Abstract
Vaccines, being one of the greatest triumphs of modern medicine, are undoubtedly the most cost-effective lifesaving device in history. Severe acute respiratory syndrome coronavirus 2 vaccines have several neurological side effects. However, they are generally mild, short-lasting, self-limiting, and manageable. Headache is reported as the most frequent neurological symptom seen after coronavirus disease 2019 (COVID-19) vaccination. It is important to differentiate vaccine-related headache from headache related to COVID-19 itself in clinical practice. Studies showed that this particular headache has heterogeneous character, being mostly moderate and bilateral, less severe, and shorter than COVID-19-related headache. Accompanying symptoms such as photophobia, phonophobia, and osmophobia typical for migraine and anosmia or ageusia typical for COVID-19-related headache are less frequently seen with COVID-19 vaccine-related headache. Female gender, primary headaches, history of COVID-19-related headache, influenza vaccine-related headache, allergy, and thyroid disorders seem to increase risk of experiencing headache after COVID-19 vaccination. Although rare, secondary headaches related to cerebral venous thrombosis following COVID-19 vaccines were also reported.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
7.1 Introduction
Vaccination against coronavirus disease 2019 (COVID-19) has a crucial role to achieve population immunity and restrict the spread of the disastrous pandemic declared on March 2020. Vaccines are indeed one of the greatest triumphs of modern medicine and undoubtedly the most cost-effective lifesaving device in the history. Numerous vaccines against COVID-19 are still in clinical development, and some of them are currently in use. Although their safety and efficacy are well documented, randomized controlled trials and further clinical studies reported various systemic adverse reactions following vaccination [1]. Neurological side effects to SARS-CoV-2 vaccines are generally mild, short-lasting, self-limiting, and mostly manageable. Headache is reported as the most frequent neurological symptom seen after SARS-CoV-2 vaccination according to the available data [2]. Moreover, headache is an overlap symptom for COVID-19 and also for COVID-19 vaccination. Therefore, knowledge of characteristics of headache seen following vaccination seems to be crucial to differentiate vaccine-related headache from headache related to COVID-19 itself.
7.2 Mechanisms of Headache Related to Vaccination
Headache related to vaccination was reported so far with various types of viral vaccines at different rates: after adenovirus-vectored (AdV) Ebola virus vaccine in 46%, measles-mumps-rubella vaccine in 35%, and inactivated influenza vaccine in 7–31.8% of the participants. It was also seen following vaccination against human papillomavirus and varicella-zoster virus [3, 4]. However, the mechanisms underlying vaccine-related headaches are not clearly illuminated.
To date, COVID-19 vaccines globally in use have one of the following technologies: viral vector-based, mRNA-based, DNA-based, whole inactivated virus, and protein subunit vaccines [5]. Numerous studies were performed to better understand the virus, and the S protein is the key target of the vaccines for SARS-CoV-2. Murine challenge models and also clinical trials detected potent humoral and cellular immune responses elicited by vaccines encoding S protein. Beside S protein, other proteins (M protein, N protein, non-structural proteins (nsps), and accessory proteins) may also serve as antigens [6].
There are several possible mechanisms underlying COVID-19 vaccine-related headache. A pro-inflammatory state occurring following vaccination, similar to that in COVID-19-related headache with high levels of pro-inflammatory cytokines, may have the predominant role [7]. Furthermore, fever accompanying more frequently COVID-19 vaccine-related headache may support the role of inflammation, leading to the development of vaccine-related headache. On the other hand, COVID-19 vaccines with inactivated virus include adjuvants, which may promote headache following vaccination. Although rare, vaccine-induced blood clotting events were also defined, and some vaccine-related secondary headache conditions were reported, which will be further presented in this chapter.
7.3 Frequency of Headache Related to COVID-19 Vaccines
Clinical trials revealed an acceptable safety profile with high antibody responses [8]. Post-authorization observational studies reported similar rates of adverse events with clinical trials. However, there were significant differences among vaccine platforms, and the dosage was closely related to the occurrence of headache as an adverse reaction (Tables 7.1 and 7.2) [5, 9, 10].
The pooled rates of systemic and local adverse events were found to be lower among inactivated vaccines (21.0%, 23.7%), protein subunit vaccines (22.3%, 33.0%), and DNA vaccines (29.3%, 39.5%), compared to mRNA vaccines (83.3%, 89.4%), non-replicating vector vaccines (66.3%, 55.9%), and virus-like particle vaccines (78.9%, 100.0%) [5]. Similar to these findings, headache was less reported in subjects vaccinated with inactivated virus, as well as all other types of side effects. This may be related to the mature technology, the mechanism, the alum-adjuvants, or other factors [9]. However, vaccine-related adverse events depend also on dosage. Regardless of the vaccine identity, subjects experienced more headache following the second dose of mRNA vaccines on both clinical trials and post-authorization studies [5]. The impact of the route of administration was also evaluated. A study evaluating the safety of an aerosolized form of adenovirus type-5 vector-based COVID-19 vaccine (Ad5-nCoV) in adults reported headache in 46 (35%) participants following vaccination, similar to other studies investigating viral vector based COVID-19 vaccines [11].
Interestingly, clinical trials showed that frequency of headache as well as other side effects decreased with age in all vaccine platforms. Binding-antibody levels were found to be low after COVID-19 vaccination in subjects older than 70 years, a finding that suggests lower immune responses in elderly, causing fewer adverse events [9].
However, findings based on the data of vaccines in real-world registries (VAERS) was somewhat surprising. Researchers exploring these big data, of 11,936 participants, found out lower rates of adverse events in the real world than those reported in clinical trials, and headache had the highest incidence (16.53%) among other adverse events [12]. In children, COVID-19 vaccines showed also good safety profiles according to VAERS reports, and mRNA vaccine-related headache was reported in only 13.9% and 19.8% of the children, being more frequent following second dose than the first dose [13].
7.4 Clinical Features of Headache Following Vaccination with COVID-19 Vaccines
To date, few studies focused on headache following COVID-19 vaccines and evaluated in detail clinical characteristics of headache. Headache starts within 24–48 h after the vaccine injection and resolves spontaneously and lasts less than 24 h, in the majority of cases. However, long-lasting headaches following vaccination are also reported. A multicenter observational study showed that headache started within 24 h after vaccination in 80% and lasted less than 22 h in 80% and more than 36 h in only 10% of the participants vaccinated with mRNA vaccine. Interestingly, headache occurred in multiple episodes in one third of the subjects [14]. Another study showed that the latency from vaccination to the occurrence of headaches was 18 h with a duration of 14 h on average [2]. In a survey study addressing healthcare workers, temporal characteristics of headache related to inactivated virus vaccine showed moderate differences from those of the headache related to mRNA vaccine. The headache occurred 1.8 ± 3.5 (median, 1; IQR, 0–2) days later after vaccination and lasted less than 24 h in 61.1% and more than 3 days in 25.9% of the subjects. However, it was overall shorter than COVID-19-related headache experienced by the same population [10].
In subjects having migraine, headache attacks began within the first 24 h of vaccine in the half of the participants following mRNA or DNA viral vector vaccines administration. These attacks lasted more than 24 h up to 7 days in 46.1% of the migraineurs [15]. It seems reasonable to suggest that COVID-19 vaccine-related headache potentiates migraine pathways and causes longer duration of attacks.
Although there are cases perceiving mild or very severe headache following vaccination, the severity of vaccine-related headache seems to be predominantly moderate. COVID-19 vaccine-related headache was less severe than COVID-19-related headache and migraine pain, whereas it was more severe than tension-type headache attacks in healthcare workers who received inactivated virus vaccine [10]. Studies investigating mRNA vaccine-related headache disclosed that the headache was moderate in nearly half of the cases (in 46.2% of the subjects). However, the pain affected routine physical activity in 42.8% of all study group [14]. A study focusing the effects of COVID-19 vaccines in migraineurs found that the character of headache following vaccination was different from migraine attacks and pain intensity was higher in more than half of the participants [15]. The severity of headache was also related to dosage; a higher severity following the second dose of the mRNA vaccine compared to the first one and between the first dose of the AdV vaccine and the second dose of the mRNA vaccine was observed [16].
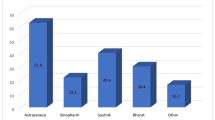
The rates of areal location of the headache showed more or less a similar distribution on frontal and temporal areas and at the back of the head (38%, 32%, 23%, respectively) [14]. COVID-19 inactivated virus vaccine-related headache was mostly bilateral (70%) similar to the location of tension-type headache and contrary to migraine attacks in the same population [10]. Headache following mRNA vaccine was also bilateral in most of the participants (73.1%). Studies investigating subjects who received mRNA or inactivated virus vaccines reported heterogeneous headache characters. It was mostly pressing in mRNA vaccine group, whereas mostly throbbing in inactivated virus vaccine group [10, 14]. Furthermore, symptoms such as fatigue, muscle pain, joint pain, or fever were found to be associated with COVID-19 vaccine-related headache [10, 14, 15]. Poor concentration and dizziness were also observed in cases with headache following vaccination [14], whereas sensitivity to noise, light, or odors typical for migraine was less frequently reported by vaccinees receiving mRNA or inactivated virus vaccines [10, 14]. Moreover, anosmia or ageusia, typical symptoms for COVID-19-related headache, accompanied rarely vaccine-related headache [10]. Clinical features associated with COVID-19 vaccine-related headache are given in Fig. 7.1.
7.5 Comorbidities and Immunization History
Preexisting primary headache disorders like migraine or tension-type headache were also found to be more often present in cases having COVID-19 vaccine-related headache after either mRNA or inactivated virus vaccines [10, 14]. The duration of headache after vaccination was also significantly longer and more severe in subjects with migraine than those without this disorder [14]. Other diseases such as thyroid, pancreas, pulmonary, and vascular disorders showed also significant association with vaccine-related headache [10, 14]. Furthermore, severe COVID-19 and COVID-19-related headache were reported more frequently in the group experiencing COVID-19 vaccine-related headache when compared to those without COVID-19 vaccine-related headache in healthcare workers [10]. Moreover, history of headache following other types of vaccines such as influenza vaccine was found to be a risk factor increasing 4.8-fold (CI, 2.84–8.23) the risk for experiencing COVID-19 inactivated virus vaccine-related headache [10].
7.6 Gender Differences and Headache Characteristics in Specific Subpopulations
Even though headache related to COVID-19 itself showed a male predilection, significant female dominance was found in subjects experiencing inactivated virus or mRNA vaccine-related headache [10, 17]. Moreover, women had a more severe headache in comparison to men. However, a gender difference in terms of the latency and the duration of headache was not notified [14]. On the other hand, this significant difference in the prevalence of headache in terms of gender disappeared in the population older than 75 years [18] and was particularly remarkable in young adults (18–30 years old) [19].
Furthermore, headache following vaccination with COVID-19 mRNA vaccines was less commonly seen among pregnant women after both doses in comparison to non-pregnant women, as well as myalgia and arthralgia. The prevalence of headache was similar in each trimester of pregnancy [20]. However, side effects following COVID-19 mRNA vaccine, such as headache, fatigue, arthralgia, and muscle pain, showed higher frequencies and tended to be more severe in individuals with different allergy histories than those without any allergy disorder. These side effects showed also a female dominance in allergic individuals [21].
7.7 Treatment
Unfortunately, there is not yet an established algorithm designated for the treatment of headache following COVID-19 vaccination. Studies focusing on COVID-19 vaccine-related headache reported that paracetamol and nonsteroidal anti-inflammatory drugs (particularly ibuprofen) were the mostly used painkillers [10, 14]. A recent survey study disclosed improvement without analgesics in almost one-third of subjects and a significant improvement of vaccine-related headache with painkillers in comparison to COVID-19-related headache [10], whereas migraineurs reported that the attacks after vaccination were different than their routine migraine attacks, with a lower responsiveness to painkillers almost in half of the patients [15].
7.8 Secondary Headaches
Secondary headaches are primarily related to underlying critical conditions, and secondary headaches associated with COVID-19 vaccines are also compatible with this rule of thumb. Although rare, life-threatening secondary headaches were also reported following COVID-19 immunization with vaccines, and emerging evidence has shown that the etiology of secondary headaches following vaccination is cerebral venous thrombosis, in most of the cases [22].
Cerebral venous thrombosis is accompanied by severe thrombocytopenia because of the presence of antiplatelet factor 4 antibodies in some cases. This clinical entity is called as vaccine-associated immune thrombosis and thrombocytopenia (VITT), in other words, thrombosis in thrombocytopenia syndrome [23, 24]. While thrombocytopenia is a key characteristic, the female to male ratio is 2:1. According to the literature, differentiation between primary and secondary type of headaches following COVID-19 vaccination may be done according to some clue clinical features. Especially delayed-onset headaches after COVID-19 vaccine should be evaluated thoroughly for secondary and life-threatening causes. Other indicators for secondary headaches seem to be female gender and being younger than 60 years old [22]. Furthermore, persistent headaches despite painkillers should raise concern regarding secondary etiologies [25].
The diagnosis of VITT requires the presence of antibodies against platelet factor 4 (PF4), determined using enzyme-linked immunosorbent assays (ELISAs) [26]. Furthermore, D-dimer levels are elevated in these patients, and it was shown that they are fourfold higher in subjects diagnosed with VITT [27]. Considering the underlying pathophysiology, treatment options targeting the immune system might be beneficial such as intravenous immunoglobulin (IVIG) and steroids in patients with VITT-related cerebral venous thrombosis [27].
Increased risk of ischemic stroke after BNT162b2 mRNA vaccination was also reported in a study including 9,513,625 participants. However, this study was a self-control retrospective study, and COVID-19-related vascular events have been found higher in the same population. Finally, there is not enough data regarding the clinical features including the presence of headaches in this population [28]. Furthermore, other etiologies underlying secondary headaches associated with COVID-19 vaccines have been rarely reported, such as a 45-year-old male patient diagnosed with Tolosa-Hunt syndrome following an mRNA-based COVID-19 vaccine [29].
7.9 Conclusion
COVID-19 vaccine-related headache is frequently seen and seems to be elicited by the pro-inflammatory state following vaccination. Fortunately, studies showed that this particular headache starting in general with a latency of 24–48 h and showing heterogeneous character has a self-limiting course and is mostly mild and short-lasting. There is not yet an established algorithm for the treatment of headache following COVID-19 vaccination, a considerable percentage of the cases recovers without medication, and standard painkillers may be effective for the relief. Although rare, secondary headaches related to COVID-19 vaccine should also be considered in patients with long-lasting and treatment-resistant headaches.
References
Gringeri M, Mosini G, Battini V, et al. Preliminary evidence on the safety profile of BNT162b2 (Comirnaty): new insights from data analysis in EudraVigilance and adverse reaction reports from an Italian health facility. Hum Vaccin Immunother. 2021;17(9):2969–71. https://doi.org/10.1080/21645515.2021.1917236.
Finsterer J. Neurological side effects of SARS-CoV-2 vaccinations. Acta Neurol Scand. 2022;145(1):5–9. https://doi.org/10.1111/ane.13550.
James SF, Chahine EB, Sucher AJ, Hanna C. Shingrix: the new adjuvanted recombinant herpes zoster vaccine. Ann Pharmacother. 2018;52(7):673–80. https://doi.org/10.1177/1060028018758431.
Pépin S, Donazzolo Y, Jambrecina A, et al. Safety and immunogenicity of a quadrivalent inactivated influenza vaccine in adults. Vaccine. 2013;31(47):5572–8. https://doi.org/10.1016/j.vaccine.2013.08.069.
Wu Q, Dudley MZ, Chen X, et al. Evaluation of the safety profile of COVID-19 vaccines: a rapid review. BMC Med. 2021;19(1):173. https://doi.org/10.1186/s12916-021-02059-5.
Bisht H, Roberts A, Vogel L, et al. Severe acute respiratory syndrome coronavirus spike protein expressed by attenuated vaccinia virus protectively immunizes mice. Proc Natl Acad Sci U S A. 2004;101(17):6641–6. https://doi.org/10.1073/pnas.0401939101.
Bolay H, Karadas Ö, Oztürk B, et al. HMGB1, NLRP3, IL-6 and ACE2 levels are elevated in COVID-19 with headache: a window to the infection-related headache mechanism. J Headache Pain. 2021;22(1):94. https://doi.org/10.1186/s10194-021-01306-7.
Folegatti PM, Ewer KJ, Aley PK, et al. Safety and immunogenicity of the ChAdOx1 nCoV-19 vaccine against SARS-CoV-2: a preliminary report of a phase 1/2, single-blind, randomised controlled trial. Lancet (London, England). 2020;396(10249):467–78. https://doi.org/10.1016/S0140-6736(20)31604-4.
Chen J, Cai Y, Chen Y, et al. Nervous and muscular adverse events after COVID-19 vaccination: a systematic review and meta-analysis of clinical trials. Vaccine. 2021;9:8. https://doi.org/10.3390/vaccines9080939.
Ekizoglu E, Gezegen H, Yalınay Dikmen P, et al. The characteristics of COVID-19 vaccine-related headache: clues gathered from the healthcare personnel in the pandemic. Cephalalgia. 2022;42(4-5):366–75. https://doi.org/10.1177/03331024211042390.
Wu S, Huang J, Zhang Z, et al. Safety, tolerability, and immunogenicity of an aerosolised adenovirus type-5 vector-based COVID-19 vaccine (Ad5-nCoV) in adults: preliminary report of an open-label and randomised phase 1 clinical trial. Lancet Infect Dis. 2021;21(12):1654–64. https://doi.org/10.1016/S1473-3099(21)00396-0.
Cai C, Peng Y, Shen E, et al. A comprehensive analysis of the efficacy and safety of COVID-19 vaccines. Mol Ther. 2021;29(9):2794–805. https://doi.org/10.1016/j.ymthe.2021.08.001.
Hause AM, Baggs J, Marquez P, et al. COVID-19 vaccine safety in children aged 5-11 years—United States, November 3-December 19, 2021. MMWR Morb Mortal Wkly Rep. 2021;70(5152):1755–60. https://doi.org/10.15585/mmwr.mm705152a1.
Göbel CH, Heinze A, Karstedt S, et al. Clinical characteristics of headache after vaccination against COVID-19 (coronavirus SARS-CoV-2) with the BNT162b2 mRNA vaccine: a multicentre observational cohort study. Brain Commun. 2021;3(3):fcab169. https://doi.org/10.1093/braincomms/fcab169.
Silvestro M, Tessitore A, Orologio I, et al. Headache worsening after COVID-19 vaccination: an online questionnaire-based study on 841 patients with migraine. J Clin Med. 2021;10:24. https://doi.org/10.3390/jcm10245914.
Perrotta A, Biondi-Zoccai G, Saade W, et al. A snapshot global survey on side effects of COVID-19 vaccines among healthcare professionals and armed forces with a focus on headache. Panminerva Med. 2021;63(3):324–31. https://doi.org/10.23736/S0031-0808.21.04435-9.
Klugar M, Riad A, Mekhemar M, et al. Side effects of mRNA-based and viral vector-based COVID-19 vaccines among German Healthcare Workers. Biology (Basel). 2021;10:8. https://doi.org/10.3390/biology10080752.
Choi YY, Kim M-K, Kwon HC, Kim GH. Safety monitoring after the BNT162b2 COVID-19 vaccine among adults aged 75 years or older. J Korean Med Sci. 2021;36(45):e318. https://doi.org/10.3346/jkms.2021.36.e318.
Riad A, Pokorná A, Klugarová J, et al. Side effects of mRNA-based COVID-19 vaccines among young adults (18-30 years old): an independent post-marketing study. Pharmaceuticals (Basel). 2021;14:10. https://doi.org/10.3390/ph14101049.
Bookstein Peretz S, Regev N, Novick L, et al. Short-term outcome of pregnant women vaccinated with BNT162b2 mRNA COVID-19 vaccine. Ultrasound Obstet Gynecol. 2021;58(3):450–6. https://doi.org/10.1002/uog.23729.
Inoue S, Igarashi A, Morikane K, et al. Adverse reactions to BNT162b2 mRNA COVID-19 vaccine in medical staff with a history of allergy. Respir Investig. 2022;60(2):248–55. https://doi.org/10.1016/j.resinv.2021.11.007.
García-Azorín D, Do TP, Gantenbein AR, et al. Delayed headache after COVID-19 vaccination: a red flag for vaccine induced cerebral venous thrombosis. J Headache Pain. 2021;22(1):108. https://doi.org/10.1186/s10194-021-01324-5.
Franchini M, Liumbruno GM, Pezzo M. COVID-19 vaccine-associated immune thrombosis and thrombocytopenia (VITT): diagnostic and therapeutic recommendations for a new syndrome. Eur J Haematol. 2021;107(2):173–80. https://doi.org/10.1111/ejh.13665.
Ropper AH, Klein JP. Cerebral venous thrombosis. N Engl J Med. 2021;385(1):59–64. https://doi.org/10.1056/NEJMra2106545.
Garg RK, Paliwal VK. Spectrum of neurological complications following COVID-19 vaccination. Neurol Sci. 2022;43(1):3–40. https://doi.org/10.1007/s10072-021-05662-9.
Schultz NH, Sørvoll IH, Michelsen AE, et al. Thrombosis and thrombocytopenia after ChAdOx1 nCoV-19 vaccination. N Engl J Med. 2021;384(22):2124–30. https://doi.org/10.1056/NEJMoa2104882.
Scully M, Singh D, Lown R, et al. Pathologic antibodies to platelet factor 4 after ChAdOx1 nCoV-19 vaccination. N Engl J Med. 2021;384(23):2202–11. https://doi.org/10.1056/NEJMoa2105385.
Hippisley-Cox J, Patone M, Mei XW, et al. Risk of thrombocytopenia and thromboembolism after covid-19 vaccination and SARS-CoV-2 positive testing: self-controlled case series study. BMJ. 2021;374:n1931. https://doi.org/10.1136/bmj.n1931.
Chuang TY, Burda K, Teklemariam E, Athar K. Tolosa-hunt syndrome presenting after COVID-19 vaccination. Cureus. 2021;13(7):e16791. https://doi.org/10.7759/cureus.16791.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Ekizoglu, E., Sezgin, M. (2023). COVID-19 Vaccine-Related Headache. In: Özge, A., Uludüz, D., Bolay, H., Karadaş, Ö. (eds) Headache Disorders in Pandemic Conditions . Headache. Springer, Cham. https://doi.org/10.1007/978-3-031-26309-5_7
Download citation
DOI: https://doi.org/10.1007/978-3-031-26309-5_7
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-26308-8
Online ISBN: 978-3-031-26309-5
eBook Packages: MedicineMedicine (R0)