Abstract
Defects of the mandible due to tumor resection, trauma, osteonecrosis of the jaw, osteomyelitis, or congenital malformation are challenging in the restoration of form, function, and aesthetics owing to the complex anatomy. The introduction of computer-aided design/computer-aided manufacturing (CAD/CAM) and patient-specific implants (PSIs) has created novel opportunities for mandibular reconstruction. Surgeons use CT scans to virtually plan and visualize the exact resection margins and the desired reconstruction leading to superior surgical procedures and predictable results. With the possibility of intraoperative navigation, resection and reconstruction of the mandible can be verified and corrected instantly. Individualized surgical guides and reconstruction plates might reduce the risk of incorrect positioning. Computer-assisted surgery (CAS) reduced complications and revision rates along with shortened surgical times compared to conventional methods. In this chapter, we present post-traumatic, oncological, and post-inflammatory defects in which mandibular reconstruction using virtual surgical planning in combination with CAD/CAM and PSI has led to superior clinical outcomes. We provide an outlook for future developments with new technologies such as virtual, augmented, and mixed realities finding their way into the operating theater and clinical routine.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction: Mandibular Defects
The loss of continuity caused by trauma, defects following tumor disease, or inflammation (i.e., osteonecrosis or osteomyelitis) significantly impairs functions such as chewing, swallowing, speaking, and breathing. Microsurgery allows restoring mandibular continuity through free bone grafting, microvascular grafts, or reconstruction with allogeneic and alloplastic materials. Technological advancements in computer-assisted surgery (CAS) provide the foundation for sustainable procedures. Depending on the localization as well as the extent of mandibular defects, there are considerable functional and aesthetic deteriorations.
Defects located in the lateral mandible or in the area of the ascending mandibular branch heavily impact the ability of chewing and swallowing. Due to a loss of continuity around the chin, the tongue loses its support. Along with issues of chewing and swallowing, patients are endangered due to possible obstruction of the upper respiratory tract. Furthermore, psychological as well as social consequences frequently occur, which might negatively influence social life and interactions. Thus, a functional and aesthetically appealing result using CAS represents an indispensable component for mandibular reconstruction.
The introduction of microvascular surgery in the 1970s completely changed the therapeutic concept. This technique allows different bone grafts, also possible in combination with soft tissue, which might be harvested from areas distant from the defect and transplanted into the head and neck region. Here, primary or secondary reconstruction is possible. Depending on the underlying diagnosis, primary osseous reconstruction is preferred in the case of fractures or chronic osteomyelitis as well as in the case of resections due to benign lesions. In the case of malignant underlying diseases, temporary fixation by means of a reconstruction plate and secondary reconstruction is rarely performed. Through CAS, the size and extent of the tumor, the safety distances, the resection margins, as well as the reconstruction can be planned preoperatively, visualized, and further implemented intraoperatively.
Unlike reconstruction in the maxilla, the option of an obturator in the sense of a defect prosthesis [1] is not possible. The continuity of the mandible can be restored using microvascular grafts from different donor regions. Besides the quality, quantity, and shape of the bone, the variability and volume of the required soft tissue, the length of the pellicle, and the morbidity of the donor region are of particular relevance when selecting the appropriate graft. For the reconstruction of the mandible, donor regions such as the fibula, the scapula, as well as the iliac crest [2] are highly suitable for clinical and surgical demands. Along with the restoration of continuity, masticatory rehabilitation following reconstruction is also of great importance.
Virtual Planning
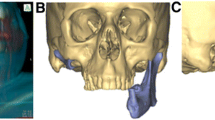
Three-dimensional virtual surgical planning (3D-VSP) uses three-dimensional imaging datasets from computed tomography (CT) and magnetic resonance imaging (MRI) of the defect site as well as CT angiography of the graft harvest site. Depending on the underlying disease, CT scans provide high resolution of both hard and soft tissues in the case of bony defects or bone erosion. In the case of soft tissue tumors and inflammatory diseases such as osteomyelitis, MRI scans can further enhance soft tissue imaging. For improving the overall image quality, the strengths of the imaging modalities can be exploited by fusing and combining CT and MRI datasets using new software programs. After merging the desired datasets, the mandible and adjacent structures are initially segmented. Intelligent software segments are semiautomatically based on algorithms, which simplifies planning and shortens the overall duration [3]. In the context of virtual planning, the size and extent of the tumor, the resection margins, and the desired reconstruction can thus be planned preoperatively, visualized, and then implemented intraoperatively. For the planning of the intraoperative navigation, special dental splints are manufactured and placed or screws are inserted before the preoperative imaging. Intraoperatively, the digital planning and the clinical situation are merged via a navigation tripod temporarily attached to the skull. During the operation, navigation can be used to directly verify the targeted resection and the correct positioning of the patient-specific implants (PSI) and graft [4]. In addition to intraoperative navigation, preoperative planning can also serve as a basis for postoperative performance monitoring. By merging the pre- and postoperative CT scans, the accuracy of fit can be validated [5]. For a functional and aesthetically appealing reconstruction, the opposite side can be used as reference or template for achieving a symmetrical result. Besides the intersegmental cutting and visualization of the microsurgical reconstruction, the dental and prosthetic rehabilitation can also be considered, simulated, and planned [6]. If the prosthetic rehabilitation is already addressed at an early stage, the procedure is referred to functional or prosthetically oriented backward planning. Thereby, the condylar position and intermaxillary relationship are reproduced, visualized preoperatively, or planned. Thus, the entire functional and dental rehabilitation is achievable. Virtual planning further allows visualization of different reconstruction possibilities enabling a completely individualized treatment concept for the patient without invasive procedures. However, not only surgeons should benefit from the possibility of visualizing the surgical site in the future. The development of virtual reality devices will also provide access to the planned operation for patients. Regarding patient education, the operation is presented in a more illustrative and tangible way, increasing the transparency of the treatment concept [7].
Patient-Specific Implants (PSIs)
Advancements in CAS, particularly regarding computer-aided design/computer-aided manufacturing (CAD/CAM) technology, are superior to more traditional methods of mandibular reconstruction with hand-bent plates [8,9,10,11,12,13,14,15]. CAD/CAM or selective laser melting (SLM) plates achieve higher accuracy compared to manually bent reconstruction plates. These plates provide greater results in terms of strength and intraoperative positioning [16]. Decisive factors for this procedure are anatomical and symmetrical bone shaping, restoration of a stable dental occlusion, and condylar repositioning into a centric relation [9, 17,18,19,20].
In the conventional technique, in contrast to CAS, the plates are bent intraoperatively or preoperatively manually before their adaptation. Depending on the complexity of the case and the skills as well as experience of the surgeon, this procedure might be very time consuming. The standard plates offered by manufacturers do not always possess the required size and number of holes for the intraoperative situation. PSIs do not need to be bent to fit the patient’s mandible and do not require predefined bending points as with conventional reconstruction [21, 22]. With improvements of CAD/CAM, it is possible to accurately plan the reconstruction of craniofacial defects preoperatively, manufacture precise PSI, and place them in less complex surgeries with shorter operating times [12, 23,24,25]. The implant can be designed and shaped by the surgeon according to the defect size, shape, and morphology [26, 27]. By selecting the appropriate design method, manufacturing process, and implant material, it is possible to perform a precise surgical procedure and reduce complications [28,29,30,31,32,33,34,35]. The integration of this technology in the pre- and intraoperative workflow has simplified the production of cutting guides and has been shown to shorten the operation time and the length of stay and to improve osseous consolidation, symmetry, and morphology [34, 36, 37]. Recent research demonstrated additional advantages, for instance, minimized interoperator variability caused by the surgeon’s experience and improved teaching possibilities for younger colleagues involved in the planning procedures/sessions with a senior consultant and/or biomedical engineer [38].
Computer-Assisted Reconstruction of the Mandible Using Microvascular Grafts
After continuity resections of the lower jaw in case of carcinoma, osteonecrosis, osteomyelitis, or trauma, a mandibular reconstruction is essential to restore function and aesthetics [6, 39]. The size of the defect is determined by the preoperative extent, the entity of the pathology, and the resulting radicality of the resection.
Defects of the mandible are reconstructible using either a reconstruction plate without bony reconstruction or immediately with a combination of reconstruction plate and primary bone flap. Despite the considerable progress in microvascular surgery, complications such as tissue necrosis, failure of the graft, infections (donor site or recipient), prolonged hospital stay, and long recovery process occur [40,41,42].
Fibula, scapula, or iliac crest grafts are suitable for the clinical requirements for reconstruction of the mandible, in the sense of a free tissue transfer.
Fibula-Tx
The microvascular fibula graft ingests a major role in computer-assisted reconstruction of the mandible and in dental rehabilitation. It provides a similar cross section as an atrophied mandible, shows good corticoid bone quality and special vascularization, and can be harvested up to a length of 20–25 cm [43].
Nevertheless, not every patient can undergo fibular grafting as in 6% of cases a nonunion of the anterior or posterior tibial artery is present [22]. In more than 1% of cases, the peroneal or fibular artery is the only vessel supplying the entire lower leg making microsurgical fibular transfer impossible. Angiography of the donor region is used to check whether transplantation is advisable, where the perforators are located, and how the graft can be obtained in an individualized manner. During virtual planning, the harvesting templates including drill holes are individually designed so that the number and position of the perforators are considered along with the planned osteotomy lines. This also facilitates the removal of a suitable skin transplant. Due to the digital and visualized planning as well as the facilitated handling, ischemia and operation time can be shortened, and an optimal aesthetic and functional result is achievable.
Scapula-Tx
The microvascular scapula graft is highly appropriate for reconstruction of the mandible [44], especially if a fibula graft is not possible due to peripheral vascular arteriopathy [45]. The lateral scapula edge with the maximal length of 14 cm can be used for bony reconstruction [46]. The circumflex scapular artery supplies the lateral scapula as the terminal branch of the subscapular artery and equips the soft tissue above the scapula via two other vascular branches. This special vascular anatomy allows two independent fasciocutaneous flaps, the scapular and the parascapular flaps, to be harvested simultaneously for soft tissue reconstruction with one pellicle [45]. Therefore, this transplant can be primarily used for the reconstruction of combined defects where intraoral and extraoral soft tissue reconstruction is necessary as well as bony reconstruction. Compared to the removal of the fibula graft, a change of position must be performed intraoperatively. When harvesting the scapula graft, it is not possible to operate in two teams and save operating time.
Iliac Crest-Tx
The iliac crest transplant is further suitable for microsurgical reconstruction of the mandible. Supplied via the circumflex ilium profunda artery, the iliac crest graft is a valid graft because of the good bone quality, the slightly curved contour, and the bone volume. The special bone structure can facilitate the desired dental rehabilitation [47]. Nevertheless, the special vascular supply (short pellicle) makes microsurgical anastomosis difficult. Thus, strict indication and concrete preoperative planning are advisable.
References
Phasuk K, Haug SP. Maxillofacial prosthetics. Oral Maxillofac Surg Clin North Am. 2018;30(4):487–97. https://doi.org/10.1016/j.coms.2018.06.009.
Mascha F, Winter K, Pietzka S, Heufelder M, Schramm A, Wilde F. Accuracy of computer-assisted mandibular reconstructions using patient-specific implants in combination with CAD/CAM fabricated transfer keys. J Craniomaxillofac Surg. 2017;45(11):1884–97. https://doi.org/10.1016/j.jcms.2017.08.028.
Rana M, Modrow D, Keuchel J, Chui C, Rana M, Wagner M, Gellrich N-C. Development and evaluation of an automatic tumor segmentation tool: a comparison between automatic, semi-automatic and manual segmentation of mandibular odontogenic cysts and tumors. J Craniomaxillofac Surg. 2015;43(3):355–9. https://doi.org/10.1016/j.jcms.2014.12.005.
Schramm A, Suarez-Cunqueiro MM, Barth EL, Essig H, Bormann K-H, Kokemueller H, et al. Computer-assisted navigation in craniomaxillofacial tumors. J Craniofac Surg. 2008;19(4):1067–74. https://doi.org/10.1097/SCS.0b013e3181760fc0.
Scolozzi P, Schouman T. Unité interventionnelle hybride multimodale: de la planification préopératoire au contrôle postopératoire immédiat. Rev Stomatol Chir Maxillofac. 2012;113(2):115–23. https://doi.org/10.1016/j.stomax.2012.01.009.
Essig H, Rana M, Kokemueller H, von See C, Ruecker M, Tavassol F, Gellrich N-C. Pre-operative planning for mandibular reconstruction—a full digital planning workflow resulting in a patient specific reconstruction. Head Neck Oncol. 2011;3(1):45. https://doi.org/10.1186/1758-3284-3-45.
Pandrangi VC, Gaston B, Appelbaum NP, Albuquerque FC, Levy MM, Larson RA. The application of virtual reality in patient education. Ann Vasc Surg. 2019;59:184–9. https://doi.org/10.1016/j.avsg.2019.01.015.
Antony AK, Chen WF, Kolokythas A, Weimer KA, Cohen MN. Use of virtual surgery and stereolithography-guided osteotomy for mandibular reconstruction with the free fibula. Plast Reconstr Surg. 2011;128(5):1080–4. https://doi.org/10.1097/PRS.0b013e31822b6723.
Eckardt A, Swennen GRJ. Virtual planning of composite mandibular reconstruction with free fibula bone graft. J Craniofac Surg. 2005;16(6):1137–40. https://doi.org/10.1097/01.scs.0000186306.32042.96.
Gil RS, Roig AM, Obispo CA, Morla A, Pagès CM, Perez JL. Surgical planning and microvascular reconstruction of the mandible with a fibular flap using computer-aided design, rapid prototype modelling, and precontoured titanium reconstruction plates: a prospective study. Br J Oral Maxillofac Surg. 2015;53(1):49–53. https://doi.org/10.1016/j.bjoms.2014.09.015.
Hallermann W, Olsen S, Bardyn T, Taghizadeh F, Banic A, Iizuka T. A new method for computer-aided operation planning for extensive mandibular reconstruction. Plast Reconstr Surg. 2006;117(7):2431–7. https://doi.org/10.1097/01.prs.0000219076.83890.e8.
Nilsson J, Hindocha N, Thor A. Time matters—differences between computer-assisted surgery and conventional planning in cranio-maxillofacial surgery: a systematic review and meta-analysis. J Craniomaxillofac Surg. 2020;48(2):132–40. https://doi.org/10.1016/j.jcms.2019.11.024.
Powcharoen W, Yang W-F, Yan Li K, Zhu W, Su Y-X. Computer-assisted versus conventional freehand mandibular reconstruction with fibula free flap: a systematic review and meta-analysis. Plast Reconstr Surg. 2019;144(6):1417–28. https://doi.org/10.1097/PRS.0000000000006261.
Rodby KA, Turin S, Jacobs RJ, Cruz JF, Hassid VJ, Kolokythas A, Antony AK. Advances in oncologic head and neck reconstruction: systematic review and future considerations of virtual surgical planning and computer aided design/computer aided modeling. J Plast Reconstr Aesthet Surg. 2014;67(9):1171–85. https://doi.org/10.1016/j.bjps.2014.04.038.
Wilde, et al. Computer-assisted mandibular reconstruction using a patient-specific reconstruction plate fabricated with computer-aided design and manufacturing techniques. Craniomaxillofac Trauma Reconstr. 2014;7(2):158–66. https://doi.org/10.1055/s-0034-1371356.
Rana M, Chin S-J, Muecke T, Kesting M, Groebe A, Riecke B, et al. Increasing the accuracy of mandibular reconstruction with free fibula flaps using functionalized selective laser-melted patient-specific implants: a retrospective multicenter analysis. J Craniomaxillofac Surg. 2017;45(8):1212–9. https://doi.org/10.1016/j.jcms.2017.04.003.
Bak M, Jacobson AS, Buchbinder D, Urken ML. Contemporary reconstruction of the mandible. Oral Oncol. 2010;46(2):71–6. https://doi.org/10.1016/j.oraloncology.2009.11.006.
Hidalgo DA, Pusic AL. Free-flap mandibular reconstruction: a 10-year follow-up study. Plast Reconstr Surg. 2002;110(2):438–49. https://doi.org/10.1097/00006534-200208000-00010.
Sonmez N, Gultekin P, Turp V, Akgungor G, Sen D, Mijiritsky E. Evaluation of five CAD/CAM materials by microstructural characterization and mechanical tests: a comparative in vitro study. BMC Oral Health. 2018;18(1):5. https://doi.org/10.1186/s12903-017-0458-2.
Wagner M, Gander T, Blumer M, Valdec S, Schumann P, Essig H, Rücker M. Die CAD/CAM-revolution in der kraniofazialen Rekonstruktion. Praxis. 2019;108(5):321–8. https://doi.org/10.1024/1661-8157/a003185.
Rendenbach C, Sellenschloh K, Gerbig L, Morlock MM, Beck-Broichsitter B, Smeets R, et al. CAD-CAM plates versus conventional fixation plates for primary mandibular reconstruction: a biomechanical in vitro analysis. J Craniomaxillofac Surg. 2017;45(11):1878–83. https://doi.org/10.1016/j.jcms.2017.08.024.
Telschow T, Wilde F, Pietzka S, Schramm A, Mascha F. Unbreakable? Frakturen patientenspezifischer Implantate nach alloplastischer Unterkieferrekonstruktion. MKG-Chirurg. 2019;12(4):263–7. https://doi.org/10.1007/s12285-019-00220-x.
Bartier S, Mazzaschi O, Benichou L, Sauvaget E. Computer-assisted versus traditional technique in fibular free-flap mandibular reconstruction: a CT symmetry study. Eur Ann Otorhinolaryngol Head Neck Dis. 2021;138(1):23–7. https://doi.org/10.1016/j.anorl.2020.06.011.
Ren W, Gao L, Li S, Chen C, Li F, Wang Q, et al. Virtual planning and 3D printing modeling for mandibular reconstruction with fibula free flap. Med Oral Patol Oral Cir Bucal. 2018;23(3):e359–66. https://doi.org/10.4317/medoral.22295.
Serrano C, van den Brink H, Pineau J, Prognon P, Martelli N. Benefits of 3D printing applications in jaw reconstruction: a systematic review and meta-analysis. J Craniomaxillofac Surg. 2019;47(9):1387–97. https://doi.org/10.1016/j.jcms.2019.06.008.
Shan X-F, Chen H-M, Liang J, Huang J-W, Cai Z-G. Surgical reconstruction of maxillary and mandibular defects using a printed titanium mesh. J Oral Maxillofac Surg. 2015;73(7):1437.e1–9. https://doi.org/10.1016/j.joms.2015.02.025.
Wilde F, Winter K, Kletsch K, Lorenz K, Schramm A. Mandible reconstruction using patient-specific pre-bent reconstruction plates: comparison of standard and transfer key methods. Int J Comput Assist Radiol Surg. 2015;10(2):129–40. https://doi.org/10.1007/s11548-014-1065-1.
Farfalli GL, Albergo JI, Ritacco LE, Ayerza MA, Milano FE, Aponte-Tinao LA. What is the expected learning curve in computer-assisted navigation for bone tumor resection? Clin Orthop Relat Res. 2017;475(3):668–75. https://doi.org/10.1007/s11999-016-4761-z.
Hanasono MM, Skoracki RJ. Computer-assisted design and rapid prototype modeling in microvascular mandible reconstruction. Laryngoscope. 2013;123(3):597–604. https://doi.org/10.1002/lary.23717.
Hsu SS-P, Gateno J, Bell RB, Hirsch DL, Markiewicz MR, Teichgraeber JF, et al. Accuracy of a computer-aided surgical simulation protocol for orthognathic surgery: a prospective multicenter study. J Oral Maxillofac Surg. 2013;71(1):128–42. https://doi.org/10.1016/j.joms.2012.03.027.
Ritschl LM, Mücke T, Hart D, Unterhuber T, Kehl V, Wolff K-D, Fichter AM. Retrospective analysis of complications in 190 mandibular resections and simultaneous reconstructions with free fibula flap, iliac crest flap or reconstruction plate: a comparative single Centre study. Clin Oral Invest. 2021;25(5):2905–14. https://doi.org/10.1007/s00784-020-03607-8.
Roser SM, Ramachandra S, Blair H, Grist W, Carlson GW, Christensen AM, et al. The accuracy of virtual surgical planning in free fibula mandibular reconstruction: comparison of planned and final results. J Oral Maxillofac Surg. 2010;68(11):2824–32. https://doi.org/10.1016/j.joms.2010.06.177.
Stirling Craig E, Yuhasz M, Shah A, Blumberg J, Salomon J, Lowlicht R, et al. Simulated surgery and cutting guides enhance spatial positioning in free fibular mandibular reconstruction. Microsurgery. 2015;35(1):29–33. https://doi.org/10.1002/micr.22229.
Weitz J, Bauer F, Hapfelmeier A, Rohleder NH, Wolff K-D, Kesting MR. Accuracy of mandibular reconstruction by three-dimensional guided vascularised fibular free flap after segmental mandibulectomy. Br J Oral Maxillofac Surg. 2016;54(5):506–10. https://doi.org/10.1016/j.bjoms.2016.01.029.
Zeller AN, Neuhaus MT, Weissbach LVM, Rana M, Dhawan A, Eckstein FM, et al. Patient-specific mandibular reconstruction plates increase accuracy and long-term stability in immediate alloplastic reconstruction of segmental mandibular defects. J Maxillofac Oral Surg. 2020;19(4):609–15. https://doi.org/10.1007/s12663-019-01323-9.
Sieira Gil R, Roig AM, Obispo CA, Morla A, Pagès CM, Perez JL. Surgical planning and microvascular reconstruction of the mandible with a fibular flap using computer-aided design, rapid prototype modelling, and precontoured titanium reconstruction plates: a prospective study. Br J Oral Maxillofac Surg. 2015;53(1):49–53. https://doi.org/10.1016/j.bjoms.2014.09.015.
Tarsitano A, Ciocca L, Scotti R, Marchetti C. Morphological results of customized microvascular mandibular reconstruction: a comparative study. J Craniomaxillofac Surg. 2016;44(6):697–702. https://doi.org/10.1016/j.jcms.2016.03.007.
Avraham T, Franco P, Brecht LE, Ceradini DJ, Saadeh PB, Hirsch DL, Levine JP. Functional outcomes of virtually planned free fibula flap reconstruction of the mandible. Plast Reconstr Surg. 2014;134(4):628e–34e. https://doi.org/10.1097/PRS.0000000000000513.
Zhou L, Shang H, He L, Bo B, Liu G, Liu Y, Zhao J. Accurate reconstruction of discontinuous mandible using a reverse engineering/computer-aided design/rapid prototyping technique: a preliminary clinical study. J Oral Maxillofac Surg. 2010;68(9):2115–21. https://doi.org/10.1016/j.joms.2009.09.033.
Chaine A, Pitak-Arnnop P, Hivelin M, Dhanuthai K, Bertrand J-C, Bertolus C. Postoperative complications of fibular free flaps in mandibular reconstruction: an analysis of 25 consecutive cases. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009;108(4):488–95. https://doi.org/10.1016/j.tripleo.2009.05.043.
Vignesh U, Mehrotra D, Howlader D, Singh PK, Gupta S. Patient specific three-dimensional implant for reconstruction of complex mandibular defect. J Craniofac Surg. 2019;30(4):e308–11. https://doi.org/10.1097/SCS.0000000000005228.
Wilkman T, Husso A, Lassus P. Clinical comparison of scapular, fibular, and iliac crest osseal free flaps in maxillofacial reconstructions. Scand J Surg. 2019;108(1):76–82. https://doi.org/10.1177/1457496918772365.
Patel A, Harrison P, Cheng A, Bray B, Bell RB. Fibular reconstruction of the maxilla and mandible with immediate implant-supported prosthetic rehabilitation: jaw in a day. Oral Maxillofac Surg Clin North Am. 2019;31(3):369–86. https://doi.org/10.1016/j.coms.2019.03.002.
Harada H, Shimamoto H, Oikawa Y, Kuroshima T, Tomioka H, Hirai H, et al. Mandibular reconstruction with scapular systems: a single-center case series involving 208 flaps. Plast Reconstr Surg. 2021;148(3):625–34. https://doi.org/10.1097/PRS.0000000000008301.
Dowthwaite SA, Theurer J, Belzile M, Fung K, Franklin J, Nichols A, Yoo J. Comparison of fibular and scapular osseous free flaps for oromandibular reconstruction: a patient-centered approach to flap selection. JAMA Otolaryngol Head Neck Surg. 2013;139(3):285–92. https://doi.org/10.1001/jamaoto.2013.1802.
Shnayder Y, Lin D, Desai SC, Nussenbaum B, Sand JP, Wax MK. Reconstruction of the lateral mandibular defect: a review and treatment algorithm. JAMA Facial Plast Surg. 2015;17(5):367–73. https://doi.org/10.1001/jamafacial.2015.0825.
Osborn TM, Helal D, Mehra P. Iliac crest bone grafting for mandibular reconstruction: 10-year experience outcomes. J Oral Biol Craniofac Res. 2018;8(1):25–9. https://doi.org/10.1016/j.jobcr.2017.12.001.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Rana, M., Möllmann, H. (2023). Microsurgical Jaw Reconstruction. In: Meyer, U. (eds) Fundamentals of Craniofacial Malformations. Springer, Cham. https://doi.org/10.1007/978-3-031-28069-6_23
Download citation
DOI: https://doi.org/10.1007/978-3-031-28069-6_23
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-28068-9
Online ISBN: 978-3-031-28069-6
eBook Packages: MedicineMedicine (R0)