Abstract
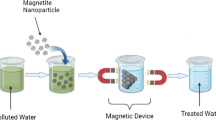
Recent advances in nanoscience suggest that the existing issues involving water quality could be resolved or greatly improved using nanomaterials, especially magnetic iron oxide nanoparticles. Magnetic nanoparticles have been synthesized for the development and use, in association with natural coagulant protein for water treatment. The nanoparticles size, morphology, structure, and magnetic properties were characterized by transmission electron microscope, X-ray diffraction, and superconducting quantum interference device magnetometry. Purified Moringa oleifera protein was attached onto microemulsions-prepared magnetic iron oxide nanoparticles (ME-MION) to form stable protein-functionalized magnetic nanoparticles (PMO+ME-MION). The turbidity removal efficiency in both synthetic and surface water samples were investigated and compared with the commonly used synthetic coagulant (alum) as well as PMO. More than 90 % turbidity could be removed from the surface waters within 12 min by magnetic separation of PMO+ME-MION; whereas gravimetrically, 70 % removal in high and low turbid waters can be achieved within 60 min. In contrast, alum requires 180 min to reduce the turbidity of low turbid water sample. These data support the advantage of separation with external magnetic field (magnetophoresis) over gravitational force. Time kinetics studies show a significant enhancement in ME-MION efficiency after binding with PMO implying the availability of large surface of the ME-MION. The coagulated particles (impurities) can be removed from PMO+ME-MION by washing with mild detergent or cleaning solution. To our knowledge, this is the first report on surface water turbidity removal using protein-functionalized magnetic nanoparticle.
This article is part of the Topical Collection on Nanotechnology for Sustainable Development
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
Keywords
References
Cloete TE (2010) Nanotechnology in water treatment applications. Caister Academic, Norfolk
Coetser SE, Heath RGM, Ndombe N (2007) Diffuse pollution associated with the mining sectors in South Africa: a first-order assessment. Water Sci Technol 55(3):9–16. doi:10.2166/Wst.2007.066
Deng YH, Wang CC, Hu JH, Yang WL, Fu SK (2005) Investigation of formation of silica-coated magnetite nanoparticles via sol-gel approach. Colloid Surface A 262(1–3):87–93. doi:10.1016/j.colsurfa.2005.04.009
Faraji M, Yamini Y, Rezaee M (2010) Magnetic nanoparticles: synthesis, stabilization, functionalization, characterization, and applications. J Iran Chem Soc 7(1):1–37
Ghebremichael KA, Gunaratna KR, Henriksson H, Brumer H, Dalhammar G (2005) A simple purification and activity assay of the coagulant protein from Moringa oleifera seed. Water Res 39(11):2338–2344. doi:10.1016/j.watres.2005.04.012
Grecu VV, Constantinescu S, Grecu MN, Olar R, Badea M, Turcu R (2008) Magnetic characterization of some nanometric iron oxides. Hyperfine Interact 183(1–3):205–214. doi:10.1007/s10751-008-9753-2
Lee EJ, Schwab KJ (2005) Deficiencies in drinking water distribution systems in developing countries. J water health 3(2):109–127
Li QL, Mahendra S, Lyon DY, Brunet L, Liga MV, Li D, Alvarez PJJ (2008) Antimicrobial nanomaterials for water disinfection and microbial control: potential applications and implications. Water Res 42(18):4591–4602. doi:10.1016/j.watres.2008.08.015
Liao MH, Chen DH (2002) Characteristics of magnetic nanoparticles-bound YADH in water/AOT/isooctane microemulsions. J Mol Catal B 18(1–3):81–87
Liu JF, Zhao ZS, Jiang GB (2008) Coating Fe3O4 magnetic nanoparticles with humic acid for high efficient removal of heavy metals in water. Environ Sci Technol 42(18):6949–6954. doi:10.1021/Es800924c
Lu AH, Salabas EL, Schuth F (2007) Magnetic nanoparticles: synthesis, protection, functionalization, and application. Angew Chem Int Ed 46(8):1222–1244. doi:10.1002/anie.200602866
Mikhaylova M, Kim DK, Bobrysheva N, Osmolowsky M, Semenov V, Tsakalakos T, Muhammed M (2004) Superparamagnetism of magnetite nanoparticles: dependence on surface modification. Langmuir 20(6):2472–2477. doi:10.1021/La035648e
Moe CL, Rheingans RD (2006) Global challenges in water, sanitation and health. J water health 4(Suppl 1):41–57
Okoli C, Boutonnet M, Mariey L, Jaras S, Rajarao G (2011a) Application of magnetic iron oxide nanoparticles prepared from microemulsions for protein purification. J Chem Technol Biot 86(11):1386–1393. doi:10.1002/Jctb.2704
Okoli C, Fornara A, Qin J, Toprak MS, Dalhammar G, Muhammed M, Rajarao GK (2011b) Characterization of superparamagnetic iron oxide nanoparticles and its application in protein purification. J Nanosci Nanotechnol 11(11):10201–10206. doi:10.1166/jnn.2011.5007
Sanchez-Martin J, Ghebremichael K, Beltran-Heredia J (2010) Comparison of single-step and two-step purified coagulants from Moringa oleifera seed for turbidity and DOC removal. Bioresour Technol 101(15):6259–6261. doi:10.1016/j.biortech.2010.02.072
Theron J, Walker JA, Cloete TE (2008) Nanotechnology and water treatment: applications and emerging opportunities. Crit Rev Microbiol 34(1):43–69. doi:10.1080/10408410701710442
Vorosmarty CJ, Green P, Salisbury J, Lammers RB (2000) Global water resources: vulnerability from climate change and population growth. Science 289(5477):284–288
Acknowledgments
The authors are grateful for the financial support provided by the Swedish research funding agency, Formas.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Additional information
Special Issue Editors: Mamadou Diallo, Neil Fromer,Myung S. Jhon
Rights and permissions
Copyright information
© 2012 Springer Science+Business Media B.V.
About this paper
Cite this paper
Okoli, C., Boutonnet, M., Järås, S., Rajarao-Kuttuva, G. (2012). Protein-functionalized magnetic iron oxide nanoparticles: time efficient potential-water treatment. In: Diallo, M.S., Fromer, N.A., Jhon, M.S. (eds) Nanotechnology for Sustainable Development. Springer, Cham. https://doi.org/10.1007/978-3-319-05041-6_10
Download citation
DOI: https://doi.org/10.1007/978-3-319-05041-6_10
Received:
Accepted:
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-05040-9
Online ISBN: 978-3-319-05041-6
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)