Abstract
Progress in the management of aneurysmal subarachnoid hemorrhage (SAH) is reflected most clearly in a continuously decreasing case fatality rate over the last decades. The purpose of the present review is to identify the relevant factors responsible for this progress and to outline future possibilities of improvement. Although data on intracerebral hemorrhage and ischemic stroke are less homogeneous, the respective data suggest that reduction of case fatalities could also be achieved with these types of stroke. Therefore, advances of general neurocritical care may be the common denominator responsible for the decreasing case fatality rates. Additionally, a change in practice with regard to treatment of elderly patients that is more active may also be a factor. Regarding SAH, the majority of unfavorable outcomes is still related to early or delayed cerebral injury. Therefore, efforts to pharmacologically prevent secondary neuronal damage are likely to play a certain role in achieving improvement in overall outcome. However, the data from previous randomized clinical trials conducted during the last three decades does not strongly support this. A clear benefit has only been proven for oral nimodipine, whereas other calcium antagonists and the rho-kinase inhibitors were not conclusively shown to have a significant effect on functional outcome, and all other tested substances disappointed in clinical trials. Regarding ischemic stroke and traumatic brain injury, intensive clinical research has also been conducted during the last 30 years to improve outcome and to minimize secondary neuronal injury. For ischemic stroke, treatment focusing on reversal of the primary pathomechanism, such as thrombolysis, proved effective, but none of the pharmacological neuroprotective concepts resulted in any benefit. To date, decompressive hemicraniectomy has been the only effective effort focused at reducing secondary damage that resulted in a clear reduction of mortality. In the case of traumatic brain injury, none of the pharmacological or other efforts to limit secondary damage met our hopes. In summary, although limited, pharmacotherapy to limit delayed neuronal injury is more effective for SAH than for ischemic stroke and traumatic brain injury. The disappointing results of most trials addressing secondary damage force one to question the general concept of mechanisms of secondary damage that do not also have a positive side in the natural course of the disease. For example, in the case of SAH, the data from the Cooperative Study from the 1960s showed that vasospasm to some degree protects against rerupture of unsecured aneurysms. Thus, one could argue from an evolutionary standpoint that the purpose of vasospasm was not exclusively a detrimental or suicide pathomechanism, but an attempt to protect against life-threating aneurysm rerupture. Because of the above-discussed arguments, SAH may indeed differ from ischemic stroke and traumatic brain injury with regard to the usefulness of blocking secondary mechanisms pharmacologically. Further efforts to limit vasospasm should therefore be made, and the most promising drugs, calcium antagonists, deserve further development. Because, with various drugs, systemic side effects counteracted the local beneficial effect, future efforts should focus on topical administration of drugs instead of systemic administration. Furthermore, efforts for a better understanding of the variations of the calcium channels and the interplay between the different types of calcium channels should be made.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Aneurysmal subarachnoid hemorrhage
- Cerebral vasospasm
- Secondary brain injury
- Delayed neurological deficit
- Secondary brain injury
- Neuroprotection
- Calcium channel vasospasm
What Has Perioperative “Management” of Subarachnoid Hemorrhage Achieved?
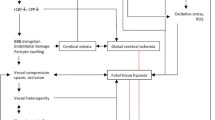
The most apparent progress in the treatment of aneurysmal subarachnoid hemorrhage (SAH) is reflected in the distinct reduction of the mortality [18]. Although data from 1980 indicated an overall case fatality rate of around 55 %, extrapolation suggests that it decreased to approximately 30 % around 2010 (Fig. 1). The reasons for this improvement are not obvious. Is the progress caused by better management of the specific complications of SAH or by better general intensive care unit (ICU) care? A recent analysis of our own cases treated during the last decade showed that at least 50 % of the case fatality rate with SAH is related to early or delayed brain injury [3]. Therefore, we concluded that changing fatality rates reflect, to a substantial degree, but not entirely, improved quality in the management of specific cerebral complications of SAH.
Decreasing case fatality rates with aneurysmal SAH over the last 3 decades (according to Lovelock et. al. [18]). Over the last 3 decades, case fatality rates continuously decreased in these population-based studies
Comparison with Other Stroke Entities
To gain a better feeling for the significance of prevention and correction of vasospasm for improved prognosis, it appears reasonable to look at other stroke entities with regard to the evolution of case fatality rates [4]. Here, we looked at the major population-based surveys on intracerebral hemorrhage and ischemic stroke (Figs. 2 and 3). In the case of intracerebral hemorrhage, the situation is confounded by massively changing demographics of this patient group [1, 2, 5, 17, 19, 20, 32]. The proportion of younger patients decreased in the Western countries clearly during the last three decades, probably mainly because of better control of arterial hypertension. Simultaneously, the proportion of older patients increased. The data found in the different surveys appear somewhat inhomogeneous. While, for example, in Dijon, a decrease of case fatalities of around 50 % was observed, no significant improvement was found in the United States or in Scotland [1, 2, 5]. For illustrative purposes, we also included the STICH data in the analysis, although the STICH cohort does not represent the population of a specific region [19, 20]. The STICH case fatality rates were substantially lower than those of the population-based surveys from the same time frame. Interestingly, the case fatality rates decreased by almost 50 % between the first and the second STICH trial, regardless of the different inclusion criteria in both trials (deep vs. lobar intracerebral hemorrhage, respectively). The relatively low case fatalities in the STICH trials, compared with other patient cohorts, confirm the assumption of a substantial selection bias in the STICH cohorts.
Inhomogeneously decreasing case fatality rates for spontaneous intracerebral hemorrhage. Overall, a similar trend is seen as with SAH, i.e., a decreasing case fatality rate. The trends at the different locations vary considerably. The low fatality rate in the STICH trials compared with population-based data is noteworthy
For ischemic stroke, there is also some heterogeneity with respect to geographical or demographical data [2, 27, 30, 31]. In general, as with cerebral hemorrhage, a trend toward decreasing case fatality rates is apparent. Persistent high case fatality rates were reported from Copenhagen and from Oxford, whereas overall data from the UK, Dijon, and Minnesota show steeply decreasing mortality rates.
Although the overall trends cannot be simplified and local factors may play a certain role, it is clear that progress with SAH may be comparable with other types of stroke. Here, improvement of neurocritical care may be the common denominator. On the other hand, it appears likely that changing attitudes accompanying aging populations are also a significant factor, e.g., treatment that is more invasive in patients older than 70–80 years or acceptance of functional deficits in elderly patients.
The Benefit of Neuroprotective Intervention in General
As highlighted, at least 50 % of the case fatality rate with SAH is related to early or delayed brain injury. Thus, a focus on the respective improvements of prevention and therapy should also result in beneficial effects on outcome. A review of the various prospective and randomized clinical trials of the last 30 years illustrates that only oral nimodipine has been definitely proven to improve outcome (Fig. 4). For other pharmacological interventions, a distinct but nonsignificant trend is seen, e.g., the rho kinase inhibitor, fasudil. However, the current smaller trials on statins seem to be promising in this respect, but larger trials are required to explore the potential of this drug type.
Summary of prospective randomized trials with aim to limit secondary damage after SAH (work in progress). In this analysis, all prospective, randomized, placebo-controlled trials addressing secondary mechanisms after SAH were included. The analysis was restricted to case fatality rate. Overall, a number of drugs proved to provide a favorable trend or clear-cut benefit, e.g., oral nimodipine and fasudil
Intensive preclinical and clinical research has been conducted during the last 30 years in the field of neuroprotection following ischemic stroke. Although efforts to reverse the initial injury, e.g., by thrombolysis, proved to be very effective, none of the pharmacologic concepts or other interventional concepts, such as hypothermia, for neuroprotection resulted in any beneficial effect on functional outcome. To date, decompressive craniotomy remains the only intervention focusing on limiting secondary brain damage that resulted in a clear reduction of mortality [11, 15].
Regarding traumatic brain injury, the data on the effect of neuroprotective strategies to limit secondary brain damage remain disappointing. To date, none of the larger clinical trials on pharmacological or interventional (hypothermia or decompressive craniectomy) treatments were able to demonstrate a beneficial effect on neurological outcome [6–8].
Common Denominators and Explanation
Looking at the clinical trials on corticosteroid therapy for meningitis may provide an explanation for the differences in benefit of neuroprotective measures (Fig. 5). Most of the few trials evaluating corticosteroids in addition to antibiotics for bacterial meningitis demonstrated a reduction of poor outcome and mortality [9, 10, 16, 21–26, 28]. It appears clear that steroids are only beneficial under concomitant treatment with antibiotics. Therefore, experimental treatment does not compare with the “natural” course for this entity, because corticosteroid treatment without antibiotic treatment would inevitably result in poor outcome or death in patients with bacterial meningitis.
Summary of prospective randomized trials on corticosteroids for meningitis (work in progress). Case fatality rate was used as the outcome parameter. In general, adding dexamethasone to antibiotic treatment reduces secondary complications. It is important to stress the point that dexamethasone is only beneficial under the protection of antibiotics. Therefore, experimental treatment does not compare with the “natural” course
There is a similar scenario in the case of SAH. Blocking secondary negative reactions by nimodipine has only been proven beneficial in the case of secured aneurysms, which means that the control group was not exposed to the true natural course of the disease. If we assume that evolution or nature has not developed “suicide pathomechanisms,” why would patients develop vasospasm during the course of this disease? For obvious reasons, there is no recent data on the natural course after aneurysmal SAH to underline this assumption, but the data from the Cooperative Study from the 1960s might provide some insight in this respect [14]. The registry dates back to the time before the introduction of early aneurysm surgery and can therefore provide information on the natural course after SAH. Although the data show a clear association between the occurrence of vasospasm and poor outcome, the data also show a clear negative association between the occurrence of vasospasm and aneurysm rerupture during the unsecured phase. Therefore, it appears that, in the natural course after SAH, macrovascular spasm might have been evolutionarily meant to protect patients against aneurysm rerupture. The protective effect of vasospasm was not readily evident in clinical practice, because risk factors to develop macrovascular spasm parallel those for rehemorrhage, i.e., severe SAH [29]. The protective effect of vasospasm against rerupture could only be distilled by multifactorial statistical correlation analysis. Thus, one could argue from an evolutionary standpoint that the purpose of macrovascular spasm was not exclusively a detrimental or suicide pathomechanism, but an attempt to protect against life-threating aneurysm rerupture.
The models of meningitis and SAH suggest that the concepts of secondary brain damage must be revised. For the natural course, nature does not appear to have developed suicide mechanisms that need to be fought, i.e., blocked pharmacologically. In contrast, nature has to limit resources for repair, and therefore also mechanisms of compensation following brain injury. Insufficient potential for repair and compensation need to be supported by medical intervention. On the other hand, current data do not indicate that blocking biochemical reactions after brain injury and stroke makes much sense. The pharmaceutical industry has realized that there is not much to gain and has largely withdrawn from drug development and clinical studies. Because of the above-discussed arguments, the situation may be different with SAH, and the most effective drugs, calcium antagonists, deserve further development.
Conclusions and Consequences
Nimodipine was introduced into clinical practice in the 1980s. Although we have a much more detailed understanding of the various types of calcium channels compared with 30 years ago, many open questions remain regarding calcium channels and their function following SAH. In particular, a variable response is seen after local application of nimodipine or intraarterial delivery. In a monocentric prospective trial at our department on intraarterial application of nimodipine for severe vasospasm, no response was seen in one of three patients [12]. The question of these nonresponders to calcium antagonists needs to be addressed. Are there substantial genetic variations of the calcium channels? How important is the balance between different types of calcium channels and how are they upregulated or downregulated?
Furthermore, in some clinical and experimental settings, the antispastic effect of nimodipine appears to decrease with increasing dose. We observed this effect in a study on the dose-related efficacy of continuous intracisternal nimodipine treatment for cerebral vasospasm in the rat double-SAH model [13]. The question of decreasing effect with increasing dose remains enigmatic and further research is needed to elucidate the underlying cause of the decreasing nimodipine effect at higher dosage. Differential effects on the individual types of calcium channels might play a role.
Last but not least, the concepts of systems theory defining the general principles governing the hierarchy and interactions between genes, cells, organs, the organism, and society should be considered more seriously by physicians and neuroscientists. A better understanding of systems theory would have made it clear that the assumption of suicide mechanisms in the brain rests on shaky grounds [8].
References
Andaluz N, Zuccarello M (2009) Recent trends in the treatment of spontaneous intracerebral hemorrhage: analysis of a nationwide inpatient database. J Neurosurg 110(3):403–410
Bejot Y, Rouaud O, Durier J, Caillier M, Marie C, Freysz M, Yeguiayan JM, Chantegret A, Osseby G, Moreau T, Giroud M (2007) Decrease in the stroke case fatality rates in a French population-based twenty-year study. A comparison between men and women. Cerebrovasc Dis 24:439–444
Beseoglu K, Holtkamp K, Steiger HJ, Hänggi D (2013) Fatal aneurysmal subarachnoid haemorrhage: causes of 30-day in-hospital case fatalities in a large single-centre historical patient cohort. Clin Neurol Neurosurg 115(1):77–81
Biotti D, Jacquin A, Boutarbouch M, Bousquet O, Durier J, Ben Salem D, Ricolfi F, Beaurain J, Osseby GV, Moreau T, Giroud M, Béjot Y (2010) Trends in case-fatality rates in hospitalized nontraumatic subarachnoid hemorrhage: results of a population-based study in Dijon, France, from 1985 to 2006. Neurosurgery 66(6):1039–1043
Christensen MC, Munro V (2008) Ischemic stroke and intracerebral hemorrhage: the latest evidence on mortality, readmissions and hospital costs from Scotland. Neuroepidemiology 30(4):239–246
Clifton GL, Miller ER, Choi SC, Levin HS, McCauley S, Smith KR Jr, Muizelaar JP, Wagner FC Jr, Marion DW, Luerssen TG, Chesnut RM, Schwartz M (2001) Lack of effect of induction of hypothermia after acute brain injury. N Engl J Med 344(8):556–563
Cooper DJ, Rosenfeld JV, Murray L, Arabi YM, Davies AR, D’Urso P, Kossmann T, Ponsford J, Seppelt I, Reilly P, Wolfe R, DECRA Trial Investigators; Australian and New Zealand Intensive Care Society Clinical Trials Group (2011) Decompressive craniectomy in diffuse traumatic brain injury. N Engl J Med 364(16):1493–1502
Drack M, Apfalter W, Pouvreau D (2007) On the making of a system theory of life: Paul A Weiss and Ludwig von Bertalanffy’s conceptual connection. Q Rev Biol 82(4):349–373
Escobar JA, Belsey MA, Dueñas A, Medina P (1975) Mortality from tuberculous meningitis reduced by steroid therapy. Pediatrics 56(6):1050–1055
Geiman BJ, Smith AL (1992) Dexamethasone and bacterial meningitis. A meta-analysis of randomized controlled trials. West J Med 157(1):27–31
Geurts M, van der Worp HB, Kappelle LJ, Amelink GJ, Algra A, Hofmeijer J, on behalf of the HAMLET Steering Committee (2013) Surgical decompression for space-occupying cerebral infarction: outcomes at 3 years in the randomized HAMLET trial. Stroke 44:2506–8
Hänggi D, Turowski B, Beseoglu K, Yong M, Steiger HJ (2008) Intra-arterial nimodipine for severe cerebral vasospasm after aneurysmal subarachnoid hemorrhage: influence on clinical course and cerebral perfusion. AJNR Am J Neuroradiol 29(6):1053–1060
Hänggi D, Eicker S, Beseoglu K, Rapp M, Perrin J, Nawatny J, Turowski B, Sommer C, Steiger HJ (2009) Dose-related efficacy of a continuous intracisternal nimodipine treatment on cerebral vasospasm in the rat double subarachnoid hemorrhage model. Neurosurgery 64(6):1155–1159
Henderson WG, Torner JC, Nibbelink DW, Henderson WG, Torner JC, Nibbelink DW (1977) Intracranial aneurysms and subarachnoid hemorrhage—report on a randomized treatment study. IV—B. Regulated bed rest— statistical evaluation. Stroke 8:579–589
Jüttler E, Schwab S, Schmiedek P, Unterberg A, Hennerici M, Woitzik J, Witte S, Jenetzky E, Hacke W, DESTINY Study Group (2007) Decompressive surgery for the treatment of malignant infarction of the middle cerebral artery (DESTINY): a randomized, controlled trial. Stroke 38(9):2518–2525
Lebel MH, Freij BJ, Syrogiannopoulos GA, Chrane DF, Hoyt MJ, Stewart SM, Kennard BD, Olsen KD, McCracken GH Jr (1988) Dexamethasone therapy for bacterial meningitis. Results of two double-blind, placebo-controlled trials. N Engl J Med 319(15):964–971
Lee S, Shafe ACE, Cowie MR (2011) UK stroke incidence, mortality and cardiovascular risk management 1999–2008: time-trend analysis from the General Practice Research Database. BMJ Open 1:e000269. doi:10.1136/bmjopen-2011-000269
Lovelock CE, Rinkel GJ, Rothwell PM (2010) Time trends in outcome of subarachnoid hemorrhage: population-based study and systematic review. Neurology 74(19):1494–1501
Mendelow AD, Gregson BA, Fernandes HM, Murray GD, Teasdale GM, Hope DT, Karimi A, Shaw MD, Barer DH, STICH investigators (2005) Early surgery versus initial conservative treatment in patients with spontaneous supratentorial intracerebral haematomas in the international surgical trial in intracerebral haemorrhage (STICH): a randomised trial. Lancet 365(9457):387–397
Mendelow AD, Gregson BA, Rowan EN, Murray GD, Gholkar A, Mitchell PM, for the STICH II Investigators (2013) Early surgery versus initial conservative treatment in patients with spontaneous supratentorial lobar intracerebral haematomas (STICH II): a randomised trial. Lancet 382:397–408. doi:10.1016/S0140-6736(13)60986-1, pii: S0140-6736(13)60986-1
Molyneux EM, Walsh AL, Forsyth H, Tembo M, Mwenechanya J, Kayira K, Bwanaisa L, Njobvu A, Rogerson S, Malenga G (2002) Dexamethasone treatment in childhood bacterial meningitis in Malawi: a randomised controlled trial. Lancet 360(9328):211–218
Mongelluzzo J, Mohamad Z, Ten Have TR, Shah SS (2008) Corticosteroids and mortality in children with bacterial meningitis. JAMA 299(17):2048–2055, PubMed [citation] PMID: 18460665
Nguyen TH, Tran TH, Thwaites G, Ly VC, Dinh XS, Ho Dang TN, Dang QT, Nguyen DP, Nguyen HP, To SD, Nguyen VC, Nguyen MD, Campbell J, Schultsz C, Parry C, Torok ME, White N, Nguyen TC, Tran TH, Stepniewska K, Farrar JJ (2007) Dexamethasone in Vietnamese adolescents and adults with bacterial meningitis. N Engl J Med 357(24):2431–2440
Odio CM, Faingezicht I, Paris M, Nassar M, Baltodano A, Rogers J, Sáez-Llorens X, Olsen KD, McCracken GH Jr (1991) The beneficial effects of early Dexamethasone administration in infants and children with bacterial meningitis. N Engl J Med 324(22):1525–1531, PubMed [citation] PMID: 2027357
Pécoul B, Varaine F, Keita M, Soga G, Djibo A, Soula G, Abdou A, Etienne J, Rey M (1991) Long-acting chloramphenicol versus intravenous ampicillin for treatment of bacterial meningitis. Lancet 338(8771):862–866
Qazi SA, Khan MA, Mughal N, Ahmad M, Joomro B, Sakata Y, Kuriya N, Matsuishi T, Abbas KA, Yamashita F (1996) Dexamethasone and bacterial meningitis in Pakistan. Arch Dis Child 75(6):482–488
Rothwell PM, Coull AJ, Giles MF, Howard SC, Silver LE, Bull LM, Gutnikov SA, Edwards P, Mant D, Sackley CM, Farmer A, Sandercock PA, Dennis MS, Warlow CP, Bamford JM, Anslow P, Oxford Vascular Study (2004) Change in stroke incidence, mortality, case-fatality, severity, and risk factors in Oxfordshire, UK from 1981 to 2004 (Oxford Vascular Study). Lancet 363(9425):1925–33
Scarborough M, Gordon SB, Whitty CJ, French N, Njalale Y, Chitani A, Peto TE, Lalloo DG, Zijlstra EE (2007) Corticosteroids for bacterial meningitis in adults in sub-Saharan Africa. N Engl J Med 357(24):2441–2450
Steiger HJ, Fritschi J, Seiler RW (1994) Current pattern of in-hospital aneurysmal rebleeds. Analysis of a series treated with individually timed surgery and intravenous nimodipine. Acta Neurochir (Wien) 127(1–2):21–26
Sturgeon JD, Folsom AR (2007) Trends in hospitalization rate, hospital case fatality, and mortality rate of stroke by subtype in Minneapolis-St. Paul, 1980–2002. Neuroepidemiology 28(1):39–45
Truelsen T, Grønbaek M, Schnohr P, Boysen G (2002) Stroke case fatality in Denmark from 1977 to 1992: the Copenhagen City Heart Study. Neuroepidemiology 21(1):22–27
van Asch CJ, Luitse MJ, Rinkel GJ, van der Tweel I, Algra A, Klijn CJ (2010) Incidence, case fatality, and functional outcome of intracerebral haemorrhage over time, according to age, sex, and ethnic origin: a systematic review and meta-analysis. Lancet Neurol 9(2):167–176
Conflict of Interest Statement
We declare that we have no conflict of interest
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Steiger, HJ., Beez, T., Beseoglu, K., Hänggi, D., Kamp, M.A. (2015). Perioperative Measures to Improve Outcome After Subarachnoid Hemorrhage—Revisiting the Concept of Secondary Brain Injury. In: Fandino, J., Marbacher, S., Fathi, AR., Muroi, C., Keller, E. (eds) Neurovascular Events After Subarachnoid Hemorrhage. Acta Neurochirurgica Supplement, vol 120. Springer, Cham. https://doi.org/10.1007/978-3-319-04981-6_36
Download citation
DOI: https://doi.org/10.1007/978-3-319-04981-6_36
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-04980-9
Online ISBN: 978-3-319-04981-6
eBook Packages: MedicineMedicine (R0)