Abstract
The majority of nonhuman primates are found in habitats impacted by humans. Therefore, conservation interventions in anthropogenic landscapes are critical for the long-term survival of primate populations. Due to their intelligence and socioecological flexibility, many primates exhibit behaviours deemed problematic such as crop feeding, property damage, and livestock depredation. Large-bodied primates may also pose a physical risk to people. In this chapter, we first revise the common criteria for selecting primate conservation priorities and consider them in the context of shared landscapes. We discuss the importance of inclusive conservation approaches and provide recommendations for addressing negative human-primate interactions based on existing information. Three case studies that illustrate conservation efforts in shared environments are presented: (1) the Bulindi Chimpanzee and Community Project in Uganda, (2) community conservation of orangutans and Javan slow lorises in Indonesia, and (3) inclusive conservation of golden lion tamarins in Brazil’s Atlantic Forest. The active participation of a diverse group of stakeholders, including local community groups, in all conservation stages is essential to fully understand the complexities of human-primate interactions in shared landscapes, address negative interactions, mitigate conservation conflicts, advocate for equity, and promote long-term human-primate coexistence.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Anthropocene
- Anthropogenic
- Brazil
- Coexistence
- Community-based conservation
- Inclusive conservation
- Human-primate interactions
- Indonesia
- Multi-stakeholder conservation
- Uganda
1 Introduction
Humans are possibly the dominant force shaping our planet’s ecosystems and environment, leading many scientists to define a new geological epoch: the Anthropocene (Lewis & Maslin, 2015). It is now estimated that nearly all of our world’s terrestrial landscapes have been altered by humans (Kennedy et al., 2020). Given that contemporary ecosystem dynamics are rarely disconnected from humans, ecologists must explicitly take their interconnectedness into account in their research frameworks (Ellis & Ramankutty, 2008). At present, 75% of the world’s nonhuman primate species (hereafter primates) have declining populations due to human activities, in particular forest conversion into agriculture, logging, and hunting (Estrada et al., 2017). Thus, the ways in which primates respond to and interact with humans are an increasingly important area of research and of growing conservation focus (Humle & Hill, 2016; Kalbitzer & Chapman, 2018; McLennan et al., 2017). Human-primate interactions within ‘shared landscapes’ may range from exposure to anthropogenic noise and infrequent encounters to high spatiotemporal overlap and direct contact, particularly where primates and humans use the same resources or in areas with high hunting pressure (Hockings et al., 2009; McKinney, 2015; McLennan & Hockings, 2016; McLennan et al., 2017; Mormile & Hill, 2017).
The majority of primates inhabit tropical or subtropical forest habitats (Galán-Acedo et al., 2019a) and are susceptible to land use change (Estrada et al., 2017). As human-induced environmental change continues to increase, primates are more often found in modified habitats including forest-agricultural/urban mosaics and commercial plantation landscapes (Spehar & Rayadin, 2017). Many primatologists are arguing for increasing recognition of the critical role of human-impacted landscapes for primate conservation in the twenty-first century, particularly for species able to adapt to some level of land use change (Estrada et al., 2012; Galán-Acedo et al., 2019b). With some level of landscape connectivity enabled by remnant forest fragments, an absence or low levels of hunting, the potential use of arboreal and diversified agriculture environments, and sufficient food sources, many primate species can temporarily or permanently persist in anthropogenic landscapes (Estrada et al., 2012; Chap. 8, this volume).
In this chapter, we (i) review criteria to select primate conservation priorities and consider them in the context of shared landscapes, (ii) discuss the importance of inclusive conservation approaches, and (iii) provide recommendations for addressing negative human-primate interactions based on ‘lessons learned’. We present three case studies across three continents that illustrate conservation efforts in shared environments: chimpanzees (Pan troglodytes schweinfurthii) in Uganda, orangutans (Pongo pygmaeus morio) and Javan slow lorises (Nycticebus javanicus) in Indonesia, and golden lion tamarins (Leontopithecus rosalia) in Brazil.
2 Primate Conservation Priorities in the Anthropocene
Resources available for conservation, including funds, time, and expertise, are limited. It is therefore necessary to develop resource- and cost-effective conservation priorities. But how can we define priorities in primate conservation? Primate conservation priorities typically focus on ‘important’ and/or threatened primate species and/or areas with high primate diversity.
‘Important’ species may consist of keystone, indicator, flagship, and/or umbrella species (Arponen, 2012) (Fig. 10.1). Keystone species play important roles in ecosystem processes and functioning, e.g. they can be critical seed dispersers (McConkey, 2018) and top-down regulators (predators) or suppress competitors (Bond, 1994). Indicator species are considered to reflect some wider aspect of environmental condition, expressing relatively rapid and consistent responses to environmental change (Lawton & Gaston, 2001). Indicator species richness can also act as surrogate to diversity of other wildlife: across Madagascar, for example, lemurs were found to predict non-primate mammal diversity (Muldoon & Goodman, 2015). Flagship species are high-profile taxa mainly used as tools for leveraging conservation, including to raise public conservation awareness and conservation funds (Simberloff, 1998). They are often considered ‘charismatic’, a definition that may differ amongst regions, cultures, and/or groups of people. Albert et al. (2018) identified 20 species considered most charismatic by the Western public. The species included chimpanzees (P. troglodytes) and gorillas (Gorilla spp.), which were considered charismatic for aesthetic reasons or because they are impressive or threatened with extinction (Albert et al., 2018). The iconic status of certain species such as great apes is not universal, however. For example, in Central Kalimantan, indigenous people consider fish more important than orangutans (P. pygmaeus) (Thornton et al., 2020). Wide-ranging, flagship species may be categorised as umbrella species, i.e. a species whose home range and/or minimum land requirements are large enough to include a high number of taxa, so that the protection of their habitat will also benefit the conservation of other, sympatric species (Simberloff, 1998). Primates identified as umbrella species include orangutans (Burivalova et al., 2020), guenons (Cercopithecus spp.: Lambert, 2011), golden lion tamarins (Ruiz-Miranda et al., 2019), and woolly monkeys (Lagothrix lagothricha: Linero et al., 2020).
(a) Infant Bornean orangutan and (b) Temminck’s red colobus in Cantanhez National Park, Guinea Bissau. Due to their charismatic appearance and threatened status, orangutans and colobines can be considered flagship species. (Photo credits: Andrew Walmsley/Yayasan Borneo Nature Indonesia (a) and Elena Bersacola (b))
Prioritisation of species or subspecies that are most vulnerable to extinction is typically based upon assessments by the International Union for Conservation of Nature’s (IUCN) Red List of Threatened Species (IUCN, 2020), which is the leading provider of conservation data, assessments, and analysis and includes the IUCN Primate Specialist Group. It is also worth noting that some species or subspecies may be classified as ‘Least Concern’ globally (IUCN, 2020), but nevertheless are threatened with extinction in some localities or regions. Additionally, classifications can be revised, meaning populations can suddenly move from being of lower to very high conservation concern, for example, due to taxonomic re-classification into distinct species, such as in the case of Tapanuli orangutan (P. tapanuliensis, which was split from P. abelii) (Nater et al., 2017). Besides the global conservation status, it is always worth considering the local context (see case studies). Another way to prioritise in primate conservation is to select areas high in (primate) biodiversity, especially where these areas are also experiencing high threat levels. ‘Biodiversity hotspots’ are threatened areas with high biodiversity and/or species endemism, where conservation action has the potential to have a large impact (Mittermeier et al., 2011).
Given broad variability in goals, scale, and scope, it is difficult to generalise about conservation priorities, particularly within anthropogenic environments (Hockings et al., 2015; Kalbitzer & Chapman, 2018; McLennan et al., 2017). For example, although many human-influenced ecosystems such as agroforests can retain high biodiversity (Estrada et al., 2012), applying the second criterion alone for selecting primate conservation priority areas may exclude opportunities for conservation interventions in important human-primate shared systems which are essential to ensure large-scale, metapopulation connectivity, given that human-influenced ecosystems may have lower biodiversity compared to pristine forests or remote locations in certain regions (Torres-Romero & Olalla-Tárraga, 2015). In addition, some highly imperilled taxa may not range in areas with high biodiversity (e.g. Barbary macaques Macaca sylvanus, Wallis et al. (2020); golden snub-nosed monkey Rhinopithecus roxellana, Long and Richardson (2020)). Considering that certain primate species may nonetheless rely on conservation policies that support and integrate these human-primate contexts, we argue that excluding shared landscapes from conservation priorities risks missing opportunities to develop inclusive, new, and effective conservation approaches that may be applicable to a significant portion of the primates’ geographical range. In addition, human-primate interactions in shared landscapes can generate considerable attention (good and/or bad), including in the media, especially where interactions are ‘negative’ such as when primates damage crops or property or pose a threat to human physical safety (e.g. @NatGeoUK, 2019). These complex interactions require conservation interventions and management for the benefit of local people, for the conservation and welfare of primates, and for the conservation of species – if negative interactions are not addressed, this can weaken public support for conservation (Chua et al., 2020). It is also argued that conservation approaches must now look beyond the one species/habitat patch interventions and should instead aim at restoring ecosystems at the large scale and integrating multi-stakeholder processes (Norris et al., 2020; Reed, 2008). However, habitat restoration might not be possible in many human-dominated landscapes. With the growing evidence that some taxa can cope with modified habitats, conservation aims may focus more on maintaining sufficient ecosystem function to allow species survival.
3 The Importance of Inclusive Primate Conservation Approaches
A crucial goal of conservation practice is to balance the costs and benefits of conservation interventions to people living in proximity to wildlife to promote long-term coexistence (Harrison et al., 2020). Importantly, practitioners must avoid colonial or ‘fortress’ conservation approaches, i.e. exclusionary and often violent conservation approaches based on the human-nature dichotomy view, which have been (and are sometimes still) prevalent across Africa and Asia (Brockington & Igoe, 2006; Colchester, 2004; Mkumbukwa, 2008). Within the scientific community, it is now widely recognised that to be successful in the long term, conservation strategies must ultimately improve local people’s lives (Adams et al., 2004). Many primate species in need of conservation occur in areas where human poverty is high. For example, over 67% of the human population in Guinea-Bissau, where much of the remaining populations of Critically Endangered Temminck’s red colobus (Piliocolobus badius temminckii) are found, live below the poverty line (Bersacola et al., 2018; World Bank, 2021). Poverty is not only economic but encompasses a range of diverse issues that are often country and context specific, such as lack of access to education and healthcare. One way to improve people’s wellbeing is through poverty alleviation and sustainable development programmes (United Nations, 2021). There are many different ways conservation programmes may be able to contribute to reduce poverty, for example, through activities that help generate financial income, but also indirectly via safeguarding Indigenous rights, education and capacity building, as well as approaches that aim to increase socioecological resilience (United Nations, 2021). However, aid-centric approaches that do not question the economic status quo have some heavy critics (Hickel, 2017; Norris et al., 2020). We must remember that widespread structural inequalities, poverty, and lack of equal opportunities not only affect more people in rural or remote areas but also exist between social groups. Economic and social inequalities such as power imbalances between conservation stakeholders and gender inequality remain a problematic issue in contemporary conservation (Chua et al., 2020; Rubis & Theriault, 2020). Primate conservationists must therefore engage with diverse stakeholders to attempt to fully understand and acknowledge existing systemic social issues and develop conservation strategies that also explicitly aim to promote social equity and human wellbeing. A cross-disciplinary, multi-stakeholder team is necessary to fully understand the complexities of human-primate interactions.
Conservation plans in human-wildlife systems can be applied at different scales. At the global/national/regional level, economic policies should reflect the needs of rural communities, including providing incentives to adopt sustainable approaches, particularly in food production, to minimise environmental damage and promote healthy human-influenced ecosystems as well as social equality (Díaz et al., 2019). Likewise, site-level conservation initiatives should incorporate the needs and perspectives of local residents, address the challenges these people experience (by living in proximity to primates), and ideally improve (or, at minimum, not worsen) local people’s lives and livelihoods. Involving different stakeholders in a co-creation process is therefore crucial when developing large-scale conservation plans, such as those at the national level, as well as small-scale projects at the site level. Multi-stakeholder participation can offer opportunities to explore equity issues, address socio-cultural and environmental complexities, and develop trust between policy makers, scientists, and citizens (Reed, 2008). In the case of primate conservation in human-influenced landscapes, stakeholders may often include local farmers, hunters, women and youth associations, traditional and Indigenous authorities, national and international NGOs, government bodies responsible for the management of wildlife or forest resources (e.g. Agriculture, Forestry, or Biodiversity departments), as well as researchers from multiple disciplines (e.g. social scientists, ecologists, economists, agronomists) and educators (Bersacola et al., 2021; Chazdon et al., 2020; Chesney et al., 2020, Case Study 3). Pre-existing socio-political power imbalances amongst conservation stakeholders can easily preclude equity and fairness in the participation process if left unaddressed (Rubis & Theriault, 2020). Multi-stakeholder approaches must ensure a fair and equal exchange of ideas throughout the conservation process, from research to planning, implementation, and monitoring and evaluation. Besides ensuring an inclusive conservation approach, the role of primatologists may also include bridging conversations between local communities and national or international organisations.
4 Addressing Negative Human-Primate Interactions in Anthropogenic Landscapes
Interactions between people and primates in shared landscapes are often complex. Due to their intelligence and socioecological flexibility, many primate species exhibit behaviours deemed problematic by local people. Risks to people living alongside primates include costs to livelihoods due to crop feeding, destruction of stored food, property damage, and livestock depredation (Campbell-Smith et al., 2010; Hill, 2017; Mormile & Hill, 2017). Aggressive interactions between large-bodied primates and humans can also result in human injury and sometimes even death, particularly in young children (Hockings et al., 2010; Hockings & McLennan, 2016; McLennan & Hockings, 2016) (Case Study 1). Primates also pose risks of disease transmission to humans (Jones-Engel et al., 2005; Pedersen & Davies, 2009; Chap. 9, this volume). These risks for humans can be a major cause of primate mortality, for example, when farmers engage in pre-emptive or retaliatory killing to protect crops or livestock (Kibaja, 2014; Kifle & Bekele, 2020; Meijaard et al., 2011; McLennan et al., 2012). It is also important to point out that human-primate interactions are not always negative. Some primates play an important role in human culture and folklore (Cormier, 2006; Fuentes & Gamerl, 2005; Parathian et al., 2018; Riley & Priston, 2010). In some areas, primates are provisioned with food by people (Fuentes & Gamerl, 2005). The presence of primates may also provide economic opportunities to local people through tourism. However, tourism revenues are often distributed unequally, and/or benefits may be limited to a selected few (Cobbinah et al., 2017; Ezebilo & Mattsson, 2010; Sabuhoro et al., 2017).
The applied field of human-wildlife interactions has traditionally focused on resolving ‘problematicFootnote 1’ wildlife behaviour such as primate crop feeding (Hill & Wallace, 2012) (Fig. 10.2). Although some technical interventions aimed at reducing crop foraging have been evaluated in a primate context (Webber et al., 2007; Frank et al., 2019), technical measures alone will not be enough in the long term. In some cases, the challenges to finding long-term solutions to primate crop feeding may be compounded by human fear of some primate species (e.g. orangutans: Campbell-Smith et al., 2010). Additionally, primates can adapt to the most sophisticated repellent devices, and traditional fences are mostly ineffective at keeping primates out (Osborn & Hill, 2005). Primates can also learn to navigate electric fences, which are expensive, need high maintenance, and are unaffordable to most farmers in developing countries (Suzuki & Muroyama, 2010; Priston & McLennan, 2013). The use of scarecrows or farmers chasing and shouting at crop feeding primates are common deterrent methods; continuous guarding during the day can sometimes help reduce crop damage (Byamukama & Asuma, 2006; Hockings & Humle, 2009). Alternative crops can be utilised to mitigate crop loss, for example, via planting of unpalatable crop buffer zones at forest edges and by changing the principal crops grown, but such techniques are only effective if these crops are economically profitable and there are existing market chains. For example, chilli is less vulnerable to baboon and other wildlife damage in Zimbabwe while also having economic value (Parker & Osborn, 2006). Likewise, tea plantations surrounding the Kibale National Park in Uganda are unappealing to wildlife, including primates, but economically valuable (Hartter & Goldman, 2009). Translocation, as a measure to move problematic primates from a particular area, is extremely invasive and requires significant money and effort. It can be lethal to animals due to stress and injuries caused while trying to capture them, particularly when using darts. Searching for and identifying suitable areas for release takes time and effort, and released animals must be monitored for years (e.g. see Palmer, 2018 for a recent detailed treatment of the ethics of rehabilitation and reintroduction in orangutans). In some cases, it may even be detrimental towards landscape-level primate conservation through removing primates in fragments that help maintain connectivity and gene flow between populations (Ancrenaz et al., 2021). Additionally, removing primates from fragmented landscapes may result in negative changes to ecosystem dynamics due to many primates’ role as seed dispersers and ‘pest’ control (e.g. rodents, snakes).
Chimpanzees readily incorporate agricultural foods introduced into their habitats. (a) Wild chimpanzees in Bulindi, Uganda, sharing a cultivated jackfruit in a village garden; behind them is a stand of exotic eucalyptus trees. (b) An adult male chimpanzee at Bulindi eats jackfruit, while a female and her offspring wait for him to finish and leave the fruit for them. (Photo credits: Matthew R. McLennan)
Negative human-primate interactions affect the conservation of a species in a certain geographic area and may also influence public support for conservation programmes, particularly when coupled with a lack of local community involvement in conservation decision-making processes (Sabuhoro et al., 2017). To develop strategies that aim to promote sustainable coexistence in the long term, besides human-wildlife dynamics, we must also fully understand the socio-political nature of conservation-related issues in shared landscapes (Fuentes, 2012). Social, political, and economic power imbalances between stakeholders (including local persons, researchers, policy makers, industry, and community stakeholder groups) underlie conservation conflicts (Temudo, 2012; Leblan, 2016; Hill et al., 2017). A large branch of ecologists now recognise that conservation and human-wildlife coexistence goals can only be achieved by understanding and addressing the socio-political dimension, as well as monitoring the impacts of human-wildlife interactions on human livelihoods and wildlife persistence (Dore et al., 2017; Hill et al., 2017; Pooley et al., 2020). Strategies may necessitate some unusual shifts in focus, for example, studying fish and fishing livelihoods in the context of peatland fires in Indonesia, as a contribution towards conservation of orangutans and other primates (Chua et al., 2020; Thornton et al., 2020). Although cross-disciplinary research on human-primate interactions (i.e. ethnoprimatology; see Fuentes, 2012; Waters et al., 2018) might not directly prevent negative human-wildlife scenarios, it forms an integral part of conservation, for example, by informing risk mitigation schemes and conservation management approaches. At both the research and conservation planning stages, cross-disciplinary research – including psychology, economics, anthropology, political sciences, and ecology – is essential to ensure that pragmatic, effective, and inclusive conservation impacts are achieved (Bartuszevige et al., 2016; Waters et al., 2018).
Box 10.1 The Bulindi Chimpanzee and Community Project: Conservation at the Human-Chimpanzee Interface in Western Uganda
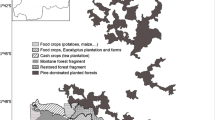
In western Uganda, wild chimpanzees occur outside, as well as within, protected areas. The Budongo and Bugoma Forest Reserves support two of Uganda’s largest chimpanzee populations, but are separated by 50 km. The intervening landscape is densely settled and dominated by agriculture, exotic timber plantations, villages, and urban centres. Since the 1990s, riverine forest, which formerly provided habitat for chimpanzees and other wildlife, was converted to farmland by landowners. About 300 chimpanzees survive in this fast-developing landscape, however, using remnant forest fragments on private land in remarkably close contact with villagers (McCarthy et al., 2015; McLennan, 2008) (Fig. 10.3). Besides habitat loss, these chimpanzees are threatened by infrastructure development including road upgrades, construction of an oil pipeline, and advancing urbanisation (McLennan et al., 2021). Given these circumstances, their long-term survival might appear doubtful. Why then, should we conserve them?
For chimpanzees living in human-dominated landscapes outside protected areas, such as in Bulindi, Uganda, encounters with humans can be unpredictable. (a) Chimpanzees looking towards the sound of people approaching in the distance. (b) Adult males crossing a village road watched by local residents. (Photo credits: Matthew R. McLennan)
First, conserving these chimpanzees is necessary to avoid the large populations in Budongo and Bugoma forests from becoming genetically isolated. Second, increasingly negative interactions between the region’s chimpanzees and human residents need addressing and mitigating. Forest clearance caused chimpanzees to feed habitually on agricultural crops, resulting in economic losses for farmers and occasional trapping or killing of chimpanzees (McLennan et al., 2012). Additionally, local people, especially children, have been seriously injured by chimpanzees and several human babies have been killed. These negative interactions have been reported in Uganda’s press, potentially reducing public support for conservation of the species.
The Bulindi Chimpanzee and Community Project (BCCP) was established in 2015 to address these problems, initially concentrating on one site (Bulindi) where chimpanzees were the focus of long-term research, but where recent deforestation had shrunk local forests by 80% (McLennan et al., 2020). Informal discussions with landowners helped to understand their priorities and constraints. Residents commonly cited their need to raise cash to fund their children’s education as a reason for clearing forest for farming or selling timber. When BCCP offered to contribute to school fees in return for an end to forest cutting, most landowners agreed. This voluntary initiative ended major forest clearance in Bulindi. BCCP helped landowners establish a formal community-based organisation, with a constitution governing conditions of membership that included entrusting members with shared responsibility for conserving local forests. After 6 years of this initiative, forest in Bulindi is regenerating. More recently, the programme was expanded to help landowners conserve unprotected forest used by other chimpanzee groups regionally.
Many landowners no longer have forest on their land, yet are still impacted by chimpanzees. Therefore, a suite of integrated programmes was developed to more widely enhance local capacity to accommodate chimpanzees and engage in conservation. Central to this effort is largescale tree planting. BCCP supplies landowners with tree seedlings as an alternative livelihood to reduce reliance on natural forest. Woodlots of fast-growing species offer an alternative (non-forest) source of wood and income from timber sales, while coffee provides a ‘chimp-friendly’ alternative to tobacco and rice cash-cropping (both major drivers of deforestation) and sugarcane growing. Unlike sugarcane, chimpanzees and other primates are not reported to eat coffee; thus, coffee farming doesn’t generate negative human-primate interactions. Indigenous trees are planted to supplement natural forest regeneration. Other initiatives include energy-efficient stoves that reduce fuelwood consumption; water wells (boreholes) that provide clean water away from forest streams, where children risk encountering chimpanzees; education clubs to promote child safety; savings groups to support alternative livelihoods; and a popular ‘chimpanzee football league’ that sponsors local teams with kits and tournaments. These community-based programmes are combined with research, yielding long-term data on chimpanzee demography, ranging, and behaviour and identifying site-specific threats to help direct conservation efforts. As of 2021, the project reached over 150 villages.
Nevertheless, these interventions offer no quick fix to the complex challenges inherent in conserving wildlife in human-dominated landscapes outside protected areas. Natural forest regionally is unlikely to ever regenerate such that chimpanzees no longer range and forage around villages. Meanwhile, the human population will continue increasing alongside expanding infrastructural development. Human-dominated environments are characterised by diverse priorities and interests of residents and other stakeholders, which create unanticipated challenges. Patience, understanding, and long-term livelihood support and economic opportunities for local residents, alongside careful management of the chimpanzees, will be required for decades to come.
Box 10.2 Primate Conservation in Shared Landscapes in Indonesia
Indonesia is home to an estimated 48 nonhuman primate species, 45 of which are threatened (Estrada et al., 2018), and the world’s fourth largest human population. It possesses diverse habitat types, land uses, and peoples, with 83% of primate ranges outside protected areas (PAs) (Estrada et al., 2018), representing a diversity of human-primate interaction contexts and conservation challenges. We illustrate two contrasting Indonesian primate conservation contexts in non-PA, multi-use landscapes.
The world’s most populated island, Java, still harbours relatively large Javan slow loris habitats, largely distributed in high-altitude agroecosystems in West Java (Nekaris et al., 2017) and lower-altitude, secondary forest with coffee agroforestry in Central Java (Sodik et al., 2020). In Central Java particularly, slow loris habitat is relatively small within existing PAs and mostly occurs in production forest (Sodik et al., 2020).
The presence of a small slow loris population (~7–9 individuals) in Kemuning lowland secondary forest (400 ha) in densely populated Central Java provides new conservation hope (Sodik et al., 2019). This forest is managed by the state-owned enterprise, PERHUTANI, and local people have access to the forest through Pengelolaan Hutan Bersama Masyarakat (PHBM) or social forestry management, for planting shade coffee. However, slow lorises only use parts of this small fragment (Sodik et al., 2019). Due to their high territoriality (Campera et al., 2020; Nekaris et al., 2020), plus the small population in Kemuning, local extinction may be occurring. Promising initiatives include the successful use of artificial canopy bridges to connect loris populations in West Java (Birot et al., 2020) and the promotion of wildlife-friendly coffee production (Campera et al., 2021) by local NGO, JAWI, and Universitas Gadjah Mada, supported by Oxford Brookes University. Local people’s involvement in these initiatives helps to reduce poaching, which is also prohibited in village regulations.
In Kalimantan (Indonesian Borneo), orangutan conservation efforts have historically focused on PAs and ‘undisturbed’ forests, yet >75% of orangutans inhabit areas open for development (Wich et al., 2012). Recent orangutan studies have revealed a high tolerance to forest disturbance in the absence of killing, generating calls to focus on integrated management of multi-use landscapes, including (connecting) orangutan populations in concessions, stakeholder engagement, and killing avoidance (Spehar et al., 2018). One area where this is relevant is Rungan Forest (1500 km2), Central Kalimantan, which supports around 2220–3275 orangutans, plus five other primate species (Buckley et al., 2018; Husson et al., 2019). The forest is bordered by 20 villages, with 22% of it currently protected, 14% allocated for oil palm, and the remainder as pulp and paper concessions (Husson et al., 2019). Borneo Nature Foundation (BNF) and partners are pursuing a multi-stakeholder conservation plan, aiming to safeguard Rungan’s orangutan population while enhancing local community wellbeing. This involves landscape-level orangutan population and habitat surveys, establishing a forest research base, supporting local community forest management rights acquisition, engaging concession managers to conserve High Conservation Value Forest, and implementing sustainable livelihood initiatives, including permaculture (BNF, 2020) (Fig. 10.4). Initial results are encouraging, though further long-term work is required to achieve desired benefits for both orangutans and people (BNF, 2020).
Box 10.3 Inclusive Conservation in Brazil’s Atlantic Forest
Brazil has the highest diversity of primate species (Estrada et al., 2018), and, except for some Amazonian primates, most (including many Endangered species) inhabit landscapes strongly influenced, if not dominated, by human activities including areas of heavy agricultural or urban development. The National Primate Action Plans show concern for dealing with the shared landscape issue. But conservation efforts must move beyond; plans must include the community as actors or stewards.
The Golden Lion Tamarin Conservation Programme, a successful Atlantic Forest flagship species project, is a good example of a conservation strategy with community participation (Kierulff et al., 2012; Ruiz-Miranda et al., 2019). Golden lion tamarins live in a landscape of forest fragments within an agricultural and urban matrix, situated between Rio de Janeiro (80 km away) and major oil and gas production areas, and it is the only water source for a major coastal tourist area in the state. From its foundation in 1984, the programme hired local people to work as research assistants and educators and provide field site logistics. Several of those original employees still work in the project, 40 years later. Environmental education was set up to foster knowledge about the golden lion tamarins and support for forest conservation (Dietz & Nagagata, 1995; Dietz, 1998; Engels & Jacobson, 2007; Pádua et al., 2002). A key strategy, the reintroduction of captive born animals, was only possible through the participation of local landowners, with all the release sites (after the initial experimental release) on private land (Kierulff et al., 2012; Ruiz-Miranda et al., 2010). In 1992, Associação Mico Leão Dourado (Golden Lion Tamarin Association, AMLD) was created (Rambaldi et al., 2002; Ruiz-Miranda et al., 2020) as a community-based NGO with local landowners serving as active and/or board members. Other members include the Chico Mendes Institute for Biodiversity Conservation and local government officials. The AMLD adaptive management strategic plan is organised around monitoring the population and reducing threats to golden lion tamarins and their habitat (Dietz et al., 2010; Ruiz-Miranda et al., 2019). Activities such as reforestation and establishment of forest corridors depend on local landowner participation (Fernandes et al., 2008). For reforestation, the AMLD established a programme to build capacity for six landowners to develop commercial nurseries for native tree species to be used in all reforestation efforts. The AMLD also assists the community to develop economic activities that rely on sustainable land use such as agroforestry and ecotourism. The reforestation and forest protection efforts of the AMLD and the Ministry of the Environment have benefited the municipal government; the region receives the largest amount of green tax funds in the State of Rio. The AMLD and local community continue to work together to make the region a multi-use conservation landscape that protects biodiversity and fosters economic activities and quality of life.
5 Conclusion
Achieving sustainable coexistence between humans and wildlife is one of the greatest challenges we face in the Anthropocene (Lewis & Maslin, 2015; Hockings et al., 2015; McLennan et al., 2017; Frank et al., 2019; Bersacola et al., 2021). Human-wildlife dynamics are influenced by direct interactions, including competition over space and resources, as well as the socio-political, economic, and environmental contexts. Primate conservation strategies in anthropogenic environments must be based on cross-disciplinary research approaches that are able to resolve these multiple, complex socioecological dimensions. Crucially, as demonstrated in the three case studies presented, conservation practitioners must work directly with local people and ensure equity in decision-making and long-term collaboration amongst stakeholders in all phases including in research and planning and within primate conservation strategies’ adaptive frameworks.
References
Adams, W. M., Aveling, R., Brockington, D., Dickson, B., Elliott, J., Hutton, J., Roe, D., Vira, B., & Wolmer, W. (2004). Biodiversity conservation and the eradication of poverty. Science, 306(5699), 1146–1149.
Albert, C., Luque, G. M., & Courchamp, F. (2018). The twenty most charismatic species. PLoS One, 13(7), e0199149. https://doi.org/10.1371/journal.pone.0199149
Ancrenaz, M., Oram, F., Nardiyono, N., et al. (2021). Importance of small forest fragments in agricultural landscapes for maintaining orangutan metapopulations. Frontiers in Forests and Global Change, 4, 560944. https://doi.org/10.3389/ffgc.2021.560944
Arponen, A. (2012). Prioritizing species for conservation planning. Biodiversity and Conservation, 21(4), 875–893.
Bartuszevige, A. M., Taylor, K., Daniels, A., & Carter, M. F. (2016). Landscape design: Integrating ecological, social, and economic considerations into conservation planning. Wildlife Society Bulletin, 40(3), 411–422.
Bersacola, E., Bessa, J., Frazão-Moreira, A., et al. (2018). Primate occurrence across a human-impacted landscape in Guinea-Bissau and neighbouring regions in West Africa: Using a systematic literature review to highlight the next conservation steps. PeerJ, 6, e4847. https://doi.org/10.7717/peerj.4847
Bersacola, E., Parathian, H., Frazão-Moreira, A., et al. (2021). Developing an evidence-based coexistence strategy to promote human and wildlife health in a biodiverse agroforest landscape. Frontiers in Conservation Science, 2, 735367. https://doi.org/10.3389/fcosc.2021.735367
Birot, H., Campera, M., Imron, M. A., & Nekaris, K. A. I. (2020). Artificial canopy bridges improve connectivity in fragmented landscapes: The case of Javan slow lorises in an agroforest environment. American Journal of Primatology, 82(4), e23076. https://doi.org/10.1002/ajp.23076
BNF. (2020). Initiating biodiversity conservation in the Rungan River Landscape, Central Kalimantan, Indonesia. Annual report 2019. Borneo Nature Foundation, Palangka Raya, Indonesia.
Bond, W. J. (1994). Keystone species. In E.-D. Schulze & H. A. Mooney (Eds.), Biodiversity and ecosystem function (pp. 237–253). Springer.
Brockington, D., & Igoe, J. (2006). Eviction for conservation: A global overview. Conservation and Society, 4(3), 424.
Buckley, B. J. W., Ripoll Capilla, B., et al. (2018). Biodiversity, forest structure & conservation importance of the Mungku Baru Education Forest, Rungan, Central Kalimantan, Indonesia. Borneo Nature Foundation in collaboration with Universitas Muhammadiyah Palangkaraya and University of Exeter, Palangka Raya, Indonesia.
Burivalova, Z., Game, E. T., Wahyudi, B., et al. (2020). Does biodiversity benefit when the logging stops? An analysis of conservation risks and opportunities in active versus inactive logging concessions in Borneo. Biological Conservation, 241, 108369. https://doi.org/10.1016/j.biocon.2019.108369
Byamukama, J., & Asuma, S. (2006). Human-gorilla conflict resolution (HuGo) – The Uganda experience. Gorilla Journal, 32, 10–12.
Campbell-Smith, G., Simanjorang, H. V. P., Leader-Williams, N., & Linkie, M. (2010). Local attitudes and perceptions toward crop-raiding by orangutans (Pongo abelii) and other nonhuman primates in northern Sumatra, Indonesia. American Journal of Primatology, 72(10), 866–876.
Campera, M., Brown, E., Imron, M. A., & Nekaris, K. A. I. (2020). Unmonitored releases of small animals? The importance of considering natural dispersal, health, and human habituation when releasing a territorial mammal threatened by wildlife trade. Biological Conservation, 242, 108404.
Campera, M., Budiadi, B., Adinda, E., et al. (2021). Fostering a wildlife-friendly program for sustainable coffee farming: The case of small-holder farmers in Indonesia. Land, 10(2), 1–16.
Chazdon, R. L., Cullen, L., Padua, S. M., & Padua, C. V. (2020). People, primates and predators in the Pontal: From endangered species conservation to forest and landscape restoration in Brazil’s Atlantic Forest. Royal Society Open Science, 7(12), 200939. https://doi.org/10.1098/rsos.200939
Chesney, C., Bolaños, N. C., Kanneh, B. A., Sillah, E., et al. (2020). Mobonda Community Conservation Project: Chimpanzees, oysters, and community engagement in Sierra Leone. American Journal of Primatology, 83(1), e23219. https://doi.org/10.1002/ajp.23219
Chua, L., Harrison, M. E., Fair, H., et al. (2020). Conservation and the social sciences: Beyond critique and co-optation. A case study from orangutan conservation. People and Nature, 2(1), 42–60.
Cobbinah, P. B., Amenuvor, D., Black, R., & Peprah, C. (2017). Ecotourism in the Kakum Conservation Area, Ghana: Local politics, practice and outcome. Journal of Outdoor Recreation and Tourism, 20, 34–44.
Colchester, M. (2004). Conservation policy and indigenous peoples. Environmental Science & Policy, 7(3), 145–153.
Cormier, L. (2006). A preliminary review of neotropical primates in the subsistence and symbolism of indigenous lowland South American peoples. Ecological and Environmental Anthropology, 2, 14–32.
Díaz, S., Settele, J., Brondízio, E. S., Ngo, H. T., et al. (2019). Pervasive human-driven decline of life on Earth points to the need for transformative change. Science, 366(6471), eaax3100. https://doi.org/10.1126/science.aax3100
Dietz, L. A. (1998). Community conservation education programme for the Golden Lion Tamarin in Brazil: Building support for habitat conservation. In Hoarge, R. J. and Moran, K. (Ed.), Culture: The missing element in conservation and development (pp. 85–94). Washington, DC: Kendal/Hunt Publishing Company and National Zoological Park, Smithsonian Institution.
Dietz, L., & Nagagata, E. Y. (1995). Golden lion tamarin conservation program: A community educational effort for forest conservation in Rio de Janeiro State, Brazil. In S. K. Jacobson (Ed.), Conserving wildlife: International education and communication approaches (pp. 64–86). Columbia University Press.
Dietz, L. A., Brown, M., & Swaminathan, V. (2010). Increasing the impact of conservation projects. American Journal of Primatology, 72(5), 425–440.
Dore, K. M., Riley, E. P., & Fuentes, A. (2017). Ethnoprimatology. Cambridge University Press.
Ellis, E. C., & Ramankutty, N. (2008). Putting people in the map: Anthropogenic biomes of the world. Frontiers in Ecology and the Environment, 6(8), 439–447.
Engels, C. A., & Jacobson, S. K. (2007). Evaluating long-term effects of the golden lion tamarin environmental education program in Brazil. The Journal of Environmental Education, 38(3), 3–14.
Estrada, A., Raboy, B. E., & Oliveira, L. C. (2012). Agroecosystems and primate conservation in the tropics: A review. American Journal of Primatology, 74(8), 696–711.
Estrada, A., Garber, P. A., Rylands, A. B., et al. (2017). Impending extinction crisis of the world’s primates: Why primates matter. Science Advances, 3(1), e1600946. https://doi.org/10.1126/sciadv.1600946
Estrada, A., Garber, P. A., Mittermeier, R. A., et al. (2018). Primates in peril: The significance of Brazil, Madagascar, Indonesia and the Democratic Republic of the Congo for global primate conservation. PeerJ, 6, e4869. https://doi.org/10.7717/peerj.4869
Ezebilo, E. E., & Mattsson, L. (2010). Socio-economic benefits of protected areas as perceived by local people around Cross River National Park, Nigeria. Forest Policy and Economics, 12(3), 189–193.
Fernandes, R. V., Rambaldi, D. M., & de Godoy Teixeira, A. M. (2008). Restauração e proteção legal da paisagem – Corredores florestais e RPPNs. In P. P. de Oliveira, A. D. Gravitol, & C. R. Miranda (Eds.), Conservação do mico-leão-dourado (pp. 160–179). Biblioteca do Centro de Biociências e Biotecnologia da Universidade Estadual do Norte Fluminense.
Frank, B., Glikman, J. A., & Marchini, S. (2019). Human–wildlife interactions: Turning conflict into coexistence. Cambridge University Press.
Fuentes, A. (2012). Ethnoprimatology and the anthropology of the human-primate interface. Annual Review of Anthropology, 41(1), 101–117.
Fuentes, A., & Gamerl, S. (2005). Disproportionate participation by age/sex classes in aggressive interactions between long-tailed macaques (Macaca fascicularis) and human tourists at Padangtegal monkey forest, Bali, Indonesia. American Journal of Primatology, 66(2), 197–204.
Galán-Acedo, C., Arroyo-Rodríguez, V., Andresen, E., & Arasa-Gisbert, R. (2019a). Ecological traits of the world’s primates. Scientific Data, 6(1), 1–5. https://doi.org/10.1038/s41597-019-0059-9
Galán-Acedo, C., Arroyo-Rodríguez, V., Andresen, E., et al. (2019b). The conservation value of human-modified landscapes for the world’s primates. Nature Communications, 10(1), 1–8. https://doi.org/10.1038/s41467-018-08139-0
Harrison, M. E., Ottay, J. B., D’Arcy, L. J., et al. (2020). Tropical forest and peatland conservation in Indonesia: Challenges and directions. People and Nature, 2(1), 4–28.
Hartter, J., & Goldman, A. C. (2009). Life on the edge: Balancing biodiversity, conservation, and sustaining rural livelihoods around Kibale National Park, Uganda. Focus on Geography, 52(1), 11–17.
Hickel, J. (2017). The divide: A brief guide to global inequality and its solutions. Random House.
Hill, C. M. (2015). Perspectives of ‘conflict’ at the wildlife–agriculture boundary: 10 years on. Human Dimensions of Wildlife, 20(4), 296–301.
Hill, C. M. (2017). Primate crop feeding behavior, crop protection, and conservation. International Journal of Primatology, 38(2), 385–400.
Hill, C. M. (2018). Crop foraging, crop losses, and crop raiding. Annual Review of Anthropology, 47(1), 377–394.
Hill, C. M., & Wallace, G. E. (2012). Crop protection and conflict mitigation: Reducing the costs of living alongside non-human primates. Biodiversity and Conservation, 21(10), 2569–2587.
Hill, C. M., Webber, A. D., & Priston, N. E. C. (2017). Understanding conflicts about wildlife: A biosocial approach. Berghahn Books.
Hockings, K. J., & Humle, T. (2009). Best practice guidelines for the prevention and mitigation of conflict between humans and great apes. IUCN.
Hockings, K. J., & McLennan, M. R. (2016). Problematic primate behaviour in agricultural landscapes: Chimpanzees as ‘pests’ and ‘predators’. In M. T. Waller (Ed.), Ethnoprimatology: Primate conservation in the 21st century (pp. 137–156). Springer International Publishing.
Hockings, K. J., Anderson, J. R., & Matsuzawa, T. (2009). Use of wild and cultivated foods by chimpanzees at Bossou, Republic of Guinea: Feeding dynamics in a human-influenced environment. American Journal of Primatology, 71(8), 636–646.
Hockings, K. J., Yamakoshi, G., Kabasawa, A., & Matsuzawa, T. (2010). Attacks on local persons by chimpanzees in Bossou, Republic of Guinea: Long-term perspectives. American Journal of Primatology, 72(10), 887–896.
Hockings, K. J., McLennan, M. R., Carvalho, S., et al. (2015). Apes in the Anthropocene: Flexibility and survival. Trends in Ecology & Evolution, 30(4), 215–222. https://doi.org/10.1016/j.tree.2015.02.002
Humle, T., & Hill, C. M. (2016). People–primate interactions: Implications for primate conservation. In S. A. Wich & A. J. Marshall (Eds.), An introduction to primate conservation (pp. 219–240). Oxford University Press.
Husson, S. J., Ripoll Capilla, B., Ottay, J. B., & Buckley, B. J. W. (2019). Initiating orangutan conservation in the Rungan Landscape, Central Kalimantan, Indonesia. 2019 Annual report. Borneo Nature Foundation, Palangka Raya, Indonesia.
IUCN. (2020). The IUCN red list of threatened species. IUCN. https://www.iucnredlist.org/en. Accessed 3 Dec 2020.
Jones-Engel, L., Engel, G. A., Schillaci, M. A., et al. (2005). Primate-to-human retroviral transmission in Asia. Emerging Infectious Diseases, 11(7), 1028–1035.
Kalbitzer, U., & Chapman, C. A. (2018). Primate responses to changing environments in the Anthropocene. In U. Kalbitzer & K. M. Jack (Eds.), Primate life histories, sex roles, and adaptability: Essays in honour of Linda M. Fedigan (pp. 283–310). Springer International Publishing.
Kennedy, C. M., Oakleaf, J. R., Theobald, D. M., et al. (2020). Global human modification of terrestrial systems. NASA Socioeconomic Data and Applications Center (SEDAC).
Kibaja, M. (2014). Diet of the ashy red colobus (Piliocolobus tephrosceles) and crop-raiding in a forest-farm mosaic, Mbuzi, Rukwa Region, Tanzania. Primate Conservation, 2014(28), 109–116.
Kierulff, M. C. M., Ruiz-Miranda, C. R., Oliveira, P. P., et al. (2012). The golden lion tamarin Leontopithecus rosalia: A conservation success story. International Zoo Yearbook, 46(1), 36–45.
Kifle, Z., & Bekele, A. (2020). Human–gelada conflict and attitude of the local community toward the conservation of the southern gelada (Theropithecus gelada obscurus) around Borena Sayint National Park, Ethiopia. Environmental Management, 65(3), 399–409.
Lambert, J. E. (2011). Primate seed dispersers as umbrella species: A case study from Kibale National Park, Uganda, with implications for Afrotropical forest conservation. American Journal of Primatology, 73(1), 9–24.
Lawton, J. H., & Gaston, K. J. (2001). Indicator species. In S. A. Levin (Ed.), Encyclopedia of biodiversity (pp. 437–450). Elsevier.
Leblan, V. (2016). Territorial and land-use rights perspectives on human-chimpanzee-elephant coexistence in West Africa (Guinea, Guinea-Bissau, Senegal, nineteenth to twenty-first centuries). Primates, 57(3), 359–366.
Lewis, S. L., & Maslin, M. A. (2015). Defining the Anthropocene. Nature, 519(7542), 171–180.
Linero, D., Cuervo-Robayo, A. P., & Etter, A. (2020). Assessing the future conservation potential of the Amazon and Andes Protected Areas: Using the woolly monkey (Lagothrix lagothricha) as an umbrella species. Journal for Nature Conservation, 58, 125926. https://doi.org/10.1016/j.jnc.2020.125926
Long, Y., & Richardson, M. (2020). Rhinopithecus roxellana. In IUCN red list threatened species. IUCN. https://www.iucnredlist.org/en. Accessed 3 Dec 2020.
McCarthy, M. S., Lester, J. D., Howe, E. J., Arandjelovic, M., Stanford, C. B., & Vigilant, L. (2015). Genetic censusing identifies an unexpectedly sizable population of an endangered large mammal in a fragmented forest landscape. BMC Ecology, 15(1), 21. https://doi.org/10.1186/s12898-015-0052-x
McConkey, K. R. (2018). Seed dispersal by primates in Asian habitats: From species, to communities, to conservation. International Journal of Primatology, 39(3), 466–492.
McKinney, T. (2015). A classification system for describing anthropogenic influence on nonhuman primate populations. American Journal of Primatology, 77(7), 715–726.
McLennan, M. R. (2008). Beleaguered chimpanzees in the agricultural district of Hoima, Western Uganda. Primate Conservation, 23(1), 45–54.
McLennan, M. R., & Hockings, K. J. (2016). The aggressive apes? Causes and contexts of great ape attacks on local persons. In F. M. Angelici (Ed.), Problematic wildlife (pp. 373–394). Springer.
McLennan, M. R., Hyeroba, D., Asiimwe, C., et al. (2012). Chimpanzees in mantraps: Lethal crop protection and conservation in Uganda. Oryx, 46(4), 598–603.
McLennan, M. R., Spagnoletti, N., & Hockings, K. J. (2017). The implications of primate behavioral flexibility for sustainable human–primate coexistence in anthropogenic habitats. International Journal of Primatology, 38(2), 105–121.
McLennan, M. R., Lorenti, G. A., Sabiiti, T., & Bardi, M. (2020). Forest fragments become farmland: Dietary response of wild chimpanzees (Pan troglodytes) to fast-changing anthropogenic landscapes. American Journal of Primatology, 82(4), e23090. https://doi.org/10.1002/ajp.23090
McLennan, M. R., Hintz, B., Kiiza, V., et al. (2021). Surviving at the extreme: Chimpanzee ranging is not restricted in a deforested human-dominated landscape in Uganda. African Journal of Ecology, 59(1), 17–28. https://doi.org/10.1111/aje.12803
Meijaard, E., Buchori, D., Hadiprakarsa, Y., et al. (2011). Quantifying killing of orangutans and human-orangutan conflict in Kalimantan, Indonesia. PLoS One, 6(11), e27491. https://doi.org/10.1371/journal.pone.0027491
Mittermeier, R. A., Turner, W. R., Larsen, F. W., et al. (2011). Global biodiversity conservation: The critical role of hotspots. In F. E. Zachos & J. C. Habel (Eds.), Biodiversity hotspots: Distribution and protection of conservation priority areas (pp. 3–22). Springer.
Mkumbukwa, A. R. (2008). The evolution of wildlife conservation policies in Tanzania during the colonial and post-independence periods. Development Southern Africa, 25(5), 589–600. https://doi.org/10.1080/03768350802447875
Mormile, J. E., & Hill, C. M. (2017). Living with urban baboons: Exploring attitudes and their implications for local baboon conservation and management in Knysna, South Africa. Human Dimensions of Wildlife, 22(2), 99–109.
Muldoon, K. M., & Goodman, S. M. (2015). Primates as predictors of mammal community diversity in the forest ecosystems of Madagascar. PLoS One, 10(9), e0136787. https://doi.org/10.1371/journal.pone.0136787
Nater, A., Mattle-Greminger, M. P., Nurcahyo, A., et al. (2017). Morphometric, behavioral, and genomic evidence for a new orangutan species. Current Biology, 27(22), 3487–3498.e10. https://doi.org/10.1016/j.cub.2017.09.047
@NatGeoUK. (2019). ‘I am scared all the time’: Chimps and people are clashing in rural Uganda. National Geographic. https://www.nationalgeographic.co.uk/animals/2019/11/i-am-scared-all-time-chimps-and-people-are-clashing-rural-uganda. Accessed 2 Feb 2021.
Nekaris, K. A. I., Poindexter, S., Reinhardt, K. D., et al. (2017). Coexistence between Javan slow lorises (Nycticebus javanicus) and humans in a dynamic agroforestry landscape in West Java, Indonesia. International Journal of Primatology, 38(2), 303–320.
Nekaris, K. A. I., Campera, M., Nijman, V., et al. (2020). Slow lorises use venom as a weapon in intraspecific competition. Current Biology, 30(20), R1252–R1253. https://doi.org/10.1016/j.cub.2020.08.084
Norris, K., Terry, A., Hansford, J. P., & Turvey, S. T. (2020). Biodiversity conservation and the Earth system: Mind the gap. Trends in Ecology & Evolution, 35(10), 919–926. https://doi.org/10.1016/j.tree.2020.06.010
Osborn, F. V., & Hill, C. M. (2005). Techniques to reduce crop loss: Human and technical dimensions in Africa. In R. Woodroffe, S. Thirgood, & A. Rabinowitz (Eds.), People and wildlife, conflict or co-existence? (pp. 72–85). Cambridge University Press.
Pádua, S., Dietz, L. A., Souza, M. G., & Santos, G. R. (2002). In situ conservation education and the lion tamarins. In D. G. Kleiman & A. B. Rylands (Eds.), Lion tamarins: Biology and conservation (pp. 315–335). Smithsonian Institution Press.
Palmer, A. (2018). Kill, incarcerate, or liberate? Ethics and alternatives to orangutan rehabilitation. Biological Conservation, 227, 181–188.
Parathian, H. E., McLennan, M. R., Hill, C. M., Frazão-Moreira, A., & Hockings, K. J. (2018). Breaking through disciplinary barriers: Human–wildlife interactions and multispecies ethnography. International Journal of Primatology, 39, 749–775.
Parker, G. E., & Osborn, F. V. (2006). Investigating the potential for chilli Capsicum spp. to reduce human-wildlife conflict in Zimbabwe. Oryx, 40(3), 343–346.
Pedersen, A. B., & Davies, T. J. (2009). Cross-species pathogen transmission and disease emergence in primates. EcoHealth, 6(4), 496–508.
Pooley, S., Bhatia, S., & Vasava, A. (2020). Rethinking the study of human–wildlife coexistence. Conservation Biology, 35, 784–793.
Priston, N. E. C., & McLennan, M. R. (2013). Managing humans, managing macaques: Human–macaque conflict in Asia and Africa. In S. Radhakrishna, M. A. Huffman, & A. Sinha (Eds.), The macaque connection: Cooperation and conflict between humans and macaques (pp. 225–250). Springer.
Rambaldi, D. M., Kleiman, D. G., Mallinson, J. J. C., et al. (2002). The role of nongovernmental organizations and the International Committee for the Conservation and Management of Leontopithecus in lion tamarin conservation. In D. G. Kleiman & A. B. Rylands (Eds.), Lion tamarins: Biology and conservation (pp. 71–94). Smithsonian Institution Press.
Reed, M. S. (2008). Stakeholder participation for environmental management: A literature review. Biological Conservation, 141(10), 2417–2431.
Riley, E. P., & Priston, N. E. C. (2010). Macaques in farms and folklore: Exploring the human–nonhuman primate interface in Sulawesi, Indonesia. American Journal of Primatology, 72(10), 848–854.
Rubis, J. M., & Theriault, N. (2020). Concealing protocols: Conservation, Indigenous survivance, and the dilemmas of visibility. Social and Cultural Geography, 21(7), 962–984.
Ruiz-Miranda, C. R., Beck, B. B., Kleiman, D. G., et al. (2010). Re-introduction and translocation of golden lion tamarins, Atlantic Coastal Forest, Brazil: The creation of a metapopulation. In Global re-introduction perspectives: Additional case-studies from around the globe (pp. 225–230). IUCN/SSC Re-introduction Specialist Group.
Ruiz-Miranda, C. R., de Morai, M. M., Jr., Dietz, L. A., et al. (2019). Estimating population sizes to evaluate progress in conservation of endangered golden lion tamarins (Leontopithecus rosalia). PLoS One, 14(6), e0216664. https://doi.org/10.1371/journal.pone.0216664
Ruiz-Miranda, C. R., Vilchis, L. I., & Swaisgood, R. R. (2020). Exit strategies for wildlife conservation: Why they are rare and why every institution needs one. Frontiers in Ecology and the Environment, 18(4), 203–210. https://doi.org/10.1002/fee.2163
Sabuhoro, E., Wright, B., Munanura, I. E., et al. (2017). The potential of ecotourism opportunities to generate support for mountain gorilla conservation among local communities neighbouring Volcanoes National Park in Rwanda. Journal of Ecotourism, 20(1), 1–17. https://doi.org/10.1080/14724049.2017.1280043
Simberloff, D. (1998). Flagships, umbrellas, and keystones: Is single-species management passé in the landscape era? Biological Conservation, 83(3), 247–257.
Sodik, M., Pudyatmoko, S., Yuwono, P. S. H., & Imron, M. A. (2019). Resource selection by Javan slow loris Nycticebus javanicus E. Geoffroy, 1812 (Mammalia: Primates: Lorisidae) in a lowland fragmented forest in Central Java, Indonesia. Journal of Threatened Taxa, 11(6), 13667–13679.
Sodik, M., Pudyatmoko, S., Yuwono, P. S. H., et al. (2020). Better providers of habitat for Javan slow loris (Nycticebus javanicus E. Geoffroy 1812): A species distribution modeling approach in Central Java, Indonesia. Biodiversitas Journal of Biological Diversity, 21(5), 1890–1900. https://doi.org/10.13057/biodiv/d210515
Spehar, S. N., & Rayadin, Y. (2017). Habitat use of Bornean orangutans (Pongo pygmaeus morio) in an industrial forestry plantation in East Kalimantan, Indonesia. International Journal of Primatology, 38(2), 358–384.
Spehar, S. N., Sheil, D., Harrison, T., et al. (2018). Orangutans venture out of the rainforest and into the Anthropocene. Science Advances, 4(6), e1701422.
Suzuki, K., & Muroyama, Y. (2010). Topic 5: Resolution of human–macaque conflicts: Changing from top-down to community-based damage management. In N. Nakagawa, M. Nakamichi, & H. Sugiura (Eds.), The Japanese macaques (pp. 359–373). Springer Japan.
Temudo, M. P. (2012). ‘The white men bought the forests’: Conservation and contestation in Guinea-Bissau, Western Africa. Conservation & Society, 10(4), 354.
Thornton, S. A., Setiana, E., Yoyo, K., et al. (2020). Towards biocultural approaches to peatland conservation: The case for fish and livelihoods in Indonesia. Environmental Science & Policy, 114, 341–351.
Torres-Romero, E. J., & Olalla-Tárraga, M. Á. (2015). Untangling human and environmental effects on geographical gradients of mammal species richness: A global and regional evaluation. The Journal of Animal Ecology, 84(3), 851–860.
United Nations. (2021). THE 17 GOALS | Sustainable Development. https://sdgs.un.org/goals. Accessed 25 Feb 2021.
Wallis, J., Benrabah, M., Pilot, M., et al. (2020). Macaca sylvanus. In The IUCN red list of threatened species. IUCN. https://www.iucnredlist.org/en. Accessed 3 Dec 2020.
Waters, S., Bell, S., & Setchell, J. M. (2018). Understanding human-animal relations in the context of primate conservation: A multispecies ethnographic approach in North Morocco. Folia Primatologica, 89(1), 13–29.
Webber, A. D., Hill, C. M., & Reynolds, V. (2007). Assessing the failure of a community-based human-wildlife conflict mitigation project in Budongo Forest Reserve, Uganda. Oryx, 41(2), 177–184.
Wich, S. A., Gaveau, D., Abram, N., et al. (2012). Understanding the impacts of land-use policies on a threatened species: Is there a future for the Bornean orangutan? PLoS One, 7(11), e49142. https://doi.org/10.1371/journal.pone.0049142
World Bank. (2021). Guinea-Bissau | Data. https://data.worldbank.org/country/GW. Accessed 25 Feb 2021.
Acknowledgements
KJH’s work was funded by the Darwin Initiative (project 26-018). MEH thanks the Indonesian Ministry of Research and Technology (RISTEK-BRIN) for research permissions and Universitas Palangka Raya and Universitas Muhammadiyah Palangkaraya for support. BNF’s work in Rungan was funded by the Arcus Foundation, Danish Civil Society in Development, Rainforest Trust, The Orangutan Project, US Fish and Wildlife Service Great Apes Conservation Fund, and Prince Bernhard Nature Fund. MRM thanks Born Free Foundation, Friends of Chimps, Jane Goodall Institute-Switzerland, Blair Drummond Safari Park, the Primate Society of Great Britain, and Uganda Biodiversity Fund for supporting the Bulindi Chimpanzee and Community Project.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Bersacola, E. et al. (2023). Primate Conservation in Shared Landscapes. In: McKinney, T., Waters, S., Rodrigues, M.A. (eds) Primates in Anthropogenic Landscapes. Developments in Primatology: Progress and Prospects. Springer, Cham. https://doi.org/10.1007/978-3-031-11736-7_10
Download citation
DOI: https://doi.org/10.1007/978-3-031-11736-7_10
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-11735-0
Online ISBN: 978-3-031-11736-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)