Abstract
Optimal lymphoma management requires accurate pretreatment staging and reliable assessment of response, both during and after therapy. Positron emission tomography with computerized tomography (PET/CT) combines functional and anatomical imaging and provides the most sensitive and accurate methods for lymphoma imaging. New guidelines for lymphoma imaging and recently revised criteria for lymphoma staging and response assessment recommend PET/CT staging, treatment monitoring, and response evaluation in all FDG-avid lymphomas, while CT remains the method of choice for non-FDG-avid histologies. Since interim PET imaging has high prognostic value in lymphoma, a number of trials investigate PET-based, response-adapted therapy for non-Hodgkin lymphomas (NHL). PET response is the main determinant of response according to the new response criteria, but PET/CT has little or no role in routine surveillance imaging, the value which is itself questionable. This review presents from a clinical point of view the evidence for the use of imaging and primarily PET/CT in NHL before, during, and after therapy. The reader is given an overview of the current PET-based interventional NHL trials and an insight into possible future developments in the field, including new PET tracers.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Over the last 2–3 decades, we have seen substantial improvements in the prognosis of patients with non-Hodgkin lymphomas (NHL) [1]. These improvements can be attributed to the following factors:
-
1.
Better understanding of the biology of the diseases and advances in molecular pathology, leading to better disease characterization and identification of valuable prognostic markers as well as therapeutic targets.
-
2.
Better imaging methods, most importantly computerized tomography (CT) with cross-sectional images providing more accurate staging, and the more recent introduction of functional imaging, notably positron emission tomography (PET) and PET/CT, which is now the cornerstone of lymphoma imaging.
-
3.
Last and not least; improved treatment regimens, including the introduction of monoclonal anti-CD20 antibodies and high-dose chemotherapy with autologous stem cell support.
The introduction of CT in the 1980s resulted in improved staging accuracy, and CT replaced cumbersome staging procedures such as lymphangiography and staging laparotomy. The introduction of CT was not based on much evidence, as opposed to the more than 1,000 studies investigating PET and PET/CT in lymphoma. During the last 15 years, a large number of studies have shown superior accuracy and sensitivity of PET/CT over CT. Along with patient symptoms and clinical examination, CT and PET/CT are now the cornerstone of all lymphoma imaging. The recent international guidelines for imaging in the staging and response assessment of lymphoma recommend PET/CT for staging and response assessment of all most NHL subtypes [2, 3]. The only PET tracer in widespread clinical use for lymphoma is still 2-[18F]fluor-2-deoxyglucose (FDG). In the following and if not otherwise specified, PET refers to FDG-PET and PET/CT refers to FDG-PET/CT.
2 Staging
2.1 PET/CT for Staging in Non-Hodgkin Lymphomas
Accurate disease staging in NHL is crucial for pretherapy risk assessment and selection of optimal therapy [4, 5]. The main purpose of the staging process, however, depends on the type of the non-Hodgkin lymphoma. Indolent NHL are often considered incurable unless localized at the time of diagnosis. Accordingly, the main question in the staging of indolent NHL is: localized or non-localized disease? Most aggressive NHL, including both B- and T-cell subtypes, are potentially curable when patients are fit for first-line treatment. However, the actual prognosis depends on the specific lymphoma subtype and the presence of high-risk features. The most widely used prognostic scores for NHL are derived from simple clinicopathological features and include disease stage or measures related to disease burden [6–10]. Thus, staging of aggressive non-Hodgkin lymphoma contributes to prognostication and eventually selection of therapy intensity.
The modified Ann Arbor classification originally developed for Hodgkin lymphoma still provides the backbone of disease staging in NHL [3, 11]. The staging workup generally includes clinical examination, blood tests, a routine bone marrow biopsy, and imaging. Contrast-enhanced CT (ceCT) of the neck, chest, and abdomen is required as a minimum, but PET/CT imaging is recommended for all FDG-avid NHL. FDG-PET is routinely used for lymphomas and measures tissue glucose metabolism by use of a short-lived radioactive glucose analog tracer. An advantage of PET-based imaging over stand-alone CT is higher sensitivity and a better discrimination between lymphoma and non-malignant lesions. When PET and CT are combined in a single procedure (PET/CT), the abnormal glucose metabolism is correlated with detailed anatomy, and the increased sensitivity comes not at the expense of decreased specificity; hence, the overall accuracy is increased [12].
2.2 PET/CT Staging in Indolent Non-Hodgkin Lymphomas
Follicular lymphoma (FL): FL is the most common subtype of indolent lymphoma, and the vast majority of FL cases are FDG-avid [13]. The main purpose of FL staging is to identify patients with true localized disease, potentially curable with radiotherapy, but also to risk stratify patients according to the follicular lymphoma international prognostic index (FLIPI) [8]. Compared to conventional stand-alone CT staging, PET/CT detects abnormal activity in normal sized lymph nodes as well as additional involvement of extranodal sites, all resulting in upstaging of 18–32 % of patients and leading to a treatment change in 11–28 % of patients [14–16]. In particular, a large number of patients considered to have limited-stage disease by conventional staging are upstaged by PET/CT to advanced-stage disease (31–62 % according to recent studies) [17, 18]. Upstaging in these cases will change therapy and therefore be clinically relevant. Use of PET/CT also assigns 18 % of the patients to poorer FLIPI risk group, whereas only 6 % are reallocated to lower-risk groups by PET/CT [17]. A replacement of the original FLIPI by a new PET/CT-based risk score has been proposed, as PET/CT-specific findings such as osteomedullar uptake and extranodal disease may be associated with a worse outcome [14].
Mantle cell lymphoma: Mantle cell lymphomas are FDG-avid, and staging with PET/CT results in change of the CT-assessed stage in almost half of the patients, most of whom are upstaged [13, 19]. Therefore, it is possible that patients with truly localized mantle cell lymphoma are more accurately identified with PET/CT. Patients with high FDG-avidity at baseline have a worse outcome than patients with low-grade uptake, which suggests that metabolic activity correlates with tumor aggressiveness [19].
Other indolent lymphomas: The value of PET/CT-based staging in MALT lymphoma and small lymphocytic lymphoma is controversial, and the reported FDG-avidity in these lymphomas is inconsistent [13, 20]. In FDG-avid cases, the uptake is usually discrete, and as for CLL, PET/CT is useful for selecting biopsy site in suspected Richter’s transformation [21, 22]. Nodal marginal zone lymphomas are usually FDG-avid, while this is rarely the case for extranodal marginal zone [13].
Summary: PET/CT for staging of indolent NHL is not well investigated, and the present evidence is derived from few retrospective series. FL, mantle cell lymphoma, and nodal marginal zone lymphoma are FDG-avid, and PET/CT is likely to discriminate between localized and non-localized diseases more accurately than CT. For other indolent lymphomas, PET/CT can guide selection of biopsy site in case of suspected Richter’s transformation. For all indolent lymphomas, the impact on patient outcome of the changes in risk stratification and treatment approach caused by PET/CT is yet largely unclear. Nevertheless, PET/CT is recommended for staging of all FDG-avid indolent lymphomas, while CT remains the recommended imaging tool for non-FDG-avid lymphomas [2].
2.3 PET/CT Staging in Staging of Aggressive Non-Hodgkin Lymphomas
Diffuse large B-cell lymphoma (DLBCL): Even though DLBCL is the most common of all lymphomas, the value of PET/CT for staging of DLBCL has mainly been studied in groups of mixed lymphoma histologies. When compared to CT, PET/CT provides more sensitive and accurate staging and results in upstaging of 10–20 % of patients, some of whom will be allocated to a more advanced treatment [23–25]. The reason for more upstaging is the high sensitivity of PET/CT for extranodal DLBCL. PET/CT accurately identifies bone marrow involvement by DLBCL, and the reported sensitivity and specificity for bone marrow involvement was 94 and 100 % in a recent study [26]. Although most cases of bone marrow involvement by DLBCL are correctly identified by focal bone marrow lesions on PET/CT, a low-grade discordant bone marrow infiltration can be missed [26, 27]. PET/CT staging impacts on pretherapy risk assessment since the number of extranodal sites and that of disease stage are both factors in the widely used IPI risk score [6], and since >1 extranodal site is a risk factor for CNS relapse which often leads to additional CNS prophylactic therapy [28]. The validity of IPI in PET/CT-staged patients is currently investigated. A number of groups have looked at different quantitative applications of PET for staging in DLBCL. While the role of the standardized uptake value (SUV) is unclear, the metabolic tumor volume (MTV) seems to be prognostic of long-term outcome [29, 30]. Furthermore, recent studies demonstrate a good prognostic value of total lesion glycolysis (TLG) which is a combined assessment of volume and metabolism [31, 32].
Burkitt: Burkitt lymphoma is the most aggressive type of non-Hodgkin lymphoma. All cases of Burkitt lymphoma are FDG-avid, and PET/CT staging detects more extranodal disease than conventional staging. Of interest, occult bone marrow involvement is found and occasional clinically important upstagings occur [33].
Peripheral T-cell lymphomas (PTCLs): PTCLs are a heterogeneous group of diseases, and FDG uptake is variable. While the cutaneous T-cell lymphomas have low or no FDG uptake, most nodal PTCLs are FDG-avid albeit with lower FDG uptake than the corresponding B-cell lymphomas [13, 34, 35]. PET/CT identifies additional disease sites in non-cutaneous PTCLs in almost 50 % of the patients, but only changes disease stage in 5 % [35]. Since most nodal PTCLs are disseminated at the time of diagnosis, the additional diagnostic information by PET/CT is less likely to impact overall stage. PET has low sensitivity for detection of bone marrow involvement in PTCLs (20 %) and cannot replace the iliac crest bone marrow biopsy [34]. Nasal-type NK/T-cell lymphoma is most often a disease localized to the head-and-neck region, but PET/CT staging will change stage in one fifth of these patients, and in few patients, disseminated disease is found by PET/CT. Change of treatment by PET/CT in nasal-type NK/T-cell lymphomas is even more common as PET/CT [often accompanied by magnetic resonance imaging (MRI)] modifies radiotherapy planning in many cases [36].
Summary: PET/CT is recommended and widely used for staging of aggressive NHL. Disease stage and pretherapy risk assessment is modified in a large proportion of patients, and for some patients, the additional diagnostic information by PET/CT will change treatment intensity. The impact of PET/CT staging on patient outcome, however, is unknown.
3 PET and PET/CT for Early Treatment Monitoring and Response-Adapted Therapy
A number of well-established pretreatment prognostic factors have been shown to predict survival in large cohort studies of lymphoma patients [6–9]. These prognostic factors are used along with the clinical stage to determine the initial treatment strategy. However, in both indolent and aggressive lymphomas, the tumor response to induction treatment is an important surrogate for other measures of clinical benefit from the treatment, including progression-free survival and overall survival. A reliable and early prediction of response to therapy can possibly separate good-risk patients who will be cured with conventional therapy or even less intensive and less toxic regimens from poor-risk patients for whom an early switch to alternative, more aggressive treatment strategies could improve the likelihood of remission and cure. This concept called risk-adapted therapy is widely recognized as a potential way to achieve higher cure rates with lower or equal risk of treatment-related morbidity and mortality.
Conventional methods for treatment response monitoring are based on morphological criteria, and a reduction in tumor size on CT is the most important determinant [37–39]. However, size reduction is not necessarily an accurate predictor of outcome. But even more importantly, tumor shrinkage takes time and depends on a number of factors in the host. So the rate of structural regression cannot form the basis for therapy response assessment until rather late during treatment, at which point a treatment modification might be less useful.
As opposed to the morphological changes of the lymphoma occurring later during therapy, functional imaging with FDG-PET enables early evaluation of the metabolic changes that take place very early during the treatment induction. Several studies of FDG-PET after 1–4 cycles of chemotherapy in aggressive NHL [40–48] and FL have shown that these early metabolic changes are highly predictive of final treatment response and progression-free survival (PFS). However, treatment-induced inflammation and concomitant infections may lead to false-positive interim scans, and this risk is particularly pertinent in patients treated with intensive regimens [49]. Most evidence is available for performing early interim FDG-PET after 2–4 cycles of chemotherapy. However, reports from Kostakoglu et al. [44, 48] strongly suggest that the predictive value may be as high after only one cycle of chemotherapy. This is of importance for early PET response-adapted treatment regimens, since the treatment adaptation is best performed as early as possible in order to increase the chance of satisfactory remission and to avoid unnecessary ineffective chemotherapy.
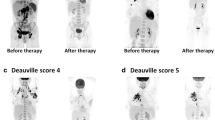
Interim treatment monitoring is more a tradition than an evidence-based practice. Even though it is well established that continued treatment in patients with progression or no response during first-line therapy will invariably lead to treatment failure, the widespread use of CT for treatment monitoring has never been proven to improve the outcome of lymphoma patients. This is true for PET/CT imaging as well, and until we have data from the studies of PET response-adapted therapy, interim imaging should only be performed if clinically justified. The recently revised lymphoma imaging recommendations state that if PET-CT is superior to CT alone to assess early response, if such an assessment is needed, but changing therapy solely on the basis of interim PET-CT is not recommended, unless there is clear evidence of progression. It is recommended that both interim and post-treatment PET scans are interpreted and reported according to the Deauville five-point scale (Table 1) [2]. Figure 1 shows examples of PET images with response scored using the five-point scale.
3.1 Interim PET in Indolent NHL
There is a relative paucity of data on the value of interim FDG-PET in indolent lymphomas. Bishu et al. reported a retrospective series of 31 patients with advanced-stage FL, of whom 11 had FDG-PET scans midway through four cycles of induction chemotherapy. The number of patients was too small for any firm conclusions. Still, the results showed interesting differences: Four patients with some persistent FDG uptake had a mean PFS of 17 months and seven interim PET-negative patients had a mean PFS of 30 months [50]. There are only casuistic reports on the value of interim FDG-PET in localized FL or the more uncommon subtypes of indolent NHL, such as marginal zone lymphoma and small lymphocytic lymphoma.
Even if a clear correlation between interim FDG-PET and treatment outcome is shown in the future, the clinical implications are uncertain, since advanced-stage indolent lymphoma is very different from aggressive NHL. First of all, current first-line therapies for advanced-stage FL are not curative, and it is unclear whether early treatment intensification with autologous stem cell transplant (ASCT) or allogeneic bone marrow transplantation is of any benefit to poor-risk patients. Secondly, the long natural history of FL and the relative success of subsequent treatment lines in inducing lasting remissions mean that a longer time to first progression does not necessarily translate into longer survival.
3.2 Interim PET in Aggressive NHL
In aggressive NHL, PFS ranges from 10 to 50 % at 1 year for early PET-positive patients and from 79 to 100 % at 1 year for early PET-negative patients. The high risk of relapse seen among PET-positive patients is consistently shown in both early and advanced stages. Mikhaeel et al. studied a large cohort of 121 high-grade NHL patients, retrospectively and with a median follow-up of 28.5 months. They confirmed that response on FDG-PET after two or three cycles strongly predicts PFS and OS. Estimated 5-year PFS was 89 % for the PET-negative group, 59 % for patients with minimal residual uptake (MRU) on FDG-PET, and 16 % for the PET-positive group. Survival analyses showed strong associations between early FDG-PET results and PFS and OS [46]. Haioun et al. prospectively studied 90 patients with aggressive NHL, performing FDG-PET after two cycles of chemotherapy. They similarly found 2-year PFS rates of 82 and 43 % in early PET-negative and early PET-positive patients, respectively, and 2-year OS rates of 90 and 60 % [47]. Spaepen et al. compared interim FDG-PET after two cycles of chemotherapy with the International Prognostic Index (IPI). In multivariate analysis, FDG-PET at mid-treatment was a stronger prognostic factor for PFS and OS than was the IPI (P < 0.58 and P < 0.03, respectively) [42]. A number of other more recent studies have investigated the value of interim PET specifically in DLBCL cohorts and found negative predictive values similar to those found in the above-mentioned studies of mixed aggressive NHL populations. But the positive predictive value is variable and generally poor [51, 52]. A possible explanation for the low positive predictive value in aggressive NHL cohorts is the higher risk of inflammation and infections among patients who are treated with the more dose-dense and dose-intensive NHL regimens, as mentioned above. In order to overcome the low positive predictive value of interim PET, some investigators have proposed a semiquantitative approach (relative or absolute change in SUV from baseline to interim scan) as superior to visual PET assessment in patients with DLBCL [53, 54].
Aggressive NHL covers a number of lymphoma subtypes where first-line treatment of both localized and advanced disease stages is given with a curative intent. Aggressive NHL patients who respond poorly to first-line treatment or relapse soon afterward generally have a very poor prognosis even with high-dose salvage regimens. Such poor-risk patients could benefit from having their treatment failure recognized early during first-line therapy in order to switch a more intensive regimen as soon as possible. Much inspired by a similar and more widespread trend in Hodgkin lymphoma, a number of trials are currently investigating early PET response-adapted treatment strategies (Table 2). Most studies test whether early or mid-treatment PET-positive patients with aggressive NHL (mainly DLBCL) will benefit from early escalation to more intensive regimens or even high-dose therapy with autologous stem cell support (ASCT) [55–60]. One de-escalation study compares standard treatment with six cycles R-CHOP to good-risk DLBCL patients to abbreviated therapy with four cycles R-CHOP in patients with a negative PET after two cycles of therapy [61].
4 PET/CT for Post-therapy Response Evaluation of Lymphomas
Response to treatment serves as an important surrogate for other measures of clinical benefit such as progression-free survival and overall survival. Response is also an important guide in decisions regarding continuation or change of therapy. Until recently, response evaluation in NHL was done according to the International Workshop Criteria [62], based mainly on morphological evaluation with a reduction in tumor size on CT being the most important factor. But after completion of therapy, CT scans will often reveal residual masses, and the presence of such masses is not highly predictive of outcome. By conventional methods, it is very difficult to assess whether this represents viable lymphoma or fibrotic scar tissue. An extensive number of studies have shown that FDG-PET performed post-treatment is highly predictive of PFS and OS with and without residual masses on CT. The vast number of studies in aggressive NHL has been summarized in a systematic review [63], while the data on indolent NHL are based on fewer studies [64–66].
Based on these findings, the International Harmonization Project developed new recommendations for response criteria for aggressive malignant lymphomas, incorporating FDG-PET into the definitions of end-of-treatment response in FDG-avid lymphomas [67, 68]. Subsequent retrospective analyses confirmed the superiority of the new PET/CT-based response criteria [69]. The recent revision of the international lymphoma staging and response criteria still uses PET response (according to the Deauville five-point scale) as the main determinant for post-treatment assessment, while measurements of the sum of the product of perpendicular diameters of measureable lesions are mainly important for the definition of progressive disease [3]. The negative predictive value of post-treatment PET/CT is high in most NHL subtypes, but due to a relatively low positive predictive value, treatment failure suspected due to a positive PET should be confirmed by biopsy before additional therapy is initiated. Alternatively, it is often preferable to wait and repeat the PET/CT after 1–2 months, provided the patient is not in clinical progression.
Unlike in Hodgkin lymphoma, the role of post-treatment PET/CT for selection of NHL patients for consolidation radiotherapy is unclear. Based on promising data showing a very high negative predictive value of PET/CT in primary mediastinal B-cell lymphoma [70], the International Extranodal Lymphoma Study Group conducts an experimental approach where consolidation radiotherapy is given only to patients with a PET-positive residual mass [71]. Also, the British Columbia Cancer Agency has since 2005 routinely used post-chemotherapy PET for all patients with residual masses larger than 2 cm. Only PET-positive patients receive consolidation radiotherapy. With 262 patients included in the series and a median follow-up of 45 months, only one of 179 PET-negative patients has received radiotherapy. The 4-year PFS and OS are 74 and 83 %, respectively. Since the data have still only been presented in abstract form, it is unfortunately unclear whether the relapses were predominantly located within or outside the sites of residual disease. If most relapses occur outside the sites of residual disease, there seems to be little value of consolidation radiotherapy in PET-negative patients, but this is not quite so clear whether a substantial number of relapses are seen within the sites of residual disease after first-line treatment [72].
5 Disease Surveillance After First Remission
5.1 Aggressive Non-Hodgkin Lymphomas
The value of routine surveillance imaging in aggressive NHL in first remission is controversial. Despite the lack of evidence, surveillance imaging is often performed with regular intervals during the first years in remission. The rationale behind this may be a presumed better outcome in patients with early and asymptomatic relapse as compared to those patients presenting with overt lymphoma symptoms. Early detection of relapse could in theory be advantageous for the following reasons: (1) lower tumor burden at relapse (and maybe reduced tumor heterogeneity and prevention of treatment resistant clones) and (2) less disease-related impairment of function and organs with better tolerability of treatment. Low disease stage and ECOG performance status 0–1 at relapse are associated with a better outcome in DLBCL [73]. With less than a quarter of the relapses being imaging detected, routine imaging did not contribute significantly to relapse detection in studies based on imaging modalities less sensitive than PET/CT [74–78]. Furthermore, the number of scans per relapse exceeded 100 per preclinical relapse for both DLBCL and PTCLs in first complete remission, and out of all patients in complete remission entering intensive follow-up protocols, only a very small minority (2–3 %) will experience imaging-detected relapse [78]. Few studies have investigated the value of PET/CT in the surveillance of aggressive NHL, mainly reporting on DLBCL. Generally, the rate of imaging-detected relapse seems to be higher when using regular PET/CT surveillance in first remission, and depending on the surveillance protocol, 31–100 % of relapses were detected by imaging [79–81]. However, with the number of PET/CT scans per relapse ranging between 35 and 120 [81, 82] and an estimated cost of up to US$85,550 per preclinical PET/CT-detected relapse, an indiscriminate use of PET/CT surveillance is not likely to be cost-effective. Restricting the use of PET/CT to high-risk patients (IPI > 2) decreases the number of PET/CT scans per relapse to 22 and may tip the balance toward better cost-effectiveness [81]. In patients with transformed indolent lymphomas undergoing routine imaging with PET/CT at least one time during follow-up, the PET/CT-detected relapses were all indolent histologies and less important from a clinical perspective. On the contrary, cases of relapsed DLBCL were symptomatic [83]. Since FDG uptake is unspecific and not restricted to lymphoma recurrence, the positive predictive value (PPV) of PET/CT surveillance in aggressive NHL has been reported low to moderate with values between 21 and 60 % [79, 81, 84]. The increasing use of PET/CT for lymphoma has probably led to safer recognition of uptake patterns characteristic for lymphoma and hence less false-positive reporting. Still, a confirmatory biopsy of lymphoma suspicious findings on PET/CT is mandatory whenever possible. As opposed to the problematic PPV of PET/CT, the negative predictive value has been reported uniformly high and often 100 % [79, 81, 84]. This means that a negative PET/CT study virtually rules out recurrent disease and the need for further investigations in patients with symptoms suggesting lymphoma relapse. Finally, the use of routine surveillance imaging in aggressive NHL can only be justified if early relapse detection is associated with improved survival. Some studies have suggested that imaging-detected relapse of aggressive non-Hodgkin lymphoma could be associated with improved outcome, whereas others found no difference in outcome [76, 78, 81]. However, the retrospective design of these studies does not allow firm conclusions about the causal role of imaging due the presence of important bias (lead time, length time, and guarantee time). Interestingly, a recent randomized trial of surveillance strategies in Hodgkin lymphoma showed that the use of chest X-ray and ultrasound was just as effective as PET/CT in early relapse detection, but at a much lower cost and with higher PPV [85].
5.2 Indolent Non-Hodgkin Lymphomas
While use of routine imaging in the follow-up of aggressive NHL is still widespread, the use of routine imaging in the follow-up of indolent NHL is not recommended. An observational approach without initial treatment is often chosen for patients with an asymptomatic relapse of an indolent lymphoma. An exception could be specific cases where an asymptomatic relapse could lead to unrecognized compression of vital organs such as vascular structures. In any case, PET/CT has no role in this setting.
6 Response Prediction with FDG-PET Before High-Dose Salvage Therapy
Duration of remission prior to relapse and the response to induction therapy are important prognostic factors that predict a good outcome after high-dose chemotherapy with autologous stem cell support (HD + ASCT) [86, 87]. A number of studies have shown that PET performed after induction therapy and before HD + ASCT is predictive of long-term remission. These studies all report short PFS in patients with persistent disease on pretransplant PET [88–93]. In relapsed Hodgkin lymphoma, Moskowitz and his group demonstrated that patients could benefit from changing to another non-cross-resistant regimen before HD + ASCT rather than proceeding straight to high-dose therapy if the PET was still positive after the initial induction therapy [94]. Even though no such data are yet available in NHL, it seems clear that achieving a negative PET before HD + ASCT should be a goal for all patients.
7 Other Imaging Methods than FDG-PET and CT
7.1 Newer PET Tracers
Like other cancers, lymphoma is characterized by deregulated cell cycle progression and most anticancer drugs are designed to inhibit cell proliferation. So a tracer enabling imaging of cell proliferation could be useful for both initial characterization and treatment monitoring of the disease. FDG uptake is somewhat correlated with cell proliferation, but this correlation is weakened by a number of factors, including FDG uptake in non-malignant lesions [95, 96]. The nucleoside [11C]thymidine was the first PET tracer to specifically address cell proliferation. Early studies showed that [11C]thymidine could determine both disease extent and early response to chemotherapy in aggressive NHL patients [97, 98]. However, the short 20-min half-life of 11C along with rapid in vivo metabolism has limited the clinical application of [11C]thymidine. The thymidine analogue 3′-deoxy-3′-[18F]fluorothymidine (FLT) offers a more suitable half-life of 110 min and is stable in vivo [99]. More recent studies have shown that FLT-PET can sensitively identify lymphoma sites [100]. FLT uptake is highly correlated with proliferation rate and may thus be able to distinguish between high- and low-grade lymphomas [101, 102]. And furthermore, recent studies have showed a potential of FLT for imaging early response to treatment in NHL [103–106]. Amino acid metabolism of cancer cells is influenced by catabolic processes favoring tumor growth [107]. It has been shown that increased uptake of amino acids reflects the increased transport and protein synthesis of malignant tissue [108, 109]. This is the background for PET imaging of amino acid metabolism with the labeled amino acids L-[methyl-11C]methionine (MET) and O-2-[18F]fluoroethyl)-L-tyrosine (FET) [110]. Nuutinen et al. [111] studied 32 lymphoma patients and found MET-PET highly sensitive for the detection of disease sites although there was no correlation between MET uptake and patient outcome. Also, MET-PET has an advantage over FDG-PET in imaging of CNS lymphoma [112]. While these results are encouraging, it should be noted that no studies have shown the usefulness or cost-effectiveness of amino acid or nucleoside tracers in large patient cohorts. Furthermore, high physiological tracer uptake in the abdomen limits the usefulness of these tracers for imaging of abdominal and pelvic lymphomas.
With CD20 expressed as a cell surface antigen in most cases of B-NHL, and with CD20 as an important therapeutic target, it is logical to also target CD20 for the purpose of lymphoma imaging. However, most of the available so-called immunoPET tracers are based on full-size, intact antibodies, and as a result of this, it takes a long time for the background activity levels to drop sufficiently to provide acceptable target-to-background ratios. Therefore, Olafsen and colleagues generated a recombinant anti-CD20 fragment which, labeled with a PET tracer, produced high-contrast images in a small-animal model [113]. This and similar targeted PET methods may in the future provide very lymphoma-specific images and prove useful in the staging and treatment monitoring of NHL.
7.2 Magnetic Resonance Imaging
MRI has long been the main structural imaging methods in pediatric lymphomas, due to the reduction in radiation exposure. MRI is used in adult patients to evaluate central nervous system disease and for disease sites in the head-and-neck region (e.g., NK/T-cell lymphomas of nasal type). Recent studies show that diffusion-weighted MRI early during treatment may identify responders before a reduction in tumor size occurs [114].
8 Conclusions and Recommendations
Supported by a large number of studies and in line with the recent consensus recommendations for lymphoma imaging as well as the revised criteria for lymphoma staging and response evaluation, PET/CT is recommended for staging and post-treatment response assessment of all FDG-avid NHLs [2, 3]. There are sufficient data to support the use of interim PET for treatment monitoring in aggressive B-NHL when interim imaging is appropriate. CT is still the preferred imaging method for non-FDG-avid lymphomas, as well as for selected cases where routine surveillance imaging is considered necessary. For interim and post-treatment imaging, the Deauville five-point scale is recommended for interpretation and reporting of PET results. The use of interim PET imaging to guide therapeutic decisions is still investigational and should preferably be done in the setting of clinical trials. Semiquantitative and quantitative PET assessments (SUV, MTV, TLG) are promising, but their role is still to be defined. Currently, much effort is put into the standardization of PET/CT methodology and interpretation, making the results of trials more comparable and ensuring a better translation into clinical practice. Upcoming results of interim PET response-adapted therapy trials are likely to impact on the management of de novo and relapsed NHL patients in the near future. The dissemination of novel and more disease-specific PET tracers may improve the basis for risk- and response-adapted therapy, hopefully in combination with better pretreatment predictive molecular pathology markers.
References
Flowers CR, Armitage JO (2010) A decade of progress in lymphoma: advances and continuing challenges. Clin Lymphoma Myeloma Leuk 10(6):414–423
Barrington SF, Mikhaeel NG, Kostakoglu L, Meignan M, Hutchings M, Mueller SP et al (2014) Role of imaging in the staging and response assessment of lymphoma: consensus of the international conference on malignant lymphomas imaging working group. J Clin Oncol 32(27):3048–3058
Cheson BD, Fisher RI, Barrington SF, Cavalli F, Schwartz LH, Zucca E et al (2014) Recommendations for initial evaluation, staging, and response assessment of hodgkin and non-Hodgkin lymphoma: the lugano classification. J Clin Oncol 32(27):3059–3068
Dreyling M, Ghielmini M, Marcus R, Salles G, Vitolo U, Ladetto M (2014) Newly diagnosed and relapsed follicular lymphoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 25(suppl 3):iii76–82
Tilly H, Vitolo U, Walewski J, da Silva MG, Shpilberg O, Andre M et al (2012) Diffuse large B-cell lymphoma (DLBCL): ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol 23(suppl 7):vii78–vii82
A predictive model for aggressive non-Hodgkin’s lymphoma. The international non-Hodgkin’s lymphoma prognostic factors project. N Engl J Med 1993; 329(14):987-994
Gallamini A, Stelitano C, Calvi R, Bellei M, Mattei D, Vitolo U et al (2004) Peripheral T-cell lymphoma unspecified (PTCL-U): a new prognostic model from a retrospective multicentric clinical study. Blood 103(7):2474–2479
Solal-Celigny P, Roy P, Colombat P, White J, Armitage JO, Arranz-Saez R et al (2004) Follicular lymphoma international prognostic index. Blood 104(5):1258–1265
Hoster E, Dreyling M, Klapper W, Gisselbrecht C, van Hoof A, Kluin-Nelemans HC et al (2008) A new prognostic index (MIPI) for patients with advanced-stage mantle cell lymphoma. Blood 111(2):558–565
Sehn LH, Berry B, Chhanabhai M, Fitzgerald C, Gill K, Hoskins P et al (2007) The revised international prognostic index (R-IPI) is a better predictor of outcome than the standard IPI for patients with diffuse large B-cell lymphoma treated with R-CHOP. Blood 109(5):1857–1861
Carbone PP, Kaplan HS, Musshoff K, Smithers DW, Tubiana M (1971) Report of the committee on Hodgkin’s disease staging classification. Cancer Res 31(11):1860–1861
Kwee TC, Kwee RM, Nievelstein RA (2008) Imaging in staging of malignant lymphoma: a systematic review. Blood 111(2):504–516
Weiler-Sagie M, Bushelev O, Epelbaum R, Dann EJ, Haim N, Avivi I et al (2010) 18F-FDG avidity in lymphoma readdressed: a study of 766 patients. J Nucl Med 51(1):25–30
Le Dortz L, de Guibert S, Bayat S, Devillers A, Houot R, Rolland Y et al (2010) Diagnostic and prognostic impact of 18F-FDG PET/CT in follicular lymphoma. Eur J Nucl Med Mol Imaging 37(12):2307–2314
Janikova A, Bolcak K, Pavlik T, Mayer J, Kral Z (2008) Value of [18F]fluorodeoxyglucose positron emission tomography in the management of follicular lymphoma: the end of a dilemma? Clin Lymphoma Myeloma 8(5):287–293
Scott AM, Gunawardana DH, Wong J, Kirkwood I, Hicks RJ, Ho SI et al (2009) Positron emission tomography changes management, improves prognostic stratification and is superior to gallium scintigraphy in patients with low-grade lymphoma: results of a multicentre prospective study. Eur J Nucl Med Mol Imaging 36(3):347–353
Luminari S, Biasoli I, Arcaini L, Versari A, Rusconi C, Merli F et al (2013) The use of FDG-PET in the initial staging of 142 patients with follicular lymphoma: a retrospective study from the FOLL05 randomized trial of the Fondazione Italiana Linfomi. Ann Oncol 24(8):2108–2112
Wirth A, Foo M, Seymour JF, MacManus MP, Hicks RJ (2008) Impact of [18F] fluorodeoxyglucose positron emission tomography on staging and management of early-stage follicular non-Hodgkin lymphoma. Int J Radiat Oncol Biol Phys 71(1):213–219
Karam M, Ata A, Irish K, Feustel PJ, Mottaghy FM, Stroobants SG et al (2009) FDG positron emission tomography/computed tomography scan may identify mantle cell lymphoma patients with unusually favorable outcome. Nucl Med Commun 30(10):770–778
Perry C, Herishanu Y, Metzer U, Bairey O, Ruchlemer R, Trejo L et al (2007) Diagnostic accuracy of PET/CT in patients with extranodal marginal zone MALT lymphoma. Eur J Haematol 79(3):205–209
Falchi LK (2014) Correlation between FDG/PET, histology, characteristics, and survival in 332 patients with chronic lymphoid leukemia. Blood 123(18):2783–2790
Papajik T, Myslivecek M, Urbanova R, Buriankova E, Kapitanova Z, Prochazka V et al (2013) 2-[18F]fluoro-2-deoxy-d-glucose positron emission tomography/computed tomography examination in patients with chronic lymphocytic leukemia may reveal Richter transformation. Leuk Lymphoma 55(2):314–319
Raanani P, Shasha Y, Perry C, Metser U, Naparstek E, Apter S et al (2006) Is CT scan still necessary for staging in Hodgkin and non-Hodgkin lymphoma patients in the PET/CT era? Ann Oncol 17(1):117–122
Elstrom RL, Leonard JP, Coleman M, Brown RK (2008) Combined PET and low-dose, noncontrast CT scanning obviates the need for additional diagnostic contrast-enhanced CT scans in patients undergoing staging or restaging for lymphoma. Ann Oncol 19(10):1770–1773
Pelosi E, Pregno P, Penna D, Deandreis D, Chiappella A, Limerutti G et al (2008) Role of whole-body [18F] fluorodeoxyglucose positron emission tomography/computed tomography (FDG-PET/CT) and conventional techniques in the staging of patients with Hodgkin and aggressive non Hodgkin lymphoma. Radiol Med 113(4):578–590
Khan AB, Barrington SF, Mikhaeel NG, Hunt AA, Cameron L, Morris T et al (2013) PET-CT staging of DLBCL accurately identifies and provides new insight into the clinical significance of bone marrow involvement. Blood 122(1):61–67
Paone G, Itti E, Haioun C, Gaulard P, Dupuis J, Lin C et al (2009) Bone marrow involvement in diffuse large B-cell lymphoma: correlation between FDG-PET uptake and type of cellular infiltrate. Eur J Nucl Med Mol Imaging 36(5):745–750
Boehme V, Zeynalova S, Kloess M, Loeffler M, Kaiser U, Pfreundschuh M et al (2007) Incidence and risk factors of central nervous system recurrence in aggressive lymphoma—a survey of 1693 patients treated in protocols of the German high-grade non-Hodgkin’s lymphoma study group (DSHNHL). Ann Oncol 18(1):149–157
Song MK, Chung JS, Shin HJ, Lee SM, Lee SE, Lee HS et al (2012) Clinical significance of metabolic tumor volume by PET/CT in stages II and III of diffuse large B cell lymphoma without extranodal site involvement. Ann Hematol 91(5):697–703
Gallicchio R, Mansueto G, Simeon V, Nardelli A, Guariglia R, Capacchione D et al (2014) F-18 FDG PET/CT quantization parameters as predictors of outcome in patients with diffuse large B-cell lymphoma. Eur J Haematol 92(5):382–389
Esfahani SA, Heidari P, Halpern EF, Hochberg EP, Palmer EL, Mahmood U (2013) Baseline total lesion glycolysis measured with (18)F-FDG PET/CT as a predictor of progression-free survival in diffuse large B-cell lymphoma: a pilot study. Am J Nucl Med Mol Imaging 3(3):272–281
Kim TM, Paeng JC, Chun IK, Keam B, Jeon YK, Lee SH et al (2013) Total lesion glycolysis in positron emission tomography is a better predictor of outcome than the international prognostic index for patients with diffuse large B cell lymphoma. Cancer 119(6):1195–1202
Carrillo-Cruz E, Marin-Oyaga VA, Rodriguez M, Borrego-Dorado I, de la Cruz Vicente F, Cantero EQ et al (2014) Role of 18F-FDG-PET/CT in the management of burkitt lymphoma. Eur J Haematol
Kako S, Izutsu K, Ota Y, Minatani Y, Sugaya M, Momose T et al (2007) FDG-PET in T-cell and NK-cell neoplasms. Ann Oncol 18(10):1685–1690
Casulo C, Schoder H, Feeney J, Lim R, Maragulia J, Zelenetz AD et al (2013) 18F-fluorodeoxyglucose positron emission tomography in the staging and prognosis of T cell lymphoma. Leuk Lymphoma 54(10):2163–2167
Moon SH, Cho SK, Kim WS, Kim SJ, Chan Ahn Y, Choe YS et al (2003) The role of 18F-FDG PET/CT for initial staging of nasal type natural killer/t-cell lymphoma: a comparison with conventional staging methods. J Nucl Med 54(7):1039–1044
Rankin SC (2003) Assessment of response to therapy using conventional imaging. Eur J Nucl Med Mol Imaging 30(1):S56–S64
Armitage JO, Weisenburger DD, Hutchins M, Moravec DF, Dowling M, Sorensen S et al (1986) Chemotherapy for diffuse large-cell lymphoma–rapidly responding patients have more durable remissions. J Clin Oncol 4(2):160–164
Gupta RK, Gospodarowicz MK, Lister TA (1999) Clinical evaluation and staging. In: Mauch P, Armitage JO, Diehl V, Hoppe R, Weiss LM (eds) Hodgkin’s disease. Lippincott Williams and Wilkins, Philadelphia, pp 223–240
Jerusalem G, Beguin Y, Fassotte MF, Najjar F, Paulus P, Rigo P et al (2000) Persistent tumor 18F-FDG uptake after a few cycles of polychemotherapy is predictive of treatment failure in non-Hodgkin’s lymphoma. Haematologica 85(6):613–618
Mikhaeel NG, Timothy AR, O’Doherty MJ, Hain S, Maisey MN (2000) 18-FDG-PET as a prognostic indicator in the treatment of aggressive non-Hodgkin’s lymphoma-comparison with CT. Leuk Lymphoma 39(5–6):543–553
Spaepen K, Stroobants S, Dupont P, Vandenberghe P, Thomas J, de Groot T et al (2002) Early restaging positron emission tomography with (18)F-fluorodeoxyglucose predicts outcome in patients with aggressive non-Hodgkin’s lymphoma. Ann Oncol 13(9):1356–1363
Hoekstra OS, Ossenkoppele GJ, Golding R, van Lingen A, Visser GW, Teule GJ et al (1993) Early treatment response in malignant lymphoma, as determined by planar fluorine-18-fluorodeoxyglucose scintigraphy. J Nucl Med 34(10):1706–1710
Kostakoglu L, Coleman M, Leonard JP, Kuji I, Zoe H, Goldsmith SJ (2002) PET predicts prognosis after 1 cycle of chemotherapy in aggressive lymphoma and Hodgkin’s disease. J Nucl Med 43(8):1018–1027
Torizuka T, Nakamura F, Kanno T, Futatsubashi M, Yoshikawa E, Okada H et al (2004) Early therapy monitoring with FDG-PET in aggressive non-Hodgkin’s lymphoma and Hodgkin’s lymphoma. Eur J Nucl Med Mol Imaging 31(1):22–28
Mikhaeel NG, Hutchings M, Fields PA, O’Doherty MJ, Timothy AR (2005) FDG-PET after two to three cycles of chemotherapy predicts progression-free and overall survival in high-grade non-Hodgkin lymphoma. Ann Oncol 16(9):1514–1523
Haioun C, Itti E, Rahmouni A, Brice P, Rain JD, Belhadj K et al (2005) [18F]fluoro-2-deoxy-D-glucose positron emission tomography (FDG-PET) in aggressive lymphoma: an early prognostic tool for predicting patient outcome. Blood 106(4):1376–1381
Kostakoglu L, Goldsmith SJ, Leonard JP, Christos P, Furman RR, Atasever T et al (2006) FDG-PET after 1 cycle of therapy predicts outcome in diffuse large cell lymphoma and classic Hodgkin disease. Cancer 107(11):2678–2687
Han HS, Escalon MP, Hsiao B, Serafini A, Lossos IS (2009) High incidence of false-positive PET scans in patients with aggressive non-Hodgkin’s lymphoma treated with rituximab-containing regimens. Ann Oncol 20(2):309–318
Bishu S, Quigley JM, Bishu SR, Olsasky SM, Stem RA, Shostrom VK et al (2007) Predictive value and diagnostic accuracy of F-18-fluoro-deoxy-glucose positron emission tomography treated grade 1 and 2 follicular lymphoma. Leuk Lymphoma 48(8):1548–1555
Cashen AF, Dehdashti F, Luo J, Homb A, Siegel BA, Bartlett NL (2011) 18F-FDG PET/CT for early response assessment in diffuse large b-cell lymphoma: poor predictive value of international harmonization project interpretation. J Nucl Med 52(3):386–392
Moskowitz CH, Schoder H, Teruya-Feldstein J, Sima C, Iasonos A, Portlock CS et al (2010) Risk-adapted dose-dense immunochemotherapy determined by interim FDG-PET in advanced-stage diffuse large B-cell lymphoma. J Clin Oncol 28(11):1896–1903
Itti E, Lin C, Dupuis J, Paone G, Capacchione D, Rahmouni A et al (2009) Prognostic value of interim 18F-FDG PET in patients with diffuse large B-cell lymphoma: SUV-based assessment at 4 cycles of chemotherapy. J Nucl Med 50(4):527–533
Casasnovas RO, Meignan M, Berriolo-Riedinger A, Bardet S, Julian A, Thieblemont C et al (2011) SUVmax reduction improves early prognosis value of interim positron emission tomography scans in diffuse large B-cell lymphoma. Blood 118(1):37–43
Fludeoxyglucose F 18 positron emission tomography in predicting risk of relapse in patients with non-Hodgkin’s lymphoma who are undergoing combination chemotherapy with or without autologous stem cell or bone marrow transplant. http://www.clinicaltrials.gov/ct2/show/NCT00238368 Accessed Dec 17 2014
Therapy for patients with untreated age-adjusted international prognostic index low-intermediate risk, high-intermediate risk, or high risk diffuse large B cell lymphoma. http://www.clinicaltrials.gov/ct2/show/NCT00712582 Accessed Dec 17 2014
Positron emission tomography guided therapy of aggressive non-Hodgkin’s lymphomas (PETAL). http://www.clinicaltrials.gov/ct2/show/NCT00554164 Accessed Dec 17 2014
Tailoring treatment for B cell non-Hodgkin’s lymphoma based on pet scan results mid treatment. http://www.clinicaltrials.gov/ct2/show/NCT00324467 Accessed Dec 17 2014
FDG-PET-stratified R-DICEP and R-beam/ASCT for diffuse large B-cell lymphoma (PET chop). http://www.clinicaltrials.gov/ct2/show/NCT00530179 Accessed Dec 17 2014
A study of two associations of rituximab and chemotherapy, with a PET-driven strategy, in lymphoma (LNH2007-3B). http://www.clinicaltrials.gov/ct2/show/NCT00498043 Accessed Dec 17 2014
Study evaluating the non-inferiority of a treatment adapted to the early response evaluated with 18f-fdg pet compared to a standard treatment, for patients aged from 18 to 80 years with low risk (aa IPI = 0) diffuse large B-cells non Hodgkin’s lymphoma CD 20+. http://www.clinicaltrials.gov/ct2/show/NCT01285765 Accessed Dec 17 2014
Cheson BD, Horning SJ, Coiffier B, Shipp MA, Fisher RI, Connors JM et al (1999) Report of an international workshop to standardize response criteria for non-Hodgkin’s lymphomas. NCI sponsored international working group. J Clin Oncol 17(4):1244
Zijlstra JM, Lindauer-van der Werf G, Hoekstra OS, Hooft L, Riphagen II, Huijgens PC (2006) 18F-fluoro-deoxyglucose positron emission tomography for post-treatment evaluation of malignant lymphoma: a systematic review. Haematologica 91(4):522–529
Trotman J, Fournier M, Lamy T, Seymour JF, Sonet A, Janikova A et al (2011) Positron emission tomography–computed tomography (PET-CT) after induction therapy is highly predictive of patient outcome in follicular lymphoma: analysis of PET-CT in a subset of PRIMA trial participants. J Clin Oncol 29(23):3194–3200
Zinzani PL, Musuraca G, Alinari L, Fanti S, Tani M, Stefoni V et al (2007) Predictive role of positron emission tomography in the outcome of patients with follicular lymphoma. Clin Lymphoma Myeloma 7(4):291–295
Tychyj-Pinel C, Ricard F, Fulham M, Fournier M, Meignan M, Lamy T et al (2014) PET/CT assessment in follicular lymphoma using standardized criteria: central review in the PRIMA study. Eur J Nucl Med Mol Imaging 41(3):408–415
Cheson BD, Pfistner B, Juweid ME, Gascoyne RD, Specht L, Horning SJ et al (2007) Revised response criteria for malignant lymphoma. J Clin Oncol 25(5):579–586
Juweid ME, Stroobants S, Hoekstra OS, Mottaghy FM, Dietlein M, Guermazi A et al (2007) Use of positron emission tomography for response assessment of lymphoma: consensus of the imaging subcommittee of international harmonization project in lymphoma. J Clin Oncol 25(5):571–578
Brepoels L, Stroobants S, De Wever W, Spaepen K, Vandenberghe P, Thomas J et al (2007) Aggressive and indolent non-Hodgkin’s lymphoma: response assessment by integrated international workshop criteria. Leuk Lymphoma 48(8):1522–1530
Martelli M, Ceriani L, Zucca E, Zinzani PL, Ferreri AsJM, Vitolo U et al (2014) [18F]Fluorodeoxyglucose positron emission tomography predicts survival after chemoimmunotherapy for primary mediastinal large B-cell lymphoma: results of the international extranodal lymphoma study group IELSG-26 study. J Clin Oncol 32(17):1769–1775
International extranodal lymphoma study group (IELSG): a randomized, open-label two-arm phase iii comparative study assessing the role of involved mediastinal radiotherapy after rituximab containing chemotherapy regimens to patients with newly diagnosed primary mediastinal large B-cell lymphoma. http://clinicaltrials.gov/ct2/show/NCT01599559 Accessed Dec 17 2014
Sehn LH, Klasa R, Shenkier T, Villa D, Slack GW, Gascoyne RD et al (2013) Long-term experience with PET-guided consolidative radiation therapy (XRT) in patients with advanced stage diffuse large B-cell lymphoma (DLBCL) treated with R-CHOP. Hematol Oncol 31(S1):137 1-6-2013
Hamlin PAZ (2003) Age-adjusted International Prognostic Index predicts autologous stem cell transplantation outcome for patients with relapsed or primary refractory diffuse large B-cell lymphoma. Blood 102(6):1989–1996
Weeks JC, Yeap BY, Canellos GP, Shipp MA (1991) Value of follow-up procedures in patients with large-cell lymphoma who achieve a complete remission. J Clin Oncol 9(7):1196–1203
Guppy AE, Tebbutt NC, Norman A, Cunningham D (2003) The role of surveillance CT scans in patients with diffuse large B-cell non-Hodgkin’s lymphoma. Leuk Lymphoma 44(1):123–125
Liedtke M, Hamlin PA, Moskowitz CH, Zelenetz AD (2006) Surveillance imaging during remission identifies a group of patients with more favorable aggressive NHL at time of relapse: a retrospective analysis of a uniformly-treated patient population. Ann Oncol 17(6):909–913
Lin TL, Kuo MC, Shih LY, Dunn P, Wang PN, Wu JH et al (2012) Value of surveillance computed tomography in the follow-up of diffuse large B-cell and follicular lymphomas. Ann Hematol 91(11):1741–1745
El Galaly TC, Mylam KJ, Boegsted M, Brown P, Rossing M, Gang AO et al (2014) Role of routine imaging in detecting recurrent lymphoma: a review of 258 patients with relapsed aggressive non-Hodgkin and Hodgkin lymphoma. Am J Hematol 89(6):575–580
El Galaly T, Prakash V, Christiansen I, Madsen J, Johansen P, Boegsted M et al (2011) Efficacy of routine surveillance with positron emission tomography/computed tomography in aggressive non-Hodgkin lymphoma in complete remission: status in a single center. Leuk Lymphoma 52(4):597–603
Zinzani PL, Stefoni V, Tani M, Fanti S, Musuraca G, Castellucci P et al (2009) Role of [18F]fluorodeoxyglucose positron emission tomography scan in the follow-up of lymphoma. J Clin Oncol 27(11):1781–1787
Cheah CY, Hofman MS, Dickinson M, Wirth A, Westerman D, Harrison SJ et al (2013) Limited role for surveillance PET-CT scanning in patients with diffuse large B-cell lymphoma in complete metabolic remission following primary therapy. Br J Cancer 109(2):312–317
Abel GA, Vanderplas A, Rodriguez MA, Crosby AL, Czuczman MS, Niland JC et al (2011) High rates of surveillance imaging for treated diffuse large B-cell lymphoma: findings from a large national database. Leuk Lymphoma, 1–4
Cheah C, Dickinson M, Hofman M, George A, Ritchie D, Prince HM et al (2014) Limited clinical benefit for surveillance PET-CT scanning in patients with histologically transformed lymphoma in complete metabolic remission following primary therapy. Ann Hematol 93(7):1193–1200
Avivi I, Zilberlicht A, Dann EJ, Leiba R, Faibish T, Rowe JM et al (2013) Strikingly high false positivity of surveillance FDG-PET/CT scanning among patients with diffuse large cell lymphoma in the rituximab era. Am J Hematol 88(5):400–405
Picardi M, Pugliese N, Cirillo M, Zeppa P, Cozzolino I, Ciancia G et al (2014) Advanced-stage Hodgkin lymphoma: US/chest radiography for detection of relapse in patients in first complete remission—randomized trial of routine surveillance imaging procedures. Radiology 272(1):262–274
Haioun C, Lepage E, Gisselbrecht C, Salles G, Coiffier B, Brice P et al (2000) Survival benefit of high-dose therapy in poor-risk aggressive non-Hodgkin’s lymphoma: final analysis of the prospective LNH87-2 protocol–a groupe d’Etude des lymphomes de l’Adulte study. J Clin Oncol 18(16):3025–3030
Stiff PJ, Dahlberg S, Forman SJ, McCall AR, Horning SJ, Nademanee AP et al (1998) Autologous bone marrow transplantation for patients with relapsed or refractory diffuse aggressive non-Hodgkin’s lymphoma: value of augmented preparative regimens–a southwest oncology group trial. J Clin Oncol 16(1):48–55
Becherer A, Mitterbauer M, Jaeger U, Kalhs P, Greinix HT, Karanikas G et al (2002) Positron emission tomography with [18F]2-fluoro-D-2-deoxyglucose (FDG-PET) predicts relapse of malignant lymphoma after high-dose therapy with stem cell transplantation. Leukemia 16(2):260–267
Terasawa T, Dahabreh IJ, Nihashi T (2010) Fluorine-18-fluorodeoxyglucose positron emission tomography in response assessment before high-dose chemotherapy for lymphoma: a systematic review and meta-analysis. Oncologist 15(7):750–759
Dickinson M, Hoyt R, Roberts AW, Grigg A, Seymour JF, Prince HM et al (2010) Improved survival for relapsed diffuse large B cell lymphoma is predicted by a negative pre-transplant FDG-PET scan following salvage chemotherapy. Br J Haematol 150(1):39–45
Cremerius U, Fabry U, Wildberger JE, Zimny M, Reinartz P, Nowak B et al (2002) Pre-transplant positron emission tomography (PET) using fluorine-18-fluoro-deoxyglucose (FDG) predicts outcome in patients treated with high-dose chemotherapy and autologous stem cell transplantation for non-Hodgkin’s lymphoma. Bone Marrow Transplant 30(2):103–111
Spaepen K, Stroobants S, Dupont P, Vandenberghe P, Maertens J, Bormans G et al (2003) Prognostic value of pretransplantation positron emission tomography using fluorine 18-fluorodeoxyglucose in patients with aggressive lymphoma treated with high-dose chemotherapy and stem cell transplantation. Blood 102(1):53–59
Filmont JE, Czernin J, Yap C, Silverman DH, Quon A, Phelps ME et al (2003) Value of F-18 fluorodeoxyglucose positron emission tomography for predicting the clinical outcome of patients with aggressive lymphoma prior to and after autologous stem-cell transplantation. Chest 124(2):608–613
Moskowitz CH, Matasar MJ, Zelenetz AD, Nimer SD, Gerecitano J, Hamlin P et al (2012) Normalization of pre-ASCT, FDG-PET imaging with second-line, non–cross-resistant, chemotherapy programs improves event-free survival in patients with Hodgkin lymphoma. Blood 119(7):1665–1670
Buck AK, Halter G, Schirrmeister H, Kotzerke J, Wurziger I, Glatting G et al (2003) Imaging proliferation in lung tumors with PET: 18F-FLT versus 18F-FDG. J Nucl Med 44(9):1426–1431
Sandherr M, von Schilling C, Link T, Stock K, von Bubnoff N, Peschel C et al (2001) Pitfalls in imaging Hodgkin’s disease with computed tomography and positron emission tomography using fluorine-18-fluorodeoxyglucose. Ann Oncol 12(5):719–722
Martiat P, Ferrant A, Labar D, Cogneau M, Bol A, Michel C et al (1988) In vivo measurement of carbon-11 thymidine uptake in non-Hodgkin’s lymphoma using positron emission tomography. J Nucl Med 29(10):1633–1637
Shields AF, Mankoff DA, Link JM, Graham MM, Eary JF, Kozawa SM et al (1998) Carbon-11-thymidine and FDG to measure therapy response. J Nucl Med 39(10):1757–1762
Shields AF, Grierson JR, Dohmen BM, Machulla HJ, Stayanoff JC, Lawhorn-Crews JM et al (1998) Imaging proliferation in vivo with [F-18]FLT and positron emission tomography. Nat Med 4(11):1334–1336
Buchmann I, Neumaier B, Schreckenberger M, Reske S (2004) [18F]3′-deoxy-3′-fluorothymidine-PET in NHL patients: whole-body biodistribution and imaging of lymphoma manifestations–a pilot study. Cancer Biother Radiopharm 19(4):436–442
Buck AK, Bommer M, Stilgenbauer S, Juweid M, Glatting G, Schirrmeister H et al (2006) Molecular imaging of proliferation in malignant lymphoma. Cancer Res 66(22):11055–11061
Kasper B, Egerer G, Gronkowski M, Haufe S, Lehnert T, Eisenhut M et al (2007) Functional diagnosis of residual lymphomas after radiochemotherapy with positron emission tomography comparing FDG- and FLT-PET. Leuk Lymphoma 48(4):746–753
Buck AK, Kratochwil C, Glatting G, Juweid M, Bommer M, Tepsic D et al (2007) Early assessment of therapy response in malignant lymphoma with the thymidine analogue [18F]FLT. Eur J Nucl Med Mol Imaging 34(11):1775–1782
Herrmann K, Buck AK, Schuster T, Rudelius M, Wester HJ, Graf N et al (2011) A pilot study to evaluate 3′-Deoxy-3′-18F-Fluorothymidine PET for initial and early response imaging in mantle cell lymphoma. J Nucl Med 52(12):1898–1902
Herrmann K, Buck AK, Schuster T, Junger A, Wieder H, Graf N et al (2011) Predictive value of initial 18F-FLT uptake in patients with aggressive non-Hodgkin lymphoma receiving R-CHOP treatment. J Nucl Med 52(5):690–696
Graf N, Herrmann K, den Hollander J, Fend F, Schuster T, Wester HJ et al (2008) Imaging proliferation to monitor early response of lymphoma to cytotoxic treatment. Mol Imaging Biol 10(6):349–355
Hoffman RM (1984) Altered methionine metabolism, DNA methylation and oncogene expression in carcinogenesis. A review and synthesis. Biochim Biophys Acta 738(1–2):49–87
Stern PH, Wallace CD, Hoffman RM (1984) Altered methionine metabolism occurs in all members of a set of diverse human tumor cell lines. J Cell Physiol 119(1):29–34
Wheatley DN (1982) On the problem of linear incorporation of amino acids into cell protein. Experientia 38(7):818–820
Leskinen-Kallio S, Ruotsalainen U, Nagren K, Teras M, Joensuu H (1991) Uptake of carbon-11-methionine and fluorodeoxyglucose in non-Hodgkin’s lymphoma: a PET study. J Nucl Med 32(6):1211–1218
Nuutinen J, Leskinen S, Lindholm P, Soderstrom KO, Nagren K, Huhtala S et al (1998) Use of carbon-11 methionine positron emission tomography to assess malignancy grade and predict survival in patients with lymphomas. Eur J Nucl Med 25(7):729–735
Kawase Y, Yamamoto Y, Kameyama R, Kawai N, Kudomi N, Nishiyama Y (2011) Comparison of 11C-Methionine PET and 18F-FDG PET in patients with primary central nervous system lymphoma. Mol Imaging Biol 13(6):1284–1289
Olafsen T, Betting D, Kenanova VE, Salazar FB, Clarke P, Said J et al (2009) Recombinant anti-CD20 antibody fragments for small-animal PET imaging of B-cell lymphomas. J Nucl Med 50(9):1500–1508
van Ufford HMEQ, Kwee TC, Beek FJ, van Leeuwen MS, Takahara T, Fijnheer R et al (2011) Newly diagnosed lymphoma: initial results with whole-body T1-weighted, STIR, and diffusion-weighted MRI compared with 18F-FDG PET/CT. Am J Roentgenol 196(3):662–669
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer International Publishing Switzerland
About this chapter
Cite this chapter
El-Galaly, T.C., Hutchings, M. (2015). Imaging of Non-Hodgkin Lymphomas: Diagnosis and Response-Adapted Strategies. In: Evens, A., Blum, K. (eds) Non-Hodgkin Lymphoma. Cancer Treatment and Research, vol 165. Springer, Cham. https://doi.org/10.1007/978-3-319-13150-4_5
Download citation
DOI: https://doi.org/10.1007/978-3-319-13150-4_5
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-13149-8
Online ISBN: 978-3-319-13150-4
eBook Packages: MedicineMedicine (R0)