Abstract
While microbial biofilms have been recognized as being ubiquitous in nature for the past 40 years, it has only been within the past 20 years that clinical practitioners have realized that biofilm play a significant role in both device-related and tissue-based infections. The global impact of surgical site infections (SSIs) is monumental and as many as 80 % of these infections may involve a microbial biofilm. Recent studies suggest that biofilm- producing organisms play a significant role in persistent skin and soft tissue wound infections in the postoperative surgical patient population. Biofilm, on an organizational level, allows bacteria to survive intrinsic and extrinsic defenses that would inactivate the dispersed (planktonic) bacteria. SSIs associated with biomedical implants are notoriously difficult to eradicate using antibiotic regimens that would typically be effective against the same bacteria growing under planktonic conditions. This biofilm-mediated phenomenon is characterized as antimicrobial recalcitrance, which is associated with the survival of a subset of cells including “persister” cells. The ideal method to manage a biofilm-mediated surgical site wound infection is to prevent it from occurring through rational use of antibiotic prophylaxis, adequate skin antisepsis prior to surgery and use of innovative in-situ irrigation procedures; together with antimicrobial suture technology in an effort to promote wound hygiene at the time of closure; once established, biofilm removal remains a significant clinical problem.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Surgical Site Infection
- Extracellular Polymeric Substance
- Periprosthetic Joint Infection
- Capsular Contraction
- Negative Pressure Wound Therapy
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
3.1 Introduction
In 2010, the Centers for Disease Control and Prevention (CDC) reported that 51.4 million in-patient surgical procedures were performed in the United States (CDC 2010). It has been estimated that approximately 400,000 surgical site infections (SSI) occur in the United States each year with an associated mortality approaching 25 % (100,000) (Reed and Kemmerly 2009; Shepard et al. 2013; De Lissovoy et al. 2009; Herwaldt et al. 2006). While these numbers have historically been extrapolated from in-patient procedures alone, the actual number of SSIs is likely to be much higher since recent CDC data suggests that more than 34 million additional surgical procedures are performed in outpatient ambulatory surgical centers (ASC) in the United States (CDC 2009). The global impact of SSIs is therefore monumental and as many as 80 % of these infections may involve a microbial biofilm (Edward and Harding 2004; Hall-Stoodley et al. 2004; Percival 2004; Romling and Balsalobre 2012; NIH 2002). Many of the microbial populations associated SSIs have been observed to exist primarily within a biofilm matrix, often as a polymicrobial (heterogeneous) community in selective disease processes (Dowd et al. 2008; Edmiston et al. 2013a). The presence of a microbial biofilm within host tissue or on the surface of a biomedical device poses a significant challenge when attempting to eradicate these infections in-situ. In addition, biofilm-mediated infections exhibits resistance to host defense, reportedly contributing to a chronic inflammatory response, leading to complement activation and formation of immune complexes which in turn leads to tissue injury through an excessive inflammatory response (Hoiby et al. 2011; Jenson et al. 2011). A recent study suggests that biofilm producing organisms play a significant (p = 0.024) role in persistent skin and soft tissue wound infections in post-surgical military personnel with deployment-related injuries compared with a cohort control group (Akers et al. 2014). The authors suggest that this presence of a polymicrobial biofilm (71.4 %) was a significant risk factor for relapsing infection in skin and soft tissue infections due to increased bioburden, severity of disease, increased antimicrobial resistance and enhanced inflammatory response within the affected-tissues. Acinetobacter baumannii and Pseudomonas aeruginosa were by far the predominant isolates recovered in both monomicrobial and polymicrobial infections, 24.0 % and 23.7 %, respectively. While microbial biofilms have been recognized as being ubiquitous in nature for the over 40 years; it has only been within the past 20 years that clinical practitioners have realized that biofilm-mediated disease plays a significant role in both device-related and tissue-based infections. The present discussion will focus on the microbial etiology, pathogenesis and treatment of selective biofilm-mediated acute and chronic surgical site infections.
3.2 Biofilm-Mediate Acute and Late Onset Infection in the Surgical Patient
What indications exist to suggest the presence of a biofilm mediated infection? Several biofilm investigators have suggested a diagnostic guideline that may serve to suggest the presence of a biofilm-based infection within the host tissue (Hall et al. 2014; Hall-Stoodley et al. 2012):
-
(a)
Microbiological evidence of a localized or foreign body-associated infection post-surgery,
-
(b)
Microscopic (light or electron optic) evidence of microbial aggregation,
-
(c)
Medical history, documenting a biofilm predisposing condition such as implanted biomedical device, infective endocarditis, previous device-related infection,
-
(d)
Recurrent infection (site specific) with organisms that are clonally identical,
-
(e)
Documented history of antimicrobial failure or therapeutic recalcitrance (persistent infection) despite selection of appropriate antimicrobial agent (both dose and duration), and
-
(f)
Presence of local or systemic signs and symptoms of infection that resolved primarily with appropriate antimicrobial therapy, only to recur following termination of therapy.
Under selective scenarios it may be assumed that a biofilm-based infection is present. However in the case of a chronic inflammatory process involving fascia (deep) or organ-space involvement, the true nature of the infection may not be evident until the wound is actually viewed at surgical revision. While selective diagnostic criteria may be helpful in elevating one’s index of suspicion that a biofilm-associated infection is present, the “heavy-lifting” involves selecting an appropriate course of therapy in the presence of an often recalcitrant disease process.
The incidence of sternotomy SSI ranges from 1 to 8 % with an associated mortality approaching 40 %, which is significant from both a patient outcome and resource utilization perspective (Mauermann et al. 2008). In the United States alone, the number of open cardiac surgery procedures exceeds 600,000 a year (Owen et al. 2010). The time period between cardiac surgery and presentation of a sterna complication can range from 2-weeks to 3-months postoperatively. The staphylococci are the most commonly reported clinical isolates associated with these postoperative infections and both Staphylococcus aureus and Staphylococcus epidermidis are recognized as organisms that possess the ability to produce biofilm (Otto 2008). A study published in 2013 examined sternal tissues and stainless steel wires extracted from infected and non-infected sternal wounds which were analyzed by traditional culture methodology, immunofluorescence and electron optics. The infections in all subjects (6) were characterized as deep sternal wound infections. Positive wound cultures were obtained from two of the patients-Methicillin-resistant S. aureus (MRSA) and Methicillin-sensitive S. aureus (MSSA)-while four were culture negative. All blood cultures collected prior to debridement were negative. Identification of staphylococci in the debrided tissues was confirmed by immunofluorescence. No evidence of staphylococci was observed in debrided tissue remote to the infected wound. Confocal laser scanning electron microscopy (CLSM) confirmed that the staphylococci were organized in three-dimensional clumps and that these clumps represented a thick biomass, occupying 70 % of the infected tissue segments. The extracted stainless steel wires from the infected cases were examined using scanning electron microscopy (SEM), and revealed a metal surface that was coated with a thick extracellular matrix which included cluster of staphylococci. It is interesting to note that all attempts to recover staphylococci from the infected sternal material and sutures using conventional culture techniques were unsuccessful (Elgharaby et al. 2013). Three observations are worthy of consideration; first sternal (deep) tissues and stainless steel wire sutures revealed a staphylococcal biofilm suggesting that there was intraoperative contamination of the surgical field prior to closure. Secondly, the presence of biofilm necessitated the complete removal of all foreign such as the stainless steel wire sutures and a wide (deep) debridement of sternal tissues to facilitate adequate control of the infected tissues. The recalcitrant nature of these infections requires that all infected tissue be excised since residual biofilm will not respond to traditional antimicrobial therapeutic measures even though the planktonic (free-floating) form of the responsible organisms will often be susceptible to traditional antimicrobial agents (Edmiston 1993). Finally, unlike other biofilm-mediated device-related infections (to be discussed later) which are traditionally viewed as device-centric infections and not tissue-based, these deep sternal wound infections often involve both tissue and prosthetic material, contributing to the significant morbidity and mortality associated with these serious infections.
Bacterial biofilms are now recognized as a causal etiology of dental peri-implantitis, leading to chronic infection in selective oral surgery patients (Subramani et al. 2009). The process of post-implant infections requires the formation of a plaque which is similar to that seen on “native” teeth. The biofilms associated with these infections are highly heterogeneous, and may involve the following bacteria; streptococci, Actinomyces, Porphyromonas, Prevotella, Capnocytophaga and Fusobacterium species. The surface characteristics of a prosthetic implant serves to foster microbial adherence and colonization by biofilm-forming oral microbiota. The rough surface of titanium implants enhances microbial adherence and plaque formation, leading to subsequent inflammation of the gums (Ray and Triplett 2011). Biofilm-mediated osteomyelitis can also be associated with dental peri-implantitis and the primary causative pathogens are S. aureus and S. epidermidis. However, biofilm-mediated osteomyelitis of the jaw can also be associated with oral streptococci, Bacillus species and Actinomyces species. Similar to other biofilm-mediated infections, antibiotic therapy has limited effectiveness and surgical debridement is still the treatment of choice.
Biofilm formation often plays a significant role in the etiology of periorbital implant infections with Micobacterium chelonae, S. aureus and P. aeruginosa being the three most common organisms recovered from explanted periorbital devices (Samini et al. 2013). The median time from implant to disease presentation is approximately 13 weeks. Implanted devices which can be affected include orbital spheres, enucleated spheres, lacrimal stents, Jones Tubes and sclera buckles. Electron microscopy optic studies have documented that the biofilm-associated with infected periorbital implants involve a heterogeneous collected of microorganisms encased in a thick exopolysaccharide matrix and the definitive therapy for treatment of these infection involves device removal (Sugita et al. 2001; Parsa et al. 2010; Holland et al. 1991).
According to the American Society of Plastic Surgeons, breast implants represent the leading type of cosmetic plastic surgery with over 300,000 breast implant surgeries performed annually in the United States (American Society of Plastic Surgeons 2011). The reported incidence of infection in breast reconstruction is between 2.8 and 28 % which suggests a high probably risk of infection for women undergoing these procedures (Neto et al. 2002; Tran et al. 2002; Vandeweyer et al. 2003; Nahabedian et al. 2003; Olsen et al. 2008). Bacterial contamination of the implant at the time of insertion can result in persistent low-grade inflammation of the surrounding tissues, leading to capsular fibrosis and capsular contraction (Rieger et al. 2013). A recent meta-analysis has documented a significant risk for infection associated with expander/implant reconstruction compared with reconstruction using autologous abdominal tissue (Tsoi et al. 2014). While most traditional (swab) cultures of explanted devices typically yield few organisms, sonication of explanted devices and tissue biopsies have yielded microbial recovery in 38.5 and 89.5 % of respective samples (Netscher 2004). S. epidermidis is hypothesized as the causative organism associated with capsular contraction, contaminating the device at the time of insertion, persisting on the device surface and resulting in subclinical infection. A recent study has documented that staphylococcal contamination of the surface of the expander/implant devices leads to biofilm-formation in 80 % of culture positive devices (Jacombs et al. 2014). The authors’ of this study have suggested that textured breast implants significantly potentiates biofilm formation compared with implants having a smooth surface. While the biologic advantages of a textured surface leads to better tissue incorporation and therefore less potential contracture, surgeons should be aware that this advantage can be negated if the device is contaminated at the time of insertion. The current treatment for an infected tissue expander or implant is device removal which is often catastrophic for the patient, potentially delaying additional therapy such as chemo or radiotherapy in patients who have undergone breast reconstruction follow mastectomy.
The practice of neurosurgery has witnessed an explosion in the number of devices that have been developed to treat patients. These include complex spinal instrumentation/hardware, pulse generator, indwelling Silastic catheters and shunts and synthetic bone flaps after delayed cranioplasty (Braxton et al. 2005). The estimated rate of infection associated with implantation of spinal hardware ranges from 2 % to approximate 9 % (Braxton et al. 2005; Massie et al. 1992). Implant infections are characterized by increased utilization of healthcare resources including prolonged length of stay, increased cost of antibiotic therapy, additional surgical revisions and extended rehabilitation post discharge. S. aureus and S. epidermidis are the two most common isolates recovered from documented spinal hardware infections. Early infections occur within approximately 2 weeks of implantation and are associated with intraoperative infection or wound contamination in the immediate postoperative period. Infections which present years postoperatively have been thought to occur following an unrelated hematogenous event. But they could also be associated with phenotypic changes of sessile bacteria to their planktonic forms in device-related biofilms. Resolution requires reoperation and removal of the infected device. The incidence of infection associated with deep brain stimulators is reported to be approximately 3.7 %, and similar rates have been suggested for dorsal column stimulators (3.4 %) (Umemura et al. 2003; Cameron 2004). Approximately 20,000 ventriculoperitoneal (VP) shunts are placed annually in the United States and the incidence of infection can exceed 15 % with virtually all infected devices harboring a biofilm (Bondurant and Jimenez 1995; Davis et al. 2002; Wood et al. 2001). The same analogy can be made for ventriculostomy catheters of which nearly 140,000 are placed yearly for many indications, including acute hydrocephalus to ICP monitoring and management of neurotrauma (Lozier et al. 2002). The reported infection rate following catheter insertion in this patient population is approximately 10 % and ventriculitis is a potentially life-threatening biofilm-mediated complication. The morbid nature of ventriculoperitoneal and ventriculostomy infections has led to the development of antimicrobial impregnated technologies that resist bacterial adherence, thereby limiting biofilm development (Braxton et al. 2005). Unfortunately infection remains a serious outcome in this patient population. Infection is a major complication of delayed bone flap cranioplasty and most cryopreserved bone grafts when cultured by conventional methodology are negative, which is suggestive of culture-resistant biofilm contamination (Braxton et al. 2005). Because of the difficulty in treating these serious life-threatening infections current emphasis focuses on fastidious technique and appropriate skin-antisepsis that can also include innovative irrigation techniques using effective biocidal agents (Barnes et al. 2014).
Over 900,000 abdominal wall hernia repairs are performed yearly in the United States (Engelsman et al. 2007). A recent meta-analysis has suggested that use of synthetic mesh prosthesis for abdominal wall closure significantly increases the risk infection (Scott et al. 2002). Contamination of the implanted mesh usually occurs at the time of implantation or exogenously in the early postoperative period. Several clinical studies suggest that the infection rate is highly variable and dependent upon the type of mesh used for abdominal repair ranging from 2.5 to >6 % with polypropylene; less than 1 to >9 % with expanded polytetrafluoroethylene (ePTFE); while polyester mesh demonstrates an infection rate similar to polypropylene (Luijendiik et al. 2000; Leber et al. 1998; Bauer et al. 1999; Hamy et al. 2003; Machairas et al. 2004). The level of microbial-contamination (bioburden) that develops upon the surface of a synthetic mesh is dependent upon the type of material and the structural surface characteristics of the device. For example, meshes made of multifilament Dacron support a luxurious and dense biofilm (Engelsman et al. 2008). Meshes which exhibit a hydrophobic surface such a ePTFE initially inhibit bacteria adherence. However with prolonged exposure to bacterial contamination both Gram-positive and Gram-negative organisms will form a dense biofilm. So it would appear that surface hydrophobicity as a deterrent against bacterial adherence has only a short-term benefit in vivo (Davidson and Lowe 2004). Biofilm-associated mesh infections adversely impact the wound healing process by interfering with the in-growth of host tissues through the mesh. S. aureus colonization of the mesh surface induces fibroblast death (apoptosis), thereby inhibiting the proliferation of these cells during the maturational period of wound healing (Bellon et al. 2004; Edds et al. 2000). Biofilm formation on the surface of a synthetic mesh can result in chronic infection, draining sinuses, mesh extrusion and enteric fistula formation. Most chronic biofilm infections has been associated with S. aureus but mesh infections following abdominal surgery may also involve selective Gram-negative bacteria. A recent analysis has suggested that relapsing infections of the type observed with chronic mesh infections is almost always associated with biofilm-forming microorganism and many of these isolates also express multi-drug resistance (Sanchez et al. 2013). Resolution of these infections requires complete mesh removal, along with removal of other foreign bodies such as residual suture material, followed by simultaneous reconstruction often using a monofilament polypropylene mesh. The affected patients will often require a prolonged period of follow-up to monitor the possibility of occult infection following mesh replacement (Birolini et al. 2014).
The pathobiology of a vascular graft infection is best understood as a biofilm-mediated infection. An excellent example of a biofilm-mediated vascular graft infection is the development of a groin sinus tract following insertion of an aortofemoral prosthetic bypass graft. These infections are characterized as late-onset, occurring weeks to months post-implantation and the presentation may be occult with no systemic signs of infection. Traditional culture methodology often fails to recover any isolates, However when the graft segment is sonicated, S. epidermidis is often recovered in numbers which exceed 6-logs (Hasanadka et al. 2007). The establishment of a biofilm-mediated vascular graft infection requires a series of sequential events (Bandyk and Black 2005; Edmiston et al. 2005). First, the device is contaminated at the time of insertion by a biofilm-forming organism; a process that is facilitated by surface conditioning by blood and tissue fluid proteins. Once the organism adheres to the surface of the graft a microcolony aggregation may to form followed by the elaboration of an extracellular matrix which can eventually progress to development of a mature biofilm. The organization and maturation of the bacterial biofilm is a dynamic process that ironically requires very few contaminating organism to initiate the process. A low metabolic activity, due to limited substrate availability and production of a luxurious extracellular matrix, contributes to the physiological conditions that foster resistance to both host immune defenses and antimicrobial therapy (recalcitrance). Typically, the biofilm spreads slowly over the exterior surface of the graft, eventually involving the graft-to-artery anastomosis, reducing anastomotic tensile strength, leading to development of a pseudoaneurysm and eventual graft failure that may be heralded by catastrophic hemorrhage. During this process there is little or no spread to the perigraft tissues nor does one observe the development of fulminant sepsis unlike early-onset vascular graft infections involving S. aureus or Gram-negative pathogens (usually Escherichia coli). The incidence of SSI following vascular surgery is reported to be in the range of 5–15 %; higher in diabetic patients, patients colonized with MRSA and after procedures requiring a groin incision (Armstrong and Bandyk 2006; Frei et al. 2011). Early-onset infections are characterized by wound dehiscence and purulent drainage often within days of surgery (Fig. 3.1). Late onset infections are more indolent, clinical recognition can be delayed for months or even years. Signs of SSI may include failure of graft healing (incorporation) within the surrounding tissues, sinus tract formation, pseudoaneurysm formation or late erosion into adjacent bowel mediated by a chronic inflammatory process (with attendant enteric hemorrhage), due to the presence of a mature bacterial biofilm (Fig. 3.2) on the surface of the graft (Frei et al. 2011). While historically, treatment of a late-onset vascular graft infection involved an extra-anatomic revascularization, this approach has been replaced by in-situ graft replacement, which is associated with significantly less morbidity and mortality (Hart et al. 2005). Future developments in graft technology to reduce microbial adherence, along with effective intraoperative risk reduction technologies are warranted.
Developmental presentation of late-onset vascular graft infection involving S. epidermidis; intraoperative contamination leads to causal contamination of external surface of vascular graft, organism down-regulates its metabolism growing slower over week to month, biofilm finally reached critical density resulting in chronic inflammatory leading to pseudoaneurysm and possible graft failure
Over the past 20 years the number of annual total hip replacements in the United States has increased twofold to >250,000 while the number of total knee replacement has increased almost fivefold to >500,000 (Del Pozo and Patel 2009). The risk of infection following total joint replacement is 0.5 % to approximately 2 % for hips and knees and 2 % to approximately 9 % following ankle replacement (Laffer et al. 2006; Kessler et al. 2012). Biofilm-mediated infection of orthopedic implants can occur either as an exogenous process, with contamination of the device occurring during surgery (or early in the postoperative period) or hematogenously via the bloodstream at any time after surgery. The vast majority of exogenous infections occur in the acute postoperative phase, especially in patients experiencing poor wound healing. Periprosthetic joint infections (PJI) can be classified as “early”, “delayed” or “late.” Early infections occur within the first 2 months; delayed infections occur between the third and 24th month postoperatively and late infections are diagnosed >2 years post-implantation. The most common organisms associated with PJI are the coagulase-negative staphylococci (30–43 %) and S. aureus (12–23 %). These are followed by streptococci (9–10 %), enterococci (3–7 %), Gram-negative bacilli (3–65 %) and miscellaneous anaerobes, such as Propioni-bacterium and Peptostreptococcus spp. (2–4 %) (Pandey et al. 2000; Steckelberg and Osmom 2000). Polymicrobial infection is observed in approximately 10 % of cases and 10–30 % of clinical cases present as culture-negative. As with other biofilm-mediated infections, traditional antibiotic therapy has limited utility in the treatment of in situ PJI. Therapeutic efficacy dictates that a sufficient concentration of antibiotic greater than the MIC90 for most likely pathogens must migrate from the blood into the tissue space (second compartment) then into the biofilm, which is essentially a third compartment. Previous studies have documented that the MIC required to inhibit or kill most microorganism within a mature biofilm is often 100–1,000 times the traditional MIC90 for selective device-related microbial pathogens (Edmiston 1993; Costerton et al. 1995). The use of antibiotics in the treatment of PJI is adjunctive to surgical management, which often involves a two-stage debridement and revision, requiring the removal of the infected prosthesis with implantation of a new device at a later operation. A temporary articulated antimicrobial-impregnated spacer is constructed for use until the second operation. The success rate of this procedure approaches 90 % but failures require additional interventions. In some scenarios debridement and retention of the original implant may be successful if: (a) the infection is detected early (<3-weeks), (b) there is absence of a sinus tract, (c) the organism is susceptible to traditional antimicrobial therapy (suggesting little if any biofilm involvement), and (d) the implant is stable (not loose). Another option involves a one-stage procedure; removal of the infected implant with immediate re-implantation of a new device, but the patient must have intact or slightly compromised soft tissue to qualify for this approach. However, the involvement of multi-drug resistant pathogens will often preempt this approach (Zimmerli and Moser 2012). A recent report has suggested that biofilm-forming organisms recovered from culture-positive cases are commonly associated with a polymicrobial infection (37.5 %), posing an additional therapeutic challenge (Fernandes and Dias 2013). These infections are among the most catastrophic for both the patient and practitioner, management may involve years of additional medical and surgical care without a guarantee of successful resolution. A proportion of device-adherent (biofilm) organisms are metabolically locked in a stationary growth phase and therefore the usual achievable antibiotic serum and tissue concentrations are inadequate to resolve the in-situ infection, leading to therapeutic failure. This most often leads to therapeutic failure. The optimal strategy is prevention which requires putting into place an appropriate interventional bundle that significantly reduces the risk of postoperative infection, therefore minimizing the opportunity for wound/device contamination (Kim et al. 2010).
Traditional studies of device-related infection have focused primarily on the device itself and little if any time is spent considering the role that sutures may play in initiating or potentiating the risk of postoperative infection. While wound closure technologies such as surgical sutures, have not always been viewed in the same light as other implantable biomedical devices, surface characteristics of these devices make them a susceptible substrate for bacterial adherence and/or contamination. The classical studies conducted by Varma, Elek and Raju documented the microbial burden required to produce an infection in a clean surgical wound (Varma et al. 1974; Elek and Cohen 1957; Raju et al. 1977). These studies further characterized the role of suture material as a foreign body, functioning as a nidus for infection in the presence of wound contamination. Recent reports by Kathju and colleagues would suggest that contamination of surgical sutures at the time of implantation by biofilm-forming organisms leads to recalcitrant infection, necessitating eventual removal of the infected material (Kathju et al. 2009, 2010). In a recent study, suture segments were explanted from 158 surgical patients, 46 (29.1 %) were recovered from documented infected cases. A bacterial biofilm was observed by scanning electron microscopy (SEM) on 100 % of infected sutures. Biofilms associated with infected explanted sutures were observed in both deep incisional and organ-site infections. In the majority of these infected cases the microbial burden exceeded >105 cfu/cm suture surface (Edmiston et al. 2013a). In three separate cases involving infected mesh segments, the primary device had been removed but recurrent infection required exploration and removal of retained suture segments. All three of these suture segments (polypropylene) exhibited a polymicrobial microbial flora comprised of Gram-positive (2 MRSA, 1 MSSA), Gram-negative aerobic (E. coli) and anaerobic bacteria (Peptostreptococcus and Bacteroides spp.) enmeshed in a luxurious biofilm (Fig. 3.3). These findings are complimentary to previous in-vitro studies, which suggest that bacterial adherence to surgical sutures is associated with the formation of a luxurious bacterial biofilm (Henry-Stanley et al. 2010; Williams and Costerton 2012). Preventing microbial adherence and biofilm formation on the surface of a multifilament or monofilament suturing devices would appear to be a beneficial risk reduction strategy. Several investigators have documented a reduction in bacterial adherence (Gram-positive and Gram-negative) to the surface of multifilament and/or monofilament sutures which are coated with the biocide, triclosan (Rothenburger et al. 2002; Ming et al. 2007; Edmiston et al. 2006a).
3.3 Mechanistic Aspects of Biofilm Formation in Host Tissues and Implantable Devices
A biofilm is an organized community of bacteria attached to a surface and enveloped within a self-produced matrix (Costerton et al. 1999). The formation of a differentiated multicellular community gives a biofilm defense against UV light, bacteriophages, biocides, antibiotics, immune system responses, and many environmental stresses. The biofilm, on an organizational level allows the bacteria to survive many intrinsic and extrinsic defenses that would inactivate the single cell (planktonic form) bacteria. The first step in establishment of a biofilm-mediate infection involves adherence of the organism to a conditioned surface such as host-tissues or implanted device. Biofilms can functions as a partial physical barrier against penetration of antibiotics, antibodies and granulocytic cell populations (Akiyama et al. 1997; Hoyle and Corsterton 1991). In the presence of host tissue protein (plasma), S. aureus forms a biofilm that has a unique composition, composed of sheaths of fibrin and glycocalyx (Nemoto et al. 2000). These substances serve to anchor the matrix to the infected cell or inert biomedical device surface.
According to a much cited model, in the process of microbial biofilm formation the microbial cells attach irreversibly to surfaces (i.e., those not removed by gentle rinsing) and will begin cell division, forming microcolonies, and produce the extracellular polymers that define structural components of the biofilm (Fig. 3.4). These extracellular polymeric substances (EPS) consist of polysaccharides, proteins, DNA and other materials of microbial and host cell origin, which and can be detected microscopically and by chemical analysis. EPS provides the matrix or structure for the biofilm. The biofilm matrices are highly hydrated (98 % water) and tenaciously bound to the underlying surface. The structure of the biofilm is not a mere homogeneous monolayer of slime but is heterogeneous, both in space and over time. The presence of “water channels” allow transport of essential nutrients and oxygen to the cells growing within the biofilm (Evans and Lewandowski 2000). In certain situations, biofilms have a propensity to act almost as filters to entrap particles of various kinds, including minerals and host components such as fibrin, RBCs, and platelets.
A simplified general model showing the sequence of events through which biofilms develop. In this scenario, a mixture of bacterial species in planktonic form adheres to the implant or tissue surface (1) and develops into a biofilm (2) with the associated deposition of enveloping matrix material. Dispersion (3) where it occurs can enable cells to colonize elsewhere. The diagram also shows coaggregation, where taxonomically distinct bacteria form aggregates through specific surface receptor interactions and aggregation (where cell clusters form through a range of mechanisms)
Phenotypic heterogeneity of biofilm-associated bacteria is an important biofilm characteristic and biofilm-associated organisms can grow more slowly than planktonic organisms occupying the same niche due to localized nutrient and/or oxygen depletion (Donlan 2001b). Bacterial cells may detach from the biofilm as a result of physical disturbance, cell growth and division, or the spontaneous release or biofilm cell aggregates (Donlan 2001a). These detached cells can potentially metastasize to distant sites in the host causing a systemic infection. Formation of a mature biofilm requires a complex series of events, involving different organisms (motile and non-motile), the material on to which the biofilm forms and the external environment including fluid flow, oxygen levels and availability of nutrients. It can be viewed as a multi-stage process beginning with adherence (adherent stage) followed by proliferation and differentiation of the attached cells (maturation stage). From a molecular perspective, in some bacteria, these two stages are controlled by surface adhesins and cell-to-cell signaling pathways, respectively. To understand attachment, it is important to closely examine the properties of the material or tissues upon which the biofilm will form. Implanted medical devices can range from various hydrophilic materials such as plastic or PTFE, which is often used for vascular prosthesis or hydrophilic materials such as various metals used in orthopedic implants (Meier-Davis 2006). Other materials that may serve as a substrate for bacterial adherence and biofilm formation can include xenografts such as implantable porcine heart valves.
Planktonic bacteria with special appendages such as fimbria, cilia, and flagella, allow for motility but also give the bacteria a sense of “touch”. Under some conditions when a bacterium encounters a surface that has been conditioned with small organic molecules, it explores the surface (Singh et al. 2002; Stoodley et al. 2002). This process is called “twitching.” According to this model, the bacterium attaches irreversibly to the surface, a radical change in the phenotype of the bacterium occurs (Sauer et al. 2002). Over 800 new proteins can be expressed within the first hour during which the bacterium attaches (Sauer et al. 2002). The microorganism undergoes division to form a young community of cells, and after only several hours, the beginning of an immature biofilm is visible (Harrison-Balestra et al. 2003). Once irreversible attachment has occurred, the bacterium is committed to a biofilm phenotype. Rapidly, the bacterium changes its phenotype and begins to grow and differentiate. The bacteria begin to excrete extracellular polymeric substances (EPS), which can contribute to the communal defense of the nascent bacterial biofilm. These cells can share a similar genotype but eventually differentiate into a variety of phenotypes. This emerging microbial population can be viewed as a of a “microcolony.” The microcolony continues to develop and mature. It takes cue from cell-to-cell signaling molecules such as quorum sensing molecules which work to guide its maturation (Parsek and Greenberg 2005). Neutrophil accumulation within the biofilm can initiate self-injury through the released of oxidants which in turn compromises host defense mechanisms. Debris from the necrotic neutrophils serves as a biological matrix that facilitates additional biofilm formation (Walker et al. 2005).
The mature biofilm releases planktonic ‘seeds’ cultures which will stimulate the host immune response to mount an intense inflammatory response. The biofilm will derive nutrients from the host exudate that accompanies this inflammatory response. In this way, the sacrifice of a few bacteria promotes the survival of the community through continual nutrient acquisition.
3.4 Why Are Biofilms Difficult to Eradicate Using Antimicrobials?
As described earlier from the clinical perspective, surgical site infections (SSIs) associated with implants are difficult to eradicate using antibiotics regimens that would typically exhibit effectiveness against the same bacteria growing under planktonic conditions (Gilbert and McBain 2001). In addition, clinical laboratory results based upon in-vitro susceptibility of planktonic cells provide little clinical therapeutic guidance (Edmiston 1993). An understanding of the mechanisms which underlie biofilm recalcitrance is useful because it contributes to the development of more effective strategies for biofilm control and aids clinicians in utilizing current therapeutic option more effectively. It is known multiple mechanisms are responsible, most of which relate to phenotypic changes and multi-cellularity, rather than the genetic adaptation responsible for antibiotic resistance exemplified by MRSA. This is evidenced by the fact that cells dispersed from a biofilm, if tested before significant cell division occurs will exhibit comparable susceptibility to planktonic cells (Gilbert et al. 2002). The following discussion outlines the main mechanism which is believed to underlie biofilm resistance and is based on the schematic presented in Fig. 3.5.
Biofilm recalcitrance is a complex and incompletely understood phenomenon. This diagram presents some of the main mechanisms believed to be responsible all of which depend on the heterogeneity afforded by the biofilm matrix. (1) gradients of nutrient and gases may be establish resulting in local variations in bacterial phenotype including areas of dormancy; (2) the penetration of antimicrobials may be perturbed by the matrix and by enzymes within the matrix including β-lactamases. This can reduce the effective concentration of active agent delivered to the deeper layers. (3) persister cells, which are phenotypically specialized recalcitrant variants can be protected from immune cells by the matrix and thus may survive sub-inhibitory antimicrobial exposure allowing regrow following treatment
3.4.1 Penetration Failure
This is probably the most intuitive reason for the tenacity of biofilms under antimicrobial stress but paradoxically it is least likely to be the main reason for antimicrobial recalcitrance. An antimicrobial agent must penetrate a biofilms effectively in order to achieve a high level of bacterial inactivation. However, diffusivity of biofilms is normally sufficient to allow significant penetration and furthermore, water channels that are a feature of some biofilms have been likened to a primitive circulatory system that will further enhance drug penetration. Cationic antimicrobials or biocides such as gentamicin and chlorhexidine can bind to anionic sites within the biofilm matrix in a process termed reaction-diffusion limitation, which perturbs penetration but probably only temporarily. Extracellular products of bacterial growth, including β-lactamases and other drug inactivation enzymes can concentrate with the biofilm matrix and thus enhance penetration failure of some antimicrobials, but in all of these situations, this protective mechanism can be overcome if the antimicrobials are delivered to the site for a sufficient length of time and concentration (Gilbert et al. 2002).
3.4.2 Phenotypic Heterogeneity and Biofilm-Specific Phenotypes
Growth-rate is a key mediator of bacterial susceptibility to many antimicrobial agents even in planktonic cells. Bacteria which are not actively dividing may be refractory to some antimicrobials through a process that has been termed “drug-indifference” (Jayaraman 2008). This is where the role of the biofilm matrix is particularly important since immobilization can confer marked phenotypic variation in cellular growth-rate within the biofilm due to localized depletion of nutrients and oxygen (for aerobic cells). This has been depicted in Fig. 3.5 as “phenotypically distinct cell clusters” but in mixed species biofilms, taxonomically distinct clusters of bacteria (also illustrated) may occur and represent pockets of bacterial survival. The reason phenotypic heterogeneity alone is unlikely to account for the totality of biofilm recalcitrance is that during antimicrobial treatment, biofilms normally undergo a considerable level of cellular death; survival being a pyrrhic victory in which small pockets of viability facilitate re-growth. In such circumstances the initial bacterial death which occurs at the biofilm periphery on exposure to antimicrobial exposure will expose deeper lying cells to nutrients and oxygen reducing heterogeneity hence lowering localized recalcitrance.
3.4.3 Persister Cells
The above explanation probably does not fully account for the extent of biofilm recalcitrance. The persister hypothesis which was originally proposed by Bigger in 1944 answers many of the questions one has when contemplating the process of biofilm recalcitrance (Bigger 1944). It was noted that strains of staphylococci could not be sterilized in situ with penicillin; an antibiotic to which it was highly susceptible. Surviving cells which were called “persisters” occur at a frequency of approximately one per million of the bacterial cell present in the original culture, for which penicillin is bacteriostatic and only very slowly, if at all, bactericidal. Another important early paper (Gunnison et al. 1964) made similar observations for S. aureus, demonstrating that persister cells were phenotypic variants that did not result from stable genetic alteration. It was noted that that the proportion of survivors was not changed even by a 1,000-fold increase in the dose of penicillin, or following the addition of streptomycin. As with the earlier study, these persister organisms were essentially drug indifferent. More recently, it has been shown that biofilm recalcitrance in P. aeruginosa is largely attributable to slow growth rate and the presence of persister cells (Spoering and Lewis 2001). Therefore persisters, which many species of bacteria can form, represents an important facet in the current explanation of why there is biofilm recalcitrance, with survival drug treatment and proliferation afterwards while being protected from phagocytosis by the biofilm matrix.
3.5 Biofilms, Infection and Wound Healing in Chronic Surgical Wounds
Biofilms can occur in practically any hydrated non-sterile environment. Their dispersed or “planktonic” counterparts, upon which microbiologists have based much of their understanding of microbial behavior, differ markedly in pathogenicity and in their responses to antimicrobials and the immune system. Biofilms are may be present in up to 70 % of open wounds, healing by secondary intention and this figure probably increases in chronic wounds particularly when there are multiple, underlying and unmet co-morbidities. There is less evidence of their presence in acute wounds but, since biofilms take time to form, it is highly likely that their incidence may be lower in acute wounds (James et al. 2008; Percival et al. 2012).
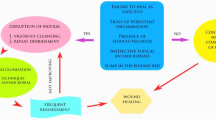
Increasing knowledge and understanding of the formation and inhibitory influence of biofilms on wound healing processes has changed many aspects of wound management. The early stages of biofilm formation may occur rapidly even in acute wounds such as burns and sutured surgical wounds healing by primary intention, and may therefore resulting in delayed healing or a greater risk of developing overt infection (Percival et al. 2012; Costerton et al. 1999). The microorganisms which can cause wound infection are diverse and in the case of chronic wound infect they are often polymicrobial. It is commonly observed that the presence of bacteria (or the bioburden) in chronic, open wounds presents as a continuum from contamination through colonization to local and systemic infection. Although the precise definition has not been agreed on, a pre-local, or covert, infection phase has been referred to as “critical colonization” (Kingsley 2001). In critically colonized wounds, there may be no clear signs of acute infection in (the Celsian calor, rubor, dolor et tumor), but instead there may be unexplained stalled or delayed healing, usually associated with increasing pain, exudate or smell, and abnormal or excessive granulation tissue, and maceration of surrounding skin (Cutting and Harding 1994; Gardner et al. 2001). This may be worsened by underlying pathological processes such as venous or arterial insufficiency, diabetes, pressure damage or an occult malignant process. The difficulty of assessing infection, particularly in these chronic wounds, is not made easier by conventional processing of microbiological swab analysis. Even when the Levine technique (based on the rotation of the swab, under pressure, in the wound swab over a 1 cm2 area) is used, to harvest organisms deep in the wound bed, or sequential biopsies are taken to assess the quantity of colony forming units/gram of tissue and progress of treatment (which has been accurate only in burn management and is unpopular with patients and Ethics Committees!) only planktonic bacteria are identified. If this report is to the caregiver and if the sensitivities are included, there is a high likelihood that an inappropriate antibiotic will be prescribed with attendant risk of developing antibiotic resistance.
Biofilms may be involved in this continuum of micro-organism proliferation (Davis et al. 2008). The establishment of bacterial biofilms in wounds may hypothetically equate to uncontrolled critical colonization and failure to prevent or manage this process, risks invasive tissue infection. The sequence of biofilm persister-cell phenotype to biofilm planktonic-cell phenotype would seem to turn the Koch postulates on infection upside down (Percival et al. 2010; Wolcott et al. 2010a). As a result conventional swabbing and microbiological testing in this situation is unlikely to reveal the “culprit” planktonic organisms, since no single bacterial species is responsible, or aid with a treatment strategy. The presence of bacterial biofilms on a wound surface can encourage and excite an underlying, inappropriate and excessive host inflammatory response (through stimulation of neutrophils and macrophages) to cause a prolonged release of nitric oxide, inflammatory cytokines and free radicals and delayed healing (Wolcott et al. 2008). This delay is not an inert biological process; quite the reverse, with persistence, the early inflammatory processes and healing cascades may be out-of-phase. This concept, which helps to explain why chronic wounds fail to heal, is not new (James et al. 2008; Percival et al. 2012; Costerton et al. 1999; Kingsley 2001; Bjarnsholt et al. 2008). The same concept likely applies to surgical wounds and explains why some surgical wounds fail to heal, often with superficial skin dehiscence, without clinical signs of acute infection (cellulitis, pus formation and pain) and a failure to identify/harvest micro-organisms from the dehisced wound. Although there is likely to be biofilm-related structures in or on most wounds these cannot be recognized without sophisticated laboratory testing. However, if there is a lack of clinical progress in the healing of an open surgical wound, or of any hard-to-heal, chronic wound, it is reasonable to assume that there is critical colonization together with reformation of biofilm. It is misguided to believe biofilms are visible to the naked eye although they may be present when the wound bed is heavily exuding or covered with fibrinous material or necrotic tissue that needs debridement.
The ideal way to manage a biofilm-mediated surgical wound would be to prevent it from occurring through the rational use of antibiotic prophylaxis in addition to adequate skin antisepsis prior to surgery or the use of antimicrobial-coated sutures. Once there is bacterial attachment to the wound bed and biofilm formation it is probable that only maintenance debridement can control it. Prevention of biofilm reformation in open wounds involves adequate wound irrigation or cleansing using antiseptics, the use of negative pressure wound therapy (NPWT), or the use of antimicrobial dressings. The diversity of biofilm phenotypes directly relates to successful attachment and infection in the wound, together with resistance to host response and antimicrobial therapy (whether it is a topical antiseptic or systemically administered antibiotic), particularly in the non-planktonic state (Sauer et al. 2002; Cho and Caparon 2005; Leid et al. 2005; Stewart and Costerton 2001; Costerton and Stewart 2001; Mah and O’Toole 2001). The role that biofilms may play in delaying healing in sutured surgical or traumatic wounds is less clear, although it may account for early dehiscence of wounds after sutures or staples have been removed. Separation of the sutured skin is often accompanied with little evidence of acute inflammation or pus formation and cultures obtained from the margins of the wound are often negative, failing to yield responsible organism. Critical colonization and biofilm formation may also be the cause of failed split thickness skin grafts. Several unanswered questions remain, particularly for acute surgical wounds: does biofilm formation turn an acute wound into a chronic one (this may be relevant in early diabetic foot ulcers); does biofilm formation precede donor or recipient site infection, or burns colonization prior to infection, particularly with Pseudomonas? Microbial attachment and biofilm formation may also occur within deep wounds and these biofilms would be clinically relevance in orthopaedic and vascular surgical site infections. For example, in a persister-cell state organisms sequestered within a biofilm attached to a prosthetic graft, may when conditions are optimal, begin to re-grow, thus perpetrating the infection. This may occur many months post-surgery and in the case of late-onset vascular graft infection occur as an occult process. The use of an antimicrobial suture technology, with a prolonged antiseptic release, may be a valid strategy for preventing initial microbial adherence in an environment of wound contamination (Edmiston et al. 2013b).
3.6 Methods for Biofilm Removal: Maintenance Debridement
Table 3.1 identifies the major strategies for wound debridement. Biofilms in wounds often defy eradication and in selective cases can only be suppressed, rather like the suppression of MRSA colonization using mupirocin plus chlorhexidine to reduce the risk of SSI; however biofilm decolonization is unlikely (Edmiston et al. 2013b). Debridement should therefore be undertaken regularly, at least weekly has been suggested in critical ischemia (Wolcott and Rhoads 2008). Biofilm suppression can be effective in surgical wounds using similar methods used for macro-debridement of slough and necrotic material in preparation of the wound bed; both should be undertaken simultaneously at dressing changes (Wolcott et al. 2009). The concept of “preparation of the wound bed” is taken from plastic surgical practice in which a recipient wound site is made as clean and receptive as possible for a split thickness skin graft. In open wounds, where the infection and biofilm is controlled by these same techniques, healing can progress successfully by secondary intention with adequate wound and dressing care alone; coupled of course with attention to holistic and correction of underling disease processes (Leaper et al. 2012a). The use of regular maintenance debridement has been shown to enhance therapeutic interventions using topical antimicrobials and dressings to reverse delayed or stalled healing, presumably by delaying biofilm reformation (Wolcott et al. 2010b; Phillips et al. 2010a).
All the methods of “macro”-debridement shown in Table 3.1 can equally be used for maintenance debridement of biofilm from critically colonized chronic, infected or dehisced surgical or burn wounds. It is probably universally accepted that open wounds heal optimally with debridement of any type; the more complete the debridement is (with removal of biofilm), the more the frequency of intervention can be lessened (Wolcott et al. 2009; Leaper et al. 2012b; Cardinal et al. 2009; Falanga et al. 2008). Methods of surgical, mechanical or sharp debridement are the most widely used at dressing changes at which the wound can also be cleansed or irrigated (with or without antiseptics). An even more effective removal of biofilm, and slowing its reformation, may be expected from the use of hydrolavage and negative pressure wound therapy (Vanwijck et al. 2010; Caputo et al. 2008; Mosti et al. 2005; Allan et al. 2010). The use of antiseptics at dressing changes to complement wound cleaning, irrigation and debridement reduces bacterial colonization, biofilm formation and reformation. This principle has also been used successfully in NPWT and instillation techniques. Polyhexamethylene biguanide (PHMB) has been shown to be effective for NPWT-instillation but other antiseptics such as cadexomer-iodine and silver as irrigants or in dressings are also effective in control of bacterial colonization and biofilm formation (Allan et al. 2010; Andriesson and Eberlein 2006; Kaehn and Eberlein 2009; Phillips et al. 2010b; Sibbald et al. 2011; Leaper et al. 2011, 2012, 2013; Back et al. 2013; Dowsett 2013; NIHCE 2013). The use of topical antimicrobials such is more effective after the biofilm has been disrupted by debridement (Dowsett 2013).
The practicality of who is responsible for maintenance debridement, or who is qualified to undertake it, may be abrogated by the use of effective but slower techniques such as enzymatic, larva-therapy or autolytic debridement. A simple technique that can be used by a wide range of general practitioners involves the use of a soft, monofilament fiber pads which can be used to regularly “brush” the wound, just as a biofilm can brushed off teeth and gingiva (White 2011). This technique however is somewhat superficial and does not compare with sharp, surgical or loop curette debridement or even the use of a surgical brush. The requirement for a qualified wound nurse to oversee this process has been questioned but regardless there are plenty of opportunities to learn and develop confidence with a myriad of debridement strategies, offered widely through wound healing societies and workshops (White 2011).
3.7 Future Strategies for Identify and Managing Chronic Surgical Wound
What’s next in biofilm research that will benefit the future of wound care? The use of a diagnostic criteria that would hasten the recognition of the presence of a biofilm at the patient bedside and give proof of adequate suppression after treatment will clearly be welcome (Alavi et al. 2012; Percival 2011). This could be based on PCR technology, which would help to “fingerprint” which micro-organisms were present in wound and in what numbers, despite a negative conventional swab and microbiological analysis. Other diagnostic strategies might include detection of signaling molecules, bacterial products or the use of host cell lines. Alternative innovative diagnostics could also aid in deciding on the best method of general and maintenance debridement, including new technologies, together with monitoring and how often debridement would be needed, and specific targeting with antimicrobials (Attinger and Wolcott 2012; Dissemond et al. 2011). The eradication of wound biofilm, once there are accurate diagnostics, may come with the development of quorum-sensing inhibitors or other anti-biofilm modalities (Rhoads et al. 2008; Sun et al. 2008; Wolcott and Cox 2013; Zhao et al. 2013). In acute wounds, which are at risk, perhaps the targeted prophylactic use of antimicrobial agents may become an appropriate element of an effective wound care bundle.
3.8 Treatment and Management of Biofilm-Mediated Infections
At present there are no evidence-based studies focusing on the therapeutic efficacy of selective strategies for managing biofilm-associated surgical site infections. However, in general the current therapeutic options can be characterized succinctly as follows:
-
(a)
Tissue-Based Infection: Surgical debridement to remove devitalized tissue, followed by copious irrigation preferable with a biocide agent followed by parenteral antibiotics (Barnes et al. 2014; Cardinal et al. 2009; Edmiston et al. 2013c; Leaper et al. 2011).
-
(b)
Device-Related Infection: Removal of an infected device followed by insertion of antimicrobial adjunctive technology such as antimicrobial spacer, beads or suture technology plus parenteral antibiotics (Del Pozo and Patel 2009; Edmiston et al. 2013d; Griffin et al. 2012).
-
(c)
Antimicrobial Agents: Selection of therapeutic or agents that appear to penetrate microbial biofilms include linezolid, daptomycin, rifampin and possibly ceftaroline (Edmiston et al. 2006b; Seaton et al. 2013; Barber et al. 2014).
References
Akers KS, Mende K, Cheatle K, Zera WC, Yu X, Beckius ML, Aggarwal D, Carlos PL, Sanchez CJ, Wenke JC, Weintrob AC, Tribble DR, Murray CK (2014) Biofilms and persistent wound infections in United States military trauma patients a case-control analysis. BMC Infect Dis 14:190. http://www.biomedcentral.com/1471-2334/14/190
Akiyama H, Ueda M, Kanwaki H, Tada J, Arata J (1997) Biofilm formation of Staphylococcus aureus strains isolated from impetigo and furuncle: role of fibrinogen and fibrin. J Dermatol Sci 16:2–10
Alavi MR, Stojadinovic A, Izadjoo MJ (2012) An overview of biofilm and its detection in clinical samples. J Wound Care 21:376–383
Allan N, Olson M, Nagel D, Martin R (2010) The impact of hydrosurgical debridement on wounds containing bacterial biofilms. Wound Repair Regen 18:A88
American Society of Plastic Surgeon (2011) Plastic surgery procedural statistics. http://www.plasticsurgery.org.news-and-resources. Accessed 20 Jan 2013
Andriesson AE, Eberlein T (2006) Assessment of a wound cleansing solution in the treatment of problem wounds. Wounds 20:171–175
Armstrong PA, Bandyk (2006) Management of infected aortic grafts by in-situ grafting. In: Pearce WH, Matsumura JS, Yao JST (eds) Trends in vascular surgery 2005. Greenwood Academic, Evanston, pp 473–48
Attinger C, Wolcott R (2012) Clinically addressing biofilm in chronic wounds. Adv Wound Care 1:127–132
Back DA, Scheuermann-Poley C, Willy C (2013) Recommendations on negative pressure wound therapy with instillation and antimicrobial solutions – when, where and how to use: what does the evidence show? Int Wound J 10:32–42
Bandyk DF, Black MR (2005) Infection in prosthetic vascular grafts. In: Rutherford RB, Johnson KW (eds) Vascular surgery, 6th edn. Elsevier Saunders, Philadelphia, pp 875–6894
Barber KE, Werth BJ, McRoberts JP, Rybak MJ (2014) A novel approach utilizing biofilm time-kill curves to assess the bactericidal activity of ceftaroline combinations against biofilm-producing methicillin-resistant Staphylococcus aureus. Antimicrob Agent Chemother 58:2989–2992
Barnes S, Spencer M, Graham D, Johnson HB (2014) Surgical wound irrigation: a call for evidence-based standardization of practice. Am J Infect Control 42:525–529
Bauer JJ, Harris MT, Kreel I, Gelernt IM (1999) Twelve-year experience with expanded polytetrafluorethylene in the repair of abdominal wall defects. Mt Sinai J Med 66:20–25
Bellon JM, Garcia-Carranza A, Garcia-Honduvilla N, Carrera-San Martin A, Bujan J (2004) Tissue integration and biomechanical behavior of contaminated experimental polypropylene and expanded polytetrafluoroethylene implants. Br J Surg 91:489–494
Bigger JW (1944) Treatment of staphylococcal infection with penicillin. Lancet 2:497–500
Birolini C, de Miranda JS, Utiyama EM, Rasslan S (2014) A retrospective review and observation over a 16-year clinical experience on the surgical treatment of chronic mesh infection. What about replacing a synthetic mesh on the infected surgical field? Hernia Feb 6 [Epub ahead of print]
Bjarnsholt T, Kirketerp-Møller K, Jensen PØ, Madsen KG, Phipps R, Krogfelt K, Høiby N, Givskov M (2008) Why chronic wounds will not heal: a novel hypothesis. Wound Repair Regen 16:2–10
Bondurant CP, Jimenez DF (1995) Epidemiology of cerebrospinal fluid shunting. Pediatr Neurosurg 23:254–258
Braxton EE, Ehrlich GD, Hall-Stoodley L, Stoodley P, Veeh R, Fux C, Hu FZ, Quigley M, Post C (2005) Role of biofilms in neurological device-related infections. Neurosurg Rev 28:249–255
Cameron T (2004) Safety and efficacy of spinal cord stimulation for the treatment of chronic pain: a 20-year literature review. J Neurosurg Spine 100:24–257
Caputo WJ, Beggs DJ, DeFede JL, Simm L, Dharma H (2008) A prospective randomized controlled clinical trial comparing hydrosurgery debridement with conventional surgical debridement in lower extremity ulcers. Int Wound J 5:288–294
Cardinal M, Eisenbud DE, Armstrong DG, Zelen C, Driver V, Attinger C, Phillips T, Harding K (2009) Serial surgical debridement: a retrospective study on clinical outcomes in chronic lower extremity wounds. Wound Repair Regen 17:306–311
Centers for Disease Control and Prevention (2009) Ambulatory surgery in the United States, 2006, National Health Statistics Report, Number 11. http://www.cdc.gov/nchs/data/nhsr/nhsr011.pdf. Revised 4 September 2009
Centers for Disease Control and Prevention (2010) National hospital discharge survey: 2010 table, procedures by selected patient characteristics – number by procedure category and age. http://www.cdc.gov/nchs/fastats/insurg.htm. Accessed 27 Aug 2013
Cho KH, Caparon MG (2005) Patterns of virulence gene expression differ between biofilm and tissue communities of Streptococcus pyogenes. Mol Microbiol 57:1545–1556
Costerton JW, Stewart PS (2001) Battling biofilms. Sci Am 285:74–81
Costerton JW, Lewandowski Z, Caldwell DE, Korber DR, Lappin-Scott HM (1995) Microbial biofilms. Annu Rev Microbiol 49:711–745
Costerton JW, Stewart PS, Greenberg EP (1999) Bacterial biofilms: a common cause of persisting infections. Science 284:1318–1322
Cutting KF, Harding KG (1994) Criteria for identifying wound infection. J Wound Care 3:198–201
Davidson CA, Lowe CR (2004) Optimization of polymeric surface pre-treatment to prevent bacterial biofilm formation for use in microfluidics. J Mol Recognit 25:2029–2037
Davis LE, Cook G, Costerton JW (2002) Biofilm on ventriculoperitoneal shunt tubing as a cause of treatment failure in coccidioidal meningitis. Emerg Infect Dis 8:376–379
Davis SC, Ricotti C, Cazzaniga, Welsh E, Eaglstein WH, Mertz PM (2008) Microscopic and physiologic evidence for biofilm-associated wound colonization in vivo. Wound Repair Regen 16:23–29
De Lissovoy G, Fraeman K, Hutchins V, Murphy D, Song D, Vaughn BB (2009) Surgical site infection: incidence and impact on hospital utilization and treatment costs. Am J Infect Control 37:387–397
Del Pozo JL, Patel R (2009) Clinical practice. Infection associated with prosthetic joints. N Eng J Med 361:787–794
Dissemond J, Assadian O, Gerber V, Kingsley A, Kramer A, Leaper DJ, Mosti G, Piatkowski DE, Grzymala A, Riepe G, Risse A, Romanelli M, Strohal R, Traber J, Vasel-Biergans A, Wild T, Eberlein T (2011) Classification of wounds at risk and their antimicrobial treatment with polyhexanide: a practice-oriented expert recommendation. Skin Pharmacol Physiol 24:245–255
Donlan RM (2001a) Biofilm formation: a clinically relevant microbiological process. Clin Infect Dis 33:1387–1392
Donlan RM (2001b) Role of biofilms in antimicrobial resistance. ASAIO J 46:547–552
Dowd SE, Wolcott RD, Sun Y, McKeehan T, Smith E, Rhoads D (2008) Polymicrobial nature of chronic diabetic foot ulcer infections determined using bacterial tag encoded FLX amplicon pyrosequencing (bTEFAP). PLoS One 3:e3326
Dowsett C (2013) Biofilms: a practice-based approach to identification and treatment. Wounds UK 9:68–92
Edds EM, Bergamini TM, Brittian KR, Kenneth R (2000) Bacterial components inhibit fibroblast proliferation in vivo. ASAIO J 46:33–37
Edmiston CE (1993) Prosthetic device infections in surgery. In: Nichols RL, Nyhus LM (eds) Update surgical sepsis. JB Lippincott, Philadelphia, pp 444–468
Edmiston CE, Seabrook GR, Cambria RA, Brown KR, Sommers JR, Krepel CJ, Wilson PJ, Sinski S, Towne JB (2005) Molecular epidemiology of microbial contamination in the operating room environment: is there a risk for infection? Surgery 138:572–588
Edmiston CE, Seabrook GR, Goheen MP, Krepel CJ, Johnson CP, Lewis BD, Brown KR, Towne JB (2006a) Bacterial adherence to surgical sutures: can antimicrobial-coated sutures reduce the risk of microbial contamination? J Am Coll Surg 203:481–489
Edmiston CE, Goheen MP, Seabrook GR, Johnson CP, Lewis BD, Brown KR, Towne JB (2006b) Impact of selective antimicrobial agents on staphylococcal adherence to biomedical devices. Am J Surg 192:344–354
Edmiston CE, Krepel CJ, Marks RM, Rossi PJ, Sanger J, Goldblatt M, Graham MB, Rotherburger S, Collier J, Seabrook GR (2013a) Microbiology of explanted sutures segments from infected and non-infected surgical cases. J Clin Microbiol 51:417–421
Edmiston CE, Kiernan M, Leaper DJ (2013b) The Ying and Yang of pre-operative screening for methicillin sensitive Staphylococcus aureus (MSSA): would the extra effort and cost of decolonization reduce surgical site infections? Wound Med 1:7–12
Edmiston CE, Daoud F, Leaper DJ (2013c) Is there an evidence-based argument for embracing an antimicrobial (triclosan)-coated suture technology to reduce the risk for surgical-site infections?: a meta-analysis. Surgery 154:89–100
Edmiston CE, Bruden B, Rucinski M, Henen C, Graham MB, Lewis BL (2013d) Reducing the risk of surgical site infections: does chlorhexidine gluconate provide a risk reduction benefit? Am J Infect Control 41:S49–S55
Edward R, Harding KG (2004) Bacteria and wound healing. Curr Opin Infect Dis 17:91–96
Elek SD, Cohen PE (1957) The virulence of S. pyogenes for man: a study of the problems of wound infection. Br J Exp Pathol 38:573–586
Elgharaby H, Mann E, Awad H, Ganesh K, Ghatak PD, Gordillo G, Sai-Sudhakar CB, Sashwati R, Wozniak DJ (2013) First evidence of sternal wound biofilm following cardiac surgery. PLoS One 8:e70360
Engelsman AF, van der Mei HC, Ploeg RJ, Busscher HJ (2007) The phenomenon of infection with abdominal wall reconstruction. Biomaterials 28:2314–2327
Engelsman AF, van der Mei HC, Busscher HJ, Ploeg RJ (2008) Morphological aspects of surgical mesh as a risk factor for bacterial colonization. Br J Surg 95:1051–1059
Evans LV, Lewandowski Z (2000) Structure and function of biofilms. In: Evans LV (ed) Biofilms: recent advances in their study and control. Harwood Academic, Amsterdam, pp 1–17
Falanga V, Brem H, Ennis WJ, Wolcott R, Gould LJ, Ayello EA (2008) Maintenance debridement in the treatment of difficult-to-heal chronic wounds. Ostomy Wound Manag S2–S13
Fernandes A, Dias M (2013) The microbiological profile of infected prosthetic implants with an emphasis on which organisms form biofilms. J Clin Diagn Res 7:219–223
Frei E, Hodgkiss-Harlow K, Rossi PJ, Edmiston CE, Bandyk DF (2011) Microbial pathogenesis of bacterial biofilms: a causative factor of vascular surgical site infection. Vasc Endovasc Surg 45:688–696
Gardner SE, Frantz RA, Doebbeling BN (2001) The validity of the clinical signs and symptoms used to identify localized chronic wound infection. Wound Repair Regen 9:178–186
Gilbert P, McBain AJ (2001) Biofilms: their impact on health and their recalcitrance toward biocides. Am J Infect Control 29:252–255
Gilbert P, Maira-Litran T, McBain AJ, Rikard AH, Whyte FW (2002) The physiology and collective recalcitrance of microbial biofilm communities. Adv Microb Physiol 46:202–256
Griffin JW, Guillot SJ, Redick JA, Browne JA (2012) Removed antibiotic-impregnated cement spacers in two-stage revision joint arthroplasty do not show biofilm formation in vivo. J Arthroplasty 27:1796–1799
Gunnison JB, Fraher MA, Jawetz E (1964) Persistence of Staphylococcus aureus in penicillin in vitro. J Gen Microbiol 35:335–349
Hall MR, McGillicuddy E, Kaplan LJ (2014) Biofilm: basic principles, pathophysiology, and implications for clinicians. Surg Infect 15:1–7
Hall-Stoodley L, Costerton JW, Stoodley P (2004) Bacterial biofilms from the natural environment to infectious disease. Nat Rev Microbiol 2:95–108
Hall-Stoodley L, Stoodley P, Kathju S, Høiby N, Moser C, Costerton JW, Moter A, Bjarnsholt T (2012) Towards diagnostic guidelines for biofilm-associated infections. FEMS Immunol Med Microbiol 65:127–145
Hamy A, Pessaux P, Mucci-Hennekinne S, Radriamananjo A, Regenet N, Arnaud JP (2003) Surgical treatment of large incisional hernias by intraperitoneal Dacron mesh and an aponeurotic graft. J Am Coll Surg 196:531–534
Harrison-Balestra C, Cazzaniga AL, Davis SC, Mertz PM (2003) A wound-isolated Pseudomonas aeruginosa grows a biofilm in vitro within 10 hours and is visualized by light microscopy. Dermatol Surg 29:631–635
Hart JP, Eginton MT, Brown KR, Seabrook GR, Lewis BD, Edmiston CE, Towne JB, Cambria RA (2005) Operative strategies in aortic graft infections: is complete graft excision always necessary? Ann Vasc Surg 19:154–160
Hasanadka R, Seabrook GR, Edmiston CE (2007) Vascular graft infections. In: Rello J, Vanes J, Kollef M (eds) Critical care infectious diseases. Kluwer Academic, Boston, pp 555–566
Henry-Stanley MJ, Hess DJ, Barnes AMT, Dunny GM, Wells CL (2010) Bacterial contamination of surgical suture resembles a biofilm. Surg Infect 11:433–439
Herwaldt LA, Cullen JJ, Scholz D, French P, Zimmerman MB, Pfaller MA, Wenzel RP, Pearl TM (2006) A prospective study of outcome, healthcare resource utilization, and cost associated with postoperative nosocomial infections. Infect Control Hosp Epidemiol 27:1291–1298
Hoiby N, Ciofu O, Johansen HK, Song ZJ, Moser C, Jensen PO, Molin S, Givskov M, Tolker-Nielsen T, Bjarnsholt T (2011) The clinical impact of bacterial biofilms. Int J Oral Sci 3:55–65
Holland SP, Pulido JS, Miller D, Ellis B, Alfonso E, Scott M, Costerton JW (1991) Biofilm and scleral buckle-associated infections: a mechanism for persistence. Ophthalmology 98:933–938
Hoyle B, Corsterton JW (1991) Bacterial resistance to antibiotics: the role of biofilm. Prog Drug Res 37:91–105
Jacombs A, Tahir S, Hu H, Deva AK, Almatroudi A, Fick WL, Bradshaw DA, Vickery K (2014) In vitro and in vivo investigation of the influence if implant surface on the formation of bacterial biofilm in mammary implants. Plast Reconstr Surg 133:e471–e2475
James GA, Swogger E, Wolcott R, Pulcini E, Secor P, Sestrich J, Costerton JW, Stewart PS (2008) Biofilms in chronic wounds. Wound Repair Regen 16:37–44
Jayaraman R (2008) Bacterial persistence: some new insights into an old phenomenon. J Biosci 33:795–805
Jenson PO, Givskov M, Bjarnsholt T, Moser C (2011) The immune system vs. Pseudomonas aeruginosa biofilms. FEMS Immunol Med Microbiol 59:292–305
Kaehn K, Eberlein T (2009) In vitro test for comparing the efficacy of wound rinsing solutions. Br J Nurs 18:S4–S10
Kathju S, Nistico L, Hall-Stoodley L, Post JC, Ehrlich GD, Stoodley P (2009) Chronic surgical site infection due to suture-associated polymicrobial biofilm. Surg Infect 10:457–461
Kathju S, Lask LA, Nistico L, Colella JJ, Stoodley P (2010) Cutaneous fistula from gastric remnant resulting from chronic suture-associated biofilm infection. Obes Surg 20:251–256
Kessler B, Sendi P, Graber P, Knupp M, Zwicky L, Hintermann B, Zimmerli W (2012) Risk factors for periprosthetic ankle joint infection: a case control study. J Bone Joint Surg Am 94:1871–1876
Kim DH, Spencer M, Davidson SM, Li L, Shaw JD, Gulczynski D, Hunter DJ, Martha JF, Miley GB, Parazin SJ, Dejoie P, Richmond JC (2010) Institutional prescreening for detection and eradication of methicillin-resistant Staphylococcus aureus in patients undergoing elective orthopaedic surgery. J Bone Joint Surg 92:1820–1826
Kingsley A (2001) A pro-active approach to wound infection. Nurs Stand 15:50–58
Laffer RR, Graber P, Ochsner PE, Zimmerli W (2006) Outcome of prosthetic knee-associated infection: evaluation of 40 consecutive episodes at a single center. Clin Microbiol Infect 15:433–443
Leaper D (2013) Silver dressings: their role in treatment of acute infected wounds. In: Willy C (ed) Antiseptics in surgery-update 2013. Lindqvist Book Publishing, Berlin, pp 151–154
Leaper DJ, Meaume S, Apelqvist J, Teot L, Gottrup F (2011) Debridement methods of non-viable tissue in wounds. In: Farrar D (ed) Advanced wound repair therapies. Woodhead Publishers, Cambridge
Leaper DJ, Ayello EA, Carville K, Fletcher J, Keast D, Lindholm C, Martinez JLL, Mavanini SD, McBain A, Moore Z, Opasanon S, Pina E (2012a) Appropriate use of silver dressings in wounds. International Consensus Document, London
Leaper DJ, Schultz G, Carville K, Fletcher F, Swanson T, Drake R (2012b) Extending the TIME concept: what have we learned in the past 10 years? Int Wound J 9:1–19
Leber GE, Garb JL, Alexander AI, Reed WP (1998) Long-term complications associated with prosthetic repair of incisional hernias. Arch Surg 133:378–382
Leid JG, Wilson CJ, Shirtliff ME, Hassett DJ, Parsek MR, Jeffers AK (2005) The exopolysaccharide alginate protects biofilm from IFN-gamma-mediated macrophage killing. J Immunol 175:7512–7518
Lozier AP, Sciacca RR, Romagnoli MF, Connolly ES (2002) Ventriculostomy-related infections; a critical review of the literature. Neurosurgery 51:170–181
Luijendiik RW, Hop WCJ, van der Tol MP, de Lange DCD, Braaksma MMJ, Ijzermans JNM, Boelhouwer RU, de Vries BC, Salu MK, Wereldsma JC, Bruijninckx CM, Jeekel J (2000) A comparison of suture repair and mesh repair for incisional hernia. N Engl J Med 343:392–398
Machairas A, Misiakos EP, Liakakos T, Karatzas G (2004) Incisional hernioplasty with extraperitoneal on-lay polyester mesh. Ann Surg 70:726–729
Mah TF, O’Toole GA (2001) Mechanisms of biofilm resistance to antimicrobial agents. Trends Microbiol 9:34–39
Massie JB, Heller JG, Abitbol JJ, McPherson D, Garfin SR (1992) Postoperative posterior spinal wound infections. Clin Orthop 284:99–108
Mauermann WJ, Sampathkumar P, Thompson RL (2008) Sternal wound infections. Best Pract Clin Anesthesiol 22:423–436
Meier-Davis S (2006) Host response to biofilms, foreign implants and devices. In: Pace JL, Mark E, Rupp Roger GF (eds) Biofilms, infection and antimicrobial therapy. Taylor & Francis Group, Boca Raton, pp 315–317
Ming X, Nichols M, Rothenburger S (2007) In-vivo antibacterial efficacy of Monocryl Plus® antibacterial (poliglecaprone 25 with triclosan) suture. Surg Infect (Larchmt) 8:1–5
Mosti G, Iabichella ML, Picerni P, Magliaro A, Mattaliano V (2005) The debridement of hard to heal leg ulcers by means of a new device based on Fluid jet technology. Int Wound J 2:307–314
Nahabedian MY, Tsangaris T, Momen B, Manson PN (2003) Infectious complications following breast reconstruction with expanders and implants. Plast Reconstr Surg 112:467–476
National Institute of Health (2002) Research on microbial biofilms: PA Number: PA-03-047. http://grants.nih.gov/grants/guide/pa-files/PA-03-047.html
National Institute for Health and Clinical Excellence (2013) NICE medical technology scope: Debrisoft for the debridement of acute and chronic wounds. Available at: www.nice.org.uk/nicemedia/live/14185/64407/64407.pdf
Nemoto K, Hirota K, Ono T, Murakami K, Nagao D, Miyake Y (2000) Effect of varidase (streptokinase) on biofilm formed by Staphylococcus aureus. Chemotherapy 46:111–115
Neto A, Lozano M, Moro MT, Keller J, Carralafuenta C (2002) Determinants of wound infection after surgery for breast cancer. Zentralbl Gynakol 124:429–433
Netscher DT (2004) Subclinical infection as a possible cause of significant breast capsules. Plast Reconstr Surg 113:2229–2230
Olsen MA, Chu-Ongsakul S, Brandt KE, Dietz JR, Mayfield J, Fraser VJ (2008) Hospital-associated cost due to surgical site infection after breast surgery. Arch Surg 143:53–56
Otto M (2008) Staphylococcal biofilm. Curr Top Microbiol Immunol 322:207–228
Owen S, Ramraj V, Wallop J (2010) The cardiac surgery advance practice group: a case study of APN and PA collaborative practice. J Nurs Pract 6:371–374
Pandey R, Berendt AR, Athanasou NA (2000) Histological and microbiological findings in non-infected and infected revision arthroplasty tissue. The OSIRIS Collaborative Study Group, Oxford Skeletal Infection Research and Intervention Service. Arch Orthop Trauma Surg 120:570–574
Parsa K, Schaudinn C, Gorur A, Sedghizadeh PP, Johnson T, Tse DT, Costerton JW (2010) Demonstration of bacterial biofilms in culture-negative silicone stent and jones tubes. Ophthal Plast Reconstr Surg 26:426–430
Parsek MR, Greenberg EP (2005) Sociomicrobiology – the connection between quorum sensing and biofilms. Trends Microbiol 13:27–33
Percival SL (2004) Biofilms and their potential role in wound healing. Wound 16:234–240
Percival SL (2011) Biofilms and their management: form concept to clinical reality. J Wound Care 20:220–226
Percival SL, Thomas JG, Williams DW (2010) Biofilms and bacterial imbalances in chronic wounds: anti-Koch. Int Wound J 7:169–175
Percival SL, Hill KE, Williams DW, Hooper SJ, Thomas DW, Costerton JW (2012) A review of the scientific evidence for biofilms in wounds. Wound Repair Regen 20:647–657
Phillips PL, Wolcott RD, Fletcher J, Schultz GS (2010a) Biofilms made easy. Wounds Int 1:1–6
Phillips PL, Yang Q, Sampson E, Schultz G (2010b) Effects of antimicrobial agents on an in vitro biofilm model of skin wounds. Adv Wound Care 1:299–304
Raju DR, Jindrak K, Weiner M, Enquist IF (1977) A study of the critical bacterial inoculums to cause a stimulus to wound healing. Surg Gynecol Obstet 144:347–350
Ray JM, Triplett G (2011) What is the role of biofilms in severe head and neck infections? Oral Maxillofac Surg Clin North Am 23:497–505
Reed D, Kemmerly SA (2009) Infection control and prevention: a review of hospital-acquired infections and the economic implications. Ochsner J 9:27–31
Rhoads DD, Wolcott RD, Percival SL (2008) Biofilms in wounds management strategies. J Wound Care 17:502–508
Rieger UM, Kalbermatten DF, Haug M, Frey HP, Pico R, Frei R, Pierer G, Luscher NJ, Trampuz A (2013) Bacterial biofilms and capsular contracture in patients with breast implants. Br J Surg 100:768–774
Romling U, Balsalobre C (2012) Biofilm infections, their resilience to therapy and innovative treatment strategies. J Intern Med 272:541–561
Rothenburger S, Spangler D, Bhende S, Burkley D (2002) In-vitro antimicrobial evaluation of coated Vicryl Plus antibacterial suture (coated polyglactin 910 with triclosan) using zone of inhibition assay. Surg Infect 3:S79–S87
Samini DB, Bielory BP, Miller D, Johnson TE (2013) Microbiologic trends and biofilm growth on explanted periorbital biomaterials: a 30-year review. Opthal Plast Reconstr Surg 29:376–381
Sanchez CJ, Mende K, Beckius ML, Akers KS, Romano DR, Wenke JC, Murray CK (2013) Biofilm formation by clinical isolates and the implication in chronic infections. BMC Infect Dis 13:47. http://www.biomedcentral.com/1571-2334/13/47
Sauer K, Camper AK, Ehrlich GD, Costerton JW, Davies DG (2002) Pseudomonas aeruginosa displays multiple phenotypes during development as a biofilm. J Bacteriol 184:1140–1154
Scott NW, McCormack K, Graham P, Go PM, Ross SJ, Grant AM (2002) Open mesh versus non-mesh repair for femoral and inguinal hernia. Cochrane Database Syst Rev CD002197
Seaton RA, Malizos KN, Viale P, Gargalianos-Kakolyris P, Santantonio T, Petrelli E, Pathan R, Heep M, Chaves R (2013) Daptomycin use in patients with osteomyelitis: a preliminary report from the EU-COREsm database. J Antimicrob Chemother 68:1642–1649
Shepard J, Ward W, Milstone A, Carlson T, Frederick J, Hadhazy E, Perl T (2013) Financial impact of surgical site infections on hospital: the hospital management perspective. JAMA Surg. doi:10.1001/jamasurg.2013.2246
Sibbald RG, Leaper DJ, Queen D (2011) Iodine made easy. Wounds Int 2:s1–s6
Singh PK, Parsek MR, Greenberg EP, Welsh MJ (2002) A component of innate immunity prevents bacterial biofilm development. Nature 417:552–555
Spoering AL, Lewis K (2001) Biofilms and planktonic cells of Pseudomonas aeruginosa has similar resistance to killing by antimicrobial. J Bacteriol 183:6746–6751
Steckelberg JM, Osmom DR (2000) In: Waldvogel FA (ed) Prosthetic joint infection: infections associated with indwelling medical devices, 3rd edn. ASM Press, Washington, DC, pp 173–205
Stewart PS, Costerton JW (2001) Antibiotic resistance of bacteria in biofilms. Lancet 358:135–138
Stoodley P, Sauer K, Davies DG, Costerton JW (2002) Review. Biofilms as complex differentiated communities. Annu Rev Microbiol 56:187–209
Subramani K, Jung RE, Molenberg A, Hammerle CH (2009) Biofilm on dental implants: a review of the literature. Int J Oral Maxillofac Implants 24:616–626
Sugita J, Yokoi N, Fullwood NJ, Quantock AJ, Takada Y, Nakamura Y, Kinoshita S (2001) The detection of bacterial biofilms in punctual plug holes. Cornea 20:362–365
Sun Y, Dowd SE, Smith E, Rhoads DD, Walcott RD (2008) In vitro multispecies Lubbock chronic wound biofilm model. Wound Repair Regen 16:805–813
Tran CL, Langer S, Broderick-Villa G, DiFronzo LA (2002) Does reoperation predispose to postoperative wound infection in women undergoing operation for breast cancer? Am Surg 69:852–856
Tsoi B, Ziolkowoski NI, Thoma A, Campbell K, O’Reilly D, Goeree R (2014) Safety of tissue expander/implant versus autologous abdominal tissue breast reconstruction in postmastectomy breast cancer patients: a systematic review and meta-analysis. Plast Reconstr Surg 133:234–249
Umemura A, Jaggi JL, Hurthi HI, Siderowf AD, Colcher A, Stern MB, Baltuch GH (2003) Deep brain stimulation for movement disorder: morbidity and mortality in 109 patients. J Neurosurg 98:779–784
Vandeweyer E, Deraemaecker R, Nogaret JM, Hertens D (2003) Immediate breast reconstruction with implants and adjuvant chemotherapy: a good option? Acta Chir Belg 103:98–101
Vanwijck R, Kaba L, Boland S, Gonzales y Azero M, Delange A, Tourbach S (2010) Immediate skin grafting of sub-acute and chronic wounds debrided by hydrosurgery. J Plast Reconstr Aesthet Surg 63:544–549
Varma S, Ferguson HC, Breen DV, Lumb WV (1974) Comparison of seven suture materials in infected wounds: an experimental study. J Surg Res 17:165–170
Walker TS, Tomlin KL, Woethern GS, Poch KR, Lieber JG, Saavedra MT, Fessler MB, Malcolm KC, Vasil ML, Nick JA (2005) Enhanced Pseudomonas biofilm development mediated by human neutrophils. Infect Immun 73:3693–3701
White W (2011) Sharp wound debridement in the management of recalcitrant, locally infected chronic venous leg ulcers: a narrative review. Wound Pract Res 19:222–228
Williams DL, Costerton JW (2012) Using biofilms as initial inocula in animal models of biofilm-related infections. J Biomed Mater Res B Appl Biomater 100:1163–1169
Wolcott RD, Cox S (2013) More effective cell-based therapy through biofilm suppression. J Wound Care 22:26–31
Wolcott RD, Rhoads DD (2008) A study of biofilm-based wound management in subjects with critical limb ischemia. J Wound Care 17:145–155
Wolcott RD, Rhoads D, Dowd SE (2008) Biofilms and chronic wound inflammation. J Wound Care 17:333–341
Wolcott RD, Kennedy JP, Dowd SE (2009) Regular debridement is the main tool for maintaining a healthy wound bed in most chronic wounds. J Wound Care 18:54–56
Wolcott RD, Rhoads DD, Bennett ME, Wolcott BM, Gogokhia L, Costerton JW, Dowd SE (2010a) Chronic wounds and the medical biofilm paradigm. J Wound Care 19:45–53
Wolcott RD, Rumbaugh KP, Stewart PS, Dowd SE (2010b) Biofilm maturity studies indicate sharp debridement opens a time-dependent therapeutic window. J Wound Care 19:320–328
Wood HL, Holden SR, Bayston R (2001) Susceptibility of Staphylococcus epidermidis biofilm in CSF shunts to bacteriophage attack. Eur J Pediatr Surg 11:S56–S57
Zhao G, Usui ML, Lippmann SI, James GA, Stewart PS, Fleckman P, Olerud JE (2013) Biofilms and inflammation in chronic wounds. Adv Wound Care 2:389–399
Zimmerli W, Moser C (2012) Pathogenesis and treatment concepts of orthopedic biofilm infections. FEMS Immunol Med Microbiol 65:158–168
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Edmiston, C.E., McBain, A.J., Roberts, C., Leaper, D. (2015). Clinical and Microbiological Aspects of Biofilm-Associated Surgical Site Infections. In: Donelli, G. (eds) Biofilm-based Healthcare-associated Infections. Advances in Experimental Medicine and Biology, vol 830. Springer, Cham. https://doi.org/10.1007/978-3-319-11038-7_3
Download citation
DOI: https://doi.org/10.1007/978-3-319-11038-7_3
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-11037-0
Online ISBN: 978-3-319-11038-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)