Abstract
Blood-material interactions are critical to the success of implantable medical devices that are used in thousands of patients every day. Some common blood-contacting devices include catheters, stents, vascular grafts, heart valve prostheses, and extracorporeal circulation/membrane oxygenation systems. Among other complications, thrombosis and clot formation remains one of the major challenges in clinical application of these devices. The initial biological response of blood to a foreign surface is the rapid adsorption of plasma proteins, which is followed by platelet adhesion and activation, and ultimately thrombus formation. The key factors in clot formation are the chemical and physical nature of the surfaces and their interactions with the blood components, such as platelets and plasma proteins. Despite decades of research, an ideal non-thrombogenic surface is still yet to be identified and clinical use of these blood-contacting devices requires use of anticoagulation agents, increasing the risk of bleeding in patients. In this chapter, we will review some of the current and most promising strategies that have been used over the years to develop polymeric materials with improved hemocompatibility, including highly hydrophilic or hydrophobic surfaces, albumin coated surfaces, zwitterionic polymers, attached endothelial cells, patterned surfaces, immobilized heparin, and nitric oxide (NO) releasing/generating surfaces. We will also discuss some of the important techniques employed (using in vitro and in vivo models) to assess the hemocompatibility of any new material, including the measurement of platelet preservation, platelet and protein adhesion, the effect of flow rates on thrombosis, and the ultimate surface clot area.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
Hemocompatibility of Common Blood-Contacting Devices
Blood-material interaction is critical to the success of implantable medical devices, including simple catheters, stents and grafts, insulation materials for electrical leads of pacemakers and defibrillators, and complex extracorporeal artificial organs, which are used in thousands of patients every day [1]. Polymers, including polyurethanes, silicone rubber (SR), and poly(vinyl chloride), are used extensively in the healthcare industry to fabricate such biomedical devices. Thrombosis is one of the primary problems associated with clinical application of blood-contacting materials, which can cause serious complications in patients and ultimately failure of the device’s functionality [2]. Thrombus formation can lead to significant consequences such as complete obstruction of blood vessels in which stents are placed [3], occlusion of catheters [4] and small diameter vascular grafts [1], errant results from implantable chemical sensors [1], embolic complications with artificial hearts [5], and extensive blood damage and platelet consumption during extracorporeal membrane oxygenation [5]. Such complications can result in significantly increased medical costs, extended hospitalization, amputation, or increased morbidity. Despite a thorough understanding of the mechanisms of blood-surface interactions and decades of bioengineering research effort, the ideal non-thrombogenic prosthetic surface remains unidentified [5]. Over the last 50 years, much has been learned about foreign surface-induced thrombosis and the attempt to prevent it with systemic anticoagulation and surface modifications. In a clinical setting, many of these devices require the use of systemic anticoagulation (e.g., heparin) to avoid device failure [6]. The long-term use of anticoagulants can also have adverse effects, including hemorrhage and thrombocytopenia [7]. Despite these complications, heparin is still used as the standard anticoagulation therapy for patients on extracorporeal circulation (ECC), but the use of heparin does not prevent platelet activation and consumption.
Blood and the Coagulation Cascade
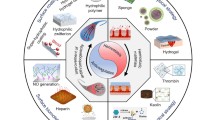
Biomaterial related thrombosis is a complex process, where the initial biological response when blood comes in contact with a foreign surface is protein adsorption, which is followed by platelet adhesion and activation, leading to thrombus formation (Fig. 16.1) [8]. Plasma proteins adsorb onto the surface of the implanted device (e.g., fibrinogen, fibronectin, von Willebrand factor (vWF), etc.) within seconds. These proteins further interact with receptors on the plasma membranes of platelets and facilitate platelet adhesion on the surface [9]. Upon contact with the foreign surface and platelet activation, a conformational change occurs, leading to the exposure of the glycoprotein GPIIb/IIIa receptor that binds platelets to fibrinogen [3]. Platelet activation also leads to conformation changes and the excretion of intracellular granulates containing agents (e.g., coagulation Factors V and VIII, adhesion molecules P-selectin and vWF, calcium ions, etc.) that further induce activation and aggregation of more platelets. This also activates the coagulation cascade, where a series of self-amplifying reactions triggered through surface contact (intrinsic pathway) or tissue factors (extrinsic pathway) ultimately converge at the final common pathway to form a thrombus (Fig. 16.2 ). Throughout the coagulation cascade, the normally inactive factors become enzymatically activated (e.g., Factor X becomes Factor Xa) by surface contact or cleavage by other activated enzymes. This sequence allows for rapid activation of other clotting factors and amplification of the entire coagulation cascade. When blood comes in contact with a foreign surface (biomedical device) protein adsorption will trigger the first step of the intrinsic pathway, with activation Factor XII to Factor XIIa. The presence of Factor XIIa will lead to the activation of Factor XI and eventually lead to the activation of Factor X and formation of thrombin (Factor II) in the common pathway [3]. Thrombin converts fibrinogen into fibrin and also activates Factor XIII, which cross-links and stabilizes fibrin into an insoluble gel that traps platelets and red blood cells in a thrombus on the surface within hours [9].
Improving Hemocompatibility of Biomedical Devices
In contrast to many polymers used in blood-contacting devices, the endothelial layer lining the human vasculature remains thrombus-free through several control mechanisms: a non-fouling phospholipid coating, membrane bound/released inhibitors of platelet and coagulation factors, as well as an efficient fibrinolytic system that removes fibrin deposits. Many of the major approaches for developing polymeric materials that are more hemocompatible are aimed at decreasing activation of key components of the coagulation cascade. Surface pacification is one approach, where the polymer surfaces aim to minimize protein adsorption (e.g., fibrinogen) and platelet adhesion/activation. Another approach is utilizing active polymer surfaces (e.g., immobilized heparin or nitric oxide (NO) release) that can interact with proteins, cells, and platelets to inhibit parts of the coagulation cascade. Research groups have worked to develop materials with suppressed blood-material interactions (e.g., polymeric materials which exhibit reduce protein and cell adhesion) and materials that mimic the non-thrombogenic endothelium. Polymers for blood-contacting devices are evaluated using various in vitro and in vivo testing methods, where protein and platelet adhesion are common markers to demonstrate the enhanced hemocompatibility properties of the material.
Overview of Current Technology/Strategies
Hydrophilic and Hydrophobic Materials
Protein adsorption on polymer surfaces is a well-recognized initiator of thrombus formation. Protein adsorption is dependent on a variety of material properties including surface charge, surface free energy, surface roughness, and a balance between hydrophobic and hydrophilic groups [10]. Ikada et al. [11] suggested that protein adhesion can be minimized when surface free energy is minimized, which occurs on strongly hydrophilic and strongly hydrophobic surfaces. One widely used approach has been to utilize hydrophilic surfaces in order to reduce protein adsorption and surface thrombogenicity. It has been concluded that hydrophilic polymers exert a steric repulsion effect towards blood proteins and cells [12, 13]. Hydrogels for blood contacting devices have been prepared using poly(vinyl alcohol) (PVA), polyacrylamides (PAAm), poly(N-vinyl pyrrolidone) (PNVP), poly(hydroxyethyl methacrylate) (PHEMA), poly(ethylene oxide) (PEO), poly(ethylene glycol) (PEG), poly(ethylene glycol) monomethyl ether (PEGME), and cellulose [14]. These hydrophilic materials strongly adsorb water, increasing its similarity to biological tissue and producing minimal interface tension with blood [15]. This strongly bound water also prevents cells and proteins from coming into contact with the polymer, reducing their adsorption to the surface [16].
Many hydrogels have poor mechanical properties, thus they have been grafted or coated onto other polymeric substrates and have improved hemocompatibility [15, 17–31]. Immobilization of PEG (–CH2CH2O–) is another popular means to make a polymer more protein and cell resistant [32]. Balakrishnan et al. demonstrated that PEGylated poly(viny chloride) had significantly less fibrinogen and platelet adsorption in comparison to a PVC control [21]. Li et al. reported that poly(poly(ethylene glycol) dimethacrylate) (P(PEGDMA)) grafted on silicone rubber reduced platelet adhesion by 90 % and also reduced plasma protein adsorption [26]. However, platelets still adsorbed on some PEG surfaces during in vivo experiments, despite the low protein adsorption observed during in vitro studies [33]. Plasma oxidation of polyethylene caused increased wettability with increased protein adsorption, but still reduced platelet adhesion [34]. Some hydrogels (e.g., cellulose) typically used as dialysis membranes/fibers are known to be thrombogenic, and therefore it can be concluded that low-protein adhesion does not necessarily result in hemocompatible surfaces [35]. Poly(2-methoxyethyl acrylate) (PMEA), which has been approved by the U.S. Food and Drug administration for medical use, not only exhibits lower protein adsorption and platelet adhesion than other hydrophilic surfaces, but it also reduces the amount of conformational changes that proteins undergo when adsorbed [16, 36]. This is of benefit since denaturation of adsorbed proteins can lead to platelet activation and subsequent thrombus formation, whereas, in contrast, when adsorbed proteins remain in their native confirmation, platelets cannot adhere (see discussion in Sect. “Albumin Coated Surfaces”).
Strongly hydrophobic polymers are also known to have good hemocompatibility properties. Some hydrophobic polymers that are commonly used for medical applications include polyurethanes, silicones, polytetrafluoroethylene (PTFE), poly(vinyl chloride) (PVC), and polyethylene [37]. Very smooth silicone rubber and polyurethanes are known to have good thromboresistant properties [38, 39]. Hydrophobic polymers are conducive to non-specific protein adsorption (e.g., fibrinogen, albumin), and the adsorbed albumin appears to prevent subsequent protein adsorption [40]. It has been shown that increasing surface hydrophobicity also increases the amount of protein adsorbed, which, in turn, decreases the amount of conformational changes, potentially having a role in pacifying the surface [41]. For example, Elast-eon polymers (polyurethane and polydimethylsiloxane copolymers) are reported to have low surface energy and stronger binding to albumin (over fibrinogen) [42]. Alkyl chains have also been grafted on relatively hydrophilic polymers in order to increase the hydrophobicity of the surface [43–45]. Khorasani et al. [46] reported that both superhydrophobic and superhydrophilic surfaces were able to reduce platelet adhesion in comparison to controls. However, materials that are hydrophobic exhibit high protein adsorption and conformational changes of the adsorbed proteins [47, 48]. Polymeric materials with both hydrophilic and hydrophobic domains have also been reported to improve hemocompatibility (see Sect. “Patterned Modifications of Surfaces”). Ultimately, there is still no consensus as to which is better, hydrophilic or hydrophobic surfaces, for blood-contacting biomedical device applications [49].
Albumin Coated Surfaces
It is a widely accepted fact that protein adsorption is the first event that occurs upon foreign surface-blood contact. Human albumin (Alb) is the most abundant protein in the body with a concentration of 35–53 mg/mL in blood plasma. Due to its high concentration and low molecular weight, it is the first protein that adsorbs on the surface of implanted materials. Unlike fibrinogen, albumin is not known to have a peptide sequence that can facilitate binding of the platelet receptors and hence has been used as a coating to block non-specific platelet-surface interactions.
Since the early finding that Alb coated surfaces prevent adhesion of proteins and platelets [50], Alb has been extensively used as one of the strategies to develop hemocompatible surfaces. In one study, Alb was shown to significantly improve short-term thrombogenicity of Dacron arterial prostheses [51]. In another study, Guidoin et al. showed that Alb treatment does not affect the strength of polyester arterial prosthesis, but also found that within 1–2 weeks of implantation the Alb coating begins to disappear [52]. In a comparison of carbon dioxide gas plasma-treated polystyrene (PS-CO2) coated Alb and PS-CO2 treated Alb-heparin conjugate, albumin treated surfaces were found to be more effective in reducing platelet adhesion [53]. Albumin-heparin conjugate surface was also found to be suitable for endothelial cell seeding, which can further improve the hemocompatibility of the surfaces. Mohammad et al. demonstrated that combining Alb with Immunoglobulin G (IgG) results in a significant reduction in platelet adhesion in a 7 day in vitro study [54].
One of the limitations of Alb coating is that other proteins can displace Alb on the surface and reduce the long-term effectiveness of this approach. To prevent this displacement, covalent immobilization of Alb has been reported [55]. Another challenge with the Alb coating approach is that it adsorbs to hydrophobic surfaces more tightly than the hydrophilic surfaces, which necessitates increasing the hydrophobicity of the surface, a modification that is considered undesirable because platelets also adhere strongly to hydrophobic surfaces [56, 57]. Some other limitations of Alb coatings include conformational changes, physical degradation, and challenges with sterilization and shelf stability. In a recent study by the Latour group, it was shown that non-activated platelets can adhere to adsorbed Alb once a critical degree of adsorption-induced unfolding is reached [58, 59]. The platelet response shows a strong correlation with the degree of adsorption induced unfolding, very similar to the platelet adhesion response to adsorbed fibrinogen. These studies demonstrate the potential challenges with Alb coated surfaces, in that, while they may pacify the surface initially, the adsorbed Alb will show a time-dependent conformational change potentially leading to increased platelet adhesion/activation.
Patterned Modifications of Surfaces
Surfaces with patterns at the micro- and nanoscale level have also been investigated for their hemocompatibility properties. Copolymers containing both hydrophilic and hydrophobic domains (ABA-type block copolymers) have exhibited good antithrombotic properties [60–66], where the balance between the hydrophilicity and hydrobphobicity is important to enhance the biocompatibility [67]. Surfaces with hydrophilic/hydrophobic microdomains are reported to create an organized protein structure, albumin adsorbing on hydrophilic domains and fibrinogen adsorbing on hydrophobic domains, which suppresses platelet adhesion/activation [64, 68]. Okano et al. reported that copolymers composed of hydrophilic monomers, 2-hydroxyethyl methacrylate (HEMA) or poly(2-hydroxyethyl methacrylate) (PHEMA), and hydrophobic styrene had excellent thromboresistance properties, in terms of preventing platelet adhesion and deformation, during in vitro experiments [61, 62]. The PHEMA-styrene copolymer was coated on vascular grafts and had an occlusion time of 20 days (vs. 2 days for controls) when implanted in rabbits [61]. However, some of these ABA-type copolymers have the disadvantage that the hydrophilic segments have high surface free energy and bury themselves in the hydrophobic segments [69]. Oyane et al. [60] has reported a new block copolymer (PS-PME3MA) where the hydrophilic blocks remain at the surface when in contact with water. The PS-PME3MA polymer was found to be highly resistant to protein adsorption, cell adhesion, and platelet adhesion/activation.
Another type of surface modification that has been shown to exhibit improved hemocompatibility is attachment of hydrophilic polymer brushes, such as PEO [70, 71] or PEG [72–75]. These hydrophilic polymer brushes create an antifouling surface on the substrate due to steric repulsion. Long-chain polymer brushes have been attached to surfaces using physical adsorption or covalent binding techniques [76]. Polymer brushes that are more densely packed (high graft density) and have longer chain lengths further reduce protein adhesion and platelet adhesion/activation on the surface (Fig. 16.3) [77–79]. Stents coated with a chitosan-PEO coating were tested in an ex vivo porcine model, and had low platelet adhesion (similar to the endothelium) [80]. Dialysis membranes grafted with PEG brushes also had reduced platelet adhesion and improved hemocompatibility [72, 81, 82]. Tetraethylene glycol dimethyl ether grafted on polyethylene tubing was able to significantly reduce plasma protein (both fibrinogen and von Willebrand’s factor) and platelet adsorption, in comparison to control tubing (PVC/Tygon, polyurethane, silicone, and polyethylene) [83]. Rodreguez-Emmenegger et al. recently reported an ultra-low fouling surface using poly[N-(2-hydroxypropyl) methacrylamide] (poly(HPMA)) brushes which was able to maintain the antifouling properties for up to two years of storage [84].
Attached Endothelial Cells
Another approach to making a surface more hemocompatible is to mimic the inner surface of blood vessels by attaching endothelial cells on the artificial surface, which provides an advantage in that the blood will come in contact with a surface that functions just like the endothelium. Endothelial cells (EC) line the inner walls of blood vessels and help in preventing thrombus formation by releasing anti-platelet agents such as nitric oxide (NO) and prostacyclin. Several research groups have used EC seeding on artificial surfaces for various biomedical applications including vascular grafts [85–87], stents [88], resorbable meshes [89], etc.
Williams and co-workers used a pressure sodding method to introduce EC cells into the luminal surface of PTFE grafts before implantation [86]. These grafts were tested for 12 weeks in carotid arteries of dogs. All of the control grafts clotted, whereas 86 % of the EC sodded grafts were found to be patent after the 12 week implantation period. To improve the hemocompatibility of vascular grafts, Taite et al. used NO releasing PU-PEG copolymer containing cell adhesive pentapeptide sequence to promote EC cell adhesion and migration [90]. In another study, Kutryk et al. [88] used xenotransplantation to seed endothelial cells on the surface of the stents to reduce blood contact with the stent surface. Zünd and co-workers, seeded fibroblasts and ECs on polyglycolic acid (PGA) resorbable mesh as a precursor to vessels or cardiac valves [89].
Despite these encouraging results, EC proliferation and seeding on artificial surfaces is complex and still has challenges to overcome. One of the challenges of using the EC seeding approach is the difficulty in growing and maintaining the cells on artificial surfaces [91]. Various studies have been conducted to modify the surfaces in such a way that they can improve and promote endothelial cell adhesion. Kawamoto et al. showed that by plasma treatment of a PU surface, EC adhesion and proliferation can be dramatically improved [92]. In another study, Li and co-workers showed that increased proliferation and cell spreading can be achieved with arg-gly-asp (RGD) peptide grafted surfaces [93]. In yet another study, Yin et al. reported that mussel adhesive polypeptide mimic, containing dihydroxyphenylalanine and L-lysine (MAPDL) with PEG spacer, improved the cell attachment and growth and also reduced platelet adhesion in comparison to the controls [94]. Other challenges associated with the EC seeding approach include complex and expensive experimental procedures and long-term stability issues which still need to be addressed.
Zwitterionic Surfaces
Another effective approach to obtain a hemocompatible surface includes introducing a zwitterionic group, such as phosphobetaine, sulfobetaine or carboxylbetaine, to the substrate material (Fig. 16.4). These zwitterionic materials have been considered as biomimetic, antifouling, and hemocompatible materials because they contain phosphorylcholine-like groups which are present on the lipid bilayers of the cell membranes [95, 96]. Zwitterionic betaines have both anionic and cationic charged moieties in the same side chain which maintains an overall neutral charge. As discussed earlier, PEG and other hydrophilic materials, such as PEO and PHEMA, become hydrated via hydrogen bonding with water to prevent fouling. Zwitterionic materials can bind water molecules even more strongly (induced by electrostatic interactions) and hence can prevent significant change in protein conformation [97, 98].
There have been numerous publications showing the potential of zwitterion-based surfaces in resisting protein adsorption [95, 99], platelet adhesion [100–103], and consequently reduced thrombus formation compared to the control materials. Atom transfer radical polymerization (ATRP) is one of the commonly used methods to graft polymer surfaces with zwitterionic groups [97, 104–106]. Recently, various research teams have modified polymers using zwitterionic groups (carboxybetaine and sulfobetaine monomers) to mimic cell membrane surfaces [100, 102, 107, 108]. These surfaces have shown significantly lower fouling and platelet adhesion properties. In an another study, a zwitterionic silane coupling agent, N,N-dimethyl, N-(2-ethyl phosphate ethyl)-aminopropyltrimethyoxysilane (DMPAMS) was synthesized and studied for in vitro hemocompatibility [96]. Surfaces grafted with DMPAMS showed a steep drop in the water contact angle from 48º to 21.5º, indicating a more hydrophilic surface. At 200 mM surface concentration of DMPAMS, activated partial thromboplastin time (APTT) was prolonged from 28.8 s for the control to 37.8 s for the modified surface. Almost no platelet attachment was seen on the DMPAMS modified surface even after 3 h of blood contact. Lee et al. combined PEG with zwitterions for stent coating applications [109]. Significant reduction in protein adsorption and platelet adhesion was observed in addition to a substantial increase (compared to controls) in blood coagulation time for the zwitterionic-PEG grafted stents [109]. Polyurethane catheters grafted with a zwitterionic sulfobetain monomer (N,N-dimethyl-N-methacryloxyethyl-N-(3-sulfopropyl) ammonium, DMMSA) were also able to prevent platelet adhesion when soaked in platelet rich plasma (PRP) for 120 min [110]. Stents coated with phosphorylcholine, such as the commercial Biodiv Ysio ™, have also been shown to exhibit enhanced hemocompatibility and increased endothelialization [111, 112].
Heparin Immobilization
Heparin (Fig. 16.5) is a highly sulfonated, anionic blood polysaccharide that binds to antithrombin III through ionic interactions, which inactivates thrombin and Factor Xa and thereby inhibits blood coagulation. Heparin has been clinically used since 1935 and is one of the most common anticoagulants employed during surgery and treatment of post-operative thrombosis and embolism [113]. However, systemic administration of anticoagulants, such as heparin, increases the risks of hemorrhage, thrombocytopenia, and thrombosis [7]. It is well known that some heparinized and sulfonated materials have anticoagulant activity, resulting in prolongation of the blood clotting time [114]. Heparin immobilized on surfaces also suppresses platelet adhesion and protein adsorption [115]. Heparinization of surfaces is one of the most popular techniques to improve hemocompatibility and has been commercialized and used in the preparation of different medical devices (e.g., catheters, extracorporeal circuits, stents, grafts, etc.) [116, 117].
Heparin has been physically adsorbed, as well as ionically and covalently immobilized, on various polymer surfaces [117–122]. Ionic immobilization techniques require cationic residual groups in the polymer which interact with the ionic groups on heparin (e.g., COO−, SO 2−,4 NHSO3 −), while covalent binding of heparin utilizes the hydroxyl, carboxyl, or amino groups of heparin [120]. Heparin that is ionically bound to a surface will slowly be released due to ion-exchange with blood components, and this ultimately shortens the usage lifetime of the material [123]. The success of heparinized materials depends on the covalently bound heparin remaining in its native confirmation and its ability to complex with antithrombin III, which is most efficient when heparin is coupled by endpoint (vs. multipoint) attachment [124]. Michanetzis et al. [120] compared common heparin immobilization techniques, direct [125] and indirect [126] covalent binding, applied to the surfaces of commercially available polymers (silicone rubber, polyethylene, polypropylene, and poly(viny chloride)). While both techniques suffered from a low yield of immobilized heparin in comparison to the initial amount of heparin, the direct method produced a better heparinization yield (10.5 %) and both methods were able to improve the hemocompatibility in terms of reduced platelet activation and, therefore, an increased platelet retention rate. The anticoagulation properties of immobilized heparin have also been improved by using hydrophilic spacers (e.g., PEG), in comparison to heparin immobilized directly to the polymer. This reduces protein adsorption [127, 128]. Polyethylene tubing was modified with immobilized heparin via the method developed by Larm et al. [119] (commercialized as Carmeda ® BioActive Surface) and was able to maintain efficacy for up to 4 months when implanted in pigs [129]. The advantage of heparinized surfaces, in addition to the binding of antithrombin III, is the reduced/selective adhesion of certain plasma proteins, which alters the composition of the surface-adsorbed layer of proteins [130–133]. Heparin-coated devices, however, also suffer challenges, especially for long-term applications, due to heparin’s short half-life and that surface-bound heparin only has ~1 % of the activity of free heparin [134]. Other immobilized direct thrombin inhibitors, such as hirudin, have demonstrated thromboresistant properties as well [116, 135].
Nitric Oxide (NO) Releasing/Generating Polymers
Nitric oxide (NO) is known to be a potent inhibitor of platelet activation and adhesion. Healthy endothelial cells exhibit an NO flux of 0.5−4.0 × 10−10 mol cm−2 min−1 [136]. Polymeric materials with an NO flux that is equivalent to this level are expected to have similar anti-thrombotic properties. Nitric oxide is highly reactive under physiological conditions, thus a wide range of NO donor molecules, with functional groups that can store and release NO, have been studied for potential biomedical applications. Various reviews have been published that are devoted to a comprehensive discussion of different NO releasing/generating materials and their many potential biomedical applications [137–144]. The two major approaches to achieving localized NO release from polymeric surfaces are to (1) incorporate NO donor molecules or functional groups (e.g. diazeniumdiolates or S-nitrosothiols) into the polymer that will release the bound NO under physiological conditions (NO releasing polymers (NOrel)), or (2) incorporate catalysts into the polymers which can react with endogenous S-nitrosothiol (RSNO) species (present in blood) to locally generate NO (NO generating polymers (NOgen)) (Fig. 16.6). Nitric oxide has many other biological roles, including its antimicrobial action, which could potentially be utilized to also reduce the infection and biofilm formation that plagues many implantable biomedical devices. Thus, NO releasing/generating chemistries have the potential to create a dual-functioning material that is both antithrombotic and antimicrobial. Duration of the NO release can be tailored to fit the specific biomedical application of the device, and NO-generating materials can continuously generate NO in the presence of endogenous RSNO species (e.g., S-nitrosoglutathione (GSNO), S-nitrosocysteine (CysNO), etc.).
Types of NO-releasing/generating polymers, where the NO that is released/generated can prevent activation of platelets that approach the polymer surface. Diazeniumdiolate-based materials undergo proton and thermal driven mechanisms to release NO. S-nitrosothiol (RSNO)-based polymers can release NO in the presence of heat, light, or metal ions (e.g., Cu+). NO-generating materials consist of immobilized catalysts (e.g., Cu or Se compounds) that generate NO from endogenous RSNOs
A wide variety of NO donor molecules have been investigated. The two most widely studied and used in combination with biomaterials are diazeniumdiolates and S-nitrosothiols. Diazeniumdiolates, also called NONOates, are relatively stable species that undergo a proton or thermally driven mechanism to release two molecules of NO per diazeniumdiolate molecule. Diazeniumdiolates have been a popular NO donor that can easily be dispersed within polymers to facilitate localized NO release [145–149]. Mowery et al. [145] reported that water-soluble diamine-based diazeniumdiolates (e.g., DMHD/N2O2, diazeniumdiolated linear polyethylenimine (LPEI/N2O2)) leached from polymer matrices. Significant leaching of the NO donor species can result in non-localized NO release where the therapeutic action of NO occurs downstream from the biomedical device. Another concern with diazeniumdiolated-based polymers is the formation and leaching of some potentially carcinogenic decomposition products (e.g., N-nitrosamines) that are not intended for release into the bloodstream [145, 150]. To overcome the leaching concerns, strategies to covalently bind diazeniumdiolated functional groups to polydimethylsiloxane (PDMS) [151], xerogels [152–154], medical-grade polyurethanes [137, 155, 156], silica nanoparticles [157, 158], dendrimers [159–161], and other nanomaterials [138] have also been reported. Zhang et al. [151] covalently linked diaminoalkyltrimethoxysilane (DACA) to polydimethylsiloxane and then loaded this materials with NO under high pressure to form the diazeniumdiolated coating, DACA/N2O2-SR, which released NO for up to 20 days. The DACA/N2O2–SR was coated on extracorporeal circuits (ECC) and was able to reduce platelet consumption and thrombus formation during the 4 h blood flow in a rabbit model.
Attempts have also been made to add polymer top coats and/or create more lipophilic diazeniumdiolated species to minimize leaching into the aqueous phase and therefore maintain the localized NO release at the blood-polymer interface [147, 150, 162, 163]. Poly(vinyl chloride) and polyurethane have been doped with the lipophilic diazeniumdiolated dibutylhexanediamine (DBHD/N2O2) [147, 148, 164, 165]. Vascular grafts coated with DBHD/N2O2–doped polyurethane had significantly less thrombus formation than controls after 21 days implantation in sheep [148]. However, the loss of NO from DBHD/N2O2 creates free lipophilic amine species within the polymer that react with water, thereby increasing the pH within the polymer phase and effectively turning off the NO release. A recent report demonstrated that NO release can be prolonged, by using poly(lactic-co-glycolic acid) (PLGA) additives, for up to 14 days from poly(vinyl chloride) (PVC) doped with diazeniumdiolated dibutylhexanediamine (DBHD/N2O2) (Fig. 16.7) [165]. The ester linkages of the PLGA hydrolyze in the presence of water, producing lactic and glycolic acids that can act as proton sources to control the NO release from DBHD/N2O2-doped polymers. PLGAs can have varying hydrolysis rates, which is primarily determined by the copolymer ratio, the end group chemistry (either a free carboxylic acid or ester end group), and molecular weight. Handa et al. also demonstrated the importance of the inherent hemocompatibility of the base polymer (in which the NO chemistry is incorporated) [166]. By incorporating the same DBHD/N2O2 and PLGA chemistry into the Elast-eon E2As polymer (a copolymer with a mixed soft segment of poly(dimethylsiloxane) and poly(hexamethylene oxide) with a methylene diphenyl isocyante (MDI) hard segment), which inherently is more hemocompatible than PVC, the platelet count after 4 h of ECC (97 ± 10 % of baseline) was significantly improved over the PVC/DOS-based coating (79 ± 11 % of baseline) [165, 166].
Another widely studied class of NO donor molecules is S-nitrosothiol (RSNO) species (examples shown in Fig. 16.8). Physiological RSNOs, including S-nitrosoalbumin, S-nitrosocysteine (CysNO), and S-nitrosoglutathione (GSNO), are considered an endogenous reservoir of NO in vivo [167–169]. Other synthetic RSNOs, such as S-nitroso-N-acetylpenicillamine (SNAP) and S-nitroso-N-acetylcysteine (SNAC), have been shown to exhibit significant antimicrobial and antithrombotic effects [170–173]. It has also been demonstrated that RSNOs are both vasodilators and potent inhibitors of platelet aggregation [174–176]. RSNOs undergo thermal decomposition to release NO and produce a corresponding disulfide species (RSSR). The NO release from RSNOs can also be catalyzed by metal ions (e.g., Cu+) [177] and by light, through irradiation at energies that correspond to the S-nitroso absorption bands at 340 and/or 590 nm [178–180]. Incorporation of RSNOs into polymers can extend the utility of these NO donors to be applicable in biomedical devices.
Low molecular weight RSNOs have been non-covalently dispersed within various polymer matrices [181–187]. Seabra et al. [182] prepared GSNO-doped poly(vinyl alcohol) and poly(vinyl pyrrolidone) blended films. This polymer matrix provided a stabilization effect on GSNO decomposition, in comparison to aqueous solutions of GSNO. However, due to leaching, 90 % of the NO was released during the first 24 h under physiological conditions. SNAP-doped polyurethanes (Tecoflex SG-80A and Tecophillic SP-60D-60) also exhibit rapid leaching of SNAP when soaked in buffer [186]. The rapid leaching of the RSNOs significantly shortened the duration of the NO release from these materials. Therefore, strategies to synthetically bind RSNO functionalities to the polymer backbone have also been explored. S-Nitrosothiol functionalities have been covalently bound to polymers such as xerogels [188, 189], polyurethanes [137, 190, 191], degradable polyesters [192–196], polyester/poly(methyl methacrylate) blends [197], poly(vinyl methyl ether-co-malic anhydride) (PVMMA) and poly(vinyl pyrrolidone) (PVP) [198], and poly(dimethylsiloxane) [199]. Dendrimers [200], fumed silica particles [180, 191], and silica nanoparticles [155, 156, 201] have also been synthetically modified with covalently bound RSNO functionalities and these materials can be doped into various polymer matrices to create coatings for biomedical devices.
Many of the RSNO-modified materials reported to date have not proven clinically useful due to their limited NO release lifetimes, low conversion to RSNO during synthesis, or lack of RSNO stability during storage. Recently, the RSNO-doping method has been revisited, utilizing more hydrophobic polymers. GSNO-doped Tygon, a proprietary plasticized poly(vinyl chloride) polymer, exhibited minimal leaching of GSNO during soaking under physiological conditions [187]. Incorporating SNAP into Elast-eon E2As created an NO-releasing polymer that is stable during storage (even at elevated temperatures), locally delivers NO under physiological conditions, significantly reduces the leaching of SNAP, and preserves platelets during a 4 h ECC study [186].
Due to the limited NO reservoir from NO donors that can be incorporated into polymers (either covalently bound or non-covalently dispersed), these NO releasing materials typically have a finite duration of NO release. Another approach to achieving localized NO delivery at a polymer/blood interface for a longer period of time is to use NO generating (NOgen) materials that utilize endogenous RSNOs and/or nitrite to locally generate NO at the blood-polymer interface. Various thiol-containing species (L-cysteine, 2-iminothiolane, and cysteine polypeptide) have been immobilized on polyethylene terephthalate and polyurethane, where the free thiol undergoes a transnitrosation reaction with circulating RSNOs (e.g., S-nitroso-albumin) [202, 203]. The resulting CysNO on the PET and PU surface decomposes to elevate the NO levels locally at the polymer/blood interface, which reduces platelet adhesion on the surface by more than 50 %. Other NOgen materials consist of catalysts (e.g., Cu (I/II) complexes or organoselenium species) that are immobilized within the polymer and are capable of locally generating NO from endogenous RSNOs and/or nitrite. Copper (II) sites within a polymer can be reduced to Cu(I) by endogenous reducing agents that are present in the blood (e.g., thiols, ascorbate). The Cu(I) sites then can reduce endogenous RSNOs in the blood (e.g., GSNO, CysNO, etc.) to NO and free thiolate anions in a catalytic manner [177]. Lipophilic copper complexes that were incorporated into polymers were able to reduce physiological levels of RSNOs and nitrite to locally generate NO [204–207]. Another reported NOgen polymer consists of copper nanoparticles (Cu0) dispersed within a hydrophilic polymer (Tecophillic SP-60D-60), which was evaluated using a rabbit model for extracorporeal circulation (ECC) [208]. However, continuous infusion of SNAP was needed to supplement the endogenous RSNO levels in order to achieve good efficacy in reducing thrombus formation.
Organoselenium species have been studied as mimics of glutathione peroxidase (GPx), a selenoenzyme that protects cells from oxidative stress by reducing hydroperoxides using glutathione (GSH) [209], and investigated for their ability to generate NO from RSNOs [210]. Selenium catalysts are highly selective for reduction of RSNOs and exhibit no catalytic activity for nitrite or nitrate reduction [211]. Low molecular weight organoselenum species, selenocystamine (SeCA) and 3,3′-diselenidedipropionic acid (SeDPA), were immobilized on cellulose filter paper and polyethylenimine (PEI), demonstrating potential dialysis membrane applications [212]. In the presence of reducing agents at physiological pH, the diselenides can be converted into selenolates, which are the key intermediates that can reduce RSNOs into NO and thiolates [212]. Selenium species were also incorporated into layer-by-layer coatings containing alternating layers of sodium alginate (Alg) and selenium-modified PEI. Alg-PEI layer-by-layer films modified with SeDPA were able to generate physiological levels of NO from GSNO and also exhibited minimal leaching of catalytic sites after soaking in blood [211]. The Alg-PEI layer-by-layer coating was also modified to immobilize ebselen (2-phenyl-1,2-benzisoselenazol-3-(2H)-one), an aromatic selenium species with good GPx activity, and coated on catheters [213]. These selenium-based materials were able to generate physiological levels of NO from RSNOs; however, some reduction of NO flux was observed after soaking in blood, potentially from catalytic sites being blocked by adsorbed plasma proteins or low levels of catalyst species leaching into the solution. These NOgen polymers have an advantage in that they could potentially generate NO at the blood-polymer interface indefinitely, provided that the blood in contact with the polymer has adequate levels of RSNOs present at all times [214].
Methods to Assess Hemocompatibility
Extensive reviews on the various methods to evaluate the hemocompatibility of polymers and medical devices have been published (summarized in Table 16.1) [1, 215–223]. Clinical devices still suffer challenges due to thrombosis, even after standard testing as developed by the International Organization for Standardization (ISO 10993-4, “Selection of Tests for Interactions with Blood”) [219, 224]. The ISO hemocompatibility testing requires evaluation of thrombosis, coagulation, hematology, platelet count and function, and immunology. Initial surface and physical-chemical characterization studies can provide useful information in order to correlate surface characteristics with hemocompatibility. Surface roughness, surface chemistry, surface charge distribution, and interfacial free energy are well known factors that can influence protein adsorption and cell-material surface interaction [225]. Many of the common hemocompatibility testing methods involve flowing blood through tubing (both in vitro and in vivo), and then conducting hemocompatibility evaluations of the blood exiting the test system. However, while they have potential to still be used as preliminary screening methods, in vitro static assays, such as fluorescent-based fibrinogen adsorption or platelet adhesion [164, 226], and dynamic systems (e.g. Chandler loops [227] ) are limited due to the blood death over time. Various medical devices (e.g., catheters, stents, vascular grafts, dialysis membranes, etc.) have been fabricated or coated with polymers for evaluation in animal models in order to mimic clinical application. Arterio-venous (A-V) and areterio-arterial (A-A) shunt models have been used to test hemocompatibility, and they allow for evaluation of both local and systemic effects of the test polymer. For example, the rabbit model of thrombogenicity is a useful model of extracorporeal circulation, where the polymer is coated on the inner walls of Tygon tubing that form the ECC circuit [164, 208]. The ECC circuit is placed on the rabbit using an A-V shunt, and blood flows through the circuit for 4 h. This model allows a comprehensive evaluation of hemocompatibility (platelet preservation/consumption, plasma fibrinogen levels, and occlusion time) as well as the thrombus area in the ECC circuit after 4 h of blood flow.
Despite the encouraging results published for many of the polymer strategies discussed above, there are still some challenges with hemocompatibility testing and clinical application. For example, counting adhered platelets on the surface is a common method to evaluate hemocompatibility and has been used as a functional test for many of the polymers discussed above. This may be a good initial test; however, it does not provide the complete picture because platelets could be activated by the foreign surface and aggregate downstream from the device [223]. Similarly, during in vivo testing of a new material, any thrombus that forms on the surface may also be carried downstream. In addition, results from short-term in vitro testing cannot necessarily be used to predict long-term in vivo results [220]. Another challenge is the fact that research groups use a wide variety of testing methods and controls, so it remains unclear as to how significant the improvement is of these new materials over other materials (either published or clinically used) [220]. Defining standardized testing methods and appropriate controls/reference materials used by researchers (commercial, clinical, and academic) will help improve the advancement of novel materials to clinical application.
Conclusions
Over the last 50 years, much has been learned about blood-material interactions, and a variety of strategies have been reported to improve the hemocompatibility of blood-contacting medical devices. Materials reported to date primarily target different parts of the coagulation cascade (e.g., protein adsorption, platelet adhesion and activation, fibrin formation, etc.). Although many of the approaches described above are promising individually, combining two or more of these approaches may prove most beneficial. When these materials are effective, they may reduce or eliminate the need for administration of systemic anti-coagulation therapeutics (e.g. heparin) which is currently clinically utilized to reduce the risk of thrombus formation and other complications during use of blood-contacting devices. However, challenges still exist (e.g., shelf life and sterilization stability) and need to be addressed for some of the polymer approaches discussed here in order to achieve widespread clinical application. Standard testing methods should be utilized to test and compare the hemocompatibility of commercially available and new polymeric materials for biomedical applications.
Abbreviations
- A-A:
-
Arterio-arterlial
- A-V:
-
Arterio-venous
- Alb:
-
Albumin
- Alg:
-
Sodium alginate
- APTT:
-
Activated partial thromboplastin time
- ATRP:
-
Atom transfer radical polymerization
- CysNO:
-
S-nitrosocysteine
- DACA:
-
Diaminoalkyltrimethoxysilane
- DBHD/N2O2 :
-
Diazeniumdiolated dibutylhexanediamine
- DBHD:
-
N,N′-dibutyl-1,6-hexanediamine
- DMHD/N2O2 :
-
Diazeniumdiolated N,N′-dimethyl-1,6-hexanediamine
- DMMSA:
-
N,N-dimethyl-N-methacryloxyethyl-N-(3-sulfopropyl) ammonium
- DMPAMS:
-
N,N-dimethyl, N-(2-ethyl phosphate ethyl)-aminopropyltrimethyoxysilane
- DOS:
-
Dioctyl sebacate
- EC:
-
Endothelial cells
- ECC:
-
Extracorporeal circulation
- GPx:
-
Glutathione peroxidase
- GSH:
-
Glutathione
- GSNO:
-
S-nitrosoglutathione
- HEMA:
-
2-hydroxyethyl methacrylate
- IgG:
-
Immunoglobulin
- LPEI/N2O2 :
-
Diazeniumdiolated linear polyethylenimine
- MDI:
-
Methylene diphenyl isocyanate
- NO:
-
Nitric oxide
- NOgen:
-
NO-generating
- NONOate:
-
Diazeniumdiolate
- NOrel:
-
NO-releasing
- P(PEGDMA):
-
Poly(poly(ethylene glycol) dimethacrylate)
- PAAm:
-
Polyacrylamides
- PDMS:
-
Poly(dimethylsiloxane)
- PEG:
-
Poly(ethylene glycol)
- PEI:
-
Polyethylenimine
- PEGME:
-
Poly(ethylene glycol) monomethyl ether
- PEO:
-
Poly(ethylene oxide)
- PGA:
-
Polyglycolic acid
- PHEMA:
-
Poly(hydroxyethyl methacrylate)
- PLGA:
-
Poly(lactic-co-glycolic acid)
- PMEA:
-
Poly(2-methoxyethyl acrylate)
- PNVP:
-
Poly(N-vinyl pyrrolidone)
- PRP:
-
Platelet-rich plasma
- PS-CO2 :
-
Carbon dioxide as plasma-treated polystyrene
- PTFE:
-
Polytetrafluoroethylene
- PVA:
-
Poly(vinyl alcohol)
- PVC:
-
Poly(vinyl chloride)
- PVMMA:
-
Poly(vinyl methyl ether-co-malic anhydride)
- PVP:
-
Poly(vinyl pyrrolidone)
- RSNO:
-
S-nitrosothiol
- RSSR:
-
Disulfide
- SeCA:
-
Selenocystamine
- SeDPA:
-
3,3′-diselenidedipropionic acid
- SNAC:
-
S-nitroso-N-acetyl-L-cysteine
- SNAP:
-
S-nitroso-N-acetylpenicillamine
- SR:
-
Silicone rubber
- vWF:
-
von Willebrand Factor
References
Ratner, B.D.: The catastrophe revisited: blood compatibility in the 21st century. Biomaterials 28(34), 5144–5147 (2007)
Colman, R.W.: Mechanisms of thrombus formation and dissolution. Cardiovasc. Pathol. 2(3), 23–31 (1993)
Gorbet, M.B., Sefton, M.V.: Biomaterial-associated thrombosis: roles of coagulation factors, complement, platelets and leukocytes. Biomaterials 25(26), 5681–5703 (2004)
Dwyer, A.: Surface-treated catheters—a review. In: Seminars in dialysis, pp 542–546. Wiley Online Library (2008)
Ratner, B.D.: Blood compatibility—a perspective. J. Biomater. Sci. Polym. Ed. 11(11), 1107–1119 (2000)
Gaffney, A.M., Wildhirt, S.M., Griffin, M.J., Annich, G.M., Radomski, M.W.: Extracorporeal life support. BMJ 341 (2010)
Robinson, T.M., Kickler, T.S., Walker, L.K., Ness, P., Bell, W.: Effect of extracorporeal membrane oxygenation on platelets in newborns. Crit. Care Med. 21(7), 1029–1034 (1993)
Horbett, T.A.: Principles underlying the role of adsorbed plasma proteins in blood interactions with foreign materials. Cardiovasc. Pathol. 2(3), 137–148 (1993)
Schopka, S., Schmid, T., Schmid, C., Lehle, K.: Current strategies in cardiovascular biomaterial functionalization. Materials 3(1), 638–655 (2010)
Brashm, J., Horbett, T.: Proteins at interfaces: physicochemical and biochemical studies. In: ACS Symposium Series, 1987, p 1. American Chemical Society, Washington, DC
Ikada, Y., Suzuki, M., Tamada, Y.: Polymer surfaces possessing minimal interaction with blood components. In: Polymers as biomaterials, pp 135–147. Springer, Heidelberg (1984)
Nagaoka, S., Mori, Y., Takiuchi, H., Yokota, K., Tanzawa, H., Nishiumi, S.: Interaction between blood components and hydrogels with poly (oxyethylene) chains. In: Polymers as biomaterials, pp 361–374. Springer, Heidelberg (1984)
Jeon, S., Lee, J., Andrade, J., De Gennes, P.: Protein—surface interactions in the presence of polyethylene oxide: I. Simplified Theo. J Colloid Interface Sci 142(1), 149–158 (1991)
Peppas, N.A., Huang, Y., Torres-Lugo, M., Ward, J.H., Zhang, J.: Physicochemical, foundations and structural design of hydrogels in medicine and biology. Annu. Rev. Biomed. Eng. 2, 9–29 (2000)
Okada, T., Ikada, Y.: Modification of silicone surface by graft polymerization of acrylamide with corona discharge. Makromol. Chem. 192(8), 1705–1713 (1991)
Tanaka, M., Hayashi, T., Morita, S.: The roles of water molecules at the biointerface of medical polymers. Polym. J. 45(7), 701–710 (2013)
Wang, H., Yu, T., Zhao, C., Du, Q.: Improvement of hydrophilicity and blood compatibility on polyethersulfone membrane by adding polyvinylpyrrolidone. Fibers Polym. 10(1), 1–5 (2009)
Hanson, S.R., Harker, L.A., Ratner, B.D., Hoffman, A.: In vivo evaluation of artificial surfaces with a nonhuman primate model of arterial thrombosis. J. Lab. Clin. Med. 95(2), 289–304 (1980)
Morra, M., Occhiello, E., Garbassi, F.: Surface modification of blood contacting polymers by poly (ethyleneoxide). Clin. Mater. 14(3), 255–265 (1993)
Kwon, O.H., Nho, Y.C., Park, K.D., Kim, Y.H.: Graft copolymerization of polyethylene glycol methacrylate onto polyethylene film and its blood compatibility. J. Appl. Polym. Sci. 71(4), 631–641 (1999)
Balakrishnan, B., Kumar, D., Yoshida, Y., Jayakrishnan, A.: Chemical modification of poly (vinyl chloride) resin using poly (ethylene glycol) to improve blood compatibility. Biomaterials 26(17), 3495–3502 (2005)
Amiji, M.M.: Synthesis of anionic poly(ethylene glycol) derivative for chitosan surface modification in blood-contacting applications. Carbohydr. Polym. 32(3–4), 193–199 (1997)
Wang, J., Pan, C.J., Huang, N., Sun, H., Yang, P., Leng, Y.X., Chen, J.Y., Wan, G.J., Chu, P.K.: Surface characterization and blood compatibility of poly(ethylene terephthalate) modified by plasma surface grafting. Surf. Coat. Technol. 196(1–3), 307–311 (2005)
Okner, R., Domb, A.J., Mandler, D.: Electrochemically deposited poly(ethylene glycol)-based sol–gel thin films on stainless steel stents. New J. Chem. 33(7), 1596–1604 (2009)
Nho, Y.C., Kwon, O.H.: Blood compatibility of AAc, HEMA, and PEGMA-grafted cellulose film. Radiat. Phys. Chem. 66(4), 299–307 (2003)
Li, M., Neoh, K.G., Xu, L.Q., Wang, R., Kang, E.T., Lau, T., Olszyna, D.P., Chiong, E.: Surface modification of silicone for biomedical applications requiring long-term antibacterial, antifouling, and hemocompatible properties. Langmuir 28(47), 16408–16422 (2012)
Jin, J., Jiang, W., Yin, J.H., Ji, X.L., Stagnaro, P.: Plasma proteins adsorption mechanism on polyethylene-grafted poly(ethylene glycol) surface by quartz crystal microbalance with dissipation. Langmuir 29(22), 6624–6633 (2013)
Chung, C.W., Kim, H.W., Kim, Y.B., Rhee, Y.H.: Poly(ethylene glycol)-grafted poly(3-hydroxyundecenoate) networks for enhanced blood compatibility. Int. J. Biol. Macromol. 32(1–2), 17–22 (2003)
Chiag, Y.C., Chang, Y., Chen, W.Y., Ruaan, R.C.: Biofouling resistance of ultrafiltration membranes controlled by surface self-assembled coating with PEGylated copolymers. Langmuir 28(2), 1399–1407 (2012)
Bajpai, A.K.: Blood protein adsorption onto a polymeric biomaterial of polyethylene glycol and poly[(2-hydroxyethyl methacrylate)-co-acrylonitrile] and evaluation of in vitro blood compatibility. Polym. Int. 54(2), 304–315 (2005)
Alibeik, S., Zhu, S., Brash, J.L.: Surface modification with PEG and hirudin for protein resistance and thrombin neutralization in blood contact. Colloids Surf. B 81(2), 389–396 (2010)
Harris, J.M.: Poly (ethylene glycol) chemistry: biotechnical and biomedical applications. Springer, Heidelberg (1992)
Llanos, G.R., Sefton, M.V.: Immobilization of poly (ethylene glycol) onto a poly (vinyl alcohol) hydrogel: 2. Evaluation of thrombogenicity. J. Biomed. Mater. Res. 27(11), 1383–1391 (1993)
Lee, J.H., Lee, H.B.: Platelet adhesion onto wettability gradient surfaces in the absence and presence of plasma proteins. J. Biomed. Mater. Res. 41(2), 304–311 (1998)
Sefton, M., Gemmell, C., Gorbet, M. Nonthrombogenic treatments and strategies. Biomaterials Science: An Introduction to Materials in Medicine, pp. 456–470. Elsevier, New York (2004)
Tanaka, M., Motomura, T., Kawada, M., Anzai, T., Yuu, K., Shiroya, T., Shimura, K., Onishi, M., Akira, M.: Blood compatible aspects of poly(2-methoxyethylacrylate) (PMEA)—relationship between protein adsorption and platelet adhesion on PMEA surface. Biomaterials 21(14), 1471–1481 (2000)
Nydegger, U., Rieben, R., Lämmle, B.: Biocompatibility in transfusion medicine. Transfus. Sci. 17(4), 481–488 (1996)
Kolobow, T., Stool, E., Weathersby, P., Pierce, J., Hayano, F., Suaudeau, J.: Superior blood compatibility of silicone rubber free of silica filler in the membrane lung. ASAIO Trans. 20, 269 (1974)
Szycher, M.: Biostability of polyurethane elastomers: a critical review. J. Biomater. Appl. 3(2), 297–402 (1988)
Lassen, B., Malmsten, M.: Structure of protein layers during competitive adsorption. J. Colloid Interface Sci. 180(2), 339–349 (1996)
Sivaraman, B., Fears, K.P., Latour, R.A.: Investigation of the effects of surface chemistry and solution concentration on the conformation of adsorbed proteins using an improved circular dichroism method. Langmuir 25(5), 3050–3056 (2009)
Cozzens, D., Luk, A., Ojha, U., Ruths, M., Faust, R.: Surface characterization and protein interactions of segmented polyisobutylene-based thermoplastic polyurethanes. Langmuir 27(23), 14160–14168 (2011)
Bahulekar, R., Tamura, N., Ito, S., Kodama, M.: Platelet adhesion and complement activation studies on poly (N-alkyl mono and disubstituted) acrylamide derivatives. Biomaterials 20(4), 357–362 (1999)
Matsuda, T., Ito, S.: Surface coating of hydrophilic-hydrophobic block co-polymers on a poly (acrylonitrile) haemodialyser reduces platelet adhesion and its transmembrane stimulation. Biomaterials 15(6), 417–422 (1994)
Hoffman, A.S., Cohn, D., Hanson, S.R., Harker, L.A., Horbett, T.A., Ratner, B.D., Reynolds, L.O.: Application of radiation-grafted hydrogels as blood-contacting biomaterials. Radiat. Phys. Chem. 22(1), 267–283 (1983)
Khorasani, M., Mirzadeh, H.: In vitro blood compatibility of modified PDMS surfaces as superhydrophobic and superhydrophilic materials. J. Appl. Polym. Sci. 91(3), 2042–2047 (2004)
Okubo, M., Hattori, H.: Competitive adsorption of fibrinogen and albumin onto polymer microspheres having hydrophilic/hydrophobic heterogeneous surface structures. Colloid Polym. Sci. 271(12), 1157–1164 (1993)
Klose, T., Welzel, P.B., Werner, C.: Protein adsorption from flowing solutions on pure and maleic acid copolymer modified glass particles. Colloids Surf., B 51(1), 1–9 (2006)
Sefton, M.V., Gemmell, C.H., Gorbet, M.B.: What really is blood compatibility? J. Biomater. Sci. Polym. Ed. 11(11), 1165–1182 (2000)
Andrade, J., Hlady, V.: Protein adsorption and materials biocompatibility: a tutorial review and suggested hypotheses. In: Biopolymers/Non-Exclusion HPLC, pp 1–63. Springer, Heidelberg (1986)
Kottke-Marchant, K., Anderson, J.M., Umemura, Y., Marchant, R.E.: Effect of albumin coating on the in vitro blood compatibility of Dacron® arterial prostheses. Biomaterials 10(3), 147–155 (1989)
Guidoin, R., Snyder, R., Martin, L., Botzko, K., Marois, M., Awad, J., King, M., Domurado, D., Bedros, M., Gosselin, C.: Albumin coating of a knitted polyester arterial prosthesis: an alternative to preclotting. Ann. Thorac. Surg. 37(6), 457–465 (1984)
Bos, G.W., Scharenborg, N.M., Poot, A.A., Engbers, G.H., Beugeling, T., Van Aken, W.G., Feijen, J.: Blood compatibility of surfaces with immobilized albumin–heparin conjugate and effect of endothelial cell seeding on platelet adhesion. J. Biomed. Mater. Res. 47(3), 279–291 (1999)
Mohammad, S., Olsen, D.: Immobilized albumin-immunoglobulin G for improved hemocompatibility of biopolymers. ASAIO J. 35(3), 384–387 (1989)
Hoffman, A., Schmer, G., Harris, C., Kraft, W.: Covalent binding of biomolecules to radiation-grafted hydrogels on inert polymer surfaces. ASAIO J. 18(1), 10–16 (1972)
Mikhalovska, L.I., Santin, M., Denyer, S.P., Lloyd, A.W., Teer, D.G., Field, S., Mikhalovsky, S.V.: Fibrinogen adsorption and platelet adhesion to metal and carbon coatings. Thromb. Haemost. 92(11), 1032–1039 (2004)
Dion, I., Roques, X., Baquey, H., Baudet, E., Basse Cathalinat, B., More, N.: Hemocompatibility of diamond-like carbon coating. Bio-Med. Mater. Eng. 3(1), 51–55 (1993)
Sivaraman, B., Latour, R.A.: The adherence of platelets to adsorbed albumin by receptor-mediated recognition of binding sites exposed by adsorption-induced unfolding. Biomaterials 31(6), 1036–1044 (2010)
Sivaraman, B., Latour, R.A.: Delineating the roles of the GPIIb/IIIa and GP-Ib-IX-V platelet receptors in mediating platelet adhesion to adsorbed fibrinogen and albumin. Biomaterials 32(23), 5365–5370 (2011)
Oyane, A., Ishizone, T., Uchida, M., Furukawa, K., Ushida, T., Yokoyama, H.: Spontaneous formation of blood-compatible surfaces on hydrophobic polymers: Surface enrichment of a block copolymer with a water-soluble block. Adv Mater 17(19), 2329–2332 (2005)
Okano, T., Aoyagi, T., Kataoka, K., Abe, K., Sakurai, Y., Shimada, M., Shinohara, I.: Hydrophilic-hydrophobic microdomain surfaces having an ability to suppress platelet aggregation and their in vitro antithrombogenicity. J. Biomed. Mater. Res. 20(7), 919–927 (1986)
Okano, T., Nishiyama, S., Shinohara, I., Akaike, T., Sakurai, Y., Kataoka, K., Tsuruta, T.: Effect of hydrophilic and hydrophobic microdomains on mode of interaction between block polymer and blood platelets. J. Biomed. Mater. Res. 15(3), 393–402 (1981)
Sato, H., Nakajima, A., Hayashi, T., Chen, G.W., Noishiki, Y.: Microheterophase structure, permeability, and biocompatibility of A-B-A triblock copolymer membranes composed of poly (γ-ethyl L-glutamate) as the A component and polybutadiene as the B component. J. Biomed. Mater. Res. 19(9), 1135–1155 (1985)
Nojiri, C., Okano, T., Jacobs, H.A., Park, K.D., Mohammad, S.F., Olsen, D.B., Kim, S.W.: Blood compatibility of PEO grafted polyurethane and HEMA/styrene block copolymer surfaces. J. Biomed. Mater. Res. 24(9), 1151–1171 (1990)
Takahara, A., Tashita, J.-I., Kajiyama, T., Takayanagi, M., Macknight, W.J.: Microphase separated structure and blood compatibility of segmented poly (urethaneureas) with different diamines in the hard segment. Polymer 26(7), 978–986 (1985)
Takahara, A., Tashita, J.-I., Kajiyama, T., Takayanagi, M., Macknight, W.J.: Microphase separated structure, surface composition and blood compatibility of segmented poly (urethaneureas) with various soft segment components. Polymer 26(7), 987–996 (1985)
Sasaki, T., Ratner, B.D., Hoffman, A.S.: Radiation-induced co-graft polymerization of 2-hydroxyethyl methacrylate and ethyl methacrylate onto silicone rubber films. In: Abstracts of Papers of ACS. ACS, Washington, DC, Jan 1975
Okano, T., Uruno, M., Sugiyama, N., Shimada, M., Shinohara, I., Kataoka, K., Sakurai, Y.: Suppression of platelet activity on microdomain surfaces of 2-hydroxyethyl methacrylate-polyether block copolymers. J. Biomed. Mater. Res. 20(7), 1035–1047 (1986)
Lewis, K.B., Ratner, B.D.: Observation of surface rearrangement of polymers using ESCA. J. Colloid Interface Sci. 159(1), 77–85 (1993)
Chen, H., Zhang, Z., Chen, Y., Brook, M.A., Sheardown, H.: Protein repellant silicone surfaces by covalent immobilization of poly (ethylene oxide). Biomaterials 26(15), 2391–2399 (2005)
Shen, M., Martinson, L., Wagner, M.S., Castner, D.G., Ratner, B.D., Horbett, T.A.: PEO-like plasma polymerized tetraglyme surface interactions with leukocytes and proteins: in vitro and in vivo studies. J. Biomater. Sci. Polym. Ed. 13(4), 367–390 (2002)
Fushimi, F., Nakayama, M., Nishimura, K., Hiyoshi, T.: Platelet adhesion, contact phase coagulation activation, and C5a generation of polyethylene glycol acid-grafted high flux cellulosic membrane with varieties of grafting amounts. Artif. Organs 22(10), 821–826 (1998)
Gorbet, M.B., Sefton, M.V.: Leukocyte activation and leukocyte procoagulant activities after blood contact with polystyrene and polyethylene glycol–immobilized polystyrene beads. J. Lab. Clin. Med. 137(5), 345–355 (2001)
Hansson, K.M., Tosatti, S., Isaksson, J., Wetterö, J., Textor, M., Lindahl, T.L., Tengvall, P.: Whole blood coagulation on protein adsorption-resistant PEG and peptide functionalised PEG-coated titanium surfaces. Biomaterials 26(8), 861–872 (2005)
Suggs, L.J., Shive, M.S., Garcia, C.A., Anderson, J.M., Mikos, A.G.: In vitro cytotoxicity and in vivo biocompatibility of poly (propylene fumarate-co-ethylene glycol) hydrogels. J. Biomed. Mater. Res. 46(1), 22–32 (1999)
Gombotz, W.R., Guanghui, W., Horbett, T.A., Hoffman, A.S.: Protein adsorption to poly (ethylene oxide) surfaces. J. Biomed. Mater. Res. 25(12), 1547–1562 (1991)
Norde, W., Gage, D.: Interaction of bovine serum albumin and human blood plasma with PEO-tethered surfaces: influence of PEO chain length, grafting density, and temperature. Langmuir 20(10), 4162–4167 (2004)
Kizhakkedathu, J.N., Janzen, J., Le, Y., Kainthan, R.K., Brooks, D.E.: Poly (oligo (ethylene glycol) acrylamide) brushes by surface initiated polymerization: effect of macromonomer chain length on brush growth and protein adsorption from blood plasma. Langmuir 25(6), 3794–3801 (2009)
Lai, B.F., Creagh, A.L., Janzen, J., Haynes, C.A., Brooks, D.E., Kizhakkedathu, J.N.: The induction of thrombus generation on nanostructured neutral polymer brush surfaces. Biomaterials 31(26), 6710–6718 (2010)
Thierry, B., Merhi, Y., Silver, J., Tabrizian, M.: Biodegradable membrane-covered stent from chitosan-based polymers. J. Biomed. Mater. Res. A 75(3), 556–566 (2005)
Wright, M.J., Woodrow, G., Umpleby, S., Hull, S., Brownjohn, A.M., Turney, J.H.: Low thrombogenicity of polyethylene glycol–grafted cellulose membranes does not influence heparin requirements in hemodialysis. Am. J. Kidney Dis. 34(1), 36–42 (1999)
Sirolli, V., Di Stante, S., Stuard, S., Di Liberato, L., Amoroso, L., Cappelli, P., Bonomini, M.: Biocompatibility and functional performance of a polyethylene glycol acid-grafted cellulosic membrane for hemodialysis. Int. J. Artif. Organs 23(6), 356–364 (2000)
Cao, L., Sukavaneshvar, S., Ratner, B.D., Horbett, T.A.: Glow discharge plasma treatment of polyethylene tubing with tetraglyme results in ultralow fibrinogen adsorption and greatly reduced platelet adhesion. J. Biomed. Mater. Res. A 79(4), 788–803 (2006)
Rodriguez-Emmenegger, C., Brynda, E., Riedel, T., Houska, M., Šubr, V., Alles, A.B., Hasan, E., Gautrot, J.E., Huck, W.T.: Polymer brushes showing non-fouling in blood plasma challenge the currently accepted design of protein resistant surfaces. Macromol. Rapid Commun. 32(13), 952–957 (2011)
Herring, M.B., Compton, R., Legrand, D.R., Gardner, A.L.: Endothelial cell seeding in the management of vascular thrombosis. Semin. Thromb. Hemost. 15(2), 200–205 (1989)
Williams, S.K., Rose, D.G., Jarrell, B.E.: Microvascular endothelial cell sodding of ePTFE vascular grafts: improved patency and stability of the cellular lining. J. Biomed. Mater. Res. 28(2), 203–212 (1994)
Zilla, P., Deutsch, M., Meinhart, J., Puschmann, R., Eberl, T., Minar, E., Dudczak, R., Lugmaier, H., Schmidt, P., Noszian, I.: Clinical in vitro endothelialization of femoropopliteal bypass grafts: an actuarial follow-up over three years. J. Vasc. Surg. 19(3), 540–548 (1994)
Kutryk, M., van Dortmont, L., de Crom, R., van der Kamp, A., Verdouw, P., van der Giessen, W.: Seeding of intravascular stents by the xenotransplantation of genetically modified endothelial cells. Semin. Interv. Cardiol. 3(3-4), 217–220 (1998)
Zünd, G., Hoerstrup, S.P., Schoeberlein, A., Lachat, M., Uhlschmid, G., Vogt, P.R., Turina, M.: Tissue engineering: a new approach in cardiovascular surgery; seeding of human fibroblasts followed by human endothelial cells on resorbable mesh. Eur. J. Cardiothorac. Surg. 13(2), 160–164 (1998)
Taite, L.J., Yang, P., Jun, H.-W., West, J.L.: Nitric oxide-releasing polyurethane–PEG copolymer containing the YIGSR peptide promotes endothelialization with decreased platelet adhesion. J. Biomed. Mater. Res. B Appl. Biomater. 84(1), 108–116 (2008)
L’heureux, N., Pâquet, S., Labbé, R., Germain, L., Auger, F.A.: A completely biological tissue-engineered human blood vessel. FASEB J 12(1), 47–56 (1998)
Kawamoto, Y., Nakao, A., Ito, Y., Wada, N., Kaibara, M.: Endothelial cells on plasma-treated segmented-polyurethane: adhesion strength, antithrombogenicity and cultivation in tubes. J. Mater. Sci. Mater. Med. 8(9), 551–557 (1997)
Li, J., Ding, M., Fu, Q., Tan, H., Xie, X., Zhong, Y.: A novel strategy to graft RGD peptide on biomaterials surfaces for endothelization of small-diamater vascular grafts and tissue engineering blood vessel. J. Mater. Sci. Mater. Med. 19(7), 2595–2603 (2008)
Yin, M., Yuan, Y., Liu, C., Wang, J.: Development of mussel adhesive polypeptide mimics coating for in-situ inducing re-endothelialization of intravascular stent devices. Biomaterials 30(14), 2764–2773 (2009)
Jiang, S., Cao, Z.: Ultralow-fouling, functionalizable, and hydrolyzable zwitterionic materials and their derivatives for biological applications. Adv. Mater. 22(9), 920–932 (2010)
Wu, L., Guo, Z., Meng, S., Zhong, W., Du, Q., Chou, L.L.: Synthesis of a zwitterionic silane and its application in the surface modification of silicon-based material surfaces for improved hemocompatibility. ACS Appl. Mater. Interfaces 2(10), 2781–2788 (2010)
Zhang, Z., Chen, S., Chang, Y., Jiang, S.: Surface grafted sulfobetaine polymers via atom transfer radical polymerization as superlow fouling coatings. J. Phys. Chem. B 110(22), 10799–10804 (2006)
Ishihara, K., Nomura, H., Mihara, T., Kurita, K., Iwasaki, Y., Nakabayashi, N.: Why do phospholipid polymers reduce protein adsorption? J. Biomed. Mater. Res. 39(2), 323–330 (1998)
Holmlin, R.E., Chen, X., Chapman, R.G., Takayama, S., Whitesides, G.M.: Zwitterionic SAMs that resist nonspecific adsorption of protein from aqueous buffer. Langmuir 17(9), 2841–2850 (2001)
Jiang, Y., Rongbing, B., Ling, T., Jian, S., Sicong, L.: Blood compatibility of polyurethane surface grafted copolymerization with sulfobetaine monomer. Colloids Surf. B Biointerfaces 36(1), 27–33 (2004)
Min, D.Y., Li, Z.Z., Shen, J., Lin, S.C.: Research and synthesis of organosilicon nonthrombogenic materials containing sulfobetaine group. Colloids Surf. B Biointerfaces 79(2), 415–420 (2010)
Yuan, J., Lin, S., Shen, J.: Enhanced blood compatibility of polyurethane functionalized with sulfobetaine. Colloids Surf. B Biointerfaces 66(1), 90–95 (2008)
West, S.L., Salvage, J.P., Lobb, E.J., Armes, S.P., Billingham, N.C., Lewis, A.L., Hanlon, G.W., Lloyd, A.W.: The biocompatibility of crosslinkable copolymer coatings containing sulfobetaines and phosphobetaines. Biomaterials 25(7–8), 1195–1204 (2004)
Lobb, E.J., Ma, I., Billingham, N.C., Armes, S.P., Lewis, A.L.: Facile synthesis of well-defined, biocompatible phosphorylcholine-based methacrylate copolymers via atom transfer radical polymerization at 20 C. J. Am. Chem. Soc. 123(32), 7913–7914 (2001)
Ma, H., Hyun, J., Stiller, P., Chilkoti, A.: “Non-fouling” oligo(ethylene glycol)- functionalized polymer brushes synthesized by surface-initiated atom transfer radical polymerization. Adv. Mater. 16(4), 338–341 (2004)
Zhang, Z., Zhang, M., Chen, S., Horbett, T.A., Ratner, B.D., Jiang, S.: Blood compatibility of surfaces with superlow protein adsorption. Biomaterials 29(32), 4285–4291 (2008)
Yuan, J., Zhang, J., Zhou, J., Yuan, Y., Shen, J., Lin, S.: Platelet adhesion onto segmented polyurethane surfaces modified by carboxybetaine. J. Biomater. Sci. Polym. Ed. 14(12), 1339–1349 (2003)
Kitano, H., Tada, S., Mori, T., Takaha, K., Gemmei-Ide, M., Tanaka, M., Fukuda, M., Yokoyama, Y.: Correlation between the structure of water in the vicinity of carboxybetaine polymers and their blood-compatibility. Langmuir 21(25), 11932–11940 (2005)
Lee, B., Shin, H.-S., Park, K., Han, D.: Surface grafting of blood compatible zwitterionic poly(ethylene glycol) on diamond-like carbon-coated stent. J. Mater. Sci. Mater. Med. 22(3), 507–514 (2011)
Yuan, Y., Ai, F., Zang, X., Zhuang, W., Shen, J., Lin, S.: Polyurethane vascular catheter surface grafted with zwitterionic sulfobetaine monomer activated by ozone. Colloids Surf. B Biointerfaces 35(1), 1–5 (2004)
Galli, M., Sommariva, L., Prati, F., Zerboni, S., Politi, A., Bonatti, R., Mameli, S., Butti, E., Pagano, A., Ferrari, G.: Acute and mid-term results of phosphorylcholine-coated stents in primary coronary stenting for acute myocardial infarction. Catheter Cardiovasc. Interv. 53(2), 182–187 (2001)
Lewis, A., Tolhurst, L., Stratford, P.: Analysis of a phosphorylcholine-based polymer coating on a coronary stent pre-and post-implantation. Biomaterials 23(7), 1697–1706 (2002)
Rabenstein, D.L.: Heparin and heparan sulfate: structure and function. Nat. Prod. Rep. 19(3), 312–331 (2002)
Hylton, D.M., Shalaby, S.W., Latour, R.A.: Direct correlation between adsorption-induced changes in protein structure and platelet adhesion. J Biomed. Mater. Res. A 73A(3), 349–358 (2005)
Ayres, N., Holt, D., Jones, C., Corum, L., Grainger, D.: Polymer brushes containing sulfonated sugar repeat units: synthesis, characterization, and in vitro testing of blood coagulation activation. J. Polym. Sci., Part A: Polym. Chem. 46(23), 7713–7724 (2008)
Kidane, A.G., Salacinski, H., Tiwari, A., Bruckdorfer, K.R., Seifalian, A.M.: Anticoagulant and antiplatelet agents: their clinical and device application (s) together with usages to engineer surfaces. Biomacromolecules 5(3), 798–813 (2004)
Sakiyama-Elbert, S.E.: Incorporation of heparin into biomaterials. Acta Biomater. 10(4), 1581–1587 (2013)
Wan Kim, S., Jacobs, H.: Design of nonthrombogenic polymer surfaces for blood-contacting medical devices. Blood Purif. 14(5), 357–372 (1996)
Larm, O., Larsson, R., Olsson, P.: A new non-thrombogenic surface prepared by selective covalent binding of heparin via a modified reducing terminal residue. Biomater. Med. Devices Artif. Organs 11(2–3), 161–173 (1983)
Michanetzis, G., Katsala, N., Missirlis, Y.: Comparison of haemocompatibility improvement of four polymeric biomaterials by two heparinization techniques. Biomaterials 24(4), 677–688 (2003)
Miyara, H., Harumiya, N., Mori, Y., Tanzawa, H.: A new antithrombogenic heparinized polymer. J. Biomed. Mater. Res. 11(2), 251–265 (1977)
Goddard, J.M., Hotchkiss, J.H.: Polymer surface modification for the attachment of bioactive compounds. Prog. Polym. Sci. 32(7), 698–725 (2007)
Engbers, G.H., Feijen, J.: Current techniques to improve the blood compatibility of biomaterial surfaces. Int. J. Artif. Organs 14(4), 199–215 (1991)
Gölander, C., Arwin, H., Eriksson, J., Lundstrom, I., Larsson, R.: Heparin surface film formation through adsorption of colloidal particles studied by ellipsometry and scanning electron microscopy. Colloids Surf. 5(1), 1–16 (1982)
Seifert, B., Romaniuk, P., Groth, T.: Bioresorbable, heparinized polymers for stent coating: in vitro studies on heparinization efficiency, maintenance of anticoagulant properties and improvement of stent haemocompatibility. J. Mater. Sci. Mater. Med. 7(8), 465–469 (1996)
Bamford, C., Al-Lamee, K.: Studies in polymer surface modification and grafting for biomedical uses: 2. Application to arterial blood filters and oxygenators. Polymer 37(22), 4885–4889 (1996)
Byun, Y., Jacobs, H.A., Kim, S.W.: Heparin surface immobilization through hydrophilic spacers: thrombin and antithrombin III binding kinetics. J. Biomater. Sci. Polym. Ed. 6(1), 1–13 (1994)
Park, K.D., Okano, T., Nojiri, C., Kim, S.W.: Heparin immobilization onto segmented polyurethaneurea surfaces—effect of hydrophilic spacers. J. Biomed. Mater. Res. 22(11), 977–992 (1988)
Arnander, C., Bagger-Sjoebaeck, D., Frebelius, S., Larsson, R., Swedenborg, J.: Long-term stability in vivo of a thromboresistant heparinized surface. Biomaterials 8(6), 496–499 (1987)
Vroman, L.: The life of an artificial device in contact with blood: initial events and their effect on its final state. Bull. N. Y. Acad. Med. 64(4), 352 (1988)
Wendel, H.P., Ziemer, G.: Coating-techniques to improve the hemocompatibility of artificial devices used for extracorporeal circulation. Eur. J. Cardiothorac. Surg. 16(3), 342–350 (1999)
Keuren, J.F., Wielders, S.J., Willems, G.M., Morra, M., Lindhout, T.: Fibrinogen adsorption, platelet adhesion and thrombin generation at heparinized surfaces exposed to flowing blood. Thromb. Haemost. 87(4), 742–747 (2002)
Weber, N., Wendel, H.P., Ziemer, G.: Hemocompatibility of heparin-coated surfaces and the role of selective plasma protein adsorption. Biomaterials 23(2), 429–439 (2002)
Sperling, C., Houska, M., Brynda, E., Streller, U., Werner, C.: In vitro hemocompatibility of albumin–heparin multilayer coatings on polyethersulfone prepared by the layer-by-layer technique. J. Biomed. Mater. Res. A 76(4), 681–689 (2006)
Werner, C., Maitz, M.F., Sperling, C.: Current strategies towards hemocompatible coatings. J. Mater. Chem. 17(32), 3376–3384 (2007)
Vaughn, M.W.: Estimation of nitric oxide production and reaction rates in tissue by use of a mathematical model. Am. J. Physiol. Heart Circ. Physiol. 274(6), H2163–H2176 (1998)
Frost, M.C., Reynolds, M.M., Meyerhoff, M.E.: Polymers incorporating nitric oxide releasing/generating substances for improved biocompatibility of blood-contacting medical devices. Biomaterials 26(14), 1685–1693 (2005)
Seabra, A.B., Marcato, P.D., De Paula, L.B., Durán, N.: New strategy for controlled release of nitric oxide. J. Nano. Res. 20, 61–67 (2012)
Riccio, D.A., Schoenfisch, M.H.: Nitric oxide release: Part I. Macromol. Scaffolds. Chem. Soc. Rev. 41(10), 3731–3741 (2012)
Kim, J., Saravanakumar, G., Choi, H.W., Park, D., Kim, W.J.: A platform for nitric oxide delivery. J. Mater. Chem. B Mater. Biol. Med. 2, 341–356 (2014)
Jen, M.C., Serrano, M.C., Van Lith, R., Ameer, G.A.: Polymer-based nitric oxide therapies: recent insights for biomedical applications. Adv. Funct. Mater. 22(2), 239–260 (2012)
Halpenny, G.M., Mascharak, P.K.: Emerging antimicrobial applications of nitric oxide (NO) and NO-releasing materials. Anti-Infect. Agents Med. Chem. 9(4), 187–197 (2010)
Eroy-Reveles, A.A., Mascharak, P.K.: Nitric oxide-donating materials and their potential in pharmacological applications for site-specific nitric oxide delivery. Future Med. Chem. 1(8), 1497–1507 (2009)
Carpenter, A.W., Schoenfisch, M.H.: Nitric oxide release: Part II. Ther. Appl. Chem. Soc. Rev. 41(10), 3742–3752 (2012)
Mowery, K.A., Schoenfisch, M.H., Saavedra, J.E., Keefer, L.K., Meyerhoff, M.E.: Preparation and characterization of hydrophobic polymeric films that are thromboresistant via nitric oxide release. Biomaterials 21(1), 9–21 (2000)
Skrzypchak, A.M., Lafayette, N.G., Bartlett, R.H., Zhou, Z., Frost, M.C., Meyerhoff, M.E., Reynolds, M.M., Annich, G.M.: Effect of varying nitric oxide release to prevent platelet consumption and preserve platelet function in an in vivo model of extracorporeal circulation. Perfusion 22(3), 193–200 (2007)
Batchelor, M.M., Reoma, S.L., Fleser, P.S., Nuthakki, V.K., Callahan, R.E., Shanley, C.J., Politis, J.K., Elmore, J., Merz, S.I., Meyerhoff, M.E.: More lipophilic dialkyldiamine-based diazeniumdiolates: synthesis, characterization, and application in preparing thromboresistant nitric oxide release polymeric coatings. J. Med. Chem. 46(24), 5153–5161 (2003)
Fleser, P.S., Nuthakki, V.K., Malinzak, L.E., Callahan, R.E., Seymour, M.L., Reynolds, M.M., Merz, S.I., Meyerhoff, M.E., Bendick, P.J., Zelenock, G.B., Shanley, C.J.: Nitric oxide-releasing biopolymers inhibit thrombus formation in a sheep model of artcriovenous bridge grafts. J. Vasc. Surg. 40(4), 803–811 (2004)
Smith, D.J., Chakravarthy, D., Pulfer, S., Simmons, M.L., Hrabie, J.A., Citro, M.L., Saavedra, J.E., Davies, K.M., Hutsell, T.C., Mooradian, D.L., Hanson, S.R., Keefer, L.K.: Nitric oxide-releasing polymers containing the [N(O)NO]- group. J. Med. Chem. 39(5), 1148–1156 (1996)
Annich, G.M., Meinhardt, J.P., Mowery, K.A., Ashton, B.A., Merz, S.I., Hirschl, R.B., Meyerhoff, M.E., Bartlett, R.H.: Reduced platelet activation and thrombosis in extracorporeal circuits coated with nitric oxide release polymers. Crit. Care Med. 28(4), 915–920 (2000)
Zhang, H., Annich, G.M., Miskulin, J., Osterholzer, K., Merz, S.I., Bartlett, R.H., Meyerhoff, M.E.: Nitric oxide releasing silicone rubbers with improved blood compatibility: preparation, characterization, and in vivo evaluation. Biomaterials 23(6), 1485–1494 (2002)
Marxer, S.M., Rothrock, A.R., Nablo, B.J., Robbins, M.E., Schoenfisch, M.H.: Preparation of nitric oxide (NO)-releasing sol-gels for biomaterial applications. Chem. Mater. 15(22), 4193–4199 (2003)
Nablo, B.J., Schoenfisch, M.H.: In vitro cytotoxicity of nitric oxide-releasing sol-gel derived materials. Biomaterials 26(21), 4405–4415 (2005)
Hetrick, E.M., Schoenfisch, M.H.: Antibacterial nitric oxide-releasing xerogels: Cell viability and parallel plate flow cell adhesion studies. Biomaterials 28(11), 1948–1956 (2007)
Koh, A., Carpenter, A.W., Slomberg, D.L., Schoenfisch, M.H.: Nitric oxide-releasing silica nanoparticle-doped polyurethane electrospun fibers. ACS Appl. Mater. Interfaces 5(16), 7956–7964 (2013)
Koh, A., Riccio, D.A., Sun, B., Carpenter, A.W., Nichols, S.P., Schoenfisch, M.H.: Fabrication of nitric oxide-releasing polyurethane glucose sensor membranes. Biosens. Bioelectron. 28(1), 17–24 (2011)
Shin, J.H., Metzger, S.K., Schoenfisch, M.H.: Synthesis of nitric oxide-releasing silica nanoparticles. J. Am. Chem. Soc. 129(15), 4612–4619 (2007)
Shin, J.H., Schoenfisch, M.H.: Inorganic/organic hybrid silica nanoparticles as a nitric oxide delivery scaffold. Chem. Mater. 20(1), 239–249 (2008)
Lu, Y., Sun, B., Li, C.H., Schoenfisch, M.H.: Structurally diverse nitric oxide-releasing poly(propylene imine) dendrimers. Chem. Mater. 23(18), 4227–4233 (2011)
Stasko, N.A., Schoenfisch, M.H.: Dendrimers as a scaffold for nitric oxide release. J. Am. Chem. Soc. 128(25), 8265–8271 (2006)
Sun, B., Slomberg, D.L., Chudasama, S.L., Lu, Y., Schoenfisch, M.H.: Nitric oxide-releasing dendrimers as antibacterial agents. Biomacromolecules 13(10), 3343–3354 (2012)
Lantvit, S.M., Barrett, B.J., Reynolds, M.M.: Nitric oxide releasing material adsorbs more fibrinogen. J. Biomed. Mater. Res. A 101(11), 3201–3210 (2013)
Wu, Y., Zhou, Z., Meyerhoff, M.E.: In vitro platelet adhesion on polymeric surfaces with varying fluxes of continuous nitric oxide release. J. Biomed. Mater. Res. A 81(4), 956–963 (2007)
Major, T.C., Brant, D.O., Reynolds, M.M., Bartlett, R.H., Meyerhoff, M.E., Handa, H., Annich, G.M.: The attenuation of platelet and monocyte activation in a rabbit model of extracorporeal circulation by a nitric oxide releasing polymer. Biomaterials 31(10), 2736–2745 (2010)
Handa, H., Brisbois, E.J., Major, T.C., Refahiyat, L., Amoako, K.A., Annich, G.M., Bartlett, R.H., Meyerhoff, M.E.: In vitro and in vivo study of sustained nitric oxide release coating using diazeniumdiolate-doped poly(vinyl chloride) matrix with poly(lactide-co-glycolide) additive. J. Mater. Chem. B Mater. Biol. Med. 1(29), 3578–3587 (2013)
Handa, H., Major, T.C., Brisbois, E.J., Amoako, K.A., Meyerhoff, M.E., Bartlett, R.H.: Hemocompatibility comparison of biomedical grade polymers using rabbit thrombogenicity model for preparing nonthrombogenic nitric oxide releasing surfaces. J. Mater. Chem. B Mater. Biol. Med. 2(8), 1059–1067 (2014)
Al-Sa’doni, H., Ferro, A.: S-nitrosothiols: a class of nitric oxide-donor drugs. Clin. Sci. 98(5), 507–520 (2000)
Hogg, N.: Biological chemistry and clinical potential of S-nitrosothiols. Free Radical Biol. Med. 28(10), 1478–1486 (2000)
Hogg, N., Singh, R.J., Kalyanaraman, B.: The role of glutathione in the transport and catabolism of nitric oxide. FEBS Lett. 382(3), 223–228 (1996)
Langford, E.J., Brown, A.S., Wainwright, R.J., Debelder, A.J., Thomas, M.R., Smith, R.E.A., Radomski, M.W., Martin, J.F., Moncada, S.: Inhibition of platelet activity by S-nitrosoglutathione during coronary angioplasty. Lancet 344(8935), 1458–1460 (1994)
Radomski, M.W., Rees, D.D., Dutra, A., Moncada, S.: S-nitroso-glutathione inhibits platelet activation in vitro and in vivo. Br. J. Pharmacol. 107(3), 745–749 (1992)
Salas, E., Moro, M.A., Askew, S., Hodson, H.F., Butler, A.R., Radomski, M.W., Moncada, S.: Comparative pharmacology of analogues of S-nitroso-N-acetyl-DL-penicillamine on human platelets. Br. J. Pharmacol. 112(4), 1071–1076 (1994)
De Souza, G.F.P., Yokoyama-Yasunaka, J.K.U., Seabra, A.B., Miguel, D.C., De Oliveira, M.G., Uliana, S.R.B.: Leishmanicidal activity of primary S-nitrosothiols against Leishmania major and Leishmania amazonensis: implications for the treatment of cutaneous leishmaniasis. Nitric Oxide 15(3), 209–216 (2006)
Salas, E., Langford, E.J., Marrinan, M.T., Martin, J.F., Moncada, S., De Belder, A.J.: S-nitrosoglutathione inhibits platelet activation and deposition in coronary artery saphenous vein grafts in vitro and in vivo. Heart 80(2), 146–150 (1998)
Albert, J., Daleskog, M., Wallen, N.H.: A comparison of the antiplatelet effect of S-nitrosoglutathione in whole blood and platelet-rich plasma. Thromb. Res. 102(2), 161–165 (2001)
Ricardo, K.F.S., Shishido, S.M., De Oliveira, M.G., Krieger, M.H.: Characterization of the hypotensive effect of S-nitroso-N-acetylcysteine in normotensive and hypertensive conscious rats. Nitric Oxide-Biol. Ch 7(1), 57–66 (2002)
Dicks, A.P., Swift, H.R., Williams, D.L.H., Butler, A.R., Al-Sa’doni, H.H., Cox, B.G.: Identification of Cu + as the effective reagent in nitric oxide formation from S-nitrosothiols (RSNO). J. Chem. Soc. Perkin Trans. 2, 481–487 (1996)
Sexton, D.J., Muruganandam, A., Mckenney, D.J., Mutus, B.: Visible light photochemical release of nitric oxide from S-nitrosoglutathione: potential photochemotherapeutic applications. Photochem. Photobiol. 59(4), 463–467 (1994)
Wood, P.D., Mutus, B., Redmond, R.W.: The mechanism of photochemical release of nitric oxide from S-nitrosoglutathione. Photochem. Photobiol. 64(3), 518–524 (1996)
Frost, M.C., Meyerhoff, M.E.: Controlled photoinitiated release of nitric oxide from polymer films containing S-Nitroso-N-acetyl-dl-penicillamine derivatized fumed silica filler. J. Am. Chem. Soc. 126(5), 1348–1349 (2004)
Shishido, S.L.M., Seabra, A.B., Loh, W., Ganzarolli De Oliveira, M.: Thermal and photochemical nitric oxide release from S-nitrosothiols incorporated in Pluronic F127 gel: potential uses for local and controlled nitric oxide release. Biomaterials 24(20), 3543–3553 (2003)
Seabra, A.B., De Oliveira, M.G.: Poly(vinyl alcohol) and poly(vinyl pyrrolidone) blended films for local nitric oxide release. Biomaterials 25(17), 3773–3782 (2004)
Seabra, A.B., De Souza, G.F.P., Da Rocha, L.L., Eberlin, M.N., De Oliveira, M.G.: S-nitrosoglutathione incorporated in poly(ethylene glycol) matrix: potential use for topical nitric oxide delivery. Nitric Oxide-Biol. Ch 11(3), 263–272 (2004)
Seabra, A.B., Da Rocha, L.L., Eberlin, M.N., De Oliveira, M.G.: Solid films of blended poly(vinyl alcohol)/poly(vinyl pyrrolidone) for topical S-nitrosoglutathione and nitric oxide release. J. Pharm. Sci. 94(5), 994–1003 (2005)
Simoes, M., De Oliveira, M.G.: Poly(vinyl alcohol) films for topical delivery of S-Nitrosoglutathione: effect of freezing-thawing on the diffusion properties. J. Biomed. Mater. Res. B Appl. Biomater. 93B(2), 416–424 (2010)
Brisbois, E.J., Handa, H., Major, T.C., Bartlett, R.H., Meyerhoff, M.E.: Long-term nitric oxide release and elevated temperature stability with S-nitroso-N-acetylpenicillamine (SNAP)-doped Elast-eon E2As polymer. Biomaterials 34(28), 6957–6966 (2013)
Joslin, J.M., Lantvit, S.M., Reynolds, M.M.: Nitric oxide releasing tygon materials: studies in donor leaching and localized nitric oxide release at a polymer-buffer interface. ACS Appl. Mater. Interfaces 5(19), 9285–9294 (2013)
Riccio, D.A., Dobmeier, K.P., Hetrick, E.M., Privett, B.J., Paul, H.S., Schoenfisch, M.H.: Nitric oxide-releasing S-nitrosothiol-modified xerogels. Biomaterials 30(27), 4494–4502 (2009)
Riccio, D.A., Coneski, P.N., Nichols, S.P., Broadnax, A.D., Schoenfisch, M.H.: Photoinitiated nitric oxide-releasing tertiary S-Nitrosothiol-modified xerogels. ACS Appl. Mater. Interfaces 4(2), 796–804 (2012)
Coneski, P.N., Schoenfisch, M.H.: Synthesis of nitric oxide-releasing polyurethanes with S-nitrosothiol-containing hard and soft segments. Polym Chem. 2(4), 906–913 (2011)
Frost, M.C., Meyerhoff, M.E.: Synthesis, characterization, and controlled nitric oxide release from S-nitrosothiol-derivatized fumed silica polymer filler particles. J. Biomed. Mater. Res. A 72A(4), 409–419 (2005)
Coneski, P.N., Rao, K.S., Schoenfisch, M.H.: Degradable nitric oxide-releasing biomaterials via post-polymerization functionalization of cross-linked polyesters. Biomacromolecules 11(11), 3208–3215 (2010)
Damodaran, V.B., Place, L.W., Kipper, M.J., Reynolds, M.M.: Enzymatically degradable nitric oxide releasing S-nitrosated dextran thiomers for biomedical applications. J. Mater. Chem. 22, 23038–23048 (2012)
Damodaran, V.B., Reynolds, M.M.: Biodegradable S-nitrosothiol tethered multiblock polymer for nitric oxide delivery. J. Mater. Chem. 21, 5870–5872 (2011)
Damodaran, V.B., Joslin, J.M., Wold, K.A., Lantvit, S.M., Reynolds, M.M.: S-Nitrosated biodegradable polymers for biomedical applications: synthesis, characterization and impact of thiol structure on the physicochemical properties. J. Mater. Chem. 22, 5990–6001 (2012)
Seabra, A.B., Martins, D., Simoes, M., Da Silva, R., Brocchi, M., De Oliveira, M.G.: Antibacterial nitric oxide-releasing polyester for the coating of blood-contacting artificial materials. Artif. Organs 34(7), E204–E214 (2010)
Seabra, A.B., Da Silva, R., De Souza, G.F.P., De Oliveira, M.G.: Antithrombogenic polynitrosated polyester/poly(methyl methacrylate) blend for the coating of blood-contacting surfaces. Artif. Organs 32(4), 262–267 (2008)
Li, Y., Lee, P.I.: Controlled nitric oxide delivery platform based on S-nitrosothiol conjugated interpolymer complexes for diabetic wound healing. Mol. Pharm. 7(1), 254–266 (2009)
Gierke, G.E., Nielsen, M., Frost, M.C.: S-Nitroso- N -acetyl-D-penicillamine covalently linked to polydimethylsiloxane (SNAP–PDMS) for use as a controlled photoinitiated nitric oxide release polymer. Sci. Technol. Adv. Mat. 12(5), 055007 (2011)
Stasko, N.A., Fischer, T.H., Schoenfisch, M.H.: S-Nitrosothiol-modified dendrimers as nitric oxide delivery vehicles. Biomacromolecules 9(3), 834–841 (2008)
Riccio, D.A., Nugent, J.L., Schoenfisch, M.H.: Stober synthesis of nitric oxide-releasing S-Nitrosothiol-modified silica particles. Chem. Mater. 23(7), 1727–1735 (2011)
Duan, X., Lewis, R.S.: Improved haemocompatibility of cysteine-modified polymers via endogenous nitric oxide. Biomaterials 23(4), 1197–1203 (2002)
Gappa-Fahlenkamp, H., Lewis, R.S.: Improved hemocompatibility of poly(ethylene terephthalate) modified with various thiol-containing groups. Biomaterials 26(17), 3479–3485 (2005)
Oh, B.K., Meyerhoff, M.E.: Spontaneous Catalytic Generation of Nitric Oxide from S-Nitrosothiols at the Surface of Polymer Films Doped with Lipophilic Copper(II) Complex. J. Am. Chem. Soc. 125(32), 9552–9553 (2003)
Oh, B.K., Meyerhoff, M.E.: Catalytic generation of nitric oxide from nitrite at the interface of polymeric films doped with lipophilic Cu (II)-complex: a potential route to the preparation of thromboresistant coatings. Biomaterials 25(2), 283–293 (2004)
Hwang, S., Meyerhoff, M.E.: Polyurethane with tethered copper (II)–cyclen complex: preparation, characterization and catalytic generation of nitric oxide from S-nitrosothiols. Biomaterials 29(16), 2443–2452 (2008)
Puiu, S.C., Zhou, Z., White, C.C., Neubauer, L.J., Zhang, Z., Lange, L.E., Mansfield, J.A., Meyerhoff, M.E., Reynolds, M.M.: Metal ion-mediated nitric oxide generation from polyurethanes via covalently linked copper(II)-cyclen moieties. J. Biomed. Mater. Res. Part B Appl. Biomater. 91B(1), 203–212 (2009)
Major, T.C., Brant, D.O., Burney, C.P., Amoako, K.A., Annich, G.M., Meyerhoff, M.E., Handa, H., Bartlett, R.H.: The hemocompatibility of a nitric oxide generating polymer that catalyzes S-nitrosothiol decomposition in an extracorporeal circulation model. Biomaterials 32(26), 5957–5969 (2011)
Mugesh, G., Singh, H.B.: Synthetic organoselenium compounds as antioxidants: glutathione peroxidase activity. Chem. Soc. Rev. 29(5), 347–357 (2000)
Hou, Y., Guo, Z., Li, J., Wang, P.G.: Seleno compounds and glutathione peroxidase catalyzed decomposition of S-nitrosothiols. Biochem. Biophys. Res. Commun. 228(1), 88–93 (1996)
Yang, J., Welby, J.L., Meyerhoff, M.E.: Generic nitric oxide (NO) generating surface by immobilizing organoselenium species via layer-by-layer assembly. Langmuir 24(18), 10265–10272 (2008)
Cha, W., Meyerhoff, M.E.: Catalytic generation of nitric oxide from S-nitrosothiols using immobilized organoselenium species. Biomaterials 28(1), 19–27 (2007)
Cai, W., Wu, J., Xi, C., Ashe Iii, A.J., Meyerhoff, M.E.: Carboxyl-ebselen-based layer-by-layer films as potential antithrombotic and antimicrobial coatings. Biomaterials 32(31), 7774–7784 (2011)
Reynolds, M.M., Frost, M.C., Meyerhoff, M.E.: Nitric oxide-releasing hydrophobic polymers: preparation, characterization, and potential biomedical applications. Free Radical Biol. Med. 37(7), 926–936 (2004)
Sefton, M.V., Gemmell, C.H., Gorbet, M.B.: What really is blood compatibility? J. Biomat. Sci-Polym. E 11(11), 1165–1182 (2000)
Lemm, W., Unger, V., Bucherl, E.S.: Blood compatibility of polymers: in vitro and in vivo tests. Med. Biol. Eng. Comput. 18(4), 521–526 (1980)
Kirkpatrick, C., Mittermayer, C.: Theoretical and practical aspects of testing potential biomaterials in vitro. J. Mater. Sci. Mater. Med. 1(1), 9–13 (1990)
Sefton, M.V.: Perspective on hemocompatibility testing. J. Biomed. Mater. Res. 55(4), 445–446 (2001)
Seyfert, U.T., Biehl, V., Schenk, J.: In vitro hemocompatibility testing of biomaterials according to the ISO 10993-4. Biomol. Eng. 19(2), 91–96 (2002)
Braune, S., Grunze, M., Straub, A., Jung, F.: Are there sufficient standards for the in vitro hemocompatibility testing of biomaterials? Biointerphases 8(1), 1–9 (2013)
Ratner, B.D.: Evaluation of the blood compatibility of synthetic polymers: consensus and significance. Contemporary Biomaterials: Material and Host Response, Clinical Applications, New Technology and Legal Aspects, pp. 193–204. Noyes Publications, Park Ridge (1984)
Hanson, S.R., Ratner, B.: Evaluation of blood-materials interactions. In: Biomaterials science: an introduction to materials in medicine, pp. 367–378. San Diego (2004)
Ratner, B.D., Horbett, T.A.: Chapter II.3.5—Evaluation of blood–materials interactions. In: Ratner BD, Hoffman AS, Schoen FJ, Lemons JE (eds) Biomaterials Science (Third Edition), pp. 617–634. Academic Press, Waltham (2013)
Sefton, M.V., Sawyer, A., Gorbet, M., Black, J.P., Cheng, E., Gemmell, C., Pottinger-Cooper, E.: Does surface chemistry affect thrombogenicity of surface modified polymers? J. Biomed. Mater. Res. 55(4), 447–459 (2001)
Gristina, A.G.: Biomaterial-centered infection: microbial adhesion versus tissue integration. Science 237, 1588–1595 (1987)
Tzoneva, R., Heuchel, M., Groth, T., Altankov, G., Albrecht, W., Paul, D.: Fibrinogen adsorption and platelet interactions on polymer membranes. J. Biomater. Sci. Polym. Ed. 13(9), 1033–1050 (2002)
Haycox, C.L., Ratner, B.D.: In vitro platelet interactions in whole human blood exposed to biomaterial surfaces: Insights on blood compatibility. J. Biomed. Mater. Res. 27(9), 1181–1193 (1993)
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Brisbois, E.J., Handa, H., Meyerhoff, M.E. (2015). Recent Advances in Hemocompatible Polymers for Biomedical Applications. In: Puoci, F. (eds) Advanced Polymers in Medicine. Springer, Cham. https://doi.org/10.1007/978-3-319-12478-0_16
Download citation
DOI: https://doi.org/10.1007/978-3-319-12478-0_16
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-12477-3
Online ISBN: 978-3-319-12478-0
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)