Abstract
Congestive heart failure is associated with essential perturbations in the autonomic nervous system. Early in the development of heart failure, there may be defective parasympathetic cardiac control. This may occur before, or in parallel with, elevation in sympathetic tone. Here, we consider alterations that occur in the parasympathetic nervous system during the initiation and development of congestive heart failure. We also consider targets in the parasympathetic nervous system at various levels that may affect and improve clinical outcomes (survival, measures of progressive heart failure and debilitation, and cardiac remodeling, to name a few) by unique mechanistic effects that the parasympathetic nervous system exerts on heart rate, inflammation, remodeling, endothelial nitric oxide synthase activity, inhibition of the sympathetic nervous system, and other potential mechanisms. We consider approaches to vagus nerve stimulation, the designs and early outcomes of trials, and some of the drug interventions that have been attempted. In this rapidly emerging field, with little clinical data, we discuss issues regarding study designs and outcome measures of importance.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Parasympathetic activation
- Heart failure
- Vagus nerve
- Inflammation
- Parasympathetic nervous system
- Baroreceptor
1 Introduction
The relationship between the sympathetic nervous system (SNS) and the development and progression of congestive heart failure (CHF) in patients with left ventricular (LV) systolic dysfunction, or heart failure with reduced ejection fraction (HFrEF), is well established. Long-term, sympathetic overstimulation is detrimental to the myocardium [1]. Beta-adrenergic blockers improve many important outcomes, including CHF hospitalization and total mortality, but also ameliorate LV remodeling and improve functional class (Chaps. 3 and 5).
On the other hand, the relationship between parasympathetic innervation and CHF is less well defined. Although there may be a dynamic inverse relationship between the SNS and parasympathetic nervous system (PNS), it is difficult to measure parasympathetic (vagus) nerve efferent and afferent activity directly. As a result, it is difficult to quantify the effects of parasympathetic activation, and its withdrawal, with regard to definable CHF outcome parameters. Furthermore, there are no specific drugs that can activate the vagus nerve preferentially, reproducibly, reliably, and measurably without having other undesirable side effects.
Measures of vagus nerve activity often include peripheral markers, such as reduced circulating levels of norepinephrine, brain natriuretic peptide (BNP), and change in heart rate and heart rate variability. These markers, however, do not necessarily provide detailed, accurate, or specific information with regard to targeted and localized vagus nerve activation in the ventricles and atria of patients with CHF. This extent of sympathetic or parasympathetic activation may differ by disease type and pathological lesion distribution. Vagus nerve activation may differ by location of myocardial infarction. It is not completely known how parasympathetic activation, and its inhibition, affects initiation and progression of CHF.
Here, we address the potential effects of parasympathetic nerve activity in the development and reversal of HFrEF. We consider the PNS as a potential target for therapeutic intervention to improve outcomes in patients with LV systolic dysfunction.
2 Organization of the Parasympathetic Nervous System
Parasympathetic innervation is complex and affected by local influences. The effects are abrupt in onset and offset (although persistent vagal tonicity also exists) which makes them distinct from the more global (slow on and slow off) effects of sympathetic activation. While the effects can be rapid and/or phasic (e.g., varying with respiration), they can be more prolonged or tonic (related to localized central processing or ganglionic gating) [2] with continuous effects on ventricular muscle, for example. Selective neural and hormonal modulation occurs at various levels from the central nervous system through the ganglia and down to the level of intracellular signaling in specific target cardiac cells.
Central efferent extensions of the PNS begin at medial medullary sites (nucleus ambiguus, nucleus tractus solitarius, and dorsal motor nucleus) and are modulated by the hypothalamus. All activity then extends through the vagus nerves to the postganglionic neurons, located in the peripheral ganglia (in fat pads around the heart), activated via nicotinic receptors and then postsynaptically via muscarinic end-organ receptors. Through cardiac mechanoreceptors, baroreceptors, and aortic arch receptors, vagal afferent activation provides feedback from the cardiovascular system to the central nervous system. Baroreceptor activation sends signals to the nucleus tractus solitarius that can be activated tonically, which in turn can then activate the dorsal nucleus of the vagus nerve to affect efferent cardiac vagal responses.
In the normal state, at rest, parasympathetic activity predominates to regulate the heart rate via the sinus node; sympathetic effects are minimal. Physical activity can enhance vagal activation and suppress resting sympathetic tone. However, in CHF, sympathetic activation can supersede to various degrees any and all effects from the PNS.
Several types of parasympathetic nerve fibers coexist in the vagus nerve. A fibers are myelinated, the largest and the fastest conducting. Afferent fibers include slow-conducting unmyelinated C fibers and small-diameter A-delta fibers, whereas small, thin, nonmyelinated postganglionic C fibers and intermediate-diameter and intermediate-conducting preganglionic myelinated B fibers in efferent fascicles contribute to cardioinhibition mediated at the heart by muscarinic receptors. Efferent fascicles contain large myelinated A-beta fibers that belong to the laryngeal bundle and cardioinhibitory A-delta fibers that excite postganglionic neurons in the cardiac fat pads via nicotinic receptors. Nicotinic receptors affect ganglionic transmission and ultimately are responsible for parasympathetic activation via local neurons. Vagal efferent activation may differ (tonic or phasic) depending on where the fibers originate (nucleus ambiguus or dorsal motor nucleus of the vagus).
Predominantly, the PNS innervates the sinus node and AV nodes (Fig. 6.1). However, extensions of fibers are also present nonuniformly in atrial and ventricular muscle in a nonuniform epicardial/endocardial and regional distribution such that anatomical relationships are not in direct relationship to sympathetic innervation. Parasympathetic activation can inhibit sympathetic activation pre- and postsynaptically. Likewise, sympathetic activation can inhibit parasympathetic activation.
Innervation of the heart by the autonomic nervous system – from central to peripheral inputs. The sympathetic and parasympathetic (vagus) nerves are marked. Vagus afferents (sensory nerves) are also present (Reproduced with permission from Sinauer and associates and Springer Publishing Company. The figure is from Dale Purves et al., Principles of Cognitive Neuroscience, second edition, Figure 21.7 page 461)
Postganglionic parasympathetic cholinergic fibers affect the heart through cardiac muscarinic (generally, M2) receptors. These, and other, muscarinic receptors (M3 and M4) are present in cardiac muscle with density varying by location and cell type. Muscarinic receptors are concentrated in the sinus and AV nodes. While fewer fibers project into the ventricles, they are denser in the endocardium than in the epicardium. Most of the effects from the PNS are modulated through M2 receptors. Under normal, physiologic conditions, parasympathetic stimulation will inhibit tonic sympathetic activation with or without exercise. This is known as “accentuated antagonism.” Muscarinic receptor stimulation opposes sympathetic activation by the Gi-mediated signaling pathway that opposes adenylyl cyclase and works via guanosine triphosphate.
In those with heart disease, there can be defective parasympathetic cardiac control, and in those with CHF, the influence of the PNS diminishes even at rest [3–5]. This may actually occur before, or in parallel with, elevation in sympathetic “tone”; the two are related [6, 7]. The mechanisms responsible for this and the causal relationships between withdrawal of parasympathetic tone and CHF progression remain uncertain. Several other changes occur during the development of CHF. There is a decrease in nicotinic receptor number with reduced ganglionic transmission and a decrease in M2 receptor density and sensitivity, partly due to formation of M2 receptor antibodies. In an animal model, autoantibodies to M2 receptors have been associated with remodeling in CHF [8] although the clinical significance of this is questioned [9, 10]. Additionally, M3 and/or M4 receptors co-localized on cardiac structures may increase in lieu of decreasing M2 receptor density [11]. The effects from these changes are uncertain clinically. Also, sympathetic activation increases, in part, due to withdrawal of parasympathetic “tone.”
Various muscarinic receptors have different cellular expression, signaling pathways, functions, and molecular subtypes. Several forms appear to be present in the heart [12]. M1, M3, and M5 receptors couple Gq/11 to phospholipase C [13] to form two key second messengers, inositol trisphosphate and diacylglycerol [14]. M2 and M4 receptors couple pertussis toxin-sensitive G protein (Gi/Go) to inhibit adenylyl cyclase [15]. The receptors have differential effects on K+ and Ca2+ channels. The interactions of the receptors and other factors may explain why muscarinic agonists at high concentrations can induce a positive inotropic effect.
During CHF, parasympathetic control of the heart is attenuated, ganglionic transmission is reduced, muscarinic receptor density is increased [16], and acetylcholinesterase activity is decreased. Perhaps, as a consequence of the withdrawal of parasympathetic tone, muscarinic receptor density is increased [16] with a shift to novel muscarinic receptors at least in animal models. Thus, during CHF, once parasympathetic withdrawal is established, several maladaptations develop which include increased heart rate, excess release of pro-inflammatory cytokines [17], development of arrhythmias, and dysregulation of the nitric oxide (NO) signaling pathways [18].
Parasympathetic activation, on the other hand, inhibits sympathetic activity presynaptically [19] and reduces the heart rate. It is anti-inflammatory, limits cytokine release, and promotes normalization of NO expression and signaling. Additionally, parasympathetic activation inhibits the renin-angiotensin system, improves baroreflex sensitivity, and reduces the burden of life-threatening arrhythmias [18].
3 Potential Benefits of Vagus Nerve Stimulation: Animal Data
Parasympathetic activation in CHF has several potential therapeutic benefits (although the targets are not completely known). Firstly, the PNS can inhibit sympathetic activation, above and beyond that accrued by beta-adrenergic blockade. Secondly, the PNS can slow the sinus rate, thus lowering myocardial oxygen consumption. Thirdly, the PNS can improve cell-to-cell conduction and thus be antiarrhythmic. Fourthly, the PNS can normalize expression of several nitric oxide synthase (NOS) isoforms and provide potent anti-inflammatory and antioxidant effects. Inhibition of sympathetic overdrive in CHF can also elicit suppression of renin and vasopressin release and enhance intracellular calcium handling.
Much of the understanding on the beneficial influences of parasympathetic nerve activation in CHF has come from animal models. In these models, the effects of vagus activation are distinct from the effects of sympathetic activation or inhibition.
3.1 Vagus Nerve Stimulation
Vagus nerve stimulation (VNS) can be antiarrhythmic and protect against ventricular fibrillation independent of muscarinic receptor activation [20]. In a canine ischemic model of ventricular fibrillation, Vanoli et al. compared a control group to those who underwent vagus stimulation [21]. Ventricular fibrillation occurred in controls who underwent a second coronary artery occlusion but was suppressed following coronary artery occlusion in almost all who had vagal stimulation beforehand. These data were also supported by murine CHF and post-myocardial infarction models showing that right vagus stimulation slowed heart rate 20–30 bpm, improved hemodynamics, and reduced the risk of death (73 % relative risk reduction) [22]. In a post-myocardial infarction model, ventricular ectopy was markedly suppressed [23].
3.2 Inflammatory Cytokines
In CHF, there is an increase in inflammatory cytokines including TNF-alpha, IL-1 beta, IL-6, and IL-18 [17, 24]. Vagus nerve activation inhibits cytokine release and attenuates the inflammatory response. Cytokine release is associated with increased mortality and morbidity in CHF patients [25]. In the dog, protein expression and plasma levels of TNF-alpha increase with induced CHF versus a normal animal. Similarly, protein expression of IL-6 and plasma levels of IL-6 increase compared with a normal animal. However, with an effective stimulation of the vagus, levels of TNF-alpha and IL-6 decrease toward baseline values. Ruble et al. in a canine model of CHF have shown that TNF-alpha is elevated compared with normal animals and is decreased toward control values by VNS [26]. Borovikova et al. have described that acetylcholine can attenuate release of cytokines (TNF-alpha, IL-1beta, IL-6, and IL-18) and that direct electrical stimulation of the vagus nerve in vivo during lethal endotoxemia in rats inhibits TNF-alpha synthesis, attenuates serum TNF-alpha, and prevents septic shock [25].
VNS inhibits cytokine release and attenuates the inflammatory response, which includes the release of TNF-alpha, IL-1-beta, IL-6, and IL-18. Tracey has considered complex peripheral/central mechanisms between the cholinergic and anti-inflammatory pathways as it involves the nicotinic alpha-7 acetylcholine receptor. Work performed by his group has shown a relationship in CHF patients between cytokine release and mortality and morbidity [27]. In fact, signal transduction by the nicotinic alpha-7 acetylcholine receptor subunit regulates intracellular signals that control cytokine transcription and translation [28] and thus is an essential regulator of inflammation [29].
Sabbah et al. have shown a relationship between TNF-alpha, IL-1-6, and CHF in an animal model [30, 31]. Vagal nerve stimulation reduced the level of inflammation back to normal. Similarly, C-reactive protein increases in a pacing model of CHF and is reduced to baseline levels with VNS. VNS reduces norepinephrine and angiotensin II. VNS, in the canine CHF model, has been associated with reduction in elevations of C-reactive protein, IL-6, TNF-alpha, pro-ANP, and NT-proBNP.
3.3 NO Modulation
Parasympathetic nerve stimulation affects NO synthases (eNOS, nNOS, and iNOS) that are altered during CHF [32–40] and that affect ventricular function [32]. eNOS, which is reduced in CHF, exerts a number of beneficial actions that promote relaxation, modulate contractility, and inhibit myocardial hypertrophy and apoptosis. On the other hand, iNOS, which increases in CHF, exerts several adverse actions that promote apoptosis, fibrosis, hypertrophy of the myocardium, heart block, and sudden death [37, 41]. nNOS, also increased in CHF, modulates calcium cycling and reduces contractility, in addition to increasing sensitivity to beta-adrenergic stimulation [39]. In an animal model of CHF, altered NOS expression is normalized by VNS.
3.4 Gap Junctions
It is known that gap junctions, essential for cell-cell communication with regard to electrical depolarization and repolarization, can be impaired in CHF. In particular, there has been focus on connexin-43 which is reduced during CHF. Slowed myocardial conduction, altered repolarization, and increased risk of arrhythmias and sudden death follow [42, 43]. VNS improves LV connexin-43 expression in a dog model of CHF and thus may be antiarrhythmic. VNS can protect against ischemia-induced arrhythmias, specifically by preserving connexin-43 [44].
3.5 Anti-apoptotic
VNS may be anti-apoptotic as shown in an animal model in which active caspase-3, elevated in CHF, is downregulated with VNS [45, 46]. VNS has been shown to improve the activity and expression of the sarcoplasmic reticulum SERCA-2a pump which is downregulated in CHF, a maladaptation that leads to both systolic and diastolic LV dysfunction. VNS can also affect the degradation and synthesis of collagens I and III in the heart and thus indirectly impact LV compliance. Furthermore, with VNS TGF-beta-1 protein expression may decrease, thereby decreasing interstitial fibrosis.
3.6 Heart Rate
A fundamental action of VNS is a reduction in heart rate and the mechanism of this is fairly well understood. Heart rate is a major determinant of myocardial oxygen consumption and is known to be associated with increased mortality and morbidity in CHF [47]. The importance of heart rate reduction in CHF is supported by the results of the BEAUTIFUL (morBidity-mortality EvAlUaTion of the I f inhibitor ivabradine in patients with coronary disease and left ventricULar dysfunction) trial and SHIFT (Systolic Heart failure treatment with the I f inhibitor ivabradine Trial) [48–51], in which ivabradine, a specific and selective inhibitor of the I f current, was studied. In the BEAUTIFUL trial, ivabradine did not influence cardiovascular death or admission to hospital for heart failure. Ivabradine did reduce admission to hospital for fatal and nonfatal myocardial infarction and coronary revascularization (Chap. 1). In the SHIFT, higher resting heart rate was shown to be a risk factor for adverse outcome, and heart rate reduction with ivabradine was an important predictor of an improvement in the primary composite endpoint of mortality and CHF hospitalization (mainly driven by CHF hospitalization). One should be aware, however, that the benefits of VNS may be mediated by effects independent of heart rate reduction.
3.7 Effects Independent of Beta-Blockade
VNS may lead to an improvement in ejection fraction over that which occurs with beta-adrenergic blockade alone. Sabbah et al. [30, 31] in a CHF model, showed that VNS, on top of beta-adrenergic blockade, caused a greater improvement in ventricular function than did beta-blockade alone. The best way to activate the PNS is not completely certain. While most data would support right vagus nerve stimulation, other approaches have been utilized including carotid sinus baroreceptor stimulation in a dog model [52], spinal cord stimulation in a canine model [53], and spinal cord stimulation in a porcine model [54]. The effects of left vagus nerve stimulation in CHF are less well studied.
VNS appears to improve ventricular remodeling in advanced CHF and thereby improve outcomes, though there are little human data as yet to substantiate this. Unanswered questions include which vagus (right, left, or both) is best to stimulate and at which intensity, frequency, and duration.
3.8 Carotid Baroreflex Activation
Alternatives to VNS as targets for PNS activation have been studied. Sabbah et al. examined the role of carotid sinus baroreflex activation in 14 dogs (eight experimental and six controls) with an LV ejection fraction of 25 % [52]. Over the course of 3 months, those receiving baroreflex activation were found to have an increase in ejection fraction (4.0 ± 2.4 %), while the controls had further decline (−2.8 ± 1 %) (p < 0.05). Biomarkers and LV remodeling also improved in the experimental group.
3.9 Other Effects of Vagus Activation
VNS (vs. control) can reduce infarct size, improve ventricular function, decrease ventricular fibrillation, and attenuate cardiac mitochondrial reactive oxygen species production, depolarization, and swelling. This effect appears modulated through muscarinic receptor modulation during VNS. The protective effects of VNS could be due to its protection of mitochondrial function during ischemia-reperfusion [55]. In another experiment in swine, VNS applied 30 min after left anterior descending occlusion, but not at reperfusion, markedly reduced ventricular fibrillation and infarct size (~59 %); improved cardiac function; attenuated cardiac mitochondrial reactive oxygen species production, depolarization, swelling, and cytochrome c release; and increased the amount of phosphorylated connexin-43 and IL-4 vs. controls [56].
4 Parasympathetic Activation in Clinical Trials
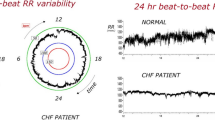
The ATRAMI (Autonomic Tone and Reflexes After Myocardial Infarction) study included patients post-myocardial infarction [57] and measured autonomic tone and reflexes to predict outcomes with regard to abnormalities in heart rate variability as measured by “standard deviation of normal to normal R-R intervals” (SDNN) >70 and baroreflex sensitivity >3 (a measure suggesting relatively high vagus nerve tone). While these particular parameters measure different things and may be associative rather than causal, the results suggested that vagus activation may improve mortality post-myocardial infarction. Low heart rate variability was associated with poor survival and low baroreflex sensitivity was associated with better survival. Other data support these findings [58]. Similarly, the CIBIS II (Cardiac Insufficiency Bisoprolol Study II) trial showed that diminished vagus nerve activity and increased heart rate predicted higher mortality in CHF [59]. The mechanisms responsible for this association remain unclear, and while increased heart rate alone may be a causative factor for higher mortality, the association is robust, but the causal nature is suspect.
Various approaches have been used to modulate the autonomic nervous system utilizing novel devices, particularly as relates to parasympathetic activation including spinal cord stimulation, selective right (or even left) vagus stimulation, baroreflex stimulation, and other innovative and as yet unapproved devices. This has led to the development of several ongoing clinical trials.
The CardioFit device, made by BioControl, attempts to selectively stimulate efferent cardioinhibitory B (A-delta) vagus fibers. An initial pilot study with a primary endpoint of safety in 32 CHF patients with LV ejection fraction ≤0.35, otherwise medically stable, was performed. VNS led to improvement in 3- and 6-month outcomes with regard to the New York Heart Association (NYHA) Functional Classification, 6-min walk, and quality of life, as well as structural/functional improvements in LV ejection fraction and end-diastolic and end-systolic volumes [60].
The INcrease Of VAgal TonE in HF (INOVATE HF) trial is a multicenter pivotal trial of CHF patients evaluating survival based on right vagus stimulation versus no vagus stimulation (no implant) and based on preliminary data [61]. The INOVATE trial is an open-label prospective, randomized comparative trial that involves 650 patients (80 investigative sites) followed for 5.5 years or until 400 events have occurred. The study is enrolling NYHA Functional Class III patients with LV ejection fractions ≤0.40 with an efficacy endpoint of total mortality or unplanned CHF hospitalization. In this trial, there is no specific measure of long-term VNS efficacy. The heart rate is not considered an important endpoint in this trial.
The Neural Cardiac TherApy foR Heart Failure (NECTAR-HF) study (Boston Scientific) of a right-sided vagus nerve implant system evaluated the primary endpoint of change in LV end-systolic dimension and mortality at 3 months, as well as a safety and several secondary endpoints, with the device “on” versus the device “off” in a 2:1 randomization of 96 patients with NYHA Functional Class III heart failure, an LV ejection fraction ≤35 %, and a narrow QRS complex [62]. The stimulation amplitude and frequency (0.1–4.0 mA and 20 Hz frequency, 300 μs pulse width, duty cycle 10 s “on” and 50 s “off”) differ from CardioFit and the device uses a different electrode design. The device itself is approved for spinal cord stimulation for pain and is being used off-label for this trial. While the patients receiving vagus stimulation were noted to have improvement in quality-of-life scores and most had at least one point improvement in NYHA Functional Class, the study failed to show improvement in primary or secondary measures of cardiac remodeling (evaluated blindly) or functional capacity (European Society of Cardiology 2014; Barcelona, Spain).
The Autonomic Neural Regulation Therapy to Enhance Myocardial Function in Heart Failure (ANTHEM-HF) trial (Cyberonics) evaluated stimulation of the left (n = 31) or right (n = 29) vagus for 6 months in 60 patients (mean age = 51) with systolic heart failure on a stable medical regimen [63]. This was an open-label feasibility trial of patients with NYHA Functional Class II–III CHF and an LV ejection fraction ≤40 %. The primary efficacy endpoint was change in left ventricular ejection fraction (LVEF) and left ventricular end-systolic volume (LVESV) at 6 months; the secondary endpoints involved evaluating the effect on (1) left ventricular end-systolic diameter (LVESD), (2) NYHA Functional Class, (3) 6-min walk distance, (4) quality of life (Minnesota Living with Heart Failure Questionnaire), (5) heart rate variability, and (6) biomarkers (brain natriuretic peptide (NT-proBNP and hs-CRP)). Stimulation was at 10 Hz, the pulse amplitude was 2.0 ± 0.6 mA, the duty cycle was 17.5 % (14 s on, 66 s off), and the pulse width was 130 ms. The intensity was titrated over a 10-week period (European Society of Cardiology 2014; Barcelona, Spain). The LV ejection fraction improved by a mean of 4.5 % (similar with left or right vagus stimulation). There was a mean improvement in the 6-min walk test of 56 m (significantly less with left vs. right vagus stimulation) and a mean improvement in Minnesota Living with Heart Failure Questionnaire scores of 18 points. There was no significant improvement in LV end-systolic volume.
A canine model of post-infarct CHF has shown to benefit from spinal cord stimulation [53]. Stimulation caused reduction in norepinephrine, serotonin, and BNP, as well as evidence for reverse remodeling with an improved LV ejection fraction when combined CHF medications. In a porcine model [54], spinal cord stimulation reduced myocardial oxygen demand substantially despite increase in LV dP/dT. The Determining the Feasibility of Spinal Cord Neuromodulation for the Treatment of Chronic Heart Failure (DEFEAT-HF) trial (Medtronic) is a feasibility trial evaluating spinal cord stimulation vs. control in 70 ICD patients with NYHA Functional Class III CHF and an LV ejection fraction <35 %, a QRS duration <120 ms, and an endpoint of LV end-diastolic volume index [64]. St Jude is working on a spinal cord stimulator study (SCS Heart, 20 patients) [65]. To be enrolled, patients must have an LV ejection fraction of 20–35 %, NYHA Functional Class III or ambulatory Class IV CHF, a St. Jude implantable cardioverter defibrillator or CRT-D device, and an LV end-diastolic diameter of 55–80 mm.
Carotid baroreflex stimulation is another approach being evaluated in the Barostim HOPE4HF (Hope for Heart Failure) trial to assesses safety, efficacy, and feasibility of baroreflex activation as a therapy in CHF with preserved LV function (ejection fraction ≥40 %) and hypertension in 60 patients with elevated brain natriuretic peptide or NT-pro-brain natriuretic peptide and creatinine ≤2.5 mg/dL [66].
There are many important issues of concern with all of these clinical trials: (1) No definitive data has yet proved that vagal activation is better than no vagal activation in patients with CHF. (2) The optimal approach to vagal activation for increasing parasympathetic tone is not well defined (intensity, frequency, duration, etc.). (3) The best endpoint to measure adequate and long-lasting VNS is not certain. (4) The proof that parasympathetic activation is actually occurring remains difficult to comprehend. (5) The proper endpoint to measure, i.e., heart rate reduction or some other parameter, such as, an inflammatory marker, is not clear. (6) It is not clear which fibers of the vagus are most important to stimulate, whether they are the A-delta, the B fibers, or the unmyelinated C fibers. Further, it is unclear if afferent activation may be of benefit as well.
5 Drugs That Can Modulate the PNS in CHF
To date, no drug acting solely to activate the PNS has been shown to benefit patients with CHF. Drugs that target the PNS in CHF are given in Table 6.1 and are briefly described below.
5.1 Digoxin
Perhaps the most widely used PNS-activating drug is digoxin. While digoxin may have a potential role as a vagal activator, the mechanism of action and the intensity of the PNS effects are uncertain. Any direct effect on the vagus by digoxin is at best speculative and not well studied. It appears, however, that vagal afferents are sensitized by drugs similar to digoxin and these drugs also inhibit renal sympathetic nerve activity [67–69]. Also, digitalis can beneficially affect baroreflex sympathetic control in normal humans [70]. However, despite an increase in plasma norepinephrine with chronic digoxin therapy, heart rate variability changes and heart rate reduction suggest a substantial increase in parasympathetic activity [71].
Digoxin inhibits sodium/potassium ATPase, thereby increasing intracellular calcium that mediates a positive inotropic effect which could nullify any direct benefit of digoxin due to vagal activation. The Digitalis Investigation Group (DIG) trial showed no benefit of digoxin regarding mortality outcomes in CHF; digoxin only had an effect on reducing the rate of hospitalization overall and CHF hospitalization [72]. Although it is likely that digoxin does have an effect on vagus activity, the clinical evidence suggests that this effect is either not adequate to affect mortality or is counterbalanced by other adverse actions.
5.2 Angiotensin-Converting Enzyme Inhibitors
Angiotensin-converting enzyme (ACE) inhibitors, which improve outcomes in CHF (see Chap. 36 for details), are associated with improvement in parasympathetic activation [73–76]. Improvements in measures of heart rate variability may be secondary to improvement in CHF in general or to inhibition of sympathetic activation. However, any causal relationship between ACE inhibitors and vagus activation remains speculative at best. Like ACE inhibitors, angiotensin receptor blockers appear to modulate and improve PNS activity [77–79]. Similar to ACE inhibitors, however, it is likely that SNS withdrawal and PNS activation are not a direct effect but, rather, are reflex mediated due to an improvement in hemodynamics.
5.3 Nitroglycerin
Nitroglycerin may have direct NO-mediated effects on vagal activity [80]. This, however, may be offset by the reflex sympathetic activation that occurs with peripheral vasodilatation. Compared with placebo, transdermal nitroglycerin had no effect on heart rate variability parameters or baroreflex sensitivity.
5.4 Omega-3 Fatty Acids
Omega-3 fatty acids may improve parasympathetic activation, enhance baroreceptor control [81], and improve heart rate variability in the elderly [82], but substantiation for this and any benefit in CHF patients remain highly speculative. Omega-3 fatty acids may slow sinus rate slightly, though the mechanism is uncertain. See Chap. 27 for details.
5.5 Nicotinic Agonists
Repeated exposure of ganglionic neurons to nicotinic agonists to prevent loss of parasympathetic control of CHF has been tested [83]. Despite decreased ganglionic function leading to reduced parasympathetic control of CHF, repeated exposure to nicotinic agonists during CHF development resulted in preserved or even supernormal effects of parasympathetic stimulation.
5.6 Acetylcholinesterase Inhibitors
Pyridostigmine bromide is an acetylcholinesterase inhibitor that does not cross the blood-brain barrier and works in the peripheral synaptic cleft. It prolongs the effect of acetylcholine. In healthy animals and humans, it appears to stimulate the parasympathetic nerves. Pyridostigmine has been evaluated in a rat model of CHF [84]. In conscious rats, pyridostigmine bromide reduces the basal heart rate, increases vagal tone, and reduces sympathetic control of the heart. In rats, pyridostigmine appears to decrease the hypertrophy seen in CHF, reduces the expression of collagen in non-infarcted myocardium, and prevents the development of systolic CHF after myocardial infarction. In patients, cholinergic stimulation by pyridostigmine appears to reduce ventricular arrhythmias and improve autonomic and hemodynamic function during dynamic exercise. Pyridostigmine can reduce the exercise heart rate by up to 60 % without changing the resting heart rate.
In a prospective, randomized, double-blind, placebo-controlled, crossover trial of 20 patients with stable CHF (mean age 55 years, mean ejection fraction 24 %), pyridostigmine (30 mg) improved heart rate recovery after maximal exercise at 1 min but not at 3 min. Ingested pyridostigmine 30 mg or a matching placebo on separate days did not affect peak heart rate, oxygen uptake, or respiratory exchange ratio or change plasma norepinephrine or brain natriuretic peptide concentrations [85].
In another study, CHF patients participated in a double-blind, crossover protocol (placebo or pyridostigmine 30 mg orally TID for 2 days). In those with frequent ventricular ectopy, pyridostigmine reduced ventricular ectopy by 65 %. For a heart rate variability group, pyridostigmine slowed heart rate and improved heart rate variability. The authors suggested that long-term trials of pyridostigmine in CHF patients should be conducted [86]. In a study of patients with chronic CHF submitted to three maximal cardiopulmonary exercise tests on different days, oral pyridostigmine (45 mg TID for 24 h) reduced cholinesterase activity by 30 %, inhibited chronotropic response throughout exercise up to 60 % of maximal effort, and improved heart rate reserve (and heart rate recovery in the first minute after exercise), whereas peak heart rate was similar to placebo [87].
Donepezil, a cholinesterase inhibitor used to treat Alzheimer’s dementia, crosses the blood-brain barrier. Donepezil has been shown to prevent LV dysfunction and neurohumoral activation and CHF in rats.
Donepezil was tested in a rat model of CHF due to healed myocardial infarction. Compared to untreated rats, those given donepezil had a smaller biventricular weight (and maximal rate of rise in LV pressure and end-diastolic LV pressure was improved). Neurohumoral factors (norepinephrine and BNP) were improved with donepezil as was heart rate variability [88].
5.7 Muscarinic Agonists and Antagonists
Carbachol, a muscarinic agonist, can prevent beta-adrenergic receptor-stimulated apoptosis in cardiac myocytes, suggesting that muscarinic activation could affect the rate of disease progression in CHF [89].
Pirenzepine, a selective muscarinic receptor antagonist, works as an anticholinergic drug but at lower doses can have a cholinergic affect that augments vagal activity in CHF.
Scopolamine likewise blocks muscarinic receptors but, at lower doses, can enhance parasympathetic activity. Scopolamine is relatively nonselective, but it can affect heart rate variability [90]. In 21 patients with moderate to severe CHF, scopolamine increased time and frequency domain parameters of heart rate variability. However, scopolamine has not been tested clinically with regard to outcomes in patients with CHF [90].
Twelve patients with NYHA Functional Class II–IV CHF and coronary artery disease (mean LV ejection fraction 26.7 %) were randomly assigned to a placebo or transdermal scopolamine patch. There was a small, but significant, increase in all time domain HRV variables with scopolamine. There was improvement in HR variability parameters [91].
In a double-blind, randomized, placebo-controlled, crossover study, 16 patients with chronic, stable CHF and ischemic cardiomyopathy (mean LV ejection fraction 28 %; NYHA Functional Class II–III) and eight age-matched healthy controls received scopolamine (500 μg/72 h) or a placebo patch. Scopolamine reduced average heart rate and increased time domain measures of heart rate variability. While exercise performance did not improve, heart rate at submaximal exercise was reduced by scopolamine [92]. In another report, low doses of transdermal scopolamine enhanced tonic and reflex cardiac vagal activity in patients in the acute phase of MI as measured by an increase in baroreflex sensitivity, heart rate variability, and respiratory sinus arrhythmia [93].
6 Standard Drugs for Heart Failure
Although various drugs that are utilized to attempt to improve outcomes in CHF patients (diuretics, statins [94, 95], aldosterone blockers [96–98], vasodilators, beta-blockers, ivabradine, and antiarrhythmic drugs) may affect sympathetic vagal balance, it is certainly possible that any positive association between these drugs and outcomes may be by other mechanisms. Even exercise training may affect vagal tone and improve outcomes in patients with CHF [99]. Recent data from the SHIFT [50] and BEAUTIFUL [48, 49] trials that evaluate the use of ivabradine in CHF patients indicate that heart rate slowing independent of autonomic interactions may also have some benefit with regard to select outcomes.
7 Can Anticholinergic Drugs Adversely Affect Heart Failure Patients?
Presently, little data substantiate any harm caused by an anticholinergic drug in patients with CHF. In part, this could be due to the fact that parasympathetic activation is already limited in these patients. However, this has not been evaluated carefully. Disopyramide, an antiarrhythmic drug with substantial anticholinergic properties, has a strong negative inotropic effect, but the mechanism of this effect has not been well tested. Nevertheless, the relationship between any anticholinergic response, i.e., increase in heart rate, has not necessarily been linked to worsening in CHF. To date, although not well tested, there are no clear-cut observational data indicating that antimuscarinic drug therapy for overactive bladder can precipitate CHF.
8 Concluding Remarks
Deranged autonomic nervous system activity plays an instrumental role in the pathophysiology of CHF. Increased activity of the sympathetic nervous system and decreased activity of the PNS contribute to disease progression. Early in the development of CHF, impaired parasympathetic activity may have a deleterious role in CHF progression.
Emerging evidence suggests that interventions that augment PNS activity can exert several beneficial effects directly and independently but in part by opposing the sympathetic nervous system. Direct measurement of PNS activity is difficult, and thus surrogate markers are often used to assess its status. Much interest is currently focused upon determining means to restore PNS activity, with devices or medications, to improve patient functionality and to reduce the morbidity and mortality of patients living with CHF.
References
Mann DL, Kent RL, Parsons B, Cooper GT. Adrenergic effects on the biology of the adult mammalian cardiocyte. Circulation. 1992;85:790–804.
McAllen RM, Salo LM, Paton JF, Pickering AE. Processing of central and reflex vagal drives by rat cardiac ganglion neurones: an intracellular analysis. J Physiol. 2011;589:5801–18.
Bibevski S, Dunlap ME. Evidence for impaired vagus nerve activity in heart failure. Heart Fail Rev. 2011;16:129–35.
Eckberg DL, Drabinsky M, Braunwald E. Defective cardiac parasympathetic control in patients with heart disease. N Engl J Med. 1971;285:877–83.
Porter TR, Eckberg DL, Fritsch JM, Rea RF, Beightol LA, Schmedtje Jr JF, Mohanty PK. Autonomic pathophysiology in heart failure patients. Sympathetic-cholinergic interrelations. J Clin Invest. 1990;85:1362–71.
Azevedo ER, Parker JD. Parasympathetic control of cardiac sympathetic activity: normal ventricular function versus congestive heart failure. Circulation. 1999;100:274–9.
Levy MN. Sympathetic-parasympathetic interactions in the heart. Circ Res. 1971;29:437–45.
Gimenez LE, Hernandez CC, Mattos EC, Brandao IT, Olivieri B, Campelo RP, Araujo-Jorge T, Silva CL, Campos de Carvalho AC, Kurtenbach E. DNA immunizations with m2 muscarinic and beta1 adrenergic receptor coding plasmids impair cardiac function in mice. J Mol Cell Cardiol. 2005;38:703–14.
Pei J, Li N, Chen J, Li X, Zhang Y, Wang Z, Zhang P, Cao K, Pu J. The predictive values of beta1-adrenergic and m2 muscarinic receptor autoantibodies for sudden cardiac death in patients with chronic heart failure. Eur J Heart Fail. 2012;14:887–94.
Talvani A, Rocha MO, Ribeiro AL, Borda E, Sterin-Borda L, Teixeira MM. Levels of anti-m2 and anti-beta1 autoantibodies do not correlate with the degree of heart dysfunction in Chagas’ heart disease. Microbes Infect/Inst Pasteur. 2006;8:2459–64.
Shi H, Wang H, Li D, Nattel S, Wang Z. Differential alterations of receptor densities of three muscarinic acetylcholine receptor subtypes and current densities of the corresponding k+ channels in canine atria with atrial fibrillation induced by experimental congestive heart failure. Cell Physiol Biochem Int J Exp Cell Physiol Biochem Pharmacol. 2004;14:31–40.
Wang Z, Shi H, Wang H. Functional m3 muscarinic acetylcholine receptors in mammalian hearts. Br J Pharmacol. 2004;142:395–408.
Takano K, Yasufuku-Takano J, Kozasa T, Singer WD, Nakajima S, Nakajima Y. Gq/11 and plc-beta 1 mediate the substance p-induced inhibition of an inward rectifier k+ channel in brain neurons. J Neurophysiol. 1996;76:2131–6.
Alea MP, Borroto-Escuela DO, Romero-Fernandez W, Fuxe K, Garriga P. Differential expression of muscarinic acetylcholine receptor subtypes in Jurkat cells and their signaling. J Neuroimmunol. 2011;237:13–22.
Krudewig R, Langer B, Vogler O, Markschies N, Erl M, Jakobs KH, van Koppen CJ. Distinct internalization of m2 muscarinic acetylcholine receptors confers selective and long-lasting desensitization of signaling to phospholipase c. J Neurochem. 2000;74:1721–30.
Dunlap ME, Bibevski S, Rosenberry TL, Ernsberger P. Mechanisms of altered vagal control in heart failure: influence of muscarinic receptors and acetylcholinesterase activity. Am J Physiol Heart Circ Physiol. 2003;285:H1632–40.
Li W, Olshansky B. Inflammatory cytokines and nitric oxide in heart failure and potential modulation by vagus nerve stimulation. Heart Fail Rev. 2011;16:137–45.
Olshansky B, Sabbah HN, Hauptman PJ, Colucci WS. Parasympathetic nervous system and heart failure: pathophysiology and potential implications for therapy. Circulation. 2008;118:863–71.
Vanhoutte PM, Levy MN. Prejunctional cholinergic modulation of adrenergic neurotransmission in the cardiovascular system. Am J Physiol. 1980;238:H275–81.
Brack KE, Patel VH, Coote JH, Ng GA. Nitric oxide mediates the vagal protective effect on ventricular fibrillation via effects on action potential duration restitution in the rabbit heart. J Physiol. 2007;583:695–704.
Vanoli E, De Ferrari GM, Stramba-Badiale M, Hull Jr SS, Foreman RD, Schwartz PJ. Vagal stimulation and prevention of sudden death in conscious dogs with a healed myocardial infarction. Circ Res. 1991;68:1471–81.
Li M, Zheng C, Sato T, Kawada T, Sugimachi M, Sunagawa K. Vagal nerve stimulation markedly improves long-term survival after chronic heart failure in rats. Circulation. 2004;109:120–4.
Zheng C, Li M, Inagaki M, Kawada T, Sunagawa K, Sugimachi M. Vagal stimulation markedly suppresses arrhythmias in conscious rats with chronic heart failure after myocardial infarction. Conf Proc IEEE Eng Med Biol Soc. 2005;7:7072–5.
Aukrust P, Gullestad L, Ueland T, Damas JK, Yndestad A. Inflammatory and anti-inflammatory cytokines in chronic heart failure: potential therapeutic implications. Ann Med. 2005;37:74–85.
Borovikova LV, Ivanova S, Zhang M, Yang H, Botchkina GI, Watkins LR, Wang H, Abumrad N, Eaton JW, Tracey KJ. Vagus nerve stimulation attenuates the systemic inflammatory response to endotoxin. Nature. 2000;405:458–62.
Ruble SB, Hamann JJ, Gupta RC, Mishra S, Sabbah HN. Chronic vagus nerve stimulation impacts biomarkers of heart failure in canines. J Am Coll Cardiol. 2010;55:A16.
Tracey KJ. The inflammatory reflex. Nature. 2002;420:853–9.
Huston JM, Tracey KJ. The pulse of inflammation: heart rate variability, the cholinergic anti-inflammatory pathway and implications for therapy. J Intern Med. 2011;269:45–53.
Wang H, Yu M, Ochani M, Amella CA, Tanovic M, Susarla S, Li JH, Wang H, Yang H, Ulloa L, Al-Abed Y, Czura CJ, Tracey KJ. Nicotinic acetylcholine receptor alpha7 subunit is an essential regulator of inflammation. Nature. 2003;421:384–8.
Sabbah HN, Ilsar I, Zaretsky A, Rastogi S, Wang M, Gupta RC. Vagus nerve stimulation in experimental heart failure. Heart Fail Rev. 2011;16:171–8.
Sabbah HN. Electrical vagus nerve stimulation for the treatment of chronic heart failure. Cleve Clin J Med. 2011;78 Suppl 1:S24–9.
Kelly RA, Balligand JL, Smith TW. Nitric oxide and cardiac function. Circ Res. 1996;79:363–80.
Patten RD, Denofrio D, El-Zaru M, Kakkar R, Saunders J, Celestin F, Warner K, Rastegar H, Khabbaz KR, Udelson JE, Konstam MA, Karas RH. Ventricular assist device therapy normalizes inducible nitric oxide synthase expression and reduces cardiomyocyte apoptosis in the failing human heart. J Am Coll Cardiol. 2005;45:1419–24.
Xu KY, Huso DL, Dawson TM, Bredt DS, Becker LC. Nitric oxide synthase in cardiac sarcoplasmic reticulum. Proc Natl Acad Sci U S A. 1999;96:657–62.
Damy T, Ratajczak P, Shah AM, Camors E, Marty I, Hasenfuss G, Marotte F, Samuel JL, Heymes C. Increased neuronal nitric oxide synthase-derived no production in the failing human heart. Lancet. 2004;363:1365–7.
Paulus WJ, Shah AM. No and cardiac diastolic function. Cardiovasc Res. 1999;43:595–606.
Mungrue IN, Gros R, You X, Pirani A, Azad A, Csont T, Schulz R, Butany J, Stewart DJ, Husain M. Cardiomyocyte overexpression of iNOS in mice results in peroxynitrite generation, heart block, and sudden death. J Clin Invest. 2002;109:735–43.
Barouch LA, Harrison RW, Skaf MW, Rosas GO, Cappola TP, Kobeissi ZA, Hobai IA, Lemmon CA, Burnett AL, O’Rourke B, Rodriguez ER, Huang PL, Lima JA, Berkowitz DE, Hare JM. Nitric oxide regulates the heart by spatial confinement of nitric oxide synthase isoforms. Nature. 2002;416:337–9.
Bendall JK, Damy T, Ratajczak P, Loyer X, Monceau V, Marty I, Milliez P, Robidel E, Marotte F, Samuel JL, Heymes C. Role of myocardial neuronal nitric oxide synthase-derived nitric oxide in beta-adrenergic hyporesponsiveness after myocardial infarction-induced heart failure in rat. Circulation. 2004;110:2368–75.
Keaney Jr JF, Hare JM, Balligand JL, Loscalzo J, Smith TW, Colucci WS. Inhibition of nitric oxide synthase augments myocardial contractile responses to beta-adrenergic stimulation. Am J Physiol. 1996;271:H2646–52.
Nisoli E, Carruba MO. Nitric oxide and mitochondrial biogenesis. J Cell Sci. 2006;119:2855–62.
Jongsma HJ, Wilders R. Gap junctions in cardiovascular disease. Circ Res. 2000;86:1193–7.
Severs NJ, Bruce AF, Dupont E, Rothery S. Remodelling of gap junctions and connexin expression in diseased myocardium. Cardiovasc Res. 2008;80:9–19.
Ando M, Katare RG, Kakinuma Y, Zhang D, Yamasaki F, Muramoto K, Sato T. Efferent vagal nerve stimulation protects heart against ischemia-induced arrhythmias by preserving connexin43 protein. Circulation. 2005;112:164–70.
Todor A, Sharov VG, Tanhehco EJ, Silverman N, Bernabei A, Sabbah HN. Hypoxia-induced cleavage of caspase-3 and dff45/icad in human failed cardiomyocytes. Am J Physiol Heart Circ Physiol. 2002;283:H990–5.
Sabbah HN, Sharov VG, Gupta RC, Todor A, Singh V, Goldstein S. Chronic therapy with metoprolol attenuates cardiomyocyte apoptosis in dogs with heart failure. J Am Coll Cardiol. 2000;36:1698–705.
Ahmadi-Kashani M, Kessler DJ, Day J, Bunch TJ, Stolen KQ, Brown S, Sbaity S, Olshansky B, Investigators IRS. Heart rate predicts outcomes in an implantable cardioverter-defibrillator population. Circulation. 2009;120:2040–5.
Fox K, Ford I, Steg PG, Tendera M, Robertson M, Ferrari R, Investigators B. Heart rate as a prognostic risk factor in patients with coronary artery disease and left-ventricular systolic dysfunction (beautiful): a subgroup analysis of a randomised controlled trial. Lancet. 2008;372:817–21.
Fox K, Ford I, Steg PG, Tendera M, Ferrari R, Investigators B. Ivabradine for patients with stable coronary artery disease and left-ventricular systolic dysfunction (beautiful): a randomised, double-blind, placebo-controlled trial. Lancet. 2008;372:807–16.
Bohm M, Swedberg K, Komajda M, Borer JS, Ford I, Dubost-Brama A, Lerebours G, Tavazzi L, Investigators S. Heart rate as a risk factor in chronic heart failure (shift): the association between heart rate and outcomes in a randomised placebo-controlled trial. Lancet. 2010;376:886–94.
Swedberg K, Komajda M, Bohm M, Borer JS, Ford I, Dubost-Brama A, Lerebours G, Tavazzi L, Investigators S. Ivabradine and outcomes in chronic heart failure (shift): a randomised placebo-controlled study. Lancet. 2010;376:875–85.
Sabbah HN, Gupta RC, Imai M, Irwin ED, Rastogi S, Rossing MA, Kieval RS. Chronic electrical stimulation of the carotid sinus baroreflex improves left ventricular function and promotes reversal of ventricular remodeling in dogs with advanced heart failure. Circ Heart Fail. 2011;4:65–70.
Lopshire JC, Zhou X, Dusa C, Ueyama T, Rosenberger J, Courtney N, Ujhelyi M, Mullen T, Das M, Zipes DP. Spinal cord stimulation improves ventricular function and reduces ventricular arrhythmias in a canine postinfarction heart failure model. Circulation. 2009;120:286–94.
Liu Y, Yue WS, Liao SY, Zhang Y, Au KW, Shuto C, Hata C, Park E, Chen P, Siu CW, Tse HF. Thoracic spinal cord stimulation improves cardiac contractile function and myocardial oxygen consumption in a porcine model of ischemic heart failure. J Cardiovasc Electrophysiol. 2012;23:534–40.
Shinlapawittayatorn K, Chinda K, Palee S, Surinkaew S, Thunsiri K, Weerateerangkul P, Chattipakorn S, KenKnight BH, Chattipakorn N. Low-amplitude, left vagus nerve stimulation significantly attenuates ventricular dysfunction and infarct size through prevention of mitochondrial dysfunction during acute ischemia-reperfusion injury. Heart Rhythm. 2013;10:1700–7.
Shinlapawittayatorn K, Chinda K, Palee S, Surinkaew S, Kumfu S, Kumphune S, Chattipakorn S, KenKnight BH, Chattipakorn N. Vagus nerve stimulation initiated late during ischemia, but not reperfusion, exerts cardioprotection via amelioration of cardiac mitochondrial dysfunction. Heart Rhythm. 2014;11:2278–87.
La Rovere MT, Bigger Jr JT, Marcus FI, Mortara A, Schwartz PJ. Baroreflex sensitivity and heart-rate variability in prediction of total cardiac mortality after myocardial infarction. ATRAMI (autonomic tone and reflexes after myocardial infarction) investigators. Lancet. 1998;351:478–84.
De Ferrari GM, Sanzo A, Bertoletti A, Specchia G, Vanoli E, Schwartz PJ. Baroreflex sensitivity predicts long-term cardiovascular mortality after myocardial infarction even in patients with preserved left ventricular function. J Am Coll Cardiol. 2007;50:2285–90.
Lechat P, Hulot JS, Escolano S, Mallet A, Leizorovicz A, Werhlen-Grandjean M, Pochmalicki G, Dargie H. Heart rate and cardiac rhythm relationships with bisoprolol benefit in chronic heart failure in CIBIS II trial. Circulation. 2001;103:1428–33.
De Ferrari GM, Crijns HJ, Borggrefe M, Milasinovic G, Smid J, Zabel M, Gavazzi A, Sanzo A, Dennert R, Kuschyk J, Raspopovic S, Klein H, Swedberg K, Schwartz PJ. Chronic vagus nerve stimulation: a new and promising therapeutic approach for chronic heart failure. Eur Heart J. 2011;32:847–55.
INOVATE-HF: Increase of vagal tone in CHF study. http://www.clinicaltrials.gov/ct2/show/NCT01303718?term=Nct01303718&rank=1, Accessed 2 Sept 2014.
NECTAR-HF: Neural cardiac therapy for heart failure study. http://www.clinicaltrials.gov/ct2/show/NCT01385176?term=Nct01385176&rank=1, Accessed 2 Sept 2014.
ANTHEM-HF: Autonomic neural regulation therapy to enhance myocardial function in heart failure study. http://www.clinicaltrials.gov/ct2/show/NCT01823887?term=Nct01823887&rank=1, Accessed 2 Sept 2014.
Defeat-HF: determining the feasibility of spinal cord neuromodulation for the treatment of chronic heart failure study. http://www.clinicaltrials.gov/ct2/show/NCT01112579?term=Nct01112579&rank=1, Accessed 2 Sept 2014.
SCS HEART: spinal cord stimulation for heart failure study. http://www.clinicaltrials.gov/ct2/show/NCT01362725?term=Nct01362725&rank=1, Accessed 2 Sept 2014.
Rheos HOPE4HF trial: device: Rheos Baroreflex activation system study. http://www.clinicaltrials.gov/ct2/show/NCT00957073?term=Nct00957073&rank=1, Accessed 2 Sept 2014.
Thames MD. Acetylstrophanthidin-induced reflex inhibition of canine renal sympathetic nerve activity mediated by cardiac receptors with vagal afferents. Circ Res. 1979;44:8–15.
Thames MD, Waickman LA, Abboud FM. Sensitization of cardiac receptors (vagal afferents) by intracoronary acetylstrophanthidin. Am J Physiol. 1980;239:H628–35.
Sleight P, Lall A, Muers M. Reflex cardiovascular effects of epicardial stimulation by acetylstrophanthidin in dogs. Circ Res. 1969;25:705–11.
Schobel HP, Oren RM, Roach PJ, Mark AL, Ferguson DW. Contrasting effects of digitalis and dobutamine on baroreflex sympathetic control in normal humans. Circulation. 1991;84:1118–29.
Krum H, Bigger Jr JT, Goldsmith RL, Packer M. Effect of long-term digoxin therapy on autonomic function in patients with chronic heart failure. J Am Coll Cardiol. 1995;25:289–94.
Digitalis IG. The effect of digoxin on mortality and morbidity in patients with heart failure. N Engl J Med. 1997;336:525–33.
Flapan AD, Nolan J, Neilson JM, Ewing DJ. Effect of captopril on cardiac parasympathetic activity in chronic cardiac failure secondary to coronary artery disease. Am J Cardiol. 1992;69:532–5.
Binkley PF, Haas GJ, Starling RC, Nunziata E, Hatton PA, Leier CV, Cody RJ. Sustained augmentation of parasympathetic tone with angiotensin-converting enzyme inhibition in patients with congestive heart failure. J Am Coll Cardiol. 1993;21:655–61.
Adigun AQ, Asiyanbola B, Ajayi AA. Cardiac autonomic function in blacks with congestive heart failure: vagomimetic action, alteration in sympathovagal balance, and the effect of ace inhibition on central and peripheral vagal tone. Cell Mol Biol (Noisy-le-Grand). 2001;47:1063–7.
Adigun AQ, Ajayi AA. The effects of enalapril-digoxin-diuretic combination therapy on nutritional and anthropometric indices in chronic congestive heart failure: preliminary findings in cardiac cachexia. Eur J Heart Fail. 2001;3:359–63.
DiBona GF, Jones SY, Sawin LL. Angiotensin receptor antagonist improves cardiac reflex control of renal sodium handling in heart failure. Am J Physiol. 1998;274:H636–41.
Tambara K, Fujita M, Sumita Y, Miyamoto S, Sekiguchi H, Eiho S, Komeda M. Beneficial effect of candesartan treatment on cardiac autonomic nervous activity in patients with chronic heart failure: simultaneous recording of ambulatory electrocardiogram and posture. Clin Cardiol. 2004;27:300–3.
Vaile JC, Chowdhary S, Osman F, Ross HF, Fletcher J, Littler WA, Coote JH, Townend JN. Effects of angiotensin II (AT1) receptor blockade on cardiac vagal control in heart failure. Clin Sci (Lond). 2001;101:559–66.
Buch AN, Chowdhary S, Coote JH, Townend JN. Effects of nitroglycerin treatment on cardiac autonomic control in heart failure. Clin Auton Res. 2004;14:9–14.
Radaelli A, Cazzaniga M, Viola A, Balestri G, Janetti MB, Signorini MG, Castiglioni P, Azzellino A, Mancia G, Ferrari AU. Enhanced baroreceptor control of the cardiovascular system by polyunsaturated fatty acids in heart failure patients. J Am Coll Cardiol. 2006;48:1600–6.
Holguin F, Tellez-Rojo MM, Lazo M, Mannino D, Schwartz J, Hernandez M, Romieu I. Cardiac autonomic changes associated with fish oil vs soy oil supplementation in the elderly. Chest. 2005;127:1102–7.
Bibevski S, Dunlap ME. Prevention of diminished parasympathetic control of the heart in experimental heart failure. Am J Physiol Heart Circ Physiol. 2004;287:H1780–5.
Lataro RM, Silva CA, Fazan Jr R, Rossi MA, Prado CM, Godinho RO, Salgado HC. Increase in parasympathetic tone by pyridostigmine prevents ventricular dysfunction during the onset of heart failure. Am J Physiol Regul Integr Comp Physiol. 2013;305:R908–16.
Androne AS, Hryniewicz K, Goldsmith R, Arwady A, Katz SD. Acetylcholinesterase inhibition with pyridostigmine improves heart rate recovery after maximal exercise in patients with chronic heart failure. Heart. 2003;89:854–8.
Behling A, Moraes RS, Rohde LE, Ferlin EL, Nobrega AC, Ribeiro JP. Cholinergic stimulation with pyridostigmine reduces ventricular arrhythmia and enhances heart rate variability in heart failure. Am Heart J. 2003;146:494–500.
Serra SM, Costa RV, Teixeira De Castro RR, Xavier SS, Nobrega AC. Cholinergic stimulation improves autonomic and hemodynamic profile during dynamic exercise in patients with heart failure. J Card Fail. 2009;15:124–9.
Okazaki Y, Zheng C, Li M, Sugimachi M. Effect of the cholinesterase inhibitor donepezil on cardiac remodeling and autonomic balance in rats with heart failure. J Physiol Sci. 2010;60:67–74.
Communal C, Singh K, Sawyer DB, Colucci WS. Opposing effects of beta(1)- and beta(2)-adrenergic receptors on cardiac myocyte apoptosis: role of a pertussis toxin-sensitive g protein. Circulation. 1999;100:2210–2.
La Rovere MT, Mortara A, Pantaleo P, Maestri R, Cobelli F, Tavazzi L. Scopolamine improves autonomic balance in advanced congestive heart failure. Circulation. 1994;90:838–43.
Venkatesh G, Fallen EL, Kamath MV, Connolly S, Yusuf S. Double blind placebo controlled trial of short term transdermal scopolamine on heart rate variability in patients with chronic heart failure. Heart. 1996;76:137–43.
Casadei B, Conway J, Forfar C, Sleight P. Effect of low doses of scopolamine on RR interval variability, baroreflex sensitivity, and exercise performance in patients with chronic heart failure. Heart. 1996;75:274–80.
Casadei B, Pipilis A, Sessa F, Conway J, Sleight P. Low doses of scopolamine increase cardiac vagal tone in the acute phase of myocardial infarction. Circulation. 1993;88:353–7.
Pliquett RU, Cornish KG, Zucker IH. Statin therapy restores sympathovagal balance in experimental heart failure. J Appl Physiol. 2003;95:700–4.
Gentlesk PJ, Wiley T, Taylor AJ. A prospective evaluation of the effect of simvastatin on heart rate variability in non-ischemic cardiomyopathy. Am Heart J. 2005;150:478–83.
Yee KM, Pringle SD, Struthers AD. Circadian variation in the effects of aldosterone blockade on heart rate variability and qt dispersion in congestive heart failure. J Am Coll Cardiol. 2001;37:1800–7.
MacFadyen RJ, Barr CS, Struthers AD. Aldosterone blockade reduces vascular collagen turnover, improves heart rate variability and reduces early morning rise in heart rate in heart failure patients. Cardiovasc Res. 1997;35:30–4.
Barr CS, Naas AA, Fenwick M, Struthers AD. Enalapril reduces QTc dispersion in mild congestive heart failure secondary to coronary artery disease. Am J Cardiol. 1997;79:328–33.
Buch AN, Coote JH, Townend JN. Mortality, cardiac vagal control and physical training – what’s the link? Exp Physiol. 2002;87:423–35.
Acknowledgments
A note of thanks to Mark Chapleau, Ph.D., Professor of Medicine at the University of Iowa, for his review and comments.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Olshansky, B., Sullivan, R.M., Colucci, W.S., Sabbah, H.N. (2015). The Parasympathetic Nervous System and Heart Failure: Pathophysiology and Potential Therapeutic Modalities for Heart Failure. In: Jagadeesh, G., Balakumar, P., Maung-U, K. (eds) Pathophysiology and Pharmacotherapy of Cardiovascular Disease. Adis, Cham. https://doi.org/10.1007/978-3-319-15961-4_6
Download citation
DOI: https://doi.org/10.1007/978-3-319-15961-4_6
Publisher Name: Adis, Cham
Print ISBN: 978-3-319-15960-7
Online ISBN: 978-3-319-15961-4
eBook Packages: MedicineMedicine (R0)