Abstract
This chapter discusses how variation in intracytoplasmic Ca2+ concentration ([Ca2+]i) over time can be considered as a signal for the contractile machinery. It presents an overview of the literature that describes the Ca2+ response pattern to different contractile or relaxant agonists in several types of airway smooth muscle cells (ASMCs) depending on the species and location in the airway tree and recording methods, insisting on the temporal aspects of this response. Since the dynamics of the Ca2+ signal depends on the dynamics of the mechanisms responsible for this signal, the chapter presents an overview of the main mechanisms responsible for Ca2+ homeodynamics in ASMCs. By analyzing some examples, it shows how the kinetics of these mechanisms determine the pattern of the Ca2+ signal. The consequence of cell-to-cell variations in the Ca2+ signal is also discussed, with special attention to oscillatory versus nonoscillatory responses. The last part of the chapter presents the relationship between the parameter of the Ca2+ signal and the pattern of the contractile response. The mechanisms of the contractile apparatus itself will not be detailed, this question being beyond the scope of the chapter, but the temporal relationship between the Ca2+ signal and the subsequent contraction is analyzed.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 [Ca2+]i Variations as a Signal
1.1 What Makes Ca2+ Variation a Signal?
A Ca2+ signal is, by definition, a variation over time or space of the intracellular Ca2+ concentration that is detected by a given intracellular machinery, so that the behavior of the cell is altered according to the variation in Ca2+ concentration. In this chapter, we consider only temporal variation of cytosolic Ca2+ concentration ([Ca2+]i), which is a major determinant of airway smooth muscle cells (ASMC) contraction. Though the Ca2+ signal may encode for several cell functions, such as contraction, proliferation, and secretion, this chapter will consider only the contractile response of ASMCs as a cell function.
Time-dependent variations in [Ca2+]i is considered a signal insofar as they carry information that the intracellular machinery is able to decipher, i.e., is able to demonstrate a behavior specifically dependent on these [Ca2+]i variations, and that is why biologists study temporal variation in [Ca2+]i. Hence, what is of interest for biologists is what makes sense for the cell. This determines which parameters should be used to characterize Ca2+ signals and the relevant correlations that can be established between the variation in the intensity of stimulation and the [Ca2+]i response on the one hand, and between the parameters of the Ca2+ signal and the cellular functional response on the other hand. The scheme of the signaling pathway can be summarized as follows:
where the first step (1) corresponds to the encoding of the Ca2+ signal and the second step (2) to the decoding of the Ca2+ signal. A signal can be basically defined as the oriented relationship through a communication channel between an issuing system (source) and a recipient system (receptor). The information is what modifies the state or the evolution of the system, and the message is the expression of this information. A given Ca2+ signal may carry several distinct messages, insofar as the Ca2+ signal can be deciphered by distinct recipient systems. For example, a Ca2+ signal may induce contraction, proliferation, or secretion, for example, each behavior being determined by a given effector system capable of deciphering the Ca2+ signal. This chapter focuses on the contractile apparatus.
According to this scheme, in ASMCs, the source of the contractile information is the agonist/agonist receptor complex and the recipient system or final receptor is the contractile apparatus. A time-dependent variation in [Ca2+]i is the signal that carries the message, i.e., the contractile information. Hence, the temporal aspects of Ca2+ signals are at the heart of the notion of Ca2+ signal.
1.2 Signal and Message
The signal is the change in time or space of the cytosolic Ca2+ concentration. The message is the contractile information encoded in the signal. Hence, it is important to determine what makes a time-dependent variation in the concentration of free Ca2+ in the cytosol a message for the contractile machinery. Whatever the complexity of the mechanisms activated by an increase in [Ca2+]i, Ca2+ ions by themselves carry a message only if they bind to a target molecule. Hence, variation in [Ca2+]i is a message only if it induces a correlated change in Ca2+ binding to a transmitter molecule, a Ca2+-binding signaling protein. This corresponds to the schematic reaction
where M is the signaling molecule, and k + and k − are the kinetic constants of the reaction. Accordingly, any change in [Ca2+]i induced by an increase or decrease in the total amount of Ca2+ in the cytosol will induce a change in the concentration of the Ca2+-M complex. Hence, variations in [Ca2+]i can be viewed as carrying a message. However, this does not mean that the temporal pattern of the Ca2+-M complex, i.e., the message, is similar to that of the Ca2+ signal. This is true only if k + and k − are such that the reaction can be considered as instantaneous. Additionally, it should be noted that a given Ca2+ signal can carry distinct messages, not only in the sense that it activates different cell systems but also by the fact that the temporal sensitivity of the signaling molecules of the different recipient systems may differ.
1.3 Deciphering the Code
A signal is an encoded message. Deciphering the code entails determining how the cell converts the spatiotemporal change in [Ca2+]i into a given change in the contractile status of the ASMCs. More precisely, the identification of the mechanisms responsible for the changes in [Ca2+]i upon ASMC stimulation corresponds to the identification of the encoding mechanisms used by the Ca2+ homeodynamic system to convert the stimulation induced by the agonist into a Ca2+ signal, and the identification of the mechanisms responsible for the Ca2+-contraction coupling means decrypting the code used by the cell to carry contractile information.
About the decryption of the Ca2+ code, it should be noted that the choice in the parameters used to describe the Ca2+ signal determines the underlying hypotheses on the nature of the coding system. Indeed, decryption of the code is done by establishing correlations between the contractile response and parameters of the Ca2+ signal. Since correlation cannot be established with unstudied parameters, exclusion of parameters should be based on explicit and relevant hypotheses on the possible encoding significance of such parameters. What may appear as a signal for the investigator may not be a message for the cell. For example, several studies use average [Ca2+]i recording on tissue containing several ASMCs. This average [Ca2+]i response can provide information on cell physiology. However, this [Ca2+]i response is the average of the [Ca2+]i signal of each ASMC but cannot be considered as a signal, even as a signal of whole tissue contraction, because it carries no information for each ASMC contractile apparatus. In addition, the area of the Ca2+ response, i.e., the integral of the [Ca2+]i curve, in some studies called the Ca2+ index [1], gives no information on the dynamics of the Ca2+ response. Hence, considering it as a parameter of the Ca2+ signal for the cell requires the underlying hypothesis that the information carried by the Ca2+ signal is independent of the dynamics of the signal. Change in the Ca2+ index can be viewed as a sign that the Ca2+ signal is altered but not as a signal of a message in itself if the contractile apparatus is sensitive to the dynamics of [Ca2+]i variations.
How can we decipher the contractile code? Description of the signal is not the same thing as understanding the message. Identification of the quantitative relationship between the parameters of the Ca2+ signal is a way of deciphering the contractile code. However, identification of correlation does not mean identification of the causal relationship between Ca2+ and contraction. Mathematical modeling is a useful tool for investigating the causal relationships between the pattern of the Ca2+ signal and that of the subsequent contraction, which is the decryption of the contractile code.
1.4 Different Temporal Aspects of Ca2+ Signal
The temporal dimension is included in the concept of a Ca2+ signal itself. The temporal aspect of a Ca2+ signal appears at different stages of the stimulation-response pathway. First, insofar as [Ca2+]i change is an intracellular message induced by the stimulation of the cell by an extracellular agonist, and is hence a consequence of the activation of a network of intracellular mechanisms that determine the pattern of [Ca2+]i variations, the temporal dimension of these interactions is critical for the shape of the Ca2+ signal and, hence, its informative content. This represents the encoding of the Ca2+ signal. Second, in as much as [Ca2+]i variations represent a signal for the contractile machinery, in the sense that they determine the contractile behavior of ASMCs, both in amplitude and time, the time-dependent parameters that describe the Ca2+ signal should be considered information on contraction, i.e., information that can be detected by the contractile system. This represents the decoding of the Ca2+ signal, which depends on the kinetics of the functional interactions in the contractile system.
Hence, the temporal aspects of the Ca2+ signal include several distinct issues: (1) the temporal aspects of the Ca2+ signal pattern itself; (2) upstream of the Ca2+ signal, the temporal aspects of Ca2+ signal encoding, i.e., the dynamics of the mechanisms involved in the Ca2+ response to cell stimulation; and (3) downstream of the Ca2+ signal, the dynamics of the decoding machinery associated with the contractile apparatus that determines the pattern of the contractile response to Ca2+ signaling.
2 Overview of ASMC Ca2+ Handling
2.1 General Scheme: Bow-Tie Architecture
Free cytosolic Ca2+ concentration is the consequence of the balance between the ON mechanisms that tend to increase [Ca2+]i and the OFF mechanisms that tend to decrease it. Signaling pathways that induce a Ca2+ signal can be viewed as acting on the ON/OFF balance and, by doing so, inducing a spatiotemporal change in [Ca2+]i corresponding to the encoding of the Ca2+ signal. This Ca2+ signal can be “read” by the contractile machinery, and the resulting change in contraction corresponds to the decoding of the Ca2+ signal. This decoding process can be viewed also as a combination of ON mechanisms that tend to increase contraction for a given Ca2+ signal and OFF mechanisms that tend to decrease contraction. The Ca2+ signal is hence at the node of encoding and decoding processes, acting upstream and downstream of the Ca2+ signal, respectively, on which cell stimulation acts, and excitation-contraction coupling can be analyzed in the conceptual framework of this general scheme [2]. Such a scheme, called a bow-tie architecture, initially used to analyze engineered systems, is also useful for analyzing the information network that commands cell behavior such as excitation-contraction coupling [3–6]. This scheme is presented in Fig. 1a. According to this scheme, the structure involved in Ca2+ homeodynamics is the ON and OFF encoding mechanisms. They are summarized in Fig. 1b.
General mechanisms of Ca2+ handling in ASMCs. (a) Representation of bow-tie architecture applied to ASMC excitation-contraction coupling. The Ca2+ signal pattern is determined by the temporal balance between ON and OFF mechanisms. The Ca2+ signal is also decoded by ON and OFF mechanisms on which depends the contraction. Contractant and relaxant agonists may act on the ON and OFF mechanisms of both encoding and decoding of the Ca2+ signal. (b) General scheme of main ON and OFF mechanisms of Ca2+ handling. The main ON mechanisms are (1) Ca2+ release from the SR through InsP3 receptors (InsP3R) activated by agonists (A) acting on membrane receptors coupled to PLC (R) or through ryanodine receptors (RyRs) and (2) extracellular Ca2+ influx through several Ca2+ channels (VOC, ROC, and SOC). The main OFF mechanisms are (1) Ca2+ extrusion through PMCA and NCX, (2) Ca2+ pumping into the SR by SERCA, (3) Ca2+ uptake by mitochondrial Ca2+ uniporter, and (4) Ca2+ binding to fast kinetics–low affinity cytosolic proteins (SPr) and slow kinetics–high affinity cytosolic proteins (BPr)
2.2 Encoding Mechanisms of Ca2+ Signal
2.2.1 ON Mechanisms
The ON mechanisms, by which [Ca2+]i is increased upon ASMC stimulation, are basically Ca2+ release in the cytosol from the sarcoplasmic reticulum (SR) and extracellular Ca2+ influx through the plasma membrane. The major contractile agonists, such as acetylcholine and histamine, act primarily via Inositol 1,4,5-trisphosphate (InsP3) production and Ca2+ release from the cytosol via InsP3 receptors (InsP3R) located in the sarcolemma [7–11]. Ca2+ release from the SR can also be due to the opening of the ryanodine receptors (RyR), which can be activated by [Ca2+]i increase, the so-called Ca2+-induced Ca2+ release (CICR), and cyclic ADP-ribose [12–14]. External Ca2+ influx can occur via different types of Ca2+ channel. L-type voltage-operated Ca2+ channels (VOCs) are present in ASMCs and are activated by membrane depolarization [15, 16]. Receptor-operated channels (ROCs) are activated by binding of the agonist on membrane receptors [17]. Both ROCs and VOCs can be physiologically involved in the Ca2+ response to airway smooth muscle stimulation [18]. For example, extracellular adenosine triphosphate (ATP) acts directly on P2X receptors, whose opening induces Ca2+ and Na+ influx, which in turn depolarizes the plasma membrane, hence indirectly activating VOCs and subsequent additional Ca2+ influx [19]. Store-operated Ca2+ channels (SOCs) are activated by the emptying of intracellular Ca2+ stores [20, 21]. Though SOCs have been detected in ASMCs and contribute to general Ca2+ homeodynamics, their direct contribution to the physiological Ca2+ signal upon cell stimulation remains unclear [21–23].
2.2.2 OFF Mechanisms
A decrease in [Ca2+]i results from Ca2+ extrusion out of the cell, Ca2+ uptake by intracellular organelles, or Ca2+ buffering by cytosolic Ca2+-binding proteins. Ca2+ extrusion against its electrochemical gradient in ASMCs is mainly due to the activity of the plasma membrane Ca2+-ATPase (PMCA) and the Na+-Ca2+ exchanger (NCX), though the contribution of the latter to the Ca2+ signal is debated [24], contributing to either Ca2+ extrusion but also, in reverse mode, to Ca2+ influx [25–28]. Ca2+ storage in intracellular compartments is due to Ca2+ pumping into the SR by sarco-/endoplasmic Ca2+-ATPase (SERCA) [24, 29] and to Ca2+ uptake into the mitochondria by the mitochondrial Ca2+ uniporter [2, 30, 31]. Another mechanism that should not be underestimated is free Ca2+ buffering by cytosolic proteins [32]. Two main types of Ca2+-binding proteins have been identified: fast kinetics–low affinity Ca2+-binding proteins, so-called signaling proteins, and slow kinetics–high affinity Ca2+-binding proteins, so-called buffering proteins. Actually, these two types of Ca2+-binding sites can coexist on the same protein [6]. Though the Ca2+ bound to these proteins remains in the cytosol, these Ca2+-binding proteins can be functionally viewed as a cell compartment of Ca2+ sequestration since they remove free Ca2+ from the cytosol [2, 31].
3 Temporal Characterization of Ca2+ Signal in ASMCs
3.1 Overview of Biological Material and Methods
For the last two decades, a large number of studies have been published on the characterization of the cytosolic Ca2+ response to several agonists in ASMCs. The heterogeneity of these studies concerns the biological material used for these investigations, the type and mode of cell stimulation, and the methodology used for [Ca2+]i recordings. Investigations were performed on ASMCs for different species, mainly rat [2, 8, 9, 19, 33–36], mouse [7, 33, 37–43], guinea-pig [25, 29, 44, 45], pig [12–14, 21, 22, 46, 47], horse [48, 49], cow [21, 50, 51], dog [24, 52–54], and human ASMCs [55–58], and interspecies differences have been demonstrated. Experiments were performed on either cultured cells or freshly isolated cells, and in some cases single cells in situ, for example in lung slides. These different conditions may be responsible for differences in the Ca2+ response pattern since it has been shown in rat tracheal cells that Ca2+ homeodynamics can be critically altered by cell culture, even for a short period (48 h) [19], and in situ recordings maintain cell-to-cell interactions that are disrupted in isolated myocytes. In most of the studies cited earlier, [Ca2+]i recordings were done using intracellular Ca2+ probes such as indo-1 or fura-2 or similar fluorescent dyes, for which [Ca2+]i variation is estimated by changes in fluorescence intensity or intensity ratio. These molecules allow real-time measurement of [Ca2+]i variations, but, insofar as they are Ca2+-binding molecules, their presence may alter the amount of free Ca2+ in the cytosol and, hence, the pattern of the Ca2+ response. Some studies have used an alternative electrophysiological method based on whole-cell patch clamp recording of Ca2+-activated Cl− currents (I ClCa ) [11, 59, 60]. Comparison of the Ca2+ signal profile obtained with both techniques indicates that fluorescent probes, at the usual concentrations, have no detectable effect on the [Ca2+]i response pattern. For example, in freshly isolated rat trachea myocytes, similar [Ca2+]i response patterns were obtained using indo-1 fluorescent probe or electrophysiological recording of I ClCa [60].
Some differences may be due to the location along the airway tree. Studies have been performed either on tracheal or intrapulmonary bronchial myocytes, but few studies have compared the responses of these different cells in similar conditions. Since the histological organization of airways differs between trachea, extralobular, and intralobular bronchi, differences in ASMC [Ca2+]i response depending on their location cannot be excluded. In rat airways, differences in contraction patterns and stimulation-contraction mechanisms have been evidenced in response to purinergic stimulation [19]. In mouse airways, a difference in Ca2+ signaling has been evidenced depending on bronchial generations [40].
3.2 General Pattern of [Ca2+]i Response
Taken together, notwithstanding the differences observed in the Ca2+ signal depending on species, agonists, location along the airway tree, and recording methods, the results obtained from the literature show that a general Ca2+ pattern can be identified. Examples of original Ca2+ traces of ASMCs from different species in response to different agonists are presented in Fig. 2. Recordings on single cells show that the response is characterized by an initial fast and transient [Ca2+]i rise. The first [Ca2+]i peak can be followed either by a progressive decay to a more or less steady-state value below the maximum of the [Ca2+]i peak but above the resting value, the so-called plateau, or by a succession of transient [Ca2+]i peaks, the so-called Ca2+ oscillations. These Ca2+ oscillations can be superimposed on the plateau phase, i.e., [Ca2+]i does not return to baseline between two oscillations. Fluorescent probes give an estimate of the variations in [Ca2+]i, but the absolute [Ca2+]i value requires calibration, which was not done in all studies that investigated the Ca2+ response. When performed, the calculations give a resting [Ca2+]i of around 100–200 nM, and the amplitude of the Ca2+ peak usually ranges up to 1 μM above the resting value. The time-to-peak, i.e., the time needed to reach the maximum [Ca2+]i, is usually not given, indicating that the authors pay more attention to the amplitude of the peak than to its timing. However, considering the few studies that have measured this value [2], and the estimation that can be made from the original traces shown in the others, the top of the Ca2+ peak is obtained within a few seconds.
Original typical traces of Ca2+ signals in freshly isolated ASMCs from several species: oscillatory (a) and nonoscillatory (b) Ca2+ response of rat tracheal myocyte to 10−5 M ACh. (c) Ca2+ response of rat tracheal myocyte to 110 mM KCl. (d) Ca2+ response of rat tracheal myocytes to 10−4 M ATP. (e) Ca2+ response of horse bronchial myocyte to 10−5 M ACh. (f) Ca2+ response of human bronchial myocyte to 10−5 M ACh. Cells were obtained from fresh tissues by enzymatic dissociation followed by mechanical dispersion and used within 1 day. [Ca2 +]i was measured by microspectrofluorimetry using the fluorescent dye Indo 1 (for the methods, see [19])
The dynamics of [Ca2+]i decay has been quantified by some studies [2, 24, 29]. They show that this decay follows a more or less exponential profile, the decay being faster when the stimulation is stopped before the decay begins, but the general pattern remains similar whether cell stimulation goes on or is stopped.
In the case of nonoscillatory response, the slow decay reaches a plateau, which is part of the Ca2+ signal, that is above the resting value. However, several studies have focused on the peak and on the oscillations, paying little attention to the plateau. Its significance in terms of contraction response is discussed later. Usually this plateau, when observed, persists as long as the stimulation continues and drops within seconds to baseline when it stops. Observations of original traces show that the plateau is not strictly constant over time and exhibits irregular variations. This can be attributable to stochastic variations in Ca2+ release and uptake, whose consequences for the overall Ca2+ signal is exacerbated by a clustering of SR Ca2+ channels [61]. In some cases, it slowly decreases even upon stimulation, for example upon ATP stimulation in rat tracheal myocytes [19]. In other cases, the Ca2+ signal may persist for several seconds after the end of the stimulation, as noticed in the same cell type with endothelin-1 [8]. In the case of oscillatory responses, the oscillations may be damped, with a progressive decay in their amplitude, or sustained as long as stimulation persists. Oscillatory responses have been evidenced in ASMCs from several species, such as mouse [42], guinea pig [29], pig [47, 62], and rat (with nonoscillatory responses as well) [9], whereas nonoscillatory responses have been observed in cow [50] and dog ASMCs [24]. In human airways, nonoscillatory Ca2+ responses have been described by some studies on isolated ASMCs [55, 57], whereas a recent in situ study describes an oscillatory Ca2+ signal [58].
The frequency of oscillations depends on species, agonists, and agonist concentration. In physiological conditions, oscillation frequency ranges between 2 and 3 per minute and 25–30 oscillations per minute. Some pathological conditions like exposure to oxidizing pollutants increase oscillation frequency [36]. Usually, oscillation frequency correlates, when recorded, with the intensity of contraction, and it is usually acknowledged that oscillation frequency encodes for the amplitude of contraction [34, 36–39, 42, 58, 63]. The mechanisms responsible for Ca2+ oscillations in ASMCs and their significance in contraction encoding are discussed later in the chapter.
3.3 Mechanisms Responsible for Ca2+ Peak and Plateau
3.3.1 ON Mechanisms
As shown earlier, the first step for the Ca2+ peak and plateau phases, for a large majority of agonists, is a fast Ca2+ increase followed by progressive decay that can usually be fitted by an exponential equation. This Ca2+ peak is observed with a quite similar profile whatever the source of Ca2+, i.e., extracellular Ca2+ influx or Ca2+ release from the SR [42, 64]. In both cases, the elevation of [Ca2+]i is due to a sudden increase in the amount of Ca2+ in the cytosol caused by the opening of Ca2+ channels, either located in the plasmalemma or in the sarcolemma. This influx is rapid because, when opened, these channels allow exergonic Ca2+ movement and the electrochemical gradient highly favors cytosolic Ca2+ influx. Mathematical modeling of Ca2+ influx through voltage-operated Ca2+ channels and InsP3R or RyR, based on experimental characterization of these channels, predicts a fast increase in [Ca2+]i, as observed in Pulmonary artery smooth muscle cell (PASMC) studies [2, 31, 65]. In the case of InsP3-induced Ca2+ release, the kinetics of the Ca2+ response depend on the kinetics of the enzymatic reactions of InsP3 production catalyzed by PLC. Hence, at the molecular level, the mechanisms responsible for [Ca2+]i increase due to InsP3R stimulation are slower than that due to VOC activation. The velocity of [Ca2+]i increase, however, depends not only on the kinetics of the molecular reactions that govern the opening and closure of Ca2+ channels but also on the ratio between the transmembrane fluxes and the volume of the cytosol. Experiments show that the kinetics of the Ca2+ peak induced by InsP3-mediated agonist are almost similar to direct InsP3 stimulation in both cases in ASMCs, indicating that, in this cell type, the enzymatic kinetics of InsP3 production are not a limiting factor for [Ca2+]i increase velocity [8].
3.3.2 OFF Mechanisms
The Ca2+ peak involves not only a rise in [Ca2+]i but also the decrease in [Ca2+]i that follows. Then, the OFF mechanisms responsible for cytosolic Ca2+ clearance are as important as the ON mechanisms responsible for [Ca2+]i increase. Experimental results on the respective roles of SR Ca2+ uptake, mitochondria, and cytosolic buffering capacity are presented in Fig. 3. Intuitively, Ca2+ decay is linked to the closure, at least partial, of Ca2+ channels and subsequent activation of mechanisms that remove Ca2+ from the cytosol, in particular Ca2+ pumping into the SR. Studies of the dynamics of Ca2+ handling in ASMCs have proven this intuitive representation to be largely wrong.
Influence of OFF mechanisms on kinetics of Ca2+ signal. (a) Kinetics of [Ca2 +]i decay following Ca2+ peak induced by 5 mM caffeine stimulation for 1 s (left trace, light gray squares), 5 s (dark gray squares, middle trace), and 30 s (right trace, black square). (b) Influence of SERCA inhibition by cyclopiazonic acid (CPA) on the kinetics of [Ca2 +]i decay following 5 mM caffeine stimulation (1 s). Black circles: control; open circles: 10−5 M CPA. (c, d) Influence of mitochondrial Ca2+ uptake inhibition by 5.10−6 M FCCP on (c) the kinetics of [Ca2+]i decay following 5 mM caffeine stimulation (1 s) (black circles: control; open circles: FCCP) and (d) the percentage of oscillating Ca2+ responses to 10−5 M ACh stimulation. (e, f) Influence of increased cytosolic Ca2+ buffering by 5.10−5 M BAPTA-AM on (e) the percentage of oscillating Ca2+ responses and (f) the frequency of Ca2+ oscillations to 10−5 M ACh stimulation. Cells were obtained from fresh tissues by enzymatic dissociation followed by mechanical dispersion and used within 1 day. [Ca2 +]i was measured by microspectrofluorimetry using the fluorescent dye Indo 1 (for the methods, see Roux and Marhl [2]). *P < 0.05 (Studentʼs t-test)
First, as far as the stimulation persists, the Ca2+ channels remain open, at least in the first step of the stimulation. The end of the Ca2+ increase and the beginning of the Ca2+ decrease do not correspond to the closure of the Ca2+ channels. It has been established, for example, that the [Ca2+]i peak, including the decay phase, induced by caffeine stimulation is not due to fast opening and closure of RyRs but to persistent RyR opening associated with simultaneous mechanisms of cytosolic Ca2+ clearance [2]. In the case of RyR closure, the exponential decay is faster than when RyRs remain open, but the profile is similar in both cases, as shown in Fig. 3a. Both experimental and theoretical studies have shown that the profile of the [Ca2+]i peak is a consequence of the balance between ON and OFF mechanisms [2, 24, 29, 31, 66]. In this balance, the kinetics of the buffering capacity of OFF mechanisms are a critical determinant of the Ca2+ peak profile.
3.3.2.1 Role of Ca2+ Pumping in SR by SERCA
In the buffering system, Ca2+ pumping back into the cytosol is usually considered the most important aspect [67]. This is actually true in the sense that the SR as a Ca2+ store is important for cell physiology, but Ca2+ pumping back by SERCA is not the major determinant of the Ca2+ decay. Actually, if Ca2+ efflux from the cytosol is activated, then Ca2+ pumping back by SERCA cannot in principle account for [Ca2+]i decreases since as soon as Ca2+ is pumped back into the cytosol it leaks through the opened Ca2+ channels. Accumulation of Ca2+ would require that the velocity of Ca2+ pumping against the concentration gradient would be higher than that of a passive Ca2+ leak, which is not the case [66]. Even when the Ca2+ channels are closed during the decay phase, as occurs for short (1 s) stimulations, inhibition of SERCA does not significantly modify the dynamics of the [Ca2+]i clearance, as shown in Fig. 3b.
3.3.2.2 Role of Ca2+ Uptake in Mitochondria by Ca2+ Uniporter
It has been shown that SERCA is neither the only nor the fastest mechanism of free cytosolic Ca2+ clearance. When [Ca2+]i increases, Ca2+ uptake into mitochondria by the mitochondrial Ca2+ uniporter plays a more important role than Ca2+ uptake by SERCA, even when SR Ca2+ channels are closed, because Ca2+ uptake by mitochondria occurs faster than that by the SR [30, 68–70]. As shown experimentally, for short caffeine stimulations, inhibition of SERCA does not significantly modify the slope of the [Ca2+]i decay, whereas inhibition of the mitochondrial uniporter by Trifluorocarbonylcyanide phenylhydrazone (FCCP) does, as illustrated in Fig. 3c. Mitochondria seem also to be involved in the occurrence of oscillations since FCCP decreases the percentage of oscillatory response to ACh stimulation in freshly isolated rat tracheal myocytes (personal data) (Fig. 3d).
3.3.2.3 Role of Ca2+ Buffering by Cytosolic Ca2+-Binding Proteins
Though significant, the effect of mitochondrial inhibition on the shape of the [Ca2+]i decay following caffeine stimulation is modest, and theoretical analysis has shown that the shape of the [Ca2+]i decay, as well as the amplitude of the Ca2+ peak, critically depends on Ca2+ binding to cytosolic proteins capable of buffering free cytosolic Ca2+ [2, 65, 66]. Though few experimental studies have been dedicated to investigating the role of cytosolic buffering capacity on the pattern of the Ca2+ signal, manipulation of the buffering capacity of the cytosol by BAPTA, a Ca2+ chelator, shows that an increase in the buffering capacity of the cytosol can alter Ca2+ signaling [71]. As shown in Fig. 3e, f, increased cytosolic Ca2+ buffering capacity by the Ca2+ chelator BAPTA decreases both the percentage of oscillating cells and the frequency of oscillations in response to cholinergic stimulation (personal data).
3.3.3 Integrated View of Ca2+ Handling upon ASMC Stimulation
Hence, besides [Ca2+]i decay following an initial [Ca2+]i increase, multiple Ca2+ buffering mechanisms act simultaneously, but not instantaneously. According to the theoretical analysis of Ca2+ handling, a fast [Ca2+]i increase is followed by free cytosolic Ca2+ buffering by cytosolic proteins and Ca2+ uptake by the mitochondria prior to Ca2+ uptake in the cytosol, even in the case of cell stimulation and Ca2+ release into the cytosol [66]. This has several consequences for our understanding of Ca2+ signaling.
First, the temporal aspects of the multiple mechanisms involved in the profile of the Ca2+ signal are critical determinants of its timing and amplitude. Indeed, the amplitude of the Ca2+ peak depends on the buffering capacity of the so-called signaling proteins (fast kinetics Ca2+-binding proteins). The sequential description of the mechanisms of the Ca2+ signal according to which Ca2+ is first released in the cytosol, then buffered by cytosolic protein, later taken up by the mitochondria, and eventually pumped back into the SR is only a metaphorical way to express the fact that these mechanisms have different kinetics that should not hide the fact that they occur simultaneously. Cell stimulation disrupts the dynamic equilibrium responsible for the [Ca2+]i resting value, and the Ca2+ peak is the result of the subsequent transitory nonequilibrium state. The Ca2+ plateau is the consequence of the new dynamics of Ca2+ handling when the new equilibrium state is reached. Fast kinetics mechanisms are critical for the peak shape, whereas the plateau value is dependent on the relative buffering capacity of each cell compartment, even if the kinetics of these buffering reactions are slow. It should be noted that even if the plateau value seems slightly different from the resting [Ca2+]i value, the equilibrium state may be quite different in both cases, with great differences in the amount of Ca2+ in the various intracellular compartments at rest versus upon cell stimulation.
The second consequence is that, although [Ca2+]i values return to baseline within seconds after the end of cell stimulation, this does not mean that the cell is already back to its initial Ca2+ homeodynamic state. Theoretical studies, in correlation with experiments, show that complete Ca2+ refilling of the SR requires several minutes [2], as illustrated in Fig. 4. As a consequence, two successive cell stimulations triggering Ca2+ release from the SR will not result in similar Ca2+ response as long as Ca2+ handling is not back at its initial state, and the time needed to recover to this initial state corresponds to a relative refractory period. The delay needed for the return to resting equilibrium, which is determined by the temporal characteristics of the mechanisms of cytosolic Ca2+ clearance, has a quite opposite consequence for cell homeostasis depending on the source of Ca2+. In the case of Ca2+ release from internal Ca2+ stores, repeated stimulations result in progressive depletion of the SR and subsequent decrease in stimulus responsiveness, whereas in the case of external Ca2+ influx, they would result in progressive Ca2+ overloading if the mechanisms of Ca2+ extrusion were saturated [2, 72].
Model prediction of Ca2+ movements upon 30 s caffeine stimulation. Simulated application of caffeine began at time 10 s. Dashed line: [Ca2+]i; full lines: concentrations of free Ca2+ in SR ([Ca2+]SR), in mitochondria ([Ca2+]m), and bound to signaling (CaSPr) and buffering (CaBPr) proteins. Though [Ca2+]i returns to baseline at the end of caffeine stimulation (time 40 s), a return to resting Ca2+ concentration in the various cell compartments requires much more time
In conclusion, it should be emphasized that the peak and the plateau involve similar mechanisms responsible for Ca2+ homeodynamics. The difference between the peak and the plateau profiles corresponds to two different phases of the transition between two equilibrium states of the Ca2+ handling, at rest and upon stimulation. The peak is a consequence of the transient nonequilibrium phase of the activated state, whereas the plateau corresponds to the state of the system when equilibrium is reached. When stimulation ends, return to the initial resting state is longer than the return of [Ca2+]i to baseline, due to silent Ca2+ fluxes between the Ca2+ buffering elements that occur at constant free cytosolic Ca2+ concentration.
3.4 Mechanisms of Ca2+ Oscillations
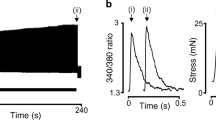
As indicated earlier, Ca2+ oscillations have been reported in a variety of ASMCs, though it should not be forgotten that in some species, including humans, the Ca2+ signal can be a nonoscillatory one. In ASMCs, Ca2+ oscillations have been mainly observed in response to contractile agonists acting on G-protein-coupled receptors and InsP3 production, such as ACh, 5-HT, endotheline-1, and ATP [9, 11, 13, 19, 42, 43, 73]. Additionally, experiments using direct stimulation by InsP3 have shown that nonoscillatory InsP3 concentration was able to generate oscillatory Ca2+ responses [8]. However, Ca2+ oscillations have also been noticed in response to stimulation acting via external Ca2+ influx [42]. Figure 5a, b compares Ca2+ oscillations induced by cholinergic stimulation and by direct InsP3 exposure.
Ca2+ oscillations. Original traces of Ca2+ response to ACh stimulation (a) and InsP3 stimulation (b). Cells were obtained from fresh tissues by enzymatic dissociation followed by mechanical dispersion and used within 1 day. [Ca2+]i was measured by microspectrofluorimetry using fluorescent dye indo-1. InsP3 was applied on β-escine-permeabilized cells [8]. (c, d) Model prediction to 1 μM InsP3 stimulation for different InsP3R isoform ratio. (c) 75 % IP3R subtype 1 and 25 % subtype 3; (d) 25 % IP3R subtype 1 and 75 % subtype 3. The model used is that of Haberichter et al. [65]
Basically, Ca2+ oscillations can be explained by asynchronous coupling between ON and OFF mechanisms. Since oscillations are observed in response to InsP3, much attention has been paid to the kinetic properties of InsP3R. Several isoforms of InsP3R have been identified, types 1, 2, and 3. These isoforms are sensitive to both cytosolic InsP3 and Ca2+. Both types 2 and 3 exhibit a sigmoid-shaped sensitivity curve to Ca2+ concentration, whereas type 1 has a bell-shaped sensitivity curve: for low [Ca2+]i, sensitivity to Ca2+ increases with [Ca2+]i, whereas for high [Ca2+]i, an increase in [Ca2+]i decreases InsP3P opening [74–77]. Moreover, this loss in sensitivity for high [Ca2+]i is delayed. InsP3R by itself seems to be an asynchronous ON/OFF switch capable of generating oscillations, an idea supported by the fact that InsP3R inhibition abolishes Ca2+ oscillations [7]. Several theoretical models have been developed to explain how InsP3R can be a molecular oscillator [78]. However, the mechanisms of Ca2+ oscillations have appeared to be more complex and could not be attributed to the properties of the InsP3R type 1 alone. First, experimental studies have shown that type 1 InsP3R expression can be associated with nonoscillatory Ca2+ signals [57]. On the other end, studies in vascular SMCs showed intercellular variation in InsP3R isoform expression and that InsP3R alone cannot generate Ca2+ oscillations, which requires a mixture of type 1 and type 2 isoform expression in the same cell [79]. In rat ASMCs, an immunofluorescence study suggested that such a heterogeneous expression is observed, with 100 % of ASMCs expressing type 1 isoform, 27 % type 2, and 40 % type 3 (personal data). Theoretical modeling of the InsP3-induced Ca2+ response indicates that both oscillatory and nonoscillatory Ca2+ signals can be predicted in cells expressing a mixture of type 1 and type 3 InsP3R, depending on the total amount of InsP3R and the type1/type3 ratio, and the presence of cytosolic Ca2+-binding proteins [65]. The role of the isoform ratio in the occurrence of Ca2+ oscillations is illustrated in Fig. 5c, d. RyRs have also been shown to contribute to ACh-induced as well as KCl-induced Ca2+ oscillations [12, 13, 72].
It appears, then, that Ca2+ oscillations in ASMCs are not due to the intrinsic property of one specific molecular oscillator whose presence determines their occurrence. Rather, the occurrence of Ca2+ oscillations is an emerging property of the machinery of Ca2+ handling, and oscillations may appear when asynchronous ON/OFF mechanisms are simultaneously active. As shown by theoretical investigations, one cell may occupy different possible positions in the phase space, at one moment being in an oscillatory phase space and at another moment in a nonoscillatory space phase, depending on the intensity of cell stimulation [65, 72]. In a cell population, interindividual variations in InsP3R expression and in the ratio of isoform expression generate the distribution of the cells in the different phases of the space, so that heterogeneous cell behavior, oscillatory and nonoscillatory Ca2+ signals, may be the consequence of a homogeneous cell type associated with inherent cell-to-cell variations [65].
Oscillatory Ca2+ responses have been reported in some cases in response to KCl stimulation [42]. In the case of forced membrane depolarization, the mechanisms responsible for the Ca2+ signal, i.e., Ca2+ influx through VOCC and CICR through RyRs, are different from that activated by agonists acting on G-coupled membrane receptors like muscarinic receptors. This emphasizes the fact that oscillations may occur as a consequence of the activation of distinct pathways. However, in the case of KCl, Ca2+ oscillations require simultaneous involvement of ON/OFF mechanisms, unlike with InsP3. Oscillations have also been reported in vascular SMCs, and theoretical modeling, including voltage-dependent Ca2+ influx and RyR activation, accounts for this oscillatory profile [80]. Though these models have been developed for vascular SMCs, the mechanisms they describe also account for what is observed in ASMCs, as was confirmed by recent modeling of KCl-induced oscillations in ASMCs [72].
Taken together, these results indicate that the pattern of the Ca2+ signal is critically determined by the kinetic constants of each element involved in ASMC Ca2+ handling. Perturbation of the functional properties of any of these structures may lead to loss of the oscillatory response, which does not mean that this structure is the element that determines the Ca2+ profile, but it is one element, among others, where functional properties contribute to shape the Ca2+ signal.
4 Decoding of Ca2+ Signals
4.1 Contractile System
How is a message carried by the Ca2+ signal? In other words, how does the Ca2+ signal determine the behavior of the contractile apparatus? What is, in the time-dependent variations of [Ca2+]i that occur upon cell stimulation, information for the contractile machinery, and what type of information is it? The object of this part of the chapter is not to detail the contractile apparatus and its regulation pathways but to present an overview of the “semantics” of the Ca2+ signal in terms of contraction. Basically, a rise in [Ca2+]i activates the contractile apparatus by the following pathways, summarized in Fig. 6. Ca2+ binds to the cytosolic protein calmodulin (CaM), which can bind to the myosin light chain kinase (MLCK), and the Ca2+-CaM-MLCK complex is the active form of MLCK. When activated, MLCK phosphorylates the 20 kDa regulatory myosin light chain (MLC20), and this phosphorylation is required for the formation of the actin-myosin cross bridge and, hence, contraction. Dephosphorylation of MLC20 is ensured by myosin light chain phosphatase (MLCP). Both MLCK and MLCP activity can be up- and downregulated by several kinase- and phosphatase-dependent pathways [81]. The canonical way that controls contraction in ASMC is the Ca2+-CaM-MLCK pathway [34, 82]. The other regulatory mechanisms are grouped into the so-called concept of regulation of Ca2+ sensitivity since they modify the contractile response for a given Ca2+ signal. Though useful, this distinction between the Ca2+-induced contraction pathway on the one hand and the regulation of Ca2+ sensitivity on the other hand is somewhat artificial since regulation of Ca2+ sensitivity can itself be Ca2+-dependent [83].
General mechanisms of Ca2+-contraction coupling in ASMCs. Ca2+ binds to calmodulin (CaM) and MLCK. The Ca2+-CaM-MLCK complex is the active form of MLCK, which catalyzes MLC20 (M) phosphorylation. Phosphorylated MLC20 (Mp) binds to actin (A) to form the actin-myosin bridge (AMp) that can later be dephosphorylated (AM). Dephosphorylation is achieved by MLCP, whose activity is modulated by several kinases, mainly PKC and RhoK
4.2 Contractile Significance of Ca2+ Signal
The semantics of the Ca2+ signal have long been investigated by correlating contraction with the Ca2+ signal parameters, mainly the Ca2+ peak, the plateau, and, when present, the oscillations [9, 19, 35–37, 42, 64]. Regarding the contraction itself, several distinct parameters are used to describe it. Usually, the intensity of the steady-state response is used to characterize the contractile response, measured by either isometric recording [19, 34, 36, 45, 51, 83–85] or changes in the trans-sectional area of the bronchial lumen [33, 40, 43], but other parameters are also used to describe the temporal aspects of the contractile response, such as shortening velocity in isotonic measurements [86, 87] or half-time maximal contraction (the time needed to achieve 50 % of the maximal contraction) in isotonic recording [34, 83, 86, 87].
Peak, plateau, and oscillation frequency all vary with the intensity of agonist stimulation, as does the amplitude of contraction. It is hence difficult to determine experimentally the specific message carried by each Ca2+ signal parameter because of the difficulty of varying one parameter independently of the others. Theoretical modeling is a way to investigate the specific significance of the distinct parameters of the Ca2+ signal since it is possible, in contrast to experimentation, to vary them independently [34, 83, 87–89].
4.2.1 Contractile Message of Initial Ca2+ Peak
Realistic computational modeling of Ca2+-contraction coupling, including both the canonical MLCK pathway and MLCP regulation by RhoK and PKC, indicates that the amplitude of the Ca2+ peak determines the velocity of the early phase of the contraction [34, 83]. This may explain why ASM contraction develops slower for low than for high levels of agonist concentration. However, according to the model, the contractile apparatus is much less sensitive to a transient [Ca2+]i rise than to a sustained one, and the peak does not determine the amplitude of contraction. This is basically due to the inertia of the MLCK-dependent activation of the contractile apparatus compared with the velocity of the Ca2+ peak, both in its increase and decrease, so that during the Ca2+ peak, the MLC phosphorylation rate decreases before phosphorylated MLC has reached its maximal value.
4.2.2 Contractile Message of Ca2+ Plateau
The amplitude of the plateau is, according to the model, the major determinant of the amplitude of contraction [34, 83]. Since the plateau is a stable [Ca2+]i value, the system can reach its equilibrium. Usually, the amplitude of the plateau is considered small compared with the maximal peak value. However, both theoretical and experimental studies indicate that the level of phosphorylated MLC is not linearly related to the steady-state [Ca2+]i value, and this explains the fact that the amplitude of the plateau is a critical determinant of the amplitude of the contraction [34].
4.2.3 Contractile Message of Ca2+ Oscillations
Experimental studies have demonstrated statistical correlations between the frequency of Ca2+ oscillations and the amplitude of contraction, suggesting that the Ca2+ oscillation frequency determines the amplitude of contraction [9, 19, 33, 35, 39, 43, 63]. However, such correlations do not establish an unequivocal causal relationship since, as stated earlier, other parameters simultaneously correlate with contraction intensity [9, 35]. Moreover, the meaning of frequency encoding should be clarified. Indeed, Ca2+ oscillations induce an average [Ca2+]i increase that may induce contraction whether this increase is oscillating or not. Thus, if we consider oscillations as carrying a specific message, this means that the contractile apparatus is specifically sensitive to the dynamics of Ca2+ oscillations. In other words, the contractile system is oscillation-frequency-sensitive only if the contractile response to an oscillatory signal differs from that of a nonoscillatory signal with an identical average [Ca2+]i value, i.e., for which the surface area under an oscillating trace is equal to that under a nonoscillating trace. It is hard, if not impossible, to design an experimental protocol to compare these two conditions. Additionally, since airway smooth muscle contraction is the response at the tissue level, the question arises as to whether the observed nonoscillatory contractile response at the tissue level is the consequence of the summation of asynchronous oscillatory responses at the cellular level or results from the integration at the cellular level of an oscillatory Ca2+ message into a nonoscillatory contractile response. For both questions, mathematical modeling is a useful tool to test these hypotheses. Computational modeling with realistic kinetic constants indicates that oscillation frequency within the range observed in experiments (5–30 oscillations min−1) generates a nonoscillatory contractile response [34, 83]. Hence, it seems that the oscillatory message is integrated at the cellular level by the contractile machinery. This integration occurs at different steps of Ca2+-contraction coupling, illustrated in Fig. 7. Model prediction indicates that the temporal pattern of MLCK activity is oscillating and follows more or less that of the Ca2+ signal, though MLCK activity decay might be slightly slower than Ca2+ decay (Fig. 7b). In contrast, the temporal pattern of MLC phosphorylation, though oscillatory, exhibits smoothed oscillations, the level of phosphorylated MLC remaining high between two oscillations (Fig. 7c). This is due to the inertia of the system, i.e., physiological MLCP activity is not sufficient to trigger an immediate drop in phosphorylated MLC when MLCK activity decreases between two Ca2+ oscillations. The second, and most important, step of the integration of the Ca2+ signal is the formation of actin-myosin bridges. Computational modeling predicts that the amount of actin-myosin bridges, which corresponds to isometric contraction, remains high between two Ca2+ peaks even if the amount of phosphorylated MLC tends to decrease. This is due to the fact that actin-myosin binding can be maintained even if MLC is dephosphorylated because of the low kinetic constant for the rupture of the dephosphorylated myosin-actin bridge, the so-called latch bridge. When the next Ca2+ peak and its subsequent rise in MLC phosphorylation occur before a significant drop in the actin-myosin bridge, which depends on the balance between oscillation frequency and the kinetics of actin-myosin bridge cycling, actin-myosin bridges tend to accumulate until an equilibrium is reached, and this corresponds to a slow additional increase in contraction (Fig. 7d). Changing the oscillation frequency within the physiological range in the model predicts an oscillation-frequency-dependent increase in contraction. It can hence be concluded that the oscillations actually encode for the intensity of ASM contraction since ASMC modeling predicts that the contractile system is specifically sensitive to oscillation frequency independent of the average [Ca2+]i value. However, if we compare, as shown in Fig. 8, the sensitivity of the contractile machinery to the peak value (or transient [Ca2+]i increase), the plateau value (or the average [Ca2+]i value), and the oscillation frequency, the model prediction is that the contractile apparatus will be much more sensitive to the plateau value than to the frequency of Ca2+ oscillation per se. Hence, the importance of the plateau phase or, in the case of oscillatory Ca2+ signals, of the average [Ca2+]i value over time independently of Ca2+ oscillations should not be underestimated. The sensitivity to a transient [Ca2+]i rise is quite low, as shown in Fig. 8b, which explains the fact that, for a standard biphasic Ca2+ response, the amplitude of the Ca2+ peak does not determine the amplitude of the contraction, which depends on the plateau, but not its velocity (Fig. 8a).
Temporal integration of oscillatory Ca2+ signal. Model prediction of MLCK activation (b), MLC phosphorylation (c), and contraction (d) upon oscillating Ca2+ signal (a), according to the model of Mbikou et al. [83]
Contraction encoding by peak, plateau, and frequency of oscillations. Model predictions are done using the model of Mbikou et al. [83]. (a) Predicted time course of force for two peak amplitudes (0.3 and 0.6 μM) for fixed resting [Ca2+]i and plateau. (b) Predicted relationship between amplitude of transient [Ca2+]i increase and force. (c) Predicted relationship between amplitude of sustained [Ca2+]i and force. (d) Predicted relationship between frequency of oscillations and force. The simulated Ca2+ signal used is similar to that of Fig. 7, with variation in oscillation frequency
It is tempting to speculate about the advantage of frequency encoding versus amplitude encoding in cell physiology. For example, it has been hypothesized that oscillation encoding may be more precise than amplitude encoding, hence ensuring more accurate functional response to cell stimulation, especially for low agonist concentrations [90]. Though this may be true in principle, one should be cautious, however, when speculating about the putative advantage of oscillatory versus nonoscillatory encoding, and at least in ASMCs, there is no clear evidence that oscillatory signaling may be advantageous. The main argument against such a speculation is that, in various species, ASMCs exhibit nonoscillatory Ca2+ signals. Additionally, analysis of cell-to-cell variability of the Ca2+ signal has shown, as a general property, high interindividual variations in the Ca2+ response to the same stimulation [91]. The Ca2+ signal appears, then, to be highly noisy, and it is hard to imagine the specific evolutionary advantage of highly precise decoding of a highly noisy signal. Robustness to the noise of the signal would, in principle, be more advantageous.
Functional analysis shows that Ca2+ oscillations can appear as an emerging property of the system responsible for intracellular Ca2+ handing and that these oscillations have functional consequences for the contractile apparatus. Whether the occurrence of an oscillating Ca2+ signal is a byproduct of the functional constraint of the system controlling Ca2+ homeodynamics [92] or a consequence of a selective pressure that has designed this system for the production of frequency-encoded Ca2+ signal is purely speculative and has no real support.
5 Conclusion
In conclusion, the temporal aspects of Ca2+ signals are inherent in the concept of Ca2+ signal, which is, by definition, a spatiotemporal change in [Ca2+]i. Temporal variations upon contractile stimulation exhibit a general pattern characterized by a transient Ca2+ peak followed either by a steady-state phase or Ca2+ oscillations. This pattern is the consequence of simultaneous, but not instantaneous, interactions between all the elements involved in Ca2+ handling, both ON and OFF mechanisms. Among the latter, Ca2+ pumping back into the SR is not the only important mechanism, and Ca2+ uptake by mitochondria and Ca2+ binding to cytosolic proteins are critical determinants of the Ca2+ signal. The transient peak is the consequence of a transitory nonequilibrium phase between the resting and the activated states of Ca2+ homeodynamics. If an equilibrium is reached, then [Ca2+]i stabilizes with respect to the plateau, whereas if asynchronous interactions between ON and OFF occur, Ca2+ oscillations appear. These different parameters of the Ca2+ signal are contractile messages. According to theoretical analysis, the amplitude of the Ca2+ peak encodes for the velocity of contraction, whereas the plateau determines the amplitude of the contraction. When they exist, oscillatory signals seem to be integrated at the cell level, leading to nonoscillatory contractions due to the temporal inertia of the MLC dephosphorylation process and dephosphorylated myosin-actin bridges. Oscillation frequency seems to encode specifically for the amplitude of contraction, but the contractile system is less sensitive to Ca2+ oscillation frequency per se than to the average Ca2+ value over time, whether or not the Ca2+ signal is oscillatory.
References
Ikeda, M., K. Kurokawa, and Y. Maruyama, Cyclic nucleotide-dependent regulation of agonist-induced calcium increases in mouse megakaryocytes. J Physiol, 1992. 447: p. 711–28.
Roux, E. and M. Marhl, Role of sarcoplasmic reticulum and mitochondria in ca(2+) removal in airway myocytes. Biophys J, 2004. 86(4): p. 2583–95.
Csete, M. and J. Doyle, Bow ties, metabolism and disease. Trends in Biotechnology, 2004. 22(9): p. 446–450.
Ma, H.W. and A.P. Zeng, The connectivity structure, giant strong component and centrality of metabolic networks. Bioinformatics, 2003. 19(11): p. 1423–1430.
Marhl, M., M. Perc, and S. Schuster, Selective regulation of cellular processes via protein cascades acting as band-pass filters for time-limited oscillations. FEBS Lett., 2005. 579(25): p. 5461–5465.
Schuster, S., B. Knoke, and M. Marhl, Differential regulation of proteins by bursting calcium oscillations - a theoretical study. Biosystems, 2005. 81(1): p. 49–63.
Bai, Y., M. Edelmann, and M.J. Sanderson, The contribution of inositol 1,4,5-trisphosphate and ryanodine receptors to agonist-induced Ca(2+) signaling of airway smooth muscle cells. Am J Physiol Lung Cell Mol Physiol, 2009. 297(2): p. L347–61.
Hyvelin, J.M., et al., Cellular mechanisms of acrolein-induced alteration in calcium signaling in airway smooth muscle. Toxicol Appl Pharmacol, 2000. 164(2): p. 176–83.
Roux, E., et al., [Ca 2+ ] i oscillations induced by muscarinic stimulation in airway smooth muscle cells: receptor subtypes and correlation with the mechanical activity. Br J Pharmacol, 1997. 120: p. 1294–1301.
Roux, E., et al., Muscarinic stimulation of airway smooth muscle cells. Gen Pharmacol, 1998. 31(3): p. 349–56.
Liu, X. and J.M. Farley, Frequency modulation of acetylcholine-induced Ca(++)-dependent Cl- current oscillations are mediated by 1, 4, 5-trisphosphate in tracheal myocytes. J Pharmacol Exp Ther, 1996. 277: p. 796–804.
Kannan, M.S., et al., Role of ryanodine receptor channels in Ca2+ oscillations of porcine tracheal smooth muscle. Am J Physiol, 1997. 272(4 Pt 1): p. L659–64.
Prakash, Y.S., M.S. Kannan, and G.C. Sieck, Regulation of intracellular calcium oscillations in porcine tracheal smooth muscle cells. Am J Physiol, 1997. 272(3 Pt 1): p. C966–75.
Prakash, Y.S., et al., Role of cyclic ADP-ribose in the regulation of [Ca2+]i in porcine tracheal smooth muscle. Am J Physiol, 1998. 274(6 Pt 1): p. C1653–60.
Marthan, R., et al., Calcium channel currents in isolated smooth muscle cells from human bronchus. J Appl Physiol, 1989. 66: p. 1706–1714.
Kotlikoff, L., Calcium currents in isolated canine airway smooth muscle cells. Am J Physiol, 1988. 254: p. C793–C801.
Rodger, I.W., Voltage-dependent and receptor-operated calcium channels, in Airways smooth muscle: biochemical control of contraction and relaxation, D. Raeburn and M.A. Giembycz, Editors. 1994, Birkhäuser Verlag: Basel. p. 155–168.
McFadzean, I. and A. Gibson, The developing relationship between receptor-operated and store-operated calcium channels in smooth muscle. Br J Pharmacol, 2002. 135(1): p. 1–13.
Mounkaila, B., R. Marthan, and E. Roux, Biphasic effect of extracellular ATP on human and rat airways is due to multiple P2 purinoceptor activation. Respir Res, 2005. 6(1): p. 143.
Sanders, K.M., Invited review: mechanisms of calcium handling in smooth muscles. J Appl Physiol, 2001. 91(3): p. 1438–49.
Helli, P.B., E. Pertens, and L.J. Janssen, Cyclopiazonic acid activates a Ca2+−permeable, nonselective cation conductance in porcine and bovine tracheal smooth muscle. J Appl Physiol, 2005. 99(5): p. 1759–68.
Ay, B., et al., Store-operated Ca2+ entry in porcine airway smooth muscle. Am J Physiol Lung Cell Mol Physiol, 2004. 286(5): p. L909–17.
Marthan, R., Store-operated calcium entry and intracellular calcium release channels in airway smooth muscle. Am J Physiol Lung Cell Mol Physiol, 2004. 286(5): p. L907–8.
Janssen, L.J., D.K. Walters, and J. Wattie, Regulation of [Ca2+]i in canine airway smooth muscle by Ca(2+)-ATPase and Na+/Ca2+ exchange mechanisms. Am J Physiol, 1997. 273(2 Pt 1): p. L322–30.
Flores-Soto, E., et al., In airways ATP refills sarcoplasmic reticulum via P2X smooth muscle receptors and induces contraction through P2Y epithelial receptors. Pflugers Arch, 2011. 461(2): p. 261–75.
Liu, B., et al., Reverse mode Na+/Ca2+ exchange mediated by STIM1 contributes to Ca2+ influx in airway smooth muscle following agonist stimulation. Respir Res, 2010. 11: p. 168.
Iwamoto, T. and M. Shigekawa, Differential inhibition of Na+/Ca2+ exchanger isoforms by divalent cations and isothiourea derivative. Am J Physiol, 1998. 275(2 Pt 1): p. C423–30.
Pitt, A. and A.J. Knox, Molecular characterization of the human airway smooth muscle Na+/Ca2+ exchanger. Am J Respir Cell Mol Biol, 1996. 15(6): p. 726–30.
Sims, S.M., Y. Jiao, and Z.G. Zheng, Intracellular calcium stores in isolated tracheal smooth muscle cells. Am J Physiol, 1996. 271(2 Pt 1): p. L300–9.
Roux, E., J.-P. Mazat, and M. Marhl, Role of mitochondria in calcium homeostasis and contraction of smooth muscle cells, in Mitochondria: Structure, Functions and Dysfunctions, O.L. Svensson, Editor. 2009, Nova Publisher: Hauppauge NY.
Roux, E., et al., Modelling of calcium handling in airway myocytes. Prog Biophys Mol Biol, 2006. 90(1–3): p. 64–87.
Marhl, M., et al., Modelling oscillations of calcium and endoplasmic reticulum transmembrane potential; role of the signalling and buffering proteins and of the size of the Ca2+ sequestering ER subcompartments. Bioelectrochem Bioenerg, 1998. 46: p. 79–90.
Bai, Y. and M.J. Sanderson, The contribution of Ca2+ signaling and Ca2+ sensitivity to the regulation of airway smooth muscle contraction is different in rats and mice. Am J Physiol Lung Cell Mol Physiol, 2009. 296(6): p. L947–58.
Mbikou, P., et al., Theoretical and experimental investigation of calcium-contraction coupling in airway smooth muscle. Cell Biochem Biophys, 2006. 46(3): p. 233–52.
Roux, E., M. Duvert, and R. Marthan, Combined effect of chronic hypoxia and in vitro exposure to gas pollutants on airway reactivity. Am J Physiol Lung Cell Mol Physiol, 2002. 283(3): p. L628–35.
Roux, E., et al., Calcium signaling in airway smooth muscle cells is altered by in vitro exposure to the aldehyde acrolein. Am J Respir Cell Mol Biol, 1998. 19(3): p. 437–44.
Perez-Zoghbi, J.F., Y. Bai, and M.J. Sanderson, Nitric oxide induces airway smooth muscle cell relaxation by decreasing the frequency of agonist-induced Ca2+ oscillations. J Gen Physiol, 2010. 135(3): p. 247–59.
Jiang, H., et al., Phosphoinositide 3-kinase gamma regulates airway smooth muscle contraction by modulating calcium oscillations. J Pharmacol Exp Ther, 2010. 334(3): p. 703–9.
Delmotte, P. and M.J. Sanderson, Effects of formoterol on contraction and Ca2+ signaling of mouse airway smooth muscle cells. Am J Respir Cell Mol Biol, 2010. 42(3): p. 373–81.
Bai, Y., M. Zhang, and M.J. Sanderson, Contractility and Ca2+ signaling of smooth muscle cells in different generations of mouse airways. Am J Respir Cell Mol Biol, 2007. 36(1): p. 122–30.
Bergner, A., et al., Ca2+−signaling in airway smooth muscle cells is altered in T-bet knock-out mice. Respir Res, 2006. 7: p. 33.
Perez, J.F. and M.J. Sanderson, The frequency of calcium oscillations induced by 5-HT, ACH, and KCl determine the contraction of smooth muscle cells of intrapulmonary bronchioles. J Gen Physiol, 2005. 125(6): p. 535–53.
Bergner, A. and M.J. Sanderson, Acetylcholine-induced calcium signaling and contraction of airway smooth muscle cells in lung slices. J Gen Physiol, 2002. 119(2): p. 187–98.
Campos-Bedolla, P., et al., Airway smooth muscle relaxation induced by 5-HT(2A) receptors: role of Na(+)/K(+)-ATPase pump and Ca(2+)-activated K(+) channels. Life Sci, 2008. 83(11–12): p. 438–46.
Oguma, T., et al., Roles of P2X receptors and Ca2+ sensitization in extracellular adenosine triphosphate-induced hyperresponsiveness in airway smooth muscle. Clin Exp Allergy, 2007. 37(6): p. 893–900.
Janssen, L.J., T. Tazzeo, and J. Zuo, Enhanced myosin phosphatase and Ca(2+)-uptake mediate adrenergic relaxation of airway smooth muscle. Am J Respir Cell Mol Biol, 2004. 30(4): p. 548–54.
Prakash, Y.S., et al., Spatial and temporal aspects of ACh-induced [Ca2+]i oscillations in porcine tracheal smooth muscle. Cell Calcium, 2000. 27(3): p. 153–62.
Fleischmann, B.K., Y.X. Wang, and M.I. Kotlikoff, Muscarinic activation and calcium permeation of nonselective cation currents in airway myocytes. Am J Physiol, 1997. 272(1 Pt 1): p. C341-9.
Wang, Y.X., B.K. Fleischmann, and M.I. Kotlikoff, M2 receptor activation of nonselective cation channels in smooth muscle cells: calcium and Gi/G(o) requirements. Am J Physiol, 1997. 273(2 Pt 1): p. C500–8.
Kajita, J. and H. Yamaguchi, Calcium mobilization by muscarinic cholinergic stimulation in bovine single airway smooth muscle. Am J Physiol, 1993. 264: p. L496–L503.
Schaafsma, D., et al., Differential Rho-kinase dependency of full and partial muscarinic receptor agonists in airway smooth muscle contraction. Br J Pharmacol, 2006. 147(7): p. 737–43.
Janssen, L.J. and S.M. Sims, Ca(2+)-dependent Cl- current in canine tracheal smooth muscle cells. Am J Physiol, 1995. 269(1 Pt 1): p. C163–9.
McCann, J.D. and M.J. Welsh, Calcium-activated potassium channels in canine airway smooth muscle. J Physiol, 1986. 372: p. 113–27.
Wade, G.R. and S.M. Sims, Muscarinic stimulation of tracheal smooth muscle cells activates large-conductance Ca(2+)-dependent K+ channel. Am J Physiol, 1993. 265(3 Pt 1): p. C658–65.
Amrani, Y., et al., Ca2+ increase and Ca(2+)-influx in human tracheal smooth muscle cells: role of Ca2+ pools controlled by sarco-endoplasmic reticulum Ca(2+)-ATPase 2 isoform. Br J Pharmacol, 1995. 115(7): p. 1204–10.
Govindaraju, V., et al., The effects of extracellular purines and pyrimidines on human airway smooth muscle cells. J Pharmacol Exp Ther, 2005. 315(2): p. 941–8.
Hyvelin, J.M., et al., Human isolated bronchial smooth muscle contains functional ryanodine/caffeine-sensitive Ca-release channels. Am J Respir Crit Care Med, 2000. 162(2 Pt 1): p. 687–94.
Ressmeyer, A.R., et al., Human airway contraction and formoterol-induced relaxation is determined by Ca2+ oscillations and Ca2+ sensitivity. Am J Respir Cell Mol Biol, 2010. 43(2): p. 179–91.
Liu, X. and J.M. Farley, Acetylcholine-induced Ca++−dependent chloride current oscillations are mediated by inositol 1,4,5-trisphosphate in tracheal myocytes. J Pharmacol Exp Ther, 1996. 277(2): p. 796–804.
Roux, E., et al., Modelling of Ca2+−activated chloride current in tracheal smooth muscle cells. Acta Biotheoretica, 2001. 49(4): p. 291–300.
Marhl, M., et al., Complex calcium oscillations and the role of mitochondria and cytosolic proteins. Biosystems, 2000. 57(2): p. 75–86.
Liu, X. and J.M. Farley, Acetylcholine-induced chloride current oscillations in swine tracheal smooth muscle cells. J Pharmacol Exp Ther, 1996. 276(1): p. 178–86.
Bai, Y. and M.J. Sanderson, Airway smooth muscle relaxation results from a reduction in the frequency of Ca2+ oscillations induced by a cAMP-mediated inhibition of the IP3 receptor. Respir Res, 2006. 7: p. 34.
Ouedraogo, N., et al., Effects of intravenous anesthetics on normal and passively sensitized human isolated airway smooth muscle. Anesthesiology, 1998. 88(2): p. 317–26.
Haberichter, T., et al., The influence of different InsP(3) receptor isoforms on Ca(2+) signaling in tracheal smooth muscle cells. Bioelectrochemistry, 2002. 57(2): p. 129.
Marhl, M., D. Noble, and E. Roux, Modeling of molecular and cellular mechanisms involved in Ca2+ signal encoding in airway myocytes. Cell Biochem Biophys, 2006. 46(3): p. 285–302.
Berridge, M.J., P. Lipp, and M.D. Bootman, The versatility and universality of calcium signalling. Nat Rev Mol Cell Biol, 2000. 1(1): p. 11–21.
Babcock, D.F., et al., Mitochondrial participation in the intracellular Ca2+ network. J Cell Biol, 1997. 136(4): p. 833–44.
Drummond, R.M. and F.S. Fay, Mitochondria contribute to Ca2+ removal in smooth muscle cells. Pflugers Arch, 1996. 431(4): p. 473–82.
Fall, C.P. and J.E. Keizer, Mitochondrial modulation of intracellular Ca(2+) signaling. J Theor Biol, 2001. 210(2): p. 151–65.
Wylam, M.E., A. Xue, and G.C. Sieck, Mechanisms of intrinsic force in small human airways. Respir Physiol Neurobiol, 2012. 181(1): p. 99–108.
Wang, I.Y., et al., A mathematical analysis of agonist- and KCl-induced Ca(2+) oscillations in mouse airway smooth muscle cells. Biophys J, 2010. 98(7): p. 1170–81.
Bergner, A. and M.J. Sanderson, ATP stimulates Ca2+ oscillations and contraction in airway smooth muscle cells of mouse lung slices. Am J Physiol Lung Cell Mol Physiol, 2002. 283(6): p. L1271–9.
Hagar, R.E. and B.E. Ehrlich, Regulation of the type III InsP(3) receptor by InsP(3) and ATP. Biophysical Journal, 2000. 79(1): p. 271–278.
Moraru, II, et al., Regulation of type 1 inositol 1,4,5-trisphosphate-gated calcium channels by InsP3 and calcium: Simulation of single channel kinetics based on ligand binding and electrophysiological analysis. J Gen Physiol, 1999. 113(6): p. 837–49.
Ramos-Franco, J., et al., Single channel function of recombinant type-1 inositol 1,4,5-trisphosphate receptor ligand binding domain splice variants. Biophys J, 1998. 75(6): p. 2783–93.
Ramos-Franco, J., M. Fill, and G.A. Mignery, Isoform-specific function of single inositol 1,4,5-trisphosphate receptor channels. Biophys J, 1998. 75(2): p. 834–9.
Dupont, G. and A. Goldbeter, One-pool model for Ca2+ oscillations involving Ca2+ and inositol 1,4,5-trisphosphate as co-agonists for Ca2+ release. Cell Calcium, 1993. 14(4): p. 311–22.
Morel, J.L., et al., Crucial role of type 2 inositol 1,4,5-trisphosphate receptors for acetylcholine-induced Ca2+ oscillations in vascular myocytes. Arterioscler Thromb Vasc Biol, 2003. 23(9): p. 1567–75.
Parthimos, D., D.H. Edwards, and T.M. Griffith, Minimal model of arterial chaos generated by coupled intracellular and membrane Ca2+ oscillators. Am J Physiol, 1999. 277(3 Pt 2): p. H1119–44.
Roux, E., P. Mbikou, and A. Fajmut, Role of Protein Kinase Network in Excitation-Contraction Coupling in Smooth Muscle Cell, in Protein Kinases, G. Da Silva Xavier, Editor. 2012, InTech: Rijeka. p. 287–320.
Mbikou, P., E. Roux, and A. Fajmut, Couplage excitation-contraction du muscle lisse des voies aeriennes. 2010, Sarrebruck: Éditions universitaires européennes. 144.
Mbikou, P., et al., Contribution of Rho kinase to the early phase of the calcium-contraction coupling in airway smooth muscle. Exp Physiol, 2011. 96(2): p. 240–58.
Hai, C.M. and B. Szeto, Agonist-induced myosin phosphorylation during isometric contraction and unloaded shortening in airway smooth muscle. Am J Physiol, 1992. 262(1 Pt 1): p. L53–62.
Ouedraogo, N., R. Marthan, and E. Roux, The effects of propofol and etomidate on airway contractility in chronically hypoxic rats. Anesth Analg, 2003. 96(4): p. 1035–41.
Hai, C.M. and R.A. Murphy, Regulation of shortening velocity by cross-bridge phosphorylation in smooth muscle. Am J Physiol Cell Physiol, 1988. 255(1): p. C86–94.
Hai, C.-M. and H.R. Kim, An expanded latch-bridge model of protein kinase C-mediated smooth muscle contraction. J Appl Physiol, 2005. 98(4): p. 1356–1365.
Hai, C.M. and R.A. Murphy, Cross-bridge phosphorylation and regulation of latch state in smooth muscle. Am. J. Physiol. Cell Physiol., 1988. 254(1): p. C99–106.
Lauzon, A.M., et al., A multi-scale approach to airway hyperresponsiveness: from molecule to organ. Front Physiol, 2012. 3: p. 191.
Berridge, M.J., P.H. Cobbold, and K.S. Cuthbertson, Spatial and temporal aspects of cell signalling. Philos Trans R Soc Lond B Biol Sci, 1988. 320(1199): p. 325–43.
Marhl, M., et al., Importance of cell variability for calcium signaling in rat airway myocytes. Biophys Chem, 2010. 148: p. 42–50.
Burdakov, D. and A. Verkhratsky, Biophysical re-equilibration of Ca2+ fluxes as a simple biologically plausible explanation for complex intracellular Ca2+ release patterns. FEBS Lett, 2006. 580(2): p. 463–8.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Roux, E. (2014). Temporal Aspects of Ca2+ Signaling in Airway Myocytes. In: Wang, YX. (eds) Calcium Signaling In Airway Smooth Muscle Cells. Springer, Cham. https://doi.org/10.1007/978-3-319-01312-1_8
Download citation
DOI: https://doi.org/10.1007/978-3-319-01312-1_8
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-01311-4
Online ISBN: 978-3-319-01312-1
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)