJ. A prospective comparison of atrio-femoral and femoro-atrial flow in adult venovenous extracorporeal life support. J Thorac Cardiovasc Surg. 1998;116(4):628–32.
Schmidt M, Tachon G, Devilliers C, Muller G, Hekimian G, Brechot N, et al. Blood oxygenation and decarboxylation determinants during venovenous ECMO for respiratory failure in adults. Intensive Care Med. 2013;39(5):838–46.
ELSO. guidelines. http://www.elsomedumichedu/Guidelineshtml.
Vlaar AP, Hofstra JJ, Determann RM, Veelo DP, Paulus F, Kulik W, et al. The incidence, risk factors, and outcome of transfusion-related acute lung injury in a cohort of cardiac surgery patients: a prospective nested case-control study. Blood. 2011;117(16):4218–25.
Gong MN, Thompson BT, Williams P, Pothier L, Boyce PD, Christiani DC. Clinical predictors of and mortality in acute respiratory distress syndrome: potential role of red cell transfusion. Crit Care Med. 2005;33(6):1191–8.
Gajic O, Rana R, Winters JL, Yilmaz M, Mendez JL, Rickman OB, et al. Transfusion-related acute lung injury in the critically ill: prospective nested case-control study. Am J Respir Crit Care Med. 2007;176(9):886–91.
Wiedemann HP, Wheeler AP, Bernard GR, Thompson BT, Hayden D, deBoisblanc B, et al. Comparison of two fluid-management strategies in acute lung injury. N Engl J Med. 2006;354(24):2564–75.
Terragni PP, Del Sorbo L, Mascia L, Urbino R, Martin EL, Birocco A, et al. Tidal volume lower than 6 ml/kg enhances lung protection: role of extracorporeal carbon dioxide removal. Anesthesiology. 2009;111(4):826–35.
Batchinsky AI, Jordan BS, Regn D, Necsoiu C, Federspiel WJ, Morris MJ, et al. Respiratory dialysis: reduction in dependence on mechanical ventilation by venovenous extracorporeal CO2 removal. Crit Care Med. 2011;39(6):1382–7.
Lindskov C, Jensen RH, Sprogoe P, Klaaborg KE, Kirkegaard H, Severinsen IK, et al. Extracorporeal membrane oxygenation in adult patients with severe acute respiratory failure. Acta Anaesthesiol Scand. 2013;57(3):303–11.
Schmid C, Philipp A, Hilker M, Rupprecht L, Arlt M, Keyser A, et al. Venovenous extracorporeal membrane oxygenation for acute lung failure in adults. J Heart Lung Transplant. 2012;31(1):9–15.
Schmidt M, Zogheib E, Roze H, Repesse X, Lebreton G, Luyt CE, et al. The PRESERVE mortality risk score and analysis of long-term outcomes after extracorporeal membrane oxygenation for severe acute respiratory distress syndrome. Intensive Care Med. 2013;39(10):1704–13.
Brogan TV, Thiagarajan RR, Rycus PT, Bartlett RH, Bratton SL. Extracorporeal membrane oxygenation in adults with severe respiratory failure: a multi-center database. Intensive Care Med. 2009;35(12):2105–14.
Forrest P, Ratchford J, Burns B, Herkes R, Jackson A, Plunkett B, et al. Retrieval of critically ill adults using extracorporeal membrane oxygenation: an Australian experience. Intensive Care Med. 2011;37(5):824–30.
Muller T, Philipp A, Luchner A, Karagiannidis C, Bein T, Hilker M, et al. A new miniaturized system for extracorporeal membrane oxygenation in adult respiratory failure. Crit Care. 2009;13(6):R205.
Schmidt M, Brechot N, Hariri S, Guiguet M, Luyt CE, Makri R, et al. Nosocomial infections in adult cardiogenic shock patients supported by venoarterial extracorporeal membrane oxygenation. Clin Infect Dis. 2012;55(12):1633–41.
Chan KK, Lee KL, Lam PK, Law KI, Joynt GM, Yan WW. Hong Kong’s experience on the use of extracorporeal membrane oxygenation for the treatment of influenza A (H1N1). Hong Kong Med J Hong Kong Acad Med. 2010;16(6):447–54.
D’Ancona G, Capitanio G, Chiaramonte G, Serretta R, Turrisi M, Pilato M, et al. Extracorporeal membrane oxygenator rescue and airborne transportation of patients with influenza A (H1N1) acute respiratory distress syndrome in a Mediterranean underserved area. Interact Cardiovasc Thorac Surg. 2011;12(6):935–7.
Freed DH, Henzler D, White CW, Fowler R, Zarychanski R, Hutchison J, et al. Extracorporeal lung support for patients who had severe respiratory failure secondary to influenza A (H1N1) 2009 infection in Canada. Can J Anaesth. 2010;57(3):240–7.
Holzgraefe B, Broome M, Kalzen H, Konrad D, Palmer K, Frenckner B. Extracorporeal membrane oxygenation for pandemic H1N1 2009 respiratory failure. Minerva Anestesiol. 2010;76(12):1043–51.
Roch A, Lepaul-Ercole R, Grisoli D, Bessereau J, Brissy O, Castanier M, et al. Extracorporeal membrane oxygenation for severe influenza A (H1N1) acute respiratory distress syndrome: a prospective observational comparative study. Intensive Care Med. 2010;36(11):1899–905.
Roncon-Albuquerque Jr R, Basilio C, Figueiredo P, Silva S, Mergulhao P, Alves C, et al. Portable miniaturized extracorporeal membrane oxygenation systems for H1N1-related severe acute respiratory distress syndrome: a case series. J Crit Care. 2012;27(5):454–63.
Takeda S, Kotani T, Nakagawa S, Ichiba S, Aokage T, Ochiai R, et al. Extracorporeal membrane oxygenation for 2009 influenza A(H1N1) severe respiratory failure in Japan. J Anesth. 2012;26(5):650–7.
Turner DA, Rehder KJ, Peterson-Carmichael SL, Ozment CP, Al-Hegelan MS, Williford WL, et al. Extracorporeal membrane oxygenation for severe refractory respiratory failure secondary to 2009 H1N1 influenza A. Respir Care. 2011;56(7):941–6.
Hou X, Guo L, Zhan Q, Jia X, Mi Y, Li B, et al. Extracorporeal membrane oxygenation for critically ill patients with 2009 influenza A (H1N1)-related acute respiratory distress syndrome: preliminary experience from a single center. Artif Organs. 2012;36(9):780–6.
Michaels AJ, Hill JG, Bliss D, Sperley BP, Young BP, Quint P, et al. Pandemic flu and the sudden demand for ECMO resources: a mature trauma program can provide surge capacity in acute critical care crises. J Trauma Acute Care Surg. 2013;74(6):1493–7.
Bonastre J, Suberviola B, Pozo JC, Guerrero JE, Torres A, Rodriguez A, et al. Extracorporeal lung support in patients with severe respiratory failure secondary to the 2010-2011 winter seasonal outbreak of influenza A (H1N1) in Spain. Med Int. 2012;36(3):193–9.
Patroniti N, Zangrillo A, Pappalardo F, Peris A, Cianchi G, Braschi A, et al. The Italian ECMO network experience during the 2009 influenza A(H1N1) pandemic: preparation for severe respiratory emergency outbreaks. Intensive Care Med. 2011;37(9):1447–57.
Beiderlinden M, Eikermann M, Boes T, Breitfeld C, Peters J. Treatment of severe acute respiratory distress syndrome: role of extracorporeal gas exchange. Intensive Care Med. 2006;32(10):1627–31.
Hemmila MR, Rowe SA, Boules TN, Miskulin J, McGillicuddy JW, Schuerer DJ, et al. Extracorporeal life support for severe acute respiratory distress syndrome in adults. Ann Surg. 2004;240(4):595–605. ; discussion -7
Roch A, Hraiech S, Masson E, Grisoli D, Forel JM, Boucekine M, et al. Outcome of acute respiratory distress syndrome patients treated with extracorporeal membrane oxygenation and brought to a referral center. Intensive Care Med. 2014;40(1):74–83.
Schmidt M, Bailey M, Sheldrake J, Hodgson C, Aubron C, Rycus PT, et al. Predicting survival after extracorporeal membrane oxygenation for severe acute respiratory failure. The Respiratory Extracorporeal Membrane Oxygenation Survival Prediction (RESP) score. Am J Respir Crit Care Med. 2014;189(11):1374–82.
Enger T, Philipp A, Videm V, Lubnow M, Wahba A, Fischer M, et al. Prediction of mortality in adult patients with severe acute lung failure receiving veno-venous extracorporeal membrane oxygenation: a prospective observational study. Crit Care. 2014;18(2):R67.
Pappalardo F, Pieri M, Greco T, Patroniti N, Pesenti A, Arcadipane A, et al. Predicting mortality risk in patients undergoing venovenous ECMO for ARDS due to influenza A (H1N1) pneumonia: the ECMOnet score. Intensive Care Med. 2013;39(2):275–81.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Schmidt, M. (2017). Indications and Physiopathology in Venovenous ECMO on Severe Acute Respiratory Distress Syndrome. In: Mossadegh, C., Combes, A. (eds) Nursing Care and ECMO. Springer, Cham. https://doi.org/10.1007/978-3-319-20101-6_3
Download citation
DOI: https://doi.org/10.1007/978-3-319-20101-6_3
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-20100-9
Online ISBN: 978-3-319-20101-6
eBook Packages: MedicineMedicine (R0)