Abstract
Identifying and listing substances or materials as contaminants of emerging concern (CECs) is not a simple task, and for the marine environment specifically is a challenge for environmental regulators, managers and researchers worldwide (Box 13.1) (Tornero V, and Hanke G (2017) Potential chemical contaminants in the marine environment: An overview of main contaminant lists. Office of the European Union Publications, Luxembourg. Available at: http://publications.jrc.ec.europa.eu/repository/bitstream/JRC108964/potential_chemical_contaminants_in_the_marine.pdf [Accessed 19 August2019].). Some of these agencies have widely different definitions of what a CEC actually is (Halden J Hazard Mater 282:2–9, 2015).
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
13.1 Introduction
Identifying and listing substances or materials as contaminants of emerging concern (CECs) is not a simple task, and for the marine environment specifically is a challenge for environmental regulators, managers and researchers worldwide (Box 13.1) (Tornero and Hanke 2017). Some of these agencies have widely different definitions of what a CEC actually is (Halden 2015).
The meaning of the term contaminant is relatively well understood and is discussed in Chapter 1 of this book. Although the text used by various authors and agencies to define contamination varies, it usually includes or implies the involvement of human-related activities and results in the production of an unnatural concentration of material in a specific environment leading to an associated adverse consequence or impairment to the natural condition for one or more attributes of that environment. However, the terms emerging and concern are more subjective, and are subject to time scales and prevailing circumstances.
13.1.1 What is Meant by “Emerging”?
A meta-analysis of 143,000 publications about 12 prominent CECs ranging from the pesticide DDT to nanoparticles and microplastics (Halden 2015) showed a common time course of emergence and subsidence of concern spanning about 29 years. That study noted that a number of factors can trigger and accelerate the emergence of new CECs, for example, new methods of detection and lowered detection limits, paradigm shifts in scientific understanding, breakthroughs in the design and manufacture of materials and changes in marketing and consumer behaviour leading to increased chemical consumption. Each of these factors can bring long-ignored environmental contaminants into the public eye and drive an increasing level of concern. This increase in the level of concern about a substance or material often triggers further research and publishing activity and the development of new regulations.
13.2 Contaminants of Emerging Concern in the Marine Environment
The list of potential candidate substances to be CECs in the marine environment is very large. In excess of 100 million chemical substances are currently registered in the Chemical Abstracts Service (CAS) and about 4000 new ones are registered every day. The number of registered and pre-registered substances in REACH (the European Union legislation for the Registration, Evaluation, Authorisation and Restriction of Chemicals) lists 30,000–50,000 industrial chemicals present in daily-use products, all of which are potentially ultimately released into the environment (Dulio et al. 2018). However, not all of these chemicals are of concern once released to the environment, and many are unlikely to become CECs. Numerous international environmental agencies and regulators have compiled individual lists of chemicals and substances they regard as being of concern but there is no common list accepted by all the relevant organizations.
The European Commission Joint Research Centre has compiled a “comprehensive list of chemical substances considered relevant” under European Union legislation and by international organizations (Tornero and Hanke 2017). Although not all of the listed contaminants are of concern for the marine environment, this list is invaluable in presenting in one table the total of approximately 2700 of concern substances (or groups of substances) identified under relevant global conventions (e.g. the Stockholm Convention on POPs), European legislation (e.g. REACH), government agencies (e.g. the United States Envrionmental Protection Agency (US EPA) Priority Pollutants legislation) and international research groups (e.g. the NORMAN Network), together with the status of each contaminant on its source list.
Several large-scale monitoring programs in the marine environment have focused on detecting and monitoring emerging contaminants of concern. The most well known (and possibly the largest, longest lasting and best resourced) of these programmes is the Mussel Watch Program conducted by the National Oceanic and Atmospheric Administration (NOAA) in North America since 1986 (NOAA 2008). (See also Chapter 2, Box 2.1).
In the marine environment, a good example of an emerging contaminant of worldwide concern at the time (some 50 years before the present) is provided by the emergence of concern over the use of tributyltin (TBT) as an active ingredient in anti-fouling coatings applied to the hulls of ships. The published scientific material on the TBT issue is very extensive, but an overview published by the European Environment Agency (EEA) is succinct and comprehensive (Santillo et al. 2002) (see also Chapter 7). In summary, the use of antifoulant protection on the submerged portion of ship hulls is essential to minimize the growth of marine life (fouling) that causes hull damage to timber vessels and reduces speed and increases fuel consumption in all affected vessels regardless of the material from which they are constructed. Initially, wooden ships were protected with metallic copper sheathing. In later times, copper-containing paints were used on vessels of all types, and in the late 1960s, organotin compounds (in particular, TBT) were found to be a very effective ingredient in anti-fouling paints and these compounds rapidly became the active ingredient of choice in hull paints and use was widespread by the early 1970s.
However, the widespread use of TBT-based antifoulant paints by commercial shipping, including fishing fleets, and by leisure craft became associated with a marked decline in many commercially important marine mollusc fisheries (for example, mussels and oysters), characterized by declining populations of many resource species especially where there was a high density of boat traffic. Research demonstrated the toxic consequence of the exposure of marine molluscs to low (ng/litre) concentrations of water-borne TBT was primarily imposex (the development of male sexual structures in females—leading to reproductive failure), but also shell deformities, failure of larval settlement and bioaccumulation of TBT. (See also Box 7.2).
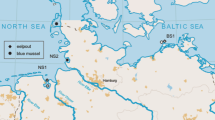
Subsequently, TBT was found to be environmentally persistent, particularly in sediments (a half-life of 4 years) but much less so in waters (half-life of 6 days), and increasing concentrations were found in the tissues of a wide range of marine life including fish and marine mammals. The sources of TBT to the marine environment were not only its release from vessel coatings, but also from poorly or non-regulated disposal of TBT-containing paint residues stripped from vessels when hulls were repaired and when regularly scheduled repainting was carried out (Figure 13.2).
The progressive introduction from 1982 by countries and international organizations (see timeline in Santillo et al. 2002) of restrictions on the use of TBT-based antifoulants, culminating in their effective phase-out by the International Maritime Organization (IMO), a specialized agency of the United Nations responsible for regulating shipping in its adoption of the International Convention on the Control of Harmful Antifouling Systems on Ships (IMO 2001). This convention imposed a global prohibition of the application of organotin compounds which act as biocides in anti-fouling systems on ships by 1 January 2003 and a complete prohibition of the presence of organotin compounds which act as biocides in anti-fouling systems on ships by 1 January 2008 and has succeeded in successfully managing the TBT contamination problem.
13.3 The Relationship Between CECs and Endocrine Disrupting Chemicals
Since the late 1980s, there has been growing evidence of the feminization of male fish in waters receiving sewage treatment plant discharges (e.g. Jobling et al. 1996, 1998) and this triggered concern in the general community and the attention of regulatory agencies and researchers concerning the presence of estrogenic chemicals in outfalls and receiving waters.
Common usage of the term endocrine disruption in the context of chemical pollution originated in 1991 as a consensus statement at a conference workshop series publication in Wisconsin, USA (Colborn and Clement 1992). The convenor of that conference, Theo Colborn along with others, subsequently published the book Our Stolen Future (Colborn et al. 1997), a landmark publication in raising public attention to the issues relating to endocrine disruption in wildlife and potentially humans.
The growing attention, given to endocrine disrupting chemicals (EDCs) phenomenon (Box 13.3), raised the concern levels about contaminants in aquatic environments, and in particular chemicals that are EDCs, and as a consequence numerous chemicals and substances became CECs. However, it is important to note that the EDC phenomenon is an expression of toxic effect, and although many CECs are associated with the EDC phenomenon, many are regarded as CECs for other reasons.
13.4 Pharmaceuticals and Personal Care Products (PPCPs) as CECs
The group of chemicals and substances collectively known as PPCPs includes both pharmaceuticals and personal care products used for personal health/well-being or for cosmetic purposes (see Chapter 12). The common usage of the term PPCPs also includes non-medicinal/non-cosmetic household products or their ingredients such as disinfectants (e.g. triclosan) and antiseptics, soaps, detergents and other cleaning products, synthetic musks and fragrances cosmetics, lotions, preservatives and sunscreen agents (e.g. oxybenzone). A recent overview of the global extent of discharges of PPCPs was provided in Dey et al. (2019).
Pharmaceuticals are defined as prescription, over-the-counter and veterinary therapeutic drugs used to prevent or treat human and animal diseases, while personal care products are used mainly to improve the quality of daily life (Boxall et al. 2012).
Pharmaceuticals can be classified by their therapeutic uses. The common uses being: anti-diabetics (e.g. alpha-glucosidase inhibitor), ß-blockers (e.g. atenolol, metoprolol), antibiotics (e.g. trimethoprim), lipid regulators (e.g. gemfibrozil), anti-epileptic (e.g. acetazolamide), tranquilizers (e.g. diazepam), anti-microbials (e.g. penicillins), anti-ulcer and anti-histamine drugs (e.g. cimetidine, famotidine), anti-anxiety or hypnotic agents (e.g. diazepam), anti-inflammatories and analgesics (e.g. ibuprofen, paracetamol, diclofenac), anti-depressants (e.g. benzodiazine-pines), anti-cancer drugs (e.g. cyclophosphamide, ifosfamide), anti-pyretics and stimulants (e.g. dexamphetamine, methylphenidate, modafinil), and estrogens and hormonal compounds (e.g. estriol, estradiol, estrone).
Currently more than 5000 manufactured pharmaceutical medicines are consumed by humans and/or domesticated animals, with an estimated total annual worldwide consumption in the range of 90,000–180,000 tonnes with the largest national consumptions being Russia, China, South Africa, India and Brazil (Van Boeckel et al. 2015; Tijani et al. 2016). A comprehensive overview of the current understanding of the extent and potential impact of contamination of the marine environment by pharmaceuticals is provided in the recent review by Ojemaye and Petrik (2019).
A large portion of medications that are ingested orally or by infusion are excreted through urine and/or faeces due to their incomplete absorption (metabolism) in humans and animals, these ultimately end up in wastewater treatment plants. Subsequently, municipal sewage treatment plants are major points of release of pharmaceuticals into the marine environment because wastewater treatment plants are not designed to decompose the vast majority of pharmaceutical compounds, which are by intent stable and robust, polar and non-volatile in nature. The most frequent and widespread pharmaceuticals in sewage and the discharge from marine outfalls are antibiotics and nonsteroidal anti-inflammatory drugs (Ojemaye and Petrik 2019). Other pathways for pharmaceuticals to be delivered into the marine system are via landfill sites, septic tanks, urban wastewater, showering and bathing, industrial effluent and agricultural runoff.
Measured concentrations of pharmaceuticals from worldwide coastal environment locations in seawater, sediments and organisms (Ojemaye and Petrik, 2019) range from 0.21 to 5000 ng/L (seawater), 0.0402 ng/g dry weight to 208 ng/g wet weight (biota) and 0.2 µg/kg dry weight to 466 µg/kg wet weight (sediments). However, despite evidence of their increasing presence, little attention has been directed towards understanding the release of pharmaceuticals into coastal-marine environments and their potential negative impact on marine ecosystems. This qualifies many pharmaceuticals as CECs in the marine environment.
Since the active ingredients in pharmaceuticals are chosen on the basis that their physicochemical and biological properties can produce specific biological effects in humans and animals, they have a high potential to trigger negative impacts on non-target organisms. In addition, anti-infection agents could create an ecological hazard by advancing the spread of resistant genes in the environment (Costanzo et al. 2005).
Other concerns are that the metabolites of many pharmaceuticals are potentially active and unsafe in the environment. For example, paracetamol and amitriptyline are mostly metabolized into highly reactive compounds (Graham et al. 2013). Also, of concern is that pharmaceuticals are discharged into the marine environment from sewage treatment plants as complex mixtures thus exposing marine life to potential synergetic environmental effects. For example, a synergistic antioxidant response in fish was demonstrated in a laboratory study involving co-exposure to a mixture of fluoxetine (an antidepressant medication) and roxithromycin (an antibiotic), and also with a mixture of fluoxetine and propranolol (a β-blocker used to treat a range of cardiac disease symptoms) (Ding et al. 2016).
Similarly, some ingredients of non-medicinal/non-cosmetic household products (e.g. triclosan—a widely used bactericide in healthcare products such as skin care ointments and lotions, mouthwashes and toothpastes, shower gels and shampoos) are not efficiently broken down in typical municipal sewage treatment plants, so that the end-of-treatment discharges from these facilities are major point sources of release into the marine environment (Cui et al. 2019). Triclosan is persistent and bioaccumulative in the aquatic environment and triggers a number of toxic responses (Maulvault et al. 2019).
The array of PPCPs in sewage discharges is extensive, but the potential for adverse effects is largely unknown for most of the active ingredients present (Ojemaye and Petrik 2019). The NORMAN Network (Box 13.2) currently lists almost 300 PPCPs substances as CECs.
13.5 Nanomaterials
The manufacture and use of nanoparticles and nanostructured materials (also known as nanomaterials) is an expanding field of modern technology. As a consequence, the perceived risks associated with potentially toxic properties of these novel materials have resulted in their attracting attention as a new class of CECs.
By their nature, nanoparticles are units of particulate materials with a maximum dimension sized in nanometres (10–9 m). Although there is no single internationally accepted definition for nanomaterials (Jeevanandam et al. 2018), they are commonly defined as materials in which a single unit is sized in the range of 1–100 nm in at least one dimension. The term aerosols is often applied to nanoparticles when they are airborne, for example, in wind-borne dust or otherwise suspended in the atmosphere. The US EPA routinely uses the term ultrafine particles when discussing natural nanomaterials and aerosols. A summary of types and classifications of nanomaterials, and common technical descriptors is at Box 13.4.
Interestingly, the use and manufacture of nanomaterials are not an entirely modern phenomenon. The Ancient Egyptians used nanoparticulate lead sulfide as a hair dye some 4000 years ago (Walter et al., 2006) and more recently (400‒100 BC) red enamels used by Ancient Celtic cultures were based on nanoparticulate copper oxides (Brun et al. 1991) and stained glass in medieval churches incorporated gold and silver nanoparticles (Schaming and Remita 2015).
The origin and source of nanoparticles and nanomaterials is diverse (Box 13.5). Naturally occurring nanoparticles (colloids) and nanomaterials are widespread in both the living and inanimate world. In addition, nanoparticles and nanomaterials may be produced as an incidental by-product of an industrial process, or they may be manufactured explicitly by an engineered process to exploit specific features that stem from their small size.
The application of nanoparticulate and nanostructured materials has increased over the past decade because they provide enhanced or unique physicochemical properties (e.g. melting point, wettability, electrical or thermal conductivity, catalytic activity, light absorbance or scattering) that are different from those of their bulk counterparts. Manufactured nanomaterials can significantly improve the characteristics of bulk materials, in terms of strength, conductivity, durability and lightness, and they can provide useful properties (e.g. self-healing, self-cleaning, anti-freezing and antibacterial) and can function as reinforcing materials for construction. By 2014, some 1814 nanotechnology-based consumer products were commercially available in over 20 countries (Vance et al. 2015). Examples of the incorporation of nanoparticles in consumer products include titanium oxide nanoparticles as a white pigment in paints, cosmetic creams and sunscreens, and silver nanoparticles used in numerous personal care products such as air sanitizers, wet wipes, shampoos and toothpastes, as well as in clothing and laundry fabric softeners (PEN 2019). Nanoscale zero-valent particulate iron (nZVI) is a widely used remediant for treating toxic wastes due to its large specific surface area and high reactivity (Stefaniuk et al. 2016).
Unfortunately, the highly sought physicochemical properties of nanomaterials that have led to their increasing applications can also have an associated environmental downside. For example, nanoparticulate zinc oxide (ZnO) used in sunscreens is toxic to marine algae largely because of its dissolution as Zn2+ (Franklin et al. 2007), and nZVI use in contaminant remediation presents a range of potentially harmful environmental consequences that are not well understood (Stefaniuk et al. 2016). The enhanced toxic potential of nanosized materials may arise from their capacity to penetrate and disturb the cells and cellular systems of living tissues.
The challenge for regulators is to determine whether nanomaterials should be regulated in the same way as micron-sized particles. Among metal nanomaterials, cerium dioxide (used as a diesel fuel additive) and nanosilver are more toxic than their micron-sized forms, whereas because of their solubility there is no difference in toxicity for zinc oxide nano- and micron-sized particles in freshwaters (Batley et al. 2013). The enhanced surface area of nanosized materials can result in different cellular uptake rates, oxidative mechanisms and processes including translocation relative to that of exposure to the same material when it is not nanosized (Oberdörster et al. 2005). In the environment, aggregation is a common feature of nanomaterials, and often coatings are used (e.g. citrate or polyvinylpyrrolidone (PVP)) to minimize this. Aggregation is greatest in marine waters due to their high ionic strength, leading to sizes > 100 nm and in many cases resulting in sedimentation (Klaine et al. 2008). The presence of organic particles such as those formed from extracellular polymeric substances can briefly stabilize nanomaterials (<48-h) (Gondikas et al. 2020). Seawater enhances the dissolution of silver from coated Ag nanomaterials, largely through chloride complexation, which reduces silver toxicity (Angel et al. 2013).
Some nanoparticles and nanomaterials are released directly into the environment from the use of consumer products (e.g. silicon nanoparticles in car tyres are released by abrasion in normal vehicle use), or indirectly (e.g. nanoparticles in pharmaceuticals and cosmetics can end up in sewage, and then be discharged to the marine environment).
13.6 PFAS (Per- and Polyfluoroalkyl Substances)
The term PFAS (per- and polyfluoroalkyl substances) applies to the set of more than 4700 synthetic substances manufactured and used in a variety of industries since the 1940s (OECD 2019), and some have been classified as POPs (Chapter 8). All PFAS constitute an array of highly persistent environmental CECs that has triggered a global response by research and regulatory organizations over the past two decades.
PFAS comprise a set of compounds each of which has a molecular structure comprising an aliphatic moiety (i.e. a group of covalently bonded carbon atoms in a straight or branched chain, and in some cases including non-aromatic rings) that is highly fluorinated and linked to a functional group moiety. This PFAS molecular structure can be conceptualized as an alkyl tail of carbon atoms with fluorine atoms attached to a a functional group head (Figure 13.3). The degree of fluorination of the aliphatic moiety in a PFAS structure can be partial or total. In polyfluoroalkyl substances, fluorine atoms replace only some of the hydrogen atoms in the aliphatic chain, whereas in perfluoroalkyl substances, fluorine atoms replace all of the hydrogen atoms in the aliphatic chain. The general formula for a perfluorinated PFAS is CnF2n+1-R where n is 3 or greater and -R is a functional group such as carboxylic acid (COOH), sulfonic acid (SO3H) or sulfonamide (SO2NH2) (Figure 13.3).
Typical perfluorinated PFAS molecules showing the basic structure comprising a perfluorinated alkyl tail attached to a functional group head. Structures here are the linear isomers. A mixture of linear and branched isomers may be present in an environmental sample. Adapted from Mueller and Yingling (2017) by M. Mortimer
Note that the term PFAS sometimes appears in print in the context of more than one fluorinated chemical, but the addition of the s is redundant since the acronym PFAS includes the plural (ATSDR 2017). Also, it is important to note that PFAS (per- and polyfluoroalkyl substances) are sometimes called perfluorinated chemicals and the acronym PFC is then used. However, this use of the PFC acronym can be confusing since PFC is also commonly used for a related, but distinctly different group of substances: the perfluorocarbons (Box 13.6).
The range of structurally related compounds comprising more than 4700 member group of PFAS substances is illustrated in the PFAS family tree in Figure 13.4.
The PFAS family tree with examples. Adapted from Wang et al. (2017) by M. Mortimer. PFCAs = Perfluoroalkyl carboxylic acids; PFSAs = Perfluoroalkane sulfonic acids; PFPAs = Perfluoroalkyl phosphonic acids; PFPiAs = Perfluoroalkyl phosphinic acids; PFECAs and PFESAs = Perfluoroether carboxylic and sulfonic acids; PASF = Perfluoroalkane sulfonyl fluoride
13.7 Summary
There is a large body of research papers and reports concerning the topic of Contaminants of Emerging Concern (CECs) but the term itself is not definitive since both emerging and concern may be subjective, and the list of materials identified as CECs changes over time and in response to community perceptions of risks to health and the environment. The NORMAN Network is a key organization in identifying such materials and coordinating meaningful related research.
In the marine environment, since the late 1900s the priority focus has moved from concern over unintended impacts from the widespread use of organic tin-based antifoulants used on the hulls of sea-going vessels to impacts relating to a wide range of material types including EDCs, PPCPs, nanomaterials, PFAS compounds as well as environmental contamination by polymer and plastic debris.
Each of these current CECs covers a large number of chemical identities. Overall this is a wide-ranging and dynamic area of risk assessment, priority setting and ongoing scientific research.
13.8 Study Questions and Activities
-
1.
In the context of the marine environment draw up a short list of up to five contaminants of emerging concern that are highlighted in recent media publications (noting that the media may not use the term contaminant of emerging concern as a descriptor) and compare this short list with contaminants which are popular topics in the programmes of recent conference presentations and journal publications. What do you suggest are reasons for similarities and differences between these two sets of CECs?
-
2.
Identify two CECs in the marine environment that have been receiving frequent attention for a period longer than two or three years. Why are they still considered emerging (for example, has the baseline of residual concern changed)?
-
3.
In this chapter, lead in the marine environment near Port Pirie, South Australia is used as an example. What other locations in Australia and other countries with territorial waters in the Pacific Ocean also have an emerging problem associated with lead mining and processing?
-
4.
Which metallic contaminants are CECs in European marine waters?
-
5.
What potential contaminants of marine waters are likely to become CECs as a consequence of the shift from fossil-fuel-based energy sources to renewables? What are some geographic locations where these may first emerge as CECs—explain why?
Abbreviations
- AFFF:
-
Aqueous film-forming firefighting foams
- ATSDR:
-
Agency for Toxic Substances and Disease Registry (USA)
- BPA:
-
Bisphenol A
- C60:
-
Fullerenes
- CAS:
-
Chemical abstracts service
- CDC:
-
Center for Disease Control and Prevention (USA)
- CEC:
-
Contaminant of emerging concern
- CFC:
-
Chlorofluorocarbon
- CNT:
-
Carbon nanotube
- DDT:
-
Dichlorodiphenyltrichloroethane, a shortened version of a former name used for 1,1'-(2,2,2-trichloroethane-1,1-diyl)bis(4-chlorobenzene)
- EDC:
-
Endocrine disrupting chemical
- EEA:
-
European Environment Agency
- FOSA:
-
Perfluorooctane sulfonamide
- Gr:
-
Graphene, an allotrope of carbon
- HCF:
-
Hydrofluorocarbon
- IMO:
-
International Maritime Organization (a United Nations Intergovernmental Body)
- IUPAC:
-
International Union of Pure and Applied Chemistry
- NOAA:
-
National Oceanic and Atmospheric Administration (USA)
- NORMAN:
-
An international network of reference laboratories, research centres and related organizations for monitoring of emerging environmental substances
- nZVI:
-
Nanoscale zero-valent particulate iron
- OECD:
-
Organisation for Economic Co-operation and Development
- PBB:
-
Polybrominated biphenyl
- PCB:
-
Polychlorinated biphenyl
- PFAA:
-
Perfluoroalkyl acid
- PFAS:
-
Per- and polyfluoroalkyl substances
- PFC:
-
Perfluorinated chemical or perfluorocarbon. These are related but distinctly different groups of substances (see Box 13.6)
- PFBA:
-
Perfluorobutanoic acid or perfluorobutanoate (see Box 13.9)
- PFCA:
-
Perfluoroalkyl carboxylic acid
- PFHxA:
-
Perfluorohexanoic acid
- PFOA:
-
Perfluorooctanoic acid
- PFOS (or PFSA):
-
Perfluorooctane sulfonic acid
- PFOSA:
-
Perfluorooctane sulfonamide
- PFSA:
-
Perfluoroalkane sulfonic acid
- POP:
-
Persistent organic pollutant
- POSF:
-
Perfluorooctane sulfonyl fluoride
- PPCP:
-
Pharmaceutical and personal care product
- REACH:
-
Registration, Evaluation, Authorisation and Restriction of Chemicals (a European Union Regulatory Organisation).
- TBT:
-
Tributyltin
- UNESCO:
-
United Nations Educational, Scientific and Cultural Organization
- US EPA:
-
United States Environmental Protection Agency
References
Ahrens L (2011) Polyfluoroalkyl compounds in the aquatic environment: a review of their occurrence and fate. J Environ Monit 13:20–31
Angel BM, Batley GE, Jarolimek C, Rogers NJ (2013) The impact of nano size on the fate and toxicity of nanoparticulate silver in aquatic systems. Chemosphere 93:359–365
ATSDR (Agency for Toxic Substances and Disease Registry) (2017) The family tree of per- and polyfluoralkyl substances (PFAS) for environmental health professionals. Available at: https://www.atsdr.cdc.gov/pfas/docs/PFAS_FamilyTree_EnvHealthPro-508.pdf. Accessed 29 Mar 2019
Basu S, Mackey K (2018) Phytoplankton as key mediators of the biological carbon pump: their responses to a changing climate. Sustainability 10(3):869
Batley GE, Kirby JK, McLaughlin MR (2013) Fate and risks of nanomaterials in the aquatic environment. Acc Chem Res 46:854–862
Boxall A, Rudd M, Brooks B, Caldwell D, Choi K, Hickmann S, Innes E, Ostapyk K, Staveley J, Verslycke T, Ankley G, Beazley K, Belanger S, Berninger J, Carriquiriborde P, Coors A, DeLeo P, Dyer S, Ericson J, Gagné F, Giesy J, Gouin T, Hallstrom L, Karlsson M, Joakim Larsson D, Lazorchak J, Mastrocco F, McLaughlin A, McMaster M, Meyerhoff R, Moore R, Parrott J, Snape J, Murray-Smith R, Servos M, Sibley P, Oliver Straub J, Szabo N, Topp E, Tetreault G, Trudeau V, Van Der Kraak G (2012) Pharmaceuticals and personal care products in the environment: what are the big questions? Environ Health Perspect 120(9):1221–1229
Bristow L, Mohr W, Ahmerkamp S, Kuypers M (2017) Nutrients that limit growth in the ocean. Curr Biol 27(11):R474–R478
Brun N, Mazerolles L, Pernot M (1991) Microstructure of opaque red glass containing copper. J Mater Sci Lett 10(23):1418–1420
Buck R, Franklin J, Berger U, Conder J, Cousins I, de Voogt P, Jensen A, Kannan K, Mabury S, van Leeuwen S (2011) Perfluoroalkyl and polyfluoroalkyl substances in the environment: terminology, classification, and origins. Integr Environ Assess Manage 7(4):513–541
Buseck P, Posfai M (1999) Airborne minerals and related aerosol particles: effects on climate and the environment. Proc Natl Acad Sci 96(7):3372–3379
Cilliers L, Retief F (2019) Lead poisoning and the downfall of Rome: reality or myth? In: Wexter P (ed) Toxicology in antiquity. Elsevier, Amsterdam, pp 135–148
Colborn T, Clement C (1992) Chemically-induced alterations in sexual and functional development: the wildlife/human connection. Princeton Scientific, Princeton, p 403
Colborn T, Dumanoski D, Myers J (1997) Our stolen future. Abacus, London, p 306
Costanzo S, Murby J, Bates J (2005) Ecosystem response to antibiotics entering the aquatic environment. Mar Pollut Bull 51(1–4):218–223
Cui Y, Wang Y, Pan C, Li R, Xue R, Guo J, Zhang R (2019) Spatiotemporal distributions, source apportionment and potential risks of 15 pharmaceuticals and personal care products (PPCPs) in Qinzhou Bay, South China. Mar Pollut Bull 141:104–111
Dey S, Bano F, Malik A (2019) Pharmaceuticals and personal care product (PPCP) contamination—a global discharge inventory. In: Prasad M, Vithanage M, Kapley A (eds) Pharmaceuticals and personal care products: waste management and treatment technology: emerging contaminants and micro pollutants. Butterworth-Heinemann, Oxford, pp 1–26
Ding J, Lu G, Li Y (2016) Interactive effects of selected pharmaceutical mixtures on bioaccumulation and biochemical status in crucian carp (Carassius auratus). Chemosphere 148:21–31
Dulio V, van Bavel B, Brorström-Lundén E, Harmsen J, Hollender J, Schlabach M, Slobodnik J, Thomas K, Koschorreck J (2018) Emerging pollutants in the EU: 10 years of NORMAN in support of environmental policies and regulations. Environ Sci Eur 30(1):1–13
Ealias A, Saravanakumar M (2017) A review on the classification, characterisation, synthesis of nanoparticles and their application. IOP Conf Ser Mater Sci Eng 263:032019
EPASA (Environment Protection Authority, South Australia) (2005) Heavy metal contamination in the Northern Spencer Gulf—a community summary. Available at: https://www.epa.sa.gov.au/files/477354_heavy_metal.pdf. Accessed 1 Oct 2021
Favre H, Powell W (2013) Nomenclature of organic chemistry. Royal Society of Chemistry, Cambridge, p 1568
Forbes M, Taylor M (2015) A review of environmental lead exposure and management in Mount Isa, Queensland. Rev Environ Health 30(3):183–189
Franklin NM, Rogers NT, Apte SC, Batley GE, Casey PE (2007) Comparative toxicity of nanoparticulate ZnO, bulk ZnO and ZnCl2 to a freshwater microalga (Pseudokirchnerilla subcapitata): the importance of particle solubility. Environ Sci Technol 41:8484–8490
Godfray H, Stephens A, Jepson P, Jobling S, Johnson A, Matthiessen P, Sumpter J, Tyler C, McLean A (2019) A restatement of the natural science evidence base on the effects of endocrine disrupting chemicals on wildlife. Proc Roy Soc B Biol Sci 286(1897):20182416
Gondikas A, Gallego-Urrea J, Halbach M, Derrien N, Hassellöv M (2020) Nanomaterial fate in seawater: a rapid sink or intermittent stabilization? Front Environ Sci 8:151
Graham G, Davies M, Day R, Mohamudally A, Scott K (2013) The modern pharmacology of paracetamol: therapeutic actions, mechanism of action, metabolism, toxicity and recent pharmacological findings. Inflammopharmacology 21(3):201–232
Jeevanandam J, Barhoum A, Chan Y, Dufresne A, Danquah M (2018) Review on nanoparticles and nanostructured materials: history, sources, toxicity and regulations. Beilstein J Nanotechnol 9:1050–1074
Jobling S, Sumpter J, Sheahan D, Osborne J, Matthiessen P (1996) Inhibition of testicular growth in rainbow trout (Oncorhynchus mykiss) exposed to estrogenic alkylphenolic chemicals. Environ Toxicol Chem 15(2):194–202
Jobling S, Nolan M, Tyler C, Brighty G, Sumpter J (1998) Widespread sexual disruption in wild fish. Environ Sci Technol 32(17):2498–2506
Klaine SJ, Alvarez PJJ, Batley GE, Fernandes TF, Handy RD, Lyon D, Mahendra S, McLaughlin MJ, Lead JR (2008) Nanomaterials in the environment: behaviour, fate, bioavailability and effects. Environ Toxicol Chem 27:1825–1851
Halden R (2015) Epistemology of contaminants of emerging concern and literature meta-analysis. J Hazard Mater 282:2–9
IMO (International Maritime Organization) (2001) International convention on the control of harmful anti-fouling systems on ships. IMO Assembly Resolution A.895 (21). Available at: http://www.imo.org/en/KnowledgeCentre/IndexofIMOResolutions/Assembly/Documents/A.895(21).pdf. Accessed 12 Mar 2019
Katsiadaki I (2019) Are marine invertebrates really at risk from endocrine-disrupting chemicals? Current Opin Environ Sci Health 11:37–42
Krafft M, Riess J (2015) Selected physicochemical aspects of poly- and perfluoroalkylated substances relevant to performance, environment and sustainability—part one. Chemosphere 129:4–19
Lafratta A, Serrano O, Masqué P, Mateo M, Fernandes M, Gaylard S, Lavery P (2019) Seagrass soil archives reveal centennial-scale metal smelter contamination while acting as natural filters. Sci Total Environ 649:1381–1392
Lead JR, Batley GE, Alvarez PJJ, Croteau MN, Handy RD, McLaughlin MJ, Judy JD, Schirmer K (2018) Nanomaterials in the environment: behaviour, fate, bioavailability and effects. An updated review. Environ Toxicol Chem 37:2029–2063
Lehmler H (2005) Synthesis of environmentally relevant fluorinated surfactants—a review. Chemosphere 58(11):1471–1496
Llorca M, Farre M, Tavano M, Alonso B, Koremblit G, Barceó D (2012) Fate of a broad spectrum of perfluorinated compounds in soils and biota from Tierra del Fuego and Antarctica. Environ Pollut 163:158–166
Maulvault A, Camacho C, Barbosa V, Alves R, Anacleto P, Cunha S, Fernandes J, Pousão-Ferreira P, Paula J, Rosa R, Diniz M, Marques A (2019) Bioaccumulation and ecotoxicological responses of juvenile white seabream (Diplodus sargus) exposed to triclosan, warming and acidification. Environ Pollut 245:427–442
Mueller R, Yingling V (2017) Naming conventions and physical and chemical properties of per- and Polyfluoroalkyl substances (PFAS). Interstate Technology Regulatory Council (ITRC), p 15. Available at: https://pfas-1.itrcweb.org/fact_sheets_page/PFAS_Fact_Sheet_Naming_Conventions_April2020.pdf. Accessed 13 Febr 2022
Mühle J, Ganesan A, Miller B, Salameh P, Harth C, Greally B, Rigby M, Porter L, Steele L, Trudinger C, Krummel P, O’Doherty S, Fraser P, Simmonds P, Prinn R, Weiss R (2010) Perfluorocarbons in the global atmosphere: tetrafluoromethane, hexafluoroethane, and octafluoropropane. Atmos Chem Phys 10(11):5145–5164
Ng C, Hungerbühler K (2014) Bioaccumulation of perfluorinated alkyl acids: observations and models. Environ Sci Technol 48(9):4637–4648
Ng C, Hungerbuehler K (2015) Exploring the use of molecular docking to identify bioaccumulative perfluorinated alkyl acids (PFAAs). Environ Sci Technol 49(20):12306–12314
NIEHS (2022) Endocrine disruptors. National Institute of Environmental Health Sciences. Available at: https://www.niehs.nih.gov/health/topics/agents/endocrine/index.cfm. Accessed 9 Mar 2022
Nilsen E, Smalling K, Ahrens L, Gros M, Miglioranza K, Picó Y, Schoenfuss H (2018) Critical review: grand challenges in assessing the adverse effects of contaminants of emerging concern on aquatic food webs. Environ Toxicol Chem 38(1):46–60
NOAA (National Oceanic and Atmospheric Administration) (2008) Mussel watch program. An assessment of two decades of contaminant monitoring in the Nation’s Coastal zone. NOAA Technical Memorandum NOS NCCOS 74. Available at: https://repository.library.noaa.gov/gsearch?related_series=NOAA%20technical%20memorandum%20NOS%20NCCOS%20%3B%2074. Accessed 29 Mar 2019
Oberdörster G, Oberdörster E, Oberdörster J (2005) Nanotoxicology: an emerging discipline evolving from studies of ultrafine particles. Environ Health Perspect 113(7):823–839
OECD (Organisation for Economic Co-operation and Development) (2013) Synthesis paper on per- and polyfluorinated chemicals (PFCs). OECD/UNEP Global PFC Group. Available at: https://www.oecd.org/env/ehs/risk-management/PFC_FINAL-Web.pdf. Accessed 29 Mar 2019
OECD (Organisation for Economic Co-operation and Development) (2019) Portal on per and poly fluorinated chemicals (Online Database). Available at: http://www.oecd.org/chemicalsafety/portal-perfluorinated-chemicals/. Accessed 13 Febr 2022
Ojemaye C, Petrik L (2019) Pharmaceuticals in the marine environment: a review. Environ Rev 27(2):151–165
PEN (Project on Emerging Nanotechnologies) (2019) The project on emerging nanotechnologies. Available at: http://www.nanotechproject.tech/search/?q=silver&x=0&y=0. Accessed 13 Febr 2022
Santillo D, Johnston P, Langston W (2002) Tributyltin (TBT) antifoulants: a tale of ships, snails and imposex. In: Harremoes P, Gee D, MacGarvin M, Stirling A, Keys J, Wynne B, Vaz S (eds) Late lessons from early warnings: the precautionary principle 1896–2000. Environment Issue Report, no. 22. European Environment Agency, Copenhagen, p 135–148. Available at: https://www.eea.europa.eu/publications#c7=en&c11=5&c14=&c12=&b_start=0&c13=Late+lessons+from+early+warnings%3A+the+precautionary+principle+1896-2000. Accessed 13 Febr 2022
Schaming D, Remita H (2015) Nanotechnology: from the ancient time to nowadays. Found Chem 17(3):187–205
Simpson SL, Yawen L, Spadaro DA, Kookana RS, Batley GE (2021) Chronic effects and thresholds for estuarine and marine benthic organism exposure to perfluorooctane sulfonic acid (PFOS)-contaminated sediments: influence of organic carbon and exposure routes. Sci Total Environ 776:146008
Stefaniuk M, Oleszczuk P, Ok Y (2016) Review on nano zerovalent iron (nZVI): from synthesis to environmental applications. Chem Eng J 287:618–632
Tijani J, Fatoba O, Babajide O, Petrik L (2016) Pharmaceuticals, endocrine disruptors, personal care products, nanomaterials and perfluorinated pollutants: a review. Environ Chem Lett 14(1):27–49
Tornero V, Hanke G (2017) Potential chemical contaminants in the marine environment: an overview of main contaminant lists. Office of the European Union Publications, Luxembourg. Available at: http://publications.jrc.ec.europa.eu/repository/bitstream/JRC108964/potential_chemical_contaminants_in_the_marine.pdf. Accessed 19 Aug 2019
UNESCO (United Nations Educational, Scientific and Cultural Organization) (2019) Emerging pollutants in water and wastewater. Available at: https://en.unesco.org/emergingpollutants. Accessed 12 Mar 2019
United Nations (1998) Kyoto protocol to the United Nations framework convention on climate change. Available at: https://unfccc.int/resource/docs/convkp/kpeng.pdf. Accessed 29 Mar 2019
Van Boeckel T, Brower C, Gilbert M, Grenfell B, Levin S, Robinson T, Teillant A, Laxminarayan R (2015) Global trends in antimicrobial use in food animals. Proc Natl Acad Sci 112(18):5649–5654
Vance M, Kuiken T, Vejerano E, McGinnis S, Hochella M, Rejeski D, Hull M (2015) Nanotechnology in the real world: redeveloping the nanomaterial consumer products inventory. Beilstein J Nanotechnol 6:1769–1780
Walter P, Welcomme E, Hallégot P, Zaluzec N, Deeb C, Castaing J, Veyssière P, Bréniaux R, Lévêque J, Tsoucaris G (2006) Early use of PbS nanotechnology for an ancient hair dyeing formula. Nano Lett 6(10):2215–2219
Wang Z, DeWitt J, Higgins C, Cousins I (2017) A never-ending story of per- and polyfluoroalkyl substances (PFASs)? Environ Sci Technol 51(5):2508–2518
Wei S, Chen L, Taniyasu S, So M, Murphy M, Yamashita N, Yeung L, Lam P (2007) Distribution of perfluorinated compounds in surface seawaters between Asia and Antarctica. Mar Pollut Bull 54(11):1813–1818
Windsor F, Ormerod S, Tyler C (2017) Endocrine disruption in aquatic systems: up-scaling research to address ecological consequences. Biol Rev 93(1):626–641
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Open Access This chapter is licensed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made.
The images or other third party material in this chapter are included in the chapter's Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the chapter's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
Copyright information
© 2023 The Author(s)
About this chapter
Cite this chapter
Mortimer, M., Batley, G. (2023). Marine Contaminants of Emerging Concern. In: Reichelt-Brushett, A. (eds) Marine Pollution – Monitoring, Management and Mitigation . Springer Textbooks in Earth Sciences, Geography and Environment. Springer, Cham. https://doi.org/10.1007/978-3-031-10127-4_13
Download citation
DOI: https://doi.org/10.1007/978-3-031-10127-4_13
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-10126-7
Online ISBN: 978-3-031-10127-4
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)