Abstract
Over the last two decades the smart polymeric materials have involved the interest of the scientific community because they can be used in the design and formulation of dosage forms that respond, with a considerable variation of their properties, to changes in their environment. As result, these materials control the drug release into specific physiological compartments. Concerning on the environmental stimuli these include pH, temperature, light, chemicals, etc. The smart polymeric materials stimuli-responsive can be synthetics or naturals and have been used in the biotechnological, medicinal and engineering fields. The present chapter is aimed to focus the importance of this category of drug delivery systems and, in particular, it provides a summarizing overview of the range of smart polymeric materials and the drug delivery systems that exploit them.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Smart polymers
- Stimuli
- pH
- Light
- Electric field
- Magnetic field
- Ultrasound
- Temperature
- Ion
- Enzyme
- Glucose
- Hydrogels
- Nanotubes
- Films
- Membranes
- Nanoparticles
- Microparticles
- Micelles
- Biosensor
Introduction
Stimuli-Responsive (SR) materials, also called “smart materials” have been attracting great interest within scientific community in the last few decades [1–4]. They possess unique properties that have made this class of materials very promising for several applications in the field of nanoscience. In particular, the smart materials undergo changes in response to small external variations in environmental conditions or to physical or biochemical stimuli. In addition, there are dual SR materials that simultaneously respond to more than one stimulus [5–7]. For instance, temperature-sensitive polymers may also respond to pH changes [8–11].
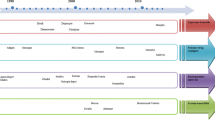
Many efforts have been carried out to find new solutions for developing SR materials for the reason that a smart response to external or internal stimuli allows a better localization of the system in the desired biological compartment and a controlled release of the loaded drug at the site interested from the pathological event. The polymers have proved interesting for the development of stimuli-responsive materials due to their chemistry that permits to modulate the properties by inserting sensible chemical moieties, a responsive compound. In this case, the polymer serves only as a carrier for that compound. On the other hand, SR systems containing polymers can be also designed with a responsive polymers. These systems strictly path the normal physiological process of the disease state where the amount of drug released is precious according to the physiological need [12]. Biopolymers such as proteins, carbohydrates and nucleic acid are all basic components of living organic systems that are responsible for the cells construction and process [13, 14]. These natural polymers have led to the development of numerous synthetic polymers that have been designed to simulate these biopolymers. In particular, a great range of polymeric materials has been synthesized to response to different stimuli such temperature, light, solvent, ionic strength, electric, magnetic, mechanical, pH, enzymes and receptors (Fig. 12.1) [15]. These polymers might recognize one or more of the listed stimuli as a signal, judge the magnitude of this signal, and then change their chain conformation in direct response [16].
The responses are manifested as variations in the shape, surface, solubility, degree of intermolecular association and others [17]. Particularly, the subsequent polymer structure and property alterations lead to the overall characteristic switching. The extraordinariness of these polymers lies not only in the fast structural macroscopic changes but also these transitions being reversible. Therefore, the polymer is capable of returning to its initial state as soon as the trigger is removed [13, 14]. There is a great deal of literature available about the different types of SR polymers. These materials have been utilized in the form of cross-linked hydrogels, reversible hydrogels, micelles, modified interfaces and conjugated solutions.
The main aim of this review is to highlight the various smart polymeric materials and the drug delivery systems (DDS) that exploit them. These are useful to realize biomimetic devices [18], intelligent medical instruments and auxiliaries [19, 20], electrochemical devices [21], artificial muscles [22], heat shrinkable materials for electronics packaging [23], microelectromechanical systems [24] and actuators and sensors [25].
Stimuli-Responsive Polymers
The stimuli that commonly drive the changes within SR polymers are represented in Fig. 12.1 [5–7]. These ones, commonly classified as physical, chemical, or biological, are also classified as both external or internal stimuli. The polymeric SR materials are too known as self-regulated devices, where the release rate is controlled by a mechanism of feedback that is produced within the body to control the structural changes in the polymer network and exhibit the desired drug release, without any external mediation [26, 27]. On the other hand, externally controlled SR polymers depend on the applied stimuli that are produced with the support of different stimuli-generating strategies, which results in pulsed drug delivery which may be defined as the rapid and transient release of a certain amount of drug within a short time period immediately after a predetermined off-release period [15, 26, 27].
Physically Dependent Stimuli
A very effective way of achieving site-specific drug targeting is by employing externally regulated polymeric drug delivery systems that are responsive to stimuli such as temperature, electric field, light, ultrasound, magnetic fields and mechanical deformation that typically modify the polymeric chain dynamics. Temperature is the most common stimulus for SR polymers.
Temperature-Responsive Polymers
Temperature-sensitive or thermo-responsive hydrogels and polymers have attracted great attention in different fields because some diseases manifest a temperature modification [28, 29]. In particular, they are characterized by a critical solution temperature (CST) around which the hydrophobic and hydrophilic interactions between the polymeric chains and the aqueous media brusquely change within a small temperature range bringing to the disruption of intra- and inter-molecular interactions and resulting in chain collapse or expansion. Typically, these polymer solutions possess a temperature above which one polymer phase exists, namely upper critical solution temperature (UCST), and below which a phase separation occurs with the formation of an hydrogel [30]. On the other hand, polymer solutions that appear as monophasic below a specific temperature and biphasic above it generally possess a so-called lower critical solution temperature (LCST). The LCST is a fascinating phenomenon found for various water soluble polymers which tend to phase-separate from solution upon heating. The most investigated temperature-responsive polymer featuring a LCST in water is the poly(N-isopropylacrylamide) (PNIPAAm). The LCST of PNIPAAm was of ~32 °C, proximate to the human body temperature. By altering the temperature of PNIPAAm solution in water, its solubility behavior can be reversibly changed from hydrophilic-soluble to hydrophobic-insoluble and thus can be induced externally (Fig. 12.2). Then, above the LCST, the polymer become increasingly insoluble leading to gel formation.
The phenomenon of transition from a solution to a gel is commonly referred to as sol-gel transition. The sol–gel transition of thermosensitive hydrogels can be experimentally verified by a number of techniques such as spectroscopy [31–33], differential scanning calorimetry (DSC) [31, 32] and rheology [33].
Other N-substituted polyacrylamides [34, 35], and further classes of polymers such as poly(oligoethyleneoxide-(meth)acrylate)s [36], poly(2-oxazoline)s [37], poly(N-vinylalkylamides), e.g. poly(N-vinylcaprolactam) (PNVC) [38], copolymers such as poly(L-lactic acid)-poly(ethylene glycol)-poly(L-lactic acid) (PLLA-PEG-PLLA) triblock copolymers [39], poly(ethylene oxide)-poly(propylene oxide)-poly(ethylene oxide) (PEO–PPO–PEO) copolymers [40] and many natural polymers [41], e.g. polysaccharides and proteins, utilize temperature change as the trigger that determines their gelling behavior without any additional external factor. Researchers have used them alone or in combination to fabricate thermally responsive hydrogels with desired properties. Hydrogels based on these polymers have been interesting for biomedical uses as they can swell in situ under physiological conditions and provide the advantage of convenient administration.
Temperature-Responsive Polymers Applications
The major advantage of thermosensitive polymeric systems is the avoidance of toxic organic solvents, the ability to transport both hydrophilic and lipophilic drugs, reduced systemic side effects, site-specific drug delivery, and sustained release properties.
Hydrogels responsive to temperature are being used to obtain an on–off drug release profile in response to a gradual change of temperature [42–44]. In particular, hydrogels PNIPAAm-BMA based loading indomethacin were analyzed for their on-off release profile. The on state was reached at low temperatures and off state at high temperatures. This behavior is due to the formation of scarcer permeable gel surface layer when the temperature was rapidly changed. This layer acts as barrier whose formation was regulated through the length of the methacrylate alkyl side-chain [45]. Thermo-responsive polymers were used also to overcome the limited therapeutic activity and insolubility of antitumoral drugs due to their toxicity and to the limited accessibility of tumors. The literature reports on the development of different systems (prodrugs, liposomes, micro- and nano-particles), such as anti-cancer drugs carriers, whose therapeutic efficacy is rather limited [46, 47]. Instead, soluble polymeric drug carriers have proved capable to increase drug tumor permeability [48] but, these carriers fail to intrinsically target a specific physiological compartment.
Thermo-responsive polymeric micelles can be used to target adriamycin at the tumoral site. In particular, block copolymers containing hydrophobic polymers, such as poly(butyl methacrylate) (PBMA) and end-functionalized PNIPAAm [49–51], forming a micellar structure in aqueous solution below the transition temperature of PNIPAAm, act as an inert material in the hydrated form. Upon 32 °C, the polymeric chains became hydrophobic due to their dehydration and aggregation and precipitation occur. The cores of micelles then acted as a reservoir for the hydrophobic drug adriamycin.
However, use of PNIPAAm is limited due to cytotoxicity attributed to the presence of quaternary ammonium in its structure, its non-biodegradability and its ability to activate platelets upon contact with body fluids. Many efforts have been made to decrease the initial burst drug release related to thermosensitive systems due to slow in vivo sol–gel transition. Literature data suggest that significant progress in release characteristics can be achieved by optimizing the chain-length ratio between hydrophilic and hydrophobic segments. A novel triblock polymeric system poly(ε-caprolactone)-block-poly(ethylene glycol)-block-poly(ε-caprolactone) (PCL-PEG-PCL) showed a noticeable decrease in initial burst release by coupling to a peptide. Moreover, in vitro drug release studies showed a wide-ranging sustained-release profile for over 1 month [52] (Fig. 12.3).
With the aim to develop a drug delivery system with thermal stimuli responsing, Nozawa et al. have been investigated liquid crystal (LC)-entrapped membranes, polymer alloyed membranes and LC-adsorbed membranes for the transport and release of indomethacin. Polymer alloyed membranes were obtained by polymerizing acrylic monomers in presence of LC and LC-adsorbed membrane were obtained by adsorbing LC into porous hydrophobic polymer membrane. Permeation experiments showed that below and above the gel-liquid crystal phase transition temperature of the LC, the extent of thermo-sensitivity for LC-adsorbed membranes was greater than that for the alloyed membrane. The permeability ratio was found to be about 120 with the LC-adsorbed membrane. The obtained results indicated that the LC-adsorbed membrane was an useful candidate as a thermo-responsive system for DDS [53].
Photo-Responsive Polymers
Photo-sensitive polymers have received increasingly attention in recent years because light can be applied straightaway and under specific conditions with high accuracy. The light is both negligibly absorbed by both cells and tissue and greatly consequently by the polymers. This makes light-responsive polymers highly advantageous for applications in wide fields including drug release, biomaterials, and artificial tissues [54–60]. Photo-responsive polymers contain light-sensitive chromophore moieties. Without external stimuli, these polymers can maintain their structures but, upon light absorption, these moieties can be broken from the polymer chain that can be degraded into smaller molecular fragments. Generally, light-sensitive polymers, UV or visible light sensitive, are classified into two main categories that are the side-chain-type polymer containing the chromophores as lateral groups of the chain backbone and the main-chain-type structure possessing single or multiple photosensitive species covalently- or non-covalently connected to the backbone. Chromophores groups are azobenzene [11, 61], spiropyran [62, 63] and nitrobenzyl [64, 65] and a variety of azobenzene or spiropyran-containing photo-responsive polymers such as poly(acrylamide) (PAA) [41, 66] and PNIPAAm [43, 44].
Photo-Responsive Polymer for the Drug Controlled Delivery
UV-sensitive hydrogels could be synthesized by introduction of bis(4-dimethylamino) phenylmethyl leucocyanide, a leuco derivative molecule, into the polymeric matrix in which ionization of the leuco derivative with UV radiation result in discontinuous swelling caused by an increase in osmotic pressure within the gel due to the appearance of cyanide ions formed by UV irradiation [67]. When the UV light is removed the hydrogels shrink. Instead, visible light–responsive materials can be prepared [68] by introducing trisodium salt of copper chlorophyllin, a light-sensitive chromophore, to PNIPAAm hydrogels. When the light is applied the chromophore absorbs it which is dissipated as heat giving an increase of the hydrogel temperature that alters its swelling promoting the drug release.
Polymeric vesicles obtained from the self-assembly of a photocleavable amphiphilic block copolymer, as a light-triggered DDS, were also investigated. The vesicles disintegrate upon UV irradiation, yielding small micellar-like structures, and simultaneously releasing their payload. The versatility of these system was tested both for low molecular weight molecules, proteins, enzymes and DNA. By varying the UV intensity, the loaded drug was released in a controlled manner [69].
Recently, spiropyran-initiated hyperbranched polyglycerol (SP-hb-PG) micelles were reported [70]. These carriers responded to UV/visible light and could dissociate due to conversion of the hydrophobic chromophore SP to zwitterionic and hydrophilic merocyanine (ME). In addition, chromophores such as coumarin, o-nitrobenzyl, stilbene, dithienylethene and 2-diazo-1,2-napthoquinone (DNQ) have been employed in light-responsive micelles, which can respond either to UV/visible or NIR irradiation to undergo structural or phase changes and trigger the drug release from micelles [71–74].
Ichimura and coworkers reported about photo-responsive polymer membranes based on poly(vinyl alcohol) derivatives having AZB side chains with different lengths, filled with liquid crystal (LC), and investigated the photo-response of LC alignment. They found that liquid crystal alignment changes were possible if the AZB unit was linked to poly(vinyl alcohol) backbone by a sufficient long spacer. The response times could be reduced by using high intensity sources [75]. Moreover, when visible light illuminates the surface of the membrane, the azobenzene moieties straighten out and the liquid crystals fall into line, which allows drug to easily flow through the holes. But when ultraviolet light illuminates the surface, the dye molecules bend into a new shape and the liquid crystals scatter into random orientations, clogging the tunnel and blocking drug from penetrating (Fig. 12.4).
Electro-Responsive Polymers
Increasing research and development efforts have been dedicated to the field of electro-responsive polymers (ERPs) due to their advantages of precise control via the magnitude of the current, the duration of an electrical pulse or the interval between pulses [76, 77]. ERPs are promising candidate materials for the design of drug delivery technologies, especially in conditions where an “on‐off” drug release mechanism is required. Typical ERPs are naturally occurring polymers such as chitosan, alginate and hyalouronic acid are commonly employed to prepare electro-responsive materials. Major synthetic polymers that have been used include polythiophene (PT) or sulphonated-polystyrene (PSS), polyaniline, polypyrrole, ethylene vinyl acetate, and polyethylene.
In some cases, combinations of natural and synthetic polymers have been used. Most polymers that exhibit electro-sensitive behavior are polyelectrolytes and they undergo deformation under an electric field due to anisotropic swelling or de-swelling as the charged ions move towards the cathode or anode (Fig. 12.5). Greatest stress is felt by the region surrounding the anode and smaller stress near the vicinity of the cathode. This stress gradient contributes to the anisotropic gel deformation under an electric field [78, 79].
Neutral polymers that exhibit electro-sensitive behaviour require the presence of a polarizable component with the ability to respond to the electric field. A rapid bending of gel in silicon oil was observed in the case of lightly cross-linked poly(dimethylsiloxane)-containing electrosensitive colloidal SiO2 particles.
These polymers can show swelling, shrinking or bending in response to an external field [80, 81] and they may be blended into responsive hydrogels in conjunction with the desired drug to obtain a patient‐controlled drug release system useful for neurotransmitters and vaccine delivery. The “on‐off” drug release mechanism can be achieved through the environmental‐responsive nature of the interpenetrating hydrogel‐EAP complex via (i) charged ions initiated diffusion of drug molecules due to an increase in osmotic pressure in the polymer; (ii) conformational changes that occur during redox switching of EAPs; or (iii) electro‐erosion. For example, when an electrochemical stimulus is applied to multilayer polyacrylamide films, the combined effects of H ions migrating to the region of the cathode and the electrostatic attraction between the anode surface and the negatively charged acrylic acid groups lead to shrinking of the film on the anode side [78, 82].
The hydrogel‐EAP composites include other implications such as, application towards biosensors, DNA hybridizations, micro‐surgical tools and miniature bioreactors and may be utilized to their full potential in the form of injectable devices as nanorobots or nano‐biosensors.
Electro-Responsive Polymeric Drug Delivery Systems
ERPs poly(2-acrylamido-2-methyl-propane sulphonic acid-co-n-butylmethacrylate) based were used to delivery edrophonium hydrochloride and hydro-cortisone in a pulsatile manner [83]. The control of drug release was achieved by varying the intensity of electric stimulation in distilled deionized water. For a positively charged drug, the release pattern depends on ion exchange between hydrogen ion produced by electrolysis of water and positively charged solute.
Liu and coworkers have been also designed and successfully realized a novel micro-electro-mechanical-systems (MEMS) based polymer drug delivery microsystem. The device consists of an array of metallic contacts, able to create an uniform electric field. In particular, a hydrogel polymer matrix loaded with hematoxylin dye, as model of hydrophilic drug, has been studied. The delivery microsystem operated at normal body temperature (37 °C) under an applied voltage of 20 V. The release rate and dose were accurately controlled. The polymer responds to the electrical stimulus by shrinking and releases the hematoxylin dye into solution. The release of hematoxylin in the media was monitored using ultraviolet-visible spectrophotometry.
Different drugs can be encapsulated within the hydrogel polymer matrix. The de-swelling of the polymer upon exposure to the applied electric field allows the encapsulated drug to be released from the matrix. The control of the applied voltage can be used to achieve pulsatile drug delivery. Alternatively, small volumes of drug may be continuously delivered to maintain the optimal therapeutic dose for the patient [84].
An electro-responsive drug delivery system was also developed using poly(acrylamide-grafted-xanthan gum) (PAAm-g-XG) hydrogel for transdermal delivery of ketoprofen [85]. When a swollen PAAm-g-XG hydrogel was placed between a pair of electrodes, a de-swelling of the hydrogel was observed in the vicinity of electrodes carrying the electric stimulus. Ketoprofen-loaded PAAm-g-XG hydrogel was also crosslinked with poly(vinyl alcohol) to prepare films as rate controlling membranes (RCM). The in vitro drug permeation study from the formulations was performed through excised rat abdominal skin. The drug permeation across the skin was significantly improved in the presence of electric stimulus as compared to passive diffusion and was found to be dependent upon the applied electric current strength and crosslink density of RCM. A pulsated pattern of drug release was observed as the electric stimulus was switched on and off. These PAAm-g-XG hydrogel could be useful as transdermal drug delivery systems actuated by an electric signal to provide on-demand release of drugs.
Pristine multi-walled carbon nanotubes (pMWNTs) were incorporated into a polymethacrylic acid (PMAA)-based hydrogel matrix by in situ radical polymerisation [86]. The effect of pMWNTs and cross-linker concentration on the electrical and mechanical properties of the hydrogel was carefully studied. The incorporation of pMWNTs into the polymeric network improved the electrical properties of the hydrogel. Moreover, the drug release from the gels was significantly enhanced at high pMWNT concentrations. But, the presence of pMWNTs within the hydrogel matrix affected the hydrogel mechanical properties by decreasing the pore size and, consequently, the swelling/de-swelling of the gels. The damage on the gel surfaces after electrical stimulation and the loss of the pulsatile release profile at high cross-linker concentrations, suggested that the mechanism of drug release involved a compacting effect and intensified the stress on the polymeric network as a result of the electrical properties of pMWNTs.
Ultrasonically Responsive Polymers
An innovative approach of exploiting ultrasound in drug delivery consists on the application of the ultrasonic waves directly at the polymers or the hydrogel matrix [87]. For the ultrasonically responsive polymers the release mechanism is driven by the cavitation. In particular, the ultrasound (ULS) energy generates both high and low pressure waves, resulting in an alternative growth and shrinkage of gas-filled microbubbles. These high-low pressure waves regulate the intermittent opening of the pores of the polymer, thus inducing the delivery of the respective drugs.
The most widely employed polymers for the ULS-responsive uses could be biodegradable or non-biodegradable. The biodegradables ones include polyglycolides, polylactides, bis(p-carboxyphen-oxy)alkane anhydride with sebacic acid. Instead, the non-biodegradables materials are ethylene-vinyl acetate copolymers or the poly(lactide-co-glycolide) microspheres, PHEMA hydrogels, the PEO-b-PPO-b-PEO micelles and the poly(HEMA-co-DMAEMA) hydrogels. The releasing agents are p-nitroaniline, p-amino-hippurate, bovine serum albumin and insulin. When exposed to ultrasound, these bioerodible polymers respond rapidly and reversibly. It is believed that the ultrasound also causes an increase in temperature in the delivery system, which allows the diffusion [88, 89]. ULS-responsive polymers are advantageous because they are non-invasive and capable of penetrating deep into the interior of the body and thus drug delivery can be focused and carefully controlled through a number of parameters including frequency, power density, duty cycles and time of application [90, 91]. Biologically the ultrasound action is related to the generation of thermal energy, perturbation of cell membranes, and enhanced permeability of blood capillaries [92].
Application of Ultrasound in Drug Delivery
Ultrasound responsive drug delivery systems have great potential for applications requiring stimulated release in vivo with a high degree of control over spatial and temporal location. Numerous carriers for ultrasonically enhanced drug delivery have been explored including polymeric ultrasound contrast agents with targeting potential, modified lipospheres and nano-/micro-bubble-enhanced chemotherapy (Fig. 12.6) [93–97]. Miyazaki and coworkers used ULS-responsive matrix ethylene-vinyl acetate copolymer (EVAc) based to achieve a 27-fold increase in the release of 5-fluoruracile. The ultrasound strength increase results in a proportional increase in the amount of 5-FU release [98]. EVAc systems were also tested as delivery device of insulin. It was found that exposure of insulin-loaded matrices to ULS significantly reduced the blood glucose levels on the rats [99].
Kwok et al. have reported on the development of ULS-sensitive barrier membranes able to give a pulsatile drug release. A co-polymer of 2-hydroxyl methacrylate (HEMA) and PEG dimethacrylate was loaded with particulate insulin and PEG, after which the surface of the polymer was coated with methylene chains. Upon exposure to ultrasound the methylene chains became disordered, permitting the insulin to diffuse out of the matrix [100, 101].
In addition, Suzuki et al. [102] achieved tumor-specific ultrasound-enhanced gene transfer with novel liposomal bubbles, which entrapped an ultrasound imaging gas. Extraordinarily, the bubble liposomes healthy transferred genes, only at the site of ultrasound exposure, into tumor cells and solid tumor tissue [103–106].
Magnetically-Responsive Polymers
The use of an oscillating magnetic field to modulate the rates of drug delivery from a polymeric matrix was one kind of method to achieve externally controlled drug delivery systems [107]. When planning a magnetic-responsive delivery system characteristics such as the magnetic properties of the carrier particles, field strength and geometry, drug/gene binding capacity and physiological parameters like the depth to target, the rate of blood flow, vascular supply and body weight need to be considered [108]. Magnetic targeting is based on the attraction of magnetic polymer carrier to an external magnetic field source that effectively traps it in the field at the target site and pulls it toward the magnet [109–113]. In order to maintain the magnetic loaded carrier at a specific location, the externally applied field must have a relatively strong gradient. When the drug is released from the magnetic matrix this is no longer responsive to the applied field.
Several carriers for magnetically enhanced drug delivery have been studied including nanoparticles with a magnetic core and a polymer shell and liposomes which have a magnetic core and an artificial liposomal shell are reported [114–119]. These materials may also be embedded in hydrogels which can carry drugs that are released upon heating [110, 120]. To produce highly efficient magnetic-responsive materials, the “doping” of polymer materials with magnetic nanoparticles (MNPs), made of inorganic matter (most often superparamagnetic iron oxide (SPION) Fe3O4 or g-Fe2O3, or ‘‘soft’’ metallic iron, but also “hard” magnetic materials e.g. Co, Ni, FeN, FePt, FePd…), appeared to be the more appealing and efficient solution.
Many hydrophilic polymers were proposed to host magnetic nanoparticles, in particular thermosensitive gels like poly(N-isopropylacrylamide) (PNIPAAm). However, PNIPAAm and other polymers exhibiting a LCST are generally less polar than polyvinyl acetate (PVA) and can be inefficient for trapping the MNPs due to the absence of hydrogen bonds and to a mesh size of typically 10–20 nm, i.e. larger than the size of superparamagnetic iron oxide MNPs (5–10 nm) [121]. To overcome this problems, strategies reported in the Fig. 12.7 concerning on a statistical copolymer network with a chelating comonomer such as 2-acetoacetoxyethylmethacrylate (AEMA) [122], a semi-interpenetrated network with alginate chains wrapping the MNPs [123] or a composite network of PNIPAAm and poly(ethylene glycol) (PEG) using PEG400–dimethacrylate as crosslinker of NIPAAm [124] were proposed.
Magnetic Field Responsive DDS
By merging magnetic and polymer materials one can obtain composites with exceptional magnetic responsive features. In this context, magnetic responsive micro- and nano-particles have been explored as possible drug carriers for site-specific drug targeting [125, 126]. Cytotoxic anti-cancer drugs were attached to these carriers and injected into the subject both via intravenous or intra-arterial injection. External magnetic fields generated by rare earth permanent magnets were then used to direct and concentrate the drug at the tumor site [127]. Subcutaneous implants of EVAc-insulin, capable of decreasing glucose levels in diabetic rats for 105 days, were also designed [128, 129]. Additionally, Hsieh et al. embedded magnetic steel beads in an EVAc copolymer matrix that was loaded with bovine serum albumin as a model of drug. They demonstrated increased rates of drug release in the presence of an oscillating magnetic field [117, 130]. During the exposure to the magnetic field, the beads oscillate within the matrix, on the other hand creating compressive and tensile forces. This consecutively acts as a pump to push an increased amount of the drug out of the polymer matrix. In vivo studies were later conducted demonstrating the effectiveness of an optimized version of this system, consisting of EVAc-protein matrices containing magnetic beads, loaded with insulin. In particular, was demonstrated that glucose levels can be repeatedly decreased on demand by applying an oscillating magnetic field [131–134].
Chemically Dependent Stimuli
Stimuli that occur internally are classified as chemical or biological. The pH as well as the ionic strength, redox and solvent are defined chemically-dependent stimuli.
pH-Responsive Polymers
pH sensitive polymers are materials which respond to the changes in the pH of the surrounding medium by varying their dimensions. Such materials increase its size (swell) or collapse depending on the pH of their environment (Fig. 12.8). This behavior is due to the presence of certain functional groups in the polymer chain such as acidic or basic groups that can either accept or release a proton in response to changes in environmental pH.
The pH is an important environmental parameter for biomedical applications, because pH changes occur in several specific or pathological sites.
The changes along the gastrointestinal tract from acidic in the stomach (pH = 2) to basic in the intestine (pH = 5–8) has to be considered for oral delivery of any kind of drug. Certain cancers as well inflamed or wound tissue exhibit a pH different from 7.4 as it is in circulation. For example chronic 7.4 and 5.4 and cancer tissue is also reported to be acidic extracellularly [135–137].
Consequently, this parameter can be exploited for a direct response at a certain tissue or in a cellular compartment. As mentioned above, pH responsive polymers consist of polymers with a large number of ionisable groups known as polyelectrolytes. Polyelectrolytes are classified into two types: weak polyacids and weak polybases. Weak polyacids accept protons at low pH and release protons at neutral and high pH [138]. Poly(acrylicacid)(PAAc) and poly(methacrylicacid) (PMAA) are commonly used pH-responsive polyacids [139, 140]. As the environmental pH changes, the pendant acidic group undergoes ionization at specific pH called as pKa. This rapid change in the charge of the attached group causes alteration in the molecular structure of the polymeric chain. This transition to expanded state is mediated by the osmotic pressure exerted by various stimuli responsible for controlling drug release from smart polymeric delivery systems [26–143]. pH responsive polymers typically include also chitosan [144], albumin [145], gelatin [146], poly(acrylic acid) (PAAc)/chitosan IPN [147], poly(methacrylic acid-g-ethylene glycol) [P(MAA-g-EG)] [148, 149], poly(ethylene imine) (PEI) [150], poly(N,N-diakylamino ethylmethacrylates) (PDAAEMA), and poly(lysine) (PL) [151, 152].
pH-Stimuli Sensitive Polymer DDS
Oral pH sensitive drug delivery systems are gaining importance because they are able to deliver the drug at specific part of the gastrointestine. Antibiotics, especially macrolide ones like erythromycin, enzymes and proteins are rapidly degraded by gastric juices. Others, such as acidic drugs like NSAID’s (e.g., diclofenac, valproic acid, or acetylsalicylic acid) cause a local irritation of the stomach mucosa.
Recently, it has been reported that aqueous dispersions or suspensions can be produced, in which the drug is present in enteric-coated form. The enteric-coated time clock system consists of a tablet core covered with a mixture of hydrophobic material and surfactant, which is applied as an aqueous dispersion [153]. The drug release from the core of the time clock system occurs after a pre-determined lag time.
Many polyanionic materials, such as PAAc, are pH sensitive and the degree of swelling of such polymers can be modulated by changing the pH. The use of these systems, too in conjunction with temperature-sensitive lipids, offers potential to target drugs to areas of inflammation or to achieve site-specific, pulsatile drug delivery [154].
Methylene-bis-acrylamide/methacrylic acid anionic microgels were also prepared by precipitation polymerization and loaded with doxorubicin and condensed by incubating in buffer at pH 5. The condensed particles were then coated with a lipid bilayer. Disruption of the lipid bilayer by electroporation was shown to cause the microgel particles to swell and release their drug. The concept of pH-sensitive liposomes emerged from the observation that certain enveloped viruses infect cells following acidification of the endosomal lumen to infect cells and from the knowledge that some pathological tissues (tumors inflamed and infected tissue) have a more acidic environment compared to normal tissues. Although, pH-sensitive liposomes are stable at physiological pH, they destabilize under acidic conditions, leading to the release of their aqueous contents [155]. The liposomes are internalized by endocytosis after binding to cell surface receptors. In addition, pH-sensitive hydrogels have been used in making biosensors and permeation switches [156]. The pH-sensitive hydrogels for these applications are usually loaded with enzymes that change the pH of the local microenvironment inside the hydrogels. One of the common enzymes used in pH-sensitive hydrogels is glucose oxidase which transforms glucose to gluconic acid. The formation of gluconic acid lowers the local pH, thus affecting the swelling of pH-sensitive hydrogels.
Soleimani and Sadeghi have reported on a super-absorbent hydrogel, starch-g-poly(sodium acrylate-co-2-hydroxy ethyl methacrylate) (starch-g—poly(NaAA-co-HEMA)) based, showing a maximum water absorbency in solutions with pH = 8. This hydrogel exhibited high sensitivity to pH, so that, several swelling changes of it or drug releasing percent were observed in lieu of pH variations in a wide range. Furthermore, the reversible swelling–deswelling behavior in solutions with acidic and basic pH, makes the hydrogels suitable candidates for controlled releasing systems [157].
Ion-Responsive Polymers and Their Applications
Ionic polymers were increasingly attractive for many applications in biotechnology and medicine such as drug delivery. They are also called polyelectrolytes, ionic gels, or ionic hydrogels. They dissociate, in a solution, into polyions and a larger number of oppositely charged counter ions. Interactions between polyions and counter ions cause phase transition, change in diffusivity and change in order of magnitude in equilibrium swelling. Typical examples of pH-sensitive polymers are PAAc, PMAA, poly(ethylene imine), and poly(N,N-dimethyl aminoethyl methacrylamide) [108–110, 158, 159].
Shahinpoor and Kim presented the fundamental properties and characteristics of ionic polymeric-metal composites (IPMCs) as biomimetic sensors, actuators and artificial muscle. The IPMC is composed of a perfluorinated ionic polymer layer, whose surfaces are coated by a conductive medium such as platinum. A strip of perfluorinated ionic membrane bends toward the anode under influence of an electric potential. The water containing counter ions moves toward anode creating a motion of the actuator [160]. Ion-exchange resins are frequently used for taste-masking, counterion-responsive drug release and sustained drug release. Polymers responding to ions in the saliva and gastrointestinal fluids are also used for controlled drug release in oral drug formulations.
Redox-Sensitive Polymers as DDS
Polymers containing labile groups present an advantageous opportunity to develop redox-responsive biodegradable systems. Disulfide linkages have been broadly applied in reduction-responsive polymeric drug delivery systems [117]. In fact, they are unstable in a reducing environment, being cleaved in favour of corresponding thiol groups [115, 116]. Polyanhydrides [111, 112], poly(lactic/glycolic acid) (PLGA) [113], and poly(b-amino esters) (PbAEs) [114], linking acid labile moieties, also induce redox responsiveness. PbAEs are useful to prepare efficient carriers for cytotoxic agents. On the other hand, poly(NiPAAm-co-Ru(bpy)3) finds application in the development of artificial muscles. Nguyen et al. reported on a polymeric nanogel useful to enhance the stability, redox responsiveness, and the efficacy for intracellular protein delivery. The thiolated heparin-Pluronic conjugate was self-assembled and oxidized to form a disulfide-crosslinked nanogel network under a diluted aqueous condition. The disulfide-crosslinked heparin-Pluronic (DHP) nanogels with encapsulated RNase A were characterized by in vitro release and cytotoxicity tests depending on the existence of glutathione (GSH). The DHP nanogels exhibited reduced hydrodynamic size, higher encapsulation degree, and augmentable release responding to the GSH concentration. The cytotoxicity data confirmed that DHP nanogels were more effective for the intracellular delivery of RNase A compared to non-crosslinked nanogel [161].
Moreover, novel redox-responsive polycationic hydrogels of N,N-diethylacrylamide (DEAAm) and 2-(dimethylamino)ethyl methacrylate (DMAEMA) were successfully synthesized by cross-linking reactions via quaternary ammonium compounds with a disulfide. The ability of the polymer network to enclose and release substances by reductive cleavage or oxidative formation of disulfide bonds was shown exemplarily using different dyes. The redox-responsive character was proven by oscillatory rheological measurements and different material properties of polycationic polymer discs were also investigated. Because of its polycationic structure, this polymeric system could be a promising compound for complexation of DNA-like substances. Its ability for selective release in reductive environments, like tumor tissues, could possibly be used in medical applications or in chemotherapy [162].
Biologically Responsive Polymers
Biologically dependent stimuli characteristically include analytes and biomacromolecules such as receptors, enzymes, glutathione, glucose, and metabolites that are over-produced in inflammation.
Glucose Responsive Polymers as Drug Carriers
Glucose sensitive materials are generated through the conjugation of glucose oxidase (GOx) to a smart pH-sensitive polymer. These systems are potentially useful for the insulin delivery [6, 119]. The incorporation of GOx may provide controlled access to the substrate and release of the product via disruption of the barrier function of the membrane. GOx oxidizes glucose to gluconic acid, which causes a pH change in the environment [4] and, subsequently, drastic changes in the polymer conformation. Hubbell et al. described glucose oxidase (GOx) encapsulated within PEG–PPS–PEG ((poly(ethylene glycol))–(poly(propylene sulfide))–PEG) polymersome [163]. This enzyme-loaded polymersome is permeable to glucose resulting in intravesicular formation of H2O2 upon generation of gluconic acid. Peroxide generation causes polymersome destabilization and particle destruction. This type of enzyme-amplified approach to particle degradation may have utility in drug delivery and the detection of biological analytes.
One of the most popular applications of glucose-sensitive polymers is the fabrication of insulin delivery systems for the treatment of diabetic patients. Delivering insulin is different from delivering other drugs, since insulin has to be delivered in an exact amount at the exact time of need. Many devices have been developed for this purpose and all of them have a glucose sensor built into the system. In a glucose-rich environment, such as the bloodstream after a meal, the oxidation of glucose to gluconic acid catalysed by glucose oxidase (GluOx) can lower the pH to approximately 5.8. This enzyme is probably the most widely used in glucose sensing, and makes possible to apply different types of pH-sensitive hydrogels for modulated insulin delivery [164].
Enzyme-Responsive Polymers
Among a range of external stimuli that have been utilized for the design of novel responsive polymers, enzymes have recently result to be a promising triggering subject. Enzyme-catalyzed reactions are highly selective and efficient toward specific substrates under mild conditions. They are involved in all biological and metabolic processes, serving as the main protagonists in the chemistry of living organisms at a molecular level. The integration of enzyme-catalyzed reactions with responsive polymers can additional increase the design of DDS characterized by high specificity and selectivity. In most enzyme-responsive polymer systems, enzymes are used to destroy the polymer or its assemblages. In addition, two other different types of systems, namely, enzyme-triggered self-assembly and aggregation of synthetic polymers and enzyme-triggered sol-to-gel and gel-to-sol transitions, are known (Fig. 12.9).
The major advantage of enzyme responsive polymers is that they do not require an external trigger. For instance, polymer systems based on alginate/chitosan or DEXS/chitosan microcapsules are responsive to chitosanase [126]. On the other hand, azo-aromatic bonds are sensitive to azo-reductase [127]. These enzymes, naturally produced by bacteria principally located in the colon, are capable of degrading polysaccharides like pectin, chitosan, amylase/amylopectin, cyclodextrin and dextrin [120, 125, 165].
Enzyme-Sensitive Polymers as DDS
Enzyme-sensitive polymers have been exploited frequently and they have become one of the most important branches of drug delivery systems. Zhang et al. prepared and synthesized mPEGylated peptide dendrimer–drug conjugate as a promising candidate of drug delivery system via a two-step click reaction with excellent characteristics and functionalities. A tetra-peptide sequence GFLG, which was cleavable in the presence of cathepsin B overexpressed in the tumor cells, was employed as linker to connect the anticancer drug DOX to mPEGylated peptide dendrimer. Due to the controlled release of drug and the particular nanoscale size, mPEGylated dendrimer–GFLG–DOX conjugate was found to have obviously improved in vivo antitumor efficacy over commercial DOX formulation at an equal dose, as well as low side effects, which were measured by changes in body weight and histological analysis. Overall, the structural design of mPEGylated peptide dendrimer–DOX conjugate-based nanoparticle in this study may provide useful strategy for design and preparation of peptide dendrimer as a safe and effective drug delivery system [166]. The use of enzyme catalysis as a tool to disassemble self-assembled hydrogels to control the release encapsulated drug provides a further opportunity to design a wide range of enzyme-specific low-molecular-weight hydrogelators (LMWGs). Vemula and coworkers reported a novel approach for controlled delivery of multiple drugs by an enzyme triggered hydrogel degradation mechanism. In particular, they described the synthesis of LMWGs (amphiphiles) from well-known drug acetaminophen (which is known as Tylenolreg), and their ability to self-assemble into nanoscale structures in aqueous solutions to form hydrogels that subsequently encapsulate a second drug such as curcumin which is a known chemopreventive hydrophobic drug. Upon enzyme triggered degradation, hydrogels showed single and double drug delivery at physiological conditions in vitro. After treating with prodrug amphiphiles, mesenchymal stem cells (MSCs) retain their stem cell properties such as maintaining their adhesive and proliferation capacities with high viability. This new platform approach will have prospective effect on hydrogel based drug delivery research through developing drug delivery vehicles from a wide range of prodrug-based gelators [167].
Inflammation-Responsive Smart Polymers
The inflammation is one of the manifestations of immune response and the chief immune components involved are the cells of the polymorphonuclear leukocytes (PML) [168]. These include the neutrophils, eosinophils and basophils. Apart from these, T-cell and B-cell lymphocytes and macrophages are also involved in the amplification of these signals. One of the hallmarks of inflammatory response is the generation of free radicals. Thus design of a system with bonds that will lyse due to the action of free radicals (for example, OH−, the hydroxyl radical) will enable release of drugs at the site of inflammation. Polyglycerolpolyglycidylether cross-linked with hyaluronic acid was used as the polymer matrix. The hyaluronic cross-links were degraded rapidly in the presence of hydroxyl radicals resulting in release of the drug. This system enables rapid release of drug at the site of inflammation [169].
Polymers with Dual Stimuli-Responsiveness
It is possible to obtain polymeric structures sensitive simultaneously to more than one stimulus. Particularly interesting are the dual temperature- and pH-responsive smart polymers that are attracting increasing attention recently for their advantages in biotechnological and biomedical applications. In this respect, Leung and coworkers have prepared smart core-shell microgels based on PNIPAAm, N,N′-methylenebisacrylamide (MBAAm) and chitosan or PEI in the absence of surfactants. The materials exhibited a well-defined core-shell structure consisting of temperature-sensitive cores, based on PNIPAAm, and pH-sensitive shells, made of cationic water-soluble polymers [170]. Moreover, Kuckling et al. prepared copolymers of NIPAAm with acrylamide derivatives bearing carboxylic groups attached to spacers with different chain length and studied the influence of both temperature and pH on their properties [171]. pH/temperature dual stimuli-responsive microcapsules have been prepared by incorporating carboxyl groups into PNIPAAm hydrogel shells by random copolymerization of NIPAAm and acrylic acid (AA) to endow the microcapsules with temperature responsiveness as well as pH responsiveness [172, 173]. The reversible change in hydrogen bonding between the two components NIPAAm and AA and water, and the ionization of carboxylate groups under different environmental condition resulted in the dual-stimuli response. Chitosan based PNIPAAm films having both thermal and pH sensitivity were prepared by combination of chitosan with PNIPAAm and PEG [174]. The resulting film had an LCST at around 32 °C, due to the presence of PNIPAAm, and showed pH responsiveness due to the amino groups of chitosan component. Poly(vinylidene fluoride) (PVDF) hydrophobic films grafted with PAA demonstrated convective permeability that changed significantly with the pH and/or the salt concentration of the surrounding fluids [175].
Nanogels with pH and temperature dual stimuli-responsive properties characterized by interpenetrating polymer network (IPN) structure, based on PNIPAAm and PAAc, were also synthesized by in situ polymerization of acrylic acid and N,N-methylenebisacrylamide (Fig. 12.10). These IPN nanogels have the advantage of less mutual interference between the temperature-responsive and pH-responsive components, which is beneficial for their applications in controlled drug release and sensors [176].
Abbreviations
- AA:
-
Acrylic acid
- AEMA:
-
2-acetoacetoxyethylmethacrylate
- BMA:
-
Butyl methacrylate
- CST:
-
Critical solution temperature
- DDS:
-
Drug delivery systems
- DEAAm:
-
N,N-diethylacrylamide
- DHP:
-
Disulfide-crosslinked heparin-Pluronic
- DMAEMA:
-
2-(dimethylamino)ethyl methacrylate
- DNQ:
-
2-diazo-1,2-napthoquinone
- DSC:
-
Differential scanning calorimetry
- EVAc:
-
Ethylene-vinyl acetate copolymer
- 5-FU:
-
5-fluoruracil
- GSH:
-
Glutathione
- HA:
-
Hyaluronic acid
- IPN:
-
Interpenetrating polymer network
- IPMCs:
-
Ionic polymeric-metal composites
- LC:
-
Liquid crystal
- LCST:
-
Lower critical solution temperature
- LMWGs:
-
Low-molecular-weight hydrogelators
- MBAAm:
-
N,N′-methylenebisacrylamide
- ME:
-
Merocyanine
- MEMS:
-
Micro-electro-mechanical-systems
- MNPs:
-
Magnetic nanoparticles
- MSCs:
-
Mesenchymal stem cells
- NIPAAm:
-
N-isopropylacrylamide
- PAA:
-
Poly(acrylamide)
- PAAc:
-
Poly(acrylicacid)
- PAAm-g-XG:
-
Poly(acrylamide-grafted-xanthan gum)
- PbAEs:
-
Poly(b-amino esters)
- PCL-PEG-PCL:
-
Poly(ε-caprolactone)-block-poly(ethyleneglycol)-block-poly(ε-caprolactone)
- PDAAEMA:
-
Poly(N,N-diakylamino ethylmethacrylates)
- PEG:
-
Poly(ethylene glycol)
- PEG–PPS–PEG:
-
((Poly(ethylene glycol))–(poly(propylene sulfide))–PEG)
- PEI:
-
Poly(ethylene imine)
- PEO–PPO–PEO:
-
Poly(ethylene oxide)-poly(propylene oxide)-poly(ethylene oxide)
- PL:
-
Poly(lysine)
- PLGA:
-
Poly(lactic/glycolic acid)
- PLLA-PEG-PLLA:
-
Poly(L-lactic acid)-poly(ethylene glycol)-poly(L-lactic acid)
- PMAA:
-
Polymethacrylic acid
- pMWNTs:
-
Pristine multi-walled carbon nanotubes
- PNIPAAm:
-
Poly(N-isopropylacrylamide)
- PNVC:
-
Poly(N-vinylcaprolactam)
- PSS:
-
Sulphonated-polystyrene
- PT:
-
Polythiophene
- PVA:
-
Polyvinyl acetate
- PVDF:
-
Poly(vinylidene fluoride)
- RCM:
-
Rate controlling membranes
- UCST:
-
Upper critical solution temperature
- SP-hb-PG:
-
Spiropyran-initiated hyperbranched polyglycerol
- SPION:
-
Superparamagnetic iron oxide
- SR:
-
Stimuli-responsive
- ULS:
-
Ultrasound
References
Galaev, I.Y., Mattiasson, B.: Smart polymers and what they could do in biotechnology and medicine. Trends Biotechnol. 17, 335–340 (1999)
Hoffman, A.S., Stayton, P.S., Bulmus, V., Chen, G., Chen, J., Cheung, C., Chilkoti, A., Ding, Z., Dong, L., Fong, R., Lackey, C.A., Long, C.J., Miura, M., Morris, J.E., Murthy, N., Nabeshima, Y., Park, T.G., Press, O.W., Shimoboji, T., Shoemaker, S., Yang, H.J., Monji, N., Nowinski, R.C., Cole, C.A., Priest, J.H., Harris, J.M., Nakamae, K., Nishino, T., Miyata, T.: Founder’s award, society for biomaterials. Sixth world biomaterials congress 2000, Kamuela, HI, May 15–20, 2000. Really smart bioconjugates of smart polymers and receptor proteins. J. Biomed. Mater. Res. 52, 577–586 (2000)
Kikuchi, A., Okano, T.: Intelligent thermoresponsive polymeric stationary phases for aqueous chromatography of biological compounds. Prog. Polym. Sci. 27, 1165–1193 (2002)
Qiu, Y., Park, K.: Environment-sensitive hydrogels for drug delivery. Adv. Drug Deliv. Rev. 53, 321–339 (2001)
Jeong, B., Gutowska, A.: Lessons from nature: stimuli-responsive polymers and their biomedical applications. Trends Biotechnol. 20, 305–311 (2002)
Gil, E.S., Hudson, S.M.: Stimuli-responsive polymers and their bioconjugates. Prog. Polym. Sci. 29, 1173–1222 (2004)
Delcea, M., Möhwald, H., Skirtach, A.G.: Stimuli-responsive LbL capsules and nanoshells for drug delivery. Adv. Drug Deliv. Rev. 63, 730–747 (2011)
Pinkrah, V.T., Snowden, M.J., Mitchell, J.C., Seidel, J., Chowdhry, B.Z., Fern, G.R.: Physicochemical properties of poly(N-isopropylacrylamide-co-4-vinylpyridine) cationic polyelectrolyte colloidal microgels. Langmuir 19, 585–590 (2003)
Peng, T., Cheng, Y.L.: PNIPAAm and PMAA co-grafted porous PE membranes: living radical co-grafting mechanism and multi-stimuli responsive permeability. Polymer 41, 2091–2100 (2001)
Bignotti, F., Penco, M., Sartore, L., Peroni, I., Mendichi, R., Casolaro, M., D’Amore, A.: Synthesis, characterisation and solution behaviour of thermo- and pH-responsive polymers bearing L-leucine residues in the side chains. Polymer 41, 8247–8256 (2000)
Gan, L.H., Gan, Y.Y., Deen, G.R.: Poly(N-acryloyl-N’-propylpiperazine): a new stimuli-responsive polymer. Macromolecules 33, 7893–7897 (2000)
Kost, J., Langer, R.: Responsive polymeric delivery systems. Adv. Drug Deliv. Rev. 46, 125–148 (2001)
Galaev, I.Y., Mattiasson, B.: Smart polymers and what they could do in biotechnology and medicine. Trends Biotechnol. 17, 335–340 (1999)
Kumar, A., Srivastava, A., Galaev, I.Y., Mattiasson, B.: Smart polymers: physical forms and bioengineering applications. Prog. Polym. Sci. 32, 1205–1237 (2007)
Liechty, W.B., Kryscio, D.R., Slaughter, B.V., Peppas, N.A.: Polymers for drug delivery systems. Annu. Rev. Chem. Biomol. Eng. 1, 149–173 (2010)
Okano, T.: Biorelated Polymers and Gels-Controlled Release and Applications in Biomedical Engineering. Academic Press, San Diago (1998)
Roy, D., Cambre, J.N., Sumerlin, B.S.: Future perspectives and recent advances in stimuli responsive materials. Prog. Polym. Sci. 35, 278–301 (2010)
Xie, T., Xiao, X.: Self-peeling reversible dry adhesive. Chem. Mater. 20, 2866–2868 (2008)
Lendlein, A., Langer, R.: Biodegradable, elastic shapememory polymers for potential biomedical applications. Science 96, 1673–1676 (2002)
Zhu, Y., Hu, J., Yeung, K.: Effect of soft segment crystallization and hard segment physical crosslink on shape memory function in antibacterial segmented polyurethane ionomers. Acta Biomater. 5, 3346–3357 (2009)
Scrosati, B.: Application of Electroactive Polymers. Chapman and Hall, London (1993)
Bar-Cohen, Y.: Proceedings of the 42nd AIAA Structures, Structural Dynamics, and Materials Conference (SDM), Gossamer Spacecraft Forum (GSF), Seattle (2001)
Charlesby, A.: Atomic Radiation and Polymers. Pergamon, New York (1960)
Gall, K., Dunn, M.L., Liu, Y., Finch, D., Lake, M., Munshi, N.A.: Shape memory polymer nanocomposites. Acta Mater. 50, 5115–5126 (2002)
Bar-Cohen, Y., Zhang, Q.: Electroactive polymer actuators and sensors. MRS Bull. 33, 173–181 (2008)
Gupta, P., Vermani, K., Garg, S.: Hydrogels: from controlled release to pH-responsive drug delivery. Drug Discov. Today 7, 569–579 (2002)
Kikuchi, A., Okano, T.: Pulsatile drug release control using hydrogels. Adv. Drug Deliv. Rev. 54, 53–77 (2002)
Nakayama, M., Okano, T., Miyazaki, T., Kohori, F., Sakai, K., Yokoyama, M.: Molecular design of biodegradable polymeric micelles for temperature-responsive drug release. J. Control Release 115, 46–56 (2006)
Bromberg, L.E., Ron, E.S.: Temperature-responsive gels and thermogelling polymer matrices for protein and peptide delivery. Adv. Drug Deliv. Rev. 31, 197–221 (1998)
Peppas, N.A., Bures, P., Leobandung, W., Ichikawa, H.: Hydrogels in pharmaceutical formulations. Eur. J. Pharm. Biopharm. 50, 27–46 (2000)
Li, L., Shan, H., Yue, C.Y., Lam, Y.C., Tam, K.C., Hu, X.: Thermally induced association and dissociation of methyl cellulose in aqueous solutions. Langmuir 18, 7291–7298 (2002)
Liu, Y.Y., Shao, Y.H., Lü, J.: Preparation properties and controlled release behaviors of pH-induced thermosensitive amphiphilic gels. Biomaterials 27, 4016–4024 (2006)
Yin, X., Hoffman, A.S., Stayton, P.S.: Poly(N-isopropylacrylamide-co-propylacrylic acid) copolymers that respond sharply to temperature and pH. Biomacromolecules 7, 1381–1385 (2006)
Kabanov, A.V., Batrakova, E.V., Alakhov, V.Y.: Pluronic block copolymers as novel polymer therapeutics for drug and gene delivery. J. Control Release 82, 189–212 (2002)
Joly-Duhamel, C., Hellio, D., Djabourov, M.: All gelatin networks: 1. Biodiversity and physical chemistry. Langmuir 18, 7208–7217 (2002)
Lutz, J.F.: Polymerization of oligo(ethylene glycol) (meth)acrylates: toward new generations of smart biocompatible materials. J. Polym. Sci. A Polym. Chem. 46, 3459–3470 (2008)
Hoogenboom, R.: Poly(2-oxazoline)s: a polymer class with numerous potential applications. Angew. Chem. Int. Ed. 48, 7978–7994 (2009)
Jeong, B., Kim, S.W., Bae, Y.H.: Thermosensitive sol-gel reversible hydrogels. Adv. Drug Deliv. Rev. 54, 37–51 (2002)
Ruel-Gariepy, E., Leroux, J.C.: In situ forming hydrogels-review of temperature-sensitive systems. Eur. J. Pharm. Biopharm. 58, 409–426 (2004)
Takahashi, M., Shimazaki, M., Yamamoto, J.: Thermoreversible gelation and phase separation in aqueous methyl cellulose solutions. J. Polym. Sci. B 39, 91–100 (2001)
Klouda, L., Mikos, A.G.: Thermoresponsive hydrogels in biomedical applications. Eur. J. Pharm. Biopharm. 68, 34–45 (2008)
Bae, Y.H., Okano, T., Kim, S.W.: ‘‘On-off’’ thermocontrol of solute transport. II. Solute release from thermosensitive hydrogels. Pharm. Res. 8, 531–537 (1991)
Bae, Y.H., Okano, T., Kim, S.W.: On-off thermocontrol of solute transport. II solute release from thermosensitive hydrogels. Pharm. Res. 8, 624–628 (1991)
Okano, T., Bae, Y.H., Jacobs, H., Kim, S.W.: Thermally on-off switching polymers for drug permeation and release. J. Controlled Release 11, 255–265 (1990)
Okuyama, Y., Yoshida, R., Sakai, K., Okano, T., Sakurai, Y.: Swelling controlled zero-order and sigmoidal drug release from thermoresponsive poly(N-isopropylacrylamide-co-butyl methacrylate) hydrogel. J. Biomater. Sci. Polym. 4, 545–556 (1993)
Jones, M., Leroux, J.: Polymeric micelles: a new generation of colloidal drug carriers. Eur. J. Pharm. Biopharm. 48, 101–111 (1999)
Allen, T.M.: Liposomal drug formulations, rationale for development and what we can. Expect for the future. Drugs 56, 747–756 (1998)
Matsumura, Y., Maeda, H.: A new concept for macromolecular therapeutics in cancer chemotherapy: mechanism of tumoritropic accumulation of proteins and the antitumor agent smancs. Cancer Res. 46, 6387–6392 (1986)
Chung, J.E., Yokoyama, M., Yamato, M., Aoyagi, T., Sakurai, Y., Okano, T.: Thermo-responsive drug delivery from polymeric micelles constructed using block copolymers of poly(N-isopropylacrylamide) and poly(butylmethacrylate). J. Controlled Release 62, 115–127 (1999)
Chung, E., Yokoyama, M., Okano, T.: Inner core segment design for drug delivery control of thermo- responsive polymeric micelles. J. Controlled Release 65, 93–103 (2000)
Kohori, F., Sakai, K., Aoyagi, T., Yokoyama, M., Yamato, M., Sakurai, Y., Okano, T.: Control of adriamycin cytotoxic activity using thermally. Colloids Surf. B 16, 195–205 (1999)
Honey, P.J., Rijo, J., Anju, A., Anoop, K.R.: Smart polymers for the controlled delivery of drugs—a concise overview. Acta Pharm. Sinica B 4, 120–127 (2014)
Nozawa, I., Suzuki, Y., Sato, S., Sugibayashi, K., Morimoto, Y.: Preparation of thermo-responsive polymer membranes. I. J. Biomed. Mat. Res. 25, 243–254 (1991)
Yan, B., Boyer, J.C., Habault, D., Branda, N.R., Zhao, Y.: Near infrared light triggered release of biomacromolecules from hydrogels loaded with upconversion nanoparticles. J. Am. Chem. Soc. 134, 16558–16561 (2012)
Fomina, N., McFearin, C.L., Sermsakdi, M., Morachis, J.M., Alrnutairi, A.: Low power, biologically benign NIR light triggers polymer disassembly. Macromolecules 44, 8590–8597 (2011)
Habault, D., Zhang, H.J., Zhao, Y.: Light-triggered self-healing and shape-memory polymers. Chem. Soc. Rev. 42, 7244–7256 (2013)
Yan, Y., Yue, L., Li, H.L., Maher, E., Li, Y.G., Wang, Y.Z., Wu, L.X., Yarn, V.W.W.: Photo-responsive self-assembly of an azobenzene-ended surfactant-encapsulated polyoxometalate complex for modulating catalytic reactions. Small 8, 3105–3110 (2012)
Schumers, J.M., Fustin, C.A., Gohy, J.F.: Light-responsive block copolymers. Macromol. Rapid Commun. 31, 1588–1607 (2010)
Gilot, J., Abbel, R., Lakhwani, G., Meijer, E.W., Schenning, A.P.H.J., Meskers, S.C.J.: Polymer photovoltaic cells sensitive to the circular polarization of light. Adv. Mater. 22, E131–E134 (2010)
Narayan, K.S., Kumar, N.: Light responsive polymer field-effect transistor. Appl. Phys. Lett. 79, 1891–1893 (2001)
Gan, L.H., Roshan, G., Loh, X.J., Gan, Y.Y.: New stimuli responsive copolymers of N-acryloyl-N′-alkyl piperazine and methyl methacrylate and their hydrogels. Polymer 42, 65–69 (2001)
Aoyagi, T., Ebara, M., Sakai, K., Sakurai, Y., Okano, T.: Novel bifunctional polymer with reactivity and temperature sensitivity. J. Biomater. Sci. Polym. 11, 101–110 (2000)
Aoki, T., Muramatsu, M., Nishina, A., Sanui, K., Ogata, N.: Thermosensitivities of optically active hydrogels constructed with N-(L)-(1-hydroxymethyl)propylmethacrylamide. Macromol. Biosci. 4, 943–949 (2004)
Bae, Y.H., Okano, T., Kim, S.W.: Insulin permeation through thermosensitive hydrogels. J. Controlled Release 9, 271–279 (1989)
Hirokawa, Y., Tanaka, T.: Volume phase transition in a nonionic gel. J. Chem. Phys. 81, 6379–6380 (1984)
Bae, Y.H., Okano, T., Kim, S.W.: A new thermo-sensitive hydrogel: Interpenetrating polymer networks from N-acryloylpyrrolidine and poly(oxyethylene). Makromol. Chem. Rapid Commun. 9, 185–189 (1988)
Mamada, A., Tanaka, T., Kungwachakun, D., Irie, M.: Photoinduced phase transition of gels. Macromolecules 23, 1517–1519 (1990)
Suzuki, A., Tanaka, T.: Phase transition in polymer gels induced by visible light. Nature 346, 345–347 (1990)
Cabane, E., Malinova, V., Menon, S., Palivan, C.G., Meier, W.: Photoresponsive polymersomes as smart, triggerable nanocarriers. Soft Matter 7, 9167–9176 (2011)
Son, S., Shin, E., Kim, B.S.: Light-responsive micelles of spiropyran initiated hyperbranched polyglycerol for smart drug delivery. Biomacromolecules 15, 628–634 (2014)
Chen, Z., He, Y., Wang, Y., Wang, X.: Amphiphilic diblock copolymer with dithienylethene pendants: synthesis and photo-modulated self-assembly. Macromol. Rapid Commun. 32, 977–982 (2011)
Jin, Q., Mitschang, F., Agarwal, S.: Biocompatible drug delivery system for photo-triggered controlled release of 5-fluorouracil. Biomacromolecules 12, 3684–3691 (2011)
Menon, S., Thekkayil, R., Varghes, S., Das, S.: Photoresponsive soft materials: synthesis and photophysical studies of a stilbene-based diblock copolymer synthesis and photophysical studies of a stilbene-based diblock copolymer. J. Polym. Sci. A Polym. Chem. 49, 5063–5073 (2011)
Liu, G.Y., Chen, C.J., Li, D.D., Wang, S.S., Ji, J.: Near-infrared light-sensitive micelles for enhanced intracellular drug delivery. J. Mater. Chem. 22, 16865–16871 (2012)
Seki, T., Fukuda, R., Tamakia, T., Ichimura, K.: Alignment photoregulation of liquid crystals on precisely area controlled azobenzene langmuir-blodgett monolayers. Thin Solid Films 243, 675–678 (1994)
Anal, A.K.: Stimuli-induced pulsatile or triggered release delivery systems for bioactive compounds. Recent Pat. Endocr. Metab. Immune Drug Discov. 1, 83–90 (2007)
Mendes, P.M.: Stimuli-responsive surfaces for bio-applications. Chem. Soc. Rev. 37, 2512–2529 (2008)
Gong, J.P., Nitta, T., Osada, Y.: Electrokinetic modeling of the contractile phenomena of polyelectrolyte gels-one dimensional capillary model. J. Phys. Chem. 98, 9583–9587 (1994)
Kwon, I.C., Bae, Y.H., Okano, T., Kim, S.W.: Drug release from electric current sensitive polymers. J. Controlled Release 17, 149–153 (1991)
Jones, D.P., Carlson, J.L., Samiec, P.S., Sternberg Jr, P., Mody Jr, V.C., Reed, R.L., Brown, L.A.S.: Glutathione measurement in human plasma. Clin. Chim. Acta 275, 175–184 (1998)
Koo, A.N., Lee, H.J., Kim, S.E., Chang, J.H., Park, C., Kim, C., Park, J.H., Lee, S.C.: Disulfide-cross-linked PEG-poly(amino acid)s copolymer micelles for glutathione-mediated intracellular drug delivery. Chem. Commun. 44, 6570–6572 (2008)
Tanaka, T., Nishio, I., Sun, S.-T., Ueno-Nishio, S.: Collapse of gels in an electric. Field. Sci. 218, 467–469 (1982)
Zhao, X., Kim, J., Cezar, C.A., Huebsch, N., Lee, K., Bouhadir, K., Mooney, D.J.: Active scaffolds for on-demand drug and cell delivery. Proc. Natl. Acad. Sci. U.S.A. 108, 67–72 (2011)
Liu, Y., Servant, A., Guy, O.J., Al-Jamal, K.T., Williams, P.R., Hawkins, K.M., Kostarelos, K.: Liposomes: from a clinically established drug delivery system to a nanoparticle platform for theranostic nanomedicine. Proc. Eng. 25, 984–987 (2011)
Kulkarni, R.V., Sa, B.: Electroresponsive polyacrylamide-grafted-xanthan hydrogels for drug delivery. J. Bioact. Compat. Polym. 24, 368–384 (2009)
Servant, A., Bussy, C., Al-Jamal, K., Kostarelos, K.: Design, engineering and structural integrity of electro-responsive carbon nanotube-poly(methyl) methacrylate hydrogels for pulsatile drug release. J. Mater. Chem. B 1, 4593–4600 (2013)
Haba, Y., Harada, A., Takagishi, T., Kono, K.: Rendering poly(amidoamine) or poly(propylenimine) dendrimers temperature sensitive. J. Am. Chem. Soc. 126, 12760–12761 (2004)
Burakowska, E., Zimmerman, S.C., Haag, R.: Photoresponsive crosslinked hyperbranched polyglycerols as smart nanocarriers for guest binding and controlled release. Small 5, 2199–2204 (2009)
Chen, W., Tomalia, D.A., Thomas, J.L.: Unusual pH-dependent polarity changes in PAMAM dendrimers: evidence for pH-responsive conformational changes. Macromolecules 33, 9169–9172 (2000)
Liu, Y., Bryantsev, V.S., Diallo, M.S., Goddard III, W.A.: PAMAM dendrimers undergo pH responsive conformational changes without swelling. J. Am. Chem. Soc. 131, 2798–2799 (2009)
Yang, K., Weng, L., Cheng, Y., Zhang, H., Zhang, J., Wu, Q., Xu, T.: Host-guest chemistry of dendrimer-drug complexes. 6. Fully acetylated dendrimers as biocompatible drug vehicles using dexamethasone 21-phosphate as a model drug. J. Phys. Chem. B 115, 2185–2195 (2011)
Kohman, R.E., Zimmerman, S.C.: Degradable dendrimers divergently synthesized via click chemistry. Chem. Commun. 45, 794–796 (2009)
Cho, S.Y., Allcock, H.R.: Dendrimers derived from polyphosphazene poly(propyleneimine) systems: encapsulation and triggered release of hydrophobic guest molecules. Macromolecules 40, 3115–3121 (2007)
Kostiainen, M.A., Rosilo, H.: Low-molecular-weight dendrons for DNA binding and release by reduction-triggered degradation of multivalent interactions. Chem. Eur. J. 15, 5656–5660 (2009)
Ong, W., McCarley, R.L.: Chemically and electrochemically mediated release of dendrimer end groups. Macromolecules 39, 7295–7301 (2006)
Azagarsamy, M.A., Sokkalingam, P., Thayumanavan, S.: Enzyme triggered disassembly of dendrimer-based amphiphilic nanocontainers. J. Am. Chem. Soc. 131, 14184–14185 (2009)
Azagarsamy, M.A., Yesilyurt, V., Thayumanavan, S.: Disassembly of dendritic micellar containers due to protein binding. J. Am. Chem. Soc. 132, 4550–4551 (2010)
Miyazaki, S., Hou, W.M., Takada, M.: Controlled drug release by ultrasound irradiation. Chem. Pharm. Bull. 33, 428–431 (1985)
Miyazaki, S., Yokouchi, C., Takada, M.: External control of drug release: controlled release of insulin from a hydrophilic polymer implants by ultrasound irradiation in diabetic rats. J. Pharm. Pharmacol. 40, 716–717 (1988)
Kwok, C.S., Mourad, P.D., Crum, L.A., Ratner, B.D.: Surface modification of polymers with self-assembled molecular structure: multitechnique surface characterization. Biomacromolecules 1, 139–148 (2000)
Kwok, C.S., Mourad, P.D., Crum, L.A., Ratner, B.D.: Self-assembled molecular structures as ultrasonically‐responsive barrier membranes for pulsatile drug delivery. J. Biomed. Mater. Res. 57, 151–164 (2001)
Kotharangannagari, V.K., Sánchez-Ferrer, A., Ruokolainen, J., Mezzenga, R.: Photoresponsive reversible aggregation and dissolution of rod-coil polypeptide diblock copolymers. Macromolecules 44, 4569–4573 (2011)
Koyama, T., Hatano, K., Matsuoka, K., Esumi, Y., Terunuma, D.: Synthesis and characterization of photo-responsive carbosilane dendrimers. Molecules 14, 2226–2234 (2009)
Chan, Y., Wong, T., Byrne, F., Kavallaris, M., Bulmus, V.: Acid-labile core cross-linked micelles for pH-triggered release of antitumor drugs. Biomacromolecules 9, 1826–1836 (2008)
Katayama, Y., Sonoda, T., Maeda, M.: A polymer micelle responding to the protein kinase A signal. Macromolecules 34, 8569–8573 (2001)
Gonzalez, D.C., Savariar, E.N., Thayumanavan, S.: Fluorescence patterns from supramolecular polymer assembly and disassembly for sensing metallo- and nonmetalloproteins. J. Am. Chem. Soc. 131, 7708–7716 (2009)
Hsieh, D.S.T., Langer, R., Folkman, J.: Magnetic modulation of release of macromolecules from polymers. Proc. Natl. Acad. Sci. U.S.A. 78, 1863–1867 (1981)
Salamone, J.C., Rodriguez, E.L., Lin, K.C., Quach, L., Watterson, A.C., Ahmed, I.: Aqueous salt absorption by ampholytic polysaccharides. Polymer 26, 1234–1238 (1985)
Morrison, M.E., Dorfman, R.C., Clendening, W.D., Kiserow, D.J., Rossky, P.J., Webber, S.E.: Quenching kinetics of anthracene covalently bound to a polyelectrolyte. 1. Effects of ionic strength. J. Phys. Chem. 98, 5534–5540 (1994)
Szczubiałka, K., Jankowska, M., Nowakowska, M.: Smart polymeric nanospheres as new materials for possible biomedical applications. J. Mater. Sci. Mater. Med. 14, 699–703 (2003)
Leong, K.W., Brott, B.C., Langer, R.: Bioerodible polyanhydrides as drug carrier matrices I: characterization, degradation and release characteristics. J. Biomed. Mater. Res. 19, 941–955 (1985)
Mathiowitz, E., Jacob, J.S., Jong, Y.S., Carino, G.P., Chickering, D.E., Chaturvedi, P., Santos, C.A., Vijayaraghavan, K., Montgomery, S., Bassett, M., Morrell, C.: Biologically erodable microspheres as potential oral drug delivery systems. Nature 386, 410–414 (1997)
Cohen, S., Yoshioka, T., Lucarelli, M., Hwang, L.H., Langer, R.: Controlled delivery systems for proteins basedon poly(lactic/glycolic acid) microspheres. Pharm. Res. 8, 713–720 (1991)
Shenoy, D., Little, S., Langer, R., Amiji, M.: Poly(ethylene oxide)-modiWed poly(beta-amino ester) nanoparticles as a pH-sensitive system for tumor-targeted delivery of hydrophobic drugs: part 3. Therapeutic efficacy and safety studies in ovarian cancer xenograft model. Pharm. Res. 22, 2107–2114 (2005)
Cerritelli, S., Velluto, D., Hubbell, J.A.: PEG-SS-PPS: reduction-sensitive disulfide block copolymer vesicles for intracellular drug delivery. Biomacromolecules 8, 1966–1972 (2007)
Oh, J.K., Siegwart, D.J., Lee, H.-I., Sherwood, G., Peteanu, L., Hollinger, J.O., Kataoka, K., Matyjaszewski, K.: Biodegradable nanogels prepared by atom transfer radical polymerization as potential drug delivery carriers: synthesis, biodegradation, in vitro release, and bioconjugation. J. Am. Chem. Soc. 129, 5939–5945 (2007)
Matsumoto, S., Christie, R.J., Nishiyama, N., Miyata, K., Ishii, A., Oba, M., Koyama, H., Yamasaki, Y., Kataoka, K.: Environment-responsive block copolymer micelles with a disulfide cross-linked core for enhanced siRNA delivery. Biomacromolecules 10, 119–127 (2008)
Yoshida, R., Yamaguchi, T., Kokufuta, E.: Self-oscillation of polymer gels coupled with the Belousov–Zhabotinsky reaction. J. Artif. Organs. 2, 135–140 (1999)
Chaterji, S., Kwon, I.K., Park, K.: Smart polymeric gels. Prog. Polym. Sci. 32, 1083–1122 (2007)
Chambin, O., Dupuis, G., Champion, D., Voilley, A., Pourcelot, Y.: Colon-specific drug delivery: influence of solution reticulation properties upon pectin beads performance. Int. J. Pharm. 321, 86–93 (2006)
Galicia, J.A., Cousin, F., Dubois, E., Sandre, O., Cabuil, V., Perzynski, R.: Static and dynamic structural probing of swollen Polyacrylamide ferrogels. Soft Matter 5, 2614–2624 (2009)
Papaphilippou, P.C., Pourgouris, A., Marinica, O., Taculescu, A., Athanasopoulos, G.I., Vekas, L., Krasia-Christoforou, T.: Fabrication and characterization of superparamagnetic and thermoresponsive hydrogels based on oleic-acid-coated Fe3O4 nanoparticles, hexa(ethylene glycol) methyl ether methacrylate and 2-(acetoacetoxy)ethyl methacrylate. J. Magn. Magn. Mater. 323, 557–563 (2011)
Li, Z., Shen, J., Ma, H., Lu, X., Shi, M., Li, N., Ye, M.: Poly(acrylamide/laponite) nanocomposite hydrogels: swelling and cationic dye adsorption properties. Polym. Bull. 68, 1153–1169 (2012)
Satarkar, N.S., Zach Hilt, J.: Magnetic hydrogel nanocomposites forremote controlled pulsatile drug release. Acta Biomater. 4, 11–16 (2007)
Vandamme, T.F., Lenourry, A., Charrueau, C., Chaumeil, J.C.: The use of polysaccharides to target drugs to the colon. Carbohydr. Polym. 48, 219–231 (2002)
Itoh, Y., Matsusaki, M., Kida, T., Akashi, M.: Enzyme-responsive release of encapsulated proteins from biodegradable hollow capsules. Biomacromolecules 7, 2715–2718 (2006)
Akala, E.O., Kopečková, P., Kopeček, J.: Enzyme-responsive release of encapsulated proteins from biodegradable hollow capsules. Biomaterials 19, 1037–1047 (1998)
Miyata, K., Kakizawa, Y., Nishiyama, N., Harada, A., Yamasaki, Y., Koyama, H., Kataoka, K.: Block catiomer polyplexes with regulated densities of charge. J. Am. Chem. Soc. 126, 2355–2361 (2004)
Boas, U., Heegaard, P.M.H.: Dendrimers in drug research. Chem. Soc. Rev. 33, 43–63 (2004)
Zrinyi, M.: Intelligent polymer gels controlled by magnetic fields. Colloid Polym. Sci. 278, 98–103 (2000)
Cheng, Y., Xu, Z., Ma, M., Xu, T.: Pharmaceutical applications of dendrimers: promising nanocarriers for drug discovery. J. Pharm. Sci. 97, 123–143 (2007)
Patri, A.K., Majoros, I.J., Baker Jr, J.R.: Dendritic polymer macromolecular carriers for drug delivery. Curr. Opin. Chem. Biol. 6, 466–471 (2002)
Zhou, J., Wang, L., Ma, J., Wang, J., Yu, H., Xiao, A.: Temperature and pH-responsive star amphiphilic block copolymer prepared by a combining strategy of ring-opening polymerization and reversible addition–fragmentation transfer polymerization. Eur Polym J 46, 1288–1298 (2010)
Yang, Z., Zhang, W., Zou, J., Shi, W.: Synthesis and thermally responsive characteristics of dendritic poly(ether-amide) grafting with PNIPAAm and PEG. Polymer 48, 931–938 (2007)
Dissemond, J., Witthoff, M., Brauns, T.C., Haberer, D., Goos, M.: Untersuchungen zum pH-Wert des Milieus chronischer Wunden im Rahmen einer modernen Wundtherapie. Hautarzt 54, 959–965 (2003)
Rofstad, E.K., Mathiesen, B., Kindem, K., Galappathi, K.: Acidic extracellular. pH promotes experimental metastasis of human melanoma cells in athymic nude mice. Cancer Res. 66, 6699–6707 (2006)
Vaupel, P., Kallinowski, F., Okunieff, P.: Blood flow, oxygen and nutrient supply, and metabolic microenvironment of human tumors: a review. Cancer Res. 49, 6449–6465 (1989)
Chan, A., Orme, R.P., Fricker, R.A., Roach, P.: Remote and local control of stimuli responsive materials for therapeutic applications. Adv. Drug Deliv. Rev. 65, 497–514 (2013)
Foss, A.C., Goto, T., Morishita, M., Peppas, N.A.: Development of acrylic-based copolymers for oral insulin delivery. Eur. J. Pharm. Biopharm. 57, 163–169 (2004)
Peng, C.L., Yang, L.Y., Luo, T.Y., Lai, P.S., Yang, S.J., Lin, W.J., Shieh, M.J.: Development of pH sensitive 2-(diisopropylamino)ethyl methacrylate based nanoparticles for photodynamic therapy. Nanotechnology 21, 155013 (2010)
Park, S.Y., Bae, Y.H.: Novel pH-sensitive polymers containing sulfonamide groups. Macromol. Rapid Commun. 20, 269–273 (1999)
Lee, Y.M., Shim, J.K.: Preparation of pH/temperature responsive polymer membrane by plasma polymerization and its riboflavin permeation. Polymer 38, 1227–1232 (1997)
Soppimath, K.S., Kulkarni, A.R., Aminabhavi, T.M.: Chemically modified Polyacrylamide-g-Guar Gum- based crosslinked anionic microgels as pH-sensitive drug delivery systems: preparation and characterization. J. Controlled Release 75, 331–345 (2001)
Abdelaal, M.Y., Abdel-Razik, E.A., Abdel-Bary, E.M., El-Sherbiny, I.M.: Study on chitosan-poly(vinyl alcohol) interpolymeric pH-responsive hydrogel films for controlled drug delivery. J. Appl. Polym. Sci. 103, 2864–2874 (2007)
Park, H.-Y., Song, I.-H., Kim, J.-H., Kim, W.-S.: Preparation of thermally denatured albumin gel and its pH-sensitive swelling. Int. J. Pharm. 175, 231–236 (1998)
Kurisawa, M., Yui, N.: Gelatin/dextran intelligent hydrogels for drug delivery: dual-stimuli-responsive degradation in relation to miscibility in interpenetrating polymer networks. Macromol. Chem. Phys. 199, 1547–1554 (1998)
Lee, J.W., Kim, S.Y., Kim, S.S., Lee, Y.M., Lee, K.H., Kim, S.J.: Synthesis and characteristics of interpenetrating polymer network hydrogel composed of chitosan and poly(acrylic acid). J. Appl. Polym. Sci. 73, 113–120 (1999)
Nakamura, K., Murray, R.J., Joseph, J.I., Peppas, N.A., Morishita, M., Lowman, A.M.: Oral insulin delivery using P(MAA-g-EG)hydrogels: effects of network morphology on insulin delivery characteristics. J. Controlled Release 95, 589–599 (2004)
Zhang, J., Peppas, N.A.: Synthesis and characterization of pH- and temperature-sensitive poly(methacrylic acid)/poly(N-isopropylacrylamide) interpenetrating polymeric network. Macromolecules 33, 102–107 (1999)
Sideratou, Z., Tsiourvas, D., Paleos, C.M.: Quaternized poly(propylene imine) dendrimers as novel pH-sensitive controlled-release systems. Langmuir 16, 1766–1769 (2000)
Burke, S.E., Barrett, C.J.: pH-responsive properties of multilayered poly(Llysine)/hyaluronic acid surfaces. Biomacromolecules 4, 1773–1783 (2003)
Toncheva, V., Wolfert, M.A., Dash, P.R., Oupicky, D., Ulbrich, K., Seymour, L.W.: Novel vectors for gene delivery formed by self-assembly of DNA with poly(L-lysine) grafted with hydrophilic polymers. Biochim. Biophys. Acta 1380, 354–368 (1998)
Wilding, R.S.S., Davis, F., Pozzi, P., Furlani, A.: Enteric coated timed release systems for colonic targeting. Int. J. Pharm. 111, 99–102 (1994)
Hongkee, S., Chien, Y.H., Park, H., Hwang, S.J., Park, K., Lloyd, A.W.: In: Drug delivery and targeting in new generation technologies. Taylor and Francis, New York (2001)
Ellens, H., Bentz, J., Szoka, F.C.: pH-induced destabilization of phosphatidylethanolamine-containing liposomes: role of bilayer contact. Biochemistry 23, 1532–1538 (1984)
Hoffman, A.S.: Intelligent polymers, controlled drug delivery: challenge and strategies. American Chemical Society, Washington, DC (1997)
Soleimani, F., Sadeghi, M.: Synthesis of pH-sensitive hydrogel based on Starch-Polyacrylate Superabsorbent. J Biomat Nanobiotech 3, 310–314 (2012)
Corpart, J.M., Candau, F.: Aqueous solution properties of ampholytic copolymers prepared in microemulsions. Macromolecules 26, 1333–1343 (1993)
Kathmann, E.E.L., White, L.A., McCormick, C.L.: Electrolyte- and pH-responsive zwitterionic copolymers of 4-[(2acrylamido-2-methylpropyl)-dimethylammonio]butanoate with 3-[(2-acrylamido-2methyl-propyl)dimethylammonio]propanesulfonate. Macromolecules 30, 5297–5304 (1997)
Shahinpoor, M., Kim, K.J.: Ionic polymer-metal composites: I fundamentals. Smart Mater Struct. J. 10, 819–833 (2001)
Nguyen, D.H., Choi, J.H., Joung, Y.K., Park, K.D.: Disulfide-crosslinked heparin-pluronic nanogels as a redox-sensitive nanocarrier for intracellular protein delivery. J. Bioact. Compat. Polym. 26, 287–300 (2011)
Dollendorf, C., Hetzer, M., Ritter, H.: Polymeric redox-responsive delivery systems bearing ammonium salts cross-linked via disulfides. Beilstein J. Org. Chem. 9, 1652–1662 (2013)
Napoli, A., Boerakker, M.J., Tirelli, N., Nolte, R.J.M., Sommerdijk, N.A.J.M., Hubbell, J.A.: Glucose-oxidase based self-destructing polymeric vesicles. Langmuir 20, 3487–3491 (2004)
Podual, K., Doyle, I.I.I., Peppas, N.A.: Preparation and dynamic response of cationic copolymer hydrogels containing glucose oxidase. Polymer 41, 3975–3983 (2000)
Sinha, V.R., Kumria, R.: Polysaccharides in colon-specific drug delivery. Int. J. Pharm. 224, 19–38 (2001)
Zhang, C., Pan, D., Luo, K., Li, N., Guo, C., Zheng, X., Gu, Z.: Dendrimer–doxorubicin conjugate as enzyme-sensitive and polymeric nanoscale drug delivery vehicle for ovarian cancer therapy. Polym. Chem. (2014). doi:10.1039/C4PY00601A
Vemula, P.K., Cruikshank, G.A., John, G., Karp, J.M.: Enzyme responsive acetaminophen hydrogels. Massachussett Institute of Technology Open Access Article (2014)
Rao, N.A.: Role of oxygen free radicals in retinal damage associated with experimental uveitis. Trans. Am. Ophthalmol. Soc. 88, 797–850 (1990)
Nobuhiko, Y., Jun, N., Teruo, O., Yasuhisa, S.: Regulated release of drug microspheres from inflammation responsive degradable matrices of crosslinked hyaluronic acid. J. Controlled Release 25, 133–143 (1993)
Leung, M.F., Zhu, J., Li, P., Harris, F.W.: Novel synthesis and properties of smart core-shell microgels. Macromol. Symp. 226, 177–185 (2005)
Kuckling, D., Adler, H.-J.P., Arndt, K.F., Ling, L., Habicher, W.D.: Temperature and pH-dependent solubility of novel poly(N-isopropylacrylamide)-copolymers. Macromol. Chem. Phys. 201, 273–280 (2000)
Gao, H.F., Yang, W.L., Min, K., Zha, L.S., Wang, C.C., Fu, S.K.: Thermosensitive poly(N-isopropylacrylamide)nanocapsules with controlled permeability. Polymer 46, 1087–1093 (2005)
Gu, J.X., Xia, F., Wu, Y., Qu, X.Z., Yang, Z.Z., Jiang, L.: Programmable delivery of hydrophilic drug using dually responsive hydrogel cages. J. Controlled Release 117, 396 (2007)
Sun, H., Liu, S., Ge, B., Xing, L., Chen, H.: Cellulose nitrate membrane formation via phase separation induced by penetration of nonsolvent from vapor phase. J. Membr. Sci. 295, 2–10 (2007)
Kontturi, K., Mafé, S., Manzanares, J.A., Svarfvar, B.L., Viinikka, P.: Modeling of the salt and pH effects on the permeability of grafted porous membranes. Macromolecules 29, 5740–5746 (1996)
Liu, X., Guoa, H., Zhaa, L.: Study of pH/ temperature dual stimuli-responsive nanogels with interpenetrating polymer network structure. Polym. Inter. 61, 1144–1150 (2012)
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Cassano, R., Trombino, S. (2015). Drug Delivery Systems: Smart Polymeric Materials. In: Puoci, F. (eds) Advanced Polymers in Medicine. Springer, Cham. https://doi.org/10.1007/978-3-319-12478-0_12
Download citation
DOI: https://doi.org/10.1007/978-3-319-12478-0_12
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-12477-3
Online ISBN: 978-3-319-12478-0
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)