Abstract
The rapid progress of nanotechnology in the manufacture of nanomedicinal compounds delivers the potential for enhancing cancer therapy strategies. Nanoparticle-based medicinal products raise the chance of developing multifunctionality and targeted delivery approaches. The use of nanoparticles in cancer therapy using plant resources is based on the efficacy of plant-based nanoparticles in the medical field, particularly in the prevention, diagnosis and treatment of various cancers. Green nanoparticles appear to be a new tool for biomarkers for cancer screening and drug delivery to tumour cells. The use of plants to produce nanoparticles has been investigated extensively. The bonding of diverse potent bioactive residues in medicinal plant-based nanoparticles has been discovered to be the most pharmacologically active material. Phytonanotechnology, now being concentrated, explores the various ways to assemble nanoparticles with potential therapeutics. This approach delivers biocompatibility, scalability and therapeutic efficacy of the synthesised nanoparticles. Therefore, plant-derived nanoparticles are green, non-toxic, and, with the use of the efficient technique, are suitable to meet the high demand in biomedicines. This review opens up possible applications of phytocompound-based nanoparticles and it also highlights cancer treatment.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Nanomedicinal compounds
- Enhancing cancer therapy
- Green nanoparticles
- Phytonanotechnology
- Biocompatibility
1 Use of Plant Resources in Cancer Therapy: Anticancer Phytochemical
Antineoplastic phytochemicals may be commonly extracted from edible plants, including medicinal plants, vegetables and fruits. The abundance and easy availability of plants make it phytochemicals to be used in different studies. In addition, phytochemicals found in these edible sources are usually safe for human consumption, even beneficial for normal body function jobs. This means that they show less or no toxic effects in normal human organs and cells when used in the corresponding physical concentration. Further research on pharmacological properties of the anti-cancer components of nature showed that they usually do not reflect only living organisms such as anti-inflammation, antioxidation and immuno-modulation, but also direct more cancer-related demonstration procedures and methods [1]. Their anti-cancer bioactivities include stimulating apoptosis, anti-growth and anti-metastatic activities, working alone or in combination killing cancer cells. In short, phytochemicals can work in many ways to fight cancer. Once bioactive phytochemicals are identified, it is often used as a structural basis modification or as a leading chemical to synthesise new compounds based on structure–function relationships, physicochemical features and pharmacodynamics for their development along with bioavailability and reduction of their toxins for continuous improvement [2]. A variety of novel phytochemicals have been found to have antineoplastic activity. They are currently in clinical trials or clinical use for the treatment of cancer and show promising clinical practice. Genistein, 5,7-dihydroxy-3-(4-hydroxyphenyl) chromen-4-one, belongs to a group of chemicals called flavones. It is found mainly in leguminous plants, which include soybeans, lupine and fava beans. Studies show that genistein exhibits a number of beneficial biological effects: heart disease, diabetes, neuropathy, osteoporosis, inflammatory diseases and cancer. In the last decade, a few cancer-related clinical trials on genistein, including colorectal, prostate, pancreatic, breast and kidney cancer, have been diagnosed worldwide. Meanwhile, the inhibition of the effects of genistein on different types of cancer and subcutaneous pathways are under investigation. Like phytoestrogen, genistein can competitively bind to oestrogen receptors (ERs) and affects oestrogen-dependent cancer (Fig. 1).
Hwang et al, reported that genistein inhibits the growth of ovarian cancer cells (BG-1) by suppressing both ER and insulin-like growth factor-1 receptor (IGF-1R) signalling methods [3]. Genistein also presses proliferation and secretion of MCF-7 and 3T3-L1 breast cancer cells by reducing regulation of ERα expression [4]. In addition, genistein induces apoptosis by inhibiting the nuclear factor-kappa B (NF-κB) pathway in LoVo and HT-29 human colon cancer cells [5] and T-cell leukaemia cells [6]. In addition, genistein is active ATM/P53-dependent mechanisms in colon cancer cells (HCT-116 and SW-480) [7]. Genistein has been identified as a tyrosine kinase inhibitor and antiangiogenic agent as well.
A DNA topoisomerase II inhibitor, contributes to cancer therapy by its antineoplastic activity. Recently, there are reports of the regulatory effect of genistein on microRNA in cancer cells. Speeches of onco-miR-1260b in prostate cancer cells [8] and kidney cancer cells [9] are reduced by genistein. On the other hand, genistein controls the expression of tumour-suppressors miR-34a and miR-574-3p [10] prostate cancer. Overall, these bioactivities of genistein result in the inhibition of cancer cell growth.
Lycopene is a carotenoid found in red fruits and vegetables, such as red carrot, watermelon, grapefruit, papaya and especially tomatoes. Because of polyunsaturated hydrocarbon chain with 13 double bonds, 11 of them combined, lycopene is very lipophilic and shows strong antioxidant activities. It can prevent oxidative DNA damage and types of active oxygen (ROS) generation. This may have implications because of its chemopreventive activities against cancer [11]. Lycopene has shown therapeutic activity in breast cancer [12], leukaemia [13], ACTH-pituitary adenoma [14] and prostate cancer [15]. Holzapfel et al. report that the effectiveness of lycopene against prostate cancer is due to its ability to detect multiple steps and signal mechanisms in cancer development [16]. In a study by Yang et al. the growth of androgen-independent prostate tumour (PC3 cells) was severely inhibited thereafter by lycopene treatment in the model of mice xenograft. At the same time, lycopene also suppressed the tumour cell proliferation by increasing the expression of increased cell nuclear antigen, reducing angiogenesis by lowering VEGF plasma levels and inhibiting IGF-1 signalling by elevating plasma IGF-binding protein level 3 [17]. An example of an anti-cancer phytochemical currently used clinically is paclitaxel, which is mitotic. The inhibitor was first discovered in the Pacific’s reef, Taxusbrevifolia in 1962, that paclitaxel strengthens cellular microtubules, thereby preventing cell division. For now, it is FDA approved as an anti-cancer drug for many types of cancer, including breast, lung and cervical cancer. At the time, its semi-synthetic analogue, docetaxel, was developed by Pierre Potier. It has been shown to have twice as much power as the parent compound, paclitaxel, because of its effect on the centrosome of the mitotic spindle. It encourages and stabilizes the assembly of microtubules, and prevents its disintegration, thereby causing a significant decrease in free tubulin, preventing the division of mitotic cells and ultimately preventing the proliferation of cancer cells. In the meantime, it is widely used in the treatment of advanced and/or metastatic cancers of the breast, stomach, prostate and lungs. Another mitotic inhibitor, vincristine, is a vinca alkaloid extracted from Catharanthus roseus. It binds mitosis to metaphase by binding to tubulin dimers. It has since been approved by the FDA as an anti- cancer drug in 1963 and is widely used in the treatment of various types of cancer.
The tumour-fighting mechanisms of natural chemical phytochemicals are very complex and require caution. In short, plant compounds can trigger apoptosis of cancer cells, promote cell adhesion, prevent the proliferation of cancer cells and direct the expression of cancer- related cells.
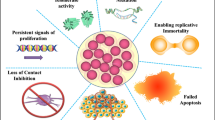
Phytochemicals can also act as suppressors of oncogenes, cancer cell invasion and metastasis inhibitors, as well as growth factor controls. Many natural compounds have been studied in the epidemiology, preclinical and early clinical stages. Although there are many challenges, phytochemicals continue to be the best source of chemotherapy novel medicines (Figs. 2 and 3).
2 Principal Steps in the Process of Nanoparticle Biosynthesis
2.1 Nano-based Drug Delivery
Nanotechnology has been shown to help the success of cancer drug development. Several phytochemical nanoparticles are commercially available. Compared to free phytochemicals, phytochemical nanoparticles show not only an increase in drug solubility but also other benefits.
Changes in pharmacokinetics and distribution of phytochemicals due to the composition of nanoparticles can lead to improving the therapeutic index of these phytochemicals in reducing toxins and increasing efficiency. For example, conjugating phytochemicals as well as antibodies to form antibodies–drug conjugates (ADCs) can produce potent cytotoxic phytochemicals selectively in cancer cells. These conjugates are about the size of a nanometre. Definition of ADCs to the tumour against other organs leads to a wide-ranging cytotoxic treatment window [18].
Improved permeability and retention (EPR) effect are the key to identifying a tumour by nanovehicles. Many hardy plants are associated with a structure that has a unique vascular structure lymphatic drainage dysfunction. EPR effect makes macromolecules and nanocarriers “leaky” especially from the blood vessels around the tumour [19]. In the meantime, lymphatic drainage dysfunction in the abscesses allows them to focus on the area of cancer cells. The effect of EPR is not present in normal muscles. The most important factors that influence the impact of EPR were the size and consistency of biocompatibility. The minimum size of the EPR result is cell size greater than 40 kDa (macromolecules) or particle size greater than 5 nm (nanocarrier) (Fig. 3) [20]. It takes a lot of rotation time to give enough time for delivery of the drug through the EPR effect. Many phytochemicals are low-weight agents with rapid approval in vivo and widespread distribution in normal organs and tissues [21]. MDR is one of the main causes of the failure of phytochemicals in cancer. Using nanovehicles, the delivery of phytochemicals is a new strategy to overcome MDR.
Modification of facial nanocarriers can improve the delivery of phytochemicals and defeat drug resistance by altering biophysical interactions between nanocarriers and cancer cell membrane lipids and increase the delivery of phytochemicals to target tissues [22]. Chemical modification of phytochemicals and recruitment of drug delivery systems are widely used for the delivery of phytochemicals. For chemical repair, we focus on selected drug candidates who have been tested in clinical trials or who have the ability to enter the clinic temptations. With the delivery programs of the phytochemical component, we will focus on the former FDA approved (Fig. 4).
3 Nanoparticles
Nanoparticles are particles at a size of 10–1000 μm, non-embedded drugs or covered at the top [23]. Based on the materials used to make them, they can be divided into biodegradable high molecular weight polymer nanoparticles and natural polymer nanoparticles [24]. The former includes poly (lactic-co-glycolic acid (PLGA) nanoparticles and polylactic acid (PLA) nanoparticles etc. The latter include albumin nanoparticles, gelatine cellulose nanoparticles and chitosan nanoparticles, etc. Abraxane (nanoparticle’s formulations for paclitaxel) is the only FDA-approved nanoparticle drug. Abraxane is made up of human serum albumin. Abraxane nanoparticles are about 130 nm wide and have a loading capacity of about 10% paclitaxel [25].
Paclitaxel loading of nanoparticles is achieved using advanced high-pressure homogenisation technology by American Bioscience. In Abraxane, albumin is illegally bound and retained by paclitaxel [26, 27]. Compared to other commercial nanocarriers, Abraxane has unique advantages over its pharmacokinetic and drug distribution. Abraxane does not depend solely on the effect of EPR for the delivery of paclitaxel in cancer cells. Abraxane binds to gp60, a 6 kDa high-density glycoprotein receptor albumin affinity, and albumin fluid phase entry for tumour endothelium in the subendothelial area. Then, SPARC (secreted protein, acid and rich in cysteine), an extracellular matrix glycoprotein highly exposed to a variety of cancers with a high concentration of gp60, also improves its plant placement (Fig. 4) [28,29,30]. Phase I Abraxane® clinical trials in 19 patients with metastatic breast cancer were performed at M. D. Anderson Cancer Centre, Texas, USA. This study focuses on the toxicity profile, magnitude tolerated dose (MTD), as well as the pharmacokinetics properties of Abraxane. The schedule was administered every 3 weeks using Abraxane dosage pump from 135 to 375 mg/m2. The results showed that Abraxane was well tolerated, producing only neuropathy in three patients, stomatitis in two patients, and external keratopathy in two patients with doses of ABI-007 at 375 mg/m2. They concluded that the Mett of Abraxane was 300 mg/m2. The plasma AUC of Abraxane was higher than that of Taxol if 260 mg/m2 of Abraxane was injected for 30 min i.e., 175 mg/m2 of Taxol injected as 3 h. Abraxane provided an example of albumin nanoparticles that will be used in clinical practice [31, 32]. Currently, Abraxane progresses to Phase II/III clinical trials for metastatic breast cancer patients with i.e., dose at 260 mg/m2 in 3-week cycles compared to the standard paclitaxel system, taken at 175 mg/m2. Compared to Taxol, Abraxane indicated that patients’ response rates were significantly higher (19% vs 33%, respectively) and much longer length of plant growth period (16.9 weeks vs 23.0, respectively) [32, 33].
3.1 Silver Nanoparticles
Plant-based silver nanoparticles have been repeatedly tested against various human cancer cell lines like breast cells, liver cells and colon cells. While compared to other routine physical and chemical approaches in the treatment of this lethal disease, green synthesis of silver nanoparticles by implementing the abilities of various secondary metabolites from plants has been proven to be more efficient, ecofriendly, cost-friendly and safer [34].
For example, using the bark and leaf extracts from the tree Ziziphus xylopyrus which is very common in the Indian sub-continent, biofabrication of silver nanoparticles (AgNPs) was possible. Not only the Zizipus tree but also several other common plants and trees like hibiscus, polyalthia, papaya, Ocimum, Coriandrum and Ficus are employed in the biosynthesis of silver nanoparticles [35].
Green synthesis of nanoparticles over synthetic nanoparticles has paved way to overcome some serious problems like aggregation of nanoparticles, toxicity and non-compatibility, which offers advantages like stability, faster reduction rate and so on.

Many research studies have proved that silver nanoparticles have beneficial toxic mechanisms, which are capable of causing DNA and mitochondrial damage and inducing apoptosis (programmed cell death) in cancer cells. For instance, green synthesis of the plant Datura inoxia, activities like cell cycle arrest, decreased DNA synthesis and apoptosis were recorded in cancer cells [36].
Interestingly enough, not just terrestrial plants but even marine corals were reported to show anti-cancer properties when used to synthesise silver nanoparticles. For example, extracts from the soft coral Cladiella pachyclados were used in producing bioactive silver nanoparticles. This coral possesses a compound called eunicellin, a diterpenoid that has a great potential against breast cancer [37].
This compound, when used to synthesise AgNPs or using silver nanoparticles as drug carriers, showed anti-inflammatory potential almost equal to the commonly used cancer drug Doxorubicin; in fact, it was able to express high selectivity comparatively.
The experiment showed that silver nanoparticles synthesised using the extracts from the mint plant as a stabilising agent exhibited a significant potential against the HCT116 human cancer cell line [36]. This analysis showed that plant-mediated AgNPs were capable of suspending cell division and minimising the further proliferation of cancer cells.
AgNPs have proved themselves very useful in the manufacture of nanodrugs. They use a mechanobiological and microenvironmental mechanism to cause apoptotic cell death in carcinogenic cells with the aid of ROS-mediated pathways. For example the chemotherapeutic drug—fructose coated angstrom silver has been reported to inhibit osteosarcoma cells (a malignant bone cancer), pancreatic and lung cancer cells. Further experiments showed the drug to be capable of inhibiting tumour growth, improving survival rate and attenuating osteolysis in osteosarcoma-bearing mice [38].
Therefore, green-synthesised silver nanoparticles are being extensively studied by researchers worldwide to be implemented in the cure of cancer effective immediately, which could be a significant breakthrough, given its properties and ecofriendly nature.
3.2 Nanodiamonds
Nanodiamonds are carbon nanoparticles with truncated octahedral architecture that are about 2–8 nm in diameter and can deliver a wide range of therapeutics, including small molecules, proteins and nucleic acids, making them very adaptable in the field of anticancer research [39].
Superior properties of nanodiamonds like hardness, high thermal conductivity, chemical stability and resistance to harsh environments have made them very compatible with several biotechnological and medical applications [40]. While compared to other nanocarbons that are synthesised from carbon blacks or fullerenes, nanodiamonds were confirmed to express comparatively less toxicity [41]. Thus, they are widely used in delivering drugs due to their biocompatible properties.
For instance, a popular drug widely used in cancer therapy is Doxorubicin [42]. It is a small-molecule drug that aids in cell death or apoptosis. This drug slows down or stops the growth of cancer cells by blocking an enzyme called topoisomerase 2. Without this enzyme, cancer cells cannot grow or divide. While working on the 4T1 breast cancer cells in mice, the nanodiamond mediated doxorubicin delivery system was well able to cease the generation of lung metastasis [43]. Thus, with the help of nanodiamonds, the drug was able to be successfully delivered in several lab trials which the researchers have speculated using Nano diamonds as intercellular carriers of plant metabolites, like the plant metabolites quercetin and citropten are intensely being studied for their pro-apoptotic and redox properties [42].
Quercetin is a natural pigment (flavonoid) that is available in most common fruits, grains and vegetables and is nowadays taken as a dietary supplement. Due to its high level of free radicals, the flavonoid promotes inflammation in human cells which is a very important anti- cancer activity.
Citropten or 5,7-dimethoxycoumarin is a metabolite (coumarin) extracted from a plant called Peleaanisata and several natural oils and citrus fruits which is known for inducing melanogenesis [44].
Extensive studies have been carried out to assess whether these plant metabolites were able to show improved antiproliferative and inflammatory effects when adsorbed on nanodiamonds.
It was speculated that the bioactivities of these plant metabolites were modifiable through their interaction with nanodiamonds.
When tests were performed on the HeLa cell line, it was evident that the conjugates were able to first penetrate the cell compartment, next, the intracellular spaces, following the areas near the nuclear region successfully.
On the whole, based on the experiments performed on the HeLa and B16F10 cell lines, it was clear that the nanodiamonds heightened the cell growth inhibitory properties of the two plant metabolites quercetin and citropten, making it an effective agent against cancer [39].
3.2.1 In Silico Studies of Plant Nanoparticles in Cancer Therapy
In silico studies are a very crucial step in the process of drug design. These are computer-aided analyses that are carried out in order to know several properties like the binding affinity which are very important in order to design an approvable drug.
4 Liposomes
Liposomes, with a diameter of 20–1200 nm, contain a water-repellent inner layer as well external phospholipids. Hydrophilic drugs can be injected into the aqueous core while hydrophobic drugs can be embedded or advertised externally with low phospholipids. Liposomes can improve in vivo biocompatibility as well as the pharmacokinetics of drugs and their properties can be improved by simply changing the composition of bilayer parts [45].
Marqibo (liposomal formulations of vincristine, FDA approved) and Lipusu (liposomal formulations for PTX, approved by China Food and Drug Administration) only 2 liposomal phytochemical products entered the medical field applications. Marqibo is made of a combination of sphingomyelin: cholesterol in the 57.4: 42.6 mol average [46].
Liposomes are approximately 100 nm in size and drug loading is done with a remote-control loading. In preclinical studies, following an injection of 2.0 mg/m2 in mice, Marqibo showed higher AUC (63,438 vs 806 ng.h/ml) and lower permit (32 vs 2488 mL/h/m2) and volume (383 vs 113,513 mL/m2) for distribution compared to Vincristine. Similar pharmacokinetic profiles have been demonstrated in mice and dogs. These data show that Marqibo has half times the long cycle of system rotation throughout three types. Of the 11 different cancer indications studied, Marqibo was found to be present as more effective than vincristine at 8 [47,48,49].
In clinical studies, Marqibo was used for leukaemia. There are 65 patients in the Phase II case. Marqibo dose of 2.25 mg/m2 once a week is associated with 63% degree of neuropathy, 20% paraesthesia and no third-degree peripheral motor events neuropathy. In contrast, vincristine injected into 1.4 mg/m2 every 21-day injection is the same associated with 92% neuropathy, 78% paraesthesia and 16% grade 3 motor weakness. Marqibo has proven the highest accessible volume for each individual and collection of vincristine compared to other vincristine products [50]. Lipusu is designed to eliminate the need for complex material pre-treatment and side effects associated with Taxol car. This construction is repaired with egg PC and cholesterol [51].
A study of preclinical acute poisonity was made available by mice comparing Lipusu and Taxol. The results showed that Lipusu has the potential to more than double LD50 (69.8 vs 33.0 mg/kg). In mice, Lipusu led to 1.5 times higher concentrations within the tumour tissue compared to Taxol 24 h after treatment. In the Phase I study, a complete comparable safety profile has been reported with Lipusu compared to Taxol®, the results show that Lipusu has twice as much MTD (375 mg/m2 vs 175 mg/m2) [52].
5 Micelle
Micelles automatically assemble themselves when the concentration of the surfactant exceeds its sensitivity micelle concentration (CMC). Compared to other nanocarriers, micelles are smaller in size (10–400 nm). Micelles, especially polymeric micelles, are gaining popularity as drug delivery carriers of phytochemicals [53]. Genexol-PM (Paclitaxel formulations, in Korea), polymeric micelle, is a product that has entered the clinic. Genexol-PM is prepared using a solid distribution process installation of paclitaxel in diblock monomethoxy poly (ethylene glycol)-block-poly (D, L-lactide) (mPEG-PDLLA) is a polymer. The micelles are about 24 nm wide and are almost identical 16% w/w paclitaxel loading [54]. In this micelle structure, a hydrophilic block (PDLLA group) forms a micellar corona and the hydrophobic block (PEG group) forms the core. The PDLA team provides hydrophobic interaction with paclitaxel resulting in high drug and spherical stable loading core, the PEG group provides a polymer coating that can prevent the binding of plasma proteins and increase circulation time in plasma. In a pre-clinical study, Genexol-PM showed strong inhibition of tumour growth and higher MTD than-Taxol®. A few of the Genexol-PM clinical trials have been in operation or in place completed, with indications including non- small cell lung cancer, metastatic breast cancer and pancreatic cancer [55]. In the first phase of clinical trials, Genexol-PM showed MTD 2.3 times higher than Taxol® in humans [56]. In additional clinical research, Genexol-PM has shown better performance than Taxol when used alone and as part of a combined treatment. However, it is interesting that Genexol-PM showed a very low T 1/2 (12 h vs 20 h) and AUC 11.58 compared to Taxol®, possibly due to Genexol-PM faster approval and better access to cancer.
6 Cladiella pachyclados
C. pachyclados is a marine soft coral that exhibits several anti-cancer properties when silver nanoparticles were synthesised using its extract eunicellin. King AbdulAziz University in Saudi Arabia conducted several experiments in order to record the anti-cancer activities of the combination mentioned above.
The extracts recovered from the coral lying in the red sea were subjected to chemical profiling with the help of LC-HRESIMS (liquid chromatography-high-resolution electrospray ionisation mass spectrometry). Several other molecular-level analyses of the silver nanoparticles were performed using UV spectroscopy, XRD, FTIR, TEM, and SEM, and an anti-proliferative assay was carried out [37]. By the aid of SwissADME, an online software which is a web tool that allows to evaluate of pharmacokinetics and drug-likeness of molecules [57] was used to analyse the drug-like characteristics and ADME profiles (adsorption, distribution, metabolism and excretion) of gastrointestinal absorption, blood-brain barrier, solubility and bioavailability were studied. These compounds were subjected to inverse docking in order to assign the potential protein targets using the PDB (protein data bank) and target platforms. The divide and conquer strategy was implied in order to run a large number of docking experiments in less time consumption. Binding affinity score was set to −7 kcal/mol to select the best targets. 26 targets were obtained for the human breast cancer cell proteins. From the experiment, it was observed that the coral extract eunicellin delivered via AgNPs showed a significant level of antiproliferative activity and cytotoxicity towards breast cancer cells almost as same as the common cancer drug doxorubicin.
(IC50 = 71.85 ± 3.57 μg/mL) [37].
MCF7 (SI) | MDA – MB – 231 (SI) | MCF10a | |
|---|---|---|---|
CE | 24.32 ± 0.52c (2.95) | 9.55 ± 0.53b (7.52) | 71.85 ± 0.5c |
AgNPs | 5.62 ± 0.26b (7.34) | 1.72 ± 0.14a (24) | 41.29 ± 0.44b |
Doxorubicin | 2.61 ± 0.03a (7.7) | 1.5 ± 0.26a (13.4) | 20.09 ± 0.72a |
Selectivity index (SI) = IC50 of tested material in a normal cell line/IC50 of the material in cancer cell line | |||
IC50 is the concentration required to kill 50% of the cell population | |||
Out of the 26 compounds that were tested with the aid of inverse molecular docking, only six were found to show binding orientations inside the corresponding binding sites. They also were reported to have the highest dG values, proving that they could be used as potential binders for several BC proteins [37].
7 Flavonoids
As we discussed the usage of flavonoids like the quercetin in the treatment of cancer, there are many other such flavonoids which possess beneficial anti-cancer properties. For example, MYRICETIN is one of the plants derived flavonoids which contains a lot of strong anticancer properties. This compound that is obtained from common plants and fruits is also one of the major constituents of several food and beverages [58]. Myricetin is proven to exhibit cytotoxic activity towards a huge number of human cancer cell lines.
Research studies show that the biosynthesis of Myr-Au NPs resulted in spherical-shaped particles of size less than 50 nm [59].
mTOR is the mammalian target of rapamycin which is a kinase molecule that regulates protein synthesis. A theoretical network analysis was carried out by the Kyoto Encyclopedia of Genes and Genomes (KEGG) database with the help of a software called Cytoscape 3.7.1 to understand the role of different genes in the mTOR signalling mechanism. The observation of the said analysis is shown below [59] (Table 1).
Molecular docking of myricetin (Myr) guided in understanding the X-ray crystal structure of human mTOR kinase enzyme obtained from the RCSB-PDB (Research Collaboratory for Structural Bioinformatics-Protein Data Bank).
The GLIDE program (v.11, Schrödinger, LLC, New York, 2016) was utilised in extra precision (XP) docking mode with the Glidescore and E-model scoring functions to perform the molecular docking analysis of myricetin for the mTOR gene.
MTT assay that is a colorimetric assay performed for assessing the cell metabolic activity was done, and ROS generation studies were carried out and the values were compared using SPSS (v. 20.0, SPSS Inc., Chicago, IL, USA).
In this analysis of Myr-AuNPs, extra precision GLIDE docking procedure was done by removing the inhibitor compound with the human mTOR receptor. The docking result of myrecitin was found to be −7.79 kcal mol−1, and its interactions were confirmed with ALA 1708, ARG 1709, LYS 1710, ILE 1711 and ASP 1712 of chain B, thereby proving a good binding activity of Myrecitin with human mTOR kinase. The molecular docking studies in mTOR kinase were understood to show a strong binding affinity with IC50 value = 13 μg mL–1. After staining, the cells treated with myr-AuNPs showed a good proportion of dead cells, thereby showing an anticancer activity [59].
8 Chitosan
Jatropha pelargoniifolia is a very famous medicinal shrub which is common in Africa, Ethiopia and in some regions of the Arabian Peninsula [60]. The plant has several therapeutic activities that make it very unique in research projects.
A recent phytochemical analysis of the plant has shown the presence of certain bioactive compounds like tannins, coumarins and flavonoids which can be exploited against cancer.
Chitosan is one of the important compounds extracted from the plant whose composition is poly [-(1,4)-2-amino-2-deoxy-D-glucopyranose]. Chitosan is popular for being the second most abundant biopolymer next to cellulose. It has several beneficial properties like biocompatibility, mucoadhesiveness and biodegradability.
By combining the constituents of Jatropha with bacterial DNA gyrase B and the kinase domain of human topo isomerase II a, a molecular docking analysis was performed with the aid of the software Autodock 4.2.
From the Chemdraw ultra 12.0 software, the suitable ligand structures which were bound to their respective proteins were downloaded and selected as a positive control. With the aid of a universal force field, the ligand energies were minimised.
Further, with the help of the Lamarck Genetic algorithm model and Solis and Wets local search method, a molecular docking experiment was performed for a total of 2,500,000 energy calculations for each docking run. By using one way ANOVA, a statistical analysis was done with graph pad prism 9.0 software [48].
9 Pro-haloacetate NPs
PDK or pyruvate dehydrogenase kinase is a mitochondrial enzyme which plays a major role in reversing the suppression of mitochondria-dependent apoptosis. If the suppression activity does not take place, the cell death process is disturbed and might later become a lead cause of cancer. In order for cancer to progress, the apoptosis process must be inhibited [49].
Inhibition of the PDK enzyme results in decreased tumour growth and angiogenesis in a variety of cancers with high selectivity. This indicates the importance of reversing the mitochondrial suppression with metabolic modulating drugs like PDK inhibitors. Recent molecular modelling studies show that the haloacetic acids are capable of selective recognition of certain PDK isoforms allowing us to recognise better haloacetate drug candidates. The quantitative docking energy data from the molecular recognition analysis indicated that MCA (mono chloroacetate) was a comparable PDK binder while DBA (di bromo acetate) showed a slightly weaker binding than the DCA [49]. For further process of molecular modelling, the above-mentioned haloacetates were prepared with the help of SKETCH module. The following requirements was set up to carry out the procedure, followed by the results [49].
Gradient convergence | – | 0.05 kcal/mol |
NB cutoff | – | 8.00 |
Dielectric constant | – | 1.00 |
Docking protocol | – | induced fit |
Score function | – | London dG |
Active site | – | L53, Y80, S83, I111, R112, H115, S153, R154, I157, R158, I161 [49] |
DBA | DCA | MCA |
|---|---|---|
− 3.463(KJ mol−1) | − 4.104 (KJ mol−1) | − 4.121 (KJ Mol−1) |
As a result of these analyses, we can interpret that PDK-modulating haloacetic acids in pro-drugs can be used for inhibition of PDK in order to reverse the suppression of before mentioned apoptosis.
10 Plants and Bioactive Component-Based Nanoparticles
With the growing necessity day by day to develop environment and economy-friendly approaches in the cure of the deadly disease cancer, the utilisation of plant nanoparticles has indeed offered a huge helping hand in this deed, given its numerous advantages over several other existing techniques.
Employment of plants in the field of medicine especially in curing diseases is not new in our civilisation. Taking this under consideration, the aid of green synthesis of nanoparticles or simply green nanoparticles has improved not just the methods but, ecofriendly and very achievable methods of treating cancer in multitudes.
Nowadays, synthesising nanoparticles with the aid of plants is a majorly discussed topic due to their unbelievably advantageous properties and safe nature. Using commonly available plants, several nanoparticles have been synthesised successfully. Few examples are given in Tables 2 and 3 [97].
11 Drug Release Nanosystem
Auxemma oncocalyx is a medicinal tree native to northeast Brazil. This tree is known to have several medicinal properties including antisepsis and antitumor activity which makes it very applicable as a part of traditional medicine in parts of Brazil till date [98].
Oncocalyxone A or simply Onco A, a meroterpenoid quinone, is a secondary metabolite extracted from the heartwood of the above-mentioned tree which possesses several pharmacological benefits like anti-platelet, anti-inflammatory, analgesic, antioxidant and more importantly several antitumor properties which help in fighting cancer [99, 100].
Several researches have shown that onco A is cytotoxic to human leukaemia cells and other cancer cell lines during the cell phases G1, G1/S and S. While exposed, the compound is capable of inhibiting DNA synthesis and cell division in the carcinogenic cells [98, 101].

Onco A was reported to show anti-proliferative activities on various cancer cell lines of the lungs, ovarian, rectal and leukemic cancer resulting in intense anti-cancer research worldwide. In spite of its medicinal properties, during clinical trials, unfortunately, the compound began to express certain toxicological side effects on vital organs like the lungs, kidneys and heart [100].
Following this, further researches theorised that the use of nanosystems can reduce those toxic side effects by a great proportion with the help of Fucoidan, a polysaccharide extracted from brown algae and certain marine invertebrates [102].
This compound is used widely in the green synthesis of nanoparticles using AEP method aiding in a few advantageous biological activities like anti-inflammatory, anticoagulant and antitumor activities as well as low toxicity [103].
Fucoidan is also claimed to show additional beneficial properties like improved antitumor activity of certain chemotherapeutic drugs when used in combination and induced cell growth inhibition and commencement of apoptosis in the breast cancer cell lines [104].
Experiments were performed to analyse the release kinetics of OncoA from the fucoidan-coated nanoparticles in vitro using dialysis technique with different solutions in order to stimulate the pH of the gastrointestinal tract. Using HPLC-UV technology, the released Onco A was quantified. Likewise, stability tests, biocompatibility tests, cytotoxicity tests and blood compatibility tests were vigorously carried out in labs, which conveniently gave satisfying results proving fucoidan was successfully able to deliver Onco A.
To conclude, with the implementation of fucoidan-coated nanoparticles possessing OncoA prepared by AEP method, researchers were successfully able to gain minimal toxicity residues and good compatibility with human blood, paving way for further studies [103].
12 Polyphenol Nanoformulations for Cancer Therapy
12.1 Coumarins
Coumarins are polyphenols with appetite-suppressing structures that prevent animals from eating plants. It can also be used in the medicine of lymphedema. It may also cause bleeding, which is their well-known feature. Fabaceae, Lamiaceae, and Rosaceae are some among the natural sources of coumarins. These phytochemicals have shown anticancer properties in several ways. 4-Methyl-7-hydroxycoumarin is a synthetic coumarin made by methylation nanoparticles of umbelliferone (7-hydroxycoumarin). 4-Methyl-7- hydroxy coumarin has shown anticancer effects on cell melanoma A culture by increasing cell apoptosis, DNA fragmentation, caspase-3 and p53 (tumour suppressor factor) and decreased cell function. Farnesiferol C is another coumarin extracted from plant species such as Ferula asafoetida.
Dendrosomal nanoformulation of farnesiferol C is indicated antineoplastic activity by reducing cell proliferation in iAGS gastric cell line. Bax’s speech (an antiapoptotic marker) and Bcl-2 (proapoptotic factor) were changed to increase Bax/Bcl-2 levels due to treatment with dendrosomal farnesiferol C.
12.2 Flavonoids
Flavonoids are a group of sub-polyphenols classes, such as chalcones, flavones, isoflavones, flavanones, flavonols and anthocyanins. Many pharmacological effects flavonoids reported, such as antioxidant, anti-inflammatory, immunomodulatory and antineoplastic activities. Flavonoids can be extracted from high plants. They can be found in yellow, orange, or red and so on is widely available as colourful fruits and vegetables in human food: Apiaceae (parsley), Ericaceae (berries), Rutaceae (orange fruit), Rosaceae (apple) and a famous sweet product of Theobroma cacao, that is, black chocolate. Flavonoids have shown antineoplastic properties work in several subjects.
12.3 Diarylheptanoid (Curcumin)
Curcumin is a major diarylheptanoid polyphenolic structure extracted from turmeric (Curcuma longa) rhizome and has many biological and pharmacological properties such as antioxidant, anti-inflammatory and anticarcinogenic functions. It has been thoroughly researched in the field of cancer treatment drugs.
References
Wang ZB, Jiang H, Xia YG, Yang BY, Kuang HX (2012) α-Glucosidase inhibitory constituents from Acanthopanax senticosus harm leaves. Molecules 17(6):6269–6276
Chin WW, Parmentier J, Widzinski M, Tan EH, Gokhale R (2014) A brief literature and patent review of nanosuspensions to a final drug product. J Pharm Sci 103(10):2980–2999. https://doi.org/10.1002/jps.24098. Epub 2014 Aug 6. PMID: 25099918
Hwang KA, Park MA, Kang NH, Yi BR, Hyun SH, Jeung EB, Choi KC (2013) Anticancer effect of genistein on BG-1 ovarian cancer growth induced by 17 β- estradiol or bisphenol A via the suppression of the crosstalk between estrogen receptor alpha and insulin-like growth factor-1 receptor signaling pathways. Toxicol Appl Pharmacol 272(3):637–646
Choi EJ, Jung JY, Kim GH (2014) Genistein inhibits the proliferation and differentiation of MCF-7 and 3T3-L1 cells via the regulation of ERα expression and induction of apoptosis. Exp Ther Med 8(2):454–458
Luo Y, Wang SX, Zhou ZQ, Wang Z, Zhang YG, Zhang Y, Zhao P (2014) Apoptotic effect of genistein on human colon cancer cells via inhibiting the nuclear factor- kappa B (NF-κB) pathway. Tumour Biol 35(11):11483–11488
Yamasaki M, Mine Y, Nishimura M, Fujita S, Sakakibara Y, Suiko M, Morishita K, Nishiyama K (2013) Genistein induces apoptotic cell death associated with inhibition of the NF-κB pathway in adult T-cell leukaemia cells. Cell Bio Int 37(7):742–747. https://doi.org/10.1002/cbin.10101. Epub 2013 Apr 18. PMID: 23526666
Zhang Z, Wang CZ, Du GJ, Qi LW, Calway T, He TC et al (2013) Genistein induces G2/M cell cycle arrest and apoptosis via ATM/p53-dependent pathway in human colon cancer cells. Int J Oncol 43(1):289–296
Hirata H, Hinoda Y, Shahryari V, Deng G, Tanaka Y, Tabatabai ZL, Dahiya R (2014) Genistein downregulates onco-miR-1260b and upregulates sFRP1 and Smad4 via demethylation and histone modification in prostate cancer cells. Br J Cancer 110(6):1645–1654
Hirata H, Ueno K, Nakajima K, Tabatabai ZL, Hinoda Y, Ishii N, Dahiya R (2013) Genistein downregulates onco-miR-1260b and inhibits Wnt-signalling in renal cancer cells. Br J Cancer 108(10):2070–2078
Chiyomaru T, Yamamura S, Fukuhara S, Hidaka H, Majid S, Saini S et al (2013) Genistein up-regulates tumour suppressor microRNA-574-3p in prostate cancer. PLoS One 8(3):e58929
Trejo-Solís C, Pedraza-Chaverrí J, Torres-Ramos M, Jiménez-Farfán D, Cruz Salgado A, Serrano-García N et al (2013) Multiple molecular and cellular mechanisms of action of lycopene in cancer inhibition. Evid Based Complement Altern Med 2013:1–17, page 45:47. http://dx.doi.org/10.1155/2013/705121
Uppala PT, Dissmore T, Lau BH, Andacht T, Rajaram S (2013) Selective inhibition of cell proliferation by lycopene in MCF-7 breast cancer cells in vitro: a proteomic analysis. Phytother Res 27(4):595–601. https://doi.org/10.1002/ptr.4764. Epub 2012 Jun 21. PMID: 22718574
Gharib A, Faezizadeh Z (2014) In vitro anti-telomerase activity of novel lycopene- loaded nanospheres in the human leukaemia cell line K562. Pharmacogn Mag 10(Suppl 1):S157
Haddad NF, Teodoro AJ, Leite de Oliveira F, Soares N, de Mattos RM, Hecht F et al (2013) Lycopene and beta-carotene induce growth inhibition and proapoptotic effects on ACTH-secreting pituitary adenoma cells. PLoS One 8(5):e62773
Elgass S, Cooper A, Chopra M (2014) Lycopene treatment of prostate cancer cell lines inhibits adhesion and migration properties of the cells. Int J Med Sci 11(9):948
Holzapfel NP, Holzapfel BM, Champ S, Feldthusen J, Clements J, Hutmacher DW (2013) The potential role of lycopene for the prevention and therapy of prostate cancer: from molecular mechanisms to clinical evidence. Int J Mol Sci 14(7):14620–14646. https://doi.org/10.3390/ijms140714620. PMID: 23857058; PMCID: PMC3742263
Yang CM, Yen YT, Huang CS, Hu ML (2011) Growth inhibitory efficacy of lycopene and β-carotene against androgen-independent prostate tumor cells xenografted in nude mice. Mol Nutr Food Res 55(4):606–612
Chari RV, Miller ML, Widdison WC (2014) Antibody–drug conjugates: an emerging concept in cancer therapy. Angew Chem Int Ed 53(15):3796–3827
Maeda H, Nakamura H, Fang J (2013) The EPR effect for macromolecular drug delivery to solid tumours: improvement of tumour uptake, lowering of systemic toxicity, and distinct tumour imaging in vivo. Adv Drug Deliv Rev 65(1):71–79
Fang J, Nakamura H, Maeda H (2011) The EPR effect: unique features of tumour blood vessels for drug delivery, factors involved, and limitations and augmentation of the effect. Adv Drug Deliv Rev 63(3):136–151
Bertrand N, Wu J, Xu X, Kamaly N, Farokhzad OC (2014) Cancer nanotechnology: the impact of passive and active targeting in the era of modern cancer biology. Adv Drug Deliv Rev 66:2–25
Patel NR, Pattni BS, Abouzeid AH, Torchilin VP (2013) Nanopreparations to overcome multidrug resistance in cancer. Adv Drug Deliv Rev 65(13–14):1748–1762
Alexis F, Rhee JW, Richie JP, Radovic-Moreno AF, Langer R, Farokhzad OC (2008) New frontiers in nanotechnology for cancer treatment. In: Urologic oncology: seminars and original investigations, 26(1):74–85, page 56:58. Elsevier, New York, United States of America. Urologic Oncology: Seminars and Original Investigations. https://doi.org/10.1016/j.urolonc.2007.03.017
Liang Y, Li Y, Wang H, Zhou J, Wang J, Regier T, Dai H (2011) Co3O4 nanocrystals on graphene as a synergistic catalyst for oxygen reduction reaction. Nat Mater 10(10):780–786
Stinchcombe TE (2007) Nanoparticle albumin-bound paclitaxel: a novel Cremphor- EL®-free formulation of paclitaxel. Nanomedicine 2(4). https://doi.org/10.2217/17435889.2.4.415
Miele E, Spinelli GP, Miele E, Tomao F, Tomao S (2009) Albumin-bound formulation of paclitaxel (Abraxane® ABI-007) in the treatment of breast cancer. Int J Nanomedicine 4:99
Singh K, Sai Nandhini R, Palanivelu J (2021) Nanosponges: in perspective to therapeutic medicine. In: Nanotechnology in medicine. Springer, Cham, pp 87–104
Kratz F, Elsadek B (2012) Clinical impact of serum proteins on drug delivery. J Control Release 161(2):429–445
Petrelli F, Borgonovo K, Barni S (2010) Targeted delivery for breast cancer therapy: the history of nanoparticle-albumin-bound paclitaxel. Expert Opin Pharmacother 11(8):1413–1432
Reddy LH, Bazile D (2014) Drug delivery design for intravenous route with integrated physicochemistry, pharmacokinetics and pharmacodynamics: illustration with the case of taxane therapeutics. Adv Drug Deliv Rev 71:34–57
Ibrahim NK, Desai N, Legha S, Soon-Shiong P, Theriault RL, Rivera E et al (2002) Phase I and pharmacokinetic study of ABI-007, a Cremophor-free, protein-stabilized, nanoparticle formulation of paclitaxel. Clin Cancer Res 8(5):1038–1044
US Food and Drug Administration (2012) ABRAXANE® for injectable suspension (paclitaxel protein-bound particles for injectable suspension) (albumin-bound)
Roy U, Chakravarty G, Zu Bentrup KH, Mondal D (2009) Montelukast is a potent and durable inhibitor of multidrug resistance protein 2-mediated efflux of taxol and saquinavir. Biol Pharm Bull 32(12):2002–2009
Javed B, Ikram M, Farooq F, Sultana T, Mashwani ZUR, Raja NI (2021) Biogenesis of silver nanoparticles to treat cancer, diabetes, and microbial infections: a mechanistic overview. Appl Microbiol Biotechnol 105(6):2261–2275
Prakash NU, Bhuvaneswari S, Nandhini RS, Azeez NA, Al-Arfaj AA, Munusamy MA (2015) Floral synthesis of silver nanoparticles using Stenolobium stans L. Asian J Chem 27(11):4089
Yesilot S, Aydin C (2019) Silver nanoparticles; a new hope in cancer therapy? East J Med 24(1):111–116
Alhadrami HA, Alkhatabi H, Abduljabbar FH, Abdelmohsen UR, Sayed AM (2021) Anticancer potential of green synthesized silver nanoparticles of the soft coral cladiellapachyclados supported by network pharmacology and in silico analyses. Pharmaceutics 13(11):1846
Hu XK, Rao SS, Tan YJ, Yin H, Luo MJ, Wang ZX et al (2020) Fructose-coated Angstrom silver inhibits osteosarcoma growth and metastasis via promoting ROS-dependent apoptosis through the alteration of glucose metabolism by inhibiting PDK. Theranostics 10(17):7710
Mengesha AE, Youan BC (2013) Nanodiamonds for drug delivery systems. In: Diamond-based materials for biomedical applications. Woodhead Publishing, Elsevier, Sawston, Cambridge, UK, pages 186–205
Mochalin VN, Shenderova O, Ho D, Gogotsi Y (2012) The properties and applications of nanodiamonds. Nat Nanotechnol 7(1):11–23
Horie M, Komaba LK, Kato H, Nakamura A, Yamamoto K, Endoh S et al (2012) Evaluation of cellular influences induced by stable nanodiamond dispersion; the cellular influences of nanodiamond are small. Diam Relat Mater 24:15–24
Gwak R, Lee GJ, Kim H, Lee MK, Rhee CK, Dae-Ro C et al (2015) Efficient doxorubicin delivery using deaggregated and carboxylatednanodiamonds for cancer cell therapy. Nanosci Nanotechnol Lett 7(9):723–728
Xiao J, Duan X, Yin Q, Zhang Z, Yu H, Li Y (2013) Nanodiamonds-mediated doxorubicin nuclear delivery to inhibit lung metastasis of breast cancer. Biomaterials 34(37):9648–9656
National Center for Biotechnology Information (2022) PubChem compound summary for CID 2775, Citropten. Retrieved February 25, 2022 from https://pubchem.ncbi.nlm.nih.gov/compound/Citropten
Hofheinz RD, Gnad-Vogt SU, Beyer U, Hochhaus A (2005) Liposomal encapsulated anti-cancer drugs. Anti-Cancer Drugs 16(7):691–707
Silverman JA, Deitcher SR (2013) Marqibo(vincristine sulfate liposome injection) improves the pharmacokinetics and pharmacodynamics of vincristine. Cancer Chemother Pharmacol 71(3):555–564
Castle MC, Mead JAR (1978) Investigations of the metabolic fate of tritiated vincristine in the rat by high-pressure liquid chromatography. Biochem Pharmacol 27(1):37–44
Krishna R, Webb MS, Onge GS, Mayer LD (2001) Liposomal and nonliposomal drug pharmacokinetics after administration of liposome-encapsulated vincristine and their contribution to drug tissue distribution properties. J Pharmacol Exp Ther 298(3):1206–1212
Webb MS, Harasym TO, Masin D, Bally MB, Mayer LD (1995) Sphingomyelin-cholesterol liposomes significantly enhance the pharmacokinetic and therapeutic properties of vincristine in murine and human tumour models. Br J Cancer 72(4):896–904
Hagemeister F, Rodriguez MA, Deitcher SR, Younes A, Fayad L, Goy A, Cabanillas F (2013) Long term results of a phase 2 study of vincristine sulfate liposome injection (M arqibo) substituted for non-liposomal vincristine in cyclophosphamide, doxorubicin, vincristine, prednisone with or without rituximab for patients with untreated aggressive non-H odgkin lymphomas. Br J Haematol 162(5):631–638
Ye L, He J, Hu Z, Dong Q, Wang H, Fu F, Tian J (2013) Antitumor effect and toxicity of Lipusu in rat ovarian cancer xenografts. Food Chem Toxicol 52:200–206
Xu X, Wang L, Xu HQ, Huang XE, Qian YD, Xiang J (2013) Clinical comparison between paclitaxel liposome (Lipusu) and paclitaxel for treatment of patients with metastatic gastric cancer. Asian Pac J Cancer Prev 14(4):2591–2594
Cabral H, Kataoka K (2014) Progress of drug-loaded polymeric micelles into clinical studies. J Control Release 190:465–476
Kim SC, Kim DW, Shim YH, Bang JS, Oh HS, Kim SW, Seo MH (2001) In vivo evaluation of polymeric micellar paclitaxel formulation: toxicity and efficacy. J Control Release 72(1–3):191–202
Lee KS, Chung HC, Im SA, Park YH, Kim CS, Kim SB et al (2008) Multicenter phase II trial of Genexol-PM, a Cremophor-free, polymeric micelle formulation of paclitaxel, in patients with metastatic breast cancer. Breast Cancer Res Treat 108(2):241–250
Kim TY, Kim DW, Chung JY, Shin SG, Kim SC, Heo DS, Kim NK, Bang YJ (2004) Phase I and pharmacokinetic study of Genexol-PM, a cremophor-free, polymeric micelle-formulated paclitaxel, in patients with advanced malignancies. Clin Cancer Res 10(11):3708–3716. https://doi.org/10.1158/1078-0432.CCR-03-0655. PMID: 15173077
DeLano WL (2002) Pymol: an open-source molecular graphics tool. CCP4 Newsl Protein Crystallogr 40(1):82–92
Semwal DK, Semwal RB, Combrinck S, Viljoen A (2016) Myricetin: a dietary molecule with diverse biological activities. Nutrients 8(2):90
Mohan UP, Sriram B, Panneerselvam T, Devaraj S, Mubarak Ali D, Parasuraman P et al (2020) Utilization of plant-derived Myricetin molecule coupled with ultrasound for the synthesis of gold nanoparticles against breast cancer. Naunyn Schmiedeberg’s Arch Pharmacol 393(10):1963–1976
Fern K (2018) Tropical plants database, Ken Fern. tropical. theferns. info
Gu FX, Karnik R, Wang AZ, Alexis F, Levy-Nissenbaum E (2007) Targeted nanoparticles for cancer treatment. Nano Today 2:14–21
Cho K, Wang X, Nie S, Chen Z, Shin DM (2008) Therapeutic nanoparticles. Clin Cancer Res 14(5):1310–1316
Mishra B, Patel BB, Tiwari S (2009) Colloidal nanocarriers: a review on formulation technology, types and applications toward targeted drug delivery. Nanomedicine: NBM 1:17
Lim KJ, Bisht S, Bar EE, Maitra A, Eberhart CG (2011) A polymeric nanoparticle formulation of curcumin inhibits growth, clonogenicity and stem-like fraction in malignant brain tumors. Cancer Biol Ther 11(5):464–473
Zu Y, Wang D, Zhao X, Jiang R, Zhang Q, Zhao D et al (2011) A novel preparation method for camptothecin (CPT) loaded folic acid conjugated dextran tumor- targeted nanoparticles. Int J Mol Sci 12(7):4237–4249
Pimple S, Manjappa AS, Ukawala M, Murthy RSR (2012) PLGA nanoparticles loaded with etoposide and quercetin dihydrate individually: in vitro cell line study to ensure advantage of combination therapy. Cancer Nanotechnol 3(1):25–36
Siu YS, Li L, Leung MF, Lee KLD, Li P (2012) Polyethylenimine-based amphiphilic core–shell nanoparticles: study of gene delivery and intracellular trafficking. Biointerphases 7(1):16
Tang X, Cai S, Zhang R, Liu P, Chen H, Zheng Y, Sun L (2013) Paclitaxel- loaded nanoparticles of star-shaped cholic acid-core PLA-TPGS copolymer for breast cancer treatment. Nanoscale Res Lett 8(1):1–12
Sundar VD, Dhanaraju MD, Sathyamoorthy N (2014) Fabrication and characterization of etoposide loaded magnetic polymeric microparticles. Int J Drug Deliv 6(1):24
Han FY, Thurecht KJ, Whittaker AK, Smith MT (2016) Bioerodable PLGA- based microparticles for producing sustained-release drug formulations and strategies for improving drug loading. Front Pharmacol 7:185
Yang A, Liu Z, Yan B, Zhou M, Xiong X (2016) Preparation of camptothecin- loaded targeting nanoparticles and their antitumor effects on hepatocellular carcinoma cell line H22. Drug Deliv 23(5):1699–1706
Zhou H, Liu X, Wu F, Zhang J, Wu Z, Yin H, Shi H (2016) Preparation, characterization, and antitumor evaluation of electrospun resveratrol loaded nanofibers. J Nanomaterials 2016
Wong HL, Bendayan R, Rauth AM, Li Y, Wu XY (2007) Chemotherapy with anticancer drugs encapsulated in solid lipid nanoparticles. Adv Drug Deliv Rev 59(6):491–504
Ekambaram P, Sathali AAH, Priyanka K (2012) Solid lipid nanoparticles: a review. Sci Rev Chem Commun 2(1):80–102
Yassin AEB, Albekairy A, Alkatheri A, Sharma RK (2013) Anticancer- loaded solid lipid nanoparticles: high potential advancement in chemotherapy. Digest J Nanomater Biostruct (DJNB) 8(2):905–916
Abd-Allah FI, Dawaba HM, Samy AM, Nutan MT (2014) Application of solvent injection method to develop stable, sustained release solid lipid nanoparticles of curcumin. Int J Dev Res 4:2734–2742
Chadha R, Kapoor VK, Thakur D, Kaur R, Arora P, Jain DVS (2008) Drug carrier systems for anticancer agents: a review. J Sci Ind Res 67:185–197
Narayanan NK, Nargi D, Randolph C, Narayanan BA (2009) Liposome encapsulation of curcumin and resveratrol in combination reduces prostate cancer incidence in PTEN knockout mice. Int J Cancer 125(1):1–8
Ramana LN, Sharma S, Sethuraman S, Ranga U, Krishnan UM (2012) Investigation on the stability of saquinavir loaded liposomes: implication on stealth, release characteristics and cytotoxicity. Int J Pharm 431(1–2):120–129
Venegas B, Zhu W, Haloupek NB, Lee J, Zellhart E, Sugár IP, Kiani MF, Chong PL (2012) Cholesterol superlattice modulates CA4P release from liposomes and CA4P cytotoxicity on mammary cancer cells. Biophys J 102(9):2086–2094. https://doi.org/10.1016/j.bpj.2012.03.063. PMID: 22824272; PMCID: PMC3341537
Shah SM, Goel PN, Jain AS, Pathak PO, Padhye SG, Govindarajan S, Ghosh SS, Chaudhari PR, Gude RP, Gopal V, Nagarsenker MS (2014) Liposomes for targeting hepatocellular carcinoma: use of conjugated arabinogalactan as targeting ligand. Int J Pharm 477(1–2):128–139. https://doi.org/10.1016/j.ijpharm.2014.10.014. Epub 2014 Oct 11. PMID: 25311181
Mehrabi M, Esmaeilpour P, Akbarzadeh A, Saffari Z, Farahnak M, Farhangi A, Chiani M (2016) Efficacy of pegylated liposomal etoposide nanoparticles on breast cancer cell lines. Turk J Med Sci 46(2):567–571
Li R, Wu RA, Zhao L, Hu Z, Guo S, Pan X, Zou H (2011) Folate and iron difunctionalized multiwall carbon nanotubes as dual-targeted drug nanocarrier to cancer cells. Carbon 49(5):1797–1805
Tian Z, Shi Y, Yin M, Shen H, Jia N (2011) Functionalized multiwalled carbon nanotubes-anticancer drug carriers: synthesis, targeting ability and antitumor activity. Nano Biomed Eng 3(3)
Popov VN (2004) Carbon nanotubes: properties and application. Mater Sci Eng R: Rep 43(3):61–102
Nakanishi T, Fukushima S, Okamoto K, Suzuki M, Matsumura Y, Yokoyama M et al (2001) Development of the polymer micelle carrier system for doxorubicin. J Control Release 74(1–3):295–302
Husseini GA, Pitt WG (2008) Micelles and nanoparticles for ultrasonic drug and gene delivery. Adv Drug Deliv Rev 60(10):1137–1152
Maeda H, Bharate GY, Daruwalla J (2009) Polymeric drugs for efficient tumor targeted drug delivery based on EPR effect. Eur J Pharm Biopharm 71(3):409
Mourya VK, Inamdar N, Nawale RB, Kulthe SS (2011) Polymeric micelles: general considerations and their applications. Indian J Pharm Educ Res 45(2):128–138
Wang C, Feng L, Yang X, Wang F, Lu W (2013) Folic acid-conjugated liposomal vincristine for multidrug resistant cancer therapy. Asian J Pharm Sci 8(2):118–127
Kore G, Kolate A, Nej A, Misra A (2014) Polymeric micelle as multifunctional pharmaceutical carriers. J Nanosci Nanotechnol 14(1):288–307. https://doi.org/10.1166/jnn.2014.9021
Naha PC, Davoren M, Lyng FM, Byrne HJ (2010) Reactive oxygen species (ROS) induced cytokine production and cytotoxicity of PAMAM dendrimers in J774A. 1 cells. Toxicol Appl Pharmacol 246(1–2):91–99
Abdel-Rahman MA, Al-Abd AM (2013) Thermoresponsive dendrimers based on oligoethylene glycols: design, synthesis and cytotoxic activity against MCF-7 breast cancer cells. Eur J Med Chem 69:848–854
Baig T, Nayak J, Dwivedi V, Singh A, Srivastava A, Tripathi PK (2015) A review about dendrimers: synthesis, types, characterization and applications. Int J Adv Pharm Biol Chem 4(1):44–59
Malar CG (2015) Dendrosomal capsaicin nanoformulation for the invitro anticancer effect on HEp 2 and MCF-7 cell lines. Int J Appl Bioeng 9(2)
Yang Q, Yang Y, Li L, Sun W, Zhu X, Huang Y (2015) Polymeric nanomedicine for tumor-targeted combination therapy to elicit synergistic genotoxicity against prostate cancer. ACS Appl Mater Interfaces 7(12):6661–6673
Kuppusamy P, Yusoff MM, Maniam GP, Govindan N (2016) Biosynthesis of metallic nanoparticles using plant derivatives and their new avenues in pharmacological applications–an updated report. Saudi Pharma J 24(4):473–484
Melo ISVD, Santos AFD, Lemos TLGD, Goulart MOF, Santana AEG (2015) Oncocalyxone A functions as an anti-glycation agent in vitro. PLoS One 10(6):e0131222
Barreto AC, Santiago VR, Freire RM, Mazzetto SE, Denardin JC, Mele G et al (2013) Magnetic nanosystem for cancer therapy using oncocalyxone a, an antitomour secondary metabolite isolated from a Brazilian plant. Int J Mol Sci 14(9):18269–18283
Cavalcanti IDL, Ximenes RM, Pessoa ODL, Magalhães NSS, de Britto Lira-Nogueira MC (2021) Fucoidan-coated PIBCA nanoparticles containing oncocalyxone A: activity against metastatic breast cancer cells. J Drug Deliv Sci Technol 65:102698
Pessoa C, Vieira FMAC, Lemos TG, Moraes MO, Lima PDL, Rabenhorst SHB et al (2003) Oncocalyxone A from Auxemmaoncocalyx lacks genotoxic activity in phytohemagglutinin-stimulated lymphocytes. Teratog Carcinog Mutagen 23(S1):215–220
Sbardelotto AB (2013) Estudo do mecanismo de citotóxicidade da oncocalixona-A emleucemiapromiolocíticahumana–linhagem HL-60
Lira MCB, Santos-Magalhães NS, Nicolas V, Marsaud V, Silva MPC, Ponchel G, Vauthier C (2011) Cytotoxicity and cellular uptake of newly synthesized fucoidan-coated nanoparticles. Eur J Pharm Biopharm 79(1):162–170
Zhang Z, Teruya K, Yoshida T, Eto H, Shirahata S (2013) Fucoidan extract enhances the anti-cancer activity of chemotherapeutic agents in MDA-MB-231 and MCF-7 breast cancer cells. Mar Drugs 11(1):81–98
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Nandhini, R.S., Kalpana Shree, S., Maity, P., Madhumathi, G.S., Bhar, A., Palanivelu, J. (2023). Use of Plant-Derived Nanoparticles in Cancer Therapy. In: Arunachalam, K., Yang, X., Puthanpura Sasidharan, S. (eds) Bioprospecting of Tropical Medicinal Plants. Springer, Cham. https://doi.org/10.1007/978-3-031-28780-0_59
Download citation
DOI: https://doi.org/10.1007/978-3-031-28780-0_59
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-28779-4
Online ISBN: 978-3-031-28780-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)