Abstract
Over the last half-century, the poultry production industry witnessed a great dependency on the in-feed use of antimicrobials to promote health and productivity. However, the continuous and uncontrolled use led to the ban of growth-promoting antibiotics (AGPs) due to resistance and residue concerns. In addition, intensive farm management procedures of modern turkey rearing systems also result in physiological, biochemical and metabolic changes that are difficult to address. Consequently, turkey growers are faced with the challenge of maintaining productivity and preventing diseases on their farms. These challenges facilitated the development and evaluation of novel antibiotic alternatives, such as probiotics, prebiotics, synbiotics, organic acids, enzymes, yeast-based products, phytogenics, bacteriophages and algal derivates. The main purported mode of action of these feed additives was to manipulate gut microbiota to achieve optimal gut function and integrity along with balanced microbiota for better growth and improved health status of turkeys. However, reports of the effect of dietary supplementation with these additives on turkey performance traits are contradictory and inconclusive. In addition, a vast number of studies with broiler chickens have revealed distinctive characteristics of these additives under intestinal health challenges or any other management stressor, but experiments with turkeys under such circumstances are considerably limited and still require elucidation. It is apparent that for a deeper understanding of the effectiveness of these additives purported to maintain gut health and production efficiency in turkey production, a more comprehensive approach is needed beyond the mere use of the additives. Therefore, the current review aims to compile results published with turkeys and suggest how the use of these novel feed additives may improve the overall health status of turkeys and production efficiency in a sustainable, economic, environmental- and animal welfare-friendly way while enabling safe food for consumers.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Commercial turkey production practices have evolved considerably over the last half-century. This has resulted from improvements in poultry genetics, nutrition, management and health maintenance, among other factors (Scott 1987; Grashorn and Bessei 2004; Huff et al. 2007; Chiang et al. 2008; Hafez and Awad 2021; Gous et al. 2022; Krautwald-Junghannsa and Janja 2022). Modern hybrid male and female turkeys weighing almost 18 kg and 14 kg, respectively, after a 112 days fattening period are the result of an intensive and continuous artificial selective process for commercial objectives, which has led to increased productivity (Huff et al. 2008; Hocking 2014). However, this continuous selection process also brought physiologic, biochemical and metabolic changes and immunodepression associated with the intensive farm management procedures of modern turkey production rearing systems (Hocking 2014; Huff et al. 2014; Erasmus 2018). For almost a half-century, turkey producers have achieved great success in addressing certain challenges, such as those hampering optimal bird health and productive output, by supplementation with AGPs (Leeson 1984; Firman and Kirn 1989; Waibel et al. 1991; Ferket 2013).
However, the emergence of antibiotic-resistant bacteria and the rise in antibiotic residue in edible meat have led to consumer concerns over their use in feed and have resulted in a move away from the use of antibiotics in livestock production worldwide (Neveling and Dicks 2021). The consideration of strict antimicrobial restrictions on their use in animal agriculture also led the turkey industry to seek efficient, cost-effective, sustainable and environmentally friendly alternatives to AGPs with no residue or resistant properties (Ferket 2013).
Consequently, a vast number of new feed additives have become continuously available in the market, with the purpose of manipulating the functionality, structure, integrity and microbial composition of the intestinal tract to manage intestinal health challenges and maintain optimal growth performance. Strategies to accomplish these crucial mechanisms of the digestive system through organic acids, probiotics, prebiotics, synbiotics, phytochemicals, yeast-based products and enzymes are showing promise in enhancing general health status and production efficiency in turkeys. Although this list includes the most researched alternatives, there are exceptional alternatives to antibiotics that have been discussed by other authors. Bacteriocins (Cole et al. 2006), antimicrobial peptides (Forkus et al. 2017), clays (Denli et al. 2009), and several trace minerals (Flores et al. 2021) have also been reviewed as antibiotic alternatives, but only organic acids, probiotics, prebiotics, yeast-based products, phytogenics and enzyme alternatives are discussed in the current literature review. These principal control strategies are likely to include combinations of products as replacements for AGPs such as synbiotics (Omidiwura et al. 2018; Czech et al. 2020). Thus, there is a great opportunity to demonstrate the efficacy of six kinds of alternative strategies to feed antibiotics in the turkey production chain, but adequate information on the use of exceptional strategies is significantly lacking.
Altogether, the available information indicates that these compounds could be used as dietary supplements with various applications, including antimicrobial, immune system stimulation, antiparasitic, anti-inflammatory and antioxidant actions (Hollister et al. 1994; Hafez and Hauck 2006; Dutta et al. 2008; Loeffler 2014), as well as several beneficial reflections on meat quality traits, including microbial and sensory actions (Botsoglou et al. 2003; Dewi et al. 2021). Furthermore, positive implications regarding environmental and turkey welfare outcomes through supplementation of these feed additives were demonstrated (Odetallah et al. 2002; Huff et al. 2011). Meeting these demands while still being profitable and successful is often a great challenge for turkey producers. This review provides a brief overview of performance-enhancing feed additive use in turkey production and introduces some examples of how dietary and other management factors interact with their mechanism of action.
2 Organic Acids
In the feeding of poultry, organic acids, known to have strong antibacterial effects, have been used as control agents to reduce the intraluminal concentration of coliform bacteria and other acid-intolerant organisms, such as Campylobacter and Salmonella, known to be involved in digestive disorders (Izat et al. 1990; Ricke 2003; Immerseel et al. 2006). Organic acids, so-called acidifiers, also have several additional effects on gastrointestinal tract (GIT) health and productive performance. These include a reduction in digesta pH, increased pancreatic secretion, and enhanced feed utilization, thereby increasing the digestion and absorption of nutrients (Patten and Waldroup 1988; Yang et al. 2018).
The majority of research studies related to the use of organic acids have been conducted on broiler chickens, often with inconsistent results (Günal et al. 2006; Hernández et al. 2006; Adewole et al. 2021). There are a limited number of studies with hybrid turkey toms; scant information is available on organic acids as feed additives in turkey diets, reporting antibacterial activity in the gut with some success but failure to promote body weight (BW), body weight gain (BWG), feed conversion rate (FCR) or meat yield.
In the study by Wajda et al. (2010), formic acid, mostly used as an acidifying agent in poultry and pig production, was administered to Big 6 turkey toms via the drinking water route over the entire experimental growth period (from 1 to 4 weeks of age—5 l/tonne, from 5 to 8 weeks of age—4 l/tonne, from 9 to 18 weeks of age—3 l/tonne). Body weight gain, FCR and mortality rate determined at 4, 8, 12 and 18 weeks of age were not affected by formic acid (Acidum formicum) intake. Slaughter characteristics (the percentage rate of carcass yield, breast muscles, thigh muscles, drumstick muscles, total leg muscles and abdominal fat), chemical composition (percentage of dry matter, total crude protein, fat and ash) and sensory properties (pH 24, brightness, water-holding capacity, palatability, juiciness and tenderness) of meat from turkeys determined at the end of the trial were comparable between birds treated with and without formic acid.
One strategy is to increase the effectiveness of organic acids by means of an antimicrobial in-feed preparation by combining them with phytogenic compounds with certain antibacterial activities (Basmacioğlu-Malayoğlu et al. 2016; Stefanello et al. 2020). Such a combination strategy showed benefits in broiler chickens in terms of growth performance indices, villus structure and absorptive surface area and reduced the pathogenic bacteria count in the intestines (Bozkurt et al. 2012; Weber et al. 2012; Gheisar et al. 2015). This synergistic mechanism of action between organic acids and plant essential oils, formerly demonstrated in broiler chickens, was tested on 400 male turkeys (Large White Big 6) from one day of age to 140 days of age (Mikulski et al. 2008). For this purpose, a four-phase basal feed mixture without any performance-enhancing feed additives was initially prepared and then supplemented with either a blend of organic acids (formic acid and propionic acid) or a mixture of organic acids (citric acid, fumaric acid, orthophosphoric acid and malic acid) and an essential oil blend from citrus fruits, cinnamon, oregano and thyme. Such a combination of specific plant bioactives with diverse antimicrobial and enzymatic activities and organic acids with strong antimicrobial properties, either alone or in combination, showed no notable effect on BWG or FCR in turkeys after a 140-day feeding period. When compared to the unsupplemented treatment, significant reductions were determined in crop pH with dietary supplementation of organic acids and blends of essential oils, but no such effect was observed for gizzard and caecal pH levels. The enzymatic activities of α-glucosidase, β-glucosidase, α-galactosidase, β-galactosidase and β-glucuronidase in the caecal content were significantly higher in birds fed the tested supplements than in those fed the unsupplemented control, while only α-glucosidase responded positively to the organic acid blend in ileal digesta. These improvements in enzymatic activities derived from the organic acid and essential oil supplementation do not seem to be reflected in nutrient utilization and, ultimately, the final growth performance of the turkeys.
In contrast to observations on the performance of hybrid turkeys reared under standard management conditions, organic acids demonstrated some beneficial effects in improving the performance of turkey poults at risk for potentially pathogenic bacteria. In this regard, successful results were illustrated by Roy et al. (2002), who used a commercial organic acid blend (primarily propionic acid; Myco Curb) to control poult enteritis and mortality syndrome (PEMS), which describes potentially lethal enteritis of young turkeys (Yu et al. 2000). Myco Curb, a mould inhibitor for processed feed ingredients and animal feeds, was maintained in the feed for the duration of the 3-week periods. At 6 days post-hatch, BUT Big 9 poults were given a 1-mL oral gavage of a 10% suspension of faeces from coronavirus-negative PEMS-positive poults. Myco Curb was supplemented into the diet at levels of 1.25 and 2.5%. Myco Curb delayed the onset of the initial mortality spike associated with the disease and reduced the cumulative mortality by more than 50%. The marked reduction in mortality in the poults treated with Myco Curb was attributed to decreased bacterial content of the gut and the maintenance of packed cell volume and haemoglobin content. However, the beneficial attributes to gut health and liveability generated by fortification of the diet with Myco Curb were not reflected in the growth performance indices of turkeys. Male and female poults had a significantly lower BW and higher FCR when they were given 2.5% Myco Curb in their feed, while no significant changes were observed at dietary inclusion levels of 0.625 and 1.25%.
Effects similar to those in the abovementioned work were reported in a study (Milbradt et al. 2014a) in which 1-day-old female commercial cross turkey poults (BUT Big 9) were fed diets supplemented with a mixture of short- and medium-chain fatty acids (acetic acid, formic acid, propionic acid, sorbic acid and vegetal fatty acids). Although the organic acid blends had no significant effects on the intestinal villus height, crypt depth, or villus: crypt ratio, negative effects on the early gain or feed intake were observed throughout the study. The same failure was witnessed in another experiment conducted for a 60-day period using white California turkey toms (Çelik et al. 2003). No significant effect of 2% dietary supplementation with a commercial organic acid blend (Biotronic® SE) comprised of formic acid (17%), ammonium format (14.1%), propionic acid (12.4%), ammonium propionate (%8.4) and carrier material (47.7%) on the BWG and FCR of turkeys was found.
Incompetence in improving BWG and FCR in turkey toms between 0 and 28 days of age with dietary organic acid administration was also reported in a more recent study (Milbradt et al. 2017), while a significant reduction in caecal numbers of Salmonella enteritis was observed. In a former study (Milbradt et al. 2014b), in-water treatment with a blend of organic acids (formic acid, acetic acid, ammonium format, mono- and diglyceride of unsaturated fatty acids and copper acetate) elicited significant reductions in the number of Salmonella enteritidis colony forming units in the caecum and crop of turkeys (BUT Big 9) that were challenged 72 and 24 hours prior to slaughter. However, it was noted that only constant treatment of organic acids in drinking water resulted in antibacterial activity, while the transient administration regimen failed to do so.
As demonstrated in a very recent study (Makowski et al. 2022), significant levels of decreases in faecal populations of Escherichia coli and Clostridium perfringens accompanying an increase in the gizzard pH were a manifestation of antimicrobial activity generated by butyric acid glycerides or coated sodium butyrate when added to turkey diets. The addition of butyric acid in different forms to turkey diets increased the butyric acid concentration in the caecal digesta and ileal protein digestibility and in turn improved FCR.
In brief, as presented in Table 1, almost all the published studies have unanimously concluded that organic acids, irrespective of their chemical characteristics and application procedures, may have the potential to be more effective in the field, where poults are subjected to repeated challenges regarding disease pressure, stocking density, litter quality, diet characteristics and management practices. The lack of reflection of the certain antimicrobial activity posed by organic acids in the growth performance of turkeys is noticeable.
3 Enzymes
The use of exogenous microbial enzymes as feed additives is a well-established practice in food-producing animal production, and their application is ubiquitous in commercial broiler and laying hen diets as a tool to enhance depolymerization of nonstarch polysaccharides (NSP), reduce digesta viscosity and modulate bird intestinal health (Bedford and Schulze 1998; Adeola and Cowieson 2011; Mahmood and Guo 2020). In particular, the application of phytase and, to a lesser extent, xylanases and ß-glucanases has been widely adopted, but many other enzyme classes can significantly improve the utilization of feed, such as proteases, pectinases, amylases, pentosanases, beta-mannanase, galactosidases, or cellulase. Evidently, supplementation with these different enzymes in poultry gives a wide range of effects on growth performance, nutrient digestibility and health (Slomonski 2011). However, in turkey feeding, there has been a lack of studies examining exogenous enzyme efficacy for over three decades (Ferket 1993, 2013; Nguyen et al. 2022).
In a study by Odetallah et al. (2002), two endoxylanase enzyme preparations (Natugrain and Lyxasan forte), with apparently different modes of action, were fed to turkey toms aged between 8 and 140 days, either alone or in combination (Natugrain Blend). Cumulative feed consumption was higher by 2 kg or more in birds treated with Natugrain Blend than in other treatments. Accordingly, Natugrain Blend markedly improved body weight at 84 and 112 days of age in comparison to the unsupplemented control diet. Lyxasan forte had the best feed/gain ratio throughout the experiment. The liveability and feather scores of birds were significantly improved in all three enzyme treatments compared to the control.
Ritz et al. (1995) found that pancreatic organ weight and pancreatic amylase activity were not consistently affected by diet in an experimental schedule in which male turkeys were administered commercial preparations of amylase (Avizyme TK0492-1®) and xylanase (TK0492-1®) from hatch to 8 weeks of age. Amylase activity within the intestinal chyme increased sporadically with dietary amylase supplementation compared to the control and xylanase-supplemented diets. The authors concluded that increased supplemental amylase activity levels may provide more conclusive evidence of an additive effect of dietary amylase and endogenous amylase activity.
Findings determined from several studies indicated that the age and metabolic needs of the turkey influence enzyme activity levels within the pancreas and small intestine. In a study by Ritz et al. (1995), pancreatic amylase activity seemed to increase in a linear fashion with age and continued to follow this progressive rise throughout the 8-week period. This continued increase in activity contradicts the results reported by Krogdahl and Sell (1989), who observed a stabilization in pancreatic amylase after 21 days. The observations of McKnight (1997) are parallel to earlier establishments that exogenous xylanase supplementation in a wheat-based diet was only effective at a young age (up to 70 days of age).
The rearing system of turkeys was found to intervene in the efficacy of in-feed enzymes in a study by García-López et al. (2011). Addition of a commercial enzyme preparation (Allzyme-Vegpro; mixture of protease, amylase, cellulase, beta-galactosidase and pentosanases) to the diet resulted in a lack of bird response in a semi-confinement rearing system with daily access for 4 hours to grassland meadows, whereas enzyme supplementation improved the BWG and FCR of turkeys kept in conventional confinements.
Zduńczyk et al. (2013) examined the relationship between dietary fibre status and supplementation with an enzyme premix containing pectinase, cellulase, xylanase, glucanase, mannanase, galactanase, amylase and protease on endogenous intestinal enzyme secretion of Big 6 male turkeys. From 1 to 56 days of age, the enzyme premix was added to a soybean-based control diet and an experimental diet in which rapeseed meal was gradually replaced with soybean up to 180 g/kg level. The results indicated that, regardless of the diet type, enzyme supplementation tended to reduce ileal viscosity, decrease ammonia concentration, increase the glycolytic activities of the intestinal microflora enzymes α-glucosidase, α-galactosidase and β-galactosidase, decrease the activity of β-glucuronidase and increase BWG in turkeys. The authors suggested that supplementation with the NSP-degrading enzyme could maintain adequate endogenous glucosidase and galactosidase activity levels for digestion in the high fibre-containing diets with rapeseed meal inclusion while maintaining gut physiology parameters of turkeys similar to those fed the SBM-based diet. However, in the previous work by the same research team (Juśkiewicz et al. 2010), significant decreases were observed in the final body weight, small intestine and caecal tissue mass, caecal digesta mass and production of volatile fatty acids in the caeca when the same enzyme preparation was used. Of note, the experimental protocol was similar between the two studies except that sunflower meal was replaced with soybean meal instead of rapeseed meal.
Within the framework of this thesis, the magnitude of the response to frost damage during seed development and dietary energy density was examined in turkey toms by dietary supplementation with a natural blend of enzymes (Natugrain Blend®) containing endoxylanases and other enzymes (β-glucanase, hemicellulase, cellulase and protease) (Santos 2002). The results from two consecutive trials demonstrated that enzyme supplementation had positive effects on the nutrient utilization of different wheat sources and cultivation conditions. In addition, different sources of supplemental enzymes had variable effects according to the age of the birds. Phospholipase alleviated the adverse effect of dietary NSP by improving fat digestion and absorption in young turkeys, whereas endoxylanase was more effective in older birds, which have greater digestive capacity and a more mature gut microbial ecosystem.
Ayoola et al. (2015) carried out two trials to examine the effects of dietary supplementation with either β-mannanase, a commercial blend of xylanase, amylase and protease, or direct-fed microbes on the gut health of turkeys. The research results demonstrated that supplements can improve gut health in 42-day-old Nicholas turkey poults, as indicated by improved morphological development of the enteric mucosa and reduction in adherent ileal mucin secretion. Similarly, in a more recent comprehensive thesis study, interactive effects between enzyme supplementation and the NSP profile of the diet and particle size distribution were assessed by feeding toms from 1 to 21 days of age (Flores Maldonado 2018). It was determined that supplementation with a commercial enzyme cocktail (Rovabio Advance, Adisseo) containing arabinofuranosidases and xylanases may improve the digestibility of feed formulated with a low crude protein soybean meal and screened feeds by reducing the negative effects of NSP in the intestinal tract.
Accumulated research over the past three decades has shown that phytase enzymes have profound effects on health, performance and skeletal development, as they influence phosphorus utilization and retention and bone mineralization in poultry. Today, nearly all broiler and layer hen feed now contain phytase enzyme products formulated in place of inorganic phosphorus preparations such as mono- and dicalcium phosphate (Selle and Ravindran 2007; Cowieson et al. 2011). The application of phytase has been widely adopted in other poultry species but has been much less investigated in turkeys. In one of the two available studies with turkeys (Brodacki et al. 2009), phytase supplementation in diets (Ronozyme P; provides 750 FTU phytase enzyme per g feed) from 6 to 16 weeks of age did not influence the BWG and FCR of female turkeys. The lack of significant effects due to phytase (regardless of source) on the performance criteria of turkey toms aged between 10 and 21 days was reported by Applegate et al. (2003). The tibia and toe ash, however, were significantly affected by the phytase source (Escherichia coli-derived vs. Aspergillus-or Peniophora-derived phytases) and concentration (0, 250, 500,750 and 1000 U/kg), indicating significant increases in ash content with heightened supplemental phytase levels over 250 U/kg feed.
Barbour et al. (2002) demonstrated that digestion of pressed turkey feathers with an enzyme mixture (protease, lipase and amylase) prior to autoclaving can improve the protein and amino acid nutritional values. Responsiveness of growing turkeys to exogenous in-feed enzyme preparations in terms of productive performance is illustrated in Table 1.
In general, the available scientific literature has demonstrated that supplementation with appropriate enzymes in poultry diets is an effective way to overcome antinutritional effects that mostly stem from the high NSP content in grains. Although relatively limited data are available, turkey poults appear to respond in the same manner as broilers and layer hens. The addition of enzyme products containing xylanase, mostly in combination with other carbohydrases, allows for the inclusion of greater amounts of wheat in diets without loss of performance (Leeson et al. 1996). In addition, glucanases and galactosidase in combination with proteinase and pectinases may work synergistically to improve the nutritive value of diets containing significant levels of soybean meal. Of note, the effect appears to be age-related, with the greatest response generally shown by young birds at 0 to 7–10 weeks.
4 Probiotics
The concept of probiotic microorganisms as in-feed preparations has become an area of much interest in poultry production, particularly for broiler and layer chickens (Rojman and Deborah 2022). However, relatively few reports have been published concerning the use of probiotics in turkey production. The favourable effects of probiotics are thought to be attributed to their ability to regulate gut microbiota balance, which in turn plays a critical role in maintaining host health (Sekirov et al. 2010). Probiotics, which are viable microorganisms with no pathogenic properties, consisting of yeast or bacteria, have also been reported to be useful under several circumstances for proper maintenance of the structure and function of the turkey gut (Marteau et al. 2001; Soccol et al. 2013).
In one of the pioneer studies, Jiraphocakul et al. (1990) compared the effectiveness of dried Bacillus subtilis culture with feed-grade antibiotics in two consecutive trials using female and male turkeys grown for 16 and 20 weeks, respectively. In the former experiment, the dietary B. subtilis culture significantly increased B. subtilis counts in the crop and caecum but failed to influence intestinal Lactobacillus or Escherichia coli counts. BW and FCR were not significantly affected by feeding the B. subtilis culture in this experiment. In the latter experiment, significant improvements in BWG at 12 weeks and FCR at 20 weeks were observed in male birds receiving the B. subtilis culture. The authors concluded that the results of the two experiments differ somewhat with respect to BWG or FCR, probably due to the bird gender difference.
Unbeneficial results from the addition of probiotic preparation [a mixture of Lactobacillus bulgaricus, Lactobacillus acidophilus, Streptococcus thermophilus, Lactococcus lactis and Propionibacterium (0.1 × 109 CFU/per gram)] at 0.05% in the diets of hybrid commercial female turkeys were reported in a recent study by Lalev et al. (2020). Supplementation of the basal diet containing 10% silkworm meal as soybean meal replacement with these probiotics did not affect the performance and meat yield characteristics of the turkeys after a 74-day feeding period (from 56 to 130 days of age). However, fortification of basal turkey grower and finisher diets with a blend of probiotic microorganisms significantly decreased serum glucose and uric acid concentrations at the end of the grow-out period.
Supplemental levels of commercial Bacillus cereus var. Bacillus toyoi strain (Toyocerin®) were the subject of another study using male middle-heavy Big 9 turkey poults (Grela et al. 2009). Addition of Toyocerin probiotic preparation to the diet increased overall BWG (0 to 16 weeks), but significant enhancements in FCR in the initial 12-week growing period disappeared thereafter. The results regarding BWG and FCR indicated that the higher the supplemental probiotic dose (0.2 × 109 versus 1 × 109 B. toyoi CFU per kg of diet), the greater the benefit, suggesting a dose-dependent effectiveness. A similar pattern in the performance traits to that observed in the study of Grela et al. (2009) was described in a later study by Batkowska et al. (2015) in which Big 9 turkey males were fed diets supplemented with 1 × 109 B. toyoi CFU/kg instead of the 0.2 × 109 level. In partial agreement with these earlier determinations, in a more recent study (Dobrowolski et al. 2019), mixed probiotic preparations at three different supplemental levels exerted a beneficial effect on the histological structure of the small intestine; however, the observed effects were dose- and region dependent. The improvements determined in absorption surface area with probiotic treatment were not reflected in turkey growth performance at market age. Similarly, significant enhancements of intestinal microbial populations and gut morphology derived from probiotic supplementation were not paired with food utilization in turkeys, although the final BW increased with probiotics (Agboola et al. 2014).
The effects of adding a microbial preparation or zinc bacitracin, either alone or in combination, to the diet were studied in an initial study by Francis et al. (1978). The addition of either Lactobacillus or zinc bacitracin to the diet resulted in poults with numerically increased BW over poults fed the control diet. The authors noticed that the improvement in growth from the combination of the two supplements was not as great as when either was administered alone. Nevertheless, probiotics have been shown to be more effective as antimicrobial agents than zinc bacitracin, as evidenced by significantly decreased coliform and total aerobe counts in the feed and the digestive system of turkey poults.
Recently, probable synergism between probiotics and prebiotic preparations, so-called synbiotics, has been the subject of several studies. In one study (Omidiwura et al. 2018), turkeys receiving a bacterial culture (B. subtilis), either alone or in combination with a prebiotic preparation, for an 8-week period showed comparable BWG and FCR to untreated control birds. In later work, Czech et al. (2020) found that Big 6 turkeys experienced limited changes in plasma biochemicals, including pro- and antioxidant compounds, enzymes, minerals and hormones, when fed a diet supplemented with fodder yeast (Yarrowia lipolytica or Saccharomyces cerevisiae; 30 g/kg diet) and probiotics after a 112-day feeding period. In this study, the addition of probiotic (a mixture of Bacillus licheniformis-1.6 × 109 CFU/g and Bacillus subtilis-1.6 × 109 CFU/g; 0.5 g/kg diet) alone to a diet generally resulted in a lack of bird response.
In a subsequent study, the effectiveness of two commercial probiotic preparations [(3.2 × 1010 CFU/g Bacillus spp. (BioPlus, Biochem) and 1.0 × 1010 CFU/g Enterococcus faecium (Cylactin, DSM)] and a synbiotic preparation [(containing 2.0 × 109 CFU/g of Lactobacillus spp., 2.0 × 107 CFU/g of Saccharomyces cerevisiae yeast and 2% inulin)] was evaluated using Big 6 turkey toms challenged with dietary mycotoxin (Śliżewska et al. 2020). The basal feed contained wheat that was naturally contaminated with mycotoxin (ochratoxin, OTA), resulting in OTA concentrations in the phase diets ranging from 198 to 462 mg/kg, and was fed for a 15-week grow-out period. The synbiotic preparation modulated the intestinal microbiota, increasing beneficial bacteria while reducing the number of potential pathogens. However, the benefits obtained from both probiotic preparations were markedly inferior to those of synbiotic application. The benefits in intestinal health resulting from these preparations did not translate into productivity in toms as measured by BWG and FCR at 6 and 14 weeks of age. This means that mycotoxin-induced performance failures could not be ameliorated by probiotic and synbiotic preparations.
In a study comprising two consecutive experiments carried out by Torres-Rodriguez et al. (2007), the effects of a commercially available lactic acid bacterium as a probiotic alone and supplemented with lactose as a prebiotic were evaluated to determine its effects on turkey BW during the brooding and grow-out phases under commercial conditions. Regardless of whether the probiotic was administered in drinking water or feed, the combination of pro- and prebiotic caused increased BW at days 26 and 28 in the first experiment and at market age in the later trial compared with the control group. However, treatment with probiotic microorganisms alone was ineffective in significantly changing the growth rate of hybrid turkeys at any age.
In terms of intestinal development, it was determined that poults differed in the response to different commercial probiotic preparations incorporated at the recommended commercial dosage (Loeffler 2014). Supplementation with one of two commercial probiotic preparations between 0 and 11 days of age increased both the villus length and area as well as the crypt depth, but the remaining treatment failed to do so. The beneficial effects of dietary supplementation with commercial direct-fed microbes (Primalac, Star Labs Inc., containing primarily Lactobacillus acidophilus and Lactobacillus casei) were reported in an earlier study by Rahimi et al. (2009). In a factorial experimental design, female one-day-old turkey poults were fed mash or crumbled feed and were subjected to a Salmonella spp. challenge by oral gavage or not. According to measurements performed at day 21, regardless of the feed form and experimental Salmonella infection, the probiotic-fed birds showed increased goblet cell numbers, total goblet cell area, goblet cell mean size, and mucosal thickness and a greater number of segmented filamentous bacteria compared with the controls. The authors postulated that changes in intestinal morphology, as observed in their study, support the optimum gut health and function concept.
Tomaszewska et al. (2016) revealed that bone characteristics and mineralization in meat-type female turkeys interact with the supplementation dose of probiotics. They found that the influence of probiotic administration on bone mineral density, bone mineral concentration, bone tissue density and bone ash was dose-dependent. The investigated properties of long bones in female turkeys were positively affected by probiotic-supplemented diets in a dose-dependent manner. In general, a review of the available data on turkeys supports the concept that poultry gut health, structure and function can be improved by probiotics, but these amendments were barely coupled with growth performance. Different components of probiotics under various methods of administration are discussed in this section and summarized in Table 2.
5 Prebiotics
Many of the yeast products prepared from the yeast Saccharomyces cerevisiae are classified as prebiotics, and were proven to be a good option as alternatives to AGPs in broiler chickens (Rosen 2007a; Hooge and Connolly 2011), turkeys (Hooge 2004; Rosen 2007b) and laying hens (Salami et al. 2022). Prebiotics are expected to decrease the pH of intestinal contents, thus inhibiting the growth of harmful bacteria such as Clostridium and Salmonella and promoting the growth of Lactobacillus spp. and Bifidobacterium spp. found in the intestines (Ofek et al. 1977; Spring et al. 2000; Rehman et al. 2007). The above specific mechanism of action has been well documented with respect to short-chain fructo oligosaccharides (FOS) and the fructose polymer inulin (Rehman et al. 2009). In contrast to the mode of action of antibiotics, which limit or suppress the growth of common gram-positive microflora, mannan oligosaccharides (MOS) and other oligosaccharides can serve as decoy attachment sites for gram-negative pathogens, thereby preventing attachment onto enterocytes and subsequent enteric refection (Oyofo et al. 1989; Newman 1994; Parks et al. 2001). Moreover, over the past three decades, a great amount of information has been gathered on the antimicrobial potential of yeast-based products, particularly for MOS, FOS and the fructose polymer inulin to a lesser extent, with reflections on overall health status and productivity in poultry (Al-Khalaifah 2018; Al-Khalaifah et al. 2019; Reuben et al. 2021; Khomayezi and Adewole 2021; Salami et al. 2022). However, the data on the use of yeast cell derivates as natural feed additives as alternatives to AGPs in turkey diets published to date are inconsistent.
Firman et al. (2013) reported improved FCR and increased pectoralis major yield in turkey toms fed a diet containing yeast culture from 15 to 18 weeks. Bradley and Savage (1995) and Hayat et al. (1993) demonstrated that dietary supplementation with a yeast fermentation product improved the reproductive performance of some turkey breeder hens. They concluded that yeast culture inclusion in turkey breeder hen diets could improve productive efficiency, such as the hatchability of fertile eggs.
Based on the results of a study with a commercial strain of large white turkeys (Nicholas Turkeys), Fritts and Waldroup (2003) concluded that a yeast fermentation product (Bio-Mos®) might be considered as part of an overall feeding program to aid in overcoming the potential loss of AGPs. Data from this study indicated that FCR from 0 to 20 weeks of age was significantly improved by 0.10% Bio-Mos® compared to the negative control diet without performance enhancer feed additive, whereas BW, mortality, and breast meat yield were not significantly (P < 0.05) influenced by either Bio-Mos® or the AGPs. This was in agreement with previously published studies (Olsen 1996; Savage and Zakrzewska 1997), which reported that turkeys fed diets provisioned with Bio-Mos® had significantly improved FCR in the absence of increased BWG. Significantly improved BWG and FCR in turkey poults fed to 8 weeks of age were reported in another study (Savage and Zakrzewska 1996).
In an early feeding study conducted at 3 weeks of age, Fairchild et al. (2001) reported that both Bio-Mos® and bambermycin significantly improved the BWG of turkey poults. Similarly, Parks et al. (2001) observed an improvement in early FCR (0–3 weeks) with the addition of Bio-Mos®, bambermycin, virginamycin and Bio-Mos® and bambermycin used in combination. The addition of Bio-Mos®, as well as the virginiamycin treatments, increased the overall BWG compared to the unsupplemented dietary treatments.
It is noteworthy that antibiotics and MOS improved the FCR of poults from 0 to 3 weeks of age, a period when gut microflora are not fully developed and stabilized. Therefore, these feed additives may provide advantages by stabilizing the gut microflora and limiting colonization by pathogens through the early stages of life.
Based on a meta-analysis of several trials, Hooge (2004) reported that dietary MOS can improve BWG and decrease mortality in turkeys compared to negative control diets, but dietary MOS supplementation does not always improve FCR. Based on the statistical similarity of the bird performance response to either MOS supplementation or antibiotic supplementation, Hooge (2004) suggested that MOS, a polysaccharide-protein complex derived from yeast, could replace AGPs.
In contrast to earlier studies reporting benefits on BWG and FCR of turkey toms, Valancony et al. (2001) observed no difference in slaughter weight or carcass yield of turkeys fed diets with added avilamycin antibiotic and Bio-Mos®; no mention was made regarding FCR. Final BW, feed intake and FCR in turkey toms at 56 days of age were comparable when treated with control, inulin, and MOS at 4 g/kg supplementation level (Zduńczyk et al. 2004a). Similarly, no significant effect of 0.1, 0.25 and 0.5% dietary inclusion of Bio-Mos® was observed on poult performance after an 8-week feeding experiment (Zduńczyk et al. 2004b). In the previous work of this research team (Juśkiewicz et al. 2003), feeding turkeys with diets supplemented with the same levels of Bio-Mos® for a 4-week period did not affect the productivity of the birds as measured by BWG, feed intake and FCR. In their following related study (Juśkiewicz et al. 2005), insignificant effects of fortifying prebiotic (inulin) in turkey diets with different supplementation levels on growth performance were determined though main indicators of gut health have remarkably enhanced.
Unbeneficial effects of feeding diets supplemented with Bio-Mos® and live yeast from 10 to 20 weeks of age on the growth performance of Big 6 male turkeys were reported by Konca et al. (2009). Likewise, Barasch (2012) reported that the inclusion of a yeast fermentation product (Original XPC™, Diamond V Mills) in the breeder hen diet during the first 2 weeks of laying did not have a carry-over effect on the performance of the male progeny compared to the unsupplemented control breeder hens. Another study provided evidence that supplementation with prebiotics containing the same amount of MOS and beta-glucans from Saccharomyces cerevisiae yeast in turkey diets with different levels of protein did not lead to any significant changes in performance, blood biochemistry, serum immunoglobulin concentrations, or haemagglutination-inhibition titres (Vahabi-Asil et al. 2017).
A similar response was seen in a study by Paiva et al. (2010), where turkey hens raised to 9 weeks of age were fed a diet with different levels of the yeast fermentation product. The birds receiving the prebiotic-supplemented diets had higher BWG at four and 6 weeks of age than the control birds. By 9 weeks of age, a significant difference was no longer observed. Likewise, supplementation with lactose as a prebiotic, either alone or in combination with lactic acid bacteria-based probiotics, was demonstrated to increase the BWG of poults by 15% and 17% after 26 and 28 days of treatment, respectively, regardless of the administration route via feed or drinking water (Torres-Rodriguez et al. 2007).
Scrutinization of the available literature on turkey toms reveals that the benefits reaped from the dietary treatment of yeast-cell wall products were notably ahead of improvements in growth performance in terms of mitigating the stress generated by management and health challenges (Juśkiewicz et al. (2006). This phenomenon was supported by two battery experiments conducted by Huff et al. (2007), where 1-week-old male poults and progeny of 33- and 40-week-old hens were subjected to cold stress and respiratory E. coli challenge. Poults were administered a commercial yeast extract feed supplement (Alphamune) for a 3-week period at 504 or 1008 g/t inclusion levels. Immunostimulation using yeast extract supplements protected poults from some of the production loss due to cold stress and E. coli.
Turkeys fed MOS during a specific challenge from Salmonella typhimurium had a decreased incidence of faecal contamination, whereas broilers fed MOS had reduced faecal counts of Salmonella dublin and E. coli (Spring 1996). Schoeni and Wong (1994) also reported a reduction in Campylobacter jejuni colonization when birds were fed MOS. As previously mentioned, Fairchild et al. (1999) observed improved performance of poults challenged with field isolates of E. coli and fed MOS. In agreement, dietary lactose, as a putative prebiotic, was found to be beneficial to intestinal health, as manifested in decreased caecal Salmonella colonization in turkey poults (Corrier et al. 1991; Hollister et al. 1994).
The success of feed supplementation with commercial yeast extract (Alphamune™) in improving the protection of turkeys against the development of Clostridial dermatitis, a production disease of commercial turkeys that is characterized by sudden mortality in market-aged male birds, was also demonstrated in another study (Huff et al. 2014). The results indicated that the addition of Alphamune™ to the feed at 1008 g/tonne may decrease the mortality rate in male poults at 2, 7 and 12 weeks of age, whereas there were no significant differences in mortality at week 16. The antimicrobial activity of another yeast cell derivate, inulin, was shown in a study by Juśkiewicz et al. (2005). They demonstrated that the supplementation of turkey diets with 1.0% inulin led to a significant reduction in E. coli populations, an insignificant but noticeable increase in Bifidobacterium and Lactobacillus counts, and the greatest SCFA production. However, the final BW of turkeys fed inulin was inferior to that of their untreated counterparts.
The association between the stress incurred due to management practice (i.e., moving birds to larger facilities three times per grow-out period and an increase in the turkey disease osteomyelitis complex) and increased colonization with foodborne pathogens was noted in several studies (Dutta et al. 2008; Huff et al. 2009, 2010). By means of healing this stress-induced health problem, it was demonstrated that Alphamune™ effectively protected turkeys from the immunosuppressive effects of incurred transport stress, as evidenced by decreased serum heterophil oxidative burst activity and increased serum corticosterone levels (Huff et al. 2011). The authors suggested that yeast derivates could be used as nutritional immunomodulators that may be applied prior to the transport of turkeys. However, the greater protective response to Alphamune™ in female turkeys than in males is noticeable.
Yeast derivates have also been shown to modulate caecal fermentation metabolites, which are accepted as reliable indicators of gut health, and the proper function and integrity of intestines (Juśkiewicz et al. 2003). As determined by Zduńczyk et al. (2004b), dietary supplementation with commercial yeast derivate (Bio-Mos®, 5 g/kg) reduced ammonia concentrations and enhanced volatile fatty acid concentrations, especially acetate and butyrate, in the caecal digesta. However, a significant enhancement of propionate, isobutyrate and isovalerate concentrations in the digesta was observed when the supplemental dosage of Bio-Mos® was halved (i.e., 2.5 g/kg). A similar MOS product (Bio-Mos®, 1.8 g/kg) showed a positive effect on early intestinal development in poults aged 9 and 11 days, increasing the villus height, villus surface area and crypt depth at both ages (Loeffler 2014).
Overall, a review of the available information stresses that management factors, including the brooding temperature during the starter period, challenge with enteric pathogens and litter quality and production stressors during the later stages of turkey production (e.g., social and psychological stress due to crowding and transportation stress), as well as hen and bird age and gender, should be considered when designing studies to evaluate the antimicrobial potential of yeast-based products as alternatives to AGPs. Table 2 also shows the growth performance responses of turkeys fed to various prebiotic preparations at different concentrations.
6 Phytogenics
Over the past two decades, plant-derived extracts have garnered interest for feed industry applications, with particular interest for pigs and poultry, along with the increase in popularity of the search for natural alternatives to in-feed antibiotics that are free from residue and resistant effects (Mountzouris et al. 2009; Wallace et al. 2010; Diaz-Sanchez et al. 2015; Giannenas et al. 2020). This is similar to the food industry’s prioritization of high-quality, clean-label meat products for consumers (Botsoglou et al. 2003; Keokamnerd et al. 2008; Dewi et al. 2021). Scientific studies with poultry are mostly devoted to broiler chickens and, to a lesser extent, layer hens and findings obtained from studies over the last two decades have been summarized in comprehensive reviews (Bozkurt et al. 2014; Harrington et al. 2020; Jin et al. 2020). However, in turkey diets, the magnitude of the response to dietary supplementation with aromatic plants and their extracts has rarely been compiled. Fortunately, the recent review of Bozkurt and Tüzün (2020) provided excellent coverage of early research appreciating metabolic, physiologic and morphologic changes along with the growth performance traits, gut function, general health status, meat yield and meat quality aspects in response to dietary consumption of plant-derived chemicals. This pioneering review also provides background information for the present review paper. The results obtained from several studies conducted after the publication of the above review are presented below.
The objectives of a recent study carried out by Zumbaugh et al. (2020) were to determine the effect of commercially available plant bioactives (Digestarom®, Biomin) on the performance of turkey poults fed a reduced protein and amino acid diet over a 6-week brooding period. Digestarom® is a proprietary phytogenic feed additive consisting of a unique blend of herbs, essential oils and functional flavours. The results showed that formulating starter and grower diets to be nutrient-deficient negatively affected the growth of poults. However, as the birds fed Digestarom® showed significantly higher (3.5%) BWG than those fed the nonsupplemented and protein-deficient diets, its addition is thought to have helped compensate for the reduced protein and limiting amino acids. However, it had minimal effect on FCR and nutrient digestion parameters, including digestive enzyme activity, pancreatic enzyme gene expression and nutrient transporter gene expression.
Salmonella Heidelberg, an invasive pathogen in humans, is the leading cause of foodborne illness in ground and processed turkey meat (Bearson et al. 2017; CDC 2019). Therefore, continued efforts have been made to mitigate the threat induced by this enteropathogen in poultry products (Antunes et al. 2016; Dewi et al. 2021). For this concept, the antimicrobial efficacy of three plant-derived antimicrobials, lemongrass essential oil, citral and trans-cinnamaldehyde, against S. Heidelberg in ground turkeys was evaluated in a very recent study (Dewi et al. 2021). The results of this study indicated that the three tested plant-derived chemicals are effective against S. Heidelberg in ground turkey during refrigerated storage, indicating their potential use as interventions to mitigate Salmonella contamination in comminuted turkeys. However, the beneficial effects of these phytogenic compounds on the flavour and appearance of the raw turkey patties were not as great as the antimicrobial activity against S. Heidelberg.
The application of oregano products as a feed additive on the performance, health and quality of turkey meat was outlined in a very recent review by Bozakova and Ivanov (2022). The efficiency of ground oregano and its main active compounds in the prevention and treatment of protozoal diseases in turkeys was suggested by the authors.
According to the available results from a limited number of studies, it can be stated that phytogenic compounds have the potential to be considered as feed additives to promote growth, health and meat quality in turkeys, and render nutrients more available for digestion as well. However, the underlying mechanism and reasons for these improvements still require clarification.
7 Mycotoxin-Counteracting Strategies
The contamination of feed with mycotoxins is a problem of critical importance in continuing feed safety issues leading to economic losses in animal production (Wu 2007). Mycotoxins are secondary metabolites produced by toxigenic fungal species. The mycotoxins produced (mostly trichothecenes, zearalenone, deoxynivalenol, aflatoxins and fumonisins) can cause deleterious effects on animal and consequently human health after oral intake (Murugesan et al. 2015; Haquea et al. 2020). Mycotoxin-detoxifying agents supplemented as feed additives are still the most promising and are therefore most commonly used in turkey feed, as is the case in other poultry species (Kolosova and Stroka 2011; Murugesan et al. 2015). These detoxifiers can be classified as mycotoxin binders and mycotoxin modifiers (EFSA 2009). Mycotoxin binders can suppress or reduce the absorption of mycotoxins in the gut, resulting in the excretion of toxin-binder complexes in faeces, whereas mycotoxin modifiers modify their mode of action and transform the toxin into nontoxic metabolites (EFSA 2010; Devreese et al. 2013, 2014). Of note, evidence from previous studies has demonstrated that poults are rather tolerant to selected mycotoxins because infected birds show comparable performance traits and biochemical, histological and immunological measurements, which are regarded as indicators of mycotoxicosis, compared to uninfected healthy birds (Giambrone et al. 1985; Olsen et al. 1986; Morris et al. 1999; Quist et al. 2000; Rauber et al. 2007; Girish et al. 2008; Grimes et al. 2010; Xu et al. 2011).
Although a range of research studies show promising results for toxin binders and toxin modifier agents in broilers, layer hens and ducks (Murugesan et al. 2015), there is a dearth of research on turkey toms. The efficacy of bentonite clay, proven to be a mycotoxin-detoxifying agent, was tested in an in vitro model using a drug interaction model (Devreese et al. 2013). In the study, based on field observations and suggestions made by other authors that mycotoxin-detoxifying agents decrease or enhance the oral absorption of drugs, the interaction between the macrolide antibiotic tylosin and a bentonite toxin binder was investigated. The results indicated that bentonite significantly reduced tylosin passage and thus bound tylosin. Therefore, the authors suggested that the combined use of bentonite in the feed with tylosin in the feed or drinking water should be avoided, as this could lead to therapy failure and eventually enhance antibiotic resistance towards tylosin due to subtherapeutic plasma concentrations. On the other hand, the modified glucomannan binder did not alter the passage of tylosin significantly, indicating safe combined use.
In their follow-up study, Devreese et al. (2014) evaluated the effect of a commercial yeast-derived glucomannan mycotoxin binder agent (Mycosorb®, Alltech Inc.) on selected nonspecific parameters and specific toxicokinetic markers after feeding hybrid turkeys a diet naturally contaminated with Fusarium mycotoxins, mainly deoxynivalenol. The mycotoxin-detoxifying agent, Mycosorb, was supplemented at 2 kg/tonne in each rearing phase diet, including starter (0–3 weeks), grower (4–6 weeks), developer (7–9 weeks) and finisher (10–12 weeks). Except for the starter phase, no significant differences in BW, BWG, feed intake, or FCR were observed. The feeding of contaminated diets reduced the duodenal villus height and apparent villus surface area. This mycotoxin-induced negative effect was prevented by glucomannan supplementation. However, the feeding of contaminated diets elevated the total duodenal CD8+ T-lymphocyte counts, but this effect was not ameliorated by Mycosorb. Thus, the provision of a diet with glucomannan was able to counteract the negative effects of Fusarium mycotoxins on duodenal morphometry but was ineffective to counter the influx of specific toxicokinetic markers and decrease deoxynivalenol absorption after naturally occurring oral toxin intake for a 3-week period. Nonetheless, in terms of the growth performance parameters, the lack of response to mycotoxicosis or mycotoxin-detoxifier agents used is noticeable.
In a more recent study, the effectiveness of in-feed mycotoxin-binder agents in turkey poult diets containing low to moderate levels of mycotoxins (aflatoxin, deoxynivalenol and zearalenone) was determined by Tilley et al. (2017). Corn, wheat and barley with the naturally occurring mycotoxins mentioned above were used to make rations for feeding turkey hen poults to 6 weeks of age. The commercial mycotoxin-detoxifier agents used in this study were Biomin BioFix (2 lb/tonne), Kemin Kallsil (4 lb/tonne) and Nutriad UNIKE (3 lb/tonne). The feed additives lessened the detrimental effect of mycotoxicosis on nutrient utilization, which manifested as improvements in FCR. The physiological effect of feeding the additives was observed as reduced relative gizzard weight for both groups and a smaller increase in the relative kidney weight for the birds fed the mycotoxin feed. The authors considered that the feed additives used in the study alleviated the harmful effect of dietary mycotoxins to some degree.
Review of the above studies shows that feed additives with mycotoxin-binding and mycotoxin-modifying activities could detoxify some mycotoxins or provide a direct benefit to poults. Considering that turkey poults are rather tolerant to different mycotoxins, it was tentatively concluded by almost all the researchers that markedly higher naturally occurring or artificially infected levels of mycotoxins, compared to those applied in the turkey trials, are warranted to establish the actual potential of mycotoxin-detoxifier agents used.
8 Conclusion
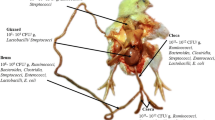
The ban on nontherapeutic antibiotics adds to the challenge of maintaining flock health, productive output and welfare in modern turkey production. Scrutiny of results from available studies indicated that, in the absence of AGPs, using various alternative feed additives with antimicrobial activity could open up options to improve nutrient utilization, absorption, metabolism and modulation of host immunity, which in turn affect the gut health of turkeys. In this sense, prebiotics, probiotics, phytogenics, organic acids, yeast-based products and enzymes appear to be regarded as practical and sustainable tools to be utilized. However, such improvements in the function and health status of the digestive system barely coincided with improved production efficiency. In general, either no differences or somewhat beneficial trends can be found in the literature on the influence of feed additives on growth performance traits, including BW, BWG, FCR and mortality rate. The same is the case for digestive organ weight and length, meat yield and processed parts with economic importance. The mechanisms of the improvement of gut health and bird resilience due to these additives may also include improvement of intestinal health and utilization of nutrients by maintaining gut integrity and enhancing antioxidant capacity. However, as far as growth performance is concerned, the feed additives evaluated herein mostly resulted in a lack of bird response, probably due to their longer grow-out period (12–20 weeks) than broilers (5–7 weeks), allowing the turkeys more time to encounter pathogens and thus perform under less stressful conditions. In addition, the relative age and bird gender may play a role, although sufficient data are not available to validate this. Taken together, a review of the current studies provides supporting evidence that performance-enhancing feed additives can help modulate the immune system and improve intestinal development in young turkeys without compromising growth performance during times of disease challenge. The figure also provides an overview of the results from a large body of research and allows the reader to glean more effective conclusions on the usefulness of these performance enhancer feed additives (Fig. 1).
9 Further Directives
Microbiota modulation by using feed additives, particularly exogenous carbohydrase enzymes and protease, is of particular importance when feeding poults on starter diets when the young birds have an immature microbiota. Thus, controlling the antinutritional effects of NSP and detrimental effects of fermentative organisms proliferating in the small intestine due to dietary supplementation of soybean meal (almost 40%) could have significantly positive impacts in antibiotic-free and no-antibiotic-ever production systems.
On the other hand, late feeding programs with low protein levels using alternative protein sources to soybean, such as sunflower meal, rapeseed cake, cotton seed cake and DDGS, may gain relevance in physiology, microbiology, metabolic function and nutrient utilization, mainly because of their high fibre content, poor nutrient digestibility, unbalanced amino acid profile and anti-nutritional factors that limit their use in rations. Certain benefits of exogenous enzymes in overcoming these nutritional issues and their contributions to economic, environmental and sustainable agriculture practices remain to be elucidated for this late growth phase, where almost half of the cumulative feed is consumed.
While these issues are relatively straightforward, the control of bacterial and protozoan diseases, including Histomonas meleagridis, Salmonella, Campylobacter, Listeria monocytogenes and coccidiosis, remains challenging in turkey production. Due to the ban on AGPs, which have been successively used to control these diseases over decades, turkey growers now face the challenge of maintaining productivity and preventing diseases on their farms. Thus, whether these additive alternatives to AGPs are likely to be highly influential in coping with these enteropathogens merits further investigation through further studies.
References
Adeola O, Cowieson AJ (2011) Board-invited review: opportunities and challenges in using exogenous enzymes to improve nonruminant animal production. J Anim Sci 89:3189–3218
Adewole DI, Oladokun S, Santin E (2021) Effect of organic acids–essential oils blend and oat fiber combination on broiler chicken growth performance, blood parameters, and intestinal health. Anim Nutr 7:1039–1051
Agboola AF, Aroniyo I, Suberu S et al (2014) Dietary supplementation of probiotics and synbiotics on intestinal microbial populations and gut morphology of Turkey poults. Afr J Livest Ext 14:13–20
Al-Khalaifah HS (2018) Benefits of probiotics and/or prebiotics for antibiotic-reduced poultry. Poult Sci 97:3807–3815. https://doi.org/10.3382/ps/pey160
Al-Khalaifah H, Al-Nasser A, Al-Surayee T et al (2019) Effect of dietary probiotics and prebiotics on the performance of broiler chickens. Poult Sci 98:4465–4479. https://doi.org/10.3382/ps/pez282
Antunes P, Mourao J, Campos J et al (2016) Salmonellosis: the role of poultry meat. Clin Microb Infect 22:110–121
Applegate TJ, Webel DM, Lei XG (2003) Efficacy of a phytase derived from Escherichia coli and expressed in yeast on phosphorus utilization and bone mineralization in Turkey poults. Poult Sci 82:1726–1732
Ayoola AA, Malheiros RD, Grimes JL et al (2015) Effect of dietary exogenous enzyme supplementation on enteric mucosal morphological development and adherent mucin thickness in turkeys. Front Vet Sci 2:1–8
Barasch IB (2012) The effects of yeast-derived feed additives and their potential as antibiotic alternative feed additives on the growth performance of Turkey toms raised to market age. Master Thesis. Graduate Faculty of North Carolina State University (USA)
Barbour GW, Werling M, Yersin AG et al (2002) The effect of enzyme predigestion on the nutritional quality of prepressed Turkey feather meal. Poult Sci 81:1032–1037
Basmacioğlu-Malayoğlu H, Özdemir P, Bağriyanik HA (2016) Influence of an organic acid blend and essential oil blend, individually or in combination, on growth performance, carcass parameters, apparent digestibility, intestinal microflora and intestinal morphology of broilers. Br Poult Sci 57:227–234. https://doi.org/10.1080/00071668.2016.1141171
Batkowska J, Brodacki A, Tomczyk G (2015) The influence of probiotic bacteria (Bacillus toyoi) on livability and performance of young meat-type turkeys. Braz J Poult Sci 17(4):433–438
Bearson BL, Bearson SMD, Looft T et al (2017) Characterization of a multidrug-resistant salmonella enterica Serovar Heidelberg outbreak strain in commercial turkeys: colonization, transmission, and host transcriptional response. Front Vet Sci 4:1–7
Bedford MR, Schulze H (1998) Exogenous enzymes for pigs and poultry. Nutr Res Rev 11:91–114
Botsoglou NA, Grigoropoulou SH, Botsoglou E et al (2003) The effects of dietary oregano essential oil and α-tocopheryl acetate on lipid oxidation in raw and cooked Turkey during refrigerated storage. Meat Sci 65:1193–1200
Bozakova N, Ivanov V (2022) Effects of oregano (Origanum vulgare) as a dietary supplement in Turkey farming – review. J Hyg Engin Des 38:219–224
Bozkurt M, Tüzün AE (2020) Application of aromatic plants and their extracts in diets of turkeys. In: Florou-Paneri P, Christaki E, Giannenas I (eds) Feed additives: aromatic plants and herbs in animal nutrition and health. Academic Press (an imprint of Elsevier), London, pp 205–220
Bozkurt M, Küçükyilmaz K, Çatli AU et al (2012) Effects of administering an essential oil mixture and an organic acid blend separately and combined to diets on broiler performance. Arch Geflügelk 76:81–87
Bozkurt M, Hippenstiel F, Abdel-Wareth AAA et al (2014) Effects of selected herbs and essential oils on performance, egg quality and some metabolic activities in laying hens-a review. Eur Poult Sci 78:1–15
Bradley GL, Savage TF (1995) The effect of autoclaving a yeast culture of Saccharomyces cerevisiae on Turkey poult performance and the retention of gross energy, and selected minerals. Anim Feed Sci Technol 55:1–7
Brodacki A, Batkowska J, Makarski B (2009) The effect of feeding with feedstuff containing microbial and plant phytase additive on performance of young slaughter turkeys. Acta Agricul Scand Sec A 59:137–141
CDC (2019) Outbreak of multidrug-resistant Salmonella infections linked to raw turkey products. 2018 Outbreaks. https://www.cdc.gov/salmonella/reading-07-18/index.html. Accessed July 2019
Çelik K, Erbil EI, Uzatici A et al (2003) The using of organic acids in California Turkey chicks and its effects on performance before pasturing. Int J Poult Sci 2:446–448
Chiang W, Booren A, Strasburg G (2008) The effect of heat stress on thyroid hormone response and meat quality in turkeys of two genetic lines. Meat Sci 80:615–622
Cole K, Farnell MB, Donoghue AM et al (2006) Bacteriocins reduce campylobacter colonization and alter gut morphology in Turkey poults. Poult Sci 85:1570–1575
Corrier DE, Hinton A Jr, Kubena LF et al (1991) Decreased salmonella colonization in Turkey poults inoculated with anaerobic cecal microflora and provided dietary lactose. Poult Sci 70:1345–1350
Cowieson AJ, Wilcock P, Bedford MR (2011) Superdosing effects of phytase in poultry and other monogastrics. World Poult Sci J 67:225–235
Czech A, Merska-Kazanowska M, Całyniuk Z (2020) Redox status, biochemical parameters and mineral elements content in blood of Turkey hens fed a diet supplemented with Yarrowia lipolytica yeast and two Bacillus species. Animals 10:459. https://doi.org/10.3390/ani10030459
Denli M, Blandon JC, Guynot ME et al (2009) Effects of dietary AflaDetox on performance, serum biochemistry, histopathological changes and AF residues in broilers exposed to AFB1. Poult Sci 88:1444–1451
Devreese M, Pasmans F, De Backer P et al (2013) An in vitro model using the IPEC-J2 cell line for efficacy and drug interaction testing of mycotoxin detoxifying agents. Toxicol Vitro 27:157–163
Devreese M, Girgis GN, Si-T T et al (2014) The effects of feed-borne fusarium mycotoxins and glucomannan in Turkey poults based on specific and non-specific parameters. Food Chem Toxicol 63:69–75
Dewi G, Nair DVT, Peichel C et al (2021) Effect of lemongrass essential oil against multidrug-resistant salmonella Heidelberg and its attachment to chicken skin and meat. Poult Sci 100:101116
Diaz-Sanchez S, D’Souza D, Biswas D et al (2015) Botanical alternatives to antibiotics for use in organic poultry production. Poult Sci 94:1419–1430
Dobrowolski P, Tomaszewska E, Klebaniuk R et al (2019) Structural changes in the small intestine of female turkeys receiving a probiotic preparation are dose and region dependent. Animal 13(12):2773–2781
Dutta V, Huff GR, Huff WE et al (2008) The effects of stress on respiratory disease and transient colonization of turkeys with Listeria monocytogenes Scott A. Avian Dis 52:581–589
EFSA (2009) Review of mycotoxin-detoxifying agents used as feed additives: mode of action, efficacy and feed/food safety. EFSA Support Publ 6(9):22E. https://doi.org/10.2903/sp.efsa.2009.EN-22
EFSA (2010) Statement on the establishment of guidelines for the assessment of additives from the functional group ‘substances for reduction of the contamination of feed by mycotoxins’. EFSA J 8(7):1693. https://doi.org/10.2903/j.efsa.2010.1693
Erasmus MA (2018) Welfare issues in Turkey production. Adv Poult Welf Pages 3:263–291. https://doi.org/10.1016/B978-0-08-100915-4.00013-0
Fairchild AS, Grimes JL, Edens FW et al (1999) Effect of hen age, Bio-Mosand Flavomycin on susceptibility of Turkey poults to oral Escherichia coli challenge. In: Lyons TP, Jacques KA (eds) Proceedings of Alltech’s 15th annual symposium, under the microscope: focal points for the new millenium. Biotechnology in the feed industry. Nottingham University Press, Nottingham, UK, pp 185–201
Fairchild AS, Grimes JL, Edens FW et al (2001) Effects of hen age, Bio-Mos, and Flavomycin on poult susceptibility to oral Escherichia coli challenge. Poult Sci 80:562–571
Ferket PR (1993) Practical use of feed enzymes for turkeys and broilers. J Appl Poult Res 2:75–81
Ferket PR (2013) Novel nutritional applications to optimize feed efficiency in turkeys. In: Paper presented at the 7th Turkey science and production conference. Proceedings, pp 1–11
Firman JD, Kirn BN (1989) Research note: effects of monensin and bambermycins on the performance of market turkeys. Poult Sci 68:1724–1726
Firman JD, Moore D, Broomhead J et al (2013) Effects of dietary inclusion of a Saccharomyces cerevisiae fermentation product on performance and gut characteristics of male turkeys to market weight. Int J Poult Sci 12:141–143. https://doi.org/10.3923/ijps.2013.141.143
Flores Maldonado KR (2018) Effect of feed quality, feed additives and litter amendments in a Turkey antibiotic free program on performance and health. Dissertation, University of North Carolina State
Flores KR, Fahrenholz A, Ferket PR et al (2021) Effect of methionine chelated Zn and Mn and corn particle size on large white male Turkey live performance and carcass yield. Poult Sci 100:101444
Forkus B, Ritter S, Vlysidis M et al (2017) Antimicrobial probiotics reduce Salmonella enterica in Turkey gastrointestinal tracts. Sci Rep 7:40695
Francis C, Janky DM, Arafa AS et al (1978) Interrelationship of lactobacillus and zinc bacitracin in the diets of Turkey poults. Poult Sci 57:1687–1698
Fritts CA, Waldroup PW (2003) Evaluation of bio-Mos mannan oligosaccharide as a replacement for growth promoting antibiotics in diets for turkeys. Int J Poult Sci 2:19–22
García-López JC, Pinos-Rodríguez JM, García-Galicia IA et al (2011) Effects of enzyme and feeding system on Turkey performance. Arch Zootec 60:297–300
Gheisar MM, Hosseindoust A, Kim IH (2015) Evaluating the effect of microencapsulated blends of organic acids and essential oils in broiler chickens diet. J Appl Poult Res 24:511–519
Giambrone JJ, Diener UL, Davis ND et al (1985) Effects of aflatoxin on young turkeys and broiler chickens. Poult Sci 64:1678–1684
Giannenas I, Sidiropoulou E, Bonos E et al (2020) The history of herbs, medicinal and aromatic plants, and their extracts: past, current situation and future perspectives. In: Florou-Paneri P, Christaki E, Giannenas I (eds) Feed additives: aromatic plants and herbs in animal nutrition and health. Academic Press (an imprint of Elsevier), London, pp 1–15
Girish CK, Smith TK, Boermans HJ et al (2008) Effects of feeding blends of grains naturally contaminated with fusarium mycotoxins on performance, hematology, metabolism, and immunocompetence of turkeys. Poult Sci 87:421–432
Gous RM, Fisher C, Tůmová E et al (2022) Dietary energy: protein ratio influences the efficiency of utilization of dietary protein by growing turkeys. Br Poult Sci. https://doi.org/10.1080/00071668.2022.211669
Grashorn MA, Bessei W (2004) Comparison of heavy Turkey breeds BUT Big 6 and hybrid Euro FP for fattening performance, slaughter yield and meat quality. Arch Geflügelk 68:2–7
Grela ER, Brodacki A, Batkowska J et al (2009) Influence of a probiotic of bacillus toyoi strain on performance of growing Turkey poults. Arch Geflügelk 73(3):160–166
Grimes JL, Koci MD, Stark CR et al (2010) Biological effect of naturally occurring mycotoxins fed to poults reared to 21 days of age. Int J Poult Sci 9:871–874
Günal M, Yayli G, Kaya O et al (2006) The effects of antibiotic growth promoter, probiotic or organic acid supplementation on performance, intestinal microflora and tissue of broilers. Int J Poult Sci 5:149–155. https://doi.org/10.3923/ijps.2006.149.155
Hafez MH, Awad AS (2021) Turkey production and health: current challenges. German J Vet Res 1:3–14
Hafez HM, Hauck R (2006) Efficacy of a herbal product against Histomonas meleagridis after experimental infection of Turkey poults. Arch Anim Nutr 60:436–442
Haquea A, Wanga Y, Shenc Z et al (2020) Mycotoxin contamination and control strategy in human, domestic animal and poultry: a review. Microb Pathog 142:104095. https://doi.org/10.1016/j.micpath.2020.104095
Harrington D, Hall H, Wilde D (2020) Application of aromatic plants and their extracts in the diets of laying hens. In: Florou-Paneri P, Christaki E, Giannenas I (eds) Feed additives: aromatic plants and herbs in animal nutrition and health. Academic Press (an imprint of Elsevier), London, pp 187–199
Hayat J, Savage TF, Mirosh LW (1993) The reproductive performance of two genetically distinct lines of medium white Turkey hens when fed breeder diets with and without a yeast culture containing Saccharomyces cerevisiae. Anim Feed Sci Technol 43:291–301
Hernández F, García V, Madrid J et al (2006) Effect of formic acid on performance, digestibility, intestinal histomorphology and plasma metabolite levels of broiler chickens. Br Poult Sci 47:50–56
Hocking PM (2014) Unexpected consequences of genetic selection in broilers and turkeys: problems and solution. Br Poult Sci 55:1–10
Hollister AG, Corrier DE, Nisbet DJ et al (1994) Comparison of effects of chicken cecal microorganisms maintained in continuous culture and provision of dietary lactose on cecal colonization by Salmonella typhimurium in Turkey poults and broiler chicks. Poult Sci 73:640–647
Hooge DM (2004) Turkey pen trials with dietary mannan oligosaccharide: meta-analysis, 1993-2003. Int J Poult Sci 3(3):179–188
Hooge DM, Connolly A (2011) Meta-analysis summary of broiler chicken trials with dietary Actigen® (2009-2011). Int J Poult Sci 10:819–824
Huff GR, Huff WE, Rath NC et al (2007) Differential effects of sex and genetics on behaviour and stress response of turkeys. Poult Sci 86:1294–1303
Huff GR, Huff WE, Rath NC et al (2008) Effects of Escherichia coli challenge and transport stress on hematology and serum chemistry values of three genetic lines of turkeys. Poult Sci 87:2234–2241
Huff GR, Dutta V, Huff WE et al (2009) Co-infection of market-age turkeys with Escherichia coli and listeria monocytogenes in two stress models. Avian Dis 53:495–501
Huff GR, Huff WR, Farnell MB et al (2010) Bacterial clearance, heterophil function, and hematological parameters of transport-stressed Turkey poults supplemented with dietary yeast extract. Poult Sci 89:447–456
Huff GR, Dutta V, Huff WE et al (2011) Effects of dietary yeast extract on Turkey stress response and heterophil oxidative burst activity. Br Poult Sci 52:446–455
Huff GR, Huff WE, Rath NC (2014) Effects of vitamin d and yeast extract supplementation on Turkey mortality and clostridial dermatitis incidence in a dexamethasone immunosuppression model. Avian Dis 58:572–578
Immerseel FV, Russell JB, Flythe MD et al (2006) The use of organic acids to combat Salmonella in poultry: a mechanistic explanation of the efficacy. Avian Pathol 35:182–188
Izat AL, Tidwell NM, Thomas RA et al (1990) Effects of a buffered propionic acid in diets on the performance of broiler chickens and on microflora of the intestine and carcass. Poult Sci 69:818–826
Jin L-Z, Dersjant-Li Y, Giannenas I (2020) Application of aromatic plants and their extracts in diets of broiler chickens. In: Florou-Paneri P, Christaki E, Giannenas I (eds) Feed Addıtıves: aromatic plants and herbs in animal nutrition and health. Academic Press (an imprint of Elsevier), London, pp 159–179
Jiraphocakul S, Sullivan TW, Shahani KM (1990) Influence of a dried Bacillus subtilis culture and antibiotics on performance and intestinal microflora in turkeys. Poult Sci 69:1966–1973
Juśkiewicz J, Zduńczyk Z, Jankowski J (2003) Effect of adding mannan-oligosaccharide to the diet on the performance, weight of digestive tract segments, and caecal digesta parameters in young turkeys. J Anim Feed Sci 12:133–142
Juśkiewicz J, Jankowski J, Zduńczyk Z et al (2005) Performance and microbial status of turkeys fed diets containing different levels of inulin. Arch Geflügelk 69(4):175–180
Juśkiewicz J, Zduńczyk Z, Jankowski J (2006) Growth performance and metabolic response of the gastrointestinal tract of turkeys to diets with different levels of mannanoligosaccharide. Worlds Poult Sci J 62:612–625
Juśkiewicz J, Jankowski J, Lecewicz A et al (2010) Effect of diets with different contents of sunflower meal without or with exogenous enzymes supplementation on gastrointestinal tract response of growing turkeys. J Anim Feed Sci 19:468483
Keokamnerd T, Acton JC, Han IY et al (2008) Effect of commercial rosemary oleoresin preparations on ground chicken thigh meat quality packaged in a high-oxygen atmosphere. Poult Sci 87:170–179
Khomayezi R, Adewole D (2021) Probiotics, prebiotics, and synbiotics: an overview of their delivery routes and effects on growth and health of broiler chickens. World Poult Sci J 78(1):57–81. https://doi.org/10.1080/00439339.2022.1988804
Kolosova A, Stroka J (2011) Substances for reduction of the contamination of feed by mycotoxins: a review. World Mycotox J 4:225–256
Konca Y, Kirkpinar F, Mert S (2009) Effects of mannan-oligo saccharides and live yeast in diets on the carcass, cut yields, meat composition and colour of finishing turkeys. Asian Aust J Anim Sci 22:550–556. https://doi.org/10.5713/ajas.2009.80350
Krautwald-Junghannsa ME, Janja S (2022) The influence of stocking density on behaviour, health, and production in commercial fattening turkeys - a review. Br Poult Sci 63:434–444. https://doi.org/10.1080/00071668.2022.2050673
Krogdahl A, Sell JL (1989) Influence of age on lipase, amylase, and protease activities in pancreatic tissue and intestinal contents of young turkeys. Poult Sci 68:1561–1568
Lalev M, Mincheva N, Oblakova M et al (2020) Effects of insect- and probiotic-based diets on turkeys’ production, health, and immune parameters. Bulg J Agric Sci 26(6):1254–1265
Leeson S (1984) Growth and carcass characteristics of chicken and Turkey broilers fed diets containing flavomycin. Can J Anim Sci 64:971–976
Leeson S, Caston L, Yungblut D (1996) Adding roxazyme to wheat diets of chicken and Turkey broilers. J Appl Poult Res 5:167–172
Loeffler SL (2014) Effects of probiotic and prebiotic supplementation in Turkey poults on intestinal morphology and MUC2 gene expression. Dissertation, The Ohio State University
Mahmood T, Guo Y (2020) Dietary fiber and chicken microbiome interaction: where will it lead to? Anim Nutr 1:1–8
Makowski Z, Lipiński K, Mazur-Kuśnirek M (2022) The effects of sodium butyrate, coated sodium butyrate, and butyric acid glycerides on nutrient digestibility, gastrointestinal function, and fecal microbiota in turkeys. Animals 12:1836. https://doi.org/10.3390/ani12141836
Marteau PR, De Vrese M, Cellier CJ (2001) Protection from gastrointestinal diseases with the use of probiotics 1–3. Am J Clin Nutr 73:430S–436S
McKnight F (1997) Use of NSP enzymes in wheat based Turkey diets. Use of Natugrain NSP enzyme in poultry nutrition. In: Paper presented at the BASF technical symposium. BASF, Ludwigshafen, Germany. Proceedings, pp 50–72
Mikulski D, Zduńczyk Z, Jankowski J et al (2008) Effects of organic acids or natural plant extracts added to diets for turkeys on growth performance, gastrointestinal tract metabolism and carcass characteristics. J Anim Feed Sci 17:233–246
Milbradt EL, Okamoto AS, Rodrigues JCZ et al (2014a) Use of organic acids and competitive exclusion product as an alternative to antibiotic as a growth promoter in the raising of commercial turkeys. Poult Sci 93:1855–1861
Milbradt EL, Zamae JR, Araujo Junior JP et al (2014b) Control of salmonella enteritidis in turkeys using organic acids and competitive exclusion product. J Appl Microbiol 117:554–563
Milbradt EL, Okamoto AS, Padovani CR et al (2017) Use of organic acids and a competitive exclusion product as growth promoter and salmonella enteritidis control in commercial turkeys. Braz J Poult Sci 19:551–558
Morris CM, Li YC, Ledoux DR et al (1999) The individual and combined effects of feeding moniliformin, supplied by fusarium fujikuroi culture material, and deoxynivalenol in young Turkey poults. Poult Sci 78:1110–1115
Mountzouris KC, Paraskevas V, Fegeros K (2009) Phytogenic compounds in broiler nutrition. In: Steiner T (ed) Phytogenics in animal nutrition: natural concepts to optimize gut health and performance. Nottingham University Press, Nottingham, pp 97–101
Murugesan GR, Ledoux DR, Naehrer K et al (2015) Prevalence and effects of mycotoxins on poultry health and performance, and recent development in mycotoxin counteracting strategies. Poult Sci 94:1298–1315
Neveling DP, Dicks LMT (2021) Probiotics: an antibiotic replacement strategy for healthy broilers and productive rearing. Prob Antimicrob Proteins 13:1–11
Newman K (1994) Mannan-oligosaccharides: natural polymers with significant impact on the gastrointestinal microflora and the immune system. In: Lyons TP, Jacques KA (eds) Biotechnology in the feed industry. Proceeding of Alltech’s Tenth Annual Symposium. Nottingham University Press, Nottingham, pp 167–175
Nguyen HT, Wu SB, Bedford MR et al (2022) Dietary soluble non-starch polysaccharide level and xylanase influence the gastrointestinal environment and nutrient utilisation in laying hens. Br Poult Sci 3:340–350
Odetallah NH, Parks CW, Ferket PR (2002) Effect of wheat enzyme preparation on the performance characteristics of tom turkeys fed wheat-based rations. Poult Sci 81:987–994
Ofek I, Mirelman D, Sharon N (1977) Adherence of Escherichia coli to human mucosal cells mediated by mannose receptors. Nature (London) 265:623–625
Olsen R (1996) Experience with mannan oligosaccharides in commercial Turkey production. Zootech Int 19:38–39
Olsen M, Mirocha CJ, Abbas HK et al (1986) Metabolism of high concentrations of dietary zearalenone by young male Turkey poults. Poult Sci 65:1905–1910
Omidiwura BRO, Agboola AF, Olaleye OO et al (2018) Influence of prebiotics, probiotics or synbiotics on performance, intestinal mucosal integrity and gut microbiota of Turkey poults. Niger J Anim Sci 20(2):64–75
Oyofo BA, DeLoach JR, Corrier DE et al (1989) Prevention of Salmonella typhimurium colonization of broiler with D-mannose. Poult Sci 68:1357–1360
Paiva DM, Walk CL, Lehman R et al (2010) Turkey response to the inclusion of a Saccharomyces cerevisiae fermentation product, original XPCTM, in antibiotic free diets following a coccidia vaccination. Poult Sci 89(Suppl 1):283–284
Parks CW, Grimes JL, Ferket PR et al (2001) The effect of mannan oligosaccharides, bambermycins, and virginianmycin on performance of large white male market turkeys. Poult Sci 80:718–723
Patten JD, Waldroup PW (1988) The use of organic acid in broiler diets. Poult Sci 67:1182–1187
Quist CF, Bounous DI, Kilburn JV et al (2000) The effect of dietary aflatoxin on wild Turkey poults. J Wild Dis 36:436–444
Rahimi S, Grimes JL, Fletcher O et al (2009) Effect of a direct-fed microbial (Primalac) on structure and ultrastructure of small intestine in Turkey poults. Poult Sci 88:491–503
Rauber RH, Dilkin P, Giacomini LZ et al (2007) Performance of Turkey poults fed different doses of aflatoxin in the diet. Poult Sci 86:1620–1624
Rehman H, Rosenkranz C, Böhm J et al (2007) Dietary inulin affects the morphology but not the sodium-dependent glucose and glutamine transport in the jejunum of broilers. Poult Sci 86:118–122
Rehman H, Vahjen W, Kohl-Parisini A et al (2009) Influence of fermentable carbohydrates on the intestinal bacteria and enteropathogens in broilers. Worlds Poult Sci J 65(1):75–90
Reuben RC, Sarkar SL, Ibnat H et al (2021) Novel multi-strain probiotics reduces Pasteurella multocida induced fowl cholera mortality in broilers. Sci Rep 11:8885. https://doi.org/10.1038/s41598-021-88299-0
Ricke SC (2003) Perspectives on the use of organic acids and short chain fatty acids as antimicrobials. Poult Sci 82:632–639
Ritz CW, Hulet RM, Self BB et al (1995) Endogenous amylase levels and response to supplemental feed enzymes in male turkeys from hatch to eight weeks of age. Poult Sci 74:1317–1322
Rojman K, Deborah A (2022) Probiotics, prebiotics, and synbiotics: an overview of their delivery routes and effects on growth and health of broiler chickens. World Poult Sci J 78(1):57–81
Rosen GD (2007a) Holo-analysis of the efficacy of Bio-Mos® in broiler nutrition. Br Poult Sci 48(1):21–26. https://doi.org/10.1080/00071660601050755
Rosen GD (2007b) Holo-analysis of the efficacy of Bio-Mos® in Turkey nutrition. Br Poult Sci 48(1):27–32. https://doi.org/10.1080/00071660601050730
Roy RD, Edens FW, Parkhurst CR et al (2002) Influence of a propionic acid feed additive on performance of Turkey poults with experimentally induced poult enteritis and mortality syndrome. Poult Sci 81:951–951
Salami SA, Ross SA, Patsiogiannis A et al (2022) Performance and environmental impact of egg production in response to dietary supplementation of mannan oligosaccharide in laying hens: a meta-analysis. Poult Sci 101:101745. https://doi.org/10.1016/j.psj.2022.101745
Santos AA (2002) Improvement of nutritional value of wheat by dietary enzyme supplementation for turkeys. Dissertation, University of North Carolina State
Savage TF, Zakrzewska EI (1996) The performance of male turkeys fed a starter diet containing a manna-oligosaccharide (Bio-Mos) from day old to eight weeks of age. In: Lyons TP, Jacques KA (eds) Biotechnology in The Feed Industry. Proceedings of Alltech’s 12 Annual Symposium. Nottingham University Press, Nottingham, UK, pp 47–54
Savage TF, Zakrzewska EI (1997) The performance of male turkeys fed a starter diet containing a mannan oligosaccharide. Zootech Int 20:30–32
Schoeni JCL, Wong ACL (1994) Inhibition of Campylobacter jejuni colonization in chicks by defined competitive exclusion cultures. Appl Environ Microbiol 60:1191–1197
Scott M (1987) Energy, protein and amino acid requirements of turkeys. In: Nutrition of the Turkey. M.L. Scott of Ithaca, Ithaca, NY, pp 28–82
Sekirov I, Russell S, Antunes L (2010) Gut microbiota in health and disease. Physiol Rev 90:859–904
Selle PH, Ravindran V (2007) Review: microbial phytase in poultry nutrition. Anim Feed Sci Technol 135:1–41
Śliżewska K, Markowiak-Kopeć P, Sip A et al (2020) The effect of using new synbiotics on the Turkey performance, the intestinal microbiota and the fecal enzymes activity in turkeys fed Ochratoxin A contaminated feed. Toxins 12:578. https://doi.org/10.3390/toxins12090578
Slomonski BA (2011) Recent advances in research on enzymes for poultry diets. Poult Sci 90:2013–2023
Soccol CR, de Souza P, Vandenberghe L, Rigon Spier M et al (2013) The potential of probiotics: a review. Food Technol Biotechnol 48:413–434
Spring P (1996) Effects of mannanoligosaccharide on different cecal parameters and on cecalconcentrations of enteric pathogens in poultry. Dissertation, Swiss Federal Institute of Technology, Zurich, Switzerland
Spring P, Wenk C, Dawson KA et al (2000) The effects of dietary mannaoligosaccharides on cecal parameters and the concentrations of enteric bacteria in the ceca of salmonella-challenged broiler chicks. Poult Sci 79:205–211
Stefanello C, Rosa DP, Dalmoro YK et al (2020) Protected blend of organic acids and essential oils improves growth performance, nutrient digestibility, and intestinal health of broiler chickens undergoing an intestinal challenge. Front Vet Sci 6:491
Tilley JEN, Grimes JL, Koci MD et al (2017) Efficacy of feed additives to reduce the effect of naturally occurring mycotoxins fed to Turkey hen poults reared to 6 weeks of age. Poult Sci 96:4236–4244
Tomaszewska E, Kwiecień M, Dobrowolski P et al (2016) Dose-dependent effects of probiotic supplementation on bone characteristics and mineralisation in meat-type female turkeys. Anim Prod Sci 58:507–516
Torres-Rodriguez A, Higgins SE, Vicente JLS et al (2007) Effect of lactose as a prebiotic on Turkey body weight under commercial conditions. J Appl Poult Res 16:635–641
Vahabi-Asil O, Bouyeh M, Qotbi A et al (2017) Effects of a prebiotic on growth performance, blood parameters and immunity response of turkeys fed low protein diets. Eur Poult Sci 81:196. https://doi.org/10.1399/eps.2017.196
Valancony H, Humbert F, Rukelibuga J et al (2001) Comparison of some substitutes for antibiotic additives in diets for Turkey poults. Effects on production and on resistance to salmonella colonization. Sci Tech Avicoles 35:25–34
Waibel PE, Halvorson JC, Noll SL et al (1991) Influence of virginiamycin on growth and efficiency of large white turkeys. Poult Sci 70:837–847
Wajda S, Śmiecińska K, Jankowski J et al (2010) The efficacy of lactic acid bacteria Pediococcus acidilactici, lactose and formic acid as dietary supplements for turkeys. Pol J Vet Sci 13:45–51
Wallace RJ, Oleszek W, Franz C et al (2010) Dietary plant bioactives for poultry health and productivity. Br Poult Sci 51:461–487
Weber GM, Michalczuk M, Huyghebaert G et al (2012) Effects of a blend of essential oil compounds and benzoic acid on performance of broiler chickens as revealed by a meta-analysis of 4 growth trials in various locations. Poult Sci 91:2820–2828
Wu F (2007) Measuring the economic impacts of fusarium toxins in animal feeds. Anim Feed Sci Technol 137:363–374
Xu L, Eicher SD, Applegate TJ (2011) Effects of increasing dietary concentrations of corn naturally contaminated with deoxynivalenol on broiler and Turkey poult performance and response to lipopolysaccharide. Poult Sci 90:2666–2774
Yang X, Xin H, Yang C et al (2018) Impact of essential oils and organic acids on the growth performance, digestive functions and immunity of broiler chickens. Anim Nutr 4:388–393. https://doi.org/10.1016/j.aninu.2018
Yu M, Ismail MM, Qureshi MA et al (2000) Viral agents associated with poult enteritis and mortality syndrome: role of a small round virus and Turkey coronavirus. Avian Dis 44:297–304
Zduńczyk Z, Jankowski J, Juśkiewicz J et al (2004a) Response of young turkeys to dietscontaining flavomycyn, mannan-oligosacharide or inulin. Vet ir Zootech 25:94–96
Zduńczyk Z, Juśkiewicz J, Jankowski J et al (2004b) Performance and caecal adaptation of turkeys to diets without or with antibiotic and with different levels of mannan oligosaccharide. Arch Anim Nutr 58:367–378
Zduńczyk Z, Jankowski J, Juśkiewicz J et al (2013) Effect of different dietary levels of low-glucosinolate rapeseed (canola) meal and non-starch polysaccharide-degrading enzymes on growth performance and gut physiology of growing turkeys. Can J Anim Sci 93:353–362
Zumbaugh CA, Murugesan GR, Wong EA et al (2020) Evaluation of a phytogenic feed additive on performance, nutrient digestion, and absorption in Turkey poults. Anim Feed Sci Technol 267:114575. https://doi.org/10.1016/j.anifeedsci.2020.114575
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Bozkurt, M., Üstündağ, A.Ö., Tüzün, A.E., Çabuk, M. (2023). Application of Feed Additives in the Diets of Turkeys. In: Arsenos, G., Giannenas, I. (eds) Sustainable Use of Feed Additives in Livestock. Springer, Cham. https://doi.org/10.1007/978-3-031-42855-5_20
Download citation
DOI: https://doi.org/10.1007/978-3-031-42855-5_20
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-42854-8
Online ISBN: 978-3-031-42855-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)