Abstract
Enhanced Recovery After Surgery (ERAS) protocols are multidisciplinary approaches to perioperative care, which aim to optimize surgical recovery by incorporating evidence-based approaches in the preoperative, intraoperative, and postoperative periods. As such, the goal of these strategies is to minimize stressors from various physiological and psychological sources. There is a compelling case for the implementation of ERAS into the routine management of spinal surgery, as many of these procedures are associated with long operative durations, considerable homeostatic stress, and significant postoperative pain which can lead to prolonged recovery, delayed mobilization, and increased opioid use. Elderly patients are a particularly vulnerable patient population in elective spinal surgery, as they tend to have more medical comorbidities and are also more sensitive to opioids. The purpose of this chapter is to summarize the components of ERAS protocols as they relate to spine surgery, and highlight advances in perioperative management such as the role of multimodal analgesia and minimally invasive techniques. A comprehensive literature review of published ERAS protocols and outcomes is provided.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
A patient’s surgical experience is comprised of different facets of perioperative care, including the preoperative, intraoperative, and postoperative phases, and is overseen by a multitude of practitioners. As such, postoperative recovery is a complex process that is not only influenced by a technically successful operation but also depends on the quality of perioperative care as coordinated by a multidisciplinary team. Such coordinated efforts are essential in reducing pain, morbidity, and recovery time. Indeed, a significant proportion of patients undergoing surgery will experience postoperative pain, with the majority reporting moderate or extreme pain [1]. Inadequate postoperative pain control has numerous adverse effects on the patient and healthcare system, including unwanted and harmful physiological side effects, poor patient satisfaction, and an increased overall cost of healthcare resource utilization [2].
Enhanced Recovery After Surgery (ERAS) protocols have thus been developed as a conceptual framework of optimizing surgical recovery. The core philosophy of ERAS consists of a multimodal approach to perioperative management, with the implementation of evidence-based approaches to treatment using a multidisciplinary team [3]. In 2001, the ERAS study group was founded by a group of European academic surgeons, who first developed a multimodal recovery protocol for colonic surgery based on the published literature [4]. Previously, the concept of “fast-track” surgery had been described in different specialties such as cardiac and general surgery [5,6,7] with an initial focus on expediting the speed of recovery, which then developed into a protocol for optimizing perioperative management to reduce complications and enhance recovery [8]. Subsequently, the ERAS Society was founded with a mission to “develop perioperative care and to improve recovery through research, education, audit and implementation of evidence-based practice” (http://www.erassociety.org).
Although the ERAS Society has published numerous guidelines and consists of multiple specialties, there is no neurosurgical representation, and no guidelines exist regarding the perioperative management of spinal surgeries; however, the Congress of Neurological Surgeons is currently developing perioperative spine surgery guidelines, which are expected to be published in 2021. Until recently, the literature lacked detailed studies outlining ERAS protocols and outcomes for spine surgery [9], and the past few years have seen a newfound enthusiasm in ERAS development for a variety of spinal procedures and pathologies [10, 11]. The purpose of this chapter is to outline the components of ERAS protocols as they relate to spine surgery, and to review the process of ERAS development and published outcomes in the literature.
Rationale for the Use of ERAS in Spinal Surgery
There is a compelling case for the implementation of ERAS into the routine management of spinal surgery. Some spine procedures are associated with long operative duration, extensive muscle retraction and dissection, and the implantation of hardware, which can lead to prolonged recovery, delayed mobilization, and significant pain. In particular, both lumbar fusion and complex spinal reconstruction procedures have been rated by patients as having the most significant pain on the first postoperative day [12]. Accordingly, postoperative pain influences several outcome measures, including length of hospitalization, time to mobilization, readmission rates, and opioid tolerance and dose escalation [13].
The complexity of pain management for spinal pathologies is derived from the diverse pain etiologies arising from nociceptive, neuropathic, and inflammatory mechanisms, with potential anatomical sources of pain including the paraspinal muscles, bone, facet joints, and the intervertebral discs [14]. In addition to delaying recovery and prolonging a patient’s initial hospital admission, the intensity of pain experienced in the early postoperative period may lead to the development of chronic postsurgical pain [15]. Pain can also be associated with kinesiophobia, or the “fear of movement” following spine surgery, which can impair early mobilization, leading to even greater pain, disability, and adverse psychological effects [16, 17].
Previously, the liberal use of opioids was favored in the treatment of acute postoperative pain; however, the rise in morbidity and mortality associated with acute and chronic opioid therapy has encouraged the development of multimodal analgesia (MMA) paradigms to both reduce perioperative opioid use and improve postoperative pain control and patient recovery [18, 19]. It is important to recognize that a majority of patients presenting for major spine surgery are taking opioids and that higher utilization of preoperative opioid use and higher pain scores are associated with chronic postoperative opioid use [20]. Interestingly, the preoperative use of high-potency opioids has also been associated with an increased reoperation rate following lumbar decompression or fusion surgeries [21]. Furthermore, opioid use in the elderly can be fraught with complications, as these patients are at an elevated risk of developing complications due to their comorbidities and higher likelihood of polypharmacy [22]. In particular, one must take into account a potentially elevated fall and fracture risk [23]. Although data on opioid abuse for adults aged 65 years and older are largely lacking [24], a number of studies have reported increasing rates of misuse and addiction [25, 26].
In the United States, the rates of surgical procedures for degenerative spine disease have rapidly increased over the past few decades, in particular the use of fusion for lumbar stenosis and spondylolisthesis [27,28,29]. The increasing complexity of cases necessitating fusion has also been associated with increased cost and risk of major complications and mortality [30]. Interestingly, some studies have suggested that postoperative, but not intraoperative, events are more predictive of increased length of stay (LOS) following lumbar fusion [31]. There is significant diversity in the nature of postoperative care among institutions and individual surgeons, with differing practices regarding the prescribing of medications, mobilization, and instructions for return to activity or work. As such, implementation of an ERAS protocol following spine surgery could potentially streamline postoperative care and improve outcomes.
ERAS Components
A multimodal ERAS management strategy focuses on optimizing the preoperative, intraoperative, and postoperative periods (Fig. 6.1). The foundation of these strategies is in minimizing stressors from a variety of physiological, psychological, and economic sources [32, 33].
Preoperative Period
Preoperative optimization begins even before the patient presents to the hospital for their procedure. Preparation begins with education, which can include the basic details of the surgical procedure, expected length of the procedure and LOS, postoperative expectations for discharge, potential restrictions on mobility and activity, and expected course of postoperative recovery. Despite the seemingly basic nature of this information, having a structured approach to providing education can empower patients. Indeed, there is evidence that such preoperative education sessions improve pain, function, and psychological outcomes following spine surgery [34, 35]. “Prehabilitation,” or the process of enhancing functional capacity prior to elective surgery, has also been researched in spine surgery [36]; however, there are few studies addressing this topic, and it is not clear if their implementation results in significant improvements in pain or function.
Additional preoperative considerations include the management and optimization of comorbidities, particularly in an elderly population with a higher incidence of heart disease and diabetes. The rising rates of obesity are concerning, and there is often an association with diabetes. Multiple studies have shown that patients with poorly controlled diabetes have a higher incidence of postoperative complications following spine surgery, including infection and poor wound healing [37, 38]. Similarly, tobacco use is associated with a higher rate of adverse events following spine surgery, including infection, fusion failure, and cardiopulmonary complications [39, 40]. Screening for such factors is essential to optimize preoperative health and function. Other considerations include the identification of nutritional insufficiency or malnutrition—this often unrecognized risk factor has been independently associated with adverse events following spine surgery including infection, increased LOS, and mortality [41, 42]. In addition to these modifiable factors, non-modifiable factors such as the use of anticoagulation medications may also contribute to the development of perioperative complications and should be managed appropriately.
As discussed, infection is an important complication in spine surgery. In addition to optimizing comorbidities, other critical considerations include the use of preoperative antibiotics, and appropriate sterile preparation and surgical technique. Preoperative bathing with chlorhexidine gluconate has received recent attention in the orthopedic literature as a means of reducing surgical site infections [43]. A recent analysis of 4266 spine surgeries reported that the implementation of a protocol requiring patients to shower at least 3 times prior to surgery with chlorhexidine significantly decreased the risk of developing an infection [44]. Interestingly, this decreased risk was only observed in patients undergoing spine surgery without fusion in univariate analysis, perhaps as a result of the increased complexity of cases requiring fusion [44].
“Preemptive” analgesia refers to the preoperative administration of pain medications to prevent postoperative pain. Mechanistically these medications inhibit or reduce autonomic reactivity and nociceptive signals generated through tissue damage and inflammation [13]. The use of gabapentinoids, including gabapentin and pregabalin in the preoperative setting, has been demonstrated to decrease opioid consumption and improve pain scores following lumbar surgery [45, 46], and both drugs may be equally efficacious [47]. The use of nonsteroidal anti-inflammatory drugs (NSAIDs) has also been demonstrated to improve pain scores and reduce opioid use in the postoperative period [48]. Although there have been concerns regarding the use of NSAIDs and the development of pseudoarthrosis or nonunion, numerous studies have reported their safety in the postoperative period when judiciously dosed [49, 50]. Many ERAS protocols will combine different agents based on institutional or provider preference, and common combinations include a gabapentinoid, NSAID, and/or acetaminophen [51,52,53]. Another consideration is timing of medication administration, as different protocols may initiate therapy on the morning of surgery and/or the night before surgery. For gabapentinoids, this may include administration of a single or divided oral dose of 300–1200 mg, 2–24 h before surgery [54]; one meta-analysis of multiple inpatient surgical procedures identified an association between the cumulative gabapentin dose and a total reduction in morphine consumption [54].
Intraoperative Period
There are numerous intraoperative considerations for ERAS implementation, including the choice of anesthetic agent, MMA with a focus on opioid-sparing medications, antimicrobial prophylaxis, and the maintenance of both normothermia and normovolemia.
A number of different anesthetic regimens have been described in conjunction with ERAS protocols. When general anesthesia is desired, propofol tends to be the agent of choice in multiple ERAS paradigms [52, 53, 55, 56]. The use of ketamine as both a pre-incisional bolus and intraoperative infusion has been reported to reduce opioid consumption and incisional hyperalgesia, and also improve the efficacy of neurophysiologic monitoring by reducing inhalational anesthetic requirements [14, 57]. Dexmedetomidine has also been investigated as a sedative and analgesic adjuvant in spine surgery, with notable reductions in both intraoperative and postoperative opioid use reported [58]. Intravenous glucocorticoids have been reported to reduce postoperative pain as well as nausea and vomiting [59], although some studies have reported a higher rate of postoperative infection [60]. The use of regional (neuraxial) anesthesia in spine surgery has also been described in a number of studies, commonly employing the use of spinal bupivacaine [61]. Compared to a general anesthetic, regional anesthesia may be associated with decreased blood loss, a lower incidence of nausea and vomiting, and reduced pain scores and LOS [61].
Infiltration of the incision with local anesthetic is a widely utilized and efficacious technique [62], and is routinely infiltrated underneath the skin prior to incision and into the muscle prior to closure. Such measures may reduce postoperative pain scores and opioid use [63]. Although local anesthetics are generally limited by their relatively short duration of action, there has been recent enthusiasm in the use of multivesicular liposomes containing bupivacaine, which allows for sustained drug release that can last for a few days. Recent studies assessing the use of liposomal bupivacaine suggest improved mobility and reduced opioid consumption when either used as a sole intervention [64] or in conjunction with an ERAS protocol [65]. The use of intrathecal morphine has also been reported to reduce pain scores and postoperative opioid use following spine surgery [66]. Complications including pruritus and respiratory depression have been reported in some studies [66]. Although pain is improved in the immediate postoperative period, the efficacy tends not to persist after 48–72 h, and is not associated with a decreased LOS [66,67,68].
Spine operations can be associated with numerous homeostatic insults, particularly in those of longer duration and requiring more exposure and/or instrumentation. Longer operations are associated with potentially extended periods of hypothermia, which has been reported to increase the incidence of infection [69]; as such, maintaining overall normothermia and targeting a core temperature of 36 °C is considered essential. Major spine surgery can also be associated with elevated blood loss, resulting in hypotension and an increased risk of end-organ damage. In one study, patients requiring a blood transfusion during lumbar fusion were significantly more likely to develop a complication, including sepsis, pulmonary embolus, or infection [70]. The maintenance of normovolemia in spine surgery is therefore essential, and has been associated with reduced blood loss and lower rates of transfusion, as well as improved respiratory and bowel function [71]. The use of tranexamic acid has been reported to be effective in reducing perioperative blood loss and the need for transfusion [72], and may provide an especially useful adjunct when used in conjunction with thorough surgical hemostasis techniques [73].
An additional consideration is the avoidance or early removal of urinary catheters and surgical drains. Urinary catheters are associated with development of urinary tract infections, and there is mixed evidence in their association with the development of a surgical site infection following spine surgery [74]. The prolonged use of surgical drains has similarly been reported as an independent risk factor for developing a surgical site infection [75]. Minimally invasive surgeries (MIS) may be able to avoid the use of catheters and drains, although they may be necessary following major spine surgery with longer durations and larger exposures. Accordingly, many ERAS protocols specify early removal to facilitate mobilization [11].
Postoperative Period
The key postoperative considerations following spine surgery focus on pain control, mobilization and the path to discharge. Whereas early “fast-track” protocols may have focused exclusively on the speed of recovery and discharge, ERAS places greater emphasis on optimizing the patient experience.
One of the most important postoperative considerations is that of pain control and the appropriate medication regimen. If implemented judiciously, the use of preemptive and intraoperative MMA as described in previous sections can improve postoperative pain control through an opioid-sparing approach. Different agents are used in combination to synergistically treat pain, and common postoperative drugs include acetaminophen, gabapentin or pregabalin, NSAIDs, and muscle relaxants. The use of scheduled NSAIDs, as opposed to the as-needed administration, may act synergistically with opioids in the postoperative period [18]. This may enable an overall decreased dose of opioids, indirectly reducing postoperative nausea and sedation [18]. Such benefits may be of particular use to the elderly population, who are particularly susceptible to opioid-related side effects [22, 76]. Of course the use of perioperative NSAIDs must be measured with the risk of potential platelet dysfunction, gastrointestinal irritation, and/or renal impairment [18].
The use of NMDA agonists, such as ketamine, in the perioperative period may also be an effective approach to surgical pain. Different administration protocols have been described, including its use as a pre-incisional bolus, an intraoperative infusion, and postoperative use in combination with a patient-controlled analgesia (PCA) pump [77]. Given as infusions, they can lower the use of opioid therapy and decrease nausea and vomiting [18, 57, 77]. However, the administration of the agent in this manner may require a coordinated effort from pharmacy, the recovery unit nursing staff, and anesthesia, who will likely ultimately be overseeing its delivery. Furthermore, ketamine is a psychoactive drug, which could potentially contribute to the development of postoperative cognitive side effects in a vulnerable elderly population [78]. In a multicenter randomized trial of patients aged 60 years or older undergoing major surgery, the use of a subanesthetic ketamine dose during surgery did not reduce the incidence of postoperative delirium, and instead increased the incidence of postoperative nightmares and hallucinations [79].
It is difficult if not impossible to avoid the use of opioids altogether, and the judicious administration of short-acting opioids and/or tramadol may be necessary. Tramadol acts weakly at the μ-opioid receptors, but also acts at non-opioid receptors, resulting in inhibition of norepinephrine and serotonin reuptake. Consequently, there is a multimodal benefit of this single agent [18]. Tramadol’s weak action at the opioid receptor also diminishes the risk of addiction and other systemic side effects seen with traditional opioids use [18]. One novel treatment strategy involves administration of pain medications based on NRS scores, with non-opioids administered for scores 4 or less, tramadol for scores between 5 and 7, and oxycodone for scores between 8 and 10 [55]. In this protocol, assessment by an anesthesiologist is required if pain is refractory and dose escalation is required [55].
PCA pumps are commonly used as postoperative adjuncts in the first 12–24 h. Following this initial period, the patient is transitioned to oral medications with dosing determined by the amount and frequency of PCA use [14]. However, PCA usage has been reported to be associated with increased total opioid use and increased adverse events [80]. Interestingly, PCA use has also been associated with equivalent or even worse postoperative pain control compared to MMA [80, 81], which suggests careful consideration for their inclusion in an ERAS protocol. When implemented, there should always be a plan for early discontinuation and transition to oral therapy [82].
An essential component of ERAS protocols is early mobilization, referring to mobilization on the day of surgery or the first postoperative day thereafter. The adverse effects of bed rest and immobilization are well-documented, in particular the elevated risk of deconditioning, cardiopulmonary events, and thromboembolism [83,84,85]. Across multiple disciplines, the benefits of early mobilization are apparent in reducing the postoperative LOS, and also as measured by an overall reduction in rates of infection, respiratory compromise, thromboembolic events, and sepsis [86]. Few studies have specifically investigated the impact of early mobilization following spine surgery; however, these studies have uniformly identified improvements in rates of perioperative complications and LOS [87,88,89,90]. Accordingly, they have been adopted with enthusiasm into spine ERAS protocols [91]. Equally important considerations are involvement with physical and occupational therapy during an inpatient admission [52, 55, 92], and to continue physical therapy on an outpatient basis.
Outcomes by Type of Spine Surgery
Few publications on ERAS and spine surgery were available prior to 2018, and the past few years have seen an exponential increase in interest and published protocols. These protocols are highly variable and tend to be institutional-specific, but have generally focused on providing improved education, early nutrition and mobilization, multimodal pain management, and a general trend toward a transition to minimally invasive techniques. These protocols have been studied in a variety of spine surgeries, from simple decompressive laminectomies to more extensive tumor and fusion surgeries. The majority of currently reported protocols are focused on lumbar surgeries. Regardless of the specific ERAS elements, nearly all studies have reported beneficial effects, often related to decreased LOS and reduced opioid usage, without an increase in complications or readmissions [93]. Table 6.1 comprehensively outlines selected studies that evaluate ERAS protocols compared to cohorts with conventional care.
Surgery by Anatomical Level
ERAS protocols have been studied in cervical spine patients [53, 99,100,101,102,103,104], although many studies report their outcomes in mixed cohorts with other spine surgery procedures. In the study described by Soffin et al., 33 patients underwent with anterior cervical discectomy and fusion or cervical arthroplasty, with each receiving an average of 18 ERAS elements. Patients were found to have minimal complications and no readmissions after 90 days [114]. In another study reported by Debono et al., two groups of patients undergoing anterior cervical discectomy and fusion were compared before and after ERAS implementation, without increased complications and with a significantly decreased LOS [100]. Sivaganesan et al. reported on pre- and post-protocol implementation results in elective degenerative spine surgeries; although there was a significant reduction in LOS with fewer 90-day complications, a subgroup analysis of cervical spine patients showed no significant changes [103]. Venkata and van Dellen also described the implementation of an ERAS protocol centered on early mobilization, opioid use reduction, patient counseling, and reduction of drains. This was a mixed cohort of lumbar and cervical patients, with the majority having undergone non-instrumented lumbar decompression surgeries. Logistic regression models showed no influence on LOS by the type of surgery performed [115]. Other similar studies with mixed cohorts of anatomical levels reported overall cost reductions or improvements in LOS [53, 101].
Lumbar spine surgery accounts for the majority of published ERAS protocols [55, 65, 82, 105,106,107,108,109,110,111,112,113]. An early fast-track protocol for lumbar spine surgery was reported by Scanlon and Richards in 2004 [116]. In this “same day laminectomy program,” patients aged 55 years or less without chronic comorbidities were subjected to a protocol that primarily involved a change in anesthesia from propofol for pentathol, the omission of long-acting muscle relaxants, and early postoperative mobilization. No preoperative changes were made. With their sample of 27 patients that were able to be discharged on the same day of surgery, they estimated an elimination of 54 hospital days and cost savings of $111,420 in costs for the hospital [116]. ERAS protocols have also been evaluated specifically in lumbar fusion surgeries. In a retrospective study, Bradywood et al. found that lumbar fusion patients who entered into a standardized care pathway had significantly decreased median LOS, with a higher percentage of patients discharged home compared to prior to implementation (75% vs. 64%) [82]. No significant differences were identified in pain scores, readmission rates, or falls between groups. In another retrospective study, Wang et al. evaluated their ERAS protocol in elderly patients undergoing one- or two-level lumbar fusions and also found an overall decreased LOS [68].
Opioid consumption following spine surgery is an important consideration that has been evaluated in a few studies. In a prospective controlled study of predominantly thoracolumbar elective spine surgery, Ali et al. compared opioid consumption, pain scores, LOS, and readmission rates [106]. The ERAS protocol that was used involved preoperative education and a carbohydrate load, as well as evaluation by various consultants including nutrition, endocrinology, sleep medicine, and pain management for evaluation and optimization if necessary. Although there was no change in the LOS, the ERAS group had significantly less opioid use immediately postoperatively and at 1 month [106]. This reduction was seen at 3 and 6 months in a follow-up publication [117]. Patients with opioid use disorders undergoing open lumbar surgery have been reported to have increased complications and overall hospitals costs, suggesting that this patient population could benefit from specialized ERAS protocols [118].
Some studies have looked at removing opioids entirely from the intraoperative period. This strategy, known as opioid-free anesthesia, does not allow for systemic, neuraxial, or tissue infiltration with opioids. In a single-surgeon series of MIS lumbar surgeries, patients who underwent such a protocol within an established ERAS pathway did not demonstrate an increase in postoperative pain compared to patients who were treated with a standard ERAS pathway using opioids [55]. Although this study is limited by its sample size, it represents a promising avenue for research and treatment.
Minimally Invasive Surgery
One of the most important innovations in spine surgery has been the proliferation of MIS techniques. Compared to conventional open spinal surgery, MIS techniques often involve smaller incisions, the use of the microscope, endoscope or tubular working channels, and implantation of expandable cages and percutaneous screws. Previous studies have demonstrated that posterior lumbar interbody fusion or transforaminal lumbar interbody fusion (TLIF) procedures done in a MIS fashion reduce both blood loss and LOS compared to open surgery [119, 120]. Despite these apparent benefits, clinical outcomes following MIS procedures are generally equivalent to open procedures [9, 121, 122]. However, multiple studies have reported clear advantages with MIS techniques including fewer postoperative infections [123], reduced opioid consumption [124], improved mobilization [125], and reduced hospital costs [105, 121, 122, 126]. As such, the true value of MIS techniques may be seen when incorporated into a rigorous ERAS framework.
Chang et al. compared endoscopic discectomy with an expandable cage to a standard MIS dissection using a microscope, and reported reduced opioid utilization and LOS in the endoscopic ERAS group [127]. Other major components of this ERAS protocol included IV sedation without intubation, and injection of liposomal bupivacaine. Similarly, Wang et al. found decreased LOS and blood loss in patients undergoing endoscopic MIS TLIF as compared to standard MIS TLIF [105]. There was also a significant cost reduction in the endoscopic ERAS group of 15.2%, approximately $3444, compared to the traditional group [105].
In ERAS protocols where MIS techniques are employed, much of the benefit is attributed to the change in surgical technique; however, that is not to say other ERAS elements are less influential. Feng et al. compared the implementation of an ERAS protocol for MIS TLIF to a historical cohort without an ERAS protocol, and without modification of the surgical technique used between groups [110]. Based on the implementation of 11 ERAS components, there was a significant reduction in LOS, blood loss, cost, and complications [110].
Deformity Surgery
ERAS pathways in fusion for the treatment of adolescent idiopathic scoliosis (AIS) have been studied by multiple groups [94,95,96,97,98]. These complex surgeries often lead to prolonged hospital courses, which is why ERAS protocols may be especially useful in this population. Muhly et al. formalized an accelerated recovery pathway with a focus on MMA, early mobilization and nutrition, and studied outcomes prior to protocol initialization, during the time of transition, and post-protocol [96]. Compared with pre-protocol patients, the LOS was significantly reduced without an increase in readmission rates, and pain in the early postoperative period was significantly reduced. Gornitzky et al. also emphasized the utilization of MMA in the perioperative and postoperative period, demonstrating a 31% reduction in inpatient hospitalization and a 34% decrease in PCA usage [95].
Sanders et al. employed a comprehensive ERAS protocol for AIS and noted a decrease in postoperative hospitalization costs [128]. This protocol utilized preoperative education, early mobilization, drain removal, and nutrition, along with early transition to oral pain medications. With this decrease in hospital usage, there was a decrease in average cost decrease by 22%, from $23,640 to $18,360. There was no increase in rate of complications despite the early discharge [128]. Fletcher et al. also emphasized early mobilization, nutrition, and drain removal following AIS surgery, and reported earlier discharge with a 33% decrease in average costs, and without an increase in the rate of complications [94]. However, the accelerated and standard discharge groups had some notable differences, including a significantly higher utilization of implants and pedicle screws in the standard group. The same group evaluated their pathway in a subsequent publication, reporting a 48% reduction in LOS [97].
Tumor Surgery
ERAS programs lend themselves to improving outcomes in high-risk populations, such as patients with cancer. Grasu et al. devised an ERAS protocol that focused on preconditioning, decreased fasting time, MMA, MIS surgical techniques, and early postoperative mobilization for patients with metastatic spinal tumors [52]. Surgeries ranged from simple decompressions to vertebrectomies. All surgeries were done in an elective manner, and emergency cases were excluded. Both control and ERAS groups had similar preoperative characteristics with similar pain scores, although the tumor location and primary tumor origin were heterogeneous. Patients in the ERAS group trended toward better postoperative pain control and a decrease in opioid consumption; however, there was no difference in LOS, readmission rates, or complications between the two groups [52].
Outcomes in Elderly Patients
Spine surgery in the aging population is becoming an increasingly relevant topic for neurosurgery as the global population of geriatric adults increases. The United Nation’s 2017 World Population Aging Report found that, from 1980 to 2017, the number of adults above age 60 doubled, to increase to an estimated 2.1 billion adults by 2050 [129]. These population trends are starting to be seen in elective spine surgery as well; population data from 2004 to 2015 indicate that the number of elective lumbar fusions increased by 138.7% in patients older than 65 years [27]. Though the literature is sparse, there are definite considerations and potential for specialized protocols, including ERAS protocols, to better address the needs of elderly patients undergoing spine surgery.
Important initial considerations when considering spine surgery in the elderly are to clarify the goal of surgery and perform the proper preoperative evaluation. It has been reported that the goals of the elderly patient are more focused on being pain-free, maintaining mobility, and maintaining the ability to live independently [130]. The preoperative evaluation of geriatric patients should take into consideration patient quality of life and the various “geriatric syndromes,” and how they contribute to the overall health and ability to undergo spine surgery. Geriatric syndrome is a term used to describe a set of diseases that are common to geriatric patients although are not necessarily linked physiologically to a specific organ system [131], and include diverse pathologies such as osteoporosis, sarcopenia, malnutrition, disability, decubitus ulcers, delirium, cognitive impairment, and a propensity for falls [129].
There have been several attempts to provide a comprehensive geriatric assessment that incorporates geriatric syndromes and frailty, and can be used to aid preoperative assessment. One such assessment is the Canadian Study of Health and Aging Frailty Index (CSHA-FI) [132], which evaluates 70 variables to measure the accumulative deficits with regard to physical, cognitive, functional, and social domains—this is a comprehensive assessment and is thus time-consuming to administer. A modified version of the CSHA-FI assessment using less variables has been termed the “modified frailty index” [133], and has been applied to predicting morbidity and mortality from spine surgery [134, 135]. In the study reported by Leven et al., patients in the oldest age group (mean of 72 ± 8.3 years) were more likely to have a higher frailty index than younger patients—this was an independent predictor of postoperative complications (need for blood transfusions, thromboembolic events, etc.), mortality, LOS, and reoperations in patients that underwent spinal fusion procedures [134]. As such, it is clear from these studies that the elderly are a vulnerable surgical population and that the development of protocols geared toward their needs is increasingly necessary in spine surgery.
The goals of ERAS protocols are to reduce the surgical stress response and minimize postoperative complications for patients. Although the role of geriatric risk factors in spinal surgery is understudied, most data on elderly patients are intermixed with younger patients, and studies evaluating the effects of ERAS protocols on elderly spine surgery patients are lacking. Few such studies have been published [104, 112]; although the protocols have different specifications, there are many commonalities geared toward meeting the needs of geriatric patients, such as preoperative education and counseling, minimizing prolonged preoperative fasting, early ambulation and oral feedings, and using multimodal perioperative analgesia.
Ifrach et al. examined the efficacy of an ERAS pathway in elderly patients undergoing elective laminectomy, discectomy, foraminotomy, thoracolumbosacral fusion, cervicothoracic fusion, and anterior cervical discectomy and fusion [104]. Relevant pain outcomes included self-reported opioid use at 1 and 3 months and patient-reported pain scores. The preoperative ERAS phase included written educational materials, smoking cessation, and the incorporation consults focused on nutrition, sleep medicine, pain, and endocrinology. Perioperative initiatives included a carbohydrate drink and gabapentin therapy. Postoperative medications included acetaminophen, ketorolac, and muscle relaxants, and limiting opioids for breakthrough pain to only postoperative day 1. Other initiatives included early ambulation, starting thromboembolism prophylaxis on day 1, and follow-up with the patient’s primary care physician within 2 weeks. These patients had a significant reduction in 1-month and 3-month self-reported narcotic use without an increase in patient-reported pain scores. Reduction of opioid use in elderly patients is an important topic, as these patients are often subject to polypharmacy due to their multiple medical comorbidities.
Wang et al. studied the efficacy of an ERAS protocol in patients 65 years and older who had lumbar disc herniations or spinal stenosis, requiring one- or two-level lumbar fusion [112]. This retrospective study examined whether such protocols affected complications, LOS, postoperative pain scores, and 30-day readmission rates, compared to a historical cohort of patients who did not receive an ERAS protocol. Preoperative initiatives included patient education and counseling, limiting preoperative fasting, fluid and carbohydrate loading, antibiotic therapy, and antithrombotic stocking. Intraoperative initiatives included the use of tranexamic acid to decrease blood loss, maintaining normothermia, and the use of local analgesia. Postoperative initiatives included early ambulation, transition to oral feeding, early removal of urinary catheters, and multimodal analgesia. Compliance rates to the ERAS protocol were 92.1%, with the poorest compliance reported with discontinuation of the urinary catheter (52.6% of patients). Overall, no differences were identified between ERAS and non-ERAS protocol patients in the number of complications or mortality rates, nor were there differences in validated outcome metrics including the Japanese Orthopaedic Association score, visual analog scale, or Oswestry Disability Index. However, there was a significant decrease in the LOS for patients in the ERAS group (12.30 ± 3.03 days vs. 15.50 ± 1.88 days). Compliance with an ERAS protocol is closely associated with prognosis; in one retrospective study of elderly patients undergoing lumbar fusion, older patients were less compliant with the protocol, and had a higher incidence of complications and a longer LOS [136].
Elderly patients represent an increasing proportion of patients with degenerative spine disease who will require surgical treatment when conservative measures fail. Their goals of surgery are often different than their younger counterparts, and are focused on their ability to maintain independence and mobility. Chakravarty et al. described an ERAS protocol used at Cleveland Clinic that included referral of all elective spine surgery patients over the age of 75 to geriatricians for frailty assessment and adequate time for optimization and prehabilitation [92]. Further study into the benefits of tailored preoperative optimization and surgical treatments aimed toward the geriatric population is needed, such as MIS procedures which generally have less blood loss and shorter LOS. Elderly patients are a vulnerable population that could benefit from tailored, multidisciplinary ERAS protocols to optimize their surgical treatment, including involvement of geriatricians, nutritionists, pain management specialists, and anesthesiologists.
ERAS Implementation
Ultimately, thoughtful delivery of an ERAS protocol for perioperative spinal surgery requires a multidisciplinary, team-based approach. This should be specific to each institution to appropriately address the needs of the patient population by incorporating readily available resources. For instance, employing a preoperative ERAS protocol with “prehabilitation,” optimization of medical comorbidities, and timely administration of gabapentinoids may necessitate the involvement of departmental nurses, nurse practitioners, physical therapists, and referring physicians. Similarly, open dialogue with the anesthesia team and operating room staff may be critical for intraoperative ERAS strategies. Postoperative ERAS implementation using pain management algorithms and early mobilization may require the development of a detailed postoperative order sets for residents, physician extenders, and hospitalists. Additionally educational materials, team-based meetings, and open communication with patients, nurses, nutritionists, physical therapists, and consulting physicians will reduce errors and unify messaging. All of this is essential to enhance both recovery after surgery and the patient experience.
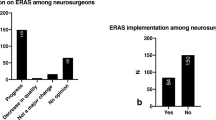
Despite the general consensus that ERAS protocols are beneficial in spine surgery, they are not universally embraced. In a multinational survey of spine surgeons, less than half of respondents were familiar with ERAS as a concept, with only about one-third utilizing ERAS protocols in their own practice [137]. Spine surgery is heterogeneous and multiple options are available for even a single pathology; therefore, no single protocol is universally applicable, making widespread utilization difficult to achieve. As the spine-specific ERAS literature becomes more robust, protocols will become more established and utilization will undoubtedly increase.
Conclusion
Due to the high level of variability and the number of simultaneous changes made in implementing ERAS protocols, the direct effect of any specific change is difficult to ascertain. A recent systematic review of the published literature from 2004 to 2019 regarding multimodality ERAS in adult elective spine surgery identified a variety of protocols, with the most common implementation being preoperative education and peri- and postoperative MMA [11]. Half of the included studies found a significant reduction in LOS, with no study identifying a worse outcome after implementation of an ERAS protocol [11].
The principle of ERAS is based on the synergistic effects of a multimodal approach in caring for a patient from the preoperative to the postoperative phase, with a focus on a multidisciplinary approach in improving surgical outcome and patient satisfaction. A single change alone would not necessarily qualify as an ERAS framework. In general, despite the wide variability in protocol elements and patient populations, ERAS protocols are associated with decreased LOS without any additional complications or readmission rates. Future research and implementation should focus on optimizations that may benefit specific surgical procedures or patient populations.
References
Gan TJ, Habib AS, Miller TE, White W, Apfelbaum JL. Incidence, patient satisfaction, and perceptions of post-surgical pain: results from a US national survey. Curr Med Res Opin. 2014;30(1):149–60. Epub 2013/11/19.
Joshi GP, Beck DE, Emerson RH, Halaszynski TM, Jahr JS, Lipman AG, et al. Defining new directions for more effective management of surgical pain in the United States: highlights of the inaugural Surgical Pain Congress. Am Surg. 2014;80(3):219–28. Epub 2014/03/29.
Ljungqvist O, Scott M, Fearon KC. Enhanced recovery after surgery: a review. JAMA Surg. 2017;152(3):292–8. Epub 2017/01/18.
Fearon KC, Ljungqvist O, Von Meyenfeldt M, Revhaug A, Dejong CH, Lassen K, et al. Enhanced recovery after surgery: a consensus review of clinical care for patients undergoing colonic resection. Clin Nutr. 2005;24(3):466–77. Epub 2005/05/18.
Engelman RM, Rousou JA, Flack JE III, Deaton DW, Humphrey CB, Ellison LH, et al. Fast-track recovery of the coronary bypass patient. Ann Thorac Surg. 1994;58(6):1742–6. Epub 1994/12/01.
Bardram L, Funch-Jensen P, Jensen P, Crawford ME, Kehlet H. Recovery after laparoscopic colonic surgery with epidural analgesia, and early oral nutrition and mobilisation. Lancet. 1995;345(8952):763–4. Epub 1995/03/25.
Kehlet H, Mogensen T. Hospital stay of 2 days after open sigmoidectomy with a multimodal rehabilitation programme. Br J Surg. 1999;86(2):227–30. Epub 1999/04/01.
Kehlet H. Multimodal approach to control postoperative pathophysiology and rehabilitation. Br J Anaesth. 1997;78(5):606–17. Epub 1997/05/01.
Wainwright TW, Immins T, Middleton RG. Enhanced recovery after surgery (ERAS) and its applicability for major spine surgery. Best Pract Res Clin Anaesthesiol. 2016;30(1):91–102. Epub 2016/04/03.
Elsarrag M, Soldozy S, Patel P, Norat P, Sokolowski JD, Park MS, et al. Enhanced recovery after spine surgery: a systematic review. Neurosurg Focus. 2019;46(4):E3. Epub 2019/04/02.
Tong Y, Fernandez L, Bendo JA, Spivak JM. Enhanced recovery after surgery trends in adult spine surgery: a systematic review. Int J Spine Surg. 2020;14(4):623–40. Epub 2020/09/29.
Gerbershagen HJ, Aduckathil S, van Wijck AJ, Peelen LM, Kalkman CJ, Meissner W. Pain intensity on the first day after surgery: a prospective cohort study comparing 179 surgical procedures. Anesthesiology. 2013;118(4):934–44. Epub 2013/02/09.
Devin CJ, McGirt MJ. Best evidence in multimodal pain management in spine surgery and means of assessing postoperative pain and functional outcomes. J Clin Neurosci. 2015;22(6):930–8. Epub 2015/03/15.
Puvanesarajah V, Liauw JA, Lo SF, Lina IA, Witham TF, Gottschalk A. Analgesic therapy for major spine surgery. Neurosurg Rev. 2015;38(3):407–18; discussion 19. Epub 2015/02/15.
Kehlet H, Jensen TS, Woolf CJ. Persistent postsurgical pain: risk factors and prevention. Lancet. 2006;367(9522):1618–25. Epub 2006/05/16.
Svensson GL, Lundberg M, Ostgaard HC, Wendt GK. High degree of kinesiophobia after lumbar disc herniation surgery: a cross-sectional study of 84 patients. Acta Orthop. 2011;82(6):732–6. Epub 2011/11/10.
Archer KR, Seebach CL, Mathis SL, Riley LH III, Wegener ST. Early postoperative fear of movement predicts pain, disability, and physical health six months after spinal surgery for degenerative conditions. Spine J. 2014;14(5):759–67. Epub 2013/11/12.
Wick EC, Grant MC, Wu CL. Postoperative multimodal analgesia pain management with nonopioid analgesics and techniques: a review. JAMA Surg. 2017;152(7):691–7. Epub 2017/06/01.
Bartels K, Mayes LM, Dingmann C, Bullard KJ, Hopfer CJ, Binswanger IA. Opioid use and storage patterns by patients after hospital discharge following surgery. PLoS One. 2016;11(1):e0147972. Epub 2016/01/30.
Dunn LK, Yerra S, Fang S, Hanak MF, Leibowitz MK, Tsang S, et al. Incidence and risk factors for chronic postoperative opioid use after major spine surgery: a cross-sectional study with longitudinal outcome. Anesth Analg. 2018;127(1):247–54. Epub 2018/03/24.
Samuel AM, Lovecchio FC, Premkumar A, Louie PK, Vaishnav AS, Iyer S, et al. Preoperative use of higher-strength opioids has a dose-dependent association with reoperations after lumbar decompression and interbody fusion surgery. Spine. 2020;46:E203. Epub 2020/10/21.
Chau DL, Walker V, Pai L, Cho LM. Opiates and elderly: use and side effects. Clin Interv Aging. 2008;3(2):273–8. Epub 2008/08/09.
Becker WC, O’Connor PG. The safety of opioid analgesics in the elderly: new data raise new concerns: comment on “The comparative safety of opioids for nonmalignant pain in older adults”. Arch Intern Med. 2010;170(22):1986–8. Epub 2010/12/15.
Le Roux C, Tang Y, Drexler K. Alcohol and opioid use disorder in older adults: neglected and treatable illnesses. Curr Psychiatry Rep. 2016;18(9):87. Epub 2016/08/05.
Carter MW, Yang BK, Davenport M, Kabel A. Increasing rates of opioid misuse among older adults visiting emergency departments. Innov Aging. 2019;3(1):igz002. Epub 2019/03/14.
Cublier M, Menecier P, von Gunten A, Lepetit A, Dorey JM, Rolland B. Mesusage d’antalgiques opioides chez le sujet age : une mise au point [Analgesic opioids misuse in the elderly: a review]. Geriatr Psychol Neuropsychiatr Vieil. 2020;18(3):343–9. Epub 2020/08/08.
Martin BI, Mirza SK, Spina N, Spiker WR, Lawrence B, Brodke DS. Trends in lumbar fusion procedure rates and associated hospital costs for degenerative spinal diseases in the United States, 2004 to 2015. Spine. 2019;44(5):369–76. Epub 2018/08/04.
Rajaee SS, Bae HW, Kanim LE, Delamarter RB. Spinal fusion in the United States: analysis of trends from 1998 to 2008. Spine. 2012;37(1):67–76. Epub 2011/02/12.
Yoshihara H, Yoneoka D. National trends in the surgical treatment for lumbar degenerative disc disease: United States, 2000 to 2009. Spine J. 2015;15(2):265–71. Epub 2014/10/05.
Deyo RA, Mirza SK, Martin BI, Kreuter W, Goodman DC, Jarvik JG. Trends, major medical complications, and charges associated with surgery for lumbar spinal stenosis in older adults. JAMA. 2010;303(13):1259–65. Epub 2010/04/08.
Gruskay JA, Fu M, Bohl DD, Webb ML, Grauer JN. Factors affecting length of stay after elective posterior lumbar spine surgery: a multivariate analysis. Spine J. 2015;15(6):1188–95. Epub 2013/11/05.
Wang MY, Chang P, Grossman J. Development of an Enhanced Recovery After Surgery (ERAS) approach for lumbar spinal fusion. J Neurosurg Spine. 2017;26(4):411–8. Epub 2016/12/24.
Ren L, Zhu D, Wei Y, Pan X, Liang L, Xu J, et al. Enhanced Recovery After Surgery (ERAS) program attenuates stress and accelerates recovery in patients after radical resection for colorectal cancer: a prospective randomized controlled trial. World J Surg. 2012;36(2):407–14. Epub 2011/11/22.
Burgess LC, Arundel J, Wainwright TW. The effect of preoperative education on psychological, clinical and economic outcomes in elective spinal surgery: a systematic review. Healthcare. 2019;7(1):48. Epub 2019/03/25.
Fleege C, Arabmotlagh M, Almajali A, Rauschmann M. Pra- und postoperative Fast-track-Behandlungskonzepte in der Wirbelsaulenchirurgie : Patienteninformation und Patientenkooperation [Pre- and postoperative fast-track treatment concepts in spinal surgery: patient information and patient cooperation]. Der Orthopade 2014;43(12):1062–4, 6–9. Epub 2014/11/13.
Gometz A, Maislen D, Youtz C, Kary E, Gometz EL, Sobotka S, et al. The effectiveness of prehabilitation (Prehab) in both functional and economic outcomes following spinal surgery: a systematic review. Cureus. 2018;10(5):e2675. Epub 2018/07/28.
Golinvaux NS, Varthi AG, Bohl DD, Basques BA, Grauer JN. Complication rates following elective lumbar fusion in patients with diabetes: insulin dependence makes the difference. Spine. 2014;39(21):1809–16. Epub 2014/07/11.
Klemencsics I, Lazary A, Szoverfi Z, Bozsodi A, Eltes P, Varga PP. Risk factors for surgical site infection in elective routine degenerative lumbar surgeries. Spine J. 2016;16(11):1377–83. Epub 2016/08/16.
Durand WM, DePasse JM, Bokshan SL, Eltorai AEM, Daniels AH. Tobacco use and complications following spinal fusion: a comparison of the national surgical quality improvement program and national inpatient sample datasets. World Neurosurg. 2019;123:e393–407. Epub 2018/12/01.
Wilson JRF, Jiang F, Badhiwala JH, Shaffrey CI, Carreon LY, Cheung KMC, et al. The effect of tobacco smoking on adverse events following adult complex deformity surgery: analysis of 270 patients from the prospective, multicenter scoli-RISK-1 study. Spine. 2020;45(1):32–7. Epub 2019/08/16.
Klein JD, Hey LA, Yu CS, Klein BB, Coufal FJ, Young EP, et al. Perioperative nutrition and postoperative complications in patients undergoing spinal surgery. Spine. 1996;21(22):2676–82. Epub 1996/11/15.
Phan K, Kim JS, Xu J, Di Capua J, Lee NJ, Kothari P, et al. Nutritional insufficiency as a predictor for adverse outcomes in adult spinal deformity surgery. Glob Spine J. 2018;8(2):164–71. Epub 2018/04/18.
Cai Y, Xu K, Hou W, Yang Z, Xu P. Preoperative chlorhexidine reduces the incidence of surgical site infections in total knee and hip arthroplasty: a systematic review and meta-analysis. Int J Surg. 2017;39:221–8. Epub 2017/02/13.
Chan AK, Ammanuel SG, Chan AY, Oh T, Skrehot HC, Edwards CS, et al. Chlorhexidine showers are associated with a reduction in surgical site infection following spine surgery: an analysis of 4266 consecutive surgeries. Neurosurgery. 2019;85(6):817–26. Epub 2018/12/28.
Pandey CK, Navkar DV, Giri PJ, Raza M, Behari S, Singh RB, et al. Evaluation of the optimal preemptive dose of gabapentin for postoperative pain relief after lumbar diskectomy: a randomized, double-blind, placebo-controlled study. J Neurosurg Anesthesiol. 2005;17(2):65–8. Epub 2005/04/21.
Khurana G, Jindal P, Sharma JP, Bansal KK. Postoperative pain and long-term functional outcome after administration of gabapentin and pregabalin in patients undergoing spinal surgery. Spine. 2014;39(6):E363–8. Epub 2014/01/05.
Yu L, Ran B, Li M, Shi Z. Gabapentin and pregabalin in the management of postoperative pain after lumbar spinal surgery: a systematic review and meta-analysis. Spine. 2013;38(22):1947–52. Epub 2013/08/08.
Rivkin A, Rivkin MA. Perioperative nonopioid agents for pain control in spinal surgery. Am J Health Syst Pharm. 2014;71(21):1845–57. Epub 2014/10/17.
Sucato DJ, Lovejoy JF, Agrawal S, Elerson E, Nelson T, McClung A. Postoperative ketorolac does not predispose to pseudoarthrosis following posterior spinal fusion and instrumentation for adolescent idiopathic scoliosis. Spine. 2008;33(10):1119–24. Epub 2008/05/02.
Dodwell ER, Latorre JG, Parisini E, Zwettler E, Chandra D, Mulpuri K, et al. NSAID exposure and risk of nonunion: a meta-analysis of case-control and cohort studies. Calcif Tissue Int. 2010;87(3):193–202. Epub 2010/06/17.
Kim SI, Ha KY, Oh IS. Preemptive multimodal analgesia for postoperative pain management after lumbar fusion surgery: a randomized controlled trial. Eur Spine J. 2016;25(5):1614–9. Epub 2015/09/02.
Grasu RM, Cata JP, Dang AQ, Tatsui CE, Rhines LD, Hagan KB, et al. Implementation of an Enhanced Recovery After Spine Surgery program at a large cancer center: a preliminary analysis. J Neurosurg Spine. 2018;29(5):588–98. Epub 2018/08/18.
Carr DA, Saigal R, Zhang F, Bransford RJ, Bellabarba C, Dagal A. Enhanced perioperative care and decreased cost and length of stay after elective major spinal surgery. Neurosurg Focus. 2019;46(4):E5. Epub 2019/04/02.
Arumugam S, Lau CS, Chamberlain RS. Use of preoperative gabapentin significantly reduces postoperative opioid consumption: a meta-analysis. J Pain Res. 2016;9:631–40. Epub 2016/09/28.
Soffin EM, Wetmore DS, Beckman JD, Sheha ED, Vaishnav AS, Albert TJ, et al. Opioid-free anesthesia within an enhanced recovery after surgery pathway for minimally invasive lumbar spine surgery: a retrospective matched cohort study. Neurosurg Focus. 2019;46(4):E8. Epub 2019/04/02.
Staartjes VE, de Wispelaere MP, Schroder ML. Improving recovery after elective degenerative spine surgery: 5-year experience with an enhanced recovery after surgery (ERAS) protocol. Neurosurg Focus. 2019;46(4):E7. Epub 2019/04/02.
Loftus RW, Yeager MP, Clark JA, Brown JR, Abdu WA, Sengupta DK, et al. Intraoperative ketamine reduces perioperative opiate consumption in opiate-dependent patients with chronic back pain undergoing back surgery. Anesthesiology. 2010;113(3):639–46. Epub 2010/08/10.
Tsaousi GG, Pourzitaki C, Aloisio S, Bilotta F. Dexmedetomidine as a sedative and analgesic adjuvant in spine surgery: a systematic review and meta-analysis of randomized controlled trials. Eur J Clin Pharmacol. 2018;74(11):1377–89. Epub 2018/07/17.
Wang F, Shi K, Jiang Y, Yang Z, Chen G, Song K. Intravenous glucocorticoid for pain control after spinal fusion: a meta-analysis of randomized controlled trials. Medicine. 2018;97(20):e10507. Epub 2018/05/17.
Nielsen RV, Siegel H, Fomsgaard JS, Andersen JD, Martusevicius R, Mathiesen O, et al. Preoperative dexamethasone reduces acute but not sustained pain after lumbar disk surgery: a randomized, blinded, placebo-controlled trial. Pain. 2015;156(12):2538–44. Epub 2015/08/14.
Zorrilla-Vaca A, Healy RJ, Mirski MA. A Comparison of regional versus general anesthesia for lumbar spine surgery: a meta-analysis of randomized studies. J Neurosurg Anesthesiol. 2017;29(4):415–25. Epub 2016/08/27.
Ong CK, Lirk P, Seymour RA, Jenkins BJ. The efficacy of preemptive analgesia for acute postoperative pain management: a meta-analysis. Anesth Analg. 2005;100(3):757–73, table of contents. Epub 2005/02/25.
Perera AP, Chari A, Kostusiak M, Khan AA, Luoma AM, Casey ATH. Intramuscular local anesthetic infiltration at closure for postoperative analgesia in lumbar spine surgery: a systematic review and meta-analysis. Spine. 2017;42(14):1088–95. Epub 2016/01/13.
Roh MS, Kucher OA, Shick KM, Knolhoff DR, McGarvey JS, Peterson SC. Intramuscular liposomal bupivacaine decreases length of stay and opioid usage following lumbar spinal fusion. Clin Spine Surg. 2020;33(8):E359–E63. Epub 2020/05/20.
Brusko GD, Kolcun JPG, Heger JA, Levi AD, Manzano GR, Madhavan K, et al. Reductions in length of stay, narcotics use, and pain following implementation of an enhanced recovery after surgery program for 1- to 3-level lumbar fusion surgery. Neurosurg Focus. 2019;46(4):E4. Epub 2019/04/02.
Pendi A, Acosta FL, Tuchman A, Movahedi R, Sivasundaram L, Arif I, et al. Intrathecal morphine in spine surgery: a meta-analysis of randomized controlled trials. Spine. 2017;42(12):E740–E7. Epub 2017/04/20.
Dhaliwal P, Yavin D, Whittaker T, Hawboldt GS, Jewett GAE, Casha S, et al. Intrathecal morphine following lumbar fusion: a randomized, placebo-controlled trial. Neurosurgery. 2019;85(2):189–98. Epub 2018/09/29.
Wang Y, Guo X, Guo Z, Xu M. Preemptive analgesia with a single low dose of intrathecal morphine in multilevel posterior lumbar interbody fusion surgery: a double-blind, randomized, controlled trial. Spine J. 2020;20(7):989–97. Epub 2020/03/18.
Guest JD, Vanni S, Silbert L. Mild hypothermia, blood loss and complications in elective spinal surgery. Spine J. 2004;4(2):130–7. Epub 2004/03/16.
Basques BA, Anandasivam NS, Webb ML, Samuel AM, Lukasiewicz AM, Bohl DD, et al. Risk factors for blood transfusion with primary posterior lumbar fusion. Spine. 2015;40(22):1792–7. Epub 2015/07/15.
Bacchin MR, Ceria CM, Giannone S, Ghisi D, Stagni G, Greggi T, et al. Goal-directed fluid therapy based on stroke volume variation in patients undergoing major spine surgery in the prone position: a cohort study. Spine. 2016;41(18):E1131–7. Epub 2016/04/06.
Hui S, Xu D, Ren Z, Chen X, Sheng L, Zhuang Q, et al. Can tranexamic acid conserve blood and save operative time in spinal surgeries? A meta-analysis. Spine J. 2018;18(8):1325–37. Epub 2017/12/17.
Mikhail C, Pennington Z, Arnold PM, Brodke DS, Chapman JR, Chutkan N, et al. Minimizing blood loss in spine surgery. Glob Spine J. 2020;10(1 Suppl):71S–83S. Epub 2020/01/15.
Yao R, Zhou H, Choma TJ, Kwon BK, Street J. Surgical site infection in spine surgery: who is at risk? Glob Spine J. 2018;8(4 Suppl):5S–30S. Epub 2018/12/24.
Pennington Z, Lubelski D, Molina C, Westbroek EM, Ahmed AK, Sciubba DM. Prolonged post-surgical drain retention increases risk for deep wound infection after spine surgery. World Neurosurg. 2019;130:e846–e53. Epub 2019/07/12.
Weesie YM, Hek K, Schermer TRJ, Schellevis FG, Leufkens HGM, Rook EJ, et al. Use of opioids increases with age in older adults: an observational study (2005-2017). Front Pharmacol. 2020;11:648. Epub 2020/06/02.
Park PJ, Makhni MC, Cerpa M, Lehman RA, Lenke LG. The role of perioperative ketamine in postoperative pain control following spinal surgery. J Spine Surg. 2020;6(3):591–7. Epub 2020/10/27.
Chou R, Gordon DB, de Leon-Casasola OA, Rosenberg JM, Bickler S, Brennan T, et al. Management of postoperative pain: a clinical practice guideline from the American Pain Society, the American Society of Regional Anesthesia and Pain Medicine, and the American Society of Anesthesiologists’ Committee on Regional Anesthesia, Executive Committee, and Administrative Council. J Pain. 2016;17(2):131–57. Epub 2016/02/02.
Avidan MS, Maybrier HR, Abdallah AB, Jacobsohn E, Vlisides PE, Pryor KO, et al. Intraoperative ketamine for prevention of postoperative delirium or pain after major surgery in older adults: an international, multicentre, double-blind, randomised clinical trial. Lancet. 2017;390(10091):267–75. Epub 2017/06/04.
Rajpal S, Gordon DB, Pellino TA, Strayer AL, Brost D, Trost GR, et al. Comparison of perioperative oral multimodal analgesia versus IV PCA for spine surgery. J Spinal Disord Tech. 2010;23(2):139–45. Epub 2010/04/09.
Patel AA, Walker CT, Prendergast V, Radosevich JJ, Grimm D, Godzik J, et al. Patient-controlled analgesia following lumbar spinal fusion surgery is associated with increased opioid consumption and opioid-related adverse events. Neurosurgery. 2020;87(3):592–601. Epub 2020/05/02.
Bradywood A, Farrokhi F, Williams B, Kowalczyk M, Blackmore CC. Reduction of inpatient hospital length of stay in lumbar fusion patients with implementation of an evidence-based clinical care pathway. Spine. 2017;42(3):169–76. Epub 2016/05/24.
Allen C, Glasziou P, Del Mar C. Bed rest: a potentially harmful treatment needing more careful evaluation. Lancet. 1999;354(9186):1229–33. Epub 1999/10/16.
Haines KJ, Skinner EH, Berney S. Association of postoperative pulmonary complications with delayed mobilisation following major abdominal surgery: an observational cohort study. Physiotherapy. 2013;99(2):119–25. Epub 2012/12/12.
Aissaoui N, Martins E, Mouly S, Weber S, Meune C. A meta-analysis of bed rest versus early ambulation in the management of pulmonary embolism, deep vein thrombosis, or both. Int J Cardiol. 2009;137(1):37–41. Epub 2008/08/12.
Epstein NE. A review article on the benefits of early mobilization following spinal surgery and other medical/surgical procedures. Surg Neurol Int. 2014;5(Suppl 3):S66–73. Epub 2014/05/21.
Rupich K, Missimer E, O’Brien D, Shafer G, Wilensky EM, Pierce JT, et al. The benefits of implementing an early mobility protocol in postoperative neurosurgical spine patients. Am J Nurs. 2018;118(6):46–53. Epub 2018/05/26.
Adogwa O, Elsamadicy AA, Fialkoff J, Cheng J, Karikari IO, Bagley C. Early ambulation decreases length of hospital stay, perioperative complications and improves functional outcomes in elderly patients undergoing surgery for correction of adult degenerative scoliosis. Spine. 2017;42(18):1420–5. Epub 2017/09/14.
Nielsen PR, Jorgensen LD, Dahl B, Pedersen T, Tonnesen H. Prehabilitation and early rehabilitation after spinal surgery: randomized clinical trial. Clin Rehabil. 2010;24(2):137–48. Epub 2010/01/28.
Qvarfordh P, Olsen KS, Bendix T, Esbensen BA. Should patients walk from the postanesthesia care unit to the general ward after a lumbar discectomy? A randomized study. J Perianesth Nurs. 2014;29(5):377–84. Epub 2014/09/28.
Burgess LC, Wainwright TW. What is the evidence for early mobilisation in elective spine surgery? A narrative review. Healthcare. 2019;7(3):92. Epub 2019/07/22.
Chakravarthy VB, Yokoi H, Coughlin DJ, Manlapaz MR, Krishnaney AA. Development and implementation of a comprehensive spine surgery enhanced recovery after surgery protocol: the Cleveland Clinic experience. Neurosurg Focus. 2019;46(4):E11. Epub 2019/04/02.
Dietz N, Sharma M, Adams S, Alhourani A, Ugiliweneza B, Wang D, et al. Enhanced recovery after surgery (ERAS) for spine surgery: a systematic review. World Neurosurg. 2019;130:415–26. Epub 2019/07/06.
Fletcher ND, Shourbaji N, Mitchell PM, Oswald TS, Devito DP, Bruce RW. Clinical and economic implications of early discharge following posterior spinal fusion for adolescent idiopathic scoliosis. J Child Orthop. 2014;8(3):257–63. Epub 2014/04/29.
Gornitzky AL, Flynn JM, Muhly WT, Sankar WN. A rapid recovery pathway for adolescent idiopathic scoliosis that improves pain control and reduces time to inpatient recovery after posterior spinal fusion. Spine Deform. 2016;4(4):288–95. Epub 2016/12/09.
Muhly WT, Sankar WN, Ryan K, Norton A, Maxwell LG, DiMaggio T, et al. Rapid recovery pathway after spinal fusion for idiopathic scoliosis. Pediatrics. 2016;137(4):e20151568. Epub 2016/03/25.
Fletcher ND, Andras LM, Lazarus DE, Owen RJ, Geddes BJ, Cao J, et al. Use of a novel pathway for early discharge was associated with a 48% shorter length of stay after posterior spinal fusion for adolescent idiopathic scoliosis. J Pediatr Orthop. 2017;37(2):92–7. Epub 2015/07/28.
Rao RR, Hayes M, Lewis C, Hensinger RN, Farley FA, Li Y, et al. Mapping the road to recovery: shorter stays and satisfied patients in posterior spinal fusion. J Pediatr Orthop. 2017;37(8):e536–e42. Epub 2016/05/04.
Li J, Li H, Xv ZK, Wang J, Yu QF, Chen G, et al. Enhanced recovery care versus traditional care following laminoplasty: a retrospective case-cohort study. Medicine. 2018;97(48):e13195. Epub 2018/12/05.
Debono B, Sabatier P, Boniface G, Bousquet P, Lescure JP, Garnaud V, et al. Implementation of enhanced recovery after surgery (ERAS) protocol for anterior cervical discectomy and fusion: a propensity score-matched analysis. Eur Spine J. 2020;30:560. Epub 2020/05/16.
Dagal A, Bellabarba C, Bransford R, Zhang F, Chesnut RM, O’Keefe GE, et al. Enhanced perioperative care for major spine surgery. Spine. 2019;44(13):959–66. Epub 2019/06/18.
Debono B, Corniola MV, Pietton R, Sabatier P, Hamel O, Tessitore E. Benefits of enhanced recovery after surgery for fusion in degenerative spine surgery: impact on outcome, length of stay, and patient satisfaction. Neurosurg Focus. 2019;46(4):E6. Epub 2019/04/02.
Sivaganesan A, Wick JB, Chotai S, Cherkesky C, Stephens BF, Devin CJ. Perioperative protocol for elective spine surgery is associated with reduced length of stay and complications. J Am Acad Orthop Surg. 2019;27(5):183–9. Epub 2018/09/08.
Ifrach J, Basu R, Joshi DS, Flanders TM, Ozturk AK, Malhotra NR, et al. Efficacy of an enhanced recovery after surgery (ERAS) pathway in elderly patients undergoing spine and peripheral nerve surgery. Clin Neurol Neurosurg. 2020;197:106115. Epub 2020/08/26.
Wang MY, Chang HK, Grossman J. Reduced Acute Care Costs With the ERAS(R) minimally invasive transforaminal lumbar interbody fusion compared with conventional minimally invasive transforaminal lumbar interbody fusion. Neurosurgery. 2018;83(4):827–34. Epub 2017/09/26.
Ali ZS, Flanders TM, Ozturk AK, Malhotra NR, Leszinsky L, McShane BJ, et al. Enhanced recovery after elective spinal and peripheral nerve surgery: pilot study from a single institution. J Neurosurg Spine. 2019;30:1–9. Epub 2019/01/27.
Tarikci Kilic E, Demirbilek T, Naderi S. Does an enhanced recovery after surgery protocol change costs and outcomes of single-level lumbar microdiscectomy? Neurosurg Focus. 2019;46(4):E10. Epub 2019/04/02.
Angus M, Jackson K, Smurthwaite G, Carrasco R, Mohammad S, Verma R, et al. The implementation of enhanced recovery after surgery (ERAS) in complex spinal surgery. J Spine Surg. 2019;5(1):116–23. Epub 2019/04/30.
Smith J, Probst S, Calandra C, Davis R, Sugimoto K, Nie L, et al. Enhanced recovery after surgery (ERAS) program for lumbar spine fusion. Perioper Med. 2019;8:4. Epub 2019/06/01.
Feng C, Zhang Y, Chong F, Yang M, Liu C, Liu L, et al. Establishment and implementation of an enhanced recovery after surgery (ERAS) pathway tailored for minimally invasive transforaminal lumbar interbody fusion surgery. World Neurosurg. 2019;129:e317–e23. Epub 2019/05/28.
Heo DH, Park CK. Clinical results of percutaneous biportal endoscopic lumbar interbody fusion with application of enhanced recovery after surgery. Neurosurg Focus. 2019;46(4):E18. Epub 2019/04/02.
Wang P, Wang Q, Kong C, Teng Z, Li Z, Zhang S, et al. Enhanced recovery after surgery (ERAS) program for elderly patients with short-level lumbar fusion. J Orthop Surg Res. 2020;15(1):299. Epub 2020/08/09.
Yang Y, Wu X, Wu W, Liu Z, Pang M, Chen Y, et al. Enhanced recovery after surgery (ERAS) pathway for microendoscopy-assisted minimally invasive transforaminal lumbar interbody fusion. Clin Neurol Neurosurg. 2020;196:106003. Epub 2020/06/20.
Soffin EM, Wetmore DS, Barber LA, Vaishnav AS, Beckman JD, Albert TJ, et al. An enhanced recovery after surgery pathway: association with rapid discharge and minimal complications after anterior cervical spine surgery. Neurosurg Focus. 2019;46(4):E9. Epub 2019/04/02.
Venkata HK, van Dellen JR. A perspective on the use of an enhanced recovery program in open, non-instrumented day surgery for degenerative lumbar and cervical spinal conditions. J Neurosurg Sci. 2018;62(3):245–54. Epub 2016/04/15.
Scanlon J, Richards B. Development of a same day laminectomy program. J Perianesth Nurs. 2004;19(2):84–8. Epub 2004/04/08.
Flanders TM, Ifrach J, Sinha S, Joshi DS, Ozturk AK, Malhotra NR, et al. Reduction of postoperative opioid use after elective spine and peripheral nerve surgery using an enhanced recovery after surgery program. Pain Med. 2020;21:3283. Epub 2020/08/08.
Martini ML, Nistal DA, Deutsch BC, Caridi JM. Characterizing the risk and outcome profiles of lumbar fusion procedures in patients with opioid use disorders: a step toward improving enhanced recovery protocols for a unique patient population. Neurosurg Focus. 2019;46(4):E12. Epub 2019/04/02.
Goldstein CL, Macwan K, Sundararajan K, Rampersaud YR. Perioperative outcomes and adverse events of minimally invasive versus open posterior lumbar fusion: meta-analysis and systematic review. J Neurosurg Spine. 2016;24(3):416–27. Epub 2015/11/14.
Ge DH, Stekas ND, Varlotta CG, Fischer CR, Petrizzo A, Protopsaltis TS, et al. Comparative analysis of two transforaminal lumbar interbody fusion techniques: open TLIF versus Wiltse MIS TLIF. Spine. 2019;44(9):E555–E60. Epub 2018/10/17.
Parker SL, Mendenhall SK, Shau DN, Zuckerman SL, Godil SS, Cheng JS, et al. Minimally invasive versus open transforaminal lumbar interbody fusion for degenerative spondylolisthesis: comparative effectiveness and cost-utility analysis. World Neurosurg. 2014;82(1–2):230–8. Epub 2013/01/17.
Sulaiman WA, Singh M. Minimally invasive versus open transforaminal lumbar interbody fusion for degenerative spondylolisthesis grades 1-2: patient-reported clinical outcomes and cost-utility analysis. Ochsner J. 2014;14(1):32–7. Epub 2014/04/02.
McGirt MJ, Parker SL, Lerner J, Engelhart L, Knight T, Wang MY. Comparative analysis of perioperative surgical site infection after minimally invasive versus open posterior/transforaminal lumbar interbody fusion: analysis of hospital billing and discharge data from 5170 patients. J Neurosurg Spine. 2011;14(6):771–8. Epub 2011/03/23.
Adogwa O, Parker SL, Bydon A, Cheng J, McGirt MJ. Comparative effectiveness of minimally invasive versus open transforaminal lumbar interbody fusion: 2-year assessment of narcotic use, return to work, disability, and quality of life. J Spinal Disord Tech. 2011;24(8):479–84. Epub 2011/02/22.
Seng C, Siddiqui MA, Wong KP, Zhang K, Yeo W, Tan SB, et al. Five-year outcomes of minimally invasive versus open transforaminal lumbar interbody fusion: a matched-pair comparison study. Spine. 2013;38(23):2049–55. Epub 2013/08/22.
Singh K, Nandyala SV, Marquez-Lara A, Fineberg SJ, Oglesby M, Pelton MA, et al. A perioperative cost analysis comparing single-level minimally invasive and open transforaminal lumbar interbody fusion. Spine J. 2014;14(8):1694–701. Epub 2013/11/21.
Chang HK, Huang M, Wu JC, Huang WC, Wang MY. Less opioid consumption with enhanced recovery after surgery transforaminal lumbar interbody fusion (TLIF): a comparison to standard minimally-invasive TLIF. Neurospine. 2020;17(1):228–36. Epub 2020/04/07.
Sanders AE, Andras LM, Sousa T, Kissinger C, Cucchiaro G, Skaggs DL. Accelerated discharge protocol for posterior spinal fusion patients with adolescent idiopathic scoliosis decreases hospital postoperative charges 22. Spine. 2017;42(2):92–7. Epub 2017/01/11.
Buehring B, Barczi S. Assessing the aging patient. In: Brooks NP, Strayer AL, editors. Spine surgery in an aging population. Stuttgart: Thieme; 2019.
Nosraty L, Jylha M, Raittila T, Lumme-Sandt K. Perceptions by the oldest old of successful aging, vitality 90+ study. J Aging Stud. 2015;32:50–8. Epub 2015/02/11.
Inouye SK, Studenski S, Tinetti ME, Kuchel GA. Geriatric syndromes: clinical, research, and policy implications of a core geriatric concept. J Am Geriatr Soc. 2007;55(5):780–91. Epub 2007/05/12.
Rockwood K, Song X, MacKnight C, Bergman H, Hogan DB, McDowell I, et al. A global clinical measure of fitness and frailty in elderly people. Can Med Assoc J. 2005;173(5):489–95. Epub 2005/09/01.
Tsiouris A, Hammoud ZT, Velanovich V, Hodari A, Borgi J, Rubinfeld I. A modified frailty index to assess morbidity and mortality after lobectomy. J Surg Res. 2013;183(1):40–6. Epub 2013/01/01.
Leven DM, Lee NJ, Kothari P, Steinberger J, Guzman J, Skovrlj B, et al. Frailty index is a significant predictor of complications and mortality after surgery for adult spinal deformity. Spine. 2016;41(23):E1394–E401. Epub 2016/09/02.
Ali R, Schwalb JM, Nerenz DR, Antoine HJ, Rubinfeld I. Use of the modified frailty index to predict 30-day morbidity and mortality from spine surgery. J Neurosurg Spine. 2016;25(4):537–41. Epub 2016/05/07.
Li ZE, Lu SB, Kong C, Sun WZ, Wang P, Zhang ST. Impact of compliance with an enhanced recovery after surgery program on the outcomes among elderly patients undergoing lumbar fusion surgery. Clin Interv Aging. 2020;15:2423–30. Epub 2021/01/01.
Corniola MV, Meling TR, Tessitore E. Enhanced recovery after spine surgery-a multinational survey assessing surgeons’ perspectives. Acta Neurochir. 2020;162(6):1371–7. Epub 2020/04/04.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Staudt, M.D., Zhou, X., Wijesekera, O., Miller, J.P., Sweet, J.A. (2023). ERAS and Spine Surgery. In: Fu, KM.G., Wang, M.Y., Virk, M.S., Dimar II, J.R., Mummaneni, P.V. (eds) Treatment of Spine Disease in the Elderly. Springer, Cham. https://doi.org/10.1007/978-3-031-12612-3_6
Download citation
DOI: https://doi.org/10.1007/978-3-031-12612-3_6
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-12611-6
Online ISBN: 978-3-031-12612-3
eBook Packages: MedicineMedicine (R0)